Abstract
The brown planthopper (BPH) is a major pest that causes serious damage to rice in countries with mild climates, including Korea, Japan, China, and Vietnam. Although a large number of BPH-resistant genes have been reported, BPH has recently evolved rapidly and in a variable manner due to rapid climate change and an unpredictable environment. The synthesis of secondary metabolites in plants is essential, as it provides resistance to various stressors, including pests such as BPH. For QTL mapping, a Samgang/Nagdong double haploid (SNDH) 113 population was used. The BPH resistance gene was screening by measure the resistance score using the phenotype that appeared after BPH inoculation in the 113 SNDH population. QTL mapping was used, and SSR marker RM584-RM225 of chromosome 6 and SSR marker RM331-S8024 of chromosome 8 were commonly identified. These locations contained transcription factors, phytohormones, signaling molecules, kinases, and secondary metabolites that defend plants from stressors in the environment. This region of chromosome 8 also contained a sequence similar to that of rice Choristmate mutase (OsCM9). CM is a gene that is not only found in rice, but also in thale cress (Arabidopsis thaliana (L.) Heynh.), maize (Zea mays L.), and soybean (Glycine max (L.) Merr.). After inoculation with BPH, the relative expression levels of OsCM9 were greater in the line resistant to BPH than in the line susceptible to BPH. The newly identified BPH-resistant gene OsCM9 can be used for the development of rice varieties that are capable of resisting sudden damage due to BPH, as the evolution of BPH due to climate change has had negative impacts on rice crops.
1. Introduction
The brown planthopper (Nilaparvata Lugens Stal.) is a major insect pest that is primarily present in Southeast Asia and East Asia, and has a significant impact on rice crops, leading to a decline in production and a large economic burden. The brown planthopper (BPH) cannot overwinter in South Korea [1]. BPH directly sucking rice, reduces nutrients in plants, destroys tissues, and interferes with the movement of nutrients. Because of these causes, the amount of photosynthesis in rice decreases and the yield decreases due to the decrease in grain filling rate. In particular, BPH has had a serious negative impact on rice yields in China, Japan, and Vietnam, which are Asian regions including Korea. In 2005 and 2008, the amount of rice was reduced by 2.7 million tons in China due to damage from BPH, and 0.4 million tons in Vietnam were damaged by BPH [2]. Rice is one of the most important crops in temperate regions of the world and is a main food source for more than one-third of the world’s population. However, many pests that damage rice grow in the fields where rice farming takes place. In particular, BPH causes direct damage to rice and triggers a ‘hopper burn’ [3]. In addition, rice can also be indirectly affected by several viruses, such as ragged stunt virus (RRSV) and grassy stunt virus (RGSV) [4,5,6]. Although the time of BPH arrival and extent of BPH infestation varies from year to year, the pest generally arrives in mid-June and propagates for 2–3 generations in the rice field, resulting in damage to rice in early September [7,8]. However, as it is difficult to understand the growth status of BPH from the field, it is challenging to establish measures to control BPH. For example, if the optimal time to implement control measures is missed, then the measures taken will be less effective [9].
In Southeast Asia, the extent and frequency of damage caused by BPH has continued to increase since the 1960s. The green revolution began in the late 1960s. Due to the Green Revolution, the use of chemical fertilizers and pesticides drastically increased rice production. Due to the excessive use of chemical fertilizers and pesticides, BPH became tolerant, which increased the incidence of damage from BPH [10]. Over the same period, the International Rice Research Institute (IRRI) actively promoted breeding of BPH resistant varieties of rice. A large number of BPH resistant varieties, such as IR26, IR32, and IR36 have been developed. Total of 32 resistance genes, including Bph1, Bph2, and Bph3, have been identified through analysis of resistance to BPH in rice germplasm resources collected from cultivated Oryza sativa and wild rice species [11]. However, most of the BPH-resistant genes discovered to date have been identified in Indica, wild rice, or native rice. In order to develop Japonica-type resistant varieties, this strain of rice must be crossed with Indica rice. However, it has been difficult to develop practical Japonica-type varieties due to the many negative traits, such as its susceptibility to many insects and disease and the differences in gene expression of resistance genes. For example, when Indica rice was introduced into Japonica, the development of Japonica-type BPH resistant variety was more difficult due to lack of expression of the resistance gene.
BPH-resistant is a quantitative trait involving a polygenic, which is a complex trait that includes various biological properties. Moreover, the quantitative trait can also be affected by the environment. In addition, quantitative traits involve various genes the breeding efficiency is very low, leading to difficulties in breeding BPH resistant varieties. Recent technology allows mapping of quantitative trait loci (QTL) for BPH-resistant using DNA markers and high-density genetic maps [12,13,14]. Previous QTL analyzes have identified BPH-resistant genes at various rice chromosomes [15,16,17]. If QTL analysis is used to identify loci associated with BPH-resistant and the marker assisted selection (MAS) system is implemented, then lines with BPH-resistant can be selected for in the early generations of breeding. This would allow breeding of resistant rice varieties due to the pyramiding of effective QTLs. As the current whole genome sequences of rice have been published, the genome database can be used to predict the putative function of a gene based on the sequence information. In the past, this has allowed the identification of candidate genes involved in specific biochemical pathways [18].
In the present research, for QTL mapping of BPH resistance genes Samgang/Nagdong double haploid (SNDH) population were established by crossing the Indica type ‘Samgang’ and Japonica type ‘Nagdong’ of rice. The DH population has been used to map genes related to disease [19], a gene conferring resistance to biotic stress [20], and a tolerance gene for abiotic stress [21]. The main purpose of this research was to screen a candidate gene that has the greatest influence on BPH-resistant, using QTL mapping on the SNDH population, and to assess the function of this gene. Although BPH-resistant genes have been reported previously, our goal is to identify a new resistance gene that confers BPH-resistant. This BPH-resistant gene could provide a convenient genetic resource that can be efficiently used for breeding rice varieties with improved resistance to unpredictable BPH infestations in the future changing climate.
2. Materials and Methods
2.1. Plant Material and Field Design
In this research, the 113 double haploid populations, SNDH, developed through the crossbreeding of Samgang (Indica, BPH-resistant variety) and Nagdong (Japonica, BPH susceptible variety), was used as the experimental rice for the analysis of BPH-resistant using QTL mapping. F1 obtained by crossing Samgang and Nagdong was anther cultured to construct 113 double haploid populations. Anther culture can process colchicine to create a double haploid population. Seeds obtained through the anther culture were sown at Kyungpook National University. The seeds were treated with a seed disinfectant such as Spotak pesticide (HANKOOKSAMGONG, Seoul, South Korea) for 4 days at 25 °C in darkness. Seeds were sowed on 23 April 2018 and 22 April 2019 at the experimental field at Kyungpook National University and transplanted on 28 May 2018 and 30 May 2019 with a planting distance of 30 × 15 cm. The amount of fertilizer used was N–P2O5–K2O = 9–4.5–5.7 kg/10a and the rice was cultivated according to the Agricultural Science and Technology Research Standards of the Rural Development Administration. The 2nd–3rd BPH instar were used for bioassays to assess BPH-resistant. The BPH resistant and the susceptible line were used to assess the BPH-resistant gene using QTL mapping. SNDH were sown in plastic boxes (32 × 25 × 10 cm), with 12 seeds per box, in rows with a spacing of 2.0 × 0.5 cm and were cultivated for 14 days prior to use for the bioassay. Plants were inoculated with 10–15 BPH insects per plant. The BPH inoculated inside breeding cage made by acrylic plate (50 × 50 × 40 cm) and 100 μm mesh net was used on the back for ventilation and water supply. The insect nursery was maintained at a temperature of 27 °C, a humidity of 60–70%, a luminous intensity of 2000 lux and over a 16 h cycle.
2.2. Brown Planthopper Resistance Evaluation of the DH Line
To identify BPH-resistant, the resistance score (RS) was assigned, according to previously described methods [22]. The resistance score was given 9 points when Nagdong, a BPH susceptible, died. And when inoculated into the 113 SNDH population, when BPH-sensitive Nagdong died, the population that died at the same time was awarded 9 points. Like Samgang, the resistant, even if BPH was inoculated, the color of the leaves continued to be dark green. 0 points were given to the SNDH population that did not collapse and stood upright. Therefore, the 113 SNDH population was inoculated with BPH, and a resistance score was given by judging the leaf color of the plant and the degree of collapse of the plant after BPH inoculation. Each day after inoculation with BPH, RS score were measured based on the changes in the phenotype of each plant, and the average value was used to measure the degree of resistance of each population. Each SNDH population was inoculated with BPH using 3 plants, and the mean and standard deviation were calculated by giving RS to each plant. Statistical analysis was performed using the SPSS program (IMMSPSS Statistics, version 22, IBMSPSS Statistics, version 22, Redmond, WC, USA).
2.3. Construction of Genetic Maps and QTL Analysis of Brown Planthopper Resistance
Total of 850 SSR markers were used to create a genetic map of the Samgang/Nagdong Double Haploid line. SSR markers with polymorphisms in the Samgang and Nagdong lines were selected. After PCR of the selected markers, 163 SSR markers with a clear band identified in gel electrophoresis and having a co-dominant were selected and used for gene mapping. MAP Manager QTXb2.0 [23] was used for linkage analysis. A genetic map was created using the MAP Manager QTX2.0 program in order to perform QTL mapping of BPH-resistant-related genes. The distance to the chromosome marker was calculated using the Kosambi map function. Win QTL cart2.5 was used for QTL mapping. Composite interval mapping (CIM) was used for the analysis of the relationship between BPH-resistant and the SSR marker, a LOD value of 3.0 or more was used to improve the accuracy of QTL mapping [24]. The percentage of phenotypic change that can be explained by each labeled QTL was determined using the R2 value, which is the ratio of the sum of squares explained by the QTL to the total sum of squares. The QTL naming protocol by Mc Couch et al. [25] was used.
2.4. Gene Information Analysis
Identification of candidate genes through QTL mapping is an important element in QTL analysis. Gramene (http://www.gramene.org/), Rapdb (https://rapdb.dna.affrc.go.jp/) and Rice X pro (https://ricexpro.dna.affrc.go.jp/) were used to create a physical map and annotate candidate genes. Open Reading Frames (ORFs) identified among the SSR markers were categorized by function and analyzed to the related BPH-resistant gene. In addition, NCBI (http://www.ncbi.nim.nih.gov) was used to search for the previously reported BPH-resistant gene.
2.5. Analysis of the Expression Level of Candidate Genes for Resistance to Brown Planthopper
The 113 SNDH population was given a resistnace score after inoculating BPH. And at this time, the most resistant to BPH (SNDH 29) and the most sensitive (SNDH 11) were selected. After seeding them again, BPH was inoculated at the seeding stage in the BPH breeding cage, and the leaves were sampled at 0, 1, 2, 4, 8, 16, 24, 48, and 72 h to analyze the relative expression level. The RNeasy plant mini kit (QIAGEN, Hilden, Germany) was used to extract total RNA from rice leaves and 2 μg of RNA was used as a template for cDNA synthesis using oligo-dt primers and transcriptase (Invitrogen, Carlsbad, CA, USA). The qRCRBIO cDNA Synthesis kit (Cat No. PB30.11-10, PCRBIOSYSTEM, Wayne, PA, USA) was used for cDNA synthesis. Quantitative real-time PCR was performed using the Eco Real-Time PCR System. For the reaction solution for quantitative real-time PCR, the mix contained: 10 μL 2x qRCRBIO SyGreen Blue Mix, 1 μL cDNA, 0.5 μL forward primer (20 pmol/μL), 0.5 μL reverse primer (20 pmol/μL), and ddH2O to a final volume of 20 μL. OsActin was used as a control group. Each reaction was run in triplicate and the mean and standard deviation were calculated.
2.6. DNA Extraction and PCR Protocol
Genomic DNA was extracted by grinding young seedling stage leaves using Tissue Layer (QIAGEN, Cat. No. 85220, Hilden, Germany) after sowing for each CNDH line. The ground sample was extracted using a DNeasy plant mini kit (QIAGEN, Cat No./ID:69104, Hilden, Germany), and the DNA concentration was measured using the Nanodrop (ND-2000; Nanodrop, Waltham, MA, USA) and diluted to a final concentration of 20 ng/μL before use. The PCR conditions were as follows: pre-denaturation 95 °C for 5 min, denaturation 94 °C for 30 s, annealing 52 °C for 30 s, extension 72 °C for 1 min, the denaturation to extension steps were repeated 35 times, and a final extension of 72 °C was carried out for 5 min. The amplified PCR product was electrophoresed on 0.8% agarose gel at 100 V for 30 min.
2.7. Statistical Analysis
All experiments were replicated at least three times, and the statistical analysis was performed using the SPSS program (IMMSPSS Statistics, version 22, IBMSPSS Statistics, version 22, Redmond, WC, USA).
3. Results
3.1. Construct of the Samgang/Nagdong Double Haploid Population Genetic Map
Polymorphic markers were selected using a marker that clearly distinguishes the parents of the SNDH population, Samgang and Nagdong. Total of 850 SSR markers were used for gene mapping in the Samgang/Nagdong double haploid line (SNDH). Among these markers, only 163 SSR markers showed polymorphisms in Samgang and Nagdong. A high number of polymorphic markers were detected due to the large differences in genetics between Samgang and Nagdong. In order to use more SSR markers to create the genetic map, 850 SSR markers were analyzed once again, and finally 222 SSR markers showing polymorphism in Samgang and Nagdong were selected. The 222 marker band selected using the polymorphism test was clearly amplified and the 222 SSR marker amplified in both Samgang and Nagdong was re-selected. Finally, using the 222 SSR marker, the average distance of the marker was set to 9.4 cM, and a related map with a total length of 2082.4 cM was created.
3.2. Analysis of QTLs Associated with Brown Planthopper Resistance
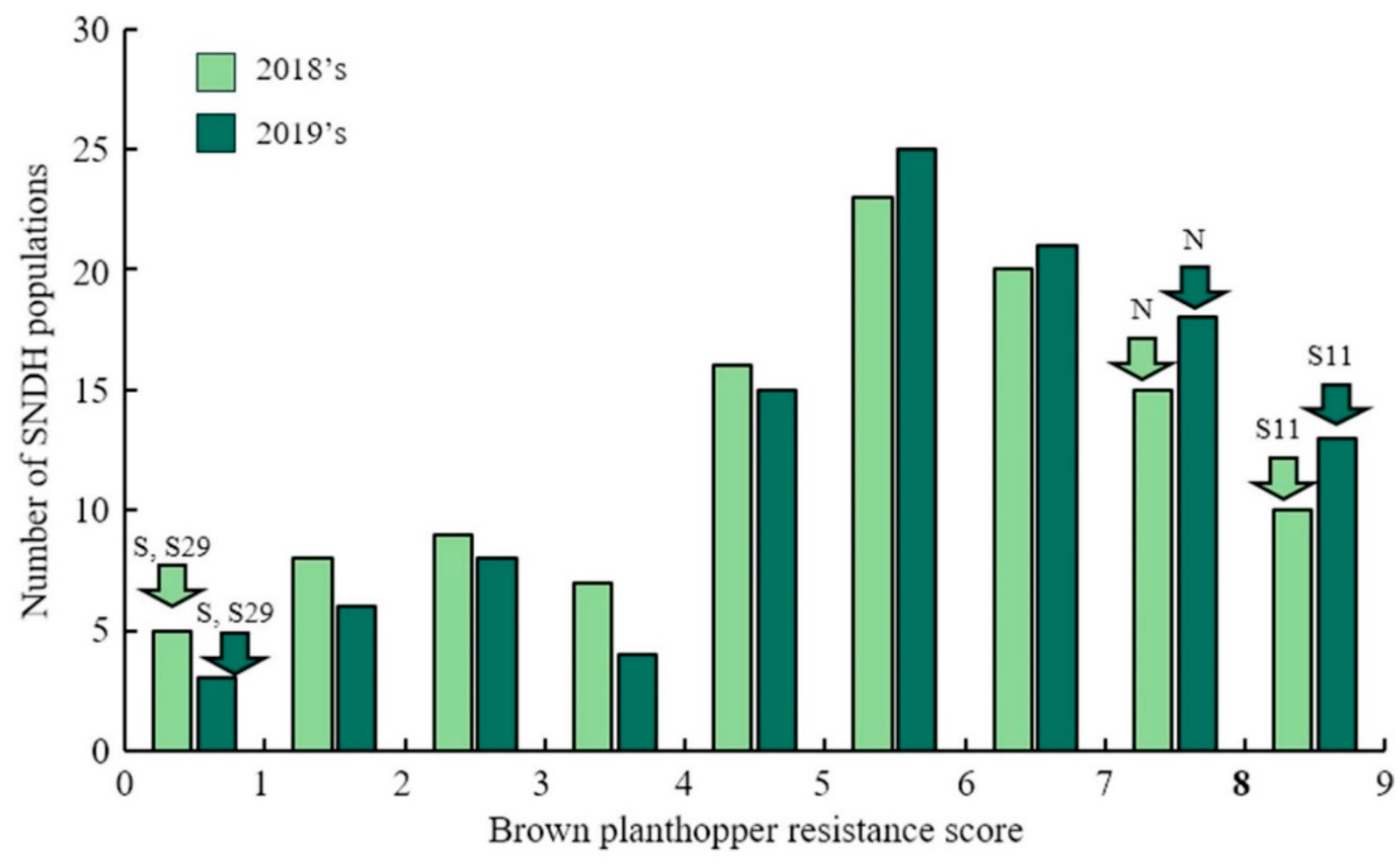
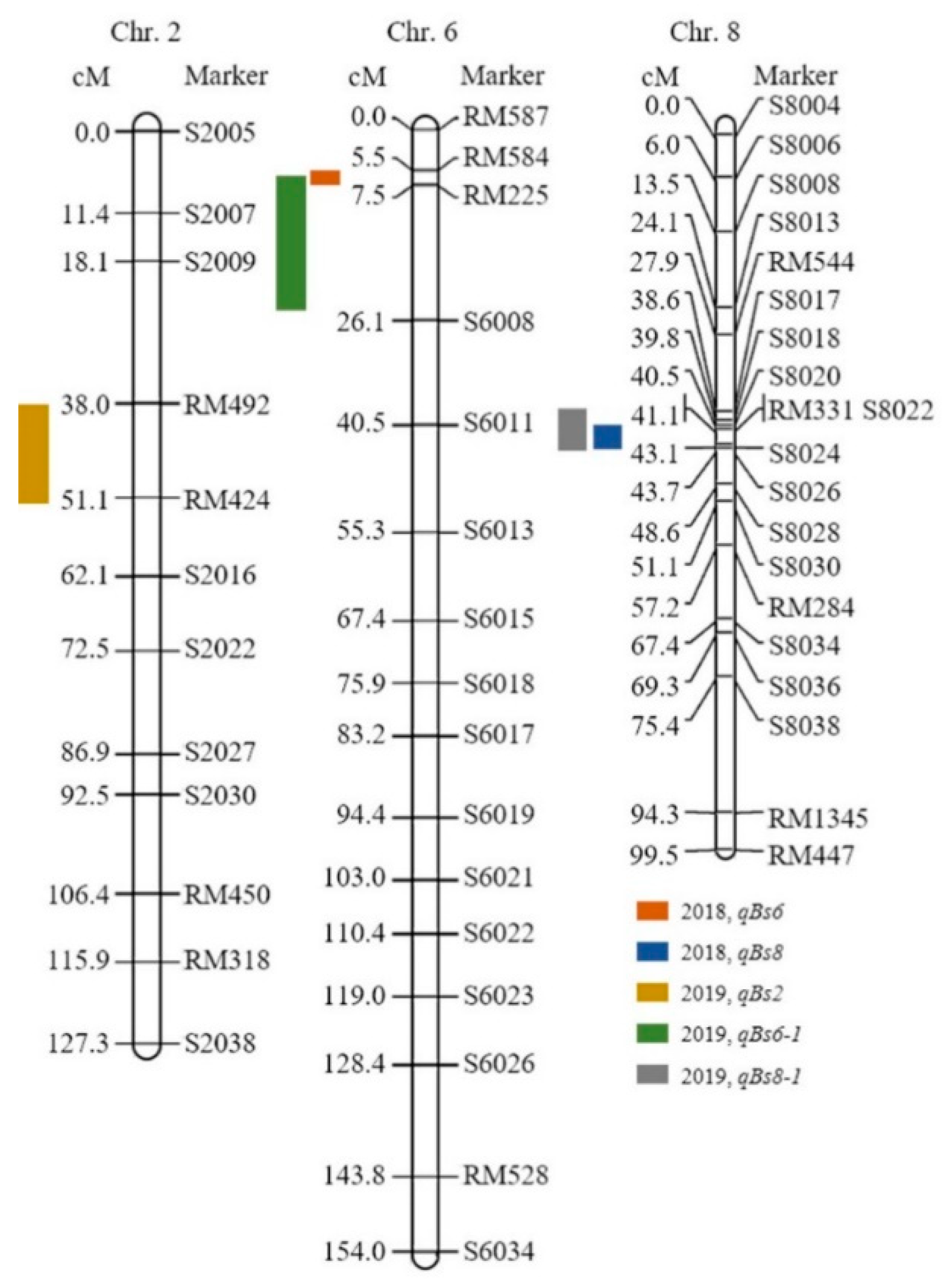
The resistance score was given to the SNDH population for the BPH-resistant gene QTL mapping. One line died earlier than Nagdong and some of lines had resistance similar to that of Samgang. However, most of the lines had a response in the middle of that of Samgang and Nagdong. A resistant score of 9 was given to the SNDH population when Nagdong, a susceptible cultivar of BPH treatment, died. In addition, plants with a resistant score of 0 continued to maintain dark green leaves even after inoculation, and the plants stood upright without falling. The BPH-resistant score of the SNDH population shows a distribution slightly skewed to the right, indicating that it has BPH-resistant, but is a quantitative trait whose expression is regulated by one or more genes (Figure 1). After BPH was inoculated on Samgang, Nagdong, and SNDH populations, BPH-resistant-related QTL mapping was performed, and results with a LOD of 3.0 included 3 QTLs. The QTLs that were detected are on chromosome 2 (qBs2), chromosome 6 (qBs6, qBs6-1), and chromosome 8 (qBs8, qBs8-1) (Supplementary Table S1). BPH QTL mapping was carried out in 2018 and 2019 using the resistance score. In 2018, qBs6 and qBs8 were detected. The qBs6 was mapped to SSR marker interval of RM584-RM225 on chromosome 6 with a LOD of 3.12 and an explainable phenotypic contribution of 29%. qBs8 was mapped in SSR marker interval of S8020-S8024 on chromosome 8 with a LOD value of 4.18 and an explainable phenotypic change of 20%. In 2019, qBs2, qBs6-1, and qBs8-1 were detected. The qBs2 was mapped to SSR marker interval of RM492-RM424 on chromosome 2, with an LOD value of 2.74 and a 20% explainable phenotypic change. The qBs6-1 was mapped to the SSR marker interval of RM584-S6008 on chromosome 6, with an LOD value of 3.35 and an explainable phenotypic change of 19%. The qBs8-1 was mapped to S8017-S8026 on chromosome 8 with a LOD value of 5.05 and an explainable phenotypic change of 22%. All BPH-resistant-related QTLs detected in 2018 and 2019 were derived from Samgang alleles. BPH-resistant QTL mapping results using a resistance score over a 2-year period were commonly detected for LOD values of 3.0 or greater on SSR marker interval of RM584-RM225 of chromosome 6 and SSR marker interval of RM331-S8024 of chromosome 8 (Figure 2). The distance between the markers of RM584-RM225 on chromosome 6 is 2.0 cM, and the distance between the markers of RM331-S8024 on chromosome 8 is 2.0 cM.
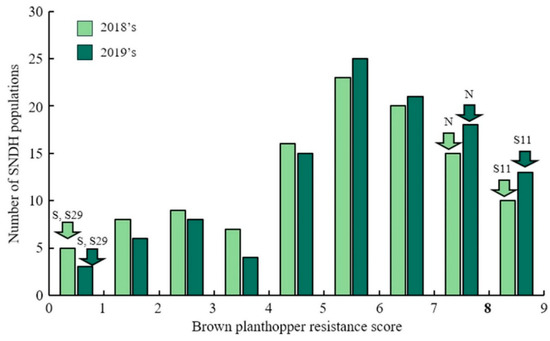
Figure 1.
The frequency distribution for resistance score in SNDH population. The resistance score on the SNDH population shows a normal distribution. Therefore, the resistance of BPH is regulated by various genes rather than a single gene. S, Samgang. N, Nagdong. S29, BPH-resistant population SNDH 29. S11, BPH susceptible population SNDH 11.
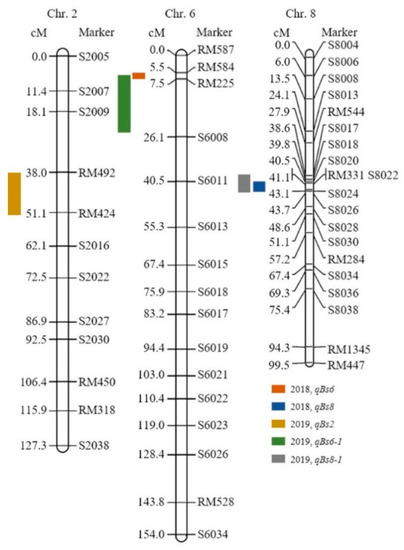
Figure 2.
The chromosomal location of the QTL associated with the BPH-resistant gene. QTL mapping results indicate that BPH-resistant genes are located on chromosomes 2, 6, and 8. Of these, RM584-RM225 on chromosome 6 and RM331-S8024 on chromosome 8 were commonly detected after two years of QTL mapping.
3.3. Search for Candidate Genes Associated with Brown Planthopper Based on QTL Mapping
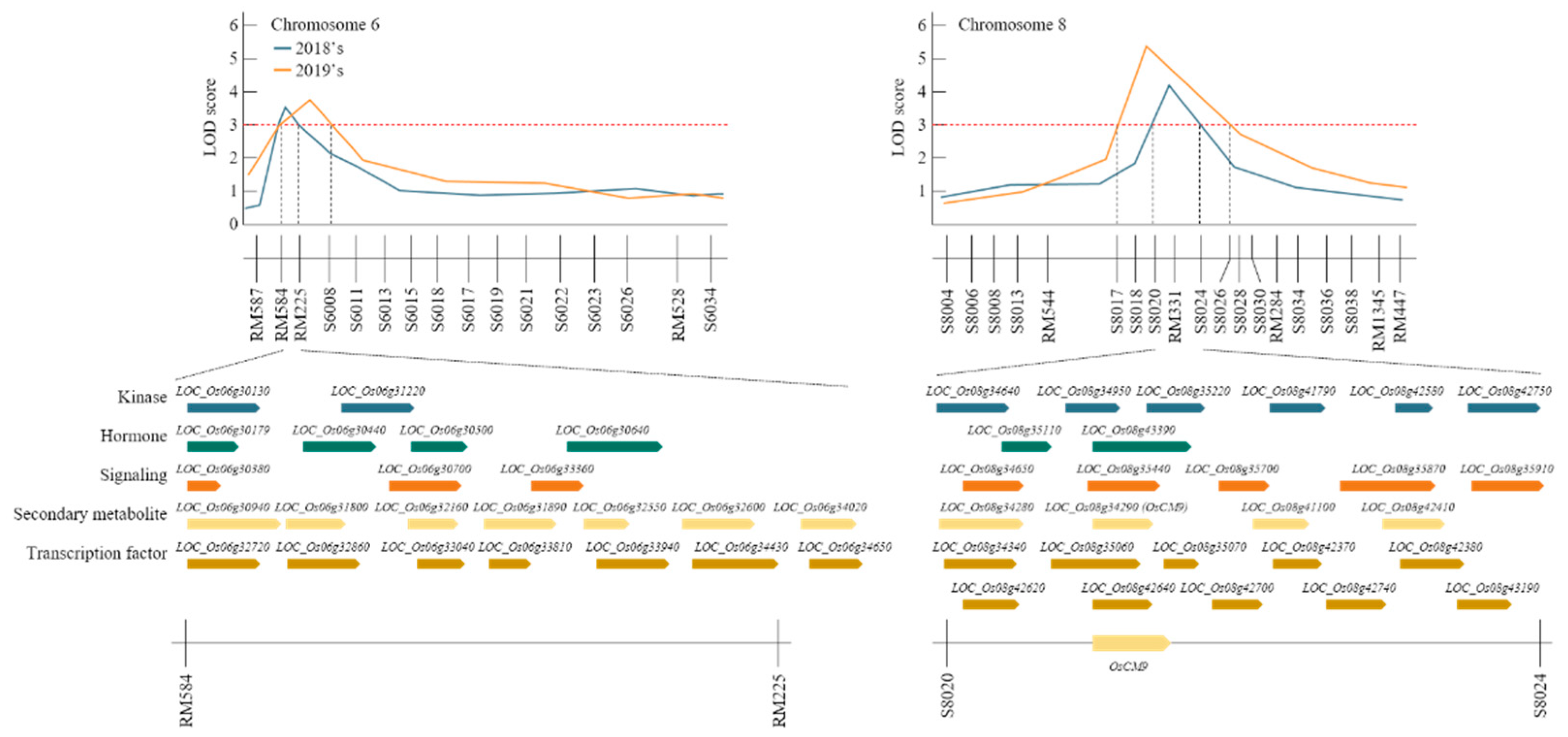
QTL mapping results using the BPH resistant score identified RM584-RM225 on chromosome 6 and RM331-S8024 on chromosome 8 (Figure 3). Analysis of these SSR markers using the NCBI database revealed 50 ORFs (Supplementary Table S2) that were associated with BPH-resistant in RM584-RM225 on chromosome 6 and RM331-S8024 on chromosome 8. These are grouped in Supplementary Table S1 according to their function [26]. RM584-RM225 and RM331-S8024 have candidate genes corresponding to kinases, phytohormones, signaling molecules, secondary metabolites, and transcription factors related to the resistance to BPH.
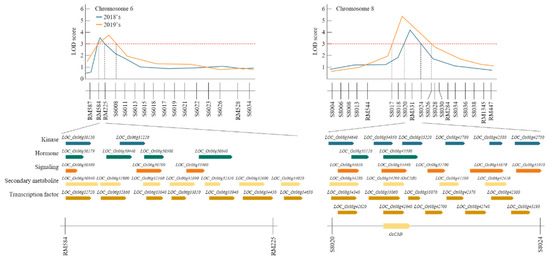
Figure 3.
Quantitative trait loci (QTL) analysis and physical mapping related to brown planthopper (BPH) resistance gene. After 2 years of QTL mapping, RM584-RM225 on chromosome 6 and S8020-S8024 on chromosome 8 were mapped consecutively. There were various candidate genes in this region, of which OsCM9 on chromosome 8 was screened as a BPH-resistant gene.
3.4. Selection of BPH-Resistant Gene and Comparison of Relative Expression Levels of Other Resistance Genes
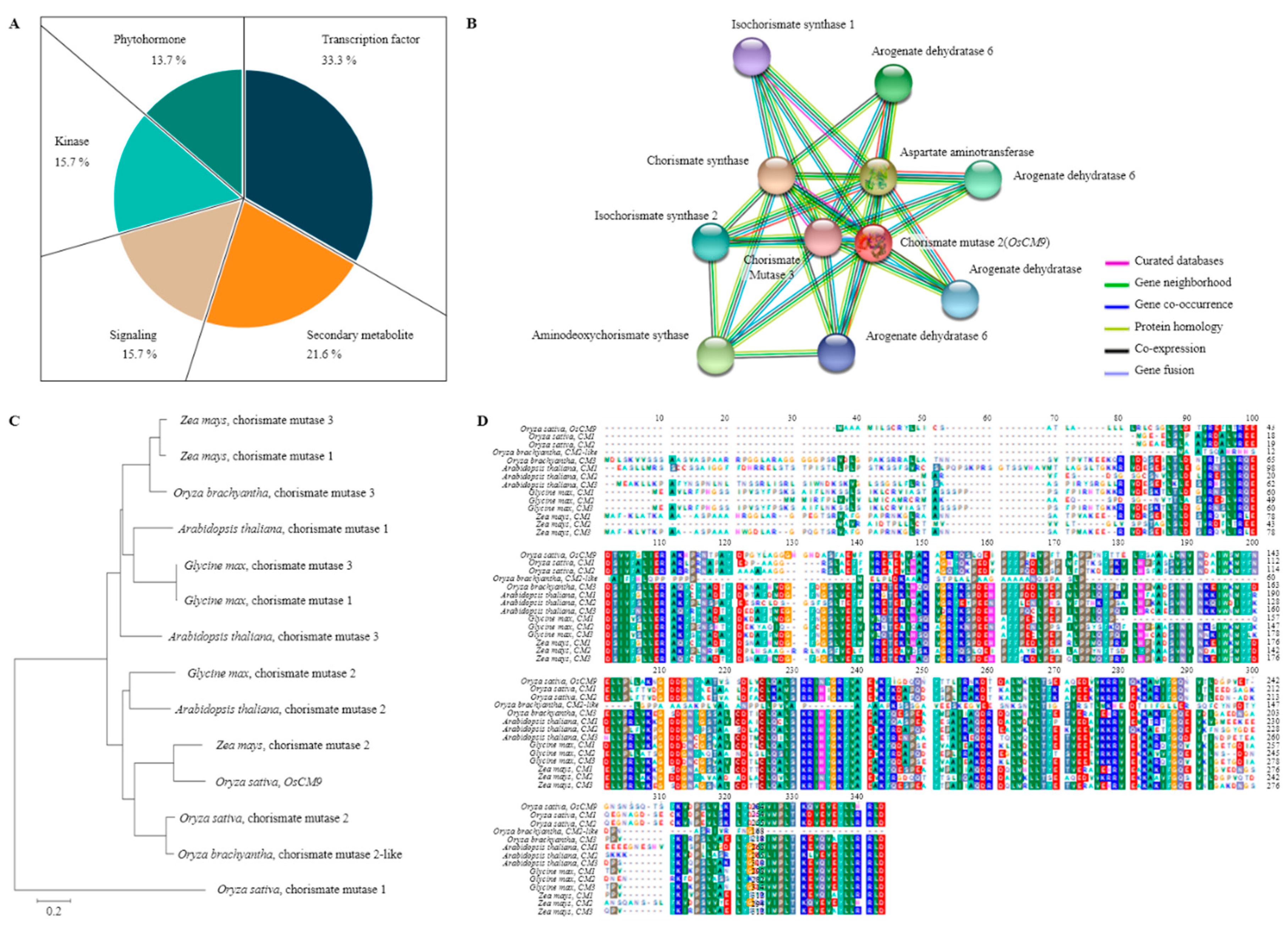
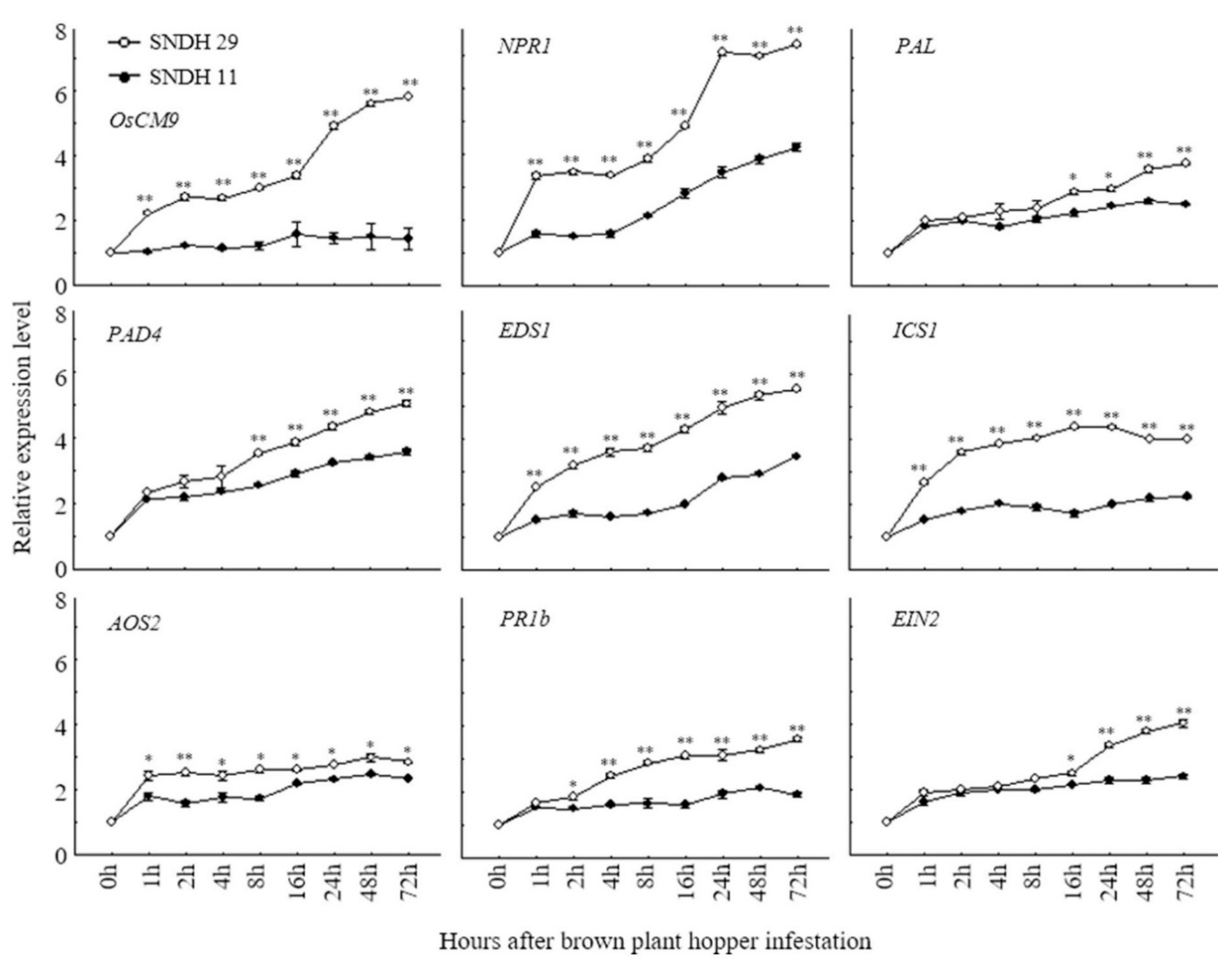
According to the rice genome annotation project databases Rapdb (https://rapdb.dna.affrc.go.jp/) and Rice X pro (https://ricexpro.dna.affrc.go.jp/), 51 candidate genes are located in the target region. Of these, 33.3% were associated with transcription factors, 21.6% with secondary metabolites, 15.7% with signaling molecules, 15.7% with kinases, and 13.7% with phytohormones (Figure 4). Among these candidate genes, LOC_Os08g34290 (OsCM9), which is located on chromosome 8, performs a similar function as chorismate mutase in rice, and is involved in the biosynthesis of the primary metabolites phenylalanine and tyrosine. We compared the relative gene expression levels of CM in BPH-resistant SNDH29 and BPH-susceptible SNDH11 with time after treatment with BPH. We also measured the relative expression levels of the non-expressor of pathogenesis-related genes 1 (NPR1), phenylalanine ammonia-lyase 6 (PAL6), phytoalexin deficient 4 (PAD4), enhanced disease susceptibility 1 (EDS1), isochorismate synthase 1 (ICS1), allene oxide synthase 2 (AOS2), basic pathogenesis-related gene 1 (PR1b), and ethylene insensitive 2 (EIN2). These genes are representative secondary metabolites of resistance, involved in the SA and JA biosynthetic pathway. Expression levels of CM were significantly different between the resistant and susceptible plants one hour after the inoculation with BPH. Among the genes responsible for SA biosynthesis, NPR1, EDS1, and ICS1 also had significant differences in the relative expression levels between the resistant and susceptible line one hour after inoculation with BPH. The expression levels of PAL6 and PAD4 were also different between the resistant and susceptible groups, but PAL6 showed a significant difference 16 h after inoculation with BPH and PAD4 was significant 8 h after the inoculation of BPH. AOS2, which is involved in JA biosynthesis, showed a significant difference in the relative expression levels one hour after inoculation with BPH, and PR1b showed a significant difference in the relative expression level after 4 h. EIN2, which is involved in ethylene signaling, showed a significant difference in the relative expression level between the resistant and susceptible lines 16 h after the inoculation of BPH (Figure 5).
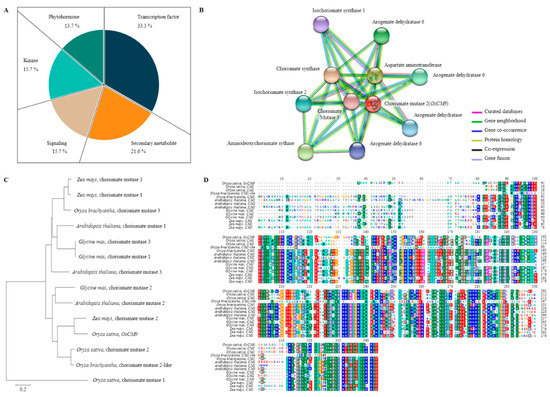
Figure 4.
Predicting the function of OsCM9. (A) QTL mapping result BPH-resistant candidate gene distribution. Among the candidate genes for BPH-resistant are Transcription factor, secondary metabolite, signaling, kinase and phytohormone. Of these, the Transcription factor accounted for 33.3%, and Secondary metabolite accounted for 21.6%. (B) Proteins that interact with OsCM9. OsCM9 interacts with five different proteins (CS; Chorismate synthase, ADCS; Aminodeoxychorismate synthase, ICS; Isochorismate synthase, CM; Chorismate mutase, PDT; Prephenate dehydratase). These are all major proteins of the shikimate pathway that synthesize secondary metabolites. (C) Phylogenetic tree of CM and homology genes. The homology of CM genes from Arabidopsis thaliana, Glycine max, Oryza and Zea mays was compared. As a result, the CM was most similar to Zea mays CM2 and was grouped near Oryza CM2. (D) Multiple sequence alignment of CM. Conserved amino acid sequences found in the CM domains of Oryza, Arabidopsis thaliana, Glycine max, Zea mays.
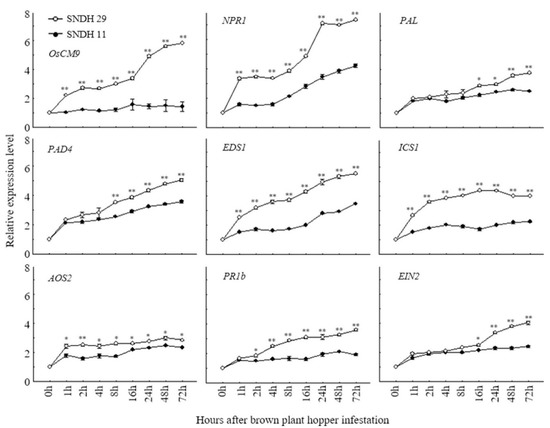
Figure 5.
Comparison of relative expression levels of CM gene according to inoculation time in resistance and sensitivity lines of BPH. Compared the newly discovered CM by QTL mapping with the relative expression levels of several BPH-resistant genes that have already been reported. All genes were expressed high in BPH-resistant line. In particular, CM expression level was not increased by BPH susceptible line over time, but BPH-resistant line showed a higher gene expression level over time. SNDH 11, BPH susceptible line. SNDH 29, BPH-resistant line. *, Significant at the 0.05 level; **, significant at the 0.01 level.
3.5. Analysis of Phylogenetic Tree and Homology Sequence, and Protein Interaction
BLAST results of the BPH-resistant gene CM showed that it has a high degree of sequence similarity with chorismate mutase (CM2). OsCM9 exhibited high levels of homology not only with rice, but also Arabidopsis thaliana, Zea mays, and Glycine max. Construction of a phylogenetic tree showed that the CM gene in Arabidopsis thaliana and Glycine max were highly similar (Figure 4). We predicted the functional partners using the domain of OsCM9. OsCM9 interacts with five proteins (CS; Chorismate synthase, ADCS; Aminodeoxychorismate synthase, ICS; Isochorismate synthase, CM; Chorismate mutase, PDT; Prephenate dehydratase) (Figure 4).
4. Discussion
BPH is a major biological stressor that impedes the stable rice production in countries in Asia, including China, Vietnam, Japan, and South Korea. To date, some of BPH resistant variety has been bred and pesticides have been used to minimize damage from BPH. However, insecticides not only lead to environmental pollution, but also cause development of populations of BPH that are resistant to insecticides. A cost-effective and environmentally friendly alternative is to develop strains of rice that are BPH resistant [27]. However, cultivating of only one resistant variety is inefficient, due to the continued differentiation of BPH. For example, in the 1980s, BPH-resistant varieties were used, but a new strain of BPH was able to overcome this resistance [28]. Therefore, it is important to identify novel BPH-resistant genes from various rice varieties in order to develop BPH resistant rice. In 1970, the BPH-resistant gene Bph1 was identified [29] and many BPH-resistant genes have subsequently been mapped.
In this research, in order to map the location of the BPH-resistant gene, a double haploid line (Samgnag/Nagdong double haploid line, SNDH) was made by crossing the BPH-resistant Indica variety, Samgang, and the susceptible Japonica variety, Nagdong. Using the BPH resistant score and QTL mapping results from 2018 and 2019, 5 QTLs on chromosomes 2, 6, and 8 were identified for two years. Of these, RM584-RM225 on chromosome 6 and RM331-S8024 on chromosome 8 were detected at the same locus in the second consecutive year and all LOD values were higher than 3.0. Myint et al. [30] mapped the BPH-resistant gene on chromosome 6. Alam and Cohen [15] used antixenosis, antibiosis, and tolerance to map the BPH-resistant gene. Ren et al. [31] mapped resistance genes on chromosomes 2, 3, and 4, and Hirabayashi et al. [16] mapped resistance genes with an LOD of 3.0 or higher on chromosomes 3, 4, and 12. The differences in QTL mapping of the same trait may be due to genetic differences in the population used and the environmental differences in the location where the studies were conducted [32].
In the present research, using the BPH resistant score, BPH QTL mapping results revealed 23 ORFs in RM584-RM225 on chromosome 6 and 27 ORFs in RM331-S8024 on chromosome 8. Of these ORFs, 33.3% were encoded transcriptional regulators, 21.6% encoded secondary metabolites, 15.7% encoded signaling molecules, 15.7% encoded kinases, and 13.7% encoded phytohormone regulators. Plants have many defense mechanisms against viral entry. As these plants respond to the biotic stress of pathogenic fungi and viruses, creating enzymes that are related to secondary metabolites is the most efficient defense against pathogens [33,34]. The secondary metabolites of plants are signaling substances that are involved in plant development, responding to environmental stress, and protecting against various pathogens. In particular, salicylic acid (SA) is a typical secondary metabolite. SA is a natural phenolic compound that plays an important role in disease resistance in plants and its production is induced by various types of pathogens [35,36]. In this research, QTL mapping results detected ORFs associated with secondary metabolites on chromosome 6 and chromosome 8. The OsCM9 gene on chromosome 8 has a similar sequence to chorismate mutase 2 (CM2). CM2 plays an important role in the synthesis of the aromatic amino acid secondary metabolites. Through QTL mapping, we compared some of the previously reported BPH-resistant genes with the newly identified BPH-resistant gene, OsCM9, to identify their effect on BPH-resistant. Similar to NPR1 which is involved in SA biosynthesis, there was a significant difference between the susceptible line and the resistance line at 1 h after BPH inoculation. These results suggest that the OsCM9 gene, which was identified in this research using QTL mapping based on the BPH-resistant score, is a putative BPH resistant gene. In addition, OsCM9 and NPR1 are genes involved in the synthesis of secondary metabolites in response to biotic stress. This suggests that secondary metabolites are important for plants to confer resistance to biotic stress. CM catalyzes the transfer of chorismate to prephenate, which is the first reaction of phenylalanine and tyrosine biosynthesis in rice. In addition, CM exists in various functional and structural forms in some organisms that produce aromatic amino acids. Three genes related to CM were identified in Arabidopsis thaliana, two were identified in Solanum lycopersicum, and four were identified in Oryza sativa. The prephenate produced by CM is an intermediate for most of the secondary metabolites of the shikimate pathway, as well as in the biosynthesis of the aromatic amino acids phenylalanine and tyrosine. In this study, the newly discovered OsCM9 interacts with CS, ADCS, ICS, CM and PDT. All of these genes act on aromatic amino acid biosynthesis by the shikimate pathway [37]. CS catalyzes the last reaction of the shikimate path that produces chorismate, has a very high homologous protein structure in various organisms, and exists in various forms [38]. The chorismate synthesized by the CS gene is the final product of the shikimate path and is used as a precursor for the synthesis of aromatic amino acids. The chorismate thus produced is used as a substrate by competition with the enzyme corresponding to the first reaction of the metabolite derived from chorismate. The well-known tetrahydrofolate for vitamin B9 is synthesized by the enzymatic reaction of ADCS [39]. The biosynthesis of phylloquinone, which is used to synthesize salicylic acid, a typical plant defense hormone, is produced by the ICS enzymatic reaction [40]. In addition, phenylalanine and tyrosine are produced by the CM enzymatic reaction [41,42]. The aromatic amino acids phenylalanine and tyrosine are synthesized from chorismate. Chorismate is converted to prephenate by CM, which is used as a substrate to produce phenylalanine and tyrosine. In particular, the interaction confirmed in this study utilizes the Phenylpyruvate path, but prephenate is dehydrated/decarboxylated by PDT and converted to phenylpyruvate in the phenylpyruvate path, and then transamination produces phenylalanine. Most microorganisms primarily use the phenylpyruvate path for the synthesis of phenylalanine and tyrosine [43]. Phenylalanine, tyrosine, and tryptophan, synthesized by the Shikimate pathway, are now widely used commercially. Phenylalanine is used as a raw material for aspartame and tyrosine is used as a treatment for people with Parkinson’s disease or with phenylketonuria. In humans, these aromatic amino acids must be absorbed in other ways, as they cannot be synthesized in the body. Therefore, the OsCM9 gene identified in this study will not only be important for the breeding of cultivars with BPH-resistant, but also in the development of human medicines. In addition, the identification of substances that confer BPH-resistant can reduce the various problems caused by the use of chemical pesticides and can contribute to the development of environmentally friendly pesticides.
5. Conclusions
In this research, brown planthopper was inoculated on the SNDH 113 line, and the brown planthopper resistance gene was screened using a resistant score and QTL mapping. Independent experiments were conducted in 2018 and 2019. In each experiment, 113 SNDH population was inoculated with BPH, and QTL was performed using the resistance score. In addition, BPH resistance candidate genes were screened centering on QTL regions that were commonly detected in 2018 and 2019. Since RM331 of chromosome 8 had a LOD score of 3.0 or higher in common for two years, BPH resistance candidate genes were selected around RM331 screened. Around RM331, various ORFs such as kinase, hormone, signaling, secondary metabolite, and transcription factor were included, and OsCM9 was finally selected among them. The RM584-RM225 region of chromosome 6 and the RM331-S8024 region of chromosome 8 are commonly detected with a value of LOD 3.0 or higher in QTL mapping for two years. By confirming the ORF of this region, a brown planthopper resistance candidate gene was searched. A total of 50 candidate genes (transcription factor 33.3%, secondary metabolite 21.6%, signaling molecules 15.7%, kinase 15.7%, phytohormone 13.7%) were detected. 21.6% of these candidate genes are genes that act to make secondary metabolites. Among these candidate genes, the expression level of OsCM9 increased in the brown planthopper resistance line after inoculation of the brown planthopper. OsCM9 is also a gene that acts to make secondary metabolites, which are necessary substances for plants to protect themselves from biotic and abiotic stress. As a result of predicting the interaction of OsCM9 proteins, the interaction with five proteins (CS; Chorismate synthase, ADCS; Aminodeoxychorismate synthase, ICS; Isochorismate synthase, CM; Chorismate mutase, PDT; Prephenate dehydratase), and these genes are involved in the biosynthesis of all aromatic amino acids phenylalanine and tyrosine. Therefore, the newly discovered brown planthopper resistance gene OsCM9 is expected to be useful not only for the development of brown planthopper resistance cultivars, but also for the pharmaceutical industry.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4395/10/12/1865/s1, Table S1: QTLs associated with resistance to BPH in Samgang/Nagdong double haploid lines, Table S2: Fifty candidate genes identified between the RM584 and RM225 markers on chromosome 6, RM331-S8024 on chromosome 8 and their ORFs, which include various proteins related to brown plant hopper resistance.
Author Contributions
Conceptualization, J.-R.P. and R.J.; methodology and investigation, J.-R.P. and S.Y.; analysis, J.-R.P. and S.Y.; writing—review and editing, J.-R.P., S.Y., and R.J.; writing—review and supervision, K.-M.K.; corresponding author, K.-M.K. All authors contributed significantly to the discussion of the results and the preparation of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
This work was supported by a grant from the New Breeding Technologies Development Program (Project No. PJ014793012020), Rural Development Administration, Republic of Korea.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sogawa, K.; Cheng, C.H. Economic Thresholds, Nature of Damage and Losses Caused by the Brown Planthopper; International Rice Research Institute: Los Banos, Philippines, 1979; pp. 125–142. [Google Scholar]
- Brar, D.S.; Virk, P.S.; Jena, K.K.; Khush, G.S. Breeding for Resistance to Planthoppers in Rice. In Plant Hoppers: New Threats to the Sustainability of Intensive Rice Production Systems in Asia; International Rice Research Institute: Los Banos, Philippines, 2009; pp. 401–409. [Google Scholar]
- Bae, S.H.; Pathak, M.D. Life history of Nilaparvata lugens (Homoptera: Delphacidae) and susceptibility of rice cultivars to its attacks. Ann. Entomol. Soc. Am. 1970, 63, 149155. [Google Scholar] [CrossRef]
- Cabauatan, P.Q.; Cabunagan, R.C.; Choi, I.R. Rice viruses transmitted by the brown planthopper Nilaparvata lugens Stål. In Planthoppers: New Threats to the Sustainability of Intensive Rice Production Systems in Asia; Heong, K.L., Hardy, B., Eds.; International Rice Research Institute: Los Baños, Philippines, 2009; pp. 357–368. [Google Scholar]
- Hibino, H.; Usugi, T.; Omura, T.; Tsuchizaki, T.; Shohara, K.; Iwasaki, M. Rice grassy stunt virus: A planthopper-borne circular filament. Phytopathology 1985, 75, 894–899. [Google Scholar] [CrossRef]
- Ling, K.C. Rice Virus Diseases; International Rice Research Institute: Manila, Philippines, 1972. [Google Scholar]
- Kim, Y.H.; Lee, J.O.; Park, H.C.; Kim, M.S. Plant Damages and Yields of the Different Rice Cultivars to Brown Planthopper (Nilaparvata lugens S.) in Fields. Korean J. Plant Protect. 1985, 24, 79–83. [Google Scholar]
- Saxena, R.C.; Barrion, A.A. Biotypes of the Brown Planthopper Nilaparvata lugens (Stål) and Strategies in Deployment of Host Plant Resistance. Int. J. Trop. Insect Sci. 1985, 6, 271–289. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, J.O. Studies on rice damage due to time migration of the brown planthopper (Nilaparvata lugens) in Korea. Rice Entomol. Newsl. 1976, 4, 17. [Google Scholar]
- Rashid, M.M.; Ahmed, N.; Jahan, M.; Islam, K.S.; Nansen, C.; Willers, J.L.; Ali, M.P. Higher fertilizer inputs increase fitness traits of brown planthopper in rice. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Li, Z.; Xue, Y.; Zhou, H.; Li, Y.; Usman, B.; Jiao, X.; Qiu, Y. High-resolution mapping and breeding application of a novel brown planthopper resistance gene derived from wild rice (Oryza. rufipogon Griff). Rice 2019, 12, 41. [Google Scholar] [CrossRef]
- Fan, C.C.; Yu, X.Q.; Xing, Y.Z.; Xu, C.G.; Luo, L.J.; Zhang, Q. The main effects, epistatic effects and environmental interactions of QTLs on the cooking and eating quality of rice in a doubled-haploid line population. Theor. Appl. Genet. 2005, 110, 1445–1452. [Google Scholar] [CrossRef]
- Qin, Y. Detection of Main-Effect QTLs, Epistasis and QTL × Environmental Interactions for Grain Quality in Rice (Oriza sativa L.). Ph.D. Thesis, Kyungpook National University, Daegu, Korea, 2007. [Google Scholar]
- Septiningsih, E.M.; Trijatmiko, K.R.; Moeljopawiro, S.; McCouch, S.R. Identification of quantitative trait loci for grain quality in an advanced backcross population derived from the Oryza sativa variety IR64 and the wild relative O. rufipogon. Theor. Appl. Genet. 2003, 107, 1433–1441. [Google Scholar] [CrossRef]
- Alam, S.N.; Cohen, M.B. Detection and analysis of QTLs for resistance to the brown planthopper, Nilaparvata lugens, in a doubled-haploid rice population. Theor. Appl. Genet. 1998, 97, 1370–1379. [Google Scholar] [CrossRef]
- Hirabayashi, H.; Kaji, R.; Okamoto, M.; Ogawa, T.; Brar, D.S.; Angeles, E.R.; Khush, G.S. Mapping QTLs for brown planthopper (BPH) resistance introgressed from Oryza officinalis in rice. In Advances in Rice Genetics; Khush, G.S., Khush, D.S., Hardy, B., Eds.; IRRI: Los Banos, Philippines, 2008; Volume 8, pp. 268–270. [Google Scholar]
- Jena, K.K.; Jeung, J.U.; Lee, J.H.; Choi, H.C.; Brar, D.S. High-resolution mapping of a new brown planthopper (BPH) resistance gene, Bph18 (t), and marker-assisted selection for BPH-resistant in rice (Oryza sativa L.). Theor. Appl. Genet. 2006, 112, 288–297. [Google Scholar] [CrossRef]
- Huh, J.; Kang, B.; Nahm, S.; Kim, S.; Ha, K.; Lee, M.H.; Kim, B.D. A candidate gene approach identified phytoene synthase as the locus for mature fruit color in red pepper (Capsicum spp.). Theor. Appl. Genet. 2001, 102, 524–530. [Google Scholar] [CrossRef]
- Seah, S.; Sivasithamparam, K.; Karakousis, A.; Lagudah, E.S. Cloning and characterisation of a family of disease resistance gene analogs from wheat and barley. Theor. Appl. Genet. 1998, 97, 937–945. [Google Scholar] [CrossRef]
- Forster, B.P.; Thomas, W.T. Doubled haploids in genetics and plant breeding. Plant Breed. 2005, 25, 57–88. [Google Scholar]
- Fan, Y.; Shabala, S.; Ma, Y.; Xu, R.; Zhou, M. Using QTL mapping to investigate the relationships between abiotic stress tolerance (drought and salinity) and agronomic and physiological traits. BMC Genom. 2015, 16, 43. [Google Scholar] [CrossRef]
- Kim, S.M.; Qin, Y.; Shon, J.K. Analysis of QTLs related to resistance to brown planthopper in rice. Korean J. Breed. Sci. 2009, 41, 236–243. [Google Scholar]
- Manly, K.F.; Olson, J.M. Overview of QTL mapping software and introduction to Map Manager QT. Mamm. Genome 1999, 10, 327–334. [Google Scholar] [CrossRef]
- Zeng, Z.B. Precision mapping of quantitative trait loci. Genetics 1994, 136, 1457–1468. [Google Scholar]
- McCouch, S.R.; CGSNL (Committee on Gene Symbolization, Nomenclature and Linkage, Rice Genetics Cooperative). Gene nomenclature system for rice. Rice 2008, 1, 72–84. [Google Scholar] [CrossRef]
- Hussain, A.; Mun, B.G.; Imran, Q.M.; Lee, S.U.; Adamu, T.A.; Shahid, M.; Kim, K.M.; Yun, B. Nitric oxide mediated transcriptome profiling reveals activation of multiple regulatory pathways in Arabidopsis thaliana. Front. Plant Sci. 2016, 7, 975. [Google Scholar] [CrossRef]
- Velusamy, R.; Ganesh, M.; Johnson, Y.S. Mechanisms of resistance to the brownplanthopper Nilaparvata lugens in wild rice (Oryza spp.) cultivars. Entomol. Exp. Appl. 1995, 74, 245–251. [Google Scholar] [CrossRef]
- Khush, G.S. Selecting rice for simply inherited resistance. In Plant Breeding in the 1990s; Stalker, H.T., Murphy, J.P., Eds.; CAB International: Wallingford, UK, 1992; pp. 303–322. [Google Scholar]
- Athwal, D.S.; Pathak, M.D.; Bacalangco, E.H.; Pura, C.D. Genetics of resistance to brown planthoppers and green leafhoppers in Oryza sativa L. Crop Sci. 1971, 11, 747–750. [Google Scholar] [CrossRef]
- Myint, K.K.M.; Fujita, D.; Matsumura, M.; Sonoda, T.; Yoshimura, A.; Yasui, H. Mapping and pyramiding of two major genes for resistance to the brown planthopper (Nilaparvata lugens [Stål]) in the rice cultivar ADR52. Theor. Appl. Genet. 2012, 124, 495–504. [Google Scholar] [CrossRef]
- Ren, X.; Wang, X.; Yuan, H.; Weng, Q.; Zhu, L.; He, G. Mapping quantitative trait loci and expressed sequence tags related to brown planthopper resistance in rice. Plant Breed. 2004, 123, 342–348. [Google Scholar] [CrossRef]
- Xiao, J.; Li, J.; Yuan, L.; Iwata, N. Identification of QTLs affecting traits of agronomic in a recombinant inbred population derived from a subspecific rice cross. Theor. Appl. Genet. 1996, 92, 230–244. [Google Scholar] [CrossRef]
- Keen, N.T. The molecular biology of disease resistance. In 10 Years Plant Molecular Biology; Springer: Dordrecht, The Netherlands, 1992; pp. 109–122. [Google Scholar]
- Lamb, C.J.; Lawton, M.A.; Dron, M.; Dixon, R.A. Signals and transduction mechanisms for activation of plant defenses against microbial attack. Cell 1989, 56, 215–224. [Google Scholar] [CrossRef]
- Janda, T.; Szalai, G.; Tari, I.; Paldi, E. Hydroponic treatment with salicylic acid decreases the effects of chilling injury in maize (Zea mays L.) plants. Planta 1999, 208, 175–180. [Google Scholar] [CrossRef]
- Raskin, I. Role of salicylic acid in plants. Annu. Rev. Plant Biol. 1992, 43, 439–463. [Google Scholar] [CrossRef]
- Schmid, J.; Amrhein, N. Molecular organization of the shikimate pathway in higher plants. Phytochemistry 1995, 39, 737–749. [Google Scholar] [CrossRef]
- Hu, P.; Meng, Y.; Wise, R.P. Functional contribution of chorismate synthase, anthranilate synthase, and chorismate mutase to penetration resistance in barley–powdery mildew interactions. Mol. Plant Microbe Interact. 2009, 22, 311–320. [Google Scholar] [CrossRef]
- Parsons, J.F.; Jensen, P.Y.; Pachikara, A.S.; Howard, A.; Eisenstein, E.; Ladner, J.E. Structure of Escherichia coli aminodeoxychorismate synthase: Architectural conservation and diversity in chorismate-utilizing enzymes. Biochemistry 2002, 41, 2198–2208. [Google Scholar] [CrossRef] [PubMed]
- Wildermuth, M.C.; Dewdney, J.; Wu, G.; Ausubel, F.M. Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nature 2001, 414, 562–565. [Google Scholar] [CrossRef] [PubMed]
- Basset, G.J.; Quinlivan, E.P.; Ravanel, S.; Rébeillé, F.; Nichols, B.P.; Shinozaki, K.; Hanson, A.D. Folate synthesis in plants: The p-aminobenzoate branch is initiated by a bifunctional PabA-PabB protein that is targeted to plastids. Proc. Natl. Acad. Sci. USA 2004, 101, 1496–1501. [Google Scholar] [CrossRef] [PubMed]
- Waller, J.C.; Akhtar, T.A.; Lara-Núñez, A.; Gregory III, J.F.; McQuinn, R.P.; Giovannoni, J.J.; Hanson, A.D. Developmental and feedforward control of the expression of folate biosynthesis genes in tomato fruit. Mol. Plant. 2010, 3, 66–77. [Google Scholar] [CrossRef]
- Yoo, H.; Widhalm, J.R.; Qian, Y.; Maeda, H.; Cooper, B.R.; Jannasch, A.S.; Dudareva, N. An alternative pathway contributes to phenylalanine biosynthesis in plants via a cytosolic tyrosine: Phenylpyruvate aminotransferase. Nat. Commun. 2013, 4, 2833. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).