1. Introduction
Wheat (
Triticum spp.) is an important crop for global food security, being the staple food for millions of people globally, especially in many developing countries. The current context of food security implies not only sufficient quantity of food but also food of high quality to ensure human health. Nutritional and health aspects of wheat grain have recently been prioritized through studying the diversity of cultivated wheat: different species like einkorn (
Triticum monococcum) and spelt (
T. spelta), landraces, old and modern cultivars, and colored wheat. The number of research publications devoted to wheat with blue and purple grain have increased dramatically over the last two years, indicating the priority and potential of these genotypes in developing wheat cultivars with specific benefits for human health [
1,
2]. The interest in purple grain wheat is due to the presence of anthocyanin, flavonoid pigments found in a wide variety of plants including fruits and vegetables. Anthocyanins act as antioxidants, scavenging free radicals [
2]. The study of Sytar et al. [
3] demonstrated a high correlation between antioxidant activities and content of bioactive phytochemicals (anthocyanin components cyanidin and pelargonin) in colored grain. Anthocyanins have a wide range of biological functions including antioxidants, enzyme inhibitors, induction of detoxification enzymes, activation of protein kinase (important for cellular energy homeostasis), increased membrane stability, induction of apoptosis, and cell cycle arrest. The anthocyanin health benefits have recently been reviewed by Daotong Lia [
1]. Crucial cellular processes, such as cell cycle, apoptosis, autophagy, and biochemical metabolism, are involved in the beneficial effects and may provide potential therapeutic targets and strategies for the improvement in treatment for a wide range of diseases in the future.
The origin of wheat genetic resources with purple grain was described in the reviews of Zeven [
4] and Grausgruber et al. [
2]. Purple tetraploid wheat (
T. durum) was first collected in Abyssinia (present Ethiopia) in the 1870s and then introduced to Europe, where it was widely distributed by botanists, and this was followed by further introductions. The purple grain color was eventually incorporated into bread wheat (
T. aestivum). In the 1960s and 1970s, purple wheat was developed worldwide to distinguish feed grain, determine outcrossing rates and develop hybrid systems. Commercial purple wheat cultivars were released in Australia, Canada, China, New Zealand, and several European countries. It is also noteworthy that purple grain landraces are still cultivated in Ethiopia and can be found in the markets in the Tigray Region [
5].
The anthocyanin pigmentation in wheat is controlled by dominant alleles of the genes
Rc (coleoptile),
Pc (culm),
Plb (leaf blades),
Pls (leaf sheaths),
Pp (grain pericarp),
Pan (anthers), and
Ra (auricles) [
6,
7,
8,
9,
10]. The alleles conferring strong pigmentation induce more intensive transcription of the structural genes, proving their role as regulators in the anthocyanin biosynthesis network. According to Tereshchenko et al. [
9], a dark purple pericarp results from complementary action of the dominant genes,
Pp-D1 (7D) and
Pp3 (2A). Presence of the dominant genes,
Pp-A1 and
Pp3, results in light purple color, and the grain will remain uncolored in the presence of the recessive
Pp3 gene even if two other genes (
Pp-A1 and
Pp-D1) are in the dominant state [
11]. Later, the complementary action of
Pp3 and
Pp-1 genes was explained by interaction of their products (the bHLH-MYC transcriptional factor encoded by the
Pp3 gene and the R2R3-MYB factor encoded by the
Pp-1 genes), resulting in a functional regulatory complex for anthocyanin biosynthesis [
12].
Breeding and genetic resource efforts to develop purple grain wheat are underway globally. The winter wheat breeding program in the Czech Republic focuses on a combination of dark grain using different genes, important agronomic traits and good baking quality [
13]. In China, five novel mutants with variable seed color were developed using gamma irradiation of the hexaploid purple wheat line K4191 [
14]. The total anthocyanin content of three mutant lines was significantly higher than the parent and white seeded check. The grain anthocyanin content can be increased by pyramiding different genes responsible for the accumulation of anthocyanins in different grain layers [
15]. Bohmdorfer et al. [
15] proposed a method of high-performance thin-layer chromatography allowing fast and efficient phenotyping of grain pigments. The same detection method was successfully used in fingerprinting of 94 colored wheat genotypes [
16]. The phenotyping cost was below 1 EUR and the processing time was less than 9 min per sample. Crosses between purple pericarp and blue aleurone genotypes were used to increase the anthocyanin content, although the majority of the progenies were within the range of the parental material [
17].
One of the main focuses of recent research has been the anthocyanin content of wheat grain, including its composition, dynamics, genotypic variation, and products. Knivel et al. [
18] studied adapted spring wheat lines with blue and purple pericarp for two seasons in Saskatoon, Canada and found that genotypic variation for anthocyanin concentration was statistically significant, while the year and genotype by year interaction were not. This facilitates breeding progress. In a more recent study with nine purple wheat genotypes in Saskatchewan, Canada, the content and composition of anthocyanin was significantly influenced by genotype and genotype by environment (G × E) interaction [
19]. The source–sink relationship has an important role in anthocyanin content [
20]. Grain position in the spike affected the anthocyanin content, decreasing at distal positions, and source reduction decreased anthocyanin content by 25% to 50%. Heat stress positively affected anthocyanin content in wheat grain [
21,
22]. Combined nitrogen and phosphorus fertilizer application results in higher antioxidant activities in six wheat cultivars, including two purple-grained cultivars [
23]. Shoeva et al. [
24] demonstrated that anthocyanins in coleoptiles protect shoots, while those in grains protect roots under drought stress (seedlings grown in 15% polyethylene glycol). In addition, anthocyanins in coleoptiles protect wheat seedlings under metal toxicity (treatment with 25 µM CdCl
2) [
25] and moderate irradiation dose (pretreatment of dry seeds with 50 Gy before sowing) [
26].
Four colored winter wheat cultivars along with two checks were studied for two years under organic and non-organic cropping systems [
27]. While total phenolic content was more affected by year, anthocyanin content was more affected by genotype and the effect of environment and genotype was comparable for total antioxidant activity. Higher anthocyanin content and antioxidant activities and lower yields were observed under an organic cropping system and in a drier year. Thirteen diverse anthocyanin pigmented wheat genotypes were studied in Austria for two seasons [
28]. Both genotype and environmental effects were important for agronomic and biochemical traits. A significant and relatively high negative correlation was observed between 1000-kernel weight and both total phenolic and anthocyanin content. However, protein content did not affect the biochemical traits measured.
Evaluation of purple grain products for end use and nutritional quality has been made for beer [
29], biscuits [
30,
31], pasta [
32], bread [
33,
34], buns [
35], powder from bran faction [
36], Chinese noodles [
37], and steamed bread [
38]. Hrivna et al. [
39] studied the rheological properties of dough containing 10, 15 and 20% of the bran from purple wheat. Addition of bran increased water loss during baking and the specific volume of the bread. Generally, higher content of bran increased dough viscosity. Detailed evaluation of flour and dough properties from purple and blue wheat grain indicated that colored wheat cultivars did not have characteristics substantially different from standard commercial cultivars or flour products [
40]. Khlestkina et al. [
34] showed that bread-making quality and organoleptic properties of bakery products made from anthocyanin-containing grains did not concede, or in some cases were higher than the corresponding properties of products obtained from control grains. It was found that the presence of anthocyanin increased the shelf life of bakery products and their resistance to the development of mold under conducive conditions [
34].
Most of the purple grain studies compared very diverse material including old and modern cultivars, cultivars with different adaptation and even different species. Using such diverse material makes it is difficult to isolate the effect of
Pp genes, because the effect of genotype background will be substantial. The Institute of Cytology and Genetics in Novosibirsk (Russia) developed isogenic lines for purple grain in the background of cv. Saratovskaya-29 [
11]. Two sources of
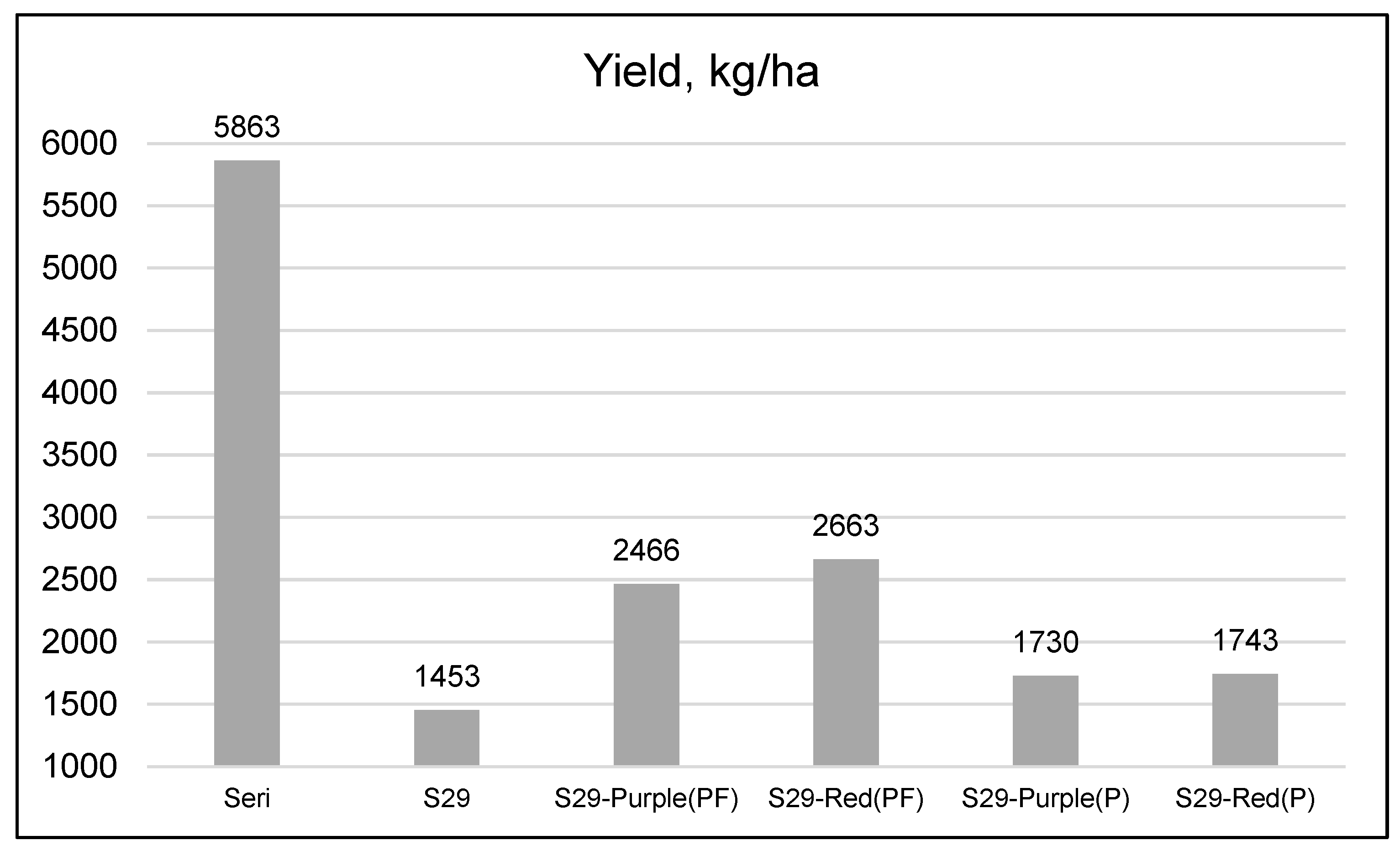
Pp genes were used: cvs Purple and Purple Feed. Morphologically identical Saratovskaya-29 isogenic lines were used in the current study to compare isogenic lines with red (recurrent parent) and purple grain for grain yield and yield components, common bread-making quality traits and composition of health-related substances under variable growing conditions to develop relevant breeding approaches.
4. Discussion
The current study addressed the important question of the relative contribution of genotype and environment for characters associated with purple color. As expected, due to the diversity of our testing sites, the effect of environment was larger compared to genotype for the studied productivity and quality traits. Nevertheless, several important traits such as grain color and anthocyanin content are closely controlled by genotype, offering the opportunity for selection. These results are similar to previous studies conducted in Canada [
56,
57]. The significant genotypic effects and genotype by environment interactions suggest that the methodology of breeding would require careful selection of sites and consideration of the genotype rank change with the change in testing locations (crossover interactions).
The current study contributed to the understanding of variation of different grain quality traits along the processing line: grain, whole wheat flour, regular flour and bread. When comparing the properties of the isogenic lines differing only in a few genes, it is not guaranteed that purple grain products will have superior and significant advantages. For example, the phenolic content in purple-grained lines was not significantly higher in whole wheat flour than in red-colored lines. However, it was significantly higher in bread. For antioxidant activities, no differences between the genotypes were detected in both years. This is a common challenge for research and breeding for health related traits in wheat. Normally grain phytochemicals are present in small quantities, and require reliable and sophisticated equipment for their accurate quantification. In this regard, while breeding colored wheat with emphasis on anthocyanin and antioxidants, it makes sense to increase the number of replicates to clearly establish the relationships between the traits and genes, and background genotypic effects. Targeting phytochemicals using genomic selection could provide an excellent opportunity to make genetic gains once relevant training populations are developed and characterized.
Two sources of
Pp genes were used in the development of the isogenic lines and were conveniently compared in two experiments of this study. The lines originating from Purple Feed were substantially better for grain yield, some productivity and grain quality traits compared to lines originating from Purple. The lines derived from Purple Feed have a donor fragment in chromosome 7D extending from the locus
Xgwm0044 to
Xgwm0111, while the donor fragment in the lines derived from Purple is larger (from
Xgwm1004 to
Xgwm0437) [
9]. This region of chromosome 7D is known as the gene-rich region [
58], including a locus for grain weight [
59]. Putative allelic differences in this locus between the isogenic lines can explain the Purple Feed-derived line superiority for grain yield. Thus, this line would be the preferred donor for further transfer of the
Pp genes into elite wheat lines. Overall, despite very clear differences in the grain, flour, and bread color observed in this study, the effects of
Pp genes on grain quality traits were not large and stable enough to be easily used in breeding. Therefore, from a breeding perspective, utilization of additional genes to strengthen
Pp gene effects on targeted traits would be a justified approach for developing germplasm for health-promoting grain. Substantial progress has already been made for biofortified wheat with higher Zn content [
60]. The combination of high Zn and purple grain can be explored to further enhance the health benefits of biofortified wheat. Likewise, as research and breeding for other traits such as Fe content and dietary fiber progress, these can also be incorporated into purple wheat improvement programs.
There is always opportunity for genetic gains provided there is genetic diversity, understanding of genotype by environment interaction, and relevant breeding methodology and technology. In this case, purple wheat improvement represents a unique opportunity to use the extensive knowledge of bread wheat to develop healthier food products.