Innovative Application of Salophen Derivatives in Organic Electronics as a Composite Film with a Poly(3,4-Ethylenedioxythiophene)-poly(styrenesulfonate) Matrix
Abstract
1. Introduction
2. Materials and Methods
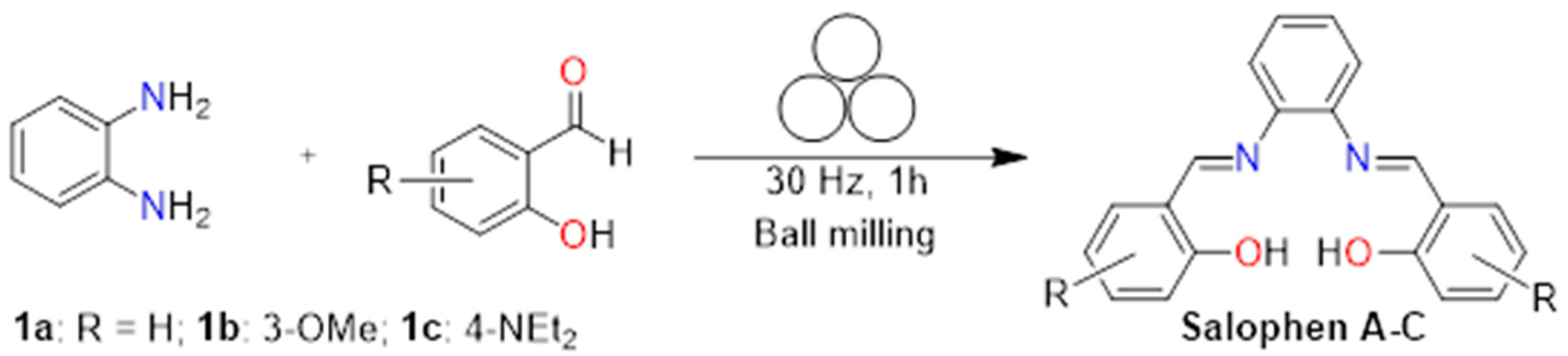
2.1. Synthesis and Characterization of Salophen Compounds
2.2. Composite Film Manufacturing
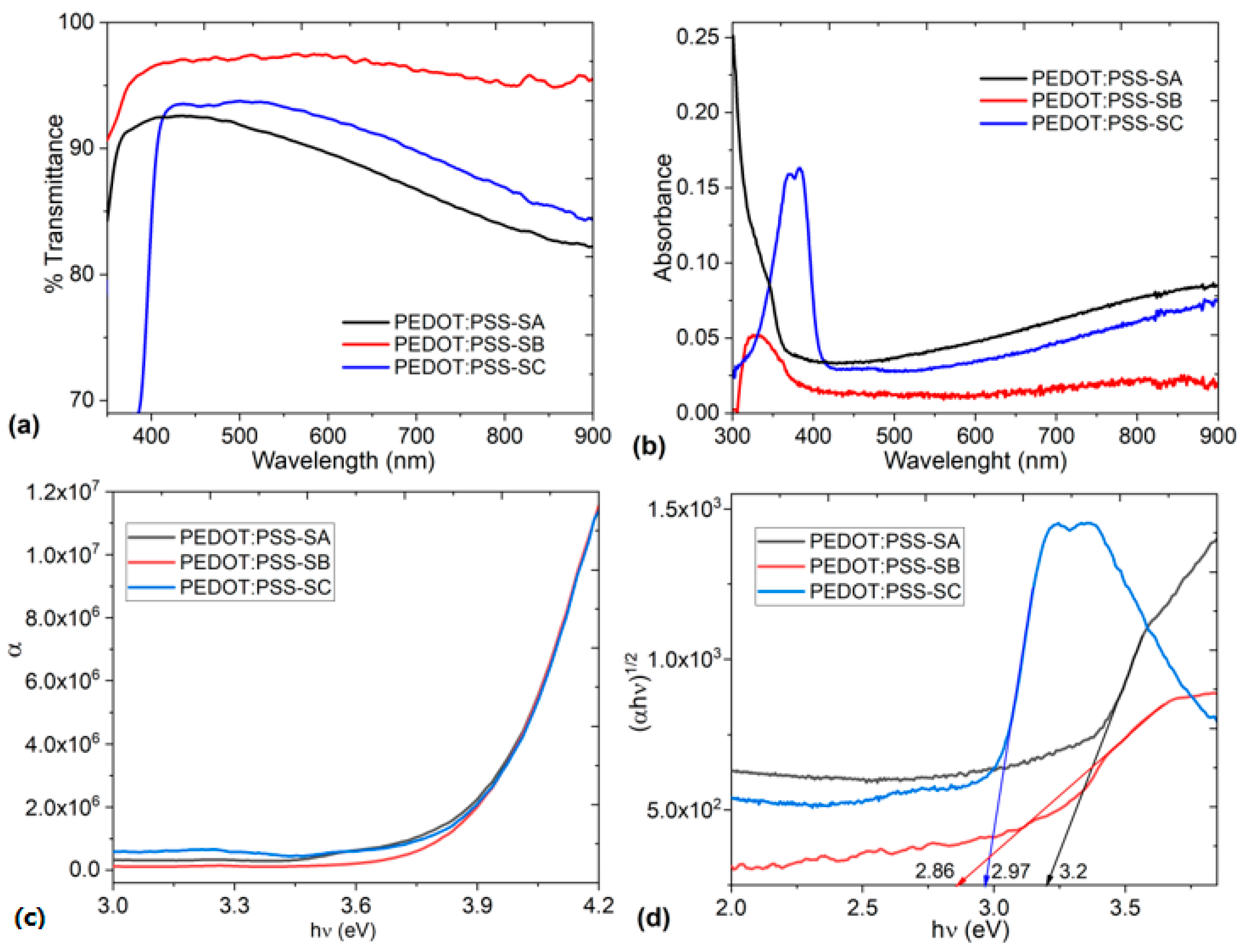
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chakraborty, A.; Herrera, D.; Fallen, P.; Hall, D.; Bampton, N.; Olivero, T.; Orlowski, M. Conductive organic electrodes for flexible electronic devices. Nature 2023, 13, 4125. [Google Scholar] [CrossRef]
- Cheng, X.-Y.; Peng, S.-Q.; Wu, L.-X.; Sun, Q.-F. 3D-printed stretchable sensor based on double network PHI/PEDOT:PSS hydrogel annealed with cosolvent of H2O and DMSO. Chem. Eng. J. 2023, 470, 144058. [Google Scholar] [CrossRef]
- Yao, X.S.; Cui, Y. A PEDOT:PSS functionalized capacitive sensor for humidity. Measurement 2020, 160, 107782. [Google Scholar] [CrossRef]
- Bhat, M.A.; Rather, R.A.; Shalla, A.H. PEDOT and PEDOT:PSS conducting polymeric hydrogels: A report on their emerging applications. Synth. Met. 2021, 273, 116709. [Google Scholar] [CrossRef]
- Furlani, F.; Montanari, M.; Sangiorgi, N.; Saracino, E.; Campodoni, E.; Sanson, A.; Benfenati, V.; Tampieri, A.; Panseria, S.; Sandri, M. Electroconductive and injectable hydrogels basedon gelatin and PEDOT:PSS for a minimally invasive approach in nervous tissue regeneration. Biomater. Sci. 2022, 10, 2040. [Google Scholar] [CrossRef]
- Li, W.; Cheng, N.; Cao, Y.; Zhao, Z.; Xiao, Z.; Zi, W.; Sun, Z. Boost the performance of inverted perovskite solar cells with PEDOT:PSS/Graphene quantum dots composite hole transporting layer. Org. Electron. 2020, 78, 105575. [Google Scholar] [CrossRef]
- Bhattacharjee, P.; Ahearne, M. Fabrication and Biocompatibility of Electroconductive Silk Fibroin/PEDOT: PSS Composites for Corneal Epithelial Regeneration. Polymers 2020, 12, 3028. [Google Scholar] [CrossRef]
- Han, J.W.; Park, J.; Kim, J.H.; Nurmaulia Entifar, S.A.; Prameswati, A.; Wibowo, A.F.; Kim, S.; Lim, D.C.; Lee, J.; Moon, M.-W.; et al. Stretchable and Conductive Cellulose/Conductive Polymer Composite Films for On-Skin Strain Sensors. Materials 2022, 15, 5009. [Google Scholar] [CrossRef]
- Li, B.; Cai, C.Y.; Liu, Y.; Wang, F.; Yang, B.; Li, Q.K.; Zhang, P.X.; Deng, B.; Hou, P.F.; Liu, W.S. Ultrasensitive mechanical/thermal response of a P(VDF-TrFE) sensor with a tailored network interconnection interface. Nature 2023, 14, 4000. [Google Scholar] [CrossRef]
- Zhu, Y.F.; Zhao, B.B.; Cheng, Z.F.; Yu, T.; Li, Y. Efficient flame-retardant and multifunctional conductive flax fabric for intelligent fire protection and human motion monitoring. Chem. Eng. J. 2023, 474, 145610. [Google Scholar] [CrossRef]
- Kim, S.; Kim, H.S.; Park, Y.D. Doped PEDOT:PSS electrodes, patterned through wettability control, and their effects on the electrical properties of polymer thin film transistors. Org. Electron. 2016, 30, 296–301. [Google Scholar] [CrossRef]
- Jang, H.-K.; Kim, J.; Park, J.-S.; Moon, J.B.; Oh, J.; Lee, W.-K.; Kang, M.-G. Synthesis and Characterization of a Conductive Polymer Blend Based on PEDOT:PSS and Its Electromagnetic Applications. Polymers 2022, 14, 393. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, E.; Kollath, V.O.; Karan, K. The key mechanism of conductivity in PEDOT:PSS thin films exposed by anomalous conduction behaviour upon solvent-doping and sulfuric acid post-treatment. J. Mater. Chem. 2020, 8, 383–385. [Google Scholar] [CrossRef]
- Olivares, A.J.; Cosme, I.; Sanchez-Vergara, M.E.; Mansurova, S.; Carrillo, J.C.; Martinez, H.E.; Itzmoyotl, A. Nanostructural Modification of PEDOT:PSS for High Charge Carrier Collection in Hybrid Frontal Interface of Solar Cells. Polymers 2019, 11, 1034. [Google Scholar] [CrossRef] [PubMed]
- Lenz, A.; Kariis, H.; Pohl, A.; Persson, P.; Ojamäe, L. The electronic structure and reflectivity of PEDOT:PSS from density functional theory. Chem. Phys. 2011, 384, 44–51. [Google Scholar] [CrossRef]
- Mantione, D.; Del Agua, I.; Sanchez-Sanchez, A.; Mecerreyes, D. Poly(3,4-ethylenedioxythiophene) (PEDOT) Derivatives: Innovative Conductive Polymers for Bioelectronics. Polymers 2017, 9, 354. [Google Scholar] [CrossRef]
- Xu, H.; Zhao, X.; Yang, G.; Ji, X.; Zhang, X.; Li, L.; Wu, B.; Ouyang, X.; Ni, Y.; Chen, L.; et al. Modification of PEDOT:PSS towards high-efficiency OLED electrode via synergistic effect of carboxy and phenol groups from biomass derivatives. Chem. Eng. J. 2022, 430, 133014. [Google Scholar] [CrossRef]
- Gaur, A.; Shrivastava, B.D.; Srivastav, K.; Prasad, J.; Singh, S.K. XAFS investigations of copper(II) complexes with tetradentate Schiff base ligands. X-ray Spectrom. 2012, 41, 384–392. [Google Scholar] [CrossRef]
- Türkoglu, G.; Berber, H.; Dal, H.; Ögretir, C. Synthesis, characterization, tautomerism and theoretical study of some new Schiff base derivatives. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 79, 1573–1583. [Google Scholar] [CrossRef]
- Mota, V.Z.; De Carvalho, G.S.G.; Corbi, P.P.; Bergamini, F.R.G.; Formiga, A.L.B.; Diniz, R.; Freitas, M.C.R.; Da Silva, A.D.; Cuin, A. Crystal structure and theoretical stidies of keto-enol isomerism of N,N’-bis(salicylidene)-o-phenylenediamine (salophen). Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 99, 110–115. [Google Scholar] [CrossRef]
- Asatkar, A.K.; Tripathi, M.; Asatkar, D. Salen and Related Ligands. In Stability and Applications of Coordination Compounds; Srivastva, A.N., Ed.; IntechOpen: London, UK, 2020; pp. 99–116. [Google Scholar] [CrossRef]
- Pessoa, J.C.; Correia, I. Salan vs. salen metal complexes in catalysis and medicinal applications: Virtues and pitfalls. Coord. Chem. Rev. 2019, 388, 227. [Google Scholar] [CrossRef]
- Glaser, T.; Gerenkamp, M.; Fröhlich, R. Targeted synthesis of ferromagnetically coupled complexes with modified 1,3,5-trihydroxybenzene ligands. Angew. Chem. Int. Ed. Engl. 2002, 41, 3823. [Google Scholar] [CrossRef] [PubMed]
- Glaser, T.; Heidemeier, M.; Grimme, S.; Bill, E. Targeted ferromagnetic coupling in a trinuclear copper(II) complex: Analysis of the St = 3/2 spin ground state. Inorg. Chem. 2004, 43, 5192. [Google Scholar] [CrossRef] [PubMed]
- Glaser, T.; Heidemeier, M.; Fröhlich, R.; Hildebrandt, P.; Bothe, E.; Bill, E. Trinuclear nickel complexes with triplesalen ligands: Simultaneous occurrence of mixed valence and valence tautomerism in the oxidized species. Inorg. Chem. 2005, 44, 5467. [Google Scholar] [CrossRef]
- Iranzo, O. Manganese complexes displaying superoxide dismutase activity: A balance between different factors. Bioorg. Chem. 2011, 39, 73. [Google Scholar] [CrossRef]
- Erxleben, A. Transition metal salen complexes in bioinorganic and medicinal chemistry. Inorg. Chim. Acta 2018, 472, 40. [Google Scholar] [CrossRef]
- Yuan, G.; Jiang, H.; Zhang, L.; Liu, Y.; Cui, Y. Metallosalen-based crystalline porous materials: Synthesis and property. Coord. Chem. Rev. 2019, 378, 483. [Google Scholar] [CrossRef]
- Crane, A.K.; MacLachlan, M.J. Portraits of porosity: Porous structures based on metal salen complexes. Eur. J. Inorg. Chem. 2011, 2012, 17. [Google Scholar] [CrossRef]
- Che, C.-M.; Kwok, C.-C.; Lai, S.-W.; Rausch, A.F.; Finkenzeller, W.J.; Zhu, N.; Yersin, H. Photophysical properties and OLED applications of phosphorescent platinum(II) Schiff Base complexes. Chem. Eur. J. 2010, 16, 233. [Google Scholar] [CrossRef]
- Asatkar, A.K.; Senanayak, S.P.; Bedi, A.; Panda, S.; Narayan, K.S.; Zade, S.S. Zn(II) and Cu(II) complexes of a new thiophene based salphen-type ligand: Solution-processable high-performance field-effect transistor materials. Chem. Commun. 2014, 50, 7036. [Google Scholar] [CrossRef]
- Elbert, S.M.; Mastalerz, M. Metal Salen- and Salphen-Containing Organic Polymers: Synthesis and Applications. Org. Mat. 2020, 2, 182. [Google Scholar] [CrossRef]
- Chepurnaya, I.A.; Karuschev, M.P.; Alekseeva, E.V.; Lukyanov, D.A.; Levin, O.V. Redox-conducting polymers based in metal-salen complexes for energy storage applications. Pure Appl. Chem. 2020, 92, 1239. [Google Scholar] [CrossRef]
- Rezaee-Adriyani, T.; Ensafi, A.A.; Rezaei, B. Flexible and sewable electrode based on Ni-Co@PANI-salphen composite-coated on textiles for wearable supercapacitor. Sci. Rep. 2023, 13, 19772. [Google Scholar] [CrossRef]
- Basoccu, F.; De Luca, L.; Porcheddu, A. Mechanochemistry in Organic Synthesis: An Italian Journey through Innovations. Eur. J. Org. Chem. 2024, 27, e202400425. [Google Scholar] [CrossRef]
- Tigineh, G.T. Electronic Impacts on the Solvent-Free Mechanochemical Synthesis of Salophen Ligands in Ball Mill. J. Chem. 2022, 2022, 1418032. [Google Scholar] [CrossRef]
- Chen, T.; Cai, C. Selective oxidation of benzyl alcohols to aldehydes with a salophen copper(II) complex and ter-butyl hydroperoxide at room temperature. Synth. Commun. 2015, 45, 1334–1341. [Google Scholar] [CrossRef]
- Li, T.T.; Chen, Y.; Li, F.-M.; Zhao, W.-L.; Wang, C.-J.; Lv, X.-J.; Xu, Q.-Q.; Fu, W.-F. Efficient Water Oxidation Catalyzed by Mononuclear Ruthenium(II) Complexes Incorporating Schiff Base Ligands. Chem. A Eur. J. 2014, 20, 8054–8061. [Google Scholar] [CrossRef]
- Wu, W.; Sun, J.; Ji, S.; Wu, W.; Zhao, J.; Guo, H. Tuning the emissive triplet excited states of platinum(II) Schiff base complexes with pyrene, and application for luminescent oxygen sensing and triplet–triplet-annihilation based up conversions. Dalton Trans. 2011, 40, 11550–11561. [Google Scholar] [CrossRef]
- More, M.S.; Pawal, S.B.; Lolage, S.R.; Chavan, S.S. Syntheses, structural characterization, luminescence and optical studies of Ni(II) and Zn(II) complexes containing salophen ligand. J. Mole. Struct. 2017, 1128, 419–427. [Google Scholar] [CrossRef]
- Abdolmaleki, A.; Tavakol, H.; Molavian, M.R.; Firouz, K. Synthesis, FT-IR, NMR and DFT analysis of a new salophen based on diaminophenazine moiety. J. Mole. Struct. 2014, 1062, 44–47. [Google Scholar] [CrossRef]
- Abdulsalam, S.; Idris, S.O.; Shallangwa, G.A.; Onu, A.D. Reaction of N, N1-phenylenebis(salicyalideneiminato)cobalt(III) and l-cysteine in mixed aqueous medium: Kinetics and mechanism. Heliyon 2020, 6, e03850. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Jamal, R.; Zhang, L.; Wang, M.; Abdiryim, T. The structure and properties of PEDOT synthesized by template-free solution method. Nanoscale Res. Lett. 2014, 9, 557. [Google Scholar] [CrossRef] [PubMed]
- Yeon, C.; Kim, G.; Lim, J.W.; Yun, S.J. Highly conductive PEDOT:PSS treated by sodium dodecyl sulfate for stretchable fabric heaters. RSC Adv. 2017, 7, 5888. [Google Scholar] [CrossRef]
- Ouyang, J.; Chu, C.-W.; Chen, F.-C.; Xu, Q.; Yang, Y. High-Conductivity Poly(3,4-ethylenedioxythiophene):Poly(styrene sulfonate) Film and Its Application in Polymer Optoelectronic Devices. Adv. Funct. Mater. 2005, 15, 203–208. [Google Scholar] [CrossRef]
- Han, Y.-K.; Chang, M.-Y.; Huang, W.-Y.; Pan, H.-Y.; Ho, K.-S.; Hsieh, T.-H.; Pan, S.-Y. Improved Performance of Polymer Solar Cells Featuring One-Dimensional PEDOT Nanorods in a Modified Buffer Layer. J. Electrochem. Soc. 2011, 158, K88. [Google Scholar] [CrossRef]
- Cho, M.S.; Yun, Y.Y.; Nam, J.D.; Son, Y.; Lee, Y. Effect of magnetic field on electrochemical polymerization of EDOT. Synth. Met. 2008, 158, 1043–1046. [Google Scholar] [CrossRef]
- Pasha, A.; Khasim, S.; Khan, F.A.; Dhananjaya, N. Fabrication of gas sensor device using poly (3, 4-ethylenedioxythiophene)-poly (styrenesulfonate)-doped reduced graphene oxide organic thin films for detection of ammonia gas at room temperature. Iran. Polym. J. 2019, 28, 183–192. [Google Scholar] [CrossRef]
- Aguirre-Macías, Y.P.; Sánchez-Vergara, M.E.; Monzón-González, C.R.; Cosme, I.; Corona-Sánchez, R.; Álvarez-Bada, J.R.; Álvarez-Toledano, C. Deposition and post-treatment of promising poly(3,4-ethylenedioxythiophene)-polystyrene sulfonate composite films for electronic applications. J. Polym. Res. 2021, 28, 478. [Google Scholar] [CrossRef]
- Cho, H.; Cho, W.; Kim, Y.; Lee, J.-G.; Kim, J.H. Influence of residual sodium ions on the structure and properties of poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate). RSC Adv. 2018, 8, 29044–29050. [Google Scholar] [CrossRef]
- Ouyang, J. “Secondary doping” methods to significantly enhance the conductivity of PEDOT:PSS for its application as transparent electrode of optoelectronic devices. Displays 2013, 34, 423–436. [Google Scholar] [CrossRef]
- De Kok, M.M.; Buechel, M.; Vulto, S.; Van de Weijer, P.; Meulenkamp, E.; De Winter, S.; Mank, A.; Vorstenbosch, H.; Weijtens, C.; Van Elsbergen, V. Modification of PEDOT:PSS as hole injection layer in polymer LEDs. Phys. Status Solidi A 2004, 201, 1342–1359. [Google Scholar] [CrossRef]
- Chang, S.H.; Chiang, C.-H.; Kao, F.-S.; Tien, C.-L.; Wu, C.-G. Unraveling the Enhanced Electrical Conductivity of PEDOT:PSS Thin Films for ITO-Free Organic Photovoltaics. IEEE Photon. J. 2014, 6, 8400307. [Google Scholar] [CrossRef]
- Hilal, M.; Han, J.I. Improving the conductivity of PEDOT:PSS to nearly 1 million S/m with graphene on an ITO-glass substrate. Synth. Met. 2018, 245, 276–285. [Google Scholar] [CrossRef]
- Ha, Y.-H.; Nikolov, N.; Pollack, S.K.; Mastrangelo, J.; Martin, B.D.; Shashidhar, R. Towards a Transparent, Highly Conductive Poly(3,4-ethylenedioxythiophene). Adv. Func. Mater. 2004, 14, 615. [Google Scholar] [CrossRef]
- Chou, T.-R.; Chen, S.-H.; Chiang, Y.-T.; Lin, Y.-T.; Chao, C.-Y. Highly conductive PEDOT:PSS films by post-treatment with dimethyl sulfoxide for ITO-free liquid crystal display. J. Mater. Chem. C 2015, 3, 3760–3766. [Google Scholar] [CrossRef]
- Tauc, J. Optical properties and electronic structure of amorphous Ge and Si. Mater. Res. Bull. 1968, 3, 37–46. [Google Scholar] [CrossRef]
- Urbach, F. The long-wavelength edge of photographic sensitivity and of the electronic absorption of solids. Phys. Rev. 1953, 92, 1324. [Google Scholar] [CrossRef]
- Maniruzzaman, M.; Abdur, R.; Kuddus Sheikh, M.A.; Singh, S.; Lee, J. Conductive MoO3–PEDOT:PSS Composite Layer in MoO3/Au/MoO3–PEDOT:PSS Multilayer Electrode in ITO-Free Organic Solar Cells. Processes 2023, 11, 594. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, W.; Wei, Q.; Liu, G.; Yuan, X.; Pen, H.; Zhang, G.; Wang, R.; Wang, C.; Yang, Y. Effect of Au@MoS2 Contacted PEDOT:PSS on Work Function of Planar Silicon Hybrid Solar Cells. Adv. Mater. Interfaces 2023, 10, 2300187. [Google Scholar] [CrossRef]
- Kirbiyik, C. Tuning the electrical and morphological characteristics of PEDOT:PSS films through mixed cosolvent addition and performance of their polymer solar cells. Turk. J. Phys. 2019, 43, 563–575. [Google Scholar] [CrossRef]
- Sánchez-Vergara, M.E.; Motomochi-Lozano, J.D.; Cosme, I.; Hamui, L.; Olivares, A.J.; Galván-Hidalgo, J.M.; Gómez, E. Growth of films with seven-coordinated diorganotin (IV) complexes and PEDOT:PSS structurally modified for electronic applications. Semicond. Sci. Technol. 2020, 35, 105016. [Google Scholar]
- Monzón-González, C.R.; Sánchez-Vergara, M.E.; Narváez, W.E.V.; Rocha-Rinza, T.; Hernández, M.; Gómez, E.; Jiménez-Sandoval, O.; Álvarez-Toledano, C. Synthesis and characterization of organotin (IV) semiconductors and their applications in optoelectronics. J. Phys. Chem. Solids 2020, 150, 109840. [Google Scholar]
- Sánchez Vergara, M.E.; Gómez, E.; Toledo Dircio, E.; Álvarez Bada, J.R.; Cuenca Pérez, S.; Galván Hidalgo, J.M.; González Hernández, A.; Hernández Ortega, S. Pentacoordinated Organotin(IV) Complexes as an Alternative in the Design of Highly Efficient Optoelectronic and Photovoltaic Devices: Synthesis and Photophysical Characterization. Int. J. Mol. Sci. 2023, 24, 5255. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Zhong, Z.; Hu, Z.; Luo, Y.; Wang, L.; Wang, J.; Cao, Y. The effect of solvent treatment on the buried PEDOT:PSS layer. Org. Electron. 2017, 43, 9–14. [Google Scholar] [CrossRef]
- Wang, Y.; Luo, Q.; Wu, N.; Wang, Q.; Zhu, H.; Chen, L.; Li, Y.Q.; Luo, L.; Ma, C.-Q. Solution-Processed MoO3:PEDOT:PSS Hybrid Hole Transporting Layer for Inverted Polymer Solar Cells. ACS Appl. Mater. Interfaces 2015, 7, 7170–7179. [Google Scholar] [CrossRef]
- Zhou, Y.; Mei, S.; Sun, D.; Liu, N.; Shi, W.; Feng, J.; Mei, F.; Xu, J.; Jiang, Y.; Cao, X. Improved Efficiency of Perovskite Light-Emitting Diodes Using a Three-Step Spin-Coated CH3NH3PbBr3 Emitter and a PEDOT:PSS/MoO3-Ammonia Composite Hole Transport Layer. Micromachines 2019, 10, 459. [Google Scholar] [CrossRef]
- Yoo, D.; Kim, J.; Kim, J.H. Direct synthesis of highly conductive poly(3,4-ethylenedioxythiophene):poly(4-styrenesulfonate) (PEDOT:PSS)/graphene composites and their applications in energy harvesting systems. Nano Res. 2014, 7, 717–730. [Google Scholar] [CrossRef]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Vergara, M.E.; Jimenez Correa, O.; Ballinas-Indilí, R.; Cosme, I.; Álvarez Bada, J.R.; Álvarez-Toledano, C. Innovative Application of Salophen Derivatives in Organic Electronics as a Composite Film with a Poly(3,4-Ethylenedioxythiophene)-poly(styrenesulfonate) Matrix. Polymers 2024, 16, 2622. https://doi.org/10.3390/polym16182622
Sánchez Vergara ME, Jimenez Correa O, Ballinas-Indilí R, Cosme I, Álvarez Bada JR, Álvarez-Toledano C. Innovative Application of Salophen Derivatives in Organic Electronics as a Composite Film with a Poly(3,4-Ethylenedioxythiophene)-poly(styrenesulfonate) Matrix. Polymers. 2024; 16(18):2622. https://doi.org/10.3390/polym16182622
Chicago/Turabian StyleSánchez Vergara, María Elena, Omar Jimenez Correa, Ricardo Ballinas-Indilí, Ismael Cosme, José Ramón Álvarez Bada, and Cecilio Álvarez-Toledano. 2024. "Innovative Application of Salophen Derivatives in Organic Electronics as a Composite Film with a Poly(3,4-Ethylenedioxythiophene)-poly(styrenesulfonate) Matrix" Polymers 16, no. 18: 2622. https://doi.org/10.3390/polym16182622
APA StyleSánchez Vergara, M. E., Jimenez Correa, O., Ballinas-Indilí, R., Cosme, I., Álvarez Bada, J. R., & Álvarez-Toledano, C. (2024). Innovative Application of Salophen Derivatives in Organic Electronics as a Composite Film with a Poly(3,4-Ethylenedioxythiophene)-poly(styrenesulfonate) Matrix. Polymers, 16(18), 2622. https://doi.org/10.3390/polym16182622




