1. Introduction
Hyaluronic acid (HA) is a glycosaminoglycan that is found in the extracellular matrix of soft connective tissues [
1]. Thanks to its high concentration of carboxylic acid and hydroxyl groups, this material possesses a wide range of applications. High-molecular-weight HA (>10 kDa) has been shown to have wide use in ophthalmology, orthopaedics, cosmetics, and tissue engineering [
2,
3,
4], whereas low-molecular-weight HA (<5 kDa) has been shown to be a versatile backbone for producing complexes that promote angiogenesis, inhibit tumour progression, or induce expression of pro-inflammatory mediators [
4]. In particular, HA has been considered of interest for the cosmetic sector as it possesses high water binding capacity [
1]. Conventionally, HA is extracted from animal tissue through a technically challenging process which involves grinding, acid treatment, and repeated extraction with organic solvents [
4]. Currently, there is a strong environmental (solvent use), social (non-vegan extraction), and economical (HA moisturisers will be valued at USD 7.36 billion by 2033 [
5]) drive towards synthesising this biopolymer through microorganisms due to the vegan nature of the resulting material. It has been shown that
Streptococcous sp., amongst others, naturally produce HA extracellularly for adherence and protection but also as a molecular imitation for evasion of the host immune system during its infection process [
4]. However,
Streptococcus sp. is still widely considered a pathogen capable of producing several endotoxins; to this end, either enzymatic (cell-free) systems are employed (limited to 2000 kDa) [
6], or the biochemical pathway for HA synthesis is engineered within non-endotoxin-producing, generally regarded as safe (GRAS) organisms (
Bacillus sp.) [
4,
7]. Nonetheless, this approach still sees high variability in molecular weights obtained for HA, relatively low yields at industrial scale (6–7 g/L) due to the high viscosity [
8], public perception of genetically modified organism (GMO) products, and very high CAPEX facilities (to handle GMOs) [
4]. Further, further broadening the application of HA is the ubiquitous nature of natural hyaluronidases, capable of efficiently depolymerising the polymer and biochemically utilising it as a natural carbon source [
9].
Similarly to Streptococcus sp., other natural organisms have been shown to produce extracellular material for adherence, protection, and immune evasion. Historically, anthrax from Bacillus antheracis was notoriously difficult to identify and treat due to native biosynthesis of poly-D-γ-glutamic acid, an immune-invisible pseudopolypeptide. Similarly to HA, poly-γ-glutamic acid is rich in water-interacting groups (carboxylic acid) and is produced between 10 and 10,000 kDa.
Numerous prokaryotic and eukaryotic microorganisms have been found to naturally synthesise poly-D/L-γ-glutamic acid (γ-PGA), a biodegradable, water-soluble, non-immunogenic biomacromolecule [
10,
11,
12]. γ-PGA is an edible biopolymer widely consumed in the Asian continent and has been used for several applications, ranging from bioremediation [
13,
14,
15] to tuneable drug delivery systems [
16,
17,
18,
19,
20]. Such suitability for different applications has been achieved due to the chemico-physical composition of the material, which can be obtained by tuning the conditions of biosynthesis [
10,
11,
21,
22,
23,
24]. In fact, as γ-PGA is synthesised by a transmembrane enzymatic complex instead of a ribosomal complex, both the molecular weight and the D/L-glutamic acid ratio can be varied. It has been previously suggested that both the molecular weight and the D/L-glutamic acid ratio can significantly impact the rheological behaviour of γ-PGA, the water absorption capabilities, and the water holding capacity [
25]. Differently to HA, γ-PGA can persist longer in natural environments due to the γ-peptide bond, which is not degradable by means of commonly produced α-proteases [
21,
24]. Further, being naturally produced by members of the
Bacillus sp., there is no need to transfer the genetic capacity to other organisms as it has highly developed biosynthetic capacity and capability to grow in industrial fermenters [
4]. These factors have been suggested to be crucial to substitute conventional moisturisers [
1,
4]. Further, γ-PGA has been suggested to possess other beneficial properties, including UV protection [
26] and pre-biotic capacity [
27], both highly desirable for the next generation of cosmetic solutions.
Similarly to GMO-mediated HA synthesis, the biosynthesis of γ-PGA is predominantly achieved by means of a defined media [
12,
24,
28]. With an outlook on industrial commercialisation and up-scaling, several more circular, complex waste media have been assessed and shown to be suitable for γ-PGA biosynthesis. For instance, ref. [
29] employed rotten tomatoes for the biosynthesis of γ-PGA, ref. [
24] successfully synthetised γ-PGA from supplemented macroalgae, and ref. [
30] employed soybean meal for the biosynthesis of γ-PGA. The use of complex waste media as a substrate for γ-PGA biosynthesis has been further described in [
12,
31,
32].
Here, we aim to demonstrate the ability to effectively alter the chemico-physical properties of γ-PGA through variation in substrate composition; compare the rheological behaviour of γ-PGA creams with commercial creams/lotions; and compare water absorption capacity of γ-PGA, HA, and glycerol. Within this context, seaweed-derived γ-PGA creams are assessed for their hydration, UV protection, and overall biocompatibility against HA, the golden standard in the industry. The aim of this work is to provide an innovative raw material which can combine both superior hygroscopicity and UV protection to the cosmetic industry. The next generation of cosmetic ingredients must be synthesised and isolated in a greener manner, and they need to be vegan friendly, biocompatible, and have comparable or improved properties to HA, the current gold standard in the cosmetic industry.
3. Results and Discussion
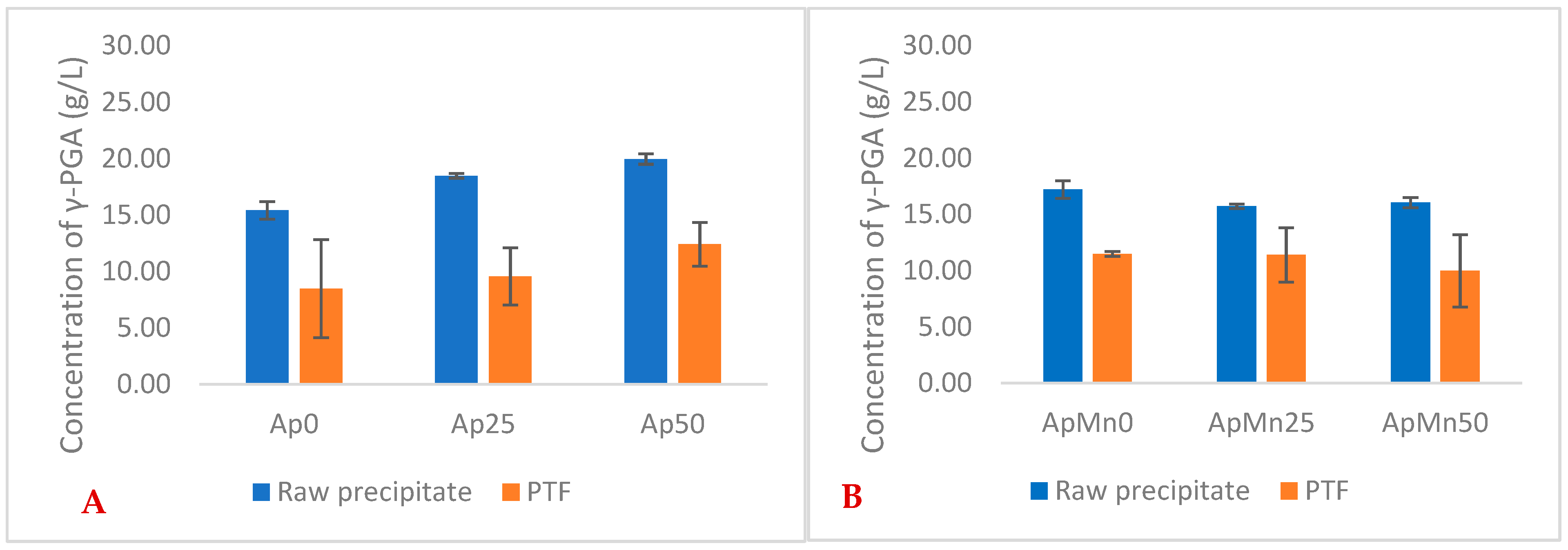
Similarly to the defined GS media, the greatest yields with seaweed-based media were observed for 25 g/L NaCl conditions (
Figure 1). Differently, higher yields of γ-PGA were obtained with 0 g/L NaCl concentration when in the presence of manganese sulphate. Overall, there was no significant difference in yields between manganese sulphate supplemented and non-supplemented samples in raw precipitates (
Figure 1) (
p = 0.574). Overall, our data suggest that manganese-supplemented substrates provided a higher material yield after tangential flow filtration compared to non-manganese-supplemented substrates.
It has been widely reported that the molecular weight, the crystallinity, and the ion profile of γ-PGA are crucial factors impacting overall material viscosity [
35,
36]. These parameters have been benchmarked against commercially available γ-PGA samples, which are specifically sold for cosmetic application. The properties of such commercially available polymers were compared to those previously obtained with
Bacillus subtilis natto cultivated on standard GS media, and algal-media-derived γ-PGA was assessed by means of gel permeation chromatography. Successful biosynthesis of γ-PGA by means of standard GS and seaweed-based media (
Laminaria digitata,
Saccharina latissimi, and
Alaria esculenta) was previously confirmed by means of FT-IR [
24]. Analogous procedure has been employed to confirm successful biosynthesis of Ap and ApMn samples reported within
Table 6. The physical properties of γ-PGA produced has been summarised within
Table 6.
From
Table 6, it can be observed that there was an inversely proportional behaviour in both samples with NaCl supplementation and in samples with NaCl + MnSO
4 supplementation for
Ascophyllum nodosum-derived substrate, meaning that, with increasing concentrations in NaCl, the molecular weight of γ-PGA synthesised was decreased (see
Table 6). Such correlation was also previously reported by [
37], but it was not upheld from previous investigations reported by [
24], for which 50 g/L NaCl yielded higher-molecular-weight γ-PGA compared to 25 g/L NaCl concentrations (see
Table 6). Such variation in NaCl dose–response can significantly be affected by the type of microorganism amongst other possible factors [
38].
Similarly to standard GS media, with an increase in concentration of NaCl, there was no significant decrease in the average molecular weight of γ-PGA (in both raw and PTF samples) [
24] (
Figure 1). Such a trend does appear to be upheld with
Ascophyllum nodosum-based substrates. This suggests that the type of substrate might have a similar NaCl effect on molecular weight. However, differently from what was reported in the literature, 50 g/L of NaCl did not seem to impact crystallinity of the γ-PGA formed. This lack of crystallinity could be due to the low molecular weight of the γ-PGA produced by
Bacillus subtilis natto in this case compared to the molecular weight of γ-PGA produced in standard GS media [
12]. We previously reported that with increases in concentration of NaCl, there is the formation of crystalline material (same microorganisms, same process conditions) [
24]. This variation in properties can be directly correlated to the lower molecular weight of the γ-PGA produced through
Ascophyllum nodosum-based media at 50 g/L NaCl (2,700,000 Da GS compared to 102,000 Da from supplemented
Ascophyllum nodosum media).
Interestingly, in standard GS media, when supplemented with MnSO4, it appears that the concentration of NaCl required to synthesise a crystalline polymer was decreased from 50 g/L to 25 g/L NaCl. This can be explained by the increased polymerisation of D-glutamic acid monomers (as suggested in literature), which results in the overall split between D- and L-glutamic acid monomers closer to 50:50 and ultimately in lower entropy for self-arrangement of γ-PGA chains in a crystalline lattice.
Differently from
Ascophyllum nodosum-based media, with standard GS media, there is not such a variation between the concentration of NaCl and the molecular weight of the material. Interestingly, in GS media, when there was a supplementation of the media with 0.415 g/L MnSO
4, there was roughly a halving in the molecular weight of the resulting γ-PGA. This result is in line with data previously reported by [
38]. Differently, in
Ascophyllum nodosum-based media, the supplementation of 0.415 g/L MnSO
4 resulted in almost a quadrupling of the molecular weight for the 0 g/L NaCl set of experiments, with comparable molecular weights at 50 g/L NaCl concentrations (
Table 2). Interestingly, across all
Ascophyllum nodosum conditions, the resulting γ-PGA presented amorphous behaviour. Such a lack of semi-crystallinity can be expected with such low-molecular-weight polymers; therefore, the effectiveness of NaCl to catalyse higher crystallinity cannot be commented on with this set of experiments.
In the literature, it has been described that, depending upon the overall ionic prevalence of γ-PGA, its physical arrangement can vary significantly, and ultimately the properties of the material change [
39,
40]. To evaluate the potential impact of algal substrate (
Ascophyllum nodosum) ionic content upon the final properties of the material, the concentration of key ions has been suggested to lie between 0.7 and 3.9 wt% of sodium, 1.68 and 3.9 wt% of chloride, 0.2 and 1.6 wt% of magnesium, 1.5 and 2.6 wt% of potassium, 0.8 and 2.6 wt% of calcium, and 0.04 and 0.45 wt% of manganese [
41]. Of the total fraction, sodium content was below 3.9 wt%, which equates to 1.56 g sodium per 40 g/L of
Ascophyllum nodosum employed. This fraction is unlikely to significantly affect the productivity or the type of seaweed seeing as within the tested dependent conditions, upwards of 50 g/L of NaCl was tested. Differently, it is possible that the combination of potassium, magnesium, and calcium could result in their association with some of the γ-PGA synthesised.
Hydration/humectant potential of γ-PGA [
25,
26,
34], and more specifically, algal-derived γ-PGA, is key to assessing its ability to displace hyaluronic acid and other synthetic polymers. To this end, the hydration/humectant capacity of three commercially available γ-PGA samples was recorded according to the method described within
Section 2.5. The values obtained from γ-PGA were compared to hyaluronic acid and glycerol, with widely recognised high hygroscopicity. The results from this investigation are summarised within
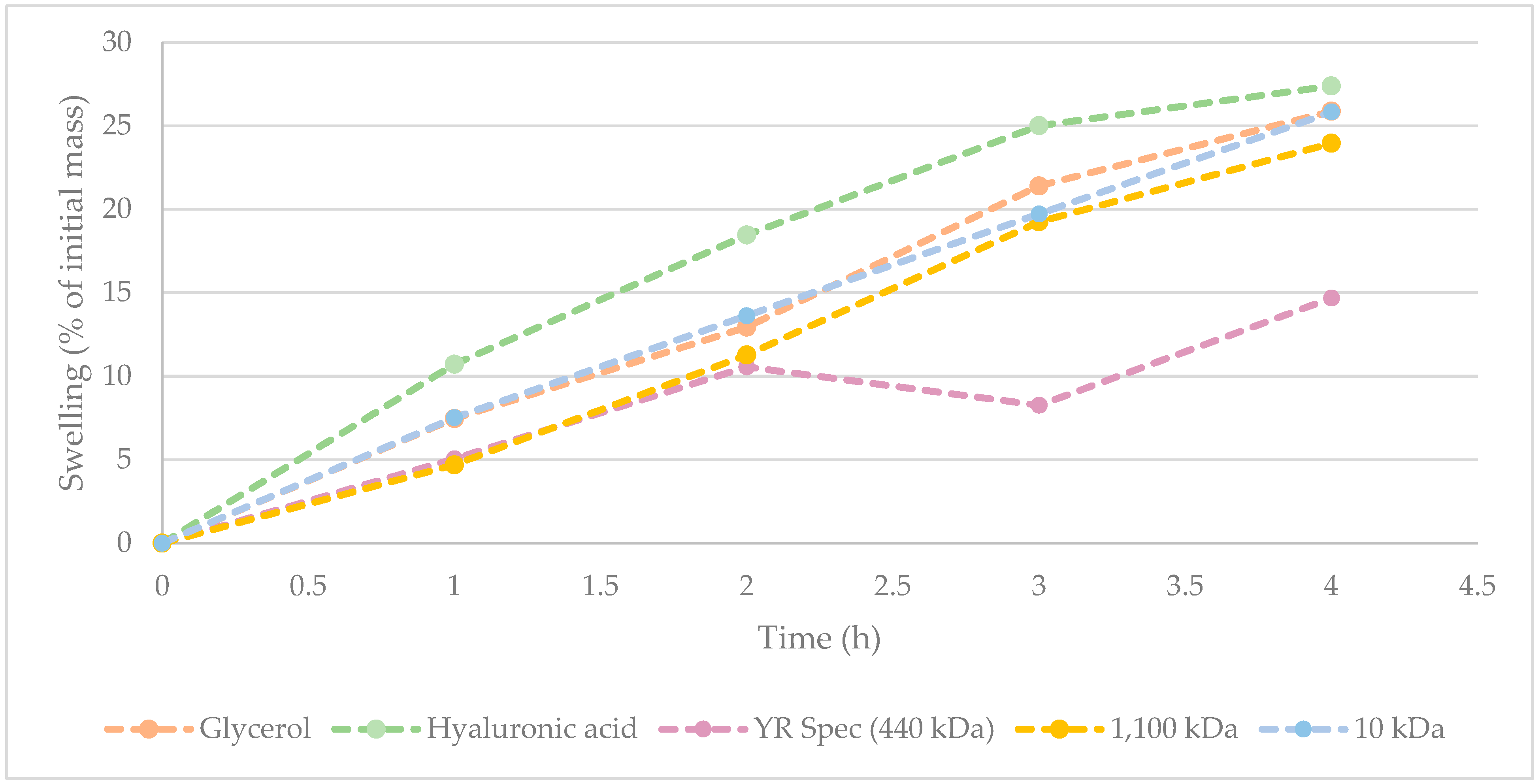
Figure 2.
Figure 2 suggests that hyaluronic acid displays the greatest hygroscopicity across from 0 to 4 h. Interestingly, hygroscopic behaviour of 10 kDa commercial sample of γ-PGA was comparable to that of glycerol. At t = 4 h, there was only a marginal difference between water absorbed by hyaluronic acid, glycerol, and 10 kDa commercial sample of γ-PGA. Supporting the data observed within
Table 6, it appears that variation in water absorbance between 10 kDa and 1100 kDa commercial samples of γ-PGA from the same supplier was marginal. Interestingly, the hygroscopicity displayed by YR Spec commercial γ-PGA was significantly lower compared to other samples at t = 3 h and 4 h, suggesting that the amorphous properties of this material might be less suitable for prolonged hygroscopic behaviour.
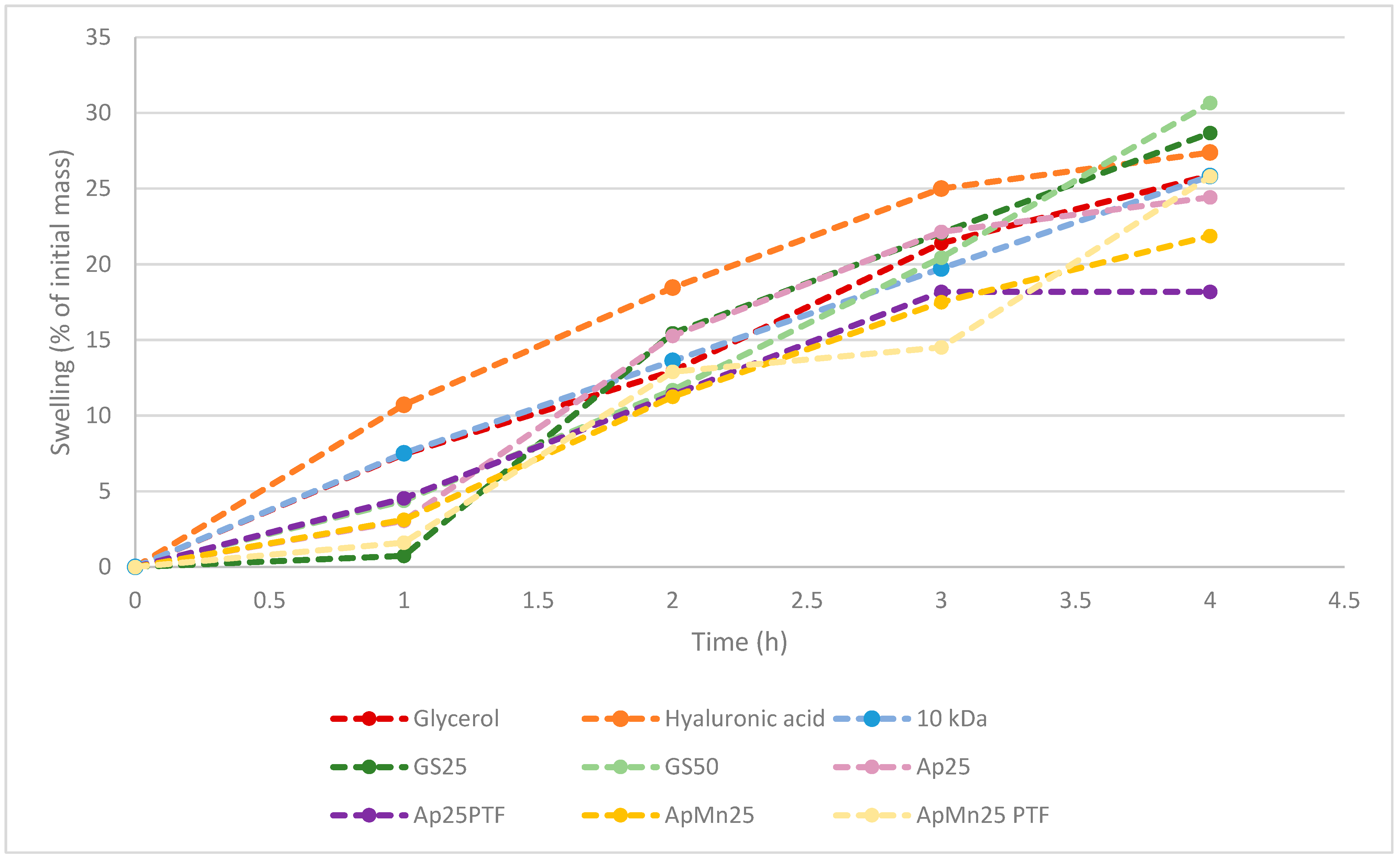
In line with the previous observation, it is evident with both standard and algal substrates that the higher crystallinity within the structure is more favourable for interaction with water compared to amorphous structures. In fact, GS25, ApMn25PTF, Ap25, Ap25PTF, and ApMn25 all displayed lower initial tendencies to absorb water compared to GS50 samples. Depending on the sample, the discrepancy between crystalline and amorphous hygroscopicity was somewhat reduced over time. Notably, it appears that the t = 4 h total hygroscopicity was significantly different between standard media GS25 and
Ascophyllum nodosum-derived media. From the data displayed within
Figure 3, it can be hypothesised that initial hygroscopicity was largely dictated by material crystallinity, whereas as time passed, hygroscopicity was further affected by polymeric molecular weight. Notably, hygroscopicity recorded at t = 4 h for both GS25 and GS50 was higher compared to that of hyaluronic acid. The data obtained for ApMn25PTF suggest only marginally lower water absorption at t = 4 h compared to hyaluronic acid.
Biocompatibility of γ-PGA samples were tested in the widely employed HaCaT human keratinocyte model. HaCaT cells were incubated with γ-PGA samples dissolved in medium at a range of concentrations from 2% downwards. Percentage viability was then measured by the XTT colorimetric assay based on metabolic activity. γ-PGA produced from standard medium (GS25) and
A. nodosum-derived medium (Ap25) both showed comparable biocompatibility to the commercial YR Spec standard γ-PGA. For all γ-PGA samples tested, some effects on viability were observed at the highest concentration tested (2%), but these were relatively minor. Some of these minor effects could be a result of physical accumulation of material on the cell rather than detrimental biochemical interaction between γ-PGA and the cell (
Figure 4).
UV protection is another key factor within the cosmetics industry [
42]. γ-PGA has not yet been reported to have either UVA or UVB photoprotective properties or photosensitisation [
43] properties. For this reason, the clonogenic survival assay was used to test whether γ-PGA could affect killing of HaCaT keratinocytes by UVA or UVB. It was observed that incubation of cells in medium containing commercial γ-PGA (YR Spec) at 1% did not lead to either photoprotection against or photosensitisation towards either UVA (50 kJ/m
2) or UVB (800 J/m
2) (
Figure 5). Although this suggests that γ-PGA is safe for inclusion in skin creams, it did not confirm the ability of these particular commercial grades to improve survival rates of keratinocytes to dangerous UV rays. Photoprotective mechanisms against UV by chemical agents can occur either by physically absorbing harmful photons [
44] or through biochemically counteracting damaging processes (e.g., through antioxidant activity) [
45]. These data suggest that commercial γ-PGA with 440 kDa when incorporated in cell culture medium at the concentration tested does not provide either biochemical protection or a sufficient physical barrier to prevent harmful effects of UV rays.
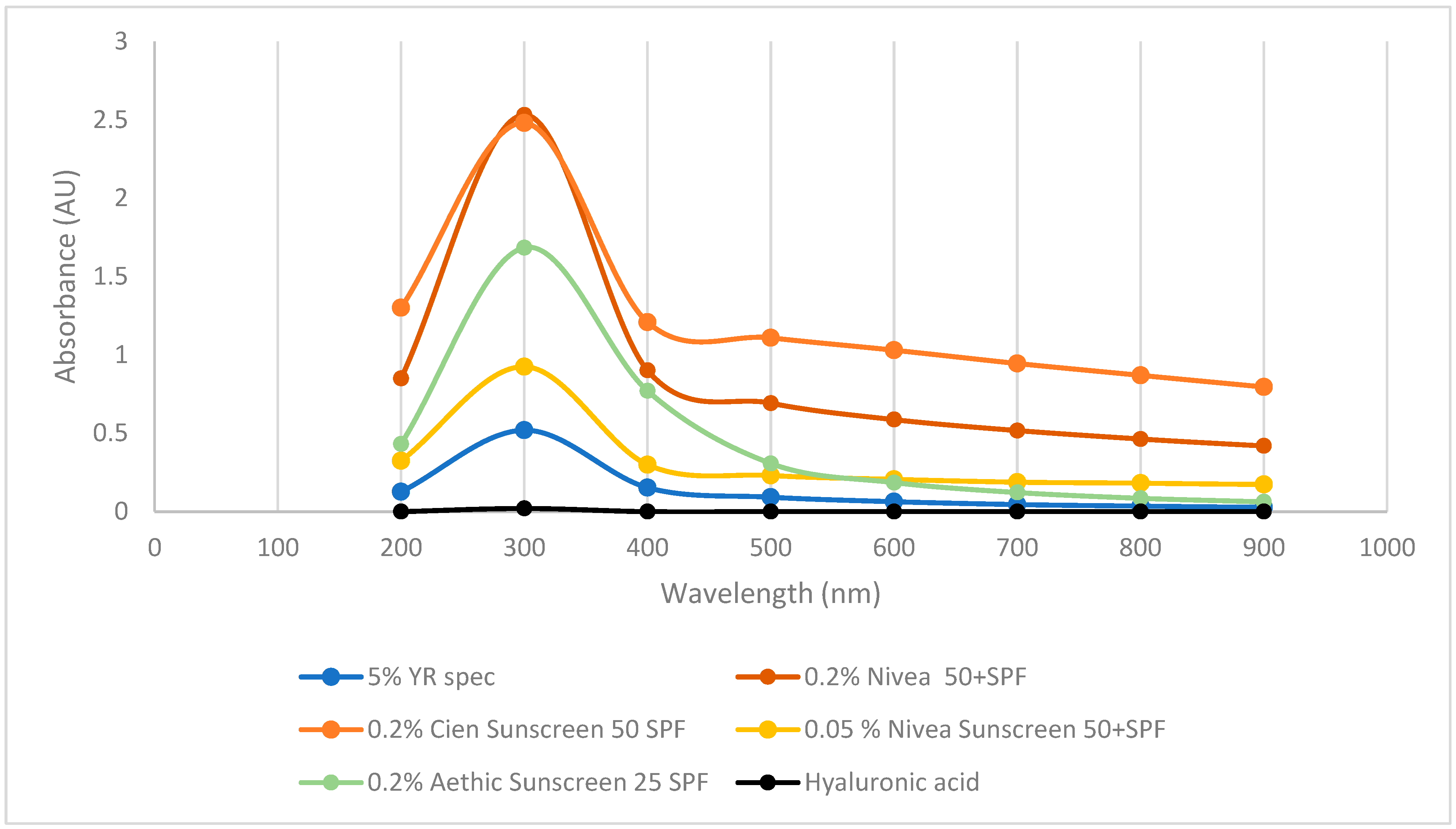
To further investigate whether γ-PGA has photoprotective properties, its UV-VIS spectrum was compared to hyaluronic acid and a range of suncreams. As previously reported in the literature, commercial hyaluronic acid did not display any absorbance across the tested wavelengths. Differently, three commercially available sunscreens displayed strong absorbance at 300 and 400 nm key ranges for blocking UVA and UVB (
Figure 6). Interestingly, commercially available YR Spec γ-PGA did show significant absorption at 300 nm and some absorption at 400 and 500 nm; however, the concentration of this component was 10 times higher compared to commercially available sunblock.
The potential efficacy of defined media and complex algal media γ-PGA as sunblock was further assessed; the results are summarised within
Figure 7.
Surprisingly, the produced γ-PGA, when dissolved at 0.2 or 0.05% concentrations, displayed significantly higher absorbance (300, 400 nm) compared to commercially available γ-PGA at 5%. Interestingly, at analogous concentration (0.2%), GS25PTF displayed higher absorbance at 300 nm compared to commercial Nivea 50+ SPF, Cien 50 SPF, and Aethic 25 SPF. Differently from hygroscopicity, it appears that UV absorption is more effective through amorphous material (GS25PTF, AP25PTF, ApMn25, AP25) compared to crystalline material (GS50, YR spec). Such observations are particularly surprising as, in the case of solid lipid nanoparticles, most of the skin protection capacity is a direct result of its crystalline structure [
46]. However, it has been described by [
47] that a superior UV protection can be achieved when an active is uniformly present within an amorphous polymeric core. This does suggest that part of the observed UV protection can be conferred by small actives associated physically with γ-PGA and not the γ-PGA itself. In fact, it has been widely reported that extracts from both brown and red seaweed do present strong UV protection properties, thanks to the abundance in microsporine-like amino acids [
46,
48]. Ap0 material also did not display particularly good UVA absorption despite being amorphous. It is possible that significant amounts of algal contaminants are present within the precipitate solution, and therefore these could impede on the three-dimensional distribution of the γ-PGA chains and ultimately on the capacity of this sample to absorb UVA.
Further, to the observed UV absorbing properties of γ-PGA (
Figure 6 and
Figure 7), spectroradiometry was used to investigate the potential UV screening effects of two creams formulated with GS25 and Ap25 γ-PGA, separately (
Figure 8). Both creams showed good potential for UV protection, decreasing transmittance most significantly in the sub-400 nm region. Notably, and consistent with the data shown in
Figure 7, the cream prepared using
A. nodosum-derived medium showed more promising optical properties than the cream containing γ-PGA prepared from GS25 medium, screening out half or more of the radiation at wavelengths in the UVB range when applied at 2.5 mg/cm
2 (
Figure 8). For example, the T
315 for GS25 γ-PGA cream was 84%, while for the Ap25cream, it was 51%. Provided that the molecular weight of GS25 was significantly higher than that of Ap25, a reasonable explanation for the reduction in transmittance is that non-γ-PGA components of Ap25 are responsible for the screening effect. This is reasonable, as γ-PGA could be strongly interacting with some algal compounds, which are not fully removed during purification.
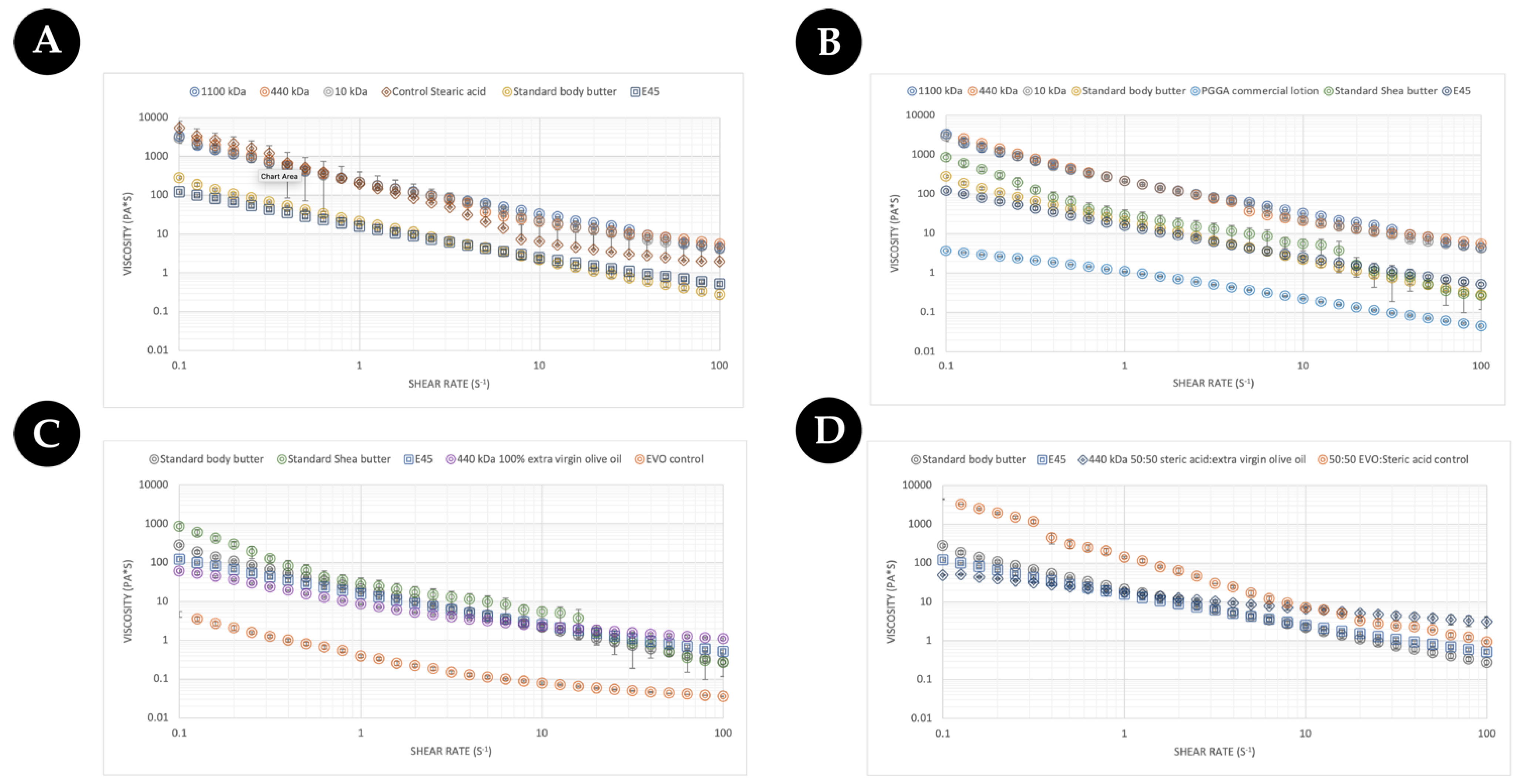
From a consumer’s point of view, the feeling of the cream/lotion/moisturiser constitutes a key part of the purchasing decision. To this end, the rheological properties of different commercial γ-PGA were assessed against an array of commercially available creams, body butters, and lotions to evaluate potential excipient integration/substitution based upon the final desired formulation. Initially, the three types of commercially available γ-PGA types were formulated with steric acid as a base material and compared to standard body butter and E45 cream. The data are summarised within (
Figure 9). As expected, the flow profile of the standard body butter and the E45 cream were very similar. As was further expected, all tested creams showed shear thinning behaviour as the viscosity decreased with the increased shear rate [
49]. Overall, the rate at which the viscosity decreased was greater for creams when compared to the lotion (
Figure 9B) and Evo control (
Figure 9C), which are less sensitive to the change in shear rate. Surprisingly the flow behaviour of steric-acid-based commercial γ-PGA samples was comparable to that of the controls but significantly different to the flow profile of commercial creams. Interestingly the behaviour of steric acid was similar to that of γ-PGA creams at shear rates below 10 (s
−1), after which the viscosity decreased significantly from 5000 (Pa*s) to approximately 9 (Pa*s) at 100 (s
−1). The viscosity at about 100 (s
−1) of shear rate has been associated with the ease of rubbing a cream [
50], and lower viscosity at a higher shear rate suggests a better spreadability upon topical application. These data suggest that the main component of the cream is significantly more viscous compared to commercial creams and that γ-PGA controls maintain the viscosity significantly higher at higher shear rates compared to the steric acid control, which would have a better spreadability.
Regardless, to better understand the variation between γ-PGA-based formulations and more cosmetic formats, the flow profile was further compared with a commercial γ-PGA-based lotion sold by Boots, UK, and another commercially available butter (
Figure 9B). The γ-PGA lotion displayed approximately Log 4 less viscosity across all shear rates compared to steric-acid-based γ-PGA formulations. Differently, the commercially available shea butter presented a rheological profile in between that of steric-acid-based γ-PGA, standard body butter, and E45.
In an attempt to further elucidate the effect of γ-PGA on the rheological of creams with different base lipids, the steric acid component was completely replaced with extra virgin olive oil. For comparison purposes, only one of the three commercial γ-PGAs was employed to formulate this new cream formulation as no significant variation in rheological profile between different commercial γ-PGAs with the same base materials were observed (
Figure 9A,B). It has been previously described within the literature the beneficial effect of extra virgin olive oil (EVO) on the skin [
51]. The effect of extra virgin γ-PGA as a rheological modifier to improve the sensation of the cream on the skin was further investigate (
Figure 9C). It can be observed from
Figure 9C that at lower shear rates, EVO-based formulations presented lower viscosity compared to E45 and body creams; differently, at higher shear rates, the viscosity became comparable or higher compared to both E45 and body creams, suggesting shear-dependent thickening commonly observed with gums, cyclodextrin, and the resulting interaction of the two species [
52,
53,
54]. From our observations in
Figure 9A–C, if the consistency of creams or butters is required, it could be further improved by substituting the lipid component (
Figure 9C) or by tailoring the ratio between more viscous steric-acid-like components and less viscous oily components such as EVO or similar (
Figure 9D).
To further validate the hypothesis on the ratio of steric acid and EVO and the resulting flow profile, a 50:50 EVO/steric acid formulation was assessed within
Figure 9D. Surprisingly, the variation in rheological profile between an EVO-only formulation and a 50:50 EVO/steric acid formulation was not as expected. Firstly, the initial viscosity of the cream was not significantly different between the two. In fact, the variation was observed in the delta viscosity between low and high shear rates, where, with the EVO-only formulation, this was quite significant and, differently from the 50:50 EVO/steric acid formulation, the viscosity was maintained quite constantly throughout the shear spectrum. This was particularly surprising as it was the opposite of what was observed with the EVO-only rheological profile (see
Figure 9C) in which the delta viscosity was lower across the shear spectrum compared to the higher delta viscosity for the 50:50 EVO/steric acid mixture, as displayed in
Figure 9D. Such variation in properties underlies the crucial effect that formulation components, including γ-PGA, exhibit on the overall formulation as a function of shear rates. It is therefore inherently more complex to create formulations which such active components compared to smaller-molecule-dominant formulations.
Further to investigating the impact of the ratio between more viscous and less viscous components, the effect of different lipids was evaluated. For instance, within
Figure 9D, we summarise the effect of beeswax on the overall cream formulation compared to beeswax- and EVO-based formulations. Interestingly, beeswax displayed even lower delta viscosity as a function of shear rate throughout the spectra. It will be therefore crucial to further investigate the behaviour of intra-formulation (gum Arabic, cyclodextrin, γ-PGA) with the lipid component and their effect upon the rheological profile of the cream.