Abstract
This work aimed to study the effects of compost (applied at two rates) and two commercial microbial biostimulants on the mobility and bioavailability of potentially toxic elements (PTEs) in an industrial soil phytostabilized by Dactylis glomerata L. or a mixed stand of grasses (Lolium perenne L., Poa pratensis L. and Festuca arundinacea Shreb.). The soil showed very high pseudototal and bioavailable concentrations of cadmium (Cd) and lead (Pb), due to improper lead-acid batteries storage. Compost amendment in combination with the two biostimulants produced the best outcomes in terms of plant growth and nutrient uptake. The same mix of beneficial microbes improved soil biological fertility enhancing soil nitrogen fixing and ammonia oxidizing bacteria, while reduced the pore water and NH4NO3 extractable concentrations of Cd and at lower extent of Pb in soil. Accordingly, the lower mobility and bioavailability of Cd in soil determined a lower uptake and accumulation of Cd in shoots of different grass species. Our results suggest that a green cap with turfgrass assisted by biostimulants and compost amendment in PTE-contaminated industrial sites could be a reliable and effective practice to protect and restore soil biological fertility and to reduce the risk of PTE dispersion in the surrounding environment.
1. Introduction
An increase in anthropogenic activities like mining, industrial activities, use of chemicals in agriculture and increasing urbanization has led to soil contamination by potentially toxic elements (PTEs) worldwide [1]. The recycling of lead-acid battery is an industry activity that allows to recover lead (Pb) and plastic from aged batteries of motor vehicles thus reducing the environmental impact deriving from Pb extraction from mines [2]. Approximately 50% of the lead consumed worldwide is derived from recycled and reused materials [3]. However, an improper recycling process or disposal may lead to environmental pollution due to Pb, cadmium (Cd Battery doping agent) and sulphuric acid solution [4,5,6]. Lead is the second among the most hazardous metals in the Priority List of the US Environmental Protection Agency [7]. Lead may affect every organ in the human body causing brain damage, mental retardation, encephalopathy, kidney and cardiovascular damage particularly in children [8,9,10,11]. Likewise, human exposure to Cd compounds may create health problems, such as damage to liver and kidneys, cardiovascular disorders and bone degradation [12].
Abiotic stress as environmental pollution could also affect bacterial populations involved in the N-cycling, altering their activity that is essential for the functioning of soil ecosystem [13,14,15].
Soil processes like biological nitrogen fixation (the reduction of atmospheric N2 to bioavailable NH4+) and biological nitrification (the oxidation of NH4+ to NO3−) are very important in N cycling to provide natural N input to plants [16]. Particular N-cycling genes involved in N-transformations such as N-fixation (nifH) and nitrification (amoA) have been extensively used to assess the soil microbial communities’ abundance and diversity [17,18]. These microbial functional groups are very sensitive to any stress and environmental change and therefore they could be considered sensitive indicators of soil heath [19].
Phytoremediation is recognized as a cheap and eco-friendly technique to remediate PTE polluted sites by accumulating soluble PTEs in easily harvestable plant organs (phytoextraction) or by stabilizing them in the rhizosphere and plant roots (phytostabilization) [20].
However, phytoextraction in lead-acid battery recycling sites has little applicability because it requires many years due to very high PTE concentrations in soil. In addition, the use of synthetic chelates proposed by some authors to enhance Pb availability to plants may dramatically increase the risk of Pb mobilization towards subsoil and groundwater [2,21].
Oppositely, aided phytostabilization is based on the use of various mineral and/or organic fertilizers and plant species adequately chosen to stabilize PTEs in the rhizosphere by root exudates or through the accumulation into root tissues [22,23].
Native plant species are preferred for phytostabilization due to their adaptation to the specific climatic conditions, soil physical and chemical features and soil PTE concentrations [24]. Furthermore, plants should ensure good soil coverage in a short time and they should limit PTE accumulation in their aboveground portions to minimize the risk of contaminant transfer to the surrounding environment [25].
Grass plants from Poaceae family such as Dactylis glomerata L., Lolium perenne L., Festuca arundinacea Shreb. and Poa pratensis L. showed a good adaptability to soil contamination and good phytostabilization capacity [6,26,27,28,29].
Dactylis glomerata L. is a perennial grass with erect or spreading culms. Leaves present entire sheaths, large ligules serrate at the apex. Blades are usually glacous, broad, folded in the shoot and strongly keeled. The plant forms erect panicles with branches close together or the lowermost distant. Spikelets have 2–5 flowers, almost sessile and arranged in dense, one-sided, ovate masses. The growth habit of D. glomerata L. may be prostrate or erect. D. glomerata L. has an extensive fibrous root system and lower nodes of the stem may form tillers and set up clonal individuals a short distance away from the parent plant [30].
Festuca arundinacea Shreb. is a perennial grass that produces large and loose tussocks. Culms are erect, unbranched and smooth. Leaves of F. arundinacea Shreb. are dark-green, usually flat, and scabrous on margin. The plant form erect or pendant panicles lanceolate to ovate. Spikelets are elliptic to oblong with 3–10 florets. F. arundinacea Shreb. is very drought resistant thanks to its dense fibrous rootstock that can form an extensive and depth root system [31].
Poa pratensis L. is a perennial plant with fibrous roots tufted or with isolated shoots. It has erected or decumbent culms and leaf sheaths moderately compressed and keeled. Leaf blades are flat or folded with smooth or sparsely scabrid surface. It produces panicles erect or slightly lax loosely, oblong to broadly pyramidal. Spikelets are ovate (green or grayish) with 2–5 florets [32].
Lolium perenne L. is a caespitose perennial plant with fibrous glabrous roots. It has erect ascending or spreading culms It has leaf sheaths somewhat flattened and not keeled. L. perenne L. leaves are glossy on underside while are dull above with flat ribs. The plant spikelets are sessile and basally in one plane and recessed into rachis each with eight florets alternating. L. perenne L. growth-form ranged from erect individuals to prostrate compact cushions [33].
Many organic amendments (e.g., compost, sewage sludge, stabilized biosolids, biochar or animal manure) were proposed for reducing PTE mobility in soil due to chelation, adsorption, binding or co-precipitation processes [34,35,36,37]. Compost is the product of the humification and microbiological stabilization of organic materials [38]. It may improve soil fertility and the activity of soil microbial communities, enhance plant growth and contribute to the immobilization of PTEs in the soil [39,40,41].
It is also suggested that beneficial soil microorganisms such as Trichoderma or mycorrhiza may assist phytoremediation of PTE polluted soils [42]. Trichoderma may increase plant growth and stimulate resistance to biotic and abiotic stresses also improving phytostabilization of polluted soil through mechanisms such as PTE reduction, oxidation, compartmentalization, and conversion to less toxic forms [43,44].
Mycorrhizae, such as arbuscular mycorrhizal fungi (AMF) could establish mutual symbiosis with a great number of plants providing nutrients to the host and ameliorating soil structure [45]. Mycorrhizae are able to develop a network of external hyphae that may extend the root surface area up to 40 times. This symbiosis allows plants to explore a great soil volume for nutrient uptake through the production of enzymes and excretions of organic substances [46]. Mycorrhizae may also increase plant tolerance to PTEs through immobilization, precipitation, adsorption and chelation [47].
Therefore, the aims of this study were: (1) to assess the influence of biostimulants and compost amendment on the phytostabilization potential of two grass swards including D. glomerata L. (native plant species of the site) and of a mixed stand of grasses in a highly contaminated soil by PTE from disposed lead-acid batteries; and (2) to evaluate the effect of the phytostabilization process on the rhizospheric N cycling bacteria and so on the biological status and quality of the soil. The comparison between the different plant-compost-biostimulant combinations, will allow us to identify the most appropriate site-specific phytostabilization strategy.
2. Materials and Methods
2.1. Study Area and Amendment Description
The soil used in this study was collected from an industrial site located in Marcianise (southern Italy—41°00′48.9” N—14°17′49.7” E) used as a temporary landfill of batteries for several years by a lead-acid battery recycling plant (Figure 1a). According to Italian legislation [48], Cd and Pb concentrations in soil were above the screening values for industrial soils (Cd: 15 mg kg−1; Pb: 1000 mg kg−1) thus the company made a risk analysis to evaluate the risks for human health and environment due to PTE pollution. Results of risk analysis showed that there was a risk of leaching of Cd and Pb in the groundwater in addition to a risk for workers linked to the inhalation and dermal contact with contaminated soil particles. A phytoremediation project was approved, using the ECOREMED protocol [20] based on ecological systems (i.e., a poplar stand with permanent meadows) to reduce PTE mobility and to reduce soil resuspension thus interrupting exposure pathways to contaminants.

Figure 1.
Map of the studied site and sampling point (a) in the industrial area of Marcianise (Campania, southern Italy) and growth chamber pot experiment with D. glomerata L. and a mixed stand of F. arundinacea Shreb., L. perenne L. and P. pratensis L (b).
Soil used for the pot experiment was collected in very high polluted area of the site (depth: 0–30 cm) (Figure 1a) and carefully homogenized before the preparation of experimental units.
The preliminary soil characterization is reported in Table 1 and can be resumed as follows: sandy loam textured and neutral soil, with medium-high contents of organic matter and total nitrogen and a moderate accumulation of soluble salts.

Table 1.
Physiochemical properties of the composite soil sample used in the pot experiment before plant sowing (number of replicates = 3).
The readily bioavailable fractions of Cd and Pb in the soil were respectively 24- and 330-fold higher than the trigger values settled by Germany and Slovak Republic [49,50,51], revealing a very high PTE mobility in soil and a high environmental risk.
The compost used in the pot experiment was well humified and stabilized, rich in nutrients (total N: 4.8%) and organic matter (58%), with a sub-alkaline pH (7.9) and medium-low electrical conductivity (4.5 mS cm−1).
2.2. Experimental Design, Compost and Biostimulants Application
Two compost doses were mixed individually with soil at 0.7% w/w (C1) and at 1.4% w/w (C2), thoroughly homogenized and transferred to pots (Ø 15 cm; V 1.5 L). The plant microbial applications, hereby noted as biostimulant BP (Panoramix commercial product; Koppert Biological Systems, Rotterdam, the Netherlands), consisted of a microbial consortium of arbuscular mycorrhizal fungi (AMF), plant growth-promoting rhizobacteria (PGPR) (Bacillus spp.), and Trichoderma; whereas BT (the commercial biostimulant Trianum-P; Koppert Biological Systems, Rotterdam, the Netherlands) contained Trichoderma harzianum strain T22. The products were applied as a coating to the surface-sterilized seed of the respective grass formulations (final concentration ca. 1 × 108 spores ml−1), using a proper volume of the product to cover the seed surface. Treated seed was left to air-dry, with constantly stirred in order to coat the surface uniformly, then stored at 4 °C until sowing. Treated seeds of Dactylis glomerata L. (98% pure live seed; 8 g seed m−2), an autochthonous species for the zone, or a commercial mix of grasses (98% pure live seed; 40 g seed m−2) including Lolium perenne L. cv. Keystone (10% w/w ) Poa pratensis L. cv. SR2100 (10% w/w) and Festuca arundinacea Shreb. (cv. SR8600 50% w/w; cv. Durango 30% w/w) were sown in individual pots (three replicates per treatment) (Figure 1b) for a total of 54 experimental units (27 experimental units for each grass sward). Pots with bare soil were used as unplanted controls (in triplicate) and kept under the same conditions of the other treatments throughout the experiment. The experiment was carried out in a growth chamber (day/night cycle = 12/12 h; T from 22 to 26 °C; relative humidity = 85%), where the pots were arranged in a completely randomized design. During the experimental period (60 days), pots were watered regularly with deionised water to keep soil moisture close to field capacity.
The following factors were tested for Dactylis glomerata L. and commercial mix of grasses:
- (i)
- 3 doses of compost from urban wastes: C0 = non-fertilized control vs. C1 = 0.7% vs. C2 = 1.4%;
- (ii)
- 3 levels of commercial biostimulants: a non-inoculated control (B0) vs. BP vs. BT.
2.3. Plant Sampling and Analysis
At the end of the experiment plant shoots were harvested, thoroughly washed with tap water and then deionized water, gently blotted, weighed (fresh weight), oven dried at 60 °C until constant dry weight to later finely-ground in an electric mill. Plant samples were analysed for N concentration (Kjeldahl method [52]) and PTE concentrations by inductively coupled plasma mass spectrometry (ICP-MS, Nexion 3000 – Perkin Elmer, USA) after 2 sequential digestions in microwave oven: firstly by concentrated HNO3 and secondly by aqua regia (HCl/HNO3, 3:1 v:v).
PTE concentrations in the two grass swards were compared to PTE thresholds (converted from concentration at 12% moisture to dry weight basis) indicated in European Union Regulation No. 1275 [53] for forages (Cd: 1.14 mg kg−1; Pb: 34.09 mg kg−1) for evaluating the PTE transfer from soil to plant. In addition, PTE “toxic ranges” in shoots (Cd: 5–30 mg kg−1; Pb: 30–300 mg kg−1) reported by Kabata-Pendias [54] were used as reference.
2.4. Soil Sampling and Analysis
At the end of the experiment, the soil of the pots was separated from the roots, oven dried at 50 °C until constant weight, homogenized and sieved at 2 mm before analyses. The following physicochemical properties were determined on soil samples (before and after plant cultivation): pH-H2O (1:2.5 soil:water solution ratio – pH meter GLP 22, Crison, Barcelona, Spain), electrical conductivity (1:5 soil:water solution ratio—Conductimeter basic 30, Crison, Barcelona, Spain), ammonium and nitrate content (Foss FIAstar 5000 continuous flow Analyzer—Foss Analytical A/S, Hillerød, Denmark), extraction of readily bioavailable fraction of PTEs by 1M NH4NO3 (1:2.5 soil:water solution ratio; [55]).
In the composite soil sample (collected before plant cultivation) they were also determined: texture (pipette and sieving method, after pre-treatment with H2O2 to oxidize organic matter and particle dispersion by sodium hexametaphosphate), total nitrogen (Kjeldahl method [52]), organic carbon [56] and pseudototal content of PTEs by aqua regia digestion [57] and ICP-MS at Acme Analytical Laboratories Ltd. (Vancouver, Canada). Certified reference material CC141 and 141R (European Reference Materials - ERM®) and lab standard materials (STD DS10 and STD OREAS45EA) were used to monitor the data quality of soil analyses, with metal recoveries around ±10% of the certified values.
Water soluble PTE concentrations were assessed by analysing soil pore water that was extracted from the pots by using rhizon-samplers (Rhizosphere Research Products, Wageningen, the Netherlands) inserted with a 45° angle in each pot. Ten ml of water were extracted from each pot just before plant harvest by applying a suction from a vacuum syringe for 24 h. Soil pore water was then analysed for pH and PTE concentrations by atomic absorption spectrometer (AAS).
2.5. Quantification of nifH and amoA Genes
The soil collected from each experimental pot as above reported were used to obtain rhizosphere samples as described by Romano et al. [58]. Total DNA from rhizosphere samples was extracted by the FastDNA Spin Kit for Soil (MP Biomedicals, Illkirch Cedex, France) according to the supplier’s recommendations. qPCRs for the bacterial functional genes nifH or amoA were performed using 50 ng of DNA template and the oligonucleotide primers nifH-F (5′-AAAGGYGGWATCGGYAARTCCACCAC-3′) and nifH-R (5′-TTGTTSGCSGCRTACATSGCCATCAT-3′) [59] or amoA-1F (5′-GGGGTTTCTACTGGTGGT-3′) and amoA-2R (5′-CCCCTCKGS AAAGCCTTCTTC-3′) [60], respectively, using a Chrom4 System Thermocycler (Bio-Rad, Milan, Italy). The PCR mixture included 50 ng of target DNA, 1× iQ SYBR Green Supermix, (Bio-Rad), and 0.2 μM of each primer. All qPCR reactions included an initial denaturing step at 95 °C for 3 min followed by 40 cycles of amplification as previously reported [61]. To calculate the abundance nifH and amoA genes, standard curves were generated by amplifying and cloning the target genes from the strains Sinorhizobium meliloti 1021 and Nitrosomonas europaea, respectively, as previously reported [61].
2.6. Statistical Analysis
Data on plants were analysed by two-way ANOVA using a general linear model and means were separated by using a least significant difference (LSD) test with p < 0.05 using the SPSS 21.0 statistical software package (SPSS Inc., Cary, NC, USA). The same statistical design was carried out for soil properties to test the main effect of tested factors and their interaction. In addition, a one-way ANOVA was carried out to compare the unplanted control to the planted pots. In this case, each compost × biostimulant combination was considered as a single treatment. Normality of distribution and homogeneity of variance were verified using the Kolmogorov–Smirnov and Levene tests, respectively. Logarithmic transformation was applied, when needed, to variables that did not accomplish normality. Pearson correlation analysis was carried out between soil and plant features by XLStat (Addinsoft, Paris, France) to evaluate general tendencies.
3. Results and Discussion
3.1. Soil Mineral Nitrogen and PTE Promptly Bioavailable Concentrations
Soil pH, mineral nitrogen and NH4NO3-extractable PTE concentrations in soil are shown in Table 2.

Table 2.
Nitrogen concentrations (NH4-N and NO3-N) and NH4NO3-extractable Cd and Pb concentrations in soil at the end of the experiment. C0 = no compost, C1 = 0.7%, C2 = 1.4%; B0 = no biostimulants, BP = Panoramix, BT = Trianum-P; Unplanted control = non-planted and untreated soil. Mean values within columns and factor with the same letter do not differ according to the LSD test (p < 0.05). * p < 0.05; ** p < 0.01; n.s., not significant.
Compost and biostimulants did not significantly affected soil pH that ranged respectively from 6.84 to 7.06 for D. glomerata L. and from 6.81 to 7.07 for the mixed stand of grasses (data not shown), similarly to the unplanted control (Table 2). The interaction of compost and biostimulants with both plant treatments showed no significant effect on soil NO3-N concentration in comparison with the control (C0-B0). Nevertheless, there was a significant reduction of soil NO3-N concentration in planted soils, if compared to unplanted control (Table 2), and this was probably due to the plant uptake of the most available form of N during the experiment [62,63].
On the contrary, compost showed a significant increase of NH4-N in soil of both the grass swards in comparison to the control (Table 2): in particular, compost application produced an increase of 34% (C1-B0) and 42% (C2-B0) in D. glomerata L. pots than control (C0-B0), and a similar behaviour was observed in the mixed stand of grasses (the higher rate of compost (C2-B0) enhanced of 44% the NH4-N concentration in soil). Compost addition may improve the soil microbial activity due to the large supply of organic carbon [64]. Accordingly, a high mineralization rate of composted organic matter triggered by soil microflora may lead to a high NH4-N content in soil, as found in C1-B0 and C2-B0 treatments [65,66]. Differently, the application of compost (C1–B0; C2-B0) showed a limited effect on NH4NO3-extractable concentrations of Cd and Pb in both plant treatments (Table 2).
According to the very high pseudototal concentrations of Cd and Pb in the studied soil, the NH4NO3-extractable concentrations of Cd and Pb for both the grass swards were above the trigger values (0.1 mg kg−1 for Cd and 0.1 mg kg−1 for Pb) settled by some European countries for contaminated soil in order to identify potential risks for the environment and plant growth [49,50,51,67,68] (Table 2).
The application of the biostimulant Panoramix (C0-BP) determined a significant reduction (−37%) of readily bioavailable Pb in soil of D. glomerata L. pots as compared with the control (C0-B0), probably due to a possible immobilization of PTEs on AMF hyphae (Table 2). Mycorrhizae may mitigate the mobility of PTEs in soil through different mechanisms, such as metal chelation by fungal activity of extra-radical mycelia, spores, intra-radical mycelia and vesicles, or through AMF secretions (polyphosphoric acid and organic acids) that can complex the PTEs at the fungus-root interface [69,70,71,72]. In addition, mycorrhizal mycelia can produce a glycoprotein called glomalin that has shown a potential role in PTEs immobilization [73]. Similarly, Trichoderma harzianum reported an immobilization effect on soil Cd and Pb through the bioaccumulation of PTEs in the fungal biomass [74,75,76].
A synergistic effect of the two compost doses and microbial biostimulants on soil NH4-N content was recorded for D. glomerata L. in BP (C1-BP and C2-BP) and BT (C2-BT and C2-BT) as compared respectively with C1-B0 and C2-B0, exerting the highest effect with the application of the highest compost dose and with both biostimulants (C2-BP and C2-BT) (Table 2). On the contrary, no synergic effect was found for the mixed stand of grasses when the highest compost rate was applied (C2-BP and C2-BT) in comparison to C2-B0, revealing a lower effect of biostimulant for the mixed stand of grasses. However, only C1-BP was effective in increasing NH4-N concentration in soil as compared with C1-B0, C1-BT and C0-B0 (Table 2).
The synergic effect showed for BP is probably due to the stimulating effect of compost on both hyphal proliferation and spore formation of AMF contained in the biostimulants BP that resulted in an increase of NH4-N content in soil [77,78]. This stimulation may lead to the release of nutrients (particularly of N-NH4) from the soil amendment through mineralization or modification of soil microbial community [79,80]. Similarly, BT exploiting nutrients provided by organic amendment may release several plant nutrients by promoting root exudation through the production of an auxin-like phytohormone. Root exudates, such as sugars and organic acids, may enhance the abundance and the interactions of soil microflora, which have a potential effect on soil nutrient cycling [81,82,83]. In all plant treatments, a higher NH4-N concentration in soil than the unplanted control was found, since the release of exudates can intensely modify soil microbial communities and influence N transformations in and near the rhizosphere [84].
The combined application of compost and biostimulants (C1-BP, C1-BT, C2-BP and C2-BT) reduced the readily bioavailable concentration of Cd in soil in comparison to the control (C0-B0) in D. glomerata L. pots, while only the combination of compost and biostimulant Panoramix (C1-BP and C2-BP) showed the same behaviour for the mixed stand of grasses (Table 2). Similarly, the combination of the high compost rate and biostimulants (C2-BP and C2-BT) reported a reduction of NH4NO3-extractable concentration of Pb in soil where D. glomerata L. grew (Table 2). Compost addition may decrease the PTE extractability through processes linked to amendment-PTEs interaction, such as adsorption, complexation, precipitation and redox reactions [85]. The mitigation of PTE toxicity by organic amendments may in turn improve the mycorrhizal abundance and root colonization by beneficial fungi [71,86,87]. In particular, organic amendments can aid AMF through the modification of soil physiochemical properties facilitating AMF growth, encouraging microorganisms that interact with AM fungi (e.g., P-solubilizing bacteria). Furthermore, organic amendments may modify plant-fungi signalling compounds that affect spore germination and mycorrhiza hyphal branching [73]. Compost can act as a substrate for microbial growth thus it can stimulate Trichoderma colonization as well. In addition, Trichoderma can provide major contribution to the decomposition of soil organic matter contained in the compost increasing its content in soil and consequently the PTE immobilization in soil [76,88].
All treatments with plants showed a lower readily bioavailable Cd in soil than unplanted control (Table 2), which reveals a strong contribution of rhizosphere processes to Cd immobilization through the interaction of soil microflora and root chelating substances (e.g., phytosiderophores) [89]. Differently, Pb NH4NO3-extractable concentration of the control (C0-B0) showed no significant differences as compared with unplanted control.
The compost addition slightly raised the pH of pore water (average effect) in D. glomerata L. (C1 = 7.61; C2 = 7.44) as compared to the control (C0 = 7.40) (data not shown). The same behaviour was reported for the mixed stand of grasses (C1 = 7.47; C2 = 7.55) as compared to the control (C0 = 7.37) (data not shown). This behaviour was also observed by Hartley et al. [90] in sub-acid or neutral soils. This increase of pH may be due to the consumption of protons during the decomposition of the organic amendment (decarboxylation of organic acid), to the proton consumption by functional groups (associated to the organic material) and to the release of OH during the specific adsorption of organic molecules on surface minerals by ligand exchange [91].
Cadmium and Pb concentrations in pore water ranged from 0.10 to 0.20 mg L−1 and from 0.15 to 1.21 mg L−1, respectively, in D. glomerata L. pots, while ranged from 0.01 to 0.20 mg L−1 and from 0.01 to 1.56 mg L−1 in the pots vegetated with the mixed stand of grasses (data not shown). Both PTE concentrations were very high if compared to similar studies in contaminated soils, indicating a high environmental risk associated with these two PTEs [92,93].
The compost and biostimulants application showed no significant effects on the Pb concentration in pore water for both the grass swards while there was a significant effect (interaction compost × biostimulants) (p < 0.05) on the Cd concentration in pore water for both the grass swards (Figure 2a,b).
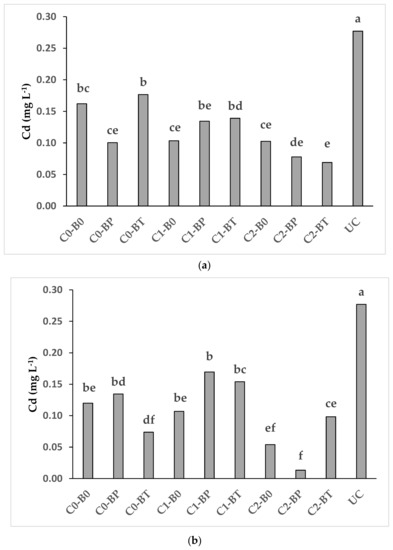
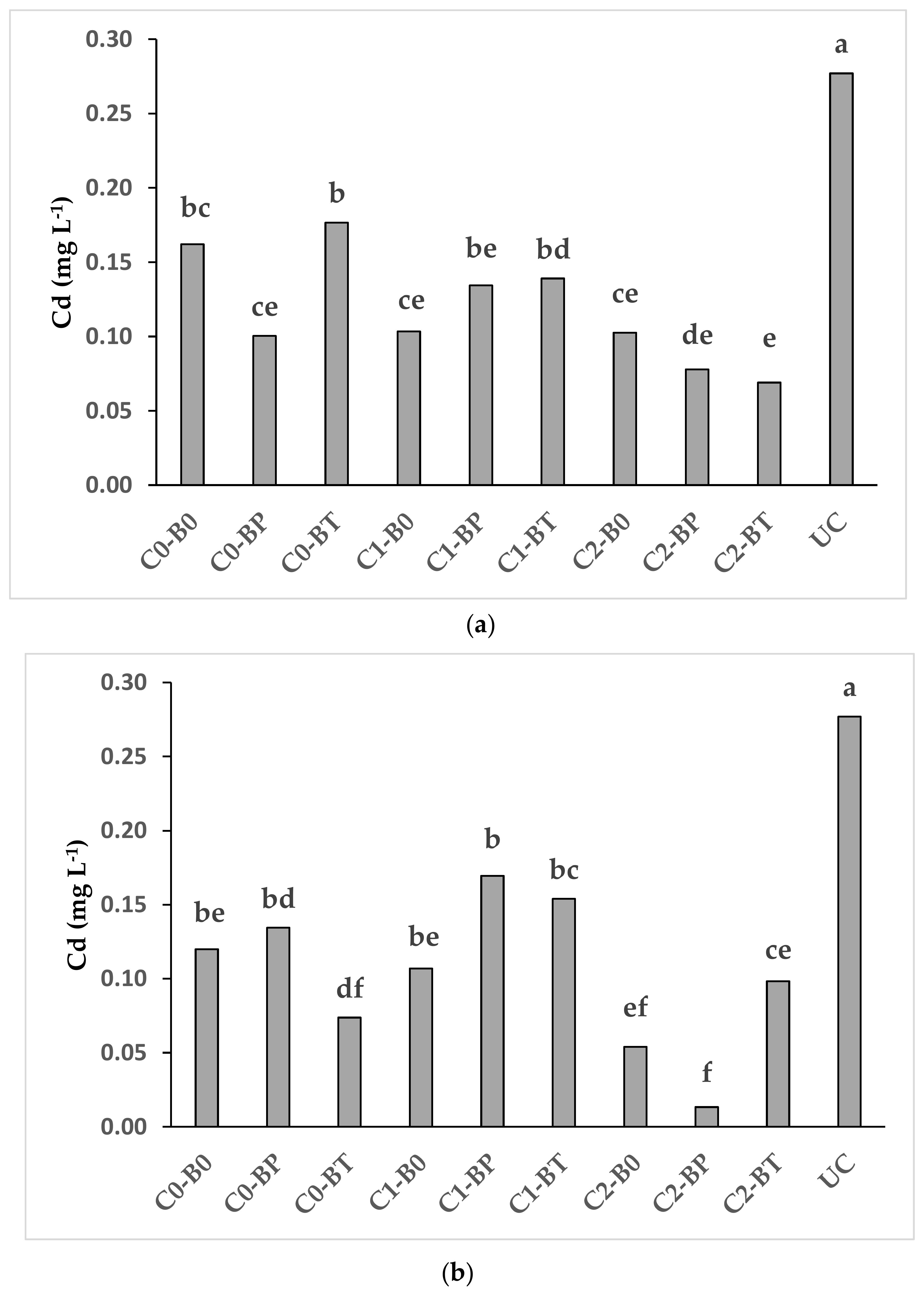
Figure 2.
Interaction (compost × biostimulants) effects on Cd soluble concentrations in D. glomerata L. (a) and in a mixed stand of F. arundinacea Shreb., L. perenne L. and P. pratensis L. (b) C0 = no compost, C1 = 0.7%, C2 = 1.4%; B0 = no biostimulants, BP = Panoramix, BT = Trianum-P; UC = non-planted and untreated soil. Bars (means) marked with the same letter do not differ significantly according to the LSD test (p < 0.05).
The combination of the high compost dose with the two biostimulants (C2-BP and C2-BT) reduced the Cd concentration in pore water of D. glomerata L. as compared with the control (C0-B0) (Figure 2a). A similar decrease of Cd concentration in pore water was recorded for the mixed stand of grasses for C2-BP in comparison to the control (C0-B0) (Figure 2b). The low Cd concentration in pore water was likely related to the increase of pH and organic matter in soil and to mycorrhizae and Trichoderma. Increased pH may modify the charge of amphoteric metal oxide materials and solid-phase organic matter becoming negative facilitates the adsorption of Cd on the surfaces or enhances surface and bulk precipitation of PTEs as sparingly soluble metal hydroxides [94,95]. Likewise, the increase of the organic matter in soil may reduce the concentrations of water-soluble Cd by increasing the availability of binding sites in soil [96]. In addition, mycorrhizae and Trichoderma may immobilize Cd in soil through the release of chelating agents as glomalina by mycorrhizae or the bioaccumulation of Cd in Trichoderma fungal biomass [73,76].
Similarly to the NH4NO3-extractable concentration of Cd (Table 2), also the pore water concentration of Cd in all the planted treatments (including control) was lower than unplanted control (Figure 2a,b). This indicate once again the important role of plant roots in the mitigation of mobility of PTEs in the soil [89].
Overall, the results on soil properties showed that compost and biostimulants were mostly effective on soil mineral N content and PTE bioavailability (NH4NO3 and pore water) when applied in combination, thanks to the stimulatory effect of compost on mycorrhizae and Trichoderma, and mitigation of PTE toxic effects.
3.2. Bacterial Functional Genes Analysis
To evaluate the influence of the different treatments (plant species, compost and biostimulants) on the N-cycling microbial populations, the abundance of specific functional genes encoding N2-fixing (nifH) and nitrifying (amoA) enzymes was estimated by qPCR (Table 3).

Table 3.
Quantification of nifH gene and amoA gene abundance in the rhizosphere of D. glomerata L. and in a mixed stand of F. arundinacea Shreb., L. perenne L. and P. pratensis L. C0 = no compost, C1 = 0.7%, C2 = 1.4%; B0 = no biostimulants, BP = Panoramix, BT = Trianum-P; Unplanted control = non-planted and untreated soil. Mean values within columns and factor with the same letter do not differ according to the LSD test (p < 0.05). * p < 0.05; ** p < 0.01; n.s., not significant.
The abundance of nifH gene seems to be affected by both biostimulants and compost application also showing an interaction between them in the rhizosphere of both the grass swards. As shown in Table 3, the application of the high compost dose (C2-B0) increased the abundance of nifH gene for both the grass swards (Table 3) as compared with control (C0-B0), probably due to contribution of compost to new microbial populations in soil [97]. A similar effect on nifH gene was recorded after the biostimulant Panoramix application (C0-BP) which increased the abundance of nifH gene by 27% for D. glomerata L. and by 34% for the mixed stand of grasses as compared to the control (C0-B0). This positive result was probably related to the improvement of the nitrogen-fixing bacteria activity by mycorrhizae that may facilitate their colonization of plant roots or may improve nutritional status of the host plant, which in turn would result in more energy available for nitrogen fixation by bacteria [98]. Diazotrophic populations were also influenced according to quantity of compost applied and biostimulant application. In fact, both biostimulants significantly increased the abundance of nifH gene in both D. glomerata L. (ranging from 13 × 104 to 71 × 104 copies g−1) and mixed stand of grasses (ranging from 13 × 104 to 73 × 104 copies g−1) rhizosphere when the highest compost dose was applied to the soil (C2-BP and C2-BT treatment; Table 3). Indeed, high values (ranging from 7.0 × 104 to 13 × 104 copies g−1) were also detected in C1-BP and C1-BT treatments in both D. glomerata L. and mixed stand of grasses rhizosphere samples (Table 3).
This result indicated that the marked increase of NH4-N in soil after compost addition (Table 2) may be attributed also to the increase of nitrogen fixing bacteria which add additional nitrogen to the soil [99].
However, the abundance of the nifH gene in all planted pots was 1 or 2 orders of magnitude higher than in the unplanted control (6.6 × 103 copies g−1; Table 3). This increase was probably due to a phenomenon known as the “rhizosphere effect” in which root-derived exudates directly affect easily degradable substances leading to proliferation of microorganisms in the rhizosphere [100,101]. This finding was in accordance with Nelson et al. [102] which reported an increase in nifH gene abundance in the rhizosphere during a phytostabilization experiment, demonstrating its suitable effectiveness to evaluate ecosystem potential and plant efficiency.
Similarly, the interaction between compost amendment and biostimulant application exerted the same effect on ammonia oxidizing bacteria in the rhizosphere of both the grass swards (Table 3). In fact, in the rhizosphere of both D. glomerata L. and mixed stand of grasses the highest abundance of amoA gene was detected in the combined C2-BP treatment (ranging from 666 x 102 to 678 × 102 copies g−1; Table 3), followed by C1-BP and C2-BT treatments in which ammonia oxidizing bacteria significantly increased approximately up to 9.2–9.3 × 103 copies g−1 compared to C1-TB (5.2–5.3 × 103 copies g−1) or C0 or B0 pots in which no treatment or a single treatment was applied (about 6 × 102 copies g−1) (Table 3). Finally, also the abundance of the amoA gene was lower in the unplanted control (1.8 × 102 copies g−1) compared to planted pots, although this difference was not significant (Table 3).
Overall results highlighted that there is a correlation between compost addition and biostimulant application, and that combined treatments significantly increased the functional genes involved in the nitrogen fixation and nitrification probably due to a decrease of NH4NO3-extractable concentrations of Cd in soil (Table 2). In addition, the highest NH4-N concentration in soil due to the combination of compost and both biostimulants (Table 2) may have contributed to high ammonia-oxidizing bacteria activity and consequently to high abundance of the amoA gene in soil [103]. This finding indicates a faster N turnover and higher N availability to plants in soils under combined treatments management, likely due to the higher release of root exudates [104]. Indeed, as previously demonstrated compost addition could promote the restoration of the biological fertility of multi-contaminated soils [97], since it could be a source of new microbial populations and improve structural stability of soil [105]. However, compost addition exerted a positive effect only when in combination with biostimulant application that could exert an evident effect on autochthonous microbial populations in the soils [101]. In particular, N2-fixing and ammonia oxidizing bacteria were strongly stimulated especially by BP compared to BT, in both the grass swards. This result could be due to the fact that unlike BT, that is a pure culture of the strain T. harzianum T22, BP consists of a combination of a mix of eukaryotic and prokaryotic microorganisms (mycorrhizae, Bacillus, and Trichoderma species) with different plant growth promoting activities that could have a synergistic interaction.
3.3. Plants Biomass, Nutrient Status and PTE Concentrations
Plant growth and nutritional status of D. glomerata L. and mixed stand of grasses are shown in Table 4, along with Cd and Pb concentrations in plant shoots.

Table 4.
Plant biomass (DW), nutrient status (N content) and PTE concentrations in shoots of D. glomerata L. and in a mixed stand of F. arundinacea Shreb., L. perenne L. and P. pratensis L. C0 = no compost, C1 = 0.7%, C2 = 1.4%; B0 = no biostimulants, BP = Panoramix, BT = Trianum-P. Mean values within columns and factor with the same letter do not differ according to the LSD test (p < 0.05); * p < 0.05; ** p < 0.01; n.s., not significant.
The application of the biostimulant BP (C0-BP) resulted in a significant increase of the mixed stand of grasses biomass and a significant increase of nitrogen uptake for both the grass swards as compared with the control (C0-B0) and BT application (C0-BT) (Table 4). This effect may be related to the improvement of plant rooting zone and nutrient uptake due to mycorrhizae infection [106]. Furthermore, the combination of PGPR (Bacillus spp.) and T. harzianum may also have a synergic effect with mycorrhizal fungi resulting in a greater promotion of plant growth, increased production of enzymes, antioxidants, nutrient solubilisation and root nodulation [107,108]. This positive effect of AMF on the growth of Lolium spp., Poa spp. and Festuca spp. cultivated alone or in combination was consistent with previous works and is associated to the improvement of plant root surface area and higher nutrient uptake [109,110].
On the contrary, BT application (C0-BT) showed no significant effect on the mixed stand of grasses growth while significantly reduced the growth of D. glomerata L. (−57%) if compared to the control (C0-B0) (Table 4). This negative effect may be related both to the competition of Trichoderma with the plant and other soil microorganism for nutrients or to a low affinity of the strain T22 with D. glomerata L. [88,107,111]. Similarly, the application of both compost doses (C1-B0 and C2-B0) reduced significantly the biomass of D. glomerata L. and a non-significant reduction was reported for the mixed stand of grasses as compared to C0-B0 (Table 4). This trend for both the grass swards suggested a nitrogen immobilization by the compost [38,112].
On the contrary, the combination of both compost doses with BP (C1-BP and C2-BP) increased the growth and N uptake for D. glomerata L. and for the mixed stand of grasses as compared to C0-B0 (Table 4). This behaviour was probably related to the provision of humic acids by compost, which may stimulate mycorrhiza hyphal growth and sporulation which in turn can improve nutrient uptake and plant growth [113,114].
Nevertheless, also the combination of both compost doses with BT (C1-BT and C2-BT) showed an improvement of D. glomerata L. biomass and N uptake than control (C0-B0), probably due to the supply of nutrients by compost for Trichoderma growth, thus reducing its competition with plants that in turn may stimulate plant root development and contribute to solubilize mineral nutrients (Table 4) [88,107].
The concentrations of Cd and Pb in the aboveground parts of D. glomerata L. and mixed stand of grasses were above the threshold for forage [53] and above the value reported by Kabata-Pendias [54] in plants grown in non-contaminated sites, suggesting a potential risk of transfer of the pollutants into the food chain (Table 4) [24]. In addition, Cd and Pb concentrations in the aboveground part of the plants were within the “toxic range” for plants indicating a good tolerance to soil pollution by the studied plants [54].
The combination of the highest compost rate and biostimulants (C2-BP and C2-BT) reduced the Cd concentration in both plant shoots in comparison to the control (C0-B0) (Table 4). In addition, also the application of the highest compost dose alone (C2-B0) showed a similar reduction of Cd content in the shoots of mixed stand of grasses as compared with the control (C0-B0) (Table 4). This trend was in accordance with Cd bioavailable concentration in soil of both the grass swards (Table 2 and Figure 2), indicating a lower Cd uptake by plants because of lower Cd mobility in soil due to the compost interaction with soil Cd and to the interaction of compost with both mycorrhiza and Trichoderma [73,76,85,87]. On the other hand, the treatments showed a low effect on Pb concentration in the aboveground part of both the grass swards (Table 4). Nevertheless, the average effect of compost application caused a reduction of Pb concentration (C1 and C2) in the aboveground part of both the grass swards compared to those observed in the control treatment (C0) (Table 4). This tendency was in accordance with NH4NO3-extractable concentration of Pb in the soil, suggesting a possible Pb immobilization in soil (Table 2). However, the immobilization of Cd and Pb in plant roots and a low PTE transfer from root to shoot may have contributed to reduce the PTE uptake by plants. In line with our results, many authors reported a reduction of Cd and Pb concentration in shoots of D. glomerata L., F. arundinacea Shreb., L. perenne L. and P. pratensis L. after the application of organic amendment as a consequence of reduced PTE availability in the soil and to the immobilization of PTEs in plant roots [115,116,117,118].
3.4. Correlations between Plant and Soil Properties
Pearson’s correlation coefficients were calculated between plant and soil properties and are presented for D. glomerata L. and mixed stand of grasses, respectively, in Table 5 and Table 6.

Table 5.
Pearson correlation coefficients showing the relationship between soil and plant features for D. glomerata L. (without the unplanted control). The asterisk after values in bold indicate significant correlations at the 0.05 level (2-tailed). PW, pore water.

Table 6.
Pearson correlation coefficients showing the relationship between soil and plant features of a mixed stand of F. arundinacea Shreb., L. perenne L. and P. pratensis L. (without the unplanted control). The asterisk after values in bold indicate significant correlations at the 0.05 level (2-tailed). PW, pore water.
A significant negative correlation was observed for D. glomerata L. between plant growth and N concentration in plant (Table 5), to indicate a dilution effect occurring when dry-weight accumulation increased at a faster rate than N accumulation [119].
Both the grass swards reported a significant positive correlation between NH4NO3-extractable concentration of Pb in the soil and Pb concentration in plant shoots, revealing that the readily bioavailable content of PTEs in the soil are generally related to the amounts taken up by the plants [68] (Table 5 and Table 6). Furthermore, a negative correlation was observed for both the grass swards between pore water pH and Cd concentration in pore water. This result was in line with the increase of pore water pH after compost application (Figure 2) which may facilitate the adsorption of Cd on the soil colloids or may enhance surface and bulk precipitation of PTEs as insoluble metal hydroxides [94,95]. The Pb concentration in D. glomerata L. shoots was negatively correlated with plant biomass probably due to a dilution effect in high biomass plants [120] (Table 5). The NH4-N concentration in soil planted with D. glomerata L. was positively correlated with both amoA and nifH gene abundance (Table 5). This correlation was probably related to the ammonium provided by treatments and by nitrogen fixing bacteria that supplied energy for ammonia oxidizing bacteria improving their abundance [99,103,121]. This result was in accordance with the highest NH4-N, amoA and nifH gene abundance reported with the application of both compost and biostimulants to soil (Table 2 and Figure 2). In addition, a high significant correlation between amoA and nifH gene abundance was reported for both the grass swards highlighting their relation in N cycling (Table 5 and Table 6).
A statistically significant negative correlation was observed between plant growth (D. glomerata L.) or nitrogen uptake (mixed stand of grasses) and NH4NO3-extractable Cd and Pb concentrations in soil, indicating a strong phytotoxic effect of the two elements in the study soil (Table 5 and Table 6). Generally, Cd was more toxic for plants than Pb mostly due to the strong affinity of Cd ions with several compounds involved in plant metabolism (sulfhydryl groups and phosphate groups) resulting in a reduction of plant biomass [122,123]. On the other hand, Pb uptake and translocation by plant is limited due to its binding to root surface also forming granules and deposits into cell walls (mainly as pyrophosphate) of leaves and steams thus reducing its toxicity [122,123]. However, treatments with compost and biostimulants showed the lowest soil PTE concentrations (Table 2) and the highest plant growth and nitrogen uptake (Table 4), indicating that these biofertilizers may alleviate the phytotoxicity of Pb, Cd and their negative effects on host plants [69,124].
Furthermore, a significant negative correlation was observed for both the grass swards between bioavailable concentrations of PTEs in soil and nifH and amoA gene abundance, suggesting a negative effect of soil PTEs on nitrogen cycling bacteria (Table 5 and Table 6). In line with our study, Liu et al. [125] and Afzal et al. [126] reported the inhibition of the activity and function of bacteria involved in the ammonium oxidation in soil contaminated by Cd and Pb. Similarly to our results, Prasad et al. [127] stated a deleterious effect of soil Cd on abundance and diversity of free living nitrogen fixing bacteria in soil. In contrast with this trend, the highest nifH and amoA abundance occurred when biostimulants and compost where applied in combination, suggesting a stimulating and protective effect of compost and biostimulants for soil microorganisms (Table 3).
4. Conclusions
This study aimed to assess the effect of compost and biostimulants combined with two grass swards on the bioavailability of PTEs and soil biological fertility. In addition, the growth of plants and N uptake were evaluated. The soil used in this study, derived from a site used as landfill of lead-acid battery, showed pseudototal contents of Cd and Pb above screening values for industrial soils. The NH4NO3-extractable and pore water concentrations of PTEs in the soil revealed that Cd and Pb were very bioavailable and showed a negative effect on both the grass swards and nitrogen cycling bacteria. However, compost and biostimulants combination increased plant growth and N uptake, showing the highest effects with the biostimulant “Panoramix”. The same combination for both the grass swards reduced Cd mobility in soil by decreasing water soluble concentration (pore water) and the readily soluble fractions (soil extraction by NH4NO3), while reported a lower effect on soil Pb. Furthermore, the combination of compost and biostimulants also reduced Cd bioavailability as compared with a bare control highlighting the importance of a plant soil covering to reduce Cd dispersion in the environment. The lower Cd mobility in soil resulted in a lower Cd bioavailability for both the grass swards that showed a lower Cd concentration in shoots. Both compost fertilization and inoculation with biostimulants improved the soil biological fertility by enhancing soil nitrogen fixing and ammonia oxidizing bacteria for both the grass swards. The highest effects on soil and plants were reported by the biostimulant “Panoramix” highlighting the importance to use a biostimulant containing a mix of eukaryotic and prokaryotic microorganisms (mycorrhiza, Bacillus, and Trichoderma species) with different soil and plant beneficial effects instead of a pure culture of a single microorganism (T. harzianum).
The findings of this work indicate that the two grass species and the combined application of compost with the commercial biostimulant “Panoramix” represent effective tools for securing the industrial site by stabilizing Cd in soil, therefore reducing the risk of PTE dispersion in the surrounding environment and the risk of their leaching towards groundwater. However, further studies are required in field conditions to test the effectiveness of the combination plant-compost-biostimulants for the phytostabilization of the industrial site.
Author Contributions
Conceptualization, D.V. and N.F.; methodology, D.V. and N.F.; formal analysis, D.V. and L.P.; investigation, D.V., A.G.C., L.P. and V.V.; writing—original draft preparation, D.V., A.G.C. and V.V.; writing—review and editing, M.F., N.F., O.P., P.A. and S.L.W.; supervision, M.F. and N.F.; project administration, M.F.; funding acquisition, M.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by EC LIFE11/ENV/IT 275 Ecoremed and Italian MIUR PRIN2017BHH84R.
Acknowledgments
We would like to thank Eugenio Cozzolino and Vincenzo Cenvinzo for their support in crop management and sampling.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Moameri, M.; Khalaki, M.A. Capability of Secale montanum trusted for phytoremediation of lead and cadmium in soils amended with nano-silica and municipal solid waste compost. Environ. Sci. Pollut. Res. 2017, 26, 24315–24322. [Google Scholar] [CrossRef]
- González–Chávez, M.; Carrillo-González, R.; Sánchez, A.C.; Delgado-Alvarado, A.; Suárez-Espinosa, J.; Ríos-Leal, E.; Solís-Domínguez, F.A.; Maldonado-Mendoza, I.E. Phytoremediation assisted by mycorrhizal fungi of a Mexican defunct lead-acid battery recycling site. Sci. Total. Environ. 2019, 650, 3134–3144. [Google Scholar] [CrossRef] [PubMed]
- Smaniotto, A.; Antunes, A.; Filho, I.D.N.; Venquiaruto, L.D.; De Oliveira, D.; Mossi, A.; Di Luccio, M.; Treichel, H.; Dallago, R. Qualitative lead extraction from recycled lead–acid batteries slag. J. Hazard. Mater. 2009, 172, 1677–1680. [Google Scholar] [CrossRef] [PubMed]
- Haefliger, P.; Mathieu-Nolf, M.; Lociciro, S.; Ndiaye, C.; Coly, M.; Diouf, A.; Faye, A.L.; Sow, A.; Tempowski, J.; Pronczuk, J.; et al. Mass Lead Intoxication from Informal Used Lead-Acid Battery Recycling in Dakar, Senegal. Environ. Heal. Perspect. 2009, 117, 1535–1540. [Google Scholar] [CrossRef] [PubMed]
- Van Der Kuijp, T.J.; Huang, L.; Cherry, C. Health hazards of China’s lead-acid battery industry: A review of its market drivers, production processes, and health impacts. Environ. Heal. 2013, 12, 61. [Google Scholar] [CrossRef]
- Visconti, D.; Fiorentino, N.; Stinca, A.; Di Mola, I.; Fagnano, M. Use of the native vascular flora for risk assessment and management of an industrial contaminated soil. Ital. J. Agron. 2018, 13, 23–33. [Google Scholar] [CrossRef]
- Agency for Toxic Substances & Disease Registry, 2019. Priority List of Hazardous Substances. Available online: https://www.atsdr.cdc.gov/spl/index.html (accessed on 20 July 2020).
- Counter, S.A.; Vahter, M.; Laurell, G.; Buchanan, L.H.; Ortega, F.; Skerfving, S. High lead exposure and auditory sensory-neural function in Andean children. Environ. Heal. Perspect. 1997, 105, 522–526. [Google Scholar] [CrossRef]
- Tong, S.; Von Schirnding, Y.E.; Prapamontol, T. Environmental lead exposure: A public health problem of global dimensions. Bull. World Heal. Organ. 2000, 78, 1068–1077. [Google Scholar]
- Sanders, T.; Liu, Y.; Buchner, V.; Tchounwou, P.B. Neurotoxic effects and biomarkers of lead exposure: A review. Rev. Environ. Heal. 2009, 24, 15–45. [Google Scholar] [CrossRef]
- Chen, L.; Xu, Z.; Liu, M.; Huang, Y.; Fan, R.; Su, Y.; Hu, G.; Peng, X.; Peng, X. Lead exposure assessment from study near a lead-acid battery factory in China. Sci. Total. Environ. 2012, 429, 191–198. [Google Scholar] [CrossRef]
- Rahimzadeh, M.R.; Rahimzadeh, M.R.; Kazemi, S.; Moghadamnia, A.A. Cadmium toxicity and treatment: An update. Casp. J. Intern. Med. 2017, 8, 135–145. [Google Scholar]
- Ventorino, V.; Chiurazzi, M.; Aponte, M.; Pepe, O.; Moschetti, G. Genetic Diversity of a Natural Population of Rhizobium leguminosarum bv. viciae Nodulating Plants of Vicia faba in the Vesuvian Area. Curr. Microbiol. 2007, 55, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Pepe, O.; Ventorino, V.; Blaiotta, G. Dynamic of functional microbial groups during mesophilic composting of agro-industrial wastes and free-living (N2)-fixing bacteria application. Waste Manag. 2013, 33, 1616–1625. [Google Scholar] [CrossRef] [PubMed]
- Ventorino, V.; Parillo, R.; Testa, A.; Viscardi, S.; Espresso, F.; Pepe, O. Chestnut green waste composting for sustainable forest management: Microbiota dynamics and impact on plant disease control. J. Environ. Manag. 2016, 166, 168–177. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Li, X.; Li, X.; Wang, J.; Zhang, H. Changes of microbial population and N-cycling function genes with depth in three Chinese paddy soils. PLoS ONE 2017, 12, e0189506. [Google Scholar] [CrossRef]
- Levy-Booth, D.J.; Prescott, C.; Grayston, S.J. Microbial functional genes involved in nitrogen fixation, nitrification and denitrification in forest ecosystems. Soil Boil. Biochem. 2014, 75, 11–25. [Google Scholar] [CrossRef]
- Francis, C.A.; Beman, J.M.; Kuypers, M.M.M. New processes and players in the nitrogen cycle: The microbial ecology of anaerobic and archaeal ammonia oxidation. ISME J. 2007, 1, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; dos Santos, C.A.; Alves, P.R.L.; de Paula, A.M.; Nakatani, A.S.; Pereira, J.D.; Nogueira, M.A. Soil health: Looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Sci. Agric. 2013, 70, 274–289. [Google Scholar] [CrossRef]
- Fiorentino, N.; Mori, M.; Cenvinzo, V.; Duri, L.G.; Gioia, L.; Visconti, D.; Fagnano, M. Assisted phytoremediation for restoring soil fertility in contaminated and degraded land. Ital. J. Agron. 2018, 13, 34–44. [Google Scholar] [CrossRef]
- De Araújo, J.D.C.T.; Nascimento, C.W.A. Phytoextraction of Lead from Soil from a Battery Recycling Site: The Use of Citric Acid and NTA. Water, Air, Soil Pollut. 2009, 211, 113–120. [Google Scholar] [CrossRef]
- Pérez-López, R.; García, B.M.; Abreu, M.M.; Nieto, J.M.; Córdoba, F. Erica andevalensis and Erica australis growing in the same extreme environments: Phytostabilization potential of mining areas. Geoderma 2014, 194–203. [Google Scholar] [CrossRef]
- Radziemska, M.; Vaverková, M.D.; Baryła, A. Phytostabilization—Management Strategy for Stabilizing Trace Elements in Contaminated Soils. Int. J. Environ. Res. Public Heal. 2017, 14, 958. [Google Scholar] [CrossRef] [PubMed]
- Visconti, D.; Fiorentino, N.; Caporale, A.G.; Stinca, A.; Adamo, P.; Motti, R.; Fagnano, M. Analysis of native vegetation for detailed characterization of a soil contaminated by tannery waste. Environ. Pollut. 2019, 252, 1599–1608. [Google Scholar] [CrossRef] [PubMed]
- Gil-Loaiza, J.; White, S.A.; Root, R.A.; Solís-Dominguez, F.A.; Hammond, C.M.; Chorover, J.; Maier, R.M. Phytostabilization of mine tailings using compost-assisted direct planting: Translating greenhouse results to the field. Sci. Total. Environ. 2016, 565, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Radziemska, M. Aided Phytostabilization of Copper Contaminated Soils with L. Perenne and Mineral Sorbents as Soil Amendments. Civ. Environ. Eng. Rep. 2017, 26, 79–89. [Google Scholar] [CrossRef][Green Version]
- Atabayeva, S. Heavy Metals Accumulation Ability of Wild Grass Species from Industrial Areas of Kazakhstan; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2016; Volume 3, pp. 157–208. [Google Scholar]
- Visconti, D.; Álvarez-Robles, M.J.; Fiorentino, N.; Fagnano, M.; Clemente, R. Use of Brassica juncea and Dactylis glomerata for the phytostabilization of mine soils amended with compost or biochar. Chemosphere 2020, 260, 127661. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Abadi, V.A.J.M.; Da Silva, J.A.T. Potential of arbuscular mycorrhizae and tall fescue in remediation of soils polluted with zinc. Chem. Ecol. 2020, 36, 122–137. [Google Scholar] [CrossRef]
- Beddows, A.R. Dactylis glomerata L. J. Ecol. 1959, 47, 223. [Google Scholar] [CrossRef]
- Gibson, D.J.; Newman, J.A. Festuca arundinacea Schreber (F. elatior L. ssp. arundinacea (Schreber) Hackel). J. Ecol. 2001, 89, 304–324. [Google Scholar] [CrossRef]
- Flora of China. eFloras.org Home Page. Available online: http://www.efloras.org/flora_page.aspx?flora_id=2 (accessed on 17 August 2020).
- Beddows, A.R. Lolium Perenne L. J. Ecol. 1967, 55, 567. [Google Scholar] [CrossRef]
- Chiu, K.; Ye, Z.; Wong, M. Growth of and on Pb/Zn and Cu mine tailings amended with manure compost and sewage sludge: A greenhouse study. Bioresour. Technol. 2006, 97, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.; Mahoney, M.; Sprenger, M. A comparison of the efficacy and ecosystem impact of residual-based and topsoil-based amendments for restoring historic mine tailings in the Tri-State mining district. Sci. Total. Environ. 2014, 485, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-S.; Min, H.-G.; Lee, S.-H.; Kim, J.-G. A Comparative Study on Poaceae and Leguminosae Forage Crops for Aided Phytostabilization in Trace-Element-Contaminated Soil. Agron. 2018, 8, 105. [Google Scholar] [CrossRef]
- Lebrun, M.; De Zio, E.; Miard, F.; Scippa, G.S.; Renzone, G.; Scaloni, A.; Bourgerie, S.; Morabito, D.; Trupiano, D. Amending an As/Pb contaminated soil with biochar, compost and iron grit: Effect on Salix viminalis growth, root proteome profiles and metal(loid) accumulation indexes. Chemosphere 2020, 244, 125397. [Google Scholar] [CrossRef]
- Alluvione, F.; Fiorentino, N.; Bertora, C.; Zavattaro, L.; Fagnano, M.; Chiarandà, F.Q.; Grignani, C. Short-term crop and soil response to C-friendly strategies in two contrasting environments. Eur. J. Agron. 2013, 45, 114–123. [Google Scholar] [CrossRef]
- Kumpiene, J.; Lagerkvist, A.; Maurice, C. Stabilization of As, Cr, Cu, Pb and Zn in soil using amendments—A review. Waste Manag. 2008, 28, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Fagnano, M.; Adamo, P.; Zampella, M.; Fiorentino, N. Environmental and agronomic impact of fertilization with composted organic fraction from municipal solid waste: A case study in the region of Naples, Italy. Agric. Ecosyst. Environ. 2011, 141, 100–107. [Google Scholar] [CrossRef]
- Clemente, R.; Pardo, T.; Madejón, P.; Madejón, E.; Bernal, M.P. Food byproducts as amendments in trace elements contaminated soils. Food Res. Int. 2015, 73, 176–189. [Google Scholar] [CrossRef]
- Fiorentino, N.; Fagnano, M.; Impagliazzo, A.; Mori, M.; Ventorino, V.; Zoina, A.; Adamo, P.; Pepe, O. Assisted phytoextraction of heavy metals: Compost and Trichoderma effects on giant reed (Arundo donax L.) uptake and soil N-cycle microflora. Ital. J. Agron. 2013, 8, 29. [Google Scholar] [CrossRef]
- Zhang, X.-H.; Wu, H.-X.; Huang, L.; Liu, C.-J. From Homogeneous to Heterogeneous: A Simple Approach to Prepare Polymer Brush Modified Surfaces for Anti-Adhesion of Bacteria. Colloid Interface Sci. Commun. 2018, 23, 21–28. [Google Scholar] [CrossRef]
- Li, X.; Zhang, X.; Wang, X.; Yang, X.; Cui, Z. Bioaugmentation-assisted phytoremediation of lead and salinity co-contaminated soil by Suaeda salsa and Trichoderma asperellum. Chemosphere 2019, 224, 716–725. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.-H.; Zhou, Z.; Gao, Y.-Q.; Yuan, X.-T.; Ai, Y.-J.; Zhang, J.-Y.; Zuo, W.-Z.; Taylor, A.A.; Nan, S.-Q.; Li, F.-P. The influences of arbuscular mycorrhizal fungus on phytostabilization of lead/zinc tailings using four plant species. Int. J. Phytoremediation 2017, 19, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Upadhyaya, H.; Panda, S.K.; Bhattacharjee, M.K.; Dutta, S. Role of arbuscular mycorrhiza in heavy metal tolerance in plants: Prospects for phytoremidiation. J. Phytol. 2010, 2, 16–27. [Google Scholar]
- Italian Legislative Decree 152. Environmental Regulations. Decree n. 152, April 3, 2006. (Norme in Materia Ambientale. Decreto n. 152, 3 Aprile 2006). Available online: https://www.gazzettaufficiale.it/dettaglio/codici/materiaAmbientale (accessed on 21 July 2020).
- BBodSchV. Bodenschutz- und Altlastenverordnung (Federal Soil Protection and Contaminated Sites Ordinance); 12 July 1999; Germany. Available online: https://www.gesetze-im-internet.de/bbodschv/BBodSchV.pdf (accessed on 20 July 2020).
- ASP. Act No. 220/2004 Coll. on the Protection and Use of the Agricultural Soil; The National Council of the Slovak Republic: Bratislava, Slovakia, 2004.
- Carlon, C. Derivation Methods of Soil Screening Values in Europe. A Review and Evaluation of National Procedures towards Harmonization, EUR 22805-EN; European Commission, Joint Research Centre, ISPRA: Rome, Italy, 2007; p. 306. [Google Scholar]
- Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern. Anal. Bioanal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- European Union Regulation No. 1275 of 6 December 2013 Amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as Regards Maximum Levels for Arsenic, Cadmium, Lead, Nitrites, Volatile Mustard Oil and Harmful Botanical Impurities Text with EEA Relevance. Available online: https://op.europa.eu/en/publication-detail/-/publication/79e4ba4c-5f1d-11e3-ab0f-01aa75ed71a1/language-en (accessed on 1 September 2020).
- Kabata-Pendias, A. Trace Elements in Soils and Plants; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2011. [Google Scholar]
- DIN 19730. Bodenbeschaffenheit-Extraktion von Spurenelementen mit Ammoniumnitratlösung; Beuth Verlag: Berlin, Germany, 1997. [Google Scholar]
- Walkley, A.; Black, I.A. An examination of Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–37. [Google Scholar] [CrossRef]
- ISO 12914. Soil Quality—Microwave-Assisted Extraction of the Aqua Regia Soluble Fraction for the Determination of Elements; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- Romano, I.; Ventorino, V.; Pepe, O. Effectiveness of Plant Beneficial Microbes: Overview of the Methodological Approaches for the Assessment of Root Colonization and Persistence. Front. Plant Sci. 2020, 11, 6. [Google Scholar] [CrossRef]
- Rösch, C.; Mergel, A.; Bothe, H. Biodiversity of Denitrifying and Dinitrogen-Fixing Bacteria in an Acid Forest Soil. Appl. Environ. Microbiol. 2002, 68, 3818–3829. [Google Scholar] [CrossRef]
- Rotthauwe, J.H.; Witzel, K.P.; Liesack, W. The ammonia monooxygenase structural gene amoA as a functional marker: Molecular fine-scale analysis of natural ammo- nia-oxidizing populations. Appl. Environ. Microbiol. 1997, 63, 4704–4712. [Google Scholar] [CrossRef]
- Fiorentino, N.; Ventorino, V.; Bertora, C.; Pepe, O.; Giancarlo, M.; Grignani, C.; Fagnano, M. Changes in soil mineral N content and abundances of bacterial communities involved in N reactions under laboratory conditions as predictors of soil N availability to maize under field conditions. Boil. Fertil. Soils 2016, 52, 523–537. [Google Scholar] [CrossRef]
- Kant, S. Understanding nitrate uptake, signaling and remobilisation for improving plant nitrogen use efficiency. Semin. Cell Dev. Boil. 2018, 74, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Visconti, D.; Fiorentino, N.; Cozzolino, E.; Woo, S.; Fagnano, M.; Rouphael, Y. Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems? Agronomy 2020, 10, 121. [Google Scholar] [CrossRef]
- Materechera, S.A.; Salagae, A.M. Use of partially decomposed cattle and chicken compost amendment with wood ash in two South Africa arable soils with contrasting texture, effect on nutrient uptake, early growth, and dry matter yield of maize. Commun. Soil Sci. Plant Anal. 2002, 33, 179–201. [Google Scholar] [CrossRef]
- Cambardella, C.; Richard, T.L.; Russell, A. Compost mineralization in soil as a function of composting process conditions. Eur. J. Soil Boil. 2003, 39, 117–127. [Google Scholar] [CrossRef]
- Binh, N.T.; Shima, K. Nitrogen Mineralization in Soil Amended with Compost and Urea as Affected by Plant Residues Supplements with Controlled C/N Ratios. J. Adv. Agric. Technol. 2018, 5, 8–13. [Google Scholar] [CrossRef]
- Duri, L.G.; Fiorentino, N.; Cozzolino, E.; Ottaiano, L.; Agrelli, D.; Fagnano, M. Bioassays for evaluation of sanitary risks from food crops cultivated in potentially contaminated sites. Ital. J. Agron. 2018, 13, 45–52. [Google Scholar]
- Duri, L.G.; Visconti, D.; Fiorentino, N.; Adamo, P.; Fagnano, M.; Caporale, A.G. Health Risk Assessment in Agricultural Soil Potentially Contaminated by Geogenic Thallium: Influence of Plant Species on Metal Mobility in Soil-Plant System. Agron. 2020, 10, 890. [Google Scholar] [CrossRef]
- Göhre, V.; Paszkowski, U. Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 2006, 223, 1115–1122. [Google Scholar] [CrossRef]
- Janoušková, M.; Pavlíková, D. Cadmium immobilization in the rhizosphere of arbuscular mycorrhizal plants by the fungal extraradical mycelium. Plant Soil 2010, 332, 511–520. [Google Scholar] [CrossRef]
- Alguacil, M.; Torrecillas, E.; Caravaca, F.; Fernandez, D.; Azcón, R.; Roldan, A. The application of an organic amendment modifies the arbuscular mycorrhizal fungal communities colonizing native seedlings grown in a heavy-metal-polluted soil. Soil Boil. Biochem. 2011, 43, 1498–1508. [Google Scholar] [CrossRef]
- Wang, F.; Liu, X.; Shi, Z.; Tong, R.; Adams, C.A.; Shi, X. Arbuscular mycorrhizae alleviate negative effects of zinc oxide nanoparticle and zinc accumulation in maize plants—A soil microcosm experiment. Chemosphere 2016, 147, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, J.; Yue, F.; Yan, X.; Wang, F.; Bloszies, S.; Wang, Y. Effects of arbuscular mycorrhizal inoculation and biochar amendment on maize growth, cadmium uptake and soil cadmium speciation in Cd-contaminated soil. Chemosphere 2018, 194, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.; Singh, P.C.; Mishra, A.; Chauhan, P.S.; Dwivedi, S.; Bais, R.T.; Tripathi, R.D. Trichoderma: A potential bioremediator for environmental clean up. Clean Technol. Environ. Policy 2013, 15, 541–550. [Google Scholar] [CrossRef]
- Mohsenzadeh, F.; Shahrokhi, F. Biological removing of Cadmium from contaminated media by fungal biomass of Trichoderma species. J. Environ. Heal. Sci. Eng. 2014, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, M.; Gul, I.; Kallerhoff, J.; Arshad, M. Fungi-assisted phytoextraction of lead: Tolerance, plant growth–promoting activities and phytoavailability. Environ. Sci. Pollut. Res. 2019, 26, 23788–23797. [Google Scholar] [CrossRef]
- Quilliam, R.S.; Hodge, A.; Jones, D. Sporulation of arbuscular mycorrhizal fungi in organic-rich patches following host excision. Appl. Soil Ecol. 2010, 46, 247–250. [Google Scholar] [CrossRef]
- Bukovská, P.; Gryndler, M.; Gryndlerová, H.; Püschel, D.; Jansa, J. Organic Nitrogen-Driven Stimulation of Arbuscular Mycorrhizal Fungal Hyphae Correlates with Abundance of Ammonia Oxidizers. Front. Microbiol. 2016, 7, 711. [Google Scholar] [CrossRef]
- Nuccio, E.E.; Herman, D.J.; Weber, P.K.; Firestone, M.K.; Pett-Ridge, J.; Hodge, A. An arbuscular mycorrhizal fungus significantly modifies the soil bacterial community and nitrogen cycling during litter decomposition. Environ. Microbiol. 2013, 15, 1870–1881. [Google Scholar] [CrossRef]
- Bukovska, P.; Bonkowski, M.; Konvalinkovã¡, T.; Beskid, O.; Hujslová, M.; Püschel, D.; Řezáčová, V.; Gutiérrez-Núñez, M.S.; Gryndler, M.; Jansa, J. Utilization of organic nitrogen by arbuscular mycorrhizal fungi—Is there a specific role for protists and ammonia oxidizers? Mycorrhiza 2018, 28, 269–283. [Google Scholar] [CrossRef]
- Zhang, F.; Huo, Y.; Cobb, A.B.; Luo, G.; Zhou, J.; Yang, G.; Wilson, G.W.T.; Zhang, Y. Trichoderma Biofertilizer Links to Altered Soil Chemistry, Altered Microbial Communities, and Improved Grassland Biomass. Front. Microbiol. 2018, 9, 848. [Google Scholar] [CrossRef]
- Zhang, F.; Huo, Y.; Xu, X.; Hu, J.; Sun, X.; Xiao, Y.; Zhang, Y. Trichoderma improves the growth of Leymus chinensis. Boil. Fertil. Soils 2018, 54, 685–696. [Google Scholar] [CrossRef]
- Ye, L.; Zhao, X.; Bao, E.; Li, J.; Zou, Z.; Cao, K. Bio-organic fertilizer with reduced rates of chemical fertilization improves soil fertility and enhances tomato yield and quality. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Coskun, D.; Britto, D.T.; Shi, W.; Kronzucker, H.J. How Plant Root Exudates Shape the Nitrogen Cycle. Trends Plant Sci. 2017, 22, 661–673. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Luo, N.; Liu, C.; Zeng, G.; Li, Z.; Huang, B.; Zhu, Y. Compost as a Soil Amendment to Remediate Heavy Metal-Contaminated Agricultural Soil: Mechanisms, Efficacy, Problems, and Strategies. Water Air Soil Pollut. 2016, 227, 359. [Google Scholar] [CrossRef]
- Köhler, J.; Caravaca, F.; Azcón, R.; Diaz, G.; Roldan, A. The combination of compost addition and arbuscular mycorrhizal inoculation produced positive and synergistic effects on the phytomanagement of a semiarid mine tailing. Sci. Total. Environ. 2015, 514, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.M.M.; Mazen, M.B.-E.-D.; Nafady, N.A.; Monsef, O.A. Bioavailability of cadmium and nickel to Daucus carota L. and Corchorus olitorius L. treated by compost and microorganisms. Soil Environ. 2017, 36, 1–12. [Google Scholar] [CrossRef]
- Vinci, G.; Cozzolino, V.; Mazzei, P.; Monda, H.; Spaccini, R.; Piccolo, A. An alternative to mineral phosphorus fertilizers: The combined effects of Trichoderma harzianum and compost on Zea mays, as revealed by 1H NMR and GC-MS metabolomics. PLoS ONE 2018, 13, e0209664. [Google Scholar] [CrossRef]
- Bali, A.S.; Sidhu, G.P.S.; Kumar, V. Root exudates ameliorate cadmium tolerance in plants: A review. Environ. Chem. Lett. 2020, 18, 1243–1275. [Google Scholar] [CrossRef]
- Hartley, W.; Dickinson, N.M.; Riby, P.; Lepp, N.W. Arsenic mobility in brownfield soils amended with green waste compost or biochar and planted with Miscanthus. Environ. Pollut. 2009, 157, 2654–2662. [Google Scholar] [CrossRef]
- Ciadamidaro, L.; Puschenreiter, M.; Santner, J.; Wenzel, W.W.; Madejón, P.; Madejón, E. Assessment of trace element phytoavailability in compost amended soils using different methodologies. J. Soil. Sedim. 2015, 17, 1251–1261. [Google Scholar] [CrossRef]
- Beesley, L.; Dickinson, N. Carbon and trace element fluxes in the pore water of an urban soil following greenwaste compost, woody and biochar amendments, inoculated with the earthworm Lumbricus terrestris. Soil Biol. Biochem. 2011, 43, 188–196. [Google Scholar] [CrossRef]
- Álvarez-Rogel, J.; Gómez, M.D.C.T.; Conesa, H.M.; Parraga-Aguado, I.; González-Alcaraz, M.N. Biochar from sewage sludge and pruning trees reduced porewater Cd, Pb and Zn concentrations in acidic, but not basic, mine soils under hydric conditions. J. Environ. Manag. 2018, 223, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, G.; Haynes, R.J.; Naidu, R.; Belyaeva, O.; Kim, K.-R.; Lamb, D.T.; Bolan, N. Natural Attenuation of Zn, Cu, Pb and Cd in Three Biosolids-Amended Soils of Contrasting pH Measured Using Rhizon Pore Water Samplers. Water Air Soil Pollut. 2011, 221, 351–363. [Google Scholar] [CrossRef]
- Rocco, C.; Seshadri, B.; Adamo, P.; Bolan, N.; Mbene, K.; Naidu, R. Impact of waste-derived organic and inorganic amendments on the mobility and bioavailability of arsenic and cadmium in alkaline and acid soils. Environ. Sci. Pollut. Res. 2018, 25, 25896–25905. [Google Scholar] [CrossRef]
- Khan, M.A.; Khan, S.; Khan, A.; Alam, M. Soil contamination with cadmium, consequences and remediation using organic amendments. Sci. Total. Environ. 2017, 601, 1591–1605. [Google Scholar] [CrossRef]
- Ventorino, V.; Pascale, A.; Fagnano, M.; Adamo, P.; Faraco, V.; Rocco, C.; Fiorentino, N.; Pepe, O. Soil tillage and compost amendment promote bioremediation and biofertility of polluted area. J. Clean. Prod. 2019, 239, 118087. [Google Scholar] [CrossRef]
- Artursson, V.; Finlay, R.D.; Jansson, J.K. Interactions between arbuscular mycorrhizal fungi and bacteria and their potential for stimulating plant growth. Environ. Microbiol. 2006, 8, 1–10. [Google Scholar] [CrossRef]
- El Sharkawi, H.; Yamamoto, S.; Honna, T. Rice yield nutrient uptake as affected by cyanobacteria soil amendments—A pot experiment. J. Plant Nutr. Soil Sci. 2006, 169, 809–815. [Google Scholar] [CrossRef]
- Ventorino, V.; De Marco, A.; Pepe, O.; De Santo, A.V.; Moschetti, G. Impact of Innovative Agricultural Practices of Carbon Sequestration on Soil Microbial Community. In Carbon Sequestration in Agricultural Soils; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2011; pp. 145–177. [Google Scholar]
- Gupta, R.; Bisaria, V.S.; Sharma, S. Response of rhizospheric bacterial communities of Cajanus cajan to application of bioinoculants and chemical fertilizers: A comparative study. Eur. J. Soil Boil. 2016, 75, 107–114. [Google Scholar] [CrossRef]
- Nelson, K.N.; Neilson, J.W.; Root, R.A.; Chorover, J.; Maier, R.M. Abundance and Activity of 16S rRNA, AmoA and NifH Bacterial Genes during Assisted Phytostabilization of Mine Tailings. Int. J. Phytoremediat. 2015, 17, 493–502. [Google Scholar] [CrossRef]
- Verhamme, D.T.; I Prosser, J.; Nicol, G.W. Ammonia concentration determines differential growth of ammonia-oxidising archaea and bacteria in soil microcosms. ISME J. 2011, 5, 1067–1071. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.; Van Nostrand, J.D.; Vangronsveld, J.; Witters, N.; Janssen, J.; Kumpiene, J.; Siebielec, G.; Galazka, R.; Giagnoni, L.; Arenella, M.; et al. Management with willow short rotation coppice increase the functional gene diversity and functional activity of a heavy metal polluted soil. Chemosphere 2015, 138, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Buyer, J.S.; Teasdale, J.R.; Roberts, D.P.; Zasada, I.A.; Maul, J.E. Factors affecting soil microbial community structure in tomato cropping systems. Soil Boil. Biochem. 2010, 42, 831–841. [Google Scholar] [CrossRef]
- Boureima, S.; Diouf, M.; Diop, T.A.; Diatta, M.; Leye, E.M.; Ndiaye, F.; Seck, D. Effects of arbuscular mycorrhizal inoculation on the growth and the development of sesame (Sesamum indicum L.). Afr. J. Agric. Res. 2008, 3, 234–238. [Google Scholar]
- Talbi, Z.; Chliyeh, M.; Mouria, B.; El Asri, A.; Ait Aguil, F.; Touhami, A.O.; Benkirane, R.; Douira, A. Effect of double inoculation with endomycorrhizae and Trichoderma harzianum on the growth of carob plants. IJAPBC 2016, 5, 44–58. [Google Scholar]
- Hashem, A.; Tabassum, B.; Fathi Abd Allah, E. Bacillus subtilis: A plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J. Biol. Sci. 2019, 26, 1291–1297. [Google Scholar] [CrossRef]
- Pelletier, S.; Dionne, J. Inoculation rate of arbuscular-mycorrhizal fungi Glomus intraradices and Glomus etunicatum affects establishment of landscape turf with no irrigation or fertilizer inputs. Crop. Sci. 2004, 44, 335–338. [Google Scholar] [CrossRef]
- Nikbakht, A.; Pessarakli, M.; Daneshvar-Hakimi-Maibodi, N.; Kafi, M. Perennial Ryegrass Growth Responses to Mycorrhizal Infection and Humic Acid Treatments. Agron. J. 2014, 106, 585–595. [Google Scholar] [CrossRef]
- Harman, G.E. Overview of Mechanisms and Uses ofTrichodermaspp. Phytopathol. 2006, 96, 190–194. [Google Scholar] [CrossRef]
- Al-Bataina, B.B.; Young, T.M.; Ranieri, E. Effects of compost age on the release of nutrients. Int. Soil Water Conserv. Res. 2016, 4, 230–236. [Google Scholar] [CrossRef]
- Medina, A.; Azcón, R. Effectiveness of the application of arbuscular mycorrhiza fungi and organic amendments to improve soil quality and plant performance under stress conditions. J. Soil Sci. Plant Nutr. 2010, 10, 354–372. [Google Scholar] [CrossRef]
- Yang, W.; Gu, S.; Xin, Y.; Bello, A.; Sun, W.; Xu, X. Compost Addition Enhanced Hyphal Growth and Sporulation of Arbuscular Mycorrhizal Fungi without Affecting Their Community Composition in the Soil. Front. Microbiol. 2018, 9, 169. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.; Mullen, G.J.; Courtney, R. Metal uptake in lolium perenne established on spent mushroom compost amended lead-zinc tailings. Land Degrad. Dev. 2009, 20, 277–282. [Google Scholar] [CrossRef]
- Padmavathiamma, P.K.; Li, L.Y. Phytoavailability and fractionation of lead and manganese in a contaminated soil after application of three amendments. Bioresour. Technol. 2010, 101, 5667–5676. [Google Scholar] [CrossRef]
- Kacprzak, M.; Grobelak, A.; Grosser, A.; Prasad, M.N.V. Efficacy of Biosolids in Assisted Phytostabilization of Metalliferous Acidic Sandy Soils with Five Grass Species. Int. J. Phytoremediat. 2013, 16, 593–608. [Google Scholar] [CrossRef]
- Özbaş, E.E.; Özcan, H.K.; Ongen, A. Efficiency of MSW compost for reducing uptake of heavy metals by plant. Environ. Prot. Eng. 2016, 42, 4. [Google Scholar] [CrossRef]
- Riedell, W.E. Mineral-nutrient synergism and dilution responses to nitrogen fertilizer in field-grown maize. J. Plant Nutr. Soil Sci. 2010, 173, 869–874. [Google Scholar] [CrossRef]
- Clemente, R.; Walker, D.J.; Bernal, M.P. Uptake of heavy metals and As by Brassica juncea grown in a contaminated soil in Aznalcóllar (Spain): The effect of soil amendments. Environ. Pollut. 2005, 138, 46–58. [Google Scholar] [CrossRef]
- Wang, J.; Ni, L.; Song, Y.; Rhodes, G.; Li, J.; Huang, Q.; Shen, Q. Dynamic Response of Ammonia-Oxidizers to Four Fertilization Regimes across a Wheat-Rice Rotation System. Front. Microbiol. 2017, 8, 630. [Google Scholar] [CrossRef]
- Påhlsson, A.-M.B. Toxicity of heavy metals (Zn, Cu, Cd, Pb) to vascular plants. Water Air Soil Pollut. 1989, 47, 287–319. [Google Scholar] [CrossRef]
- Wang, F.; Wang, L.; Shi, Z.Y.; Li, Y.J.; Song, Z.M. Effects of AM Inoculation and Organic Amendment, Alone or in Combination, on Growth, P Nutrition, and Heavy-Metal Uptake of Tobacco in Pb-Cd-Contaminated Soil. J. Plant Growth Regul. 2012, 31, 549–559. [Google Scholar] [CrossRef]
- Mau, A.E.; Utami, S.R. Effects of biochar amendment and arbuscular mycorrhizal fungi inoculation on availability of soil phosphorus and growth of maize. J. Degr. Min. Lands Manag. 2014, 1, 69–74. [Google Scholar]
- Liu, Y.; Liu, Y.; Ding, Y.; Zheng, J.; Zhou, T.; Pan, G.; Crowley, D.; Li, L.; Zheng, J.; Zhang, X.; et al. Abundance, Composition and Activity of Ammonia Oxidizer and Denitrifier Communities in Metal Polluted Rice Paddies from South China. PLoS ONE 2014, 9, e102000. [Google Scholar] [CrossRef]
- Afzal, M.; Yu, M.; Tang, C.; Zhang, L.; Muhammad, N.; Zhao, H.; Feng, J.; Yu, L.; Xu, J. The negative impact of cadmium on nitrogen transformation processes in a paddy soil is greater under non-flooding than flooding conditions. Environ. Int. 2019, 129, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Bolla, K.; Prasad, D.; Subrahmanyam, G. Effect of Cadmium on Abundance and Diversity of Free Living Nitrogen Fixing Azotobacter spp. J. Environ. Sci. Technol. 2012, 5, 184–191. [Google Scholar] [CrossRef][Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).