Sorption Mechanisms and Behavior of Benzene Series Compounds by Microplastics in Aqueous Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sorbent Characterization
2.3. Sorption Batch Experiments
2.4. Sorption Models and Statistics
3. Results and Discussion
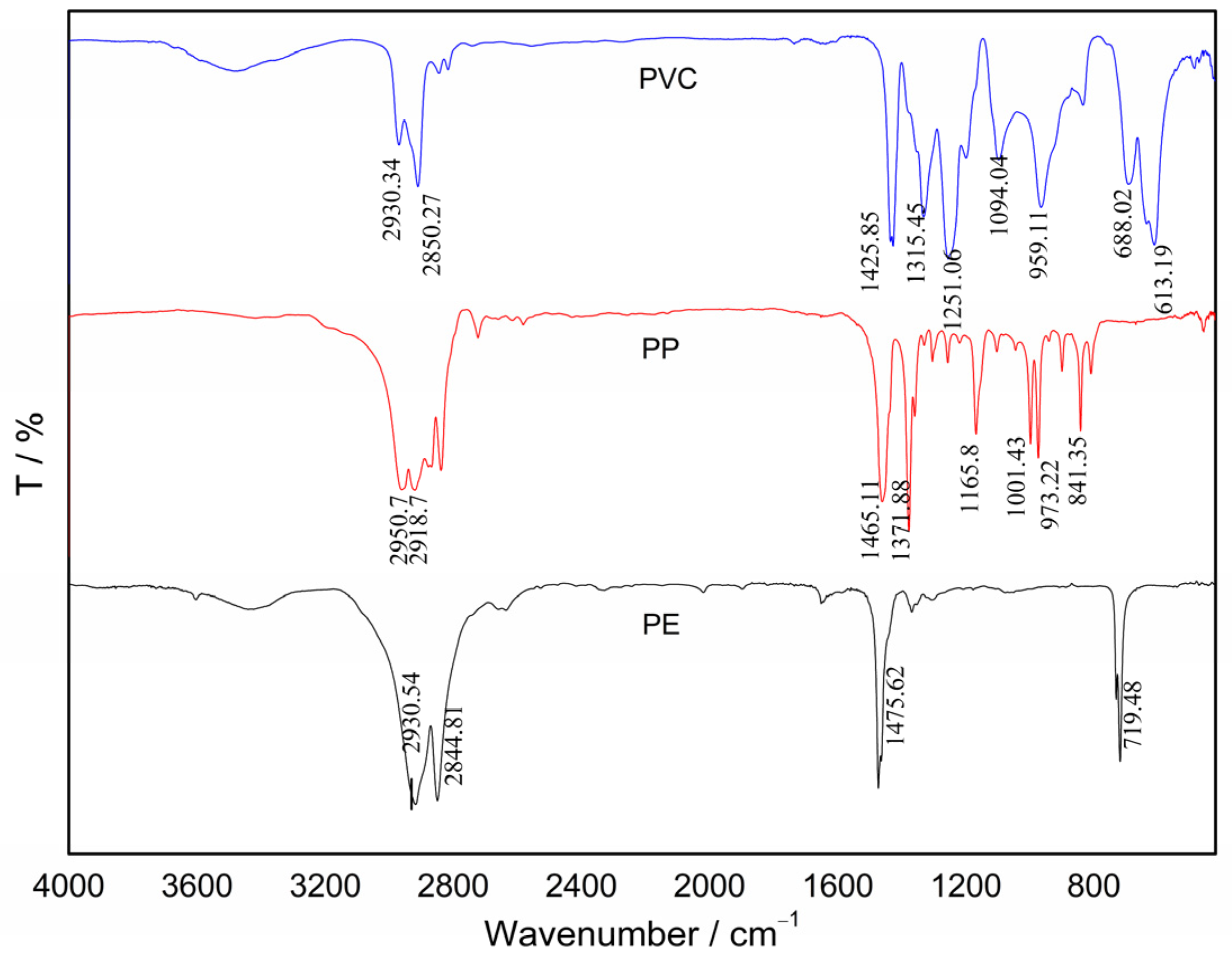
3.1. Sorbent Characterization
3.2. Sorption Kinetics
3.3. Sorption Isotherm Fit
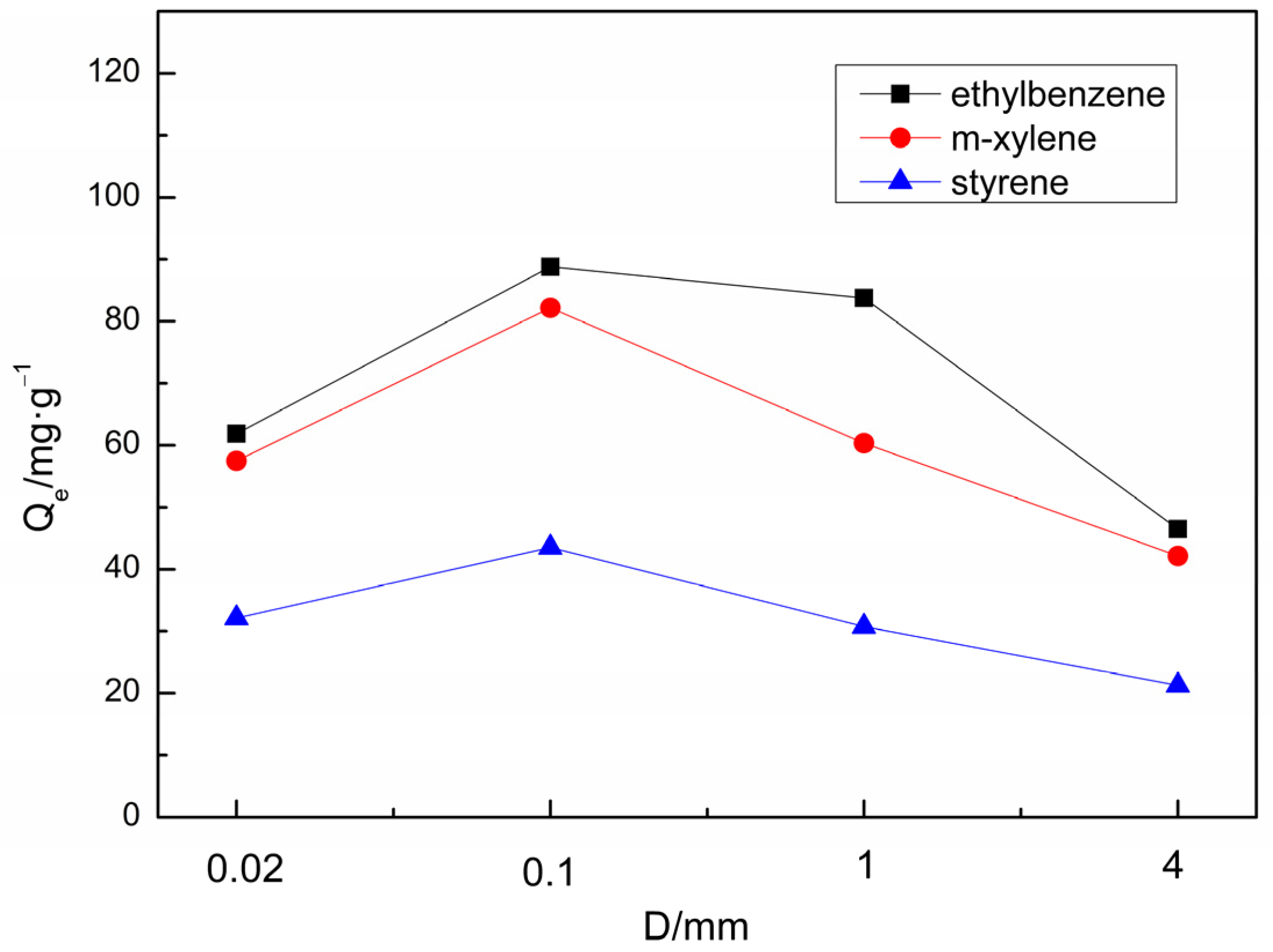
3.4. Influence of Sorbent Properties on Sorption
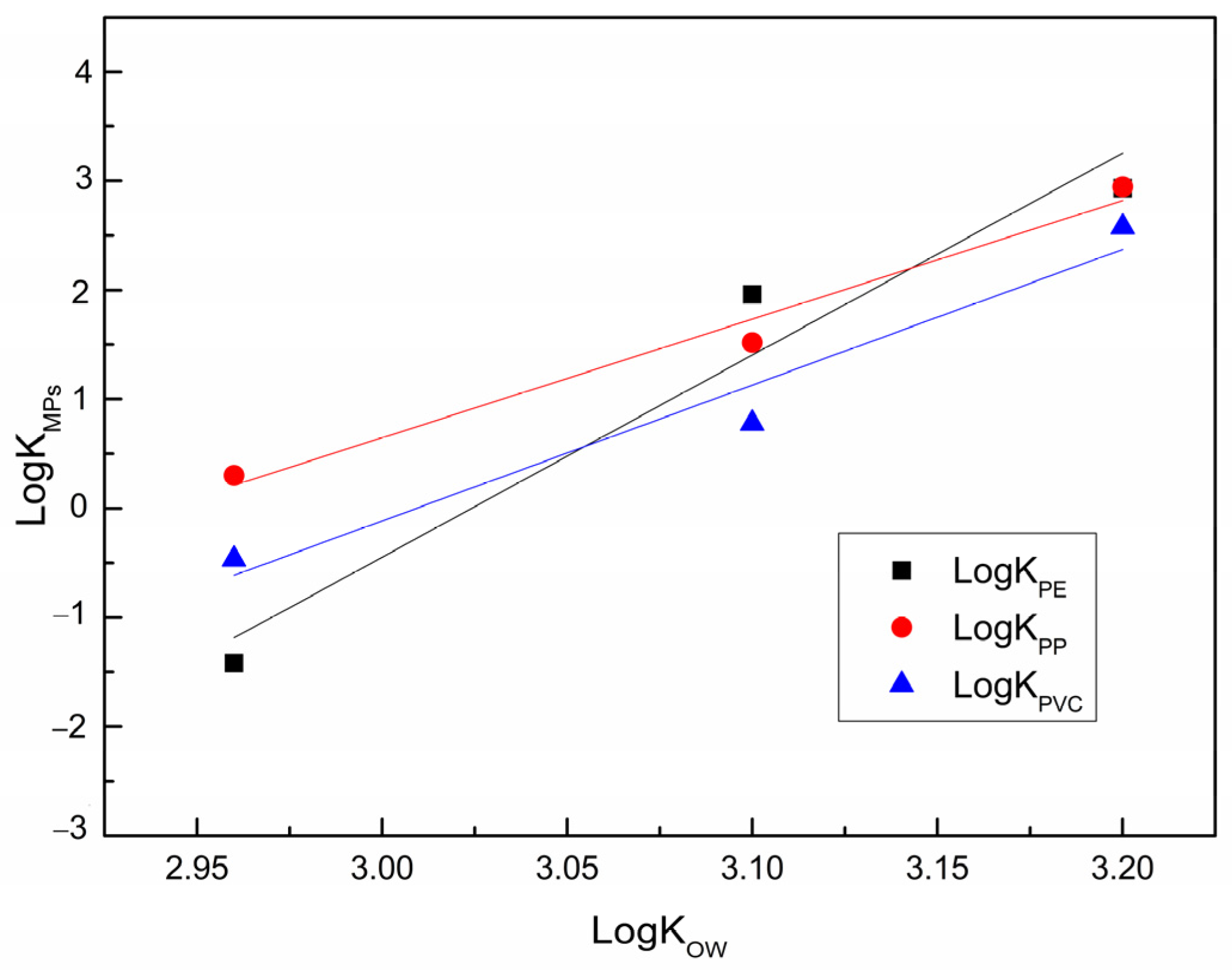
3.5. Influence of Sorbate Properties on Sorption
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Wen, H.; Wang, P.; Mai, L.; Xu, X.; Yu, K.; Zeng, E.Y. Distribution, sorption patterns, and outflows of riverine microplastics-affiliated linear alkylbenzenes and polycyclic aromatic hydrocarbons in a dynamic coastal zone. Environ. Pollut. 2025, 364, 125295. [Google Scholar] [CrossRef]
- Mohsin, M.A.; Abd Zaid, A.H. Microplastic Pollution in the Environment: A Chemical Engineering Perspective on Sources, Fate, and Mitigation Strategies. Polymers 2026, 18, 29. [Google Scholar] [CrossRef] [PubMed]
- Eerkes-Medrano, D.; Thompson, R.C.; Aldridge, D.C. Microplastics in freshwater systems: A review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Res. 2015, 75, 63–82. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, L.; Du, Y.; Yang, X.; He, X.; Zhang, J.; Jia, B. Microplastics in china’s surface water systems: Distribution, driving forces and ecological risk. J. Hazard. Mater. 2025, 485, 136864. [Google Scholar] [CrossRef] [PubMed]
- Rendón-von Osten, J.; Sosa-Rodríguez, E.; Borges-Ramírez, M.M. Ecological risk assessment for metal(oid)s in microplastics and sediments at different depths from cenotes (sinkholes) of Yucatan Mexico. Environ. Res. 2025, 285, 122262. [Google Scholar] [CrossRef]
- Van Cauwenberghe, L.; Devriese, L.; Galgani, F.; Robbens, J.; Janssen, C.R. Microplastics in sediments: A review of techniques, occurrence and effects. Mar. Environ. Res. 2015, 111, 5–17. [Google Scholar] [CrossRef]
- de Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an emerging threat to terrestrial ecosystems. Glob. Change Biol. 2017, 24, 1405–1416. [Google Scholar] [CrossRef]
- Rashid, S.; Majeed, L.R.; Mehta, N.; Radu, T.; Martín-Fabiani, I.; Bhat, M.A. Microplastics in terrestrial ecosystems: Sources, transport, fate, mitigation, and remediation strategies. Euro-Mediterr. J. Environ. Integr. 2025, 10, 2633–2659. [Google Scholar] [CrossRef]
- Jeong, E.; Lee, J.Y.; Redwan, M. Animal exposure to microplastics and health effects: A review. Emerg. Contam. 2024, 10, 100369. [Google Scholar] [CrossRef]
- Duda, A.; Petka, K. The Presence of Micro- and Nanoplastics in Food and the Estimation of the Amount Consumed Depending on Dietary Patterns. Molecules 2025, 30, 3666. [Google Scholar] [CrossRef]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the marine environment: A review of the methods used for identification and quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef]
- Gigault, J.; Halle, A.T.; Baudrimont, M.; Pascal, P.Y.; Gauffre, F.; Phi, T.L.; Hadri, H.E.; Grassl, B.; Reynaud, S. Current opinion: What is a nanoplastic? Environ. Pollut. 2018, 235, 1030–1034. [Google Scholar] [CrossRef]
- Cruz-Salas, A.A.; Velasco-Pérez, M.; Mendoza-Muñoz, N.; Vázquez-Morillas, A.; Beltrán-Villavicencio, M.; Alvarez-Zeferino, J.C.; Ojeda-Benítez, S. Sorption of Total Petroleum Hydrocarbons in Microplastics. Polymers 2023, 15, 2050. [Google Scholar] [CrossRef]
- Nauth, S.D.; Campiglia, A.D. Sorption of Polycyclic Aromatic Sulfur Heterocycles (PASH) on Nylon Microplastics at Environmentally Relevant Concentrations. Molecules 2024, 29, 1653. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, M.; Sha, W.; Wang, Y.; Hao, H.; Dou, Y.; Li, Y. Sorption Behavior and Mechanisms of Organic Contaminants to Nano and Microplastics. Molecules 2020, 25, 1827. [Google Scholar] [CrossRef]
- Bakir, A.; Rowland, S.J.; Thompson, R.C. Transport of persistent organic pollutants by microplastics in estuarine conditions. Estuar. Coast. Shelf Sci. 2014, 140, 14–21. [Google Scholar] [CrossRef]
- Wardrop, P.; Shimeta, J.; Nugegoda, D.; Morrison, P.D.; Miranda, A.; Tang, M.; Clarke, B.O. Chemical pollutants sorbed to ingested microbeads from personal care products accumulate in fish. Environ. Sci. Technol. 2016, 50, 4037–4044. [Google Scholar] [CrossRef]
- Hüffer, T.; Hofmann, T. Sorption of non-polar organic compounds by micro-sized plastic particles in aqueous solution. Environ. Pollut. 2016, 214, 194–201. [Google Scholar] [CrossRef]
- Zhao, L.; Rong, L.; Xu, J.; Lian, J.; Wang, L.; Sun, H. Sorption of five organic compounds by polar and nonpolar microplastics. Chemosphere 2020, 257, 127206. [Google Scholar] [CrossRef]
- Rochman, C.M.; Hoh, E.; Hentschel, B.T.; Kaye, S. Long-term field measurement of sorption of organic contaminants to five types of plastic pellets: Implications for plastic marine debris. Environ. Sci. Technol. 2012, 47, 1646–1654. [Google Scholar] [CrossRef]
- Guo, X.; Wang, X.; Zhou, X.; Kong, X.; Tao, S.; Xing, B. Sorption of four hydrophobic organic compounds by three chemically distinct polymers: Role of chemical and physical composition. Environ. Sci. Technol. 2012, 46, 7252–7259. [Google Scholar] [CrossRef]
- Velzeboer, I.; Kwadijk, C.J.; Koelmans, A.A. Strong sorption of PCBs to nanoplastics, microplastics, carbon nanotubes, and fullerenes. Environ. Sci. Technol. 2014, 48, 4869–4876. [Google Scholar] [CrossRef]
- Shevchenko, N.; Iakobson, O.; Isakov, V.; Zorin, I. Effects of Particle Shape and Surface Structure on the Adsorption Properties of Polystyrene Microplastics. Polymers 2024, 16, 3159. [Google Scholar] [CrossRef]
- Guo, X.; Pang, J.; Chen, S.; Jia, H. Sorption properties of tylosin on four different microplastics. Chemosphere 2018, 209, 240–245. [Google Scholar] [CrossRef]
- Hu, B.; Li, Y.; Jiang, L.; Chen, X.; Wang, L.; An, S.; Zhang, F. Influence of microplastics occurrence on the adsorption of 17β-estradiol in soil. J. Hazard. Mater. 2020, 400, 123325. [Google Scholar] [CrossRef]
- Na, Y.; Weon, S.H.; Lee, G.-W.; Kim, H.J.; Lee, S.H.; Kim, Y.-H.; Kim, J.E.; Kang, G.; Park, S.; Choi, Y.-K. Evaluation of Benzene Adsorption onto Grass-Derived Biochar and Comparison of Adsorption Capacity via RSM (Response Surface Methodology). J. Compos. Sci. 2024, 8, 132. [Google Scholar] [CrossRef]
- Deng, L.; Xi, H.; Wan, C.; Fu, L.; Wang, Y.; Wu, C. Is the petrochemical industry an overlooked critical source of environmental microplastics? J. Hazard. Mater. 2023, 451, 131199. [Google Scholar] [CrossRef]
- Liu, X.; Xu, J.; Zhao, Y.; Shi, H.; Huang, C.-H. Hydrophobic sorption behaviors of 17β-Estradiol on environmental microplastics. Chemosphere 2019, 226, 726–735. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, M.; Zhang, L.; Wang, K.; Yu, X.; Zheng, Z.; Zheng, R. Sorption behaviors of phenanthrene on the microplastics identified in a mariculture farm in Xiangshan Bay, southeastern China. Sci. Total Environ. 2018, 628–629, 1617–1626. [Google Scholar] [CrossRef]
- Ji, Y.; Gao, H.; Sun, J.; Cai, F. Experimental probation on the binding kinetics and thermodynamics of Au(III) onto Bacillus subtilis. Chem. Eng. J. 2011, 172, 122–128. [Google Scholar] [CrossRef]
- Liu, F.; Liu, G.; Zhu, Z.; Wang, S.; Zhao, F. Interactions between microplastics and phthalate esters as affected by microplastics characteristics and solution chemistry. Chemosphere 2019, 214, 688–694. [Google Scholar] [CrossRef]
- Vrentas, J.S.; Duda, J.L. Diffusion in polymer-solvent systems. I. Reexamination of the free-volume theory. J. Appl. Polym. Sci. 1977, 15, 403–416. [Google Scholar] [CrossRef]
- Seidensticker, S.; Grathwohl, P.; Lamprecht, J.; Zarfl, C. A combined experimental and modeling study to evaluate pH-dependent sorption of polar and non-polar compounds to polyethylene and polystyrene microplastics. Environ. Sci. Eur. 2018, 30, 30. [Google Scholar] [CrossRef]
- Endo, S.; Koelmans, A.A. Sorption of hydrophobic organic compounds to plastics in the marine environment: Equilibrium. In Hazardous Chemicals Associated with Plastics in the Marine Environment; Springer: Cham, Switzerland, 2018; pp. 185–204. [Google Scholar]
- Xing, B.S.; Pignatello, J.J. Dual-mode sorption of low-polarity compounds in glassy poly(vinyl chloride) and soil organic matter. Environ. Sci. Technol. 1997, 31, 792–799. [Google Scholar] [CrossRef]
- Llorca, M.; Schirinzi, G.; Martínez, M.; Barceló, D.; Farré, M. Adsorption of perfluoroalkyl substances on microplastics under environmental conditions. Environ. Pollut. 2018, 235, 680–691. [Google Scholar] [CrossRef]
- Wang, F.; Shih, K.M.; Li, X.Y. The partition behavior of perfluorooctanesulfonate (PFOS) and perfluorooctanesulfonamide (FOSA) on microplastics. Chemosphere 2015, 119, 841–847. [Google Scholar] [CrossRef]
- Town, R.M.; van Leeuwen, H.P.; Duval, J.F.L. Sorption kinetics of metallic and organic contaminants on micro- and nanoplastics: Remarkable dependence of the intraparticulate contaminant diffusion coefficient on the particle size and potential role of polymer crystallinity. Environ. Sci. Process. Impacts 2025, 27, 634–648. [Google Scholar] [CrossRef]
- Tumwesigye, E.; Nnadozie, C.F.; Akamagwuna, F.C.; Noundou, X.S.; Nyakairu, G.W.; Odume, O.N. Microplastics as vectors of chemical contaminants and biological agents in freshwater ecosystems: Current knowledge status and future perspectives. Environ. Pollut. 2023, 330, 121829. [Google Scholar] [CrossRef]
- Dong, Y.; Gao, M.; Song, Z.; Qiu, W. Adsorption mechanism of As(III) on polytetrafluoroethylene particles of different size. Environ. Pollut. 2019, 254, 112950. [Google Scholar] [CrossRef]
- Lee, H.; Shim, W.J.; Kwon, J.-H. Sorption capacity of plastic debris for hydrophobic organic chemicals. Sci. Total Environ. 2014, 470–471, 1545–1552. [Google Scholar] [CrossRef]
- Wang, F.; Pan, G.; Li, L. Effects of free iron oxyhydrates and soil organic matter on copper sorption-desorption behavior by size fractions of aggregates from two paddy soils. J. Environ. Sci. 2009, 21, 618–624. [Google Scholar] [CrossRef]






| Compound | MW * | Density * | MV * | LogKaw * | Sw * | LogKow * | Polarizability * |
|---|---|---|---|---|---|---|---|
| ethylbenzene | 106.17 | 0.865–0.869 | 122.251 ± 3.00 | −0.78 | 184 | 3.1 | 14.16 |
| m-xylene | 106.17 | 0.860–0.862 | 121.984 ± 3.00 | −0.74 | 200 | 3.2 | 14.23 |
| styrene | 104.15 | 0.906–1.105 | 115.381 ± 3.00 | −0.95 | 320 | 2.96 | 14.73 |
| MPs | Polarity | Diameter (μm) | Density (g/cm3) | Surface Area (m2/g) | Micropore Volume (mm3/g) | Crystallinity (%) | Glass Transition Temperature (Tg/°C) | Contact Angle (°) | pHPZC |
|---|---|---|---|---|---|---|---|---|---|
| PE | non-polar | 100 | 0.92 | 0.511 | 0.137 | 74.95 | −120~−100 | 134.9 | 1.93 |
| PP | weak-polar | 100 | 0.90 | 0.567 | 0.073 | 44.31 | −20~−10 | 131.8 | 1.24 |
| PVC | strong-polar | 100 | 1.15 | 0.46 | 0.149 | 7.76 | 60~100 | 120.4 | 0.54 |
| MPs | BTEX | Pseudo-First-Order Model | Pseudo-Second-Order Model | Weber-Morris Intraparticle Diffusion Model | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Qm1 | K1 | R12 | Qm2 | K2 | R22 | C | K3 | R32 | ||
| PE | ethylbenzene | 27.42678 | 0.15 | 0.91 | 59.41771 | 0.010949 | 0.99 | 17.76 | 9.47 | 0.96 |
| m-xylene | 42.40431 | 0.19 | 0.92 | 85.10638 | 0.008203 | 0.99 | 22.76 | 14.47 | 0.99 | |
| styrene | 62.26752 | 0.20 | 0.85 | 84.38819 | 0.004941 | 0.99 | 16.53 | 14.38 | 0.98 | |
| PP | ethylbenzene | 53.26609 | 0.13 | 0.87 | 63.09148 | 0.006389 | 0.99 | 9.08 | 12.09 | 0.92 |
| m-xylene | 36.75227 | 0.09 | 0.95 | 87.26003 | 0.008087 | 0.99 | 22.29 | 16.15 | 0.87 | |
| styrene | 33.58905 | 0.11 | 0.98 | 66.5779 | 0.009071 | 0.99 | 15.03 | 12.39 | 0.94 | |
| PVC | ethylbenzene | 46.72129 | 0.15 | 0.99 | 59.63029 | 0.006854 | 0.99 | 6.64 | 12.1 | 0.94 |
| m-xylene | 47.90405 | 0.15 | 0.92 | 68.21282 | 0.009099 | 0.99 | 14.75 | 13.053 | 0.86 | |
| styrene | 34.10691 | 0.08 | 0.95 | 58.30904 | 0.005554 | 0.99 | 4.06 | 11.83 | 0.96 | |
| MPs | BTEX | Henry Model | Langmuir Model | Freundlich Model | D–R Model | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Kd/(L·g−1) | R2 | KL/(L·mg−1) | R2 | KF/(mg·L−1) | n−1 | R2 | KDR | R2 | ||
| PE | ethylbenzene | 0.457 | 0.973 | 4.61 × 10−5 | 0.97 | 0.102 | 1.338 | 0.971 | 0.0002 | 0.919 |
| m-xylene | 0.854 | 0.985 | 3.11 × 10−5 | 0.956 | 0.388 | 1.188 | 0.967 | 9 × 10−5 | 0.888 | |
| styrene | 0.526 | 0.841 | 4.93 × 10−9 | 0.982 | 3.81 × 10−5 | 3.148 | 0.985 | 0.0004 | 0.802 | |
| PP | ethylbenzene | 0.582 | 0.948 | 4.03 × 10−6 | 0.986 | 0.033 | 1.658 | 0.989 | 0.0001 | 0.764 |
| m-xylene | 0.972 | 0.96 | 1.34 × 10−3 | 0.946 | 0.884 | 1.023 | 0.96 | 0.0001 | 0.941 | |
| styrene | 0.424 | 0.917 | 2.01 × 10−6 | 0.997 | 0.002 | 2.141 | 0.997 | 0.0003 | 0.849 | |
| PVC | ethylbenzene | 0.384 | 0.882 | 5.55 × 10−8 | 0.915 | 0.006 | 1.934 | 0.921 | 0.0002 | 0.777 |
| m-xylene | 0.577 | 0.956 | 1.87 × 10−5 | 0.935 | 0.378 | 1.096 | 0.952 | 0.0001 | 0.927 | |
| styrene | 0.313 | 0.849 | 1.91 × 10−7 | 0.927 | 3.418 × 10−4 | 2.482 | 0.945 | 0.0005 | 0.835 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yan, X.; Xie, Y.; Zhang, S.; Zhang, Z.; Dou, X.; Liu, J.; Che, S. Sorption Mechanisms and Behavior of Benzene Series Compounds by Microplastics in Aqueous Solution. Polymers 2026, 18, 526. https://doi.org/10.3390/polym18040526
Yan X, Xie Y, Zhang S, Zhang Z, Dou X, Liu J, Che S. Sorption Mechanisms and Behavior of Benzene Series Compounds by Microplastics in Aqueous Solution. Polymers. 2026; 18(4):526. https://doi.org/10.3390/polym18040526
Chicago/Turabian StyleYan, Xi, Yan Xie, Shucai Zhang, Zhiqing Zhang, Xiaohan Dou, Jingru Liu, and Shun Che. 2026. "Sorption Mechanisms and Behavior of Benzene Series Compounds by Microplastics in Aqueous Solution" Polymers 18, no. 4: 526. https://doi.org/10.3390/polym18040526
APA StyleYan, X., Xie, Y., Zhang, S., Zhang, Z., Dou, X., Liu, J., & Che, S. (2026). Sorption Mechanisms and Behavior of Benzene Series Compounds by Microplastics in Aqueous Solution. Polymers, 18(4), 526. https://doi.org/10.3390/polym18040526







