Synthesis of Novel Hyperbranched Polybenzo-Bisthiazole Amide with Donor–Acceptor (D-A) Architecture, High Fluorescent Quantum Yield and Large Stokes Shift
Abstract
:1. Introduction
2. Materials and Methods
2.1. Starting Materials
2.2. Polymerization
2.3. Measurements
3. Results and Discussion
3.1. NMR Analysis
3.2. FTIR Analysis
3.3. GPC Analysis
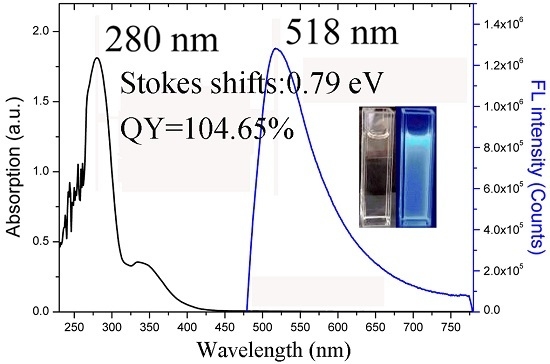
3.4. UV–Vis Absorption and Fluorescence Properties of HP–COOH and HP–NH2
3.5. XRD Analysis
3.6. Thermal Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Huang, W.; Smarsly, E.; Han, J.S.; Bender, M.; Seehafer, K.; Wacker, I.; Schröder, R.R.; Bunz, U.H.F. Truxene-Based Hyperbranched Conjugated Polymers: Fluorescent Micelles Detect Explosives in Water. ACS Appl. Mater. Interfaces 2017, 9, 3068–3074. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Wu, X.F.; Xu, Y.X.; Tong, H.; Wang, L.X. Dicyanovinyl-functionalized fluorescent hyperbranched conjugated polymer nanoparticles for sensitive naked-eye cyanide ion detection. Polym. Chem. 2014, 5, 5949–5956. [Google Scholar] [CrossRef]
- Liu, J.Z.; Zhong, Y.C.; Lu, P.; Hong, Y.N.; Lam, J.W.Y.; Faisal, M.; Yu, Y.; Wong, K.S.; Tang, B.Z. A superamplification effect in the detection of explosives by a fluorescent hyperbranched poly(silylenephenylene) with aggregation-enhanced emission characteristics. Polym. Chem. 2010, 1, 426–429. [Google Scholar] [CrossRef]
- Chen, L.; Gao, Y.X.; Fu, Y.Y.; Zhu, D.F.; He, Q.G.; Cao, H.M.; Cheng, J.G. Borate ester endcapped fluorescent hyperbranched conjugated polymer for trace peroxide explosive vapor detection. RSC Adv. 2015, 5, 29624–29630. [Google Scholar] [CrossRef]
- Jiang, H.B.; Wu, P.C.; Zhang, Y.; Jiao, Z.N.; Xu, W.; Zhang, X.T.; Fu, Y.Y.; He, Q.G.; Cao, H.M.; Cheng, J.G. Hyperbranched polymer based fluorescent probes for ppt level nerve agent simulant vapor detection. Anal. Methods 2017, 9, 1748–1754. [Google Scholar] [CrossRef]
- Armelin, E.; Whelan, R.; Martínez-Triana, Y.M.; Aleman, C.; Finn, M.G.; Díaz, D.D. Protective Coatings for Aluminum Alloy Based on Hyperbranched 1,4-Polytriazoles. ACS Appl. Mater. Interfaces 2017, 9, 4231–4243. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, J.S.; Pavía-Sanders, A.; Russell, J.D.; Wooley, K.L. Dynamic Anti-Icing Coatings: Complex, Amphiphilic Hyperbranched Fluoropolymer Poly(ethylene glycol) Cross-Linked Networks with an Integrated Liquid Crystalline Comonomer. Chem. Mater. 2016, 28, 5471–5479. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Li, B.; Zhang, M.C.; Wen, R.; Guo, X.H.; Li, X.; Zhang, J.; Li, S.J.; Ma, L.J. Pore-Free Matrix with Cooperative Chelating of Hyperbranched Ligands for High-Performance Separation of Uranium. ACS Appl. Mater. Interfaces 2016, 8, 28853–28861. [Google Scholar] [CrossRef] [PubMed]
- Duan, S.; Yu, B.G.; Gao, C.X.; Yuan, W.; Ma, J.; Xu, F.-J. A Facile Strategy to Prepare Hyperbranched Hydroxyl-Rich Polycations for Effective Gene Therapy. ACS Appl. Mater. Interfaces 2016, 8, 29334–29342. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Chen, R.J. pH-Responsive, Lysine-Based, Hyperbranched Polymers Mimicking Endosomolytic Cell-Penetrating Peptides for Efficient Intracellular Delivery. Chem. Mater. 2017. [Google Scholar] [CrossRef]
- Ma, N.; Zhao, L.R.; Hu, X.Y.; Yin, Z.; Zhang, Y.F.; Meng, J.Q. Protein Transport Properties of PAN Membranes Grafted with Hyperbranched Polyelectrolytes and Hyperbranched Zwitterions. Ind. Eng. Chem. Res. 2017, 56, 1019–1028. [Google Scholar] [CrossRef]
- Liu, G.H.; Zhang, G.F.; Hu, J.M.; Wang, X.R.; Zhu, M.Q.; Liu, S.Y. Hyperbranched Self-Immolative Polymers (hSIPs) for Programmed Payload Delivery and Ultrasensitive Detection. J. Am. Chem. Soc. 2015, 137, 11645–11655. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Ishitobi, H.; Inouye, Y. Formation of fluorescent platinum nanoclusters using hyper-branched polyethylenimine and their conjugation to antibodies for bio-imaging. RSC Adv. 2016, 6, 9709–9716. [Google Scholar] [CrossRef]
- Sun, P.; Chen, D.; Deng, H.P.; Wang, N.; Huang, P.; Jin, X.; Zhu, X.Y. “Bottom-up” Construction of Multi-Polyprodrug-Arm Hyperbranched Amphiphiles for Cancer Therapy. Bioconj. Chem. 2017, 28, 1470–1480. [Google Scholar] [CrossRef] [PubMed]
- Tian, K.; Jia, X.; Zhao, X.; Liu, P. Biocompatible Reduction and pH Dual-Responsive Core Cross-Linked Micelles Based on Multifunctional Amphiphilic Linear–Hyperbranched Copolymer for Controlled Anticancer Drug Delivery. Mol. Pharm. 2017, 14, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Heckert, B.; Banerjee, T.; Sulthana, S.; Naz, S.; Alnasser, R.; Thompson, D.; Normand, G.; Grimm, J.; Perez, J.M.; Santra, S. Design and Synthesis of New Sulfur-Containing Hyperbranched Polymer and Theranostic Nanomaterials for Bimodal Imaging and Treatment of Cancer. ACS Macro Lett. 2017, 6, 235–240. [Google Scholar] [CrossRef]
- Wu, Y.G.; Hao, X.H.; Wu, J.L.; Jin, J.; Ba, X.W. Pure Blue-Light-Emitting Materials: Hyperbranched Ladder-Type Poly(p-phenylene)s Containing Truxene Units. Macromolecules 2010, 43, 731–738. [Google Scholar] [CrossRef]
- Li, Z.A.; Ye, S.H.; Liu, Y.Q.; Yu, G.; Wu, W.B.; Qin, J.G.; Li, Z. New Hyperbranched Conjugated Polymers Containing Hexaphenylbenzene and Oxadiazole Units: Convenient Synthesis and Efficient Deep Blue Emitters for PLEDs Application. J. Phys. Chem. B 2010, 114, 9101–9108. [Google Scholar] [CrossRef] [PubMed]
- Qin, A.J.; Lam, J.W.Y.; Jim, C.K.W.; Zhang, L.; Yan, J.J.; Häussler, M.; Liu, J.Z.; Dong, Y.Q.; Liang, D.H.; Chen, E.Q.; et al. Hyperbranched Polytriazoles: Click Polymerization, Regioisomeric Structure, Light Emission, and Fluorescent Patterning. Macromolecules 2008, 41, 3808–3822. [Google Scholar] [CrossRef]
- Wen, X.; Zhang, D.X.; Ren, T.C.; Xiao, J.C.; Wu, Y.G.; Bai, L.B.; Ba, X.W. Yellow emitting materials: Truxene-based conjugated hyperbranched polymer containing difluoroboron-diketonate complexes. Dyes Pigments 2017, 137, 437–444. [Google Scholar] [CrossRef]
- Sun, J.; Yang, J.L.; Zhang, C.Y.; Wang, H.; Li, J.; Su, S.J.; Xu, H.X.; Zhang, T.M.; Wu, Y.L.; Wong, W.-Y.; et al. A novel white-light-emitting conjugated polymer derived from polyfluorene with a hyperbranched structure. New J. Chem. 2015, 39, 5180–5188. [Google Scholar] [CrossRef]
- Wu, Y.L.; Li, J.; Liang, W.Q.; Yang, J.L.; Sun, J.; Wang, H.; Liu, X.G.; Xu, B.S.; Huang, W. Hyperbranched fluorene-alt-carbazole copolymers with spiro[3.3]heptane-2,6-dispirofluorene as the core and their application in white polymer light-emitting devices. RSC Adv. 2015, 5, 49662–49670. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.; Jin, S.-H.; Lee, T.S. Synthesis of conjugated, hyperbranched copolymers for tunable multicolor emissions in light-emitting diodes. Polym. Chem. 2015, 6, 5062–5069. [Google Scholar] [CrossRef]
- Xing, L.-B.; Wang, X.-J.; Zhang, J.-L.; Zhou, Z.Y.; Zhuo, S.P. Tetraphenylethene-containing supramolecular hyperbranched polymers: Aggregation-induced emission by supramolecular polymerization in aqueous solution. Polym. Chem. 2016, 7, 515–518. [Google Scholar] [CrossRef]
- Du, Y.Q.; Yan, H.X.; Niu, S.; Bai, L.H.; Chai, F. Facile one-pot synthesis of novel water-soluble fluorescent hyperbranched poly(amino esters). RSC Adv. 2016, 6, 88030–88037. [Google Scholar] [CrossRef]
- Fu, X.; Zhang, Q.W.; Wu, G.; Zhou, W.; Wang, Q.-C.; Qu, D.-H. BODIPY-based conjugated microporous polymers as reusable heterogeneous photosensitisers in a photochemical flow reactor. Polym. Chem. 2014, 5, 6662–6670. [Google Scholar] [CrossRef]
- Scott, M.G.; Fréchet, J.M.J. Convergent Dendrons and Dendrimers: From Synthesis to Applications. Chem. Rev. 2001, 101, 3819–3868. [Google Scholar] [CrossRef]
- Matthew, R.R.; Wang, S.; Guillermo, C.B.; Cao, Y. Electroluminescence from Well-Defined Tetrahedral Oligophenylenevinylene Tetramers. Adv. Mater. 2000, 12, 1701–1704. [Google Scholar] [CrossRef]
- Chen, H.; Kong, J. Hyperbranched polymers from A2 + B3 strategy: Recent advances in description and control of fine topology. Polym. Chem. 2016, 7, 3643–3663. [Google Scholar] [CrossRef]
- Wu, W.B.; Ye, C.; Qin, J.G.; Li, Z. The Utilization of Isolation Chromophore in an “A3+B2” Type Second-Order Nonlinear Optical Hyperbranched Polymer. Macromol. Rapid Commun. 2013, 34, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Jikei, M.; Chon, S.-H.; Kakimoto, M.; Kawauchi, S.; Imase, T.; Watanebe, J. Synthesis of Hyperbranched Aromatic Polyamide from Aromatic Diamines and Trimesic Acid. Macromolecules 1999, 32, 2061–2064. [Google Scholar] [CrossRef]
- Young, H.K. Hyperbranched polymers 10 years after. J. Polym. Sci. Part A Polym. Chem. 1998, 36, 1685–1698. [Google Scholar] [CrossRef]
- Kudo, H.; Matsubara, S.; Yamamoto, H.; Kozawa, T. Synthesis of hyperbranched polyacetals via an + b2-type polyaddition (n = 3, 8, 18, and 21): Candidate resists for extreme ultraviolet lithography. J. Polym. Sci. Part A Polym. Chem. 2015, 53, 2343–2350. [Google Scholar] [CrossRef]
- Herrema, J.K.; Wildeman, J.; Gil, R.E.; van Hutten, P.F.; Wieringa, R.H.; Hadziioannou, G. Tuning of the Luminescence in Multiblock Alternating Copolymers. 1. Synthesis and Spectroscopy of Poly[(silanylene)thiophene]s. Macromolecules 1995, 28, 8102–8116. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.W.; Wang, C.S. Processing of optical quality rigid-rod polymer thin films. Polymer 1994, 35, 3673–3678. [Google Scholar] [CrossRef]
- Roberts, M.F.; Jenekhe, S.A.; Cameron, A.; Mcmillan, M.; Perlstein, J. Lewis Acid Coordination Complexes of Polymers. 2. Computational Modeling of Single-Chain and Aggregate Structures of Rigid-Rod Poly(p-phenylenebenzobisthiazole). Chem. Mater. 1994, 6, 658–670. [Google Scholar] [CrossRef]
- Bai, S.J.; Spry, R.J.; Zelmon, D.E.; Ramabadran, U.; Jackson, J. Optical anisotropy of polymeric films measured by waveguide propagation mode determination. J. Polym. Sci. Part B Polym. Phys. 1992, 30, 1507–1514. [Google Scholar] [CrossRef]
- Vanherzeele, H.; Meth, J.S.; Jenekhe, S.A.; Roberts, M.F. Third-order nonlinear optical properties of thin films of poly(p-phenylene benzobisthiazole) and its molecular composites with polyamides. Appl. Phys. Lett. 1991, 58, 663–665. [Google Scholar] [CrossRef]
- Hu, X.B.; Yu, D.M. Hyperbranched Polybenzobisthiazole with High Thermal Stability, Good Organosolubility, and Interesting Optical Performance. Macromol. Chem. Phys. 2012, 213, 738–746. [Google Scholar] [CrossRef]
- Hu, X.B. Synthesis and properties of novel benzobisthiazole-containing hyperbranched polyamides derived from 2,6-diaminobenzo[1,2-d:4,5-d’]bisthiazole. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Li, J.; Bo, Z.S. “AB2 + AB” Approach to Hyperbranched Polymers Used as Polymer Blue Light Emitting Materials. Macromolecules 2004, 37, 2013–2015. [Google Scholar] [CrossRef]
- Dutta, T.; Woody, K.B.; Parkin, S.R.; Watson, M.D.; Gierschner, J. Conjugated Polymers with Large Effective Stokes Shift: Benzobisdioxole-Based Poly(phenylene ethynylene)s. J. Am. Chem. Soc. 2009, 131, 17321–17327. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.R.; Sarkar, S.K.; Thilagar, P. Aggregation-Induced Emission and Sensing Characteristics of Triarylborane–Oligothiophene–Dicyanovinyl Triads. Chem. Eur. J. 2016, 22, 17215–17225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.S.; Kuwabara, H.; Potscavage, W.J.; Huang, S.P.; Hatae, Y.; Shibata, T.; Adachi, C. Anthraquinone-Based Intramolecular Charge-Transfer Compounds: Computational Molecular Design, Thermally Activated Delayed Fluorescence, and Highly Efficient Red Electroluminescence. J. Am. Chem. Soc. 2014, 136, 18070–18081. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.F.; Luo, W.W.; He, B.R.; Chen, L.; Nie, H.; Hu, R.G.; Qin, A.J.; Zhao, Z.J.; Tang, B.Z. Integration of aggregation-induced emission and delayed fluorescence into electronic donor–acceptor conjugates. J. Mater. Chem. C 2016, 4, 3705–3708. [Google Scholar] [CrossRef]
- Hu, X.B. Novel fluorescent porous hyperbranched aromatic polyamide containing 1,3,5-triphenylbenzene moieties: Synthesis and characterization. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Wolfe, F.; Loo, B.H.; Arnold, F.E. Rigid-rod polymers. 2. Synthesis and thermal properties of para-aromatic polymers with 2,6-benzobisthiazole units in the main chain. Macromolecules 1981, 14, 915–920. [Google Scholar] [CrossRef]
- Osaheni, J.A.; Jenekhe, S.A. Synthesis and processing of heterocyclic polymers as electronic, optoelectronic, and nonlinear optical materials. 1. New conjugated rigid-rod benzobisthiazole polymers. Chem. Mater. 1992, 4, 1282–1290. [Google Scholar] [CrossRef]
- Komber, H.; Voit, B.; Monticelli, O.; Russo, S. 1H and 13C-NMR Spectra of a Hyperbranched Aromatic Polyamide from p-Phenylenediamine and Trimesic Acid. Macromolecules 2001, 34, 5487–5493. [Google Scholar] [CrossRef]
- Hawker, C.J.; Lee, R.; Fréchet, J.M.J. One-step synthesis of hyperbranched dendritic polyesters. J. Am. Chem. Soc. 1991, 113, 4583–4588. [Google Scholar] [CrossRef]
- Xu, H.J.; Chen, K.C.; Guo, X.X.; Fang, J.H.; Yin, J. Synthesis and properties of hyperbranched polybenzimidazoles via A2 + B3 approach. J. Polym. Sci. Part A Polym. Chem. 2007, 45, 1150–1158. [Google Scholar] [CrossRef]
- Chen, B.; Zhang, H.; Luo, W.; Nie, H.; Hu, R.; Qin, A.; Zhao, Z.; Tang, B.Z. Oxidation-enhanced emission: Exploring novel AIEgens from thieno[3,2-b]thiophene S,S-dioxide. J. Mater. Chem. C 2017, 5, 960–968. [Google Scholar] [CrossRef]
- Gao, M.; Su, H.; Lin, Y.; Ling, X.; Li, S.; Qin, A.; Tang, B.Z. Photoactivatable aggregation-induced emission probes for lipid droplets-specific live cell imaging. Chem. Sci. 2017, 8, 1763–1768. [Google Scholar] [CrossRef]
- Chen, B.; Nie, H.; Hu, R.; Qin, A.; Zhao, Z.; Tang, B.Z. Insights into the correlation between the molecular conformational change and AIE activity of 2,5-bis(dimesitylboryl)-3,4-diphenylsiloles. J. Mater. Chem. C 2016, 4, 7541–7545. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, C.-Y. Simple and Accurate Quantification of Quantum Yield at the Single-Molecule/Particle Level. Anal. Chem. 2013, 85, 2000–2004. [Google Scholar] [CrossRef] [PubMed]
- Melhuish, W.H. Quantum efficiencies of fluorescence of organic substances: Effect of solvent and concentration of the fluorescent solute. J. Phys. Chem. 1961, 65, 229–235. [Google Scholar] [CrossRef]
- Dexter, D.L. Possibility of Luminescent Quantum Yields Greater than Unity. Phys. Rev. 1957, 108, 630–633. [Google Scholar] [CrossRef]
- Lorbeer, C.; Cybinska, J.; Mudring, A.V. Reaching quantum yields ≫100% in nanomaterials. J. Mater. Chem. C 2014, 2, 1862–1868. [Google Scholar] [CrossRef]
- Wegh, R.T.; Donker, H.; Oskam, K.D.; Meijerink, A. Visible Quantum Cutting in LiGdF4:Eu3+ Through Downconversion. Science 1999, 283, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Sasanka, D.; Enquan, J.; Matthew, A.; Thomas, H.; Donglin, J. Highly Emissive Covalent Organic Frameworks. J. Am. Chem. Soc. 2016, 138, 5797–5800. [Google Scholar] [CrossRef]
- Banal, J.L.; Zhang, B.L.; Jones, D.J.; Ghiggino, K.P.; Wong, W.W.H. Emissive Molecular Aggregates and Energy Migration in Luminescent Solar Concentrators. Acc. Chem. Res. 2017, 50, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, K.; Chio, T.I.; Gu, H.; Banerjee, A.; Sorrentino, A.M.; Sackett, D.L.; Bane, S.L. Benzocoumarin Hydrazine: A Large Stokes Shift Fluorogenic Sensor for Detecting Carbonyls in Isolated Biomolecules and in Live Cells. ACS Sens. 2017, 2, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Shizu, K.; Nakanotani, H.; Adachi, C. Twisted Intramolecular Charge Transfer State for Long-Wavelength Thermally Activated Delayed Fluorescence. Chem. Mater. 2013, 25, 3766–3771. [Google Scholar] [CrossRef]
- Kim, B.-G.; Ma, X.; Chen, C.; Le, Y.; Coir, E.W.; Hashemi, H.; Aso, Y.; Green, P.F.; Kieffer, J.; Kim, J. Energy Level Modulation of HOMO, LUMO, and Band-Gap in Conjugated Polymers for Organic Photovoltaic Applications. Adv. Funct. Mater. 2013, 23, 439–445. [Google Scholar] [CrossRef]











| Samples | Monomer Feed Ratio of A2 to B3 | Mn | Mw | Mp | Mz | Mz+1 | PDI a |
|---|---|---|---|---|---|---|---|
| HP–COOH | 1:1 | 7426 | 10,195 | 12,314 | 13,387 | 16,547 | 1.37 |
| HP–NH2 | 9:4 | 10,280 | 19,291 | 11,035 | 33,938 | 49,610 | 1.88 |
| Samples | λabs (nm) | λem (nm) | Stokes Shift (nm/eV) a | Egopt (eV) b | ФFL c |
|---|---|---|---|---|---|
| HP–COOH in DMSO | 285, 336 | 427, 517 | 137/0.86 | 3.13 | 77.75% |
| HP–COOH in DMF | 280, 333 | 518 | 128/0.79 | 2.55 | 104.65% |
| HP–COOH Film | 305 | 436, 462 | 131/0.42 | 3.02 | 2.01% |
| HP–NH2 in DMSO | 283, 336 | 529 | 149/0.92 | 2.97 | 81.14% |
| HP–NH2 in DMF | 280, 342 | 537 | 147/0.87 | 2.76 | 118.72% |
| HP–NH2 Film | 301, 339 | 480 | 179/0.86 | 2.55 | 4.89% |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X. Synthesis of Novel Hyperbranched Polybenzo-Bisthiazole Amide with Donor–Acceptor (D-A) Architecture, High Fluorescent Quantum Yield and Large Stokes Shift. Polymers 2017, 9, 304. https://doi.org/10.3390/polym9080304
Hu X. Synthesis of Novel Hyperbranched Polybenzo-Bisthiazole Amide with Donor–Acceptor (D-A) Architecture, High Fluorescent Quantum Yield and Large Stokes Shift. Polymers. 2017; 9(8):304. https://doi.org/10.3390/polym9080304
Chicago/Turabian StyleHu, Xiaobing. 2017. "Synthesis of Novel Hyperbranched Polybenzo-Bisthiazole Amide with Donor–Acceptor (D-A) Architecture, High Fluorescent Quantum Yield and Large Stokes Shift" Polymers 9, no. 8: 304. https://doi.org/10.3390/polym9080304