The Emerging Roles of Diacylglycerol Kinase (DGK) in Plant Stress Tolerance, Growth, and Development
Abstract
:1. Introduction
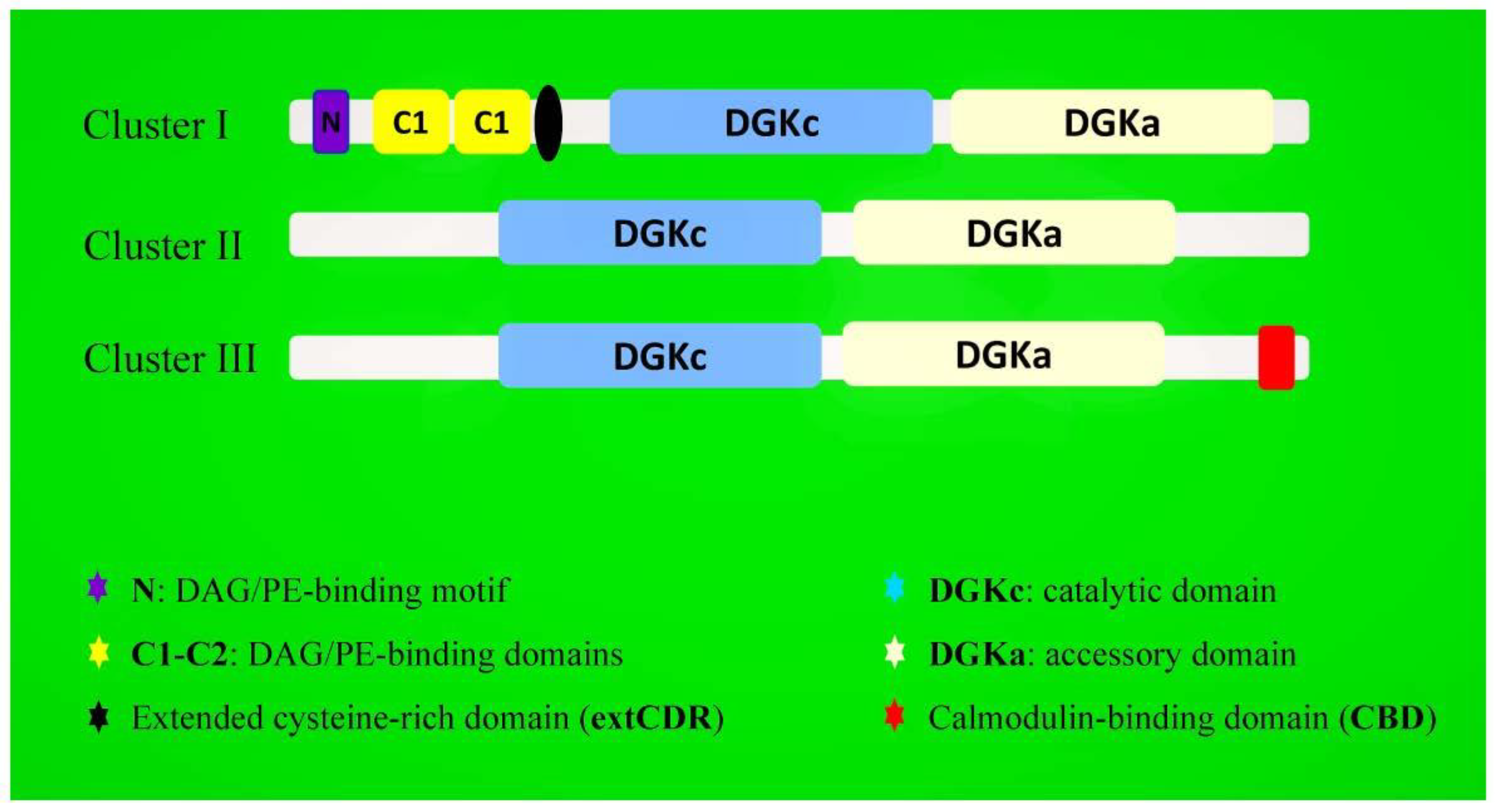
2. Structure and Isoforms of Plant DGK Genes
3. Subcellular Localization and Tissue Distribution of DGK Genes
4. DGK in Plant Development
4.1. DGK in Growth and Development
4.2. DGK in Nitric Oxide-Dependent Pollen Tube Guidance and Fertilization
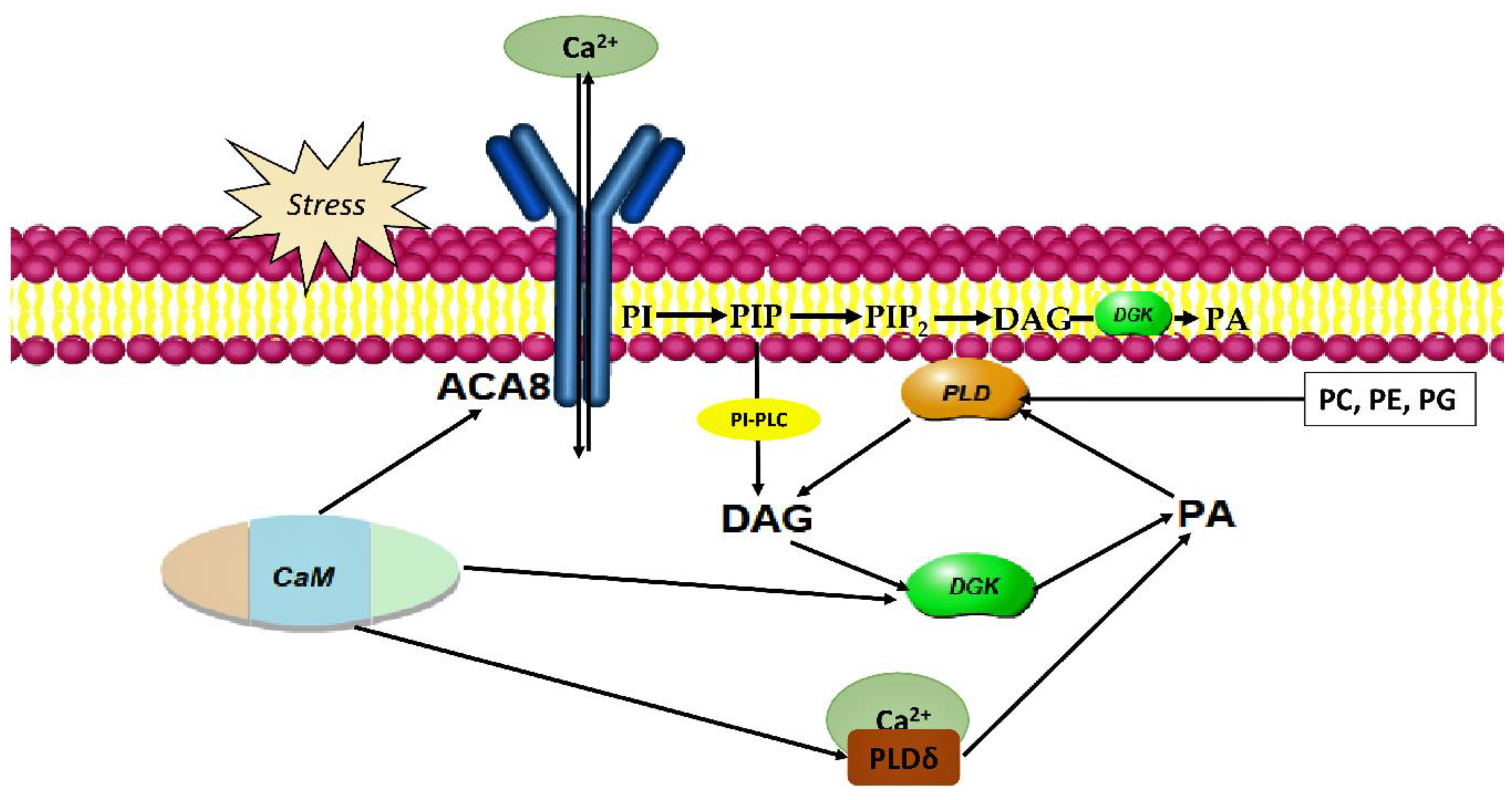
5. DGK in Plant Stress Adaptation
5.1. Biotic Stress
5.2. Abiotic Stress
5.2.1. Drought Stress
5.2.2. Cold Stress
5.2.3. Saline and Osmotic Stresses
5.2.4. Heavy Metal Stress
6. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Ramegowda, V.; Senthil-Kumar, M. The interactive effects of simultaneous biotic and abiotic stresses on plants: Mechanistic understanding from drought and pathogen combination. J. Plant Physiol. 2015, 176, 47–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- AbuQamar, S.; Luo, H.; Laluk, K.; Mickelbart, M.V.; Mengiste, T. Crosstalk between biotic and abiotic stress responses in tomato is mediated by the AIM1 transcription factor. Plant J. 2009, 58, 347–360. [Google Scholar] [CrossRef]
- Mao, X.; Zhang, H.; Tian, S.; Chang, X.; Jing, R. TaSnRK2. 4, an SNF1-type serine/threonine protein kinase of wheat (Triticum aestivum L.), confers enhanced multistress tolerance in Arabidopsis. J. Exp. Bot. 2009, 61, 683–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sreenivasulu, N.; Sopory, S.; Kishor, P.K. Deciphering the regulatory mechanisms of abiotic stress tolerance in plants by genomic approaches. Gene 2007, 388, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Narsai, R.; Wang, C.; Chen, J.; Wu, J.; Shou, H.; Whelan, J. Antagonistic, overlapping and distinct responses to biotic stress in rice (Oryza sativa) and interactions with abiotic stress. BMC Genom. 2013, 14, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, M.; Liang, X.Q.; Dang, P.; Holbrook, C.C.; Bausher, M.G.; Lee, R.D.; Guo, B.Z. Microarray-based screening of differentially expressed genes in peanut in response to Aspergillus parasiticus infection and drought stress. Plant Sci. 2005, 169, 695–703. [Google Scholar] [CrossRef]
- Shaik, R.; Ramakrishna, W. Genes and co-expression modules common to drought and bacterial stress responses in Arabidopsis and rice. PLoS ONE 2013, 8, e77261. [Google Scholar] [CrossRef] [Green Version]
- Sharma, R.; De Vleesschauwer, D.; Sharma, M.K.; Ronald, P.C. Recent advances in dissecting stress-regulatory crosstalk in rice. Mol. Plant 2013, 6, 250–260. [Google Scholar] [CrossRef] [Green Version]
- Tippmann, H.F.; Schlüter, U.; Collinge, D.B. Common themes in biotic and abiotic stress signalling in plants. In Floriculture, Ornamental and Plant Biotechnology; Global Science Books: London, UK, 2006; pp. 52–67. [Google Scholar]
- Arisz, S.A.; Testerink, C.; Munnik, T. Plant PA signaling via diacylglycerol kinase. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2009, 1791, 869–875. [Google Scholar] [CrossRef]
- Barbaglia, A.M.; Hoffmann-Benning, S. Long-distance lipid signaling and its role in plant development and stress response. In Lipids in Plant and Algae Development; Springer: Berlin/Heidelberg, Germany, 2016; pp. 339–361. [Google Scholar]
- Ohlrogge, J.; Browse, J. Lipid biosynthesis. Plant Cell 1995, 7, 957. [Google Scholar]
- Testerink, C.; Munnik, T. Phosphatidic acid: A multifunctional stress signaling lipid in plants. Trends Plant Sci. 2005, 10, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Testerink, C.; Munnik, T. Molecular, cellular, and physiological responses to phosphatidic acid formation in plants. J. Exp. Bot. 2011, 62, 2349–2361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Lin, F.; Mao, T.; Nie, J.; Yan, M.; Yuan, M.; Zhang, W. Phosphatidic acid regulates microtubule organization by interacting with MAP65-1 in response to salt stress in Arabidopsis. Plant Cell 2012, 24, 4555–4576. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Qin, C.; Zhao, J.; Wang, X. Phospholipase Dα1-derived phosphatidic acid interacts with ABI1 phosphatase 2C and regulates abscisic acid signaling. Proc. Natl. Acad. Sci. USA 2004, 101, 9508–9513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munnik, T.; Meijer, H.J.; Ter Riet, B.; Hirt, H.; Frank, W.; Bartels, D.; Musgrave, A. Hyperosmotic stress stimulates phospholipase D activity and elevates the levels of phosphatidic acid and diacylglycerol pyrophosphate. Plant J. 2000, 22, 147–154. [Google Scholar] [CrossRef]
- Testerink, C.; Dekker, H.L.; Lim, Z.Y.; Johns, M.K.; Holmes, A.B.; de Koster, C.G.; Ktistakis, N.T.; Munnik, T. Isolation and identification of phosphatidic acid targets from plants. Plant J. 2004, 39, 527–536. [Google Scholar] [CrossRef]
- Li, Y.; Tan, Y.; Shao, Y.; Li, M.; Ma, F. Comprehensive genomic analysis and expression profiling of diacylglycerol kinase gene family in Malus prunifolia (Willd.) Borkh. Gene 2015, 561, 225–234. [Google Scholar] [CrossRef]
- Munnik, T. Phosphatidic acid: An emerging plant lipid second messenger. Trends Plant Sci. 2001, 6, 227–233. [Google Scholar] [CrossRef]
- Ruelland, E.; Cantrel, C.; Gawer, M.; Kader, J.C.; Zachowski, A. Activation of phospholipases C and D is an early response to a cold exposure in Arabidopsis suspension cells. Plant Physiol. 2002, 130, 999–1007. [Google Scholar] [CrossRef] [Green Version]
- Arisz, S.A.; van Wijk, R.V.; Roels, W.; Zhu, J.K.; Haring, M.A.; Munnik, T. Rapid phosphatidic acid accumulation in response to low temperature stress in Arabidopsis is generated through diacylglycerol kinase. Front. Plant Sci. 2013, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Xiao, S. Lipids in salicylic acid-mediated defense in plants: Focusing on the roles of phosphatidic acid and phosphatidylinositol 4-phosphate. Front. Plant Sci. 2015, 6, 387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyodo, K.; Taniguchi, T.; Manabe, Y.; Kaido, M.; Mise, K.; Sugawara, T.; Taniguchi, H.; Okuno, T. Phosphatidic acid produced by phospholipase D promotes RNA replication of a plant RNA virus. PLoS Pathog. 2015, 11, e1004909. [Google Scholar] [CrossRef] [PubMed]
- Villasuso, A.L.; Di Palma, M.A.; Aveldaño, M.; Pasquaré, S.J.; Racagni, G.; Giusto, N.M.; Machado, E.E. Differences in phosphatidic acid signalling and metabolism between ABA and GA treatments of barley aleurone cells. Plant Physiol. Biochem. 2013, 65, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pleskot, R.; Li, J.; Žárský, V.; Potocký, M.; Staiger, C.J. Regulation of cytoskeletal dynamics by phospholipase D and phosphatidic acid. Trends Plant Sci. 2013, 18, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Pleskot, R.; Pejchar, P.; Bezvoda, R.; Lichtscheidl, I.K.; Wolters-Arts, M.; Marc, J.; Žárský, V.; Potocký, M. Turnover of phosphatidic acid through distinct signaling pathways affects multiple aspects of pollen tube growth in tobacco. Front. Plant Sci. 2012, 3, 54. [Google Scholar] [CrossRef] [Green Version]
- Baldanzi, G.; Bettio, V.; Malacarne, V.; Graziani, A. Diacylglycerol kinases: Shaping diacylglycerol and phosphatidic acid gradients to control cell polarity. Front. Cell Dev. Biol. 2016, 4, 140. [Google Scholar] [CrossRef] [Green Version]
- Eichmann, T.O.; Lass, A. DAG tales: The multiple faces of diacylglycerol—Stereochemistry, metabolism, and signaling. Cell. Mol. Life Sci. 2015, 72, 3931–3952. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Su, Y.; Wang, X. Phosphatidic acid-mediated signaling. In Lipid-Mediated Protein Signaling; Springer: Berlin/Heidelberg, Germany, 2013; pp. 159–176. [Google Scholar]
- Wang, X.; Chapman, K.D. Lipid signaling in plants. Front. Plant Sci. 2013, 4, 216. [Google Scholar] [CrossRef] [Green Version]
- Yamagami, A.; Chieko, S.; Sakuta, M.; Shinozaki, K.; Osada, H.; Nakano, A.; Asami, T.; Nakano, T. Brassinosteroids regulate vacuolar morphology in root meristem cells of Arabidopsis thaliana. Plant Signal. Behav. 2018, 13, e1417722. [Google Scholar] [CrossRef] [Green Version]
- Derevyanchuk, M.; Kretynin, S.; Iakovenko, O.; Litvinovskaya, R.; Zhabinskii, V.; Martinec, J.; Blume, Y.; Khripach, V.; Kravets, V. Effect of 24-epibrassinolide on Brassica napus alternative respiratory pathway, guard cells movements and phospholipid signaling under salt stress. Steroids 2017, 117, 16–24. [Google Scholar] [CrossRef]
- Gómez-Merino, F.C.; Brearley, C.A.; Ornatowska, M.; Abdel-Haliem, M.E.; Zanor, M.I.; Mueller-Roeber, B. AtDGK2, a novel diacylglycerol kinase from Arabidopsis thaliana, phosphorylates 1-stearoyl-2-arachidonoyl-sn-glycerol and 1, 2-dioleoyl-sn-glycerol and exhibits cold-inducible gene expression. J. Biol. Chem. 2004, 279, 8230–8241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaz Dias, F.; Serrazina, S.; Vitorino, M.; Marchese, D.; Heilmann, I.; Godinho, M.; Rodrigues, M.; Malhó, R. A role for diacylglycerol kinase 4 in signalling crosstalk during Arabidopsis pollen tube growth. New Phytol. 2019, 222, 1434–1446. [Google Scholar] [CrossRef] [PubMed]
- Angkawijaya, A.E.; Nguyen, V.C.; Gunawan, F.; Nakamura, Y. A Pair of Arabidopsis Diacylglycerol Kinases Essential for Gametogenesis and ER Phospholipid Metabolism in Leaves and Flowers. Plant Cell 2020. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Wang, L. MpDGK2, a Novel Diacylglycerol Kinase from Malus prunifolia, Confers Drought Stress Tolerance in Transgenic Arabidopsis. Plant Mol. Biol. Rep. 2020, 1–9. [Google Scholar] [CrossRef]
- Katagiri, T.; Mizoguchi, T.; Shinozaki, K. Molecular cloning of a cDNA encoding diacylglycerol kinase (DGK) in Arabidopsis thaliana. Plant Mol. Biol. 1996, 30, 647–653. [Google Scholar] [CrossRef]
- Arana-Ceballos, F.A. Biochemical and Physiological Studies of Arabidopsis Thaliana Diacylglycerol Kinase 7 (AtDGK7). Ph.D. Thesis, University of Potsdam, Potsdam, Germany, 2006. [Google Scholar]
- Cacas, J.L.; Gerbeau-Pissot, P.; Fromentin, J.; Cantrel, C.; Thomas, D.; Jeannette, E.; Kalachova, T.; Mongrand, S.; Simon-Plas, F.; Ruelland, E. Diacylglycerol kinases activate tobacco NADPH oxidase-dependent oxidative burst in response to cryptogein. Plant Cell Environ. 2017, 40, 585–598. [Google Scholar] [CrossRef]
- Sagi, M.; Fluhr, R. Production of reactive oxygen species by plant NADPH oxidases. Plant Physiol. 2006, 141, 336–340. [Google Scholar] [CrossRef] [Green Version]
- Masai, I.; Hosoya, T.; Kojima, S.I.; Hotta, Y. Molecular cloning of a Drosophila diacylglycerol kinase gene that is expressed in the nervous system and muscle. Proc. Natl. Acad. Sci. USA 1992, 89, 6030–6034. [Google Scholar] [CrossRef] [Green Version]
- Gupta, R.S.; Epand, R.M. Phylogenetic analysis of the diacylglycerol kinase family of proteins and identification of multiple highly-specific conserved inserts and deletions within the catalytic domain that are distinctive characteristics of different classes of DGK homologs. PLoS ONE 2017, 12, e0182758. [Google Scholar] [CrossRef]
- Mérida, I.; Avila-Flores, A.; Merino, E. Diacylglycerol kinases: At the hub of cell signalling. Biochem. J. 2007, 409, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Sakane, F.; Imai, S.I.; Yamada, K.; Murakami, T.; Tsushima, S.; Kanoh, H. Alternative splicing of the human diacylglycerol kinase δ gene generates two isoforms differing in their expression patterns and in regulatory functions. J. Biol. Chem. 2002, 277, 43519–43526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakane, F.; Imai, S.I.; Kai, M.; Yasuda, S.; Kanoh, H. Diacylglycerol kinases: Why so many of them? Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2007, 1771, 793–806. [Google Scholar] [CrossRef]
- Sasaki, H.; Hozumi, Y.; Hasegawa, H.; Ito, T.; Takagi, M.; Ogino, T.; Watanabe, M.; Goto, K. Gene expression and localization of diacylglycerol kinase isozymes in the rat spinal cord and dorsal root ganglia. Cell Tissue Res. 2006, 326, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Merino, F.C.; Arana-Ceballos, F.A.; Trejo-Téllez, L.I.; Skirycz, A.; Brearley, C.A.; Dörmann, P.; Mueller-Roeber, B. Arabidopsis AtDGK7, the Smallest Member of Plant Diacylglycerol Kinases (DGKs), Displays Unique Biochemical Features and Saturates at Low Substrate Concentration the DGK Inhibitor R59022 Differentially Affects ATDGK2 and AtDGK7 Activity in vitro and Alters Plant Growth and Development. J. Biol. Chem. 2005, 280, 34888–34899. [Google Scholar] [PubMed] [Green Version]
- Ge, H.; Chen, C.; Jing, W.; Zhang, Q.; Wang, H.; Wang, R.; Zhang, W. The rice diacylglycerol kinase family: Functional analysis using transient RNA interference. Front. Plant Sci. 2012, 3, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carther, K.F.; Ketehouli, T.; Ye, N.; Yang, Y.H.; Wang, N.; Dong, Y.Y.; Yao, N.; Liu, X.M.; Liu, W.C.; Li, X.W.; et al. Comprehensive Genomic Analysis and Expression Profiling of Diacylglycerol Kinase (DGK) Gene Family in Soybean (Glycine max) under Abiotic Stresses. Int. J. Mol. Sci. 2019, 20, 1361. [Google Scholar] [CrossRef] [Green Version]
- Gu, Y.; Zhao, C.; He, L.; Yan, B.; Dong, J.; Li, Z.; Yang, K.; Xu, J. Genome-wide identification and abiotic stress responses of DGK gene family in maize. J. Plant Biochem. Biotechnol. 2018, 27, 156–166. [Google Scholar] [CrossRef]
- Sui, Z.; Niu, L.; Yue, G.; Yang, A.; Zhang, J. Cloning and expression analysis of some genes involved in the phosphoinositide and phospholipid signaling pathways from maize (Zea mays L.). Gene 2008, 426, 47–56. [Google Scholar] [CrossRef]
- Snedden, W.A.; Blumwald, E. Alternative splicing of a novel diacylglycerol kinase in tomato leads to a calmodulin-binding isoform. Plant J. 2000, 24, 317–326. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, J.; Zhang, H.; Song, F. Overexpression of a rice diacylglycerol kinase gene OsBIDK1 enhances disease resistance in transgenic tobacco. Mol. Cells Springer Sci. Bus. Media BV 2008, 26, 258–264. [Google Scholar]
- Liu, X.; Zhai, S.; Zhao, Y.; Sun, B.; Liu, C.; Yang, A.; Zhang, J. Overexpression of the phosphatidylinositol synthase gene (ZmPIS) conferring drought stress tolerance by altering membrane lipid composition and increasing ABA synthesis in maize. Plant Cell Environ. 2013, 36, 1037–1055. [Google Scholar] [CrossRef] [PubMed]
- Vaultier, M.N.; Cantrel, C.; Guerbette, F.; Boutté, Y.; Vergnolle, C.; Çiçek, D.; Bolte, S.; Zachowski, A.; Ruelland, E. The hydrophobic segment of Arabidopsis thaliana cluster I diacylglycerol kinases is sufficient to target the proteins to cell membranes. FEBS Lett. 2008, 582, 1743–1748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGrath, C.L.; Gout, J.F.; Johri, P.; Doak, T.G.; Lynch, M. Differential retention and divergent resolution of duplicate genes following whole-genome duplication. Genome Res. 2014, 24, 1665–1675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, M.; O’Hely, M.; Walsh, B.; Force, A. The probability of preservation of a newly arisen gene duplicate. Genetics 2001, 159, 1789–1804. [Google Scholar] [PubMed]
- Massart, J.; Zierath, J.R. Role of diacylglycerol kinases in glucose and energy homeostasis. Trends Endocrinol. Metab. 2019, 30, 603–617. [Google Scholar] [CrossRef]
- Kobayashi, N.; Hozumi, Y.; Ito, T.; Hosoya, T.; Kondo, H.; Goto, K. Differential subcellular targeting and activity-dependent subcellular localization of diacylglycerol kinase isozymes in transfected cells. Eur. J. Cell Biol. 2007, 86, 433–444. [Google Scholar] [CrossRef]
- Vermeer, J.E.; van Wijk, R.; Goedhart, J.; Geldner, N.; Chory, J.; Gadella, T.W., Jr.; Munnik, T. In vivo imaging of diacylglycerol at the cytoplasmic leaflet of plant membranes. Plant Cell Physiol. 2017, 58, 1196–1207. [Google Scholar] [CrossRef] [Green Version]
- Hendrix, K.W.; Assefa, H.; Boss, W.F. The polyphosphoinositides, phosphatidylinositol monophosphate and phosphatidylinositol bisphosphate, are present in nuclei isolated from carrot protoplast. Protoplasma 1989, 151, 62–72. [Google Scholar] [CrossRef]
- Xie, S.; Naslavsky, N.; Caplan, S. Diacylglycerol kinases in membrane trafficking. Cell. Logist. 2015, 5, e1078431. [Google Scholar] [CrossRef] [Green Version]
- Lundberg, G.A.; Sommarin, M. Diacylglycerol kinase in plasma membranes from wheat. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1992, 1123, 177–183. [Google Scholar] [CrossRef]
- Tan, W.J.; Yang, Y.C.; Zhou, Y.; Huang, L.P.; Xu, L.; Chen, Q.F.; Yu, L.J.; Xiao, S. Diacylglycerol Acyltransferase and Diacylglycerol KINASE modulate triacylglycerol and phosphatidic acid production in the plant response to freezing stress. Plant Physiol. 2018, 177, 1303–1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Savojardo, C.; Martelli, P.L.; Fariselli, P.; Profiti, G.; Casadio, R. BUSCA. An integrative web server to predict subcellular localization of proteins. Nucleic Acids Res. 2018, 46, W459–W466. [Google Scholar] [CrossRef] [PubMed]
- Sperschneider, J.; Catanzariti, A.M.; DeBoer, K.; Petre, B.; Gardiner, D.M.; Singh, K.B.; Dodds, P.N.; Taylor, J.M. LOCALIZER: Subcellular localization prediction of both plant and effector proteins in the plant cell. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shirai, Y.; Segawa, S.; Kuriyama, M.; Goto, K.; Sakai, N.; Saito, N. Subtype-specific translocation of diacylglycerol kinase α and γ and its correlation with protein kinase C. J. Biol. Chem. 2000, 275, 24760–24766. [Google Scholar] [CrossRef] [Green Version]
- Wissing, J.B.; Wagner, K.G. Diacylglycerol kinase from suspension cultured plant cells: Characterization and subcellular localization. Plant Physiol. 1992, 98, 1148–1153. [Google Scholar] [CrossRef] [Green Version]
- Yunus, I.S.; Cazenave-Gassiot, A.; Liu, Y.C.; Lin, Y.C.; Wenk, M.R.; Nakamura, Y. Phosphatidic acid is a major phospholipid class in reproductive organs of Arabidopsis thaliana. Plant Signal. Behav. 2015, 10, e1049790. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.C.; Meng, F.R.; Zhang, C.Y.; Zhang, N.; Sun, M.S.; Ren, J.P.; Niu, H.B.; Wang, X.; Yin, J. Comparative analysis of water stress-responsive transcriptomes in drought-susceptible and-tolerant wheat (Triticum aestivum L.). J. Plant Biol. 2012, 55, 349–360. [Google Scholar] [CrossRef]
- Möbius, K.; Kazemi, S.; Güntert, P.; Jakob, A.; Heckel, A.; Becker-Baldus, J.; Glaubitz, C. Global response of diacylglycerol kinase towards substrate binding observed by 2D and 3D MAS NMR. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Delage, E.; Ruelland, E.; Guillas, I.; Zachowski, A.; Puyaubert, J. Arabidopsis type-III phosphatidylinositol 4-kinases β1 and β2 are upstream of the phospholipase C pathway triggered by cold exposure. Plant Cell Physiol. 2012, 53, 565–576. [Google Scholar] [CrossRef]
- De Jong, C.F.; Laxalt, A.M.; Bargmann, B.O.; De Wit, P.J.; Joosten, M.H.; Munnik, T. Phosphatidic acid accumulation is an early response in the Cf-4/Avr4 interaction. Plant J. 2004, 39, 1–12. [Google Scholar] [CrossRef]
- Andersson, M.X.; Kourtchenko, O.; Dangl, J.L.; Mackey, D.; Ellerström, M. Phospholipase-dependent signalling during the AvrRpm1-and AvrRpt2-induced disease resistance responses in Arabidopsis thaliana. Plant J. 2006, 47, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Haucke, V.; di Paolo, G. Lipids and lipid modifications in the regulation of membrane traffic. Curr. Opin. Cell Biol. 2007, 19, 426–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hodgkin, M.N.; Pettitt, T.R.; Martin, A.; Michell, R.H.; Pemberton, A.J.; Wakelam, M.J. Diacylglycerols and phosphatidates: Which molecular species are intracellular messengers? Trends Biochem. Sci. 1998, 23, 200–204. [Google Scholar] [CrossRef]
- McLoughlin, F.; Arisz, S.A.; Dekker, H.L.; Kramer, G.; de Koster, C.G.; Haring, M.A.; Munnik, T.; Testerink, C. Identification of novel candidate phosphatidic acid-binding proteins involved in the salt-stress response of Arabidopsis thaliana roots. Biochem. J. 2013, 450, 573–581. [Google Scholar] [CrossRef] [Green Version]
- Muthan, B.; Roston, R.L.; Froehlich, J.E.; Benning, C. Arabidopsis chloroplast diacylglycerol pools by selectively targeting bacterial diacylglycerol kinase to suborganellar membranes. Plant Physiol. 2013, 163, 61–74. [Google Scholar] [CrossRef] [Green Version]
- Escobar-Sepúlveda, H.F.; Trejo-Téllez, L.I.; Pérez-Rodríguez, P.; Hidalgo-Contreras, J.V.; Gómez-Merino, F.C. Diacylglycerol kinases are widespread in higher plants and display inducible gene expression in response to beneficial elements, metal, and metalloid ions. Front. Plant Sci. 2017, 8, 129. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, P.; Hirsch-Hoffmann, M.; Hennig, L.; Gruissem, W. GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol. 2004, 136, 2621–2632. [Google Scholar] [CrossRef] [Green Version]
- Yuan, S.; Kim, S.C.; Deng, X.; Hong, Y.; Wang, X. Diacylglycerol kinase and associated lipid mediators modulate rice root architecture. New Phytol. 2019, 223, 261–276. [Google Scholar] [CrossRef]
- Hepler, P.K. Calcium: A central regulator of plant growth and development. Plant Cell 2005, 17, 2142–2155. [Google Scholar] [CrossRef]
- Wood, N.T. Production of a novel calmodulin-binding DGK by alternative splicing. Trends Plant Sci. 2001, 6, 50. [Google Scholar] [CrossRef]
- Gaur, R.K.; Sharma, P. Approaches to Plant Stress and Their Management; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Suhita, D.; Raghavendra, A.S.; Kwak, J.M.; Vavasseur, A. Cytoplasmic alkalization precedes reactive oxygen species production during methyl jasmonate-and abscisic acid-induced stomatal closure. Plant Physiol. 2004, 134, 1536–1545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Islam, M.M.; Munemasa, S.; Hossain, M.A.; Nakamura, Y.; Mori, I.C.; Murata, Y. Roles of AtTPC1, vacuolar two pore channel 1, in Arabidopsis stomatal closure. Plant Cell Physiol. 2010, 51, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hong, Y.; Wang, X. Phospholipase D-and phosphatidic acid-mediated signaling in plants. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2009, 1791, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Prado, A.M.; Colaço, R.; Moreno, N.; Silva, A.C.; Feijó, J.A. Targeting of pollen tubes to ovules is dependent on nitric oxide (NO) signaling. Mol. Plant 2008, 1, 703–714. [Google Scholar] [CrossRef]
- Prado, A.M.; Porterfield, D.M.; Feijó, J.A. Nitric oxide is involved in growth regulation and re-orientation of pollen tubes. Development 2004, 131, 2707–2714. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Chen, T.; Zhang, C.; Hao, H.; Liu, P.; Zheng, M.; Baluška, F.; Šamaj, J.; Lin, J. Nitric oxide modulates the influx of extracellular Ca2+ and actin filament organization during cell wall construction in Pinus bungeana pollen tubes. New Phytol. 2009, 182, 851–862. [Google Scholar] [CrossRef]
- Wong, A.; Donaldson, L.; Portes, M.T.; Eppinger, J.; Feijó, J.A.; Gehring, C. The Arabidopsis Diacylglycerol Kinase 4 is involved in nitric oxide-dependent pollen tube guidance and fertilization. Development 2020. [Google Scholar] [CrossRef]
- Alonso, R.; Rodríguez, M.C.; Pindado, J.; Merino, E.; Mérida, I.; Izquierdo, M. Diacylglycerol kinase α regulates the secretion of lethal exosomes bearing Fas ligand during activation-induced cell death of T lymphocytes. J. Biol. Chem. 2005, 280, 28439–28450. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, P.; Bleuler, S.; Laule, O.; Martin, F.; Ivanov, N.V.; Campanoni, P.; Oishi, K.; Lugon-Moulin, N.; Wyss, M.; Hruz, T.; et al. ExpressionData-A public resource of high quality curated datasets representing gene expression across anatomy, development and experimental conditions. BioData Min. 2014, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Pilon-Smits, E.A.; Quinn, C.F.; Tapken, W.; Malagoli, M.; Schiavon, M. Physiological functions of beneficial elements. Curr. Opin. Plant Biol. 2009, 12, 267–274. [Google Scholar] [CrossRef]
- Poschenrieder, C.; Cabot, C.; Martos, S.; Gallego, B.; Barceló, J. Do toxic ions induce hormesis in plants? Plant Sci. 2013, 212, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Topham, M.K.; Prescott, S.M. Diacylglycerol kinases. Diacylglycerol kinases. In Handbook of Cell Signaling; Elsevier: Amsterdam, The Netherlands, 2010; pp. 1177–1182. [Google Scholar]
- Hong, S.; Kim, S.A.; Guerinot, M.L.; McClung, C.R. Reciprocal interaction of the circadian clock with the iron homeostasis network in Arabidopsis. Plant Physiol. 2013, 161, 893–903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, L.; Nie, J.; Cao, C.; Jin, Y.; Yan, M.; Wang, F.; Liu, J.; Xiao, Y.; Liang, Y.; Zhang, W. Phosphatidic acid mediates salt stress response by regulation of MPK6 in Arabidopsis thaliana. New Phytol. 2010, 188, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Newton, A.C.; Johnson, S.N.; Gregory, P.J. Implications of climate change for diseases, crop yields and food security. Euphytica 2011, 179, 3–18. [Google Scholar] [CrossRef]
- van der Luit, A.H.; Piatti, T.; van Doorn, A.; Musgrave, A.; Felix, G.; Boller, T.; Munnik, T. Elicitation of suspension-cultured tomato cells triggers the formation of phosphatidic acid and diacylglycerol pyrophosphate. Plant Physiol. 2000, 123, 1507–1516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, S.A.; Wang, X.; Leach, J.E. Changes in the plasma membrane distribution of rice phospholipase D during resistant interactions with Xanthomonas oryzae pv oryzae. Plant Cell 1996, 8, 1079–1090. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Minami, E.; Shibuya, N. Activation of phospholipases by N-acetylchitooligosaccharide elicitor in suspension-cultured rice cells mediates reactive oxygen generation. Physiol. Plant. 2003, 118, 361–370. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Minami, E.; Ueki, J.; Shibuya, N. Elicitor-induced activation of phospholipases plays an important role for the induction of defense responses in suspension-cultured rice cells. Plant Cell Physiol. 2005, 46, 579–587. [Google Scholar] [CrossRef]
- Suzuki, K.; Yano, A.; Nishiuchi, T.; Nakano, T.; Kodama, H.; Yamaguchi, K.; Shinshi, H. Comprehensive analysis of early response genes to two different microbial elicitors in tobacco cells. Plant Sci. 2007, 173, 291–301. [Google Scholar] [CrossRef]
- De Torres Zabela, M.; Fernandez-Delmond, I.; Niittyla, T.; Sanchez, P.; Grant, M. Differential expression of genes encoding Arabidopsis phospholipases after challenge with virulent or avirulent Pseudomonas isolates. Mol. Plant-Microbe Interact. 2002, 15, 808–816. [Google Scholar] [CrossRef] [Green Version]
- Kalachova, T.; Puga-Freitas, R.; Kravets, V.; Soubigou-Taconnat, L.; Repellin, A.; Balzergue, S.; Zachowski, A.; Ruelland, E. The inhibition of basal phosphoinositide-dependent phospholipase C activity in Arabidopsis suspension cells by abscisic or salicylic acid acts as a signalling hub accounting for an important overlap in transcriptome remodelling induced by these hormones. Environ. Exp. Bot. 2016, 123, 37–49. [Google Scholar] [CrossRef]
- Toyoda, K.; Kawahara, T.; Ichinose, Y.; Yamada, T.; Shiraishi, T. Potentiation of phytoalexin accumulation in elicitor-treated epicotyls of pea (Pisum sativum) by a diacylglycerol kinase inhibitor. J. Phytopathol. 2000, 148, 633–636. [Google Scholar]
- Domingos, P.; Prado, A.M.; Wong, A.; Gehring, C.; Feijo, J.A. Nitric oxide: A multitasked signaling gas in plants. Mol. Plant 2015, 8, 506–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sousa, E.; Kost, B.; Malhó, R. Arabidopsis phosphatidylinositol-4-monophosphate 5-kinase 4 regulates pollen tube growth and polarity by modulating membrane recycling. Plant Cell 2008, 20, 3050–3064. [Google Scholar] [CrossRef] [Green Version]
- Elmore, J.M.; Liu, J.; Smith, B.; Phinney, B.; Coaker, G. Quantitative proteomics reveals dynamic changes in the plasma membrane during Arabidopsis immune signaling. Mol. Cell. Proteom. 2012, 11. [Google Scholar] [CrossRef] [Green Version]
- Laxalt, A.M.; Munnik, T. Phospholipid signalling in plant defence. Curr. Opin. Plant Biol. 2002, 5, 332–338. [Google Scholar] [CrossRef]
- Den Hartog, M.; Musgrave, A.; Munnik, T. Nod factor-induced phosphatidic acid and diacylglycerol pyrophosphate formation: A role for phospholipase C and D in root hair deformation. Plant J. 2001, 25, 55–65. [Google Scholar] [CrossRef]
- Den Hartog, M.; Verhoef, N.; Munnik, T. Nod factor and elicitors activate different phospholipid signaling pathways in suspension-cultured alfalfa cells. Plant Physiol. 2003, 132, 311–317. [Google Scholar] [CrossRef] [Green Version]
- Xiong, J.; Zhang, L.; Fu, G.; Yang, Y.; Zhu, C.; Tao, L. Drought-induced proline accumulation is uninvolved with increased nitric oxide, which alleviates drought stress by decreasing transpiration in rice. J. Plant Res. 2012, 125, 155–164. [Google Scholar] [CrossRef]
- Vierling, E. The roles of heat shock proteins in plants. Annu. Rev. Plant Biol. 1991, 42, 579–620. [Google Scholar] [CrossRef]
- Lipiec, J.; Doussan, C.; Nosalewicz, A.; Kondracka, K. Effect of drought and heat stresses on plant growth and yield: A review. Int. Agrophys. 2013, 27, 463–477. [Google Scholar] [CrossRef]
- Banerjee, A.; Roychoudhury, A. Group II late embryogenesis abundant (LEA) proteins: Structural and functional aspects in plant abiotic stress. Plant Growth Regul. 2016, 79, 1–17. [Google Scholar] [CrossRef]
- Sakamoto, A.; Murata, N. The role of glycine betaine in the protection of plants from stress: Clues from transgenic plants. Plant Cell Environ. 2002, 25, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Murata, N. Glycinebetaine protects plants against abiotic stress: Mechanisms and biotechnological applications. Plant Cell Environ. 2011, 34, 1–20. [Google Scholar] [CrossRef]
- Finkelstein, R. Abscisic acid synthesis and response. Arab. Book Am. Soc. Plant Biol. 2013, 11. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Dong, W.; Zhang, N.; Ai, X.; Wang, M.; Huang, Z.; Xiao, L.; Xia, G. A wheat allene oxide cyclase gene enhances salinity tolerance via jasmonate signaling. Plant Physiol. 2014, 164, 1068–1076. [Google Scholar] [CrossRef] [Green Version]
- Bohnert, H.J.; Nelson, D.E.; Jensen, R.G. Adaptations to environmental stresses. Plant Cell 1995, 7, 1099. [Google Scholar] [CrossRef]
- Juszczak, I.; Bartels, D. LEA gene expression, RNA stability and pigment accumulation in three closely related Linderniaceae species differing in desiccation tolerance. Plant Sci. 2017, 255, 59–71. [Google Scholar] [CrossRef]
- Bargmann, B.O.; Laxalt, A.M.; Riet, B.T.; Van Schooten, B.; Merquiol, E.; Testerink, C.; Haring, M.A.; Bartels, D.; Munnik, T. Multiple PLDs required for high salinity and water deficit tolerance in plants. Plant Cell Physiol. 2008, 50, 78–89. [Google Scholar] [CrossRef] [Green Version]
- Gasulla, F.; vom Dorp, K.; Dombrink, I.; Zähringer, U.; Gisch, N.; Dörmann, P.; Bartels, D. The role of lipid metabolism in the acquisition of desiccation tolerance in C raterostigma plantagineum: A comparative approach. Plant J. 2013, 75, 726–741. [Google Scholar] [CrossRef]
- Drøbak, B.K.; Watkins, P.A. Inositol (1, 4, 5) trisphosphate production in plant cells: An early response to salinity and hyperosmotic stress. FEBS Lett. 2000, 481, 240–244. [Google Scholar] [CrossRef]
- Savchenko, T.; Kolla, V.A.; Wang, C.Q.; Nasafi, Z.; Hicks, D.R.; Phadungchob, B.; Chehab, W.E.; Brandizzi, F.; Froehlich, J.; Dehesh, K. Functional convergence of oxylipin and abscisic acid pathways controls stomatal closure in response to drought. Plant Physiol. 2014, 164, 1151–1160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ng, C.K.; Carr, K.; McAinsh, M.R.; Powell, B.; Hetherington, A.M. Drought-induced guard cell signal transduction involves sphingosine-1-phosphate. Nature 2001, 410, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Topham, M.K. Signaling roles of diacylglycerol kinases. J. Cell. Biochem. 2006, 97, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Chakradhar, T.; Reddy, R.A.; Chandrasekhar, T. Protein Kinases and Phosphatases in Stress Transduction: Role in Crop Improvement. In Plant Signaling Molecules; Elsevier: Amsterdam, The Netherlands, 2019; pp. 533–547. [Google Scholar]
- Distéfano, A.M.; Scuffi, D.; García-Mata, C.; Lamattina, L.; Laxalt, A.M. Phospholipase Dδ is involved in nitric oxide-induced stomatal closure. Planta 2012, 236, 1899–1907. [Google Scholar] [CrossRef]
- Distefano, A.M.; GARCÍA-MATACA, C.; Lamattina, L.; Laxalt, A.M. Nitric oxide-induced phosphatidic acid accumulation: A role for phospholipases C and D in stomatal closure. Plant Cell Environ. 2008, 31, 187–194. [Google Scholar] [CrossRef]
- Guo, L.; Devaiah, S.P.; Narasimhan, R.; Pan, X.; Zhang, Y.; Zhang, W.; Wang, X. Cytosolic glyceraldehyde-3-phosphate dehydrogenases interact with phospholipase Dδ to transduce hydrogen peroxide signals in the Arabidopsis response to stress. Plant Cell 2012, 24, 2200–2212. [Google Scholar] [CrossRef] [Green Version]
- Villasuso, A.L.; Molas, M.L.; Racagni, G.; Abdala, G.; Machado-Domenech, E. Gibberellin signal in barley aleurone: Early activation of PLC by G protein mediates amylase secretion. Plant Growth Regul. 2003, 41, 197–205. [Google Scholar] [CrossRef]
- Daszkowska-Golec, A. The role of abscisic acid in drought stress: How aba helps plants to cope with drought stress. In Drought Stress Tolerance in Plants; Springer: Berlin/Heidelberg, Germany, 2016; Volume 2, pp. 123–151. [Google Scholar]
- Djafi, N.; Vergnolle, C.; Cantrel, C.; Wietrzyński, W.; Delage, E.; Cochet, F.; Puyaubert, J.; Soubigou-Taconnat, L.; Gey, D.; Collin, S.; et al. The Arabidopsis DREB2 genetic pathway is constitutively repressed by basal phosphoinositide-dependent phospholipase C coupled to diacylglycerol kinase. Front. Plant Sci. 2013, 4, 307. [Google Scholar] [CrossRef] [Green Version]
- Fowler, S.; Thomashow, M.F. Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 2002, 14, 1675–1690. [Google Scholar] [CrossRef] [Green Version]
- Thomashow, M.F. Plant cold acclimation: Freezing tolerance genes and regulatory mechanisms. Annu. Rev. Plant Biol. 1999, 50, 571–599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Buskirk, H.A.; Thomashow, M.F. Arabidopsis transcription factors regulating cold acclimation. Physiol. Plant. 2006, 126, 72–80. [Google Scholar] [CrossRef]
- Amir, R.; Hussain, S.; Noor-ul-Ain, H.; Hussain, A.; Yun, B.W. ROS Mediated Plant Defense against Abiotic Stresses. In Plant Biotechnology: Progress in Genomic Era; Springer: Berlin/Heidelberg, Germany, 2019; pp. 481–515. [Google Scholar]
- Lv, X.; Li, H.; Chen, X.; Xiang, X.; Guo, Z.; Yu, J.; Zhou, Y. The role of calcium-dependent protein kinase in hydrogen peroxide, nitric oxide and ABA-dependent cold acclimation. J. Exp. Bot. 2018, 69, 4127–4139. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Schumaker, K.S.; Zhu, J.-K. Cell signaling during cold, drought, and salt stress. Plant Cell 2002, 14 (Suppl. 1), S165–S183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirayama, T.; Ohto, C.; Mizoguchi, T.; Shinozaki, K. A gene encoding a phosphatidylinositol-specific phospholipase C is induced by dehydration and salt stress in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 1995, 92, 3903–3907. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Li, M.; Zhang, W.; Welti, R.; Wang, X. The plasma membrane–bound phospholipase Dδ enhances freezing tolerance in Arabidopsis thaliana. Nat. Biotechnol. 2004, 22, 427–433. [Google Scholar] [CrossRef]
- Lee, B.-H.; Henderson, D.A.; Zhu, J.-K. The Arabidopsis cold-responsive transcriptome and its regulation by ICE1. Plant Cell 2005, 17, 3155–3175. [Google Scholar] [CrossRef] [Green Version]
- Gilmour, S.J.; Thomashow, M.F. Cold acclimation and cold-regulated gene expression in ABA mutants of Arabidopsis thaliana. Plant Mol. Biol. 1991, 17, 1233–1240. [Google Scholar] [CrossRef]
- Mantyla, E.; Lang, V.; Palva, E.T. Role of abscisic acid in drought-induced freezing tolerance, cold acclimation, and accumulation of LT178 and RAB18 proteins in Arabidopsis thaliana. Plant Physiol. 1995, 107, 141–148. [Google Scholar] [CrossRef] [Green Version]
- Brandt, B.; Munemasa, S.; Wang, C.; Nguyen, D.; Yong, T.; Yang, P.G.; Poretsky, E.; Belknap, T.F.; Waadt, R.; Alemán, F.; et al. Calcium specificity signaling mechanisms in abscisic acid signal transduction in Arabidopsis guard cells. Elife 2015, 4, e03599. [Google Scholar] [CrossRef]
- Huang, X.; Shi, H.; Hu, Z.; Liu, A.; Amombo, E.; Chen, L.; Fu, J. ABA is involved in regulation of cold stress response in bermudagrass. Front. Plant Sci. 2017, 8, 1613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, M.; Zhang, J. Water stress-induced abscisic acid accumulation triggers the increased generation of reactive oxygen species and up-regulates the activities of antioxidant enzymes in maize leaves. J. Exp. Bot. 2002, 53, 2401–2410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, X.; Jiang, M.; Zhang, J.; Zhang, A.; Lin, F.; Tan, M. Calcium–calmodulin is required for abscisic acid-induced antioxidant defense and functions both upstream and downstream of H2O2 production in leaves of maize (Zea mays) plants. New Phytol. 2007, 173, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Neill, S.; Barros, R.; Bright, J.; Desikan, R.; Hancock, J.; Harrison, J.; Morris, P.; Ribeiro, D.; Wilson, I. Nitric oxide, stomatal closure, and abiotic stress. J. Exp. Bot. 2008, 59, 165–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, A.; Jiang, M.; Zhang, J.; Tan, M.; Hu, X. Mitogen-activated protein kinase is involved in abscisic acid-induced antioxidant defense and acts downstream of reactive oxygen species production in leaves of maize plants. Plant Physiol. 2006, 141, 475–487. [Google Scholar] [CrossRef] [Green Version]
- Xing, Y.; Jia, W.; Zhang, J. AtMKK1 mediates ABA-induced CAT1 expression and H2O2 production via AtMPK6-coupled signaling in Arabidopsis. Plant J. 2008, 54, 440–451. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Yu, X.C.; Wang, X.J.; Zhao, R.; Li, Y.; Fan, R.C.; Shang, Y.; Du, S.Y.; Wang, X.F.; Wu, F.Q.; et al. Two calcium-dependent protein kinases, CPK4 and CPK11, regulate abscisic acid signal transduction in Arabidopsis. Plant Cell 2007, 19, 3019–3036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, Y.; Cao, J.; Ni, L.; Zhu, Y.; Zhang, A.; Tan, M.; Jiang, M. ZmCPK11 is involved in abscisic acid-induced antioxidant defence and functions upstream of ZmMPK5 in abscisic acid signalling in maize. J. Exp. Bot. 2012, 64, 871–884. [Google Scholar] [CrossRef] [Green Version]
- Zou, J.J.; Li, X.D.; Ratnasekera, D.; Wang, C.; Liu, W.X.; Song, L.F.; Zhang, W.Z.; Wu, W.H. Arabidopsis CALCIUM-DEPENDENT PROTEIN KINASE8 and CATALASE3 function in abscisic acid-mediated signaling and H2O2 homeostasis in stomatal guard cells under drought stress. Plant Cell 2015, 27, 1445–1460. [Google Scholar] [CrossRef] [Green Version]
- Vaultier, M.N.; Cantrel, C.; Vergnolle, C.; Justin, A.M.; Demandre, C.; Benhassaine-Kesri, G.; Çiçek, D.; Zachowski, A.; Ruelland, E. Desaturase mutants reveal that membrane rigidification acts as a cold perception mechanism upstream of the diacylglycerol kinase pathway in Arabidopsis cells. FEBS Lett. 2006, 580, 4218–4223. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Carlsson, A.S.; Francis, T.; Zhang, M.; Hoffman, T.; Giblin, M.E.; Taylor, D.C. Triacylglycerol synthesis by PDAT1 in the absence of DGAT1 activity is dependent on re-acylation of LPC by LPCAT2. BMC Plant Biol. 2012, 12, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Q.F.; Xu, L.; Tan, W.J.; Chen, L.; Qi, H.; Xie, L.J.; Chen, M.X.; Liu, B.Y.; Yu, L.J.; Yao, N.; et al. Disruption of the Arabidopsis defense regulator genes SAG101, EDS1, and PAD4 confers enhanced freezing tolerance. Mol. Plant 2015, 8, 1536–1549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vergnolle, C.; Vaultier, M.N.; Taconnat, L.; Renou, J.P.; Kader, J.C.; Zachowski, A.; Ruelland, E. The cold-induced early activation of phospholipase C and D pathways determines the response of two distinct clusters of genes in Arabidopsis cell suspensions. Plant Physiol. 2005, 139, 1217–1233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, W.-J.; Xiao, S.; Chen, Q.-F. Potential role of salicylic acid in modulating diacylglycerol homeostasis in response to freezing temperatures in Arabidopsis. Plant Signal. Behav. 2015, 10, e1082698. [Google Scholar] [CrossRef] [Green Version]
- Rozema, J.; Schat, H. Salt tolerance of halophytes, research questions reviewed in the perspective of saline agriculture. Environ. Exp. Bot. 2013, 92, 83–95. [Google Scholar] [CrossRef] [Green Version]
- Reddy, P.S.; Jogeswar, G.; Rasineni, G.K.; Maheswari, M.; Reddy, A.R.; Varshney, R.K.; Kishor, P.K. Proline over-accumulation alleviates salt stress and protects photosynthetic and antioxidant enzyme activities in transgenic sorghum [Sorghum bicolor (L.) Moench]. Plant Physiol. Biochem. 2015, 94, 104–113. [Google Scholar] [CrossRef] [Green Version]
- Meringer, M.V.; Villasuso, A.L.; Margutti, M.P.; Usorach, J.; Pasquaré, S.J.; Giusto, N.M.; Machado, E.E.; Racagni, G.E. Saline and osmotic stresses stimulate PLD/diacylglycerol kinase activities and increase the level of phosphatidic acid and proline in barley roots. Environ. Exp. Bot. 2016, 128, 69–78. [Google Scholar] [CrossRef]
- Hou, Q.; Ufer, G.; Bartels, D. Lipid signalling in plant responses to abiotic stress. Plant Cell Environ. 2016, 39, 1029–1048. [Google Scholar] [CrossRef]
- Julkowska, M.M.; Testerink, C. Tuning plant signaling and growth to survive salt. Trends Plant Sci. 2015, 20, 586–594. [Google Scholar] [CrossRef]
- Meringer, M.V.; Villasuso, A.L.; Pasquaré, S.J.; Giusto, N.M.; Machado, E.E.; Racagni, G.E. Comparative phytohormone profiles, lipid kinase and lipid phosphatase activities in barley aleurone, coleoptile, and root tissues. Plant Physiol. Biochem. 2012, 58, 83–88. [Google Scholar] [CrossRef]
- McLoughlin, F.; Galvan-Ampudia, C.S.; Julkowska, M.M.; Caarls, L.; van der Does, D.; Laurière, C.; Munnik, T.; Haring, M.A.; Testerink, C. The Snf1-related protein kinases SnRK2. 4 and SnRK2. 10 are involved in maintenance of root system architecture during salt stress. Plant J. 2012, 72, 436–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsugane, K.; Kobayashi, K.; Niwa, Y.; Ohba, Y.; Wada, K.; Kobayashi, H. A recessive Arabidopsis mutant that grows photoautotrophically under salt stress shows enhanced active oxygen detoxification. Plant Cell 1999, 11, 1195–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernández, J.A.; Ferrer, M.A.; Jiménez, A.; Barceló, A.R.; Sevilla, F. Antioxidant systems and O2.−/H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 2001, 127, 817–831. [Google Scholar] [CrossRef]
- Isayenkov, S. Physiological and molecular aspects of salt stress in plants. Cytol. Genet. 2012, 46, 302–318. [Google Scholar] [CrossRef] [Green Version]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [Green Version]
- Flowers, T. Improving crop salt tolerance. J. Exp. Bot. 2004, 55, 307–319. [Google Scholar] [CrossRef]
- Munns, R. Genes and salt tolerance: Bringing them together. New Phytol. 2005, 167, 645–663. [Google Scholar] [CrossRef]
- Ashraf, M.; Akram, N.A. Improving salinity tolerance of plants through conventional breeding and genetic engineering: An analytical comparison. Biotechnol. Adv. 2009, 27, 744–752. [Google Scholar] [CrossRef]
- Agarwal, P.K.; Shukla, P.S.; Gupta, K.; Jha, B. Bioengineering for salinity tolerance in plants: State of the art. Mol. Biotechnol. 2013, 54, 102–123. [Google Scholar] [CrossRef]
- Baena-González, E.; Rolland, F.; Thevelein, J.M.; Sheen, J. A central integrator of transcription networks in plant stress and energy signalling. Nature 2007, 448, 938–942. [Google Scholar] [CrossRef]
- Jaspers, P.; Kangasjärvi, J. Reactive oxygen species in abiotic stress signaling. Physiol. Plant. 2010, 138, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.A.; Suzuki, N.; Ciftci-Yilmaz, S.U.; Mittler, R.O. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lee, B.H.; Dellinger, M.; Cui, X.; Zhang, C.; Wu, S.; Nothnagel, E.A.; Zhu, J.K. A cellulose synthase-like protein is required for osmotic stress tolerance in Arabidopsis. Plant J. 2010, 63, 128–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darwish, E.; Testerink, C.; Khalil, M.; El-Shihy, O.; Munnik, T. Phospholipid signaling responses in salt-stressed rice leaves. Plant Cell Physiol. 2009, 50, 986–997. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez, J.-P.; Chua, N.-H. Arabidopsis PLC1 is required for secondary responses to abscisic acid signals. Plant Cell 2001, 13, 1143–1154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arisz, S.A.; Munnik, T. The salt stress-induced LPA response in Chlamydomonas is produced via PLA2 hydrolysis of DGK-generated phosphatidic acid. J. Lipid Res. 2011, 52, 2012–2020. [Google Scholar] [CrossRef] [Green Version]
- König, S.; Mosblech, A.; Heilmann, I. Stress-inducible and constitutive phosphoinositide pools have distinctive fatty acid patterns in Arabidopsis thaliana. FASEB J. 2007, 21, 1958–1967. [Google Scholar] [CrossRef] [Green Version]
- Gendron, J.M.; Liu, J.S.; Fan, M.; Bai, M.Y.; Wenkel, S.; Springer, P.S.; Barton, M.K.; Wang, Z.Y. Brassinosteroids regulate organ boundary formation in the shoot apical meristem of Arabidopsis. Proc. Natl. Acad. Sci. USA 2012, 109, 21152–21157. [Google Scholar] [CrossRef] [Green Version]
- González-García, M.P.; Vilarrasa-Blasi, J.; Zhiponova, M.; Divol, F.; Mora-García, S.; Russinova, E.; Caño-Delgado, A.I. Brassinosteroids control meristem size by promoting cell cycle progression in Arabidopsis roots. Development 2011, 138, 849–859. [Google Scholar] [CrossRef] [Green Version]
- Lucini, L.; Rouphael, Y.; Cardarelli, M.; Bonini, P.; Baffi, C.; Colla, G. A vegetal biopolymer-based biostimulant promoted root growth in melon while triggering brassinosteroids and stress-related compounds. Front. Plant Sci. 2018, 9, 472. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Yang, Q.; Wang, Y.; Wang, L.; Fu, Y.; Wang, X. Brassinosteroids regulate pavement cell growth by mediating BIN2-induced microtubule stabilization. J. Exp. Bot. 2018, 69, 1037–1049. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Gao, H.B.; Zhang, L.L.; Xue, H.W.; Lin, W.H. Phosphatidic acid regulates BZR1 activity and brassinosteroid signal of Arabidopsis. Mol. Plant 2014, 7, 445–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kagale, S.; Divi, U.K.; Krochko, J.E.; Keller, W.A.; Krishna, P. Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta 2007, 225, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Sharma, I.; Kaur, N.; Pati, P.K. Brassinosteroids: A promising option in deciphering remedial strategies for abiotic stress tolerance in rice. Front. Plant Sci. 2017, 8, 2151. [Google Scholar] [CrossRef] [PubMed]
- Derevyanchuk, M.; Kretynin, S.; Kolesnikov, Y.; Litvinovskaya, R.; Martinec, J.; Khripach, V.; Kravets, V. Seed germination, respiratory processes and phosphatidic acid accumulation in Arabidopsis diacylglycerol kinase knockouts–The effect of brassinosteroid, brassinazole and salinity. Steroids 2019, 147, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Shameer, K.; Ambika, S.; Varghese, S.M.; Karaba, N.; Udayakumar, M.; Sowdhamini, R. STIFDB—Arabidopsis stress responsive transcription factor dataBase. Int. J. Plant Genom. 2009, 2009. [Google Scholar] [CrossRef] [Green Version]
- LeDuc, D.L.; AbdelSamie, M.; Móntes-Bayon, M.; Wu, C.P.; Reisinger, S.J.; Terry, N. Overexpressing both ATP sulfurylase and selenocysteine methyltransferase enhances selenium phytoremediation traits in Indian mustard. Environ. Pollut. 2006, 144, 70–76. [Google Scholar] [CrossRef]
- Singh, S.; Parihar, P.; Singh, R.; Singh, V.P.; Prasad, S.M. Heavy metal tolerance in plants: Role of transcriptomics, proteomics, metabolomics, and ionomics. Front. Plant Sci. 2016, 6, 1143. [Google Scholar] [CrossRef] [Green Version]
- Calabrese, E.J. Hormesis: A fundamental concept in biology. Microb. Cell 2014, 1, 145. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, M.E.; Castro, P.R.; Azevedo, R.A. Hormesis in plants under Cd exposure: From toxic to beneficial element? J. Hazard. Mater. 2020, 384, 121–434. [Google Scholar] [CrossRef]
- Barker, A.V.; Pilbeam, D.J. Handbook of Plant Nutrition; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Hannun, Y.A.; Obeid, L.M. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Van Blitterswijk, W.J.; Houssa, B. Properties and functions of diacylglycerol kinases. Cell. Signal. 2000, 12, 595–605. [Google Scholar] [CrossRef]



| Genes | Nucleus | Cytoplasm | Peroxisome | Cell Membrane | Mitochondrion | Lysosome | Golgi Apparatus | Endoplasmic Reticulum | Extra Cellular | Plastid |
|---|---|---|---|---|---|---|---|---|---|---|
| GmDGK2 | 0.0002 | 0 | 0.0001 | 0.7858 | 0.0008 | 0.0152 | 0.0935 | 0.1044 | 0 | 0 |
| GmDGK11 | 0.0046 | 0.0004 | 0.0112 | 0.4733 | 0.0278 | 0.0728 | 0.1536 | 0.2536 | 0.001 | 0.0017 |
| GmDGK12 | 0.0013 | 0.0003 | 0.0013 | 0.6059 | 0.0041 | 0.0395 | 0.1734 | 0.1736 | 0.0001 | 0.0006 |
| AtDGK1 | 0.0003 | 0 | 0.0002 | 0.8015 | 0.0004 | 0.0044 | 0.1111 | 0.0821 | 0.0001 | 0 |
| ATDGK2 | 0.0001 | 0 | 0 | 0.7834 | 0.0001 | 0.0007 | 0.1733 | 0.0422 | 0 | 0 |
| GmDGK5 | 0.0781 | 0.5242 | 0.1985 | 0.0734 | 0.0589 | 0.0187 | 0.0095 | 0.0266 | 0.0055 | 0.0066 |
| GmDGK6 | 0.2288 | 0.6091 | 0.0339 | 0.0066 | 0.0414 | 0.0133 | 0.0105 | 0.0124 | 0.0088 | 0.0351 |
| GmDGK7 | 0.2419 | 0.4886 | 0.034 | 0.0586 | 0.092 | 0.0273 | 0.0196 | 0.0254 | 0.0053 | 0.0072 |
| GmDGK10 | 0.463 | 0.3372 | 0.0123 | 0.0482 | 0.0688 | 0.0125 | 0.0087 | 0.0046 | 0.0067 | 0.0379 |
| AtDGK3 | 0.4414 | 0.2774 | 0.1085 | 0.0666 | 0.0377 | 0.0174 | 0.0164 | 0.0151 | 0.0105 | 0.009 |
| AtDGK4 | 0.3848 | 0.3591 | 0.0154 | 0.1309 | 0.008 | 0.043 | 0.0172 | 0.0117 | 0.0279 | 0.0021 |
| AtDGK7 | 0.5091 | 0.3567 | 0.0071 | 0.0567 | 0.0048 | 0.0175 | 0.0139 | 0.0112 | 0.0222 | 0.0001 |
| GmDGK1 | 0.227 | 0.4039 | 0.1129 | 0.0527 | 0.1044 | 0.0145 | 0.0408 | 0.0241 | 0.0071 | 0.0126 |
| GmDGK3 | 0.2122 | 0.3607 | 0.2087 | 0.0677 | 0.0782 | 0.0097 | 0.0187 | 0.0174 | 0.0091 | 0.0187 |
| GmDGK4 | 0.212 | 0.49 | 0.112 | 0.0663 | 0.0553 | 0.0173 | 0.014 | 0.0114 | 0.0109 | 0.0108 |
| GmDGK8 | 0.2563 | 0.5964 | 0.0152 | 0.0569 | 0.021 | 0.0169 | 0.0117 | 0.0089 | 0.013 | 0.0036 |
| GmDGK9 | 0.2012 | 0.5483 | 0.0758 | 0.0989 | 0.0142 | 0.0202 | 0.0154 | 0.0096 | 0.0141 | 0.0022 |
| AtDGK5 | 0.1692 | 0.6145 | 0.0836 | 0.0434 | 0.0313 | 0.0186 | 0.0154 | 0.0112 | 0.0087 | 0.004 |
| AtDGK6 | 0.287 | 0.6074 | 0.0077 | 0.0558 | 0.0059 | 0.0143 | 0.0076 | 0.0043 | 0.0081 | 0.00019 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kue Foka, I.C.; Ketehouli, T.; Zhou, Y.; Li, X.-W.; Wang, F.-W.; Li, H. The Emerging Roles of Diacylglycerol Kinase (DGK) in Plant Stress Tolerance, Growth, and Development. Agronomy 2020, 10, 1375. https://doi.org/10.3390/agronomy10091375
Kue Foka IC, Ketehouli T, Zhou Y, Li X-W, Wang F-W, Li H. The Emerging Roles of Diacylglycerol Kinase (DGK) in Plant Stress Tolerance, Growth, and Development. Agronomy. 2020; 10(9):1375. https://doi.org/10.3390/agronomy10091375
Chicago/Turabian StyleKue Foka, Idrice Carther, Toi Ketehouli, Yonggang Zhou, Xiao-Wei Li, Fa-Wei Wang, and Haiyan Li. 2020. "The Emerging Roles of Diacylglycerol Kinase (DGK) in Plant Stress Tolerance, Growth, and Development" Agronomy 10, no. 9: 1375. https://doi.org/10.3390/agronomy10091375
APA StyleKue Foka, I. C., Ketehouli, T., Zhou, Y., Li, X.-W., Wang, F.-W., & Li, H. (2020). The Emerging Roles of Diacylglycerol Kinase (DGK) in Plant Stress Tolerance, Growth, and Development. Agronomy, 10(9), 1375. https://doi.org/10.3390/agronomy10091375





