Abstract
Insect pests cause significant global agricultural damage and lead to major financial and environmental costs. Crops contain intrinsic defenses to protect themselves from such pests, including a wide array of specialized secondary metabolite-based defense chemicals. These chemicals can be induced upon attack (phytoalexins) or are constitutive (phytoanticipins), and can have a direct impact on the pests or be used indirectly to attract their natural enemies. They form part of a global arms race between the crops and their insect pests, with the insects developing methods of suppression, avoidance, detoxification, or even capture of their hosts defensive chemicals. Harnessing and optimizing the chemical defense capabilities of crops has the potential to aid in the continuing struggle to enhance or improve agricultural pest management. Such strategies include breeding for the restoration of defense chemicals from ancestral varieties, or cross-species transfer of defense metabolite production.
1. Introduction
In order to survive and reproduce, plants must be able to defend themselves from attack. Such attacks come from a range of organisms including microbes and animals, but some of the most predominant consumers of plants are insect herbivores. Around two-thirds of known insect species are phytophagous (use live plants as a food source, excluding pollen and nectar), and insect pests cause significant global crop losses both by direct damage and by the transmission of plant diseases. Insect herbivores can be broadly categorized as having either chewing or pierce-and-suck feeding styles. Each predicts the type of crop damage they cause, with chewing insects causing potentially massive mechanical damage, and piercing/sucking insects causing nutrient loss and often vectoring diseases. Furthermore, insects can have either a broad host range using many different types of plants as food (generalists) or have a more limited specialized range of hosts (specialists). Often the more specialized the insect is, the more adapted it is at dealing with specialized host defenses [1].
Plants have a variety of inducible and constitutive defense mechanisms with which to defend themselves from attack. These include structural defenses such as thorns and waxy cuticles as well as protein-based and chemical defenses [2,3,4]. Plant defense chemicals consist of secondary metabolites, the core structures of which are predominantly terpenes, benzenoids, phenylpropanoids, flavonoids, or N-containing compounds, which are modified by hydroxylation, glycosylation, methylation, acylation, and prenylation at various positions to produce a vast array of compounds [5,6]. The identification and functional characterization of plant defense compounds is ongoing with a focus on compounds found in important crops and model systems, or compounds with known medicinal properties. However, many compounds have yet to be structurally confirmed, and others have limited information about their production or function.
Plant defense chemicals can be grouped by whether they are inducible or constitutively produced. Initially, these classes were characterized based on their response to microbial pathogens and were termed phytoalexins and phytoanticipins, respectively. They were defined as follows: phytoalexins “low molecular weight, antimicrobial compounds that are both synthesized by and accumulated in plants after exposure to microorganisms” [7], and phytoanticipins “low molecular weight antimicrobial compounds present in plants before challenge by microorganisms or produced after infection solely from preexisting constituents” [8]. As many of these defense compounds are effective against multiple types of organisms, these definitions have since been broadened to include molecules that respond to various biological threats including insect pests. In this perspective, we provide an overview of how phytoalexins and phytoanticipins function in crops against insect pests. We provide illustrative examples of the types of chemicals used in direct defense and describe their known function. We also discuss how volatiles are used in indirect defense to attract the natural enemies of pests. It is important to recognize that defense chemicals are not always effective against all insect pests, as specialized insects can detoxify specific chemicals and even exploit them for their own benefit. Some examples are provided of how insects achieve this, and strategies are discussed for enhancing crop defense chemicals through breeding, engineering, and cross-species transfer of genes.
2. Phytoalexins
Plants need to respond rapidly to herbivory and produce defense chemicals at the site of attack. One method to do this is for the plants to recognize the attack and then induce the transcription of genes encoding enzymes that synthesize defense compounds. These insect-inducible defense compounds (phytoalexins) can have significant functional roles as feeding deterrents. An early example of phytoalexin insect-inducibility was found in peas (Pisum sativum), where the elicitor Bruchin B from the pea weevil (Bruchus pisorum) was shown to induce a cytochrome P450 isoflavone synthase, resulting in successive accumulation of the chromanocoumarane pisatin [9]. In maize (Zea mays), metabolic analysis of European corn borer (Ostrinia nubilalis) feeding tunnels in stems led to the identification of maize diterpenoid phytoalexins [10,11]. Schmelz et al. observed that prolonged infestation of maize stems by the European corn borer elicited the accumulation of metabolites closely related to ent-kaurane diterpenoids, namely ent-kauran-17-oic acid, ent-kauran-17,19-dioic acid, and ent-kauran-19-al-17-oic acid, termed kauralexins A1 through A3 [10]. Additional observations and chemical isolations identified ent-kaur-15-en-17-oic acid, ent-kaur-15-en-17,19-dioic acid, and ent-kaur-15-en-19-al-17-oic acid, termed kauralexins B1 through B3, respectively. Quantitative analysis of European corn borer infested tissues showed that kauralexins A1, B1, B2, and B3 were elicited to significantly higher levels than those observed in damaged controls. Interestingly, kauralexins A2 and A3 were not significantly different from damaged controls, suggesting that the insect-inducibility of phytoalexins can be specific to different compounds within the same class of metabolites. Subsequent studies in maize also demonstrated that European corn borer stem tunneling also elicited the accumulation of sesquiterpenoid zealexins A1–A4 and B1 [11,12].
In rice (Oryza sativa), insect studies have provided examples of how phytoalexin insect-inducibility can be herbivore-specific. For example, the white backed planthopper (Sogatella furcifera) elicits a strong phytohormone response accompanied by accumulation of the diterpenoid momilactone A, which occurs over the 7-day infestation period [13]. Moreover, continuous feeding by the loreyi leafworm (Mythimna loreyi) larvae elicits momilactone A and momilactone B production as a defense response [14]. In contrast, feeding by the lawn armyworm (Spodoptera mauritia) and the common straight swift butterfly (Parnara guttata) elicits low and inconsistent levels of momilactone A and momilactone B [15,16], suggesting that phytoalexin responses to insect attack can be variable and dependent on the specific plant-insect interaction. General insect-inducibility of phytoalexins has also been observed in soybean (Glycine max), including heightened levels of the isoflavones daidzein, formononetin, and genistein in response to aphid infestation. However, not all isoflavones are inducible as glyceollins and are not significantly changed during aphid infestation [17]. The above examples illustrate the inducible nature of crop phytoalexins in response to diverse herbivore attackers and emphasize their potential as direct defense compounds.
A primary benefit of insect-inducible phytoalexins is their direct feeding deterrent action against herbivory. Early studies investigating this activity showed that the isoflavans 3R-(-)-vestitol and sativan from a member of the pea crop family (Lotus pendunculatus) had substantial feeding deterrent activity against larvae of the grass grub beetle (Costelytra zealandica) [18]. It was subsequently found that pastures with >20% L. pendunculatus had virtually no grass grub beetle presence, indicating a strong role for pea phytoalexins in insect defense [9]. Another early study testing the feeding deterrent activity of lupin (Lupinus angustifolius) root-derived flavonoids showed that the isoflavones maackiain and medicarpin, the isoflavan vestitol, the chromanocoumarane pisatin, the phenolic compound phaseollin, and the isoflavins phaseollinisoflavin and 2′-methoxy phaseollinisoflavin all displayed significant feeding deterrent activity against the grass grub beetle and the African black beetle (Heteronychus arator) [19].
Importantly, some phytoalexins have demonstrated bioactivity beyond agricultural practices. The soybean phytoalexin glyceollin has shown biomedical importance across various human health areas including antiproliferative, antiestrogenic, anti-inflammatory, antioxidative, and anticholesterolemic applications [20]. The potential for glyceollin to have an important role in agronomic applications also exists. When purified glyceollin was applied to common bean (Phaseolus vulgarus) at varying concentrations, strong feeding deterrent activity against the spotted cucumber beetle (Diabrotica undecimpunctata howardi) and the Mexican bean beetle (Epilachna varivestis) was observed [21]. In maize, the agronomic function of phytoalexin bioactivity against insects has also been demonstrated. Exogenous application of kauralexins A3 and B3 on the excised surface of maize stems significantly reduced European corn borer maize consumption compared to controls. An additional example in maize was observed in a class of compounds less commonly associated with phytoalexin activity, namely oxylipins. Oxylipins, such as jasmonic acid, are broadly known for their role in defense signaling. However, a recent study demonstrated that the 9-oxylipin, 10-oxo-11-phytoenoic acid, was strongly damage-inducible in maize silks and caused a concentration-dependent inhibition of corn earworm (Helicoverpa zea) growth [22], illustrating that diverse phytoalexins can have anti-herbivore activity.
3. Phytoanticipins
The disadvantage of inducible defense systems is the delay for the synthesis of new compounds. An alternate strategy is to produce the compounds constitutively in the tissues that are susceptible to attack. Disadvantages of this strategy include the metabolic energy required to produce compounds even in the absence of the insect threat, and that the active form of certain compounds are toxic not only to the insects, but also to the plant itself. A common alternate approach to bypass the toxicity issue is to store the compounds as readily activated non-toxic forms and activate them upon insect attack. These compounds are known as phytoanticipins, as they are produced in anticipation of a threat.
Some pathways have a hybrid approach, where the pathway to produce the compounds is constitutive and the last activating step is inducible. For instance, maysin is a C-glycosyl flavone that is produced constitutively at high levels in the silks of maize and correlates with increased resistance to insect pests including corn earworm [23,24]. Maysin itself, however, is not thought to directly have anti-insect properties, rather it is activated by conversion into quinones by polyphenol oxidases that are induced during tissue damage [25,26]. Quinones formed from maysin and related compounds reduce the availability of proteins and amino acids as a food source by binding to their -SH and -NH2 groups. The plants are, therefore, less nutritive to insect larvae [27,28].
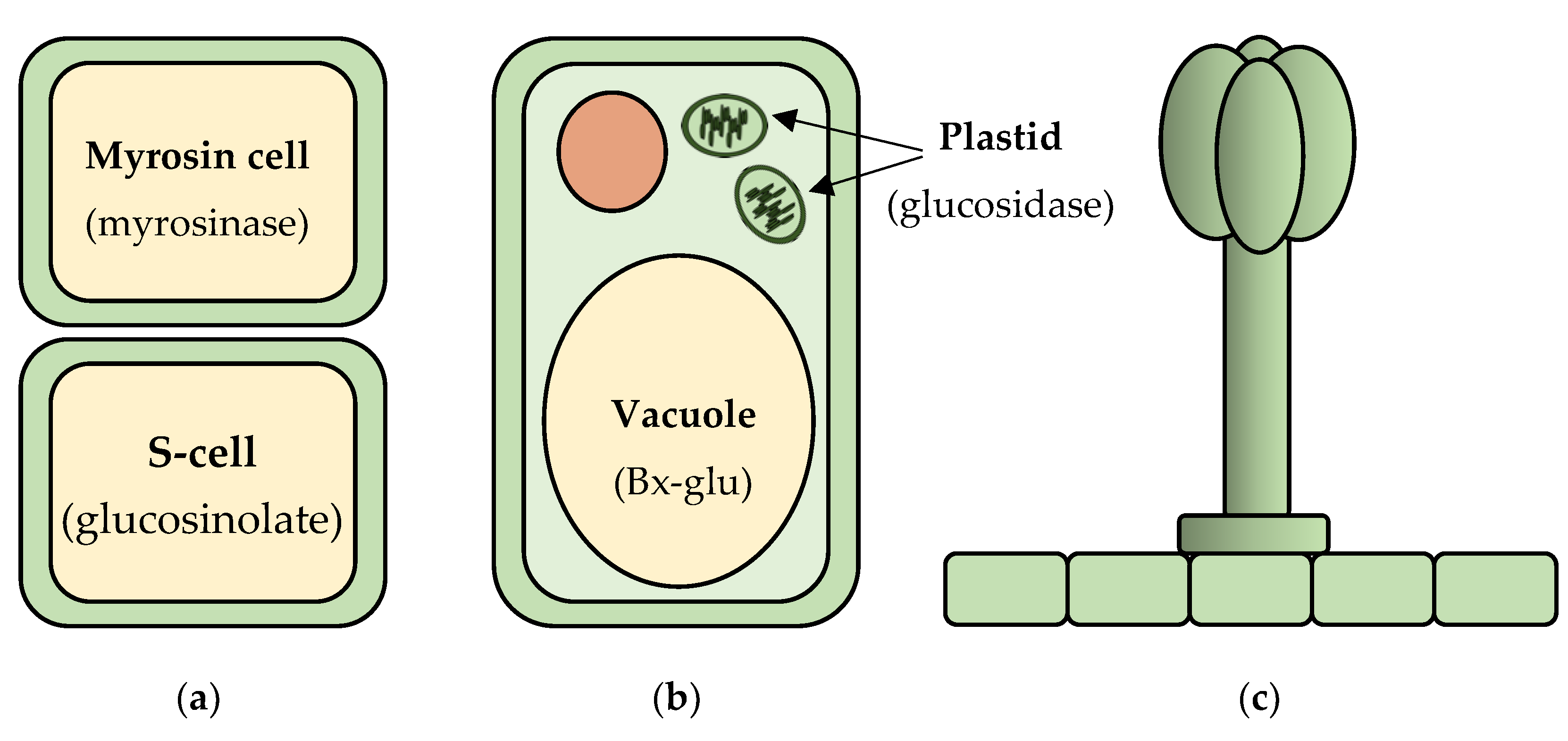
Alternate strategies to deal with toxic compound production utilize the fact that plant cells are mechanically disrupted during herbivory, which leads to the mixing of contents from different cells and different cellular compartments (Figure 1). An example of phytoanticipins that are separated from their activating enzymes in different cells are glucosinolates, which form the so-called mustard bomb. These compounds accumulate under pressure in specialized S-cells [29]. They are activated via hydrolysis by myrosinases, which are stored separately in specialized vegetative vacuoles of specific myrosin cells [30]. Rupture of the plant tissue during herbivory leads to the mixing of the components of the two cell types, and the cleavage of the glucosinolates by the myrosinases to produce unstable aglycones. The aglycones spontaneously rearrange to form a variety of toxic products including isothiocyanates, thiocyanates, nitriles, and epithionitriles [31,32].
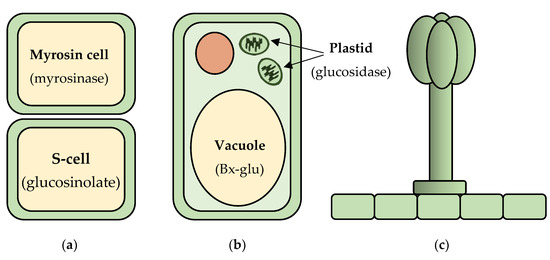
Figure 1.
Mechanisms for isolation and activation of phytoanticipins. (a) In glucosinolate containing plants, the glucosinolates are stored in the vacuoles of sulfur rich S-cells and their activating myrosinase enzymes in adjacent myrosin cells. Tissue rupture during herbivory leads to the mixing of the components of the two cell types and glucosinolate activation; (b) In benzoxazinoid containing plants, the benzoxazinoids are stored as glucose conjugates (Bx-glu) in the vacuoles, and their activating glucosidase enzymes are stored in the plastids of the same cell. Cell rupture during herbivory leads to removal of the glucose moiety by the glucosidase and release of the active aglycone; (c) Glandular trichomes are found on a range of species and are specialized secretory or storage cell structures on the epidermis that physically separate defense molecules from the plant tissues and aid in their exposure to the attacking insect or surrounding environment.
Other phytoanticipins are separated from their activating enzymes by different subcellular compartments. Sorghum (Sorghum bicolor) accumulates the cyanogenic glucoside dhurrin in the cytoplasm [33], whereas the endogenous β-glucosidase, dhurrinase, that activates it is in the chloroplasts [34]. Upon cell rupture during herbivory, cyanogenic glucosides become exposed to their activating hydrolases, which converts them to cyanohydrin aglycones. These compounds spontaneously, or with the assistance of hydroxynitrile lyases, can be converted into hydrogen cyanide (HCN) [35,36,37]. A similar approach is observed for the benzoxazinoids found in maize, wheat (Triticum aestivum), and rye (Secale cereale) that are stored as glucosides in the vacuole. These glucosides are hydrolyzed upon tissue disruption by plastid localized glucosidases, releasing toxic aglycones [38].
An alternate approach to keeping the compounds inactive is to produce them in specialized compartments and secrete them externally in protected tissues. These are often complex mixtures of compounds and can be defined as essential oils, resins, or latex depending on their production from glandular trichomes, laticifers, or resin ducts, respectively [39]. Glandular trichomes, for example, can contain a range of defensive metabolites depending on the species and type of trichome. These can include toxins and terpene volatiles, as well as sticky compounds that act to physically entrap or suffocate insects [40]. For instance, glandular trichomes of the wild tomato Lycopersicon pennellii contain acylsugars that can deter feeding and oviposition of pests such as the leafminer Liriomyza trifolii [41].
4. Mechanisms of Action of Anti-Insect Defense Chemicals
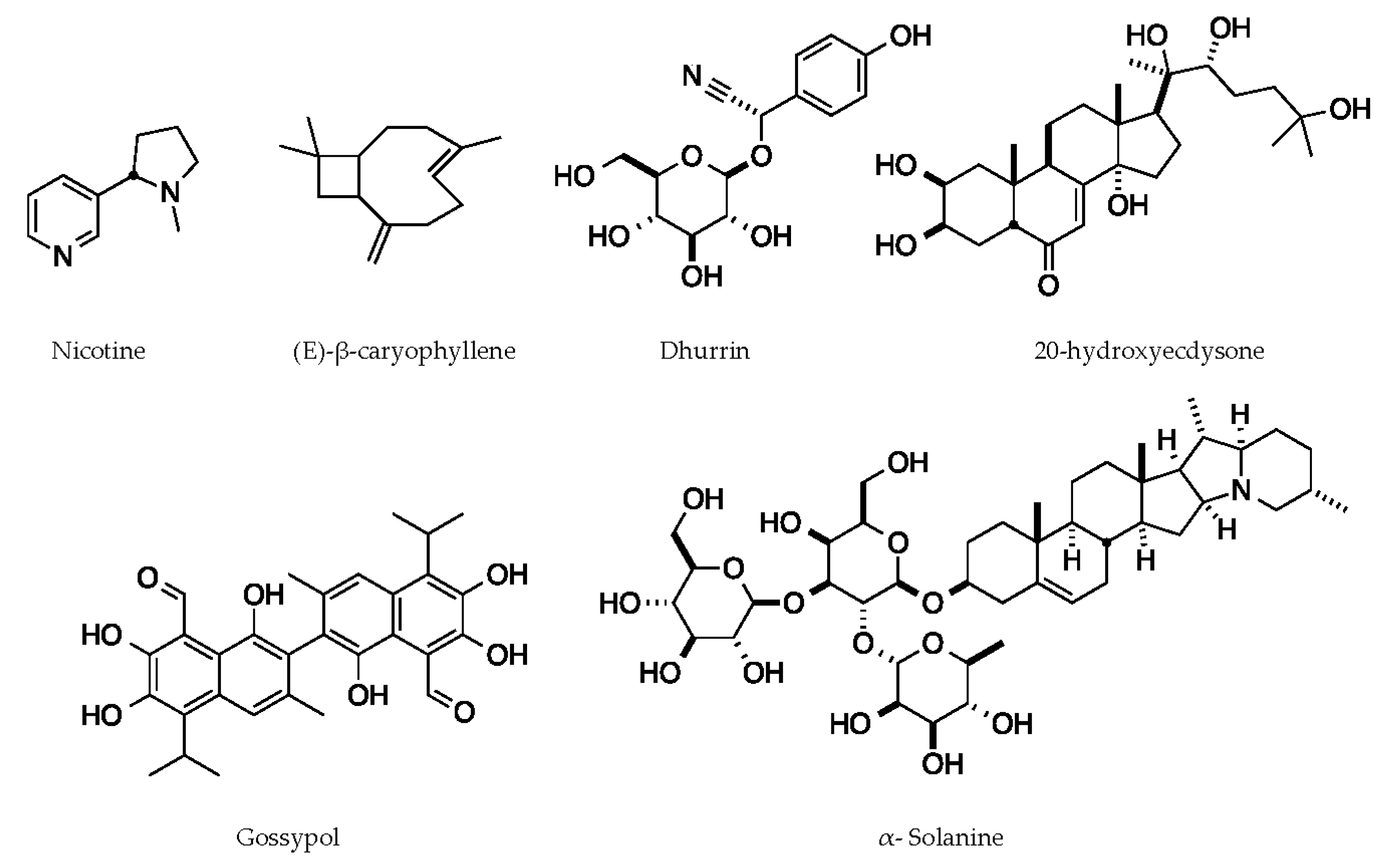
Crops contain a wide variety of defense chemicals that have anti-insect properties. In this section, we present examples of compounds that have a direct impact on their target insects and provide details for the mechanism of action, even if it has only been partially characterized. These chemicals include dhurrin, a cyanogenic glycoside produced by sorghum; α-solanine, a steroidal alkaloid saponin from nightshades such as tomato (Solanum lycopersicum) and potato (Solanum tuberosum) [42]; Gossypol, a sesquiterpene aldehyde from cotton (Gossypium spp.) [43]; 20-hydroxyecdysone, a phytoecdysteroid from spinach (Spinacia oleracea) [44]; Nicotine, an alkaloid from tobacco (Nicotiana tabacum); and (E)-β-caryophyllene, a sesquiterpene volatile from maize (discussed in the following section) [45] (Figure 2).
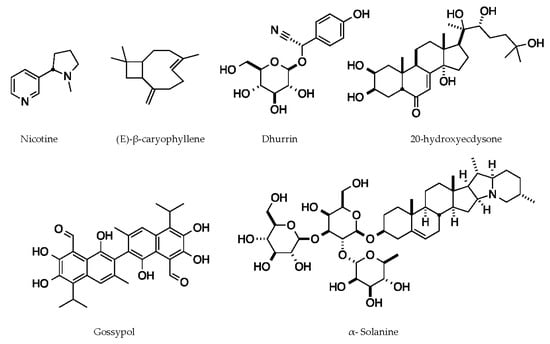
Figure 2.
Representative chemical structures of plant defense compounds with known functions.
Cyanogenic glycosides are a group of amino acid derived compounds that release hydrogen cyanide (HCN) [36], which prevents the binding of oxygen to cytochrome-c oxidase in the mitochondrion electron transfer chain, thus inhibiting respiration. HCN can be toxic to insects. For example, a correlation was established between reduced feeding of grasshopper (Acrida exaltata) nymphs or spotted stalk borer (Chilo partellus) larvae and dhurrin concentrations in sorghum lines [46]. These compounds are also toxic to mammals, as high doses of or prolonged exposure to HCN can cause critical illness or death in livestock fed on high dhurrin sorghum forage [47].
Gossypol is a sesquiterpene aldehyde that was originally isolated from cotton (Gossypium spp.). It has two central aldehyde groups that crosslink to proteins via the amino acid lysine, enabling it to deactivate enzymes [48]. For instance, it can inhibit protease and amylase activities in African cotton leafworm (Spodoptera littoralis) larvae, thus affecting larval weight gain [49]. Gossypol can also bind to membranes, which could affect electron transport in the mitochondria [50]. The critical role of the aldehyde groups was proven when Krempl et al. utilized SB-gossypol, a gossypol derivative in which the = O of the -CHO was replaced by = N, in a feeding experiment with cotton bollworm (Helicoverpa armigera) and tobacco budworm (Heliothis virescens). Larvae fed on a SB-gossypol diet did not suffer the weight loss seen in those fed on the gossypol supplemented diet [48].
Alkaloids, such as nicotine from tobacco, have a chemical ring structure containing a nitrogen atom. To date, there are more than 10,000 known alkaloids [51]. Nicotine and its related compounds exert their toxicity by binding to the nicotine acetylcholine receptor in the insect nervous systems. The binding induces a conformation shift in the receptor that allows an influx of cations into the cell, disrupting the function of the nervous system [52]. Mutations in genes of the nicotine biosynthetic pathway in the wild tobacco Nicotiana attenuate led to three times more damage by native herbivores, especially beet armyworm (Spodoptera exigua), in field trials [53].
Saponins, found in legumes and other plants, are a diverse group of compounds that can form stable, soap-like foams in aqueous solutions [54]. The generally accepted mode of action for saponins relates to their amphipathic properties resulting from the combination of a hydrophobic triterpene or sterol backbone and a hydrophilic carbohydrate chain, which induces formation of pores and vesicles, and causes general membrane disruption [55,56]. Some compounds fall into multiple of these compound categories. For instance, α-solanine and α-chaconine from potato are both a steroidal alkaloids, and saponins that can have anti-insect properties as well as animal toxicity [42].
Plant compounds can also impact insect development. For example, phytoecdysteroids are analogues of the insect hormone ecdystreroid, which controls the phases of development in insects from larva to adult. Phytoecdysteroids such as 20-hydroxyecdysone can disrupt insect development by binding to the ecdysone receptor [57]. These compounds have been identified in an estimated 6% of plants including asparagus (Asparagus spp.), spinach, and quinoa (Chenopodium quinoa) [58,59,60].
5. Volatiles and Indirect Defenses
The preceding section focuses on defense chemicals that have direct effects on insect physiology. Plants produce an array of small-molecule volatile compounds, termed herbivore-induced plant volatiles (HIPVs) [61]. The identity of these compounds varies depending on the species and cultivar of plant. The types of HIPVs produced can include terpenoids, volatile fatty-acid derivatives, benzenoids, phenylpropanoids, and volatile amino-acid derivatives. Natural enemies of many plant pests can use HIPVs as cues to locate infested plants and, thus, their prey/host [62]. HIPVs are, therefore, an indirect defense mechanism, as they attract predatory or parasitic insects, which have a negative impact on the plant pest.
In many cases, it is still not clear precisely which volatiles are responsible for the indirect defense, but specialized parasitic wasps (parasitoids) can clearly distinguish between HIPVs induced by different pests. For example, the specialist parasitoid Cardiochiles nigriceps can differentiate between volatiles induced by its host, tobacco budworm, from that by its non-host, corn earworm [63]. While the use of HIPVs by some parasitoids for host location can be considered innate, several species also demonstrate learned behavioral responses to chemical cues. An example of this is the parasitoid Aphidius rhopalosiphi, which can learn distinct wheat cultivar odors depending on which cultivar their host aphids are reared on [64]. The general trend appears to be that specialized parasitoids have more innate cues and generalists have more learned cues, allowing them to better exploit available hosts.
HIPVs can act in indirect defense both above and below ground, as certain volatiles produced in the roots can diffuse through the soil matrix. For example, maize uses volatiles in both above and below ground defense (Figure 3). Above ground beet armyworm infestation leads to the production of HIPVs from the leaves of maize. This blend of HIPVs contains green leaf volatiles ((Z)-3-hexenal, (Z)-2-hexenal, (Z)-3-hexen-l-ol, and (Z)-3-hexen-1-yl acetate), terpenoids (β-linalool, (E)-4,8-dimethyl-1,3,7-nonatriene, (E)-α-trans-bergamotene, (E)-β-farnesene; (E)-nerolidol; and (E,E)-4,8,12-trimethyl-1,3,7,11-tridecatetraene), and the amino-acid derived volatile indole. These HIPVs diffuse through the air where they attract the parasitoid Cotesia marginiventris, for which beet armyworm is a host [65]. C. marginiventris then locates the beet armyworm on the infested maize plant and oviposits in the larvae. Parasitism of the beet armyworm larvae significantly retards its growth and ability to feed and, thus, protects the maize plant. Below ground infestation of maize with larvae of the beetle western corn rootworm (Diabrotica virgifera virgifera) leads to the production of the volatile sesquiterpene (E)-β-caryophyllene. This compound diffuses through the soil and acts as an attractant to the entomopathogenic nematode Heterorhabditis megidis. Production of (E)-β-caryophyllene by maize roots in response to western corn rootworm damage, therefore, attracts the nematodes to infiltrate the rootworm larvae and kill them, leading to control of the rootworm and less damage to the plant [66].

Figure 3.
Volatile compounds in maize are induced in response to above or below-ground herbivory and act as an indirect defense mechanism via the attraction of predatory or parasitic organisms that help control the attacking pest.
HIPVs can also have direct effects against insect pests; for example, indole can negatively impact African cotton leafworm food consumption and survival. Interestingly though, indole also leads to increased larval growth, even though the leafworm avoids indole-producing plants [67]. HIPVs can often have repellent activities to insect pests. For instance, the terpenes zingiberene, curcumene, p-cymene, α-terpinene, and α-phellandrene from wild tomato species are repellent to whitefly (Bemisia tabaci) [68]. Insects are not without their defenses against HIPVs, as some species such as fall armyworm (Spodoptera frugiperda) can suppress HIPV production in their host plants [69].
6. Detoxification and Utilization of Plant Defense Chemicals by Insects
The evolutionary pressure for plants to continue to develop new and modified defense chemicals is maintained by the ability of insects to evolve to overcome or avoid these defenses. Various insects have developed specialized methods of overcoming their host plant’s defense chemicals. These methods include suppressing host defense responses via the use of secreted effector proteins [70], and avoidance, target site insensitivity, detoxification, disarming, sequestering, or excretion of host defense chemicals [71]. These methods have not only allowed the insects to feed on their hosts, they have also allowed some insects to adopt the defense systems of their host plants. An example of this can be seen in glucosinolate producing brassica crops such cultivars of Brassica oleracea including broccoli and cabbage [72]. Brassica specialist insects, such as the larvae of the diamondback moth (Plutella xylostella) and the desert locust (Schistocerca gregaria), possess sulfatases in their gut, which allow them to feed on glucosinolate-harboring plants [73,74]. These sulfatases disarm glucosinolates by converting them into an inactive form that does not react with myrosinases, allowing the insects to feed safely. Cabbage white butterflies (Pieris rapae) are also specialist herbivores on glucosinolate-containing plants. Their larvae are exposed to cyanide during metabolism of the benzylglucosinolate breakdown product phenylacetonitrile. The larvae have β-cyanoalanine synthases that enable them to metabolize cyanide to β-cyanoalanine and thiocyanate, thus preventing its accumulation and toxicity [75].
Another strategy is to avoid the cell rupture that is necessary for the activation of these compounds. As pierce-and-suck herbivores, cabbage aphids (Brevicoryne brassicae) can bypass the mixing of glucosinolates and myrosinases altogether due to their method of feeding, which avoids damaging multiple cells at once. Furthermore, these aphids can sequester their host plant’s glucosinolates completely intact. This, in addition to the fact that these aphids possess their own myrosinase-containing microbodies, allows the aphids to effectively copy the chemical defense system of their host plants [76,77,78].
Larvae of moths of the genus Zygaena, which have evolved to specialize in cyanogenic plants, can produce their own cyanogenic glucosides, but have also demonstrated the ability to sequester the compounds intact from their host plants [79,80]. Larvae feeding on cyanogenic plants were shown to develop faster, most likely due to the metabolic benefit of not having to produce their own cyanogenic glucosides [81]. A further example is the western corn rootworm, whose larvae are attracted to and sequester intact benzoxazinoids, and assimilate the compounds into their hemolymph [82]. The larvae can also glycosylate the benzoxazinoid breakdown products and use them as defense compounds against entomopathogenic nematodes. The sequestered benzoxazinoid 2-hydroxy-4,7-dimethoxy-1,4-benzoxazin-3-one O-glucoside (HDMBOA-Glc) is directly toxic to the nematodes and their entomopathogenic bacteria, while the converted benzoxazinoid breakdown product 6-methoxy-2-benzoxazolinone N-glucoside (MBOA-Glc), which accumulates on their cuticle and in their frass, makes them less attractive to the nematodes [83]. However, extended exposure of multiple generations of nematodes to benzoxazinoid-harboring rootworms led to their eventual adaptation to the benzoxazinoid containing rootworm larvae [84], illustrating the amazing adaptability of nematodes and insects to defense chemicals.
7. Breeding, Engineering, and Cross Species Transfer of Defense Chemicals
During crop domestication, metabolites involved in pest resistance were often lost due to the lack of selection for these traits in the wake of selecting for improved yield. One such compound is the sesquiterpene 7-epizingiberene that is present in wild tomato (Solanum habrochaites) but not domesticated tomato. When the genes responsible for producing this compound were expressed in domesticated tomato, the plants showed improved herbivore resistance [85]. A similar success was seen with (E)-β-caryophyllene, a sesquiterpene volatile whose production is lacking in many North American maize varieties, yet is present in European and ancestral maize lines [45]. Maize plants were engineered to express (E)-β-caryophyllene synthase from Origanum vulgare. These plants emitted (E)-β-caryophyllene and were shown in field trials to be more effective at attracting entomopathogenic nematodes to control western corn rootworm, resulting in reduced root damage to the plants [86]. A helpful benefit of the re-capture of lost metabolites from wild relatives is that it can now be accomplished either via traditional breeding or transgenic approaches.
As many insect pests have co-evolved with their host plants, they have naturally developed mechanisms to overcome the defense chemicals of their hosts. An alternate strategy for enhancing host defenses is, therefore, to engineer the production of metabolites from different species by cross-species transfer of metabolic pathways. A proof of concept of this approach can be seen in the transfer of dhurrin biosynthesis from sorghum to the model plant Arabidopsis thaliana. This process required the expression of only three sorghum genes, and Arabidopsis was found to contain native glucosidases that could activate dhurrin upon tissue damage. The resultant plants were resistant to the flea beetle Phyllotreta nemorum [87]. A similar effect is also seen in tobacco, where the same sorghum genes produced dhurrin, though at a lower level than in sorghum. In this case, these genes also resulted in negative phenotypes including paler leaves, slower growth, and delayed flowering, possibly due to inadvertent HCN release due to the incomplete channeling of metabolites [88]. Positive anti-insect activity in model plants also does not necessarily translate to success in crop plants. For instance, the sorghum genes, when transferred to grapevine (Vitis vinifera) hairy roots, also led to dhurrin production, but no protection was provided against the specialist insect, grapevine phylloxera (Daktulosphaira vitifoliae) [89].
Dhurrin was not the only compound successfully transferred into crops that ran into problems in the field. For instance, wheat was engineered to express a plastid-targeted gene from peppermint (Mentha × piperita). These plants successfully produced the aphid alarm pheromone, (E)-β-farnesene, and in laboratory tests was repellant to three species of cereal aphids. This activity, however, did not translate to more resistant plants in field trials [90]. A similar strategy in Arabidopsis that led to the constitutive production of (E)-β-farnesene had no direct effect on aphids. The authors suggested that the insects may have become habituated to the continual release of the compound [91]. These studies illustrate the challenges of the cross-species transfer of defense metabolites and reflect the tight control, specialized storage, and flux regulation needed to produce these compounds properly in their native plants. An increased understanding of these factors will likely increase the number of successfully engineered plants with enhanced defense chemistry. However, all such engineering approaches must be carefully considered in terms of the identity and location of the metabolites added, such that they pose no added risk of toxicity or adverse effects to the consumer or environment.
8. Conclusions
Advances in metabolomics and genomics have led to an increased awareness of the sheer scope and possibilities inherent in plant defense chemicals. Substantial progress has been made in determining the structures, biosynthetic pathways, and functions of defense chemicals, and will continue over the coming decades. Genetic engineering studies have led to the successful transfer of the production of defense compounds into new species, elegantly demonstrating that modification of a crop’s defense chemical arsenal is possible. However, successful attempts to translate this into real insect resistance under field conditions remain limited. The current roadblocks to successful cross-species transfer appear to derive from problems in the production of the compounds, such as constitutive production of phytoalexins, insufficient substrates, or incomplete metabolite channeling of toxic intermediates. Other issues include lack of appropriate storage compartments for phytoanticipins, and ineffective action of defense compounds against the target insect. Our improved understanding of defense chemicals, their regulation, range of action, and effects at an ecosystem level will help future attempts to use them to increase insect resistance in crops. Finally, this perspective has focused on the defense chemicals in crop plants; however, many non-crop species have extensive defense chemicals, some of which have provided template compounds for insecticide development. The ability to leverage these defenses for endogenous crop protection would greatly enhance our ability to control insect pests without relying solely on pesticide applications.
Author Contributions
Conceptualization, A.K.B.; writing—original draft preparation, J.P.Y.-C., H.V.T., J.M., S.A.C., A.K.B.; writing—review and editing, all authors; funding acquisition, A.K.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the United States Department of Agriculture (USDA) Agricultural Research Service (ARS) Project number 6036-11210-001-00D, USDA National Institute of Food and Agriculture (NIFA) grant 2018-51181-28419, and the Florida Department of Agriculture and Consumer Services (FDACS) (Specialty Crop Block Grant) AM180100XXXXG046.
Acknowledgments
We thank J. Beck for rendering the structures used in Figure 2 using the ChemDraw software. J.M. is supported in part by an appointment to the Program Research Participation Program administered by the Oak Ridge Institute for Science and Education (ORISE) through an interagency agreement between the U.S. Department of Energy (DOE) and the USDA-ARS. ORISE is managed by ORAU under DOE contract number DE-SC0014664. The USDA is an equal opportunity employer.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Douglas, A.E. Strategies for enhanced crop resistance to insect pests. Ann. Rev. Plant Biol. 2017, 69, 637–660. [Google Scholar] [CrossRef] [PubMed]
- Pedras, M.S.C.; Yaya, E.E. Plant chemical defenses: Are all constitutive antimicrobial metabolites phytoanticipins? Nat. Prod. Commun. 2015, 10, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Wittstock, U.; Gershenzon, J. Constitutive plant toxins and their role in defense against herbivores and pathogens. Curr. Opin. Plant Biol. 2002, 5, 300–307. [Google Scholar] [CrossRef]
- Lev-Yadun, S. Plants are not sitting ducks waiting for herbivores to eat them. Plant Signal. Behav. 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Alseekh, S.; Fernie, A.R. Metabolomics 20 years on: What have we learned and what hurdles remain? Plant J. 2018, 94, 933–942. [Google Scholar] [CrossRef]
- Wang, S.; Alseekh, S.; Fernie, A.R.; Luo, J. The structure and function of major plant metabolite modifications. Mol. Plant 2019, 12, 899–919. [Google Scholar] [CrossRef]
- Paxton, J.D. A new working definition of the term “phytoalexin”. Plant Dis. 1980, 64, 734. [Google Scholar]
- VanEtten, H.D.; Mansfield, J.W.; Bailey, J.A.; Farmer, E.E. Two classes of plant antibiotics: Phytoalexins versus “phytoanticipins”. Plant Cell 1994, 6, 1191–1192. [Google Scholar] [CrossRef]
- Cooper, L.D.; Doss, R.P.; Price, R.; Peterson, K.; Oliver, J.E. Application of Bruchin B to pea pods results in the up-regulation of CYP93C18, a putative isoflavone synthase gene, and an increase in the level of pisatin, an isoflavone phytoalexin. J. Exp. Bot. 2005, 56, 1229–1237. [Google Scholar] [CrossRef]
- Schmelz, E.A.; Kaplan, F.; Huffaker, A.; Dafoe, N.J.; Vaughan, M.M.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E.A. Identity, regulation, and activity of inducible diterpenoid phytoalexins in maize. Proc. Natl. Acad. Sci. USA 2011, 108, 5455–5460. [Google Scholar] [CrossRef]
- Huffaker, A.; Kaplan, F.; Vaughan, M.V.; Dafoe, N.J.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E.A.; Schmelz, E.A. Novel acidic sesquiterpenoids constitute a dominant class of pathogen-induced phytoalexins in maize. Plant Physiol. 2011, 156, 2082–2097. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S.A.; Huffaker, A.; Sims, J.; Hunter, C.T.; Block, A.; Vaughan, M.M.; Willett, D.; Romero, M.; Mylroie, J.E.; Williams, W.P.; et al. Fungal and herbivore elicitation of the novel maize sesquiterpenoid, zealexin A4, is attenuated by elevated CO2. Planta 2018, 247, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Kanno, H.; Hasegawa, M.; Kodama, O. Accumulation of salicylic acid, jasmonic acid and phytoalexins in rice, Oryza sativa, infested by the white-backed planthopper, Sogatella furcifera (Hemiptera: Delphacidae). Appl. Entomol. Zool. 2012, 47, 27–34. [Google Scholar] [CrossRef]
- Shinya, T.; Hojo, Y.; Desaki, Y.; Christeller, J.T.; Okada, K.; Shibuya, N.; Galis, I. Modulation of plant defense responses to herbivores by simultaneous recognition of different herbivore-associated elicitors in rice. Sci. Rep. 2016, 6, 32537. [Google Scholar] [CrossRef] [PubMed]
- Alamgir, K.M.; Hojo, Y.; Christeller, J.T.; Fukumoto, K.; Ishhiki, R.; Shinya, T.; Baldwin, I.T.; Galis, I. Systematic analysis of rice (Oryza sativa) metabolic responses to herbivory. Plant Cell Environ. 2016, 39, 453–466. [Google Scholar] [CrossRef]
- Tanabe, K.; Hojo, Y.; Shinya, T.; Galis, I. Molecular evidence for biochemical diversification of phenolamide biosynthesis in rice plants. J. Int. Plant Biol. 2016, 58, 901–913. [Google Scholar] [CrossRef]
- Hohenstein, J.D.; Studham, M.E.; Klein, A.; Kovinich, N.; Barry, N.; Lee, Y.; MacIntosh, G.C. Transcriptional and chemical changes in soybean leaves in response to long-term aphid colonization. Front. Plant Sci. 2019, 10, 310. [Google Scholar] [CrossRef]
- Russell, G.B.; Sutherland, O.W.R.; Hutchins, R.F.N.; Christmas, P.E. A phytoalexin with insect feeding-deterrent activity. J. Chem. Ecol. 1978, 4, 571–579. [Google Scholar] [CrossRef]
- Lane, G.A.; Sutherland, O.R.W.; Skipp, R.A. Isoflavonoids as insect feeding deterrents and antifungal components from root of Lupinus angustifolius. J. Chem. Ecol. 1987, 13, 771–783. [Google Scholar] [CrossRef]
- Pham, T.H.; Lecomte, S.; Efstathiou, T.; Ferriere, F.; Pakdel, F. An update on the effects of glyceollins on human health: Possible anticancer effects and underlying mechanisms. Nutrients 2019, 11, 79. [Google Scholar] [CrossRef]
- Fischer, D.C.; Kogan, M.; Paxton, J. Deterrency of Mexican bean beetle (Coleoptera: Coccinellidae) feeding by free phenolic acids. J. Entomol. Sci. 1990, 25, 230–238. [Google Scholar] [CrossRef]
- Christensen, S.A.; Huffaker, A.; Kaplan, F.; Sims, J.; Ziemann, S.; Doehlemann, G.; Ji, L.; Schmitz, R.J.; Kolomiets, M.V.; Alborn, H.T.; et al. Maize death acids, 9-lipoxygenase-derived cyclopente(a)nones, display activity as cytotoxic phytoalexins and transcriptional mediators. Proc. Natl. Acad. Sci. USA 2015, 112, 11407–11412. [Google Scholar] [CrossRef] [PubMed]
- Waiss, A.C.; Chan, B.G.; Elliger, C.A.; Wiseman, B.R.; McMillian, W.W.; Widstrom, N.W.; Zuber, M.S.; Keaster, A.J. Maysin, a flavone glycoside from corn silks with antibiotic activity toward corn earworm. J. Econ. Entomol. 1979, 72, 256–258. [Google Scholar] [CrossRef]
- Elliger, C.A.; Chan, B.G.; Waiss, A.C.; Lundin, R.E.; Haddon, W.F. C-glycosylflavones from Zea mays that inhibit insect development. Phytochemistry 1980, 19, 293–297. [Google Scholar] [CrossRef]
- Levings, C.S.; Stuber, C.W. A maize gene controlling silk browning in response to wounding. Genetics 1971, 69, 491–498. [Google Scholar]
- Byrne, P.F.; Darrah, L.L.; Snook, M.E.; Wiseman, B.R.; Widstrom, N.W.; Moellenbeck, D.J.; Barry, B.D. Maize silk-browning, maysin content, and antibiosis to the corn earworm, Helicoverpa zea (Boddie). Maydica 1996, 41, 13–18. [Google Scholar]
- Felton, G.W.; Donato, K.; Del Vecchio, R.J.; Duffey, S.S. Activation of plant foliar oxidases by insect feeding reduces nutritive quality of foliage for noctuid herbivores. J. Chem. Ecol. 1989, 15, 2667–2694. [Google Scholar] [CrossRef]
- Wiseman, B.R.; Carpenter, J.E. Growth inhibition of corn earworm (Lepidoptera: Noctuidae) larvae reared on resistant corn silk diets. J. Econ. Entomol. 1995, 88, 1037–1043. [Google Scholar] [CrossRef]
- Koroleva, O.A.; Gibson, T.M.; Cramer, R.; Stain, C. Glucosinolate-accumulating S-cells in Arabidopsis leaves and flower stalks undergo programmed cell death at early stages of differentiation. Plant J. 2010, 64, 456–469. [Google Scholar] [CrossRef]
- Shirakawa, M.; Hara-Nishimura, I. Specialized vacuoles of myrosin cells: Chemical defense strategy in Brassicales plants. Plant Cell Physiol. 2018, 59, 21. [Google Scholar] [CrossRef]
- Burow, M.; Halkier, B.A. How does a plant orchestrate defense in time and space? Using glucosinolates in Arabidopsis as case study. Curr. Opin. Plant Biol. 2017, 38, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Halkier, B.A.; Gershenzon, J. Biology and Biochemistry of Glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Heraud, P.; Cowan, M.F.; Marzec, K.M.; Moller, B.L.; Blomstedt, C.K.; Gleadow, R. Label-free raman hyperspectral imaging analysis localizes the cyanogenic glucoside dhurrin to the cytoplasm in sorghum cells. Sci. Rep. 2018, 8, 2691. [Google Scholar] [CrossRef] [PubMed]
- Thayer, S.S.; Conn, E.E. Subcellular localization of dhurrin beta-glucosidase and hydroxynitrile lyase in the mesophyll cells of sorghum leaf blades. Plant Physiol. 1981, 67, 617–622. [Google Scholar] [CrossRef]
- Poulton, J.E. Cyanogenesis in Plants. Plant Physiol. 1990, 94, 401–405. [Google Scholar] [CrossRef]
- Gleadow, R.M.; Møller, B.L. Cyanogenic glycosides: Synthesis, physiology, and phenotypic plasticity. Annu. Rev. Plant Biol. 2014, 65, 155–185. [Google Scholar] [CrossRef]
- Vetter, J. Plant cyanogenic glycosides. Toxicon 2000, 38, 11–36. [Google Scholar] [CrossRef]
- Frey, M.; Schullehner, K.; Dick, R.; Fiesselmann, A.; Gierl, A. Benzoxazinoid biosynthesis, a model for evolution of secondary metabolic pathways in plants. Phytochemistry 2009, 70, 1645–1651. [Google Scholar] [CrossRef]
- Abarca, L.F.S.; Klinkhamer, P.G.L.; Choi, Y.H. Plant latex, from ecological interests to bioactive chemical resources. Planta Med. 2019, 85, 856–868. [Google Scholar] [CrossRef]
- Glas, J.J.; Schimmel, B.C.J.; Alba, J.M.; Escobar-Bravo, R.; Schuurink, R.C.; Kant, M. R Plant glandular trichomes as targets for breeding or engineering of resistance to herbivores. Int. J. Mol. Sci. 2012, 13, 17077–17103. [Google Scholar] [CrossRef]
- Hawthorne, D.J.; Shapiro, J.A.; Tingey, W.M.; Mutschler, M.A. Trichome-borne and artificially applied acylsugars of wild tomato deter feeding and oviposition of the leafminer Liriomyza trifolii. Entomol. Exp. Appl. 1992, 65, 65–73. [Google Scholar] [CrossRef]
- Paudel, J.R.; Davidson, C.; Song, J.; Maxim, I.; Aharoni, A.; Tai, H.H. Pathogen and pest responses are altered due to RNAi-mediated knockdown of GLYCOALKALOID METABOLISM 4 in Solanum tuberosum. Mol. Plant Microbe Interact. 2017, 30, 876–885. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Ruan, J.; Huang, J.; Fang, X.; Mao, Y.; Wang, L.; Chen, X.; Yang, C. Gossypol: Phytoalexin of cotton. Sci. China Life Sci. 2016, 59, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Soriano, I.R.; Riley, I.T.; Potter, M.J.; Bowers, W.S. Phytoecdysteroids: A novel defense against plant-parasitic nematodes. J. Chem. Ecol. 2004, 30, 1885–1899. [Google Scholar] [CrossRef] [PubMed]
- Köllner, T.G.; Held, M.; Lenk, C.; Hiltpold, I.; Turlings, T.C.J.; Gershenzon, J.; Degenhardt, J. A maize (E)-β-caryophyllene synthase implicated in indirect defense responses against herbivores is not expressed in most American maize varieties. Plant Cell 2008, 20, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Woodhead, S.; Padgham, D.E.; Bernays, E.A. Insect feeding on different sorghum cultivars in relation to cyanide and phenolic acid content. Ann. Appl. Biol. 1980, 95, 151–157. [Google Scholar] [CrossRef]
- Hayes, C.M.; Burrow, G.B.; Brown, P.J.; Thurber, C.; Xin, Z.; Burke, J.J. Natural variation in synthesis and catabolism genes influences dhurrin content in sorghum. Plant Genome 2015, 8, 1–9. [Google Scholar] [CrossRef]
- Krempl, C.; Heidel-Fischer, H.M.; Jimemez-Alemán, G.H.; Reichelt, M.; Menezes, R.C.; Boland, W.; Vogel, H.; Heckel, D.; Jouβen, N. Gossypol toxicity and detoxification in Helicoverpa armigera and Heliothis virescens. Insect Biochem. Mol. Biol. 2016, 78, 69–77. [Google Scholar] [CrossRef]
- Meisner, J.; Ishaaya, I.; Ascher, K.R.S.; Zur, M. Gossypol inhibits protease and amylase activity of Spodoptera littoralis larvae. Ann. Entomol. Soc. Am. 1978, 71, 5–9. [Google Scholar] [CrossRef]
- Reyes, J.; Allen, J.; Tanphaicgitr, N.; Bellve, A.; Benos, D. Molecular mechanism of gossypol action on lipid membranes. J. Biol. Chem. 1984, 259, 9607–9615. [Google Scholar]
- Chromel, M.; Guittonny-Larchevêque, M.; Fernandez, C.; Gallet, C.; DesRocher, A.; Paré, D.; Jackson, B.; Baldy, V. Plant secondary metabolites: A key driver of litter decomposition and soil nutrient cycling. J. Ecol. 2016, 104, 1527–1541. [Google Scholar] [CrossRef]
- Ihara, M.; Buckingham, S.D.; Matsuda, K.; Sattelle, D.B. Modes of action, resistance and toxicity of insecticides targeting nicotinic acetylcholine receptors. Curr. Med. Chem. 2017, 24, 2925–2934. [Google Scholar] [CrossRef] [PubMed]
- Steppuhn, A.; Gase, K.; Krock, B.; Halitschke, R.; Baldwin, I.T. Nicotine’s defensive function in nature. PLoS Biol. 2004, 2, e217. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Arunasalam, K.; Yeung, D.; Kakuda, Y.; Mittal, G.; Jiang, Y. Saponins from edible legumes: Chemistry, processing, and health benefits. J. Med. Food 2004, 7, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Augustin, J.M.; Kuzina, V.; Andersen, S.B.; Bak, S. Molecular activities, biosynthesis, and evolution of triterpenoid saponins. Phytochemistry 2011, 72, 435–457. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Debnath, B.; Qasim, M.; Bamisile, B.S.; Islam, W.; Hameed, M.S.; Wang, L.; Qiu, D. Role of saponins in plant defense against specialist herbivores. Molecules 2019, 24, 2067. [Google Scholar] [CrossRef]
- Chaubey, M.K. Role of phytoecdysteroids in insect pest management: A Review. J. Agron. 2018, 17, 1–10. [Google Scholar] [CrossRef][Green Version]
- Dinan, L.; Savchenko, T.; Whiting, P. Phytoecdysteroids in the genus Asparagus (Asparagaceae). Phytochemistry 2001, 56, 569–576. [Google Scholar] [CrossRef]
- Bokov, D.O.; Yus, S.S.; Mazo, V.K.; Bessonov, V.V. Prospects for the use of spinach (Spinacia oleracea L.) containing phytoecdysteroids and polyphenols. Pharm. J. 2020, 12, 246–250. [Google Scholar] [CrossRef]
- Graf, B.L.; Poulev, A.; Kuhn, P.; Grace, M.H.; Lila, M.A.; Raskin, I. Quinoa seeds leach phytoecdysteroids and other compounds with anti-diabetic properties. Food Chem. 2012, 163, 178–185. [Google Scholar] [CrossRef]
- War, A.R.; Sharma, H.C.; Paulraj, M.G.; War, M.Y.; Ignacimuthu, S. Herbivore induced plant volatiles. Their role in plant defense for pest management. Plant Signal. Behav. 2011, 6, 1973–1978. [Google Scholar] [CrossRef] [PubMed]
- Turlings, T.C.J.; Erb, M. Tritrophic Interactions Mediated by Herbivore-Induced Plant Volatiles: Mechanisms, Ecological Relevance, and Application Potential. Annu. Rev. Entomol. 2018, 63, 433–452. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, C.; Lewis, W.; Paré, P.; Alborn, H.T.; Tumlinson, J.H. Herbivore-infested plants selectively attract parasitoids. Nature 1998, 393, 570–573. [Google Scholar] [CrossRef]
- Van Emden, H.F.; Sponagl, B.; Wagner, E.; Baker, T.; Ganguly, S.; Doulhoumpaka, S. Hopkins ‘host selection principle’, another nail in its coffin. Physiol. Entomol. 1996, 21, 325–328. [Google Scholar] [CrossRef]
- Turlings, T.C.J.; Tumlinson, J.H.; Lewis, W.J. Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 1990, 250, 1251–1253. [Google Scholar] [CrossRef] [PubMed]
- Rasmann, S.; Köllner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C.J. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434, 732–737. [Google Scholar] [CrossRef]
- Verat, N.; Robert, C.A.M.; Turlings, T.C.T.; Erb, M. Herbivore intoxication as a potential primary function of an inducible volatile plant signal. J. Ecol. 2015, 104, 591–600. [Google Scholar] [CrossRef]
- Bleeker, P.M.; Diergaarde, P.J.; Ament, K.; Guerra, J.; Weidner, M.; Schütz, S.; de Both, M.T.J.; Haring, M.A.; Schuurink, R.C. The role of specific tomato volatiles in tomato-whitefly interaction. Plant Physiol. 2009, 151, 925–935. [Google Scholar] [CrossRef]
- De Lange, E.S.; Laplanche, D.; Guo, H.; Xu, W.; Vlimant, M.; Erb, M.; Ton, J.; Turlings, T.C.J. Spodoptera frugiperda caterillars suppress herbivore-induced volatile emissions in maize. J. Chem. Ecol. 2020, 46, 344–360. [Google Scholar] [CrossRef]
- Hogenhout, S.A.; Bos, J.I.B. Effector proteins that modulate plant–insect interactions. Curr. Opin. Plant Biol. 2011, 14, 422–428. [Google Scholar] [CrossRef]
- Pentzold, S.; Zagrobelny, M.; Bjarnholt, N.; Kroymann, J.; Vogel, H.; Olsen, E.; Moller, B.L.; Bak, S. Metabolism, excretion and avoidance of cyanogenic glucosides in insects with different feeding specializations. Insect Biochem. Mol. Biol. 2015, 66, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, I.; Rohloff, J.; Bones, A.M. Defence mechanisms of Brassicaceae: Implications for plant–insect interactions and potential for integrated pest management. Agron. Sustain. Dev. 2010, 30, 311–348. [Google Scholar] [CrossRef]
- Ratzka, A.; Vogel, H.; Kliebenstein, D.J.; Mitchell-Olds, T.; Kroymann, J. Disarming the mustard oil bomb. Proc. Natl. Acad. Sci. USA 2002, 99, 11223–11228. [Google Scholar] [CrossRef] [PubMed]
- Falk, K.L.; Gershenzon, J. The desert locust, Schistocerca gregaria, detoxifies the glucosinolates of Schouwia purpurea by desulfation. J. Chem. Ecol. 2007, 33, 1542–1555. [Google Scholar] [CrossRef]
- Van Ohlen, M.; Herfurth, A.; Kerbstadt, H.; Wittstock, U. Cyanide detoxification in an insect herbivore: Molecular identification of β-cyanoalanine synthases from Pieris rapae. Insect Biochem. Mol. Biol. 2016, 70, 99–110. [Google Scholar] [CrossRef]
- Francis, F.; Lognay, G.; Wathelet, J.P.; Haubruge, E. Characterisation of aphid myrosinase and degradation studies of glucosinolates. Arch. Insect Biochem. Physiol. 2002, 50, 173–182. [Google Scholar] [CrossRef]
- Kazana, E.; Pope, T.W.; Tibbles, L.; Bridges, M.; Pickett, J.A.; Bones, A.M.; Powell, G.; Rossiter, J.T. The cabbage aphid: A walking mustard oil bomb. Proc. R. Soc. Ser. B 2007, 274, 2271–2277. [Google Scholar] [CrossRef]
- Pontoppidan, P.; Ekbom, B.; Eriksson, S.; Meijer, J. Purification and characterization of myrosinase from the cabbage aphid (Brevicoryne brassicae), a brassica herbivore. Eur. J. Biochem. 2001, 268, 1041–1048. [Google Scholar] [CrossRef]
- Nahrstedt, A. Cyanogenesis and the role of cyanogenic compounds in insects. Ciba Found. Symp. 1988, 140, 131–150. [Google Scholar] [CrossRef]
- Zagrobelny, M.; Bak, S.; Møller, B.L. Cyanogenesis in plants and arthropods. Phytochemistry 2008, 69, 1457–1468. [Google Scholar] [CrossRef]
- Zagrobelny, M.; Bak, S.; Ekstrøm, C.T.; Olsen, C.E.; Møller, B.L. The cyanogenic glucoside composition of Zygaena filipendulae (Lepidoptera: Zygaenidae) as effected by feeding on wild-type and transgenic lotus populations with variable cyanogenic glucoside profiles. Insect Biochem. Mol. Biol. 2007, 37, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Mateo, P.; Ye, M.; Zhang, X.; Berset, J.D.; Handrick, V.; Radisch, D.; Grabe, V.; Gershenzon, J.; Robert, C.A.M.; et al. Plant iron acquisition strategy exploited by an insect herbivore. Science 2018, 361, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.A.M.; Zhang, X.; Machado, R.A.R.; Schirmer, S.; Lori, M.; Matero, P.; Erb, M.; Gershenzon, J. Sequestration and activation of plant toxins protect the western corn rootworm from enemies at multiple trophic levels. Elife 2017, 6, e29307. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Doan, C.; Arce, C.C.M.; Hu, L.; Gruenig, S.; Parisod, C.; Hibbard, B.E.; Herve, M.R.; Nielson, C.; Robert, A.M.; et al. Plant defense resistance in natural enemies of a specialist insect herbivore. Proc. Natl. Acad. Sci. USA 2019, 116, 23174–23181. [Google Scholar] [CrossRef]
- Bleeker, P.M.; Mirabella, R.; Diergaarde, P.J.; VanDoorn, A.; Tissier, A.; Kant, M.R.; Prins, M.; de Vos, M.; Haring, M.A.; Schuurink, R.C. Improved herbivore resistance in cultivated tomato with the sesquiterpene biosynthetic pathway from a wild relative. Proc. Natl. Acad. Sci. USA 2012, 109, 20124–20129. [Google Scholar] [CrossRef]
- Degenhardt, J.; Hiltpold, I.; Kollner, T.G.; Frey, M.; Gierl, A.; Gershenzon, J.; Hibbard, B.E.; Ellersieck, M.R.; Turlings, T.C.J. Restoring a maize root signal that attracts insect-killing nematodes to control a major pest. Proc. Natl. Acad. Sci. USA 2009, 106, 13213–13218. [Google Scholar] [CrossRef]
- Tattersall, D.B.; Bak, S.; Jones, P.R.; Olsen, C.E.; Nielsen, J.K.; Hansen, M.L.; Hoj, P.B.; Lindberg Moller, B. Resistance to an herbivore through engineered cyanogenic glucoside synthesis. Science 2001, 293, 1826–1828. [Google Scholar] [CrossRef]
- Gnanasekaran, T.; Karcher, D.; Nielsen, A.Z.; Martens, H.J.; Ruf, S.; Kroop, X.; Olsen, C.E.; Motawie, M.S.; Pribil, M.; Lindberg Moller, B.; et al. Transfer of the cytochrome P450-dependent dhurrin pathway from Sorghum bicolor into Nicotiana tabacum chloroplasts for light-driven synthesis. J. Exp. Bot. 2016, 67, 2495–2506. [Google Scholar] [CrossRef]
- Franks, T.K.; Powell, K.S.; Choimes, S.; Marsh, E.; Iocco, P.; Sinclair, B.J.; Ford, C.M.; van Heeswijck, R. Consequences of transferring three sorghum genes for secondary metabolite (cyanogenic glucoside) biosynthesis to grapevine hairy roots. Transgenic Res. 2006, 15, 181–195. [Google Scholar] [CrossRef]
- Bruce, T.; Aradottir, G.; Smart, L.; Martin, J.L.; Caulfield, J.C.; Doherty, A.; Sparks, C.A.; Woodcock, C.M.; Birkett, M.A.; Napier, J.A.; et al. The first crop plant genetically engineered to release an insect pheromone for defence. Sci. Rep. 2015, 5, 11183. [Google Scholar] [CrossRef]
- Kunert, G.; Reinhold, C.; Gershenzon, J. Constitutive emission of the aphid alarm pheromone, (E)-β-farnesene, from plants does not serve as a direct defense against aphids. BMC Ecol. 2010, 10, 23. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).