The Seed–Seedling Transition in Commercial Soybean Cultivars with the Presence of Greenish Seeds in the Sample: A Perspective from Classical Genetic Parameters
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seed Production and Sample Processing
2.2. Seed–Seedling Transition
2.2.1. Seed Germination
2.2.2. Phenotyping of Seedlings
2.2.3. Seedling Growth
2.3. Hierarchical Model or Nested Analysis
2.4. Genetic Parameters Associated with Seed–Seedling Transition Characterization
2.5. Statistical Analysis
3. Results
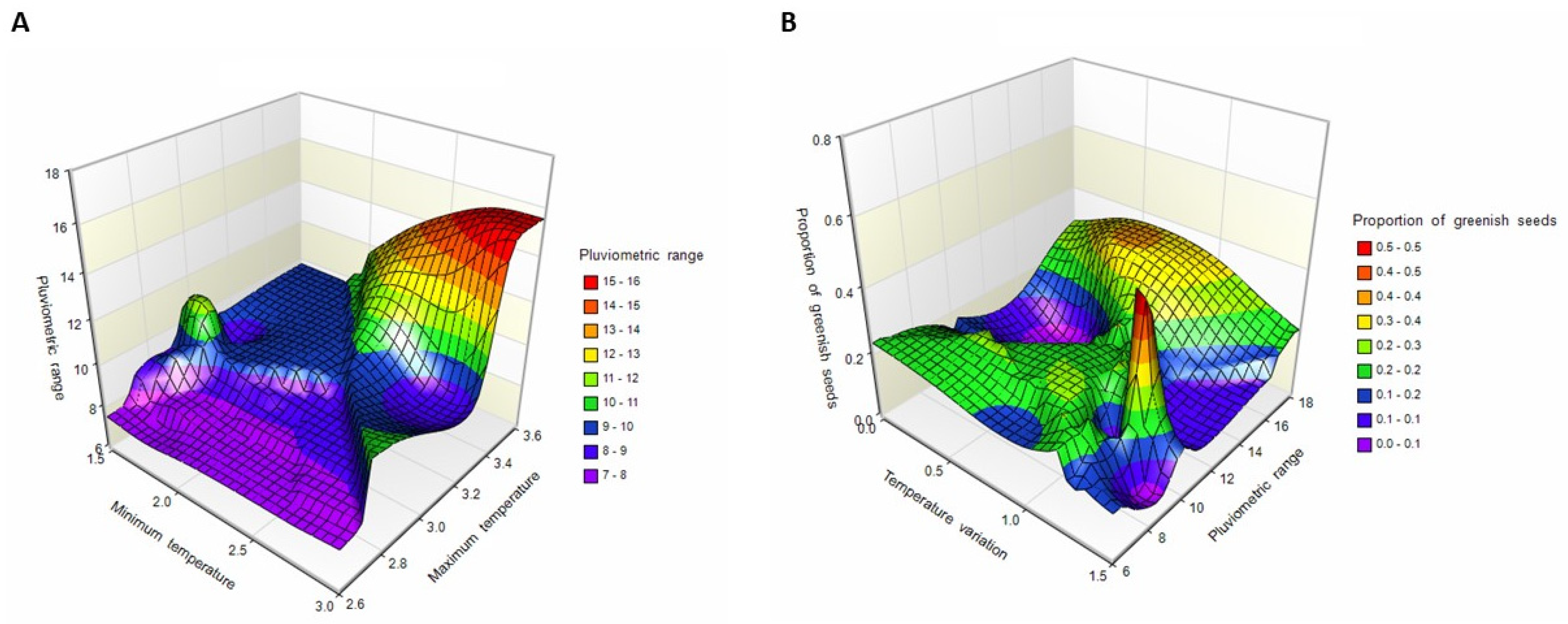
3.1. Seed Production and Maternal Environment
3.2. Seed–Seedling Transition and Classical Genetic Parameters
3.3. Relationship between the Seed–Seedling Transition Measurements
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Pádua, G.P.; França-Neto, J.D.B.; de Carvalho, M.L.M.; Costa, O.; Krzyzanowski, F.C.; Costa, N.P. Da Tolerance Level of Green Seed in Soybean Seed Lots after Storage. Rev. Bras. Sementes 2007, 29, 128–138. [Google Scholar] [CrossRef] [Green Version]
- De Pádua, G.P.; França-Neto, J.D.B.; De Carvalho, M.L.M.; Krzyzanowski, F.C.; Guimarães, R.M. Incidence of Green Soybean Seeds as a Function of Environmental Stresses during Seed Maturation. Rev. Bras. Sementes 2009, 31, 150–159. [Google Scholar] [CrossRef]
- Zorato, M.d.F.; Peske, S.T.; Takeda, C.; França Neto, J. de B. Presença de Sementes Esverdeadas Em Soja e Seus Efeitos Sobre Seu Potencial Fisiológico. Rev. Bras. Sementes 2007, 29, 11–19. [Google Scholar] [CrossRef] [Green Version]
- de Pádua, G.P.; de Carvalho, M.L.M.; França-Neto, J.D.B.; Guerreiro, M.C.; Guimarães, R.M. Response of Soybean Genotypes to the Expression of Green Seed under Temperature and Water Stresses. Rev. Bras. Sement. 2009, 31, 140–149. [Google Scholar] [CrossRef]
- Rangel, M.A.S.; Minuzzi, A.; Pierezan, L.; Teodósio, T.K.C.; Ono, F.B.; Cardoso, P.C. Presença e Qualidade de Sementes Esverdeadas de Soja Na Região Sul Do Estado Do Mato Grosso Do Sul. Acta Sci. Agron. 2011, 33, 127–132. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, S.B.; Da Silva, J.G.; Meneguzzo, M.R.R.; Martins, A.B.N.; Meneghello, G.E.; De Tunes, L.V.M. Green Soybean Seeds: Effect on Physiological Quality. Ciência Rural. 2020, 50. [Google Scholar] [CrossRef] [Green Version]
- Werner, F.; Junior, A.A.B.; Ferreira, A.S.; Silva, M.A.A.E.; Mandarino, J.M.G.; Zucareli, C. Size, Chlorophyll Retention and Protein and Oil Contents of Grains from Soybean Plants Grown in Different Spatial Arrangements. Semin. Cienc Agrar. 2017, 38, 85. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, R.N.; Ligterink, W.; França-Neto, J.d.B.; Hilhorst, H.W.; da Silva, E.A.A. Gene Expression Profiling of the Green Seed Problem in Soybean. BMC Plant Biol. 2016, 16, 37. [Google Scholar] [CrossRef] [Green Version]
- De Pádua, G.P.; França-Neto, J.D.B.; Rossi, R.F.; Cândido, H.G. Agroclimatic Zoning of the State of Minas Gerais for the Production of High Quality Soybean Seeds. J. Seed Sci. 2014, 36, 413–418. [Google Scholar] [CrossRef] [Green Version]
- França-Neto, J.B.; Kryzanowski, F.C.; Henning, A.A.; Pádua, G.P.; Lorini, I.; Henning, F.A. Tecnologia Da Produção de Semente de Soja de Alta Qualidade. Inf. ABRATES 2010, 20, 20–32. [Google Scholar]
- Mandarino, J.M.G. Grãos Verdes: Influência Na Qualidade Dos Produtos à Base de Soja; EMBRAPA: Londrina, SC, Brazil, 2012. [Google Scholar]
- van Wart, J.; van Bussel, L.G.J.; Wolf, J.; Licker, R.; Grassini, P.; Nelson, A.; Boogaard, H.; Gerber, J.; Mueller, N.D.; Claessens, L.; et al. Use of Agro-Climatic Zones to Upscale Simulated Crop Yield Potential. Field Crops Res. 2013, 143, 44–55. [Google Scholar] [CrossRef] [Green Version]
- Neres, D.C.D.C.; Amorim, R.S.S.; Albuquerque, M.C.D.F.E.; De Pádua, G.P.; França-Neto, J.D.B. Agroclimatic Zoning of the Brazilian State of Mato Grosso for the Production of Soybean Seeds of Early Cultivars. J. Seed Sci. 2019, 41, 205–212. [Google Scholar] [CrossRef]
- Ferreira, W.R.; Ranal, M.A.; de Santana, D.G.; Nogueira, A.P.O. Germination and Emergence Measurements Could Group Individuals and Species? Braz. J. Bot. 2015, 38, 457–468. [Google Scholar] [CrossRef]
- Ribeiro-Oliveira, J.P.; Ranal, M.A.; Boselli, M.A. Water Dynamics on Germinating Diaspores: Physiological Perspectives from Biophysical Measurements. Plant Phenomics 2020, 2020, 5196176. [Google Scholar] [CrossRef]
- Ranal, M.A.; De Santana, D.G. How and Why to Measure the Germination Process? Braz. J. Bot. 2006, 29, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Brasil. Regras Para Análise de Sementes, 3rd ed.; MAPA/ACS: Ministério da Agricultura, Pecuária e Abastecimento: Brasília, DF, Brazil, 2009.
- Ranal, M.A.; de Santana, D.G.; Ferreira, W.R.; Mendes-Rodrigues, C. Calculating Germination Measurements and Organizing Spreadsheets. Rev. Bras. De Botânica 2009, 32, 849–855. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, J. Testes de Vigor Baseados Na Avaliação de Plântulas. In Vigor de Sementes: Conceitos e Testes; Abrates: Londrina, SC, Brazil, 1999; pp. 1–10. [Google Scholar]
- Cruz, C.D. Genes: A Software Package for Analysis in Experimental Statistics and Quantitative Genetics. Acta Sci. Agron. 2013, 35, 271–276. [Google Scholar] [CrossRef]
- Cruz, C.D. Princípios de Genética Quantitativa, 1st ed.; Editora UFV: Viçosa, MG, Brazil, 2005. [Google Scholar]
- Ribeiro-Oliveira, J.P.; De Santana, D.G.; Pereira, V.J.; Santos, C. Data Transformation: An Underestimated Tool by Inappropriate Use. Acta Sci. Agron. 2018, 40, 35300. [Google Scholar] [CrossRef] [Green Version]
- Davis, J.A. Elementary Survey Analysis., 1st ed.; Prentice-Hall. Prentice-Hall Methods of Social Science: Hoboken, NJ, USA, 1971. [Google Scholar]
- Clerkx, E.J.M.; Vries, H.B.-D.; Ruys, G.J.; Groot, S.P.C.; Koornneef, M. Characterization of Green Seed, an Enhancer of Abi3-1 in Arabidopsis That Affects Seed Longevity. Plant Physiol. 2003, 132, 1077–1084. [Google Scholar] [CrossRef] [Green Version]
- Delmas, F.; Sankaranarayanan, S.; Deb, S.; Widdup, E.; Bournonville, C.; Bollier, N.; Northey, J.G.B.; McCourt, P.; Samuel, M.A. ABI3 Controls Embryo Degreening through Mendel’s I Locus. Proc. Natl. Acad. Sci. USA 2013, 110, E3888–E3894. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, J.; Qiu, K.; Kuai, B. Phytohormone and Light Regulation of Chlorophyll Degradation. Front. Plant Sci. 2017, 8, 1911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreira, T.R.; da Silva, S.F.; da Silva, N.B.; dos Santos, G.M.A.D.A.; dos Santos, A.R. Global Warming and the Effects of Climate Change on Coffee Production. In Quality Determinants in Coffee Production; Springer: Cham, Switzerland, 2021; pp. 65–100. [Google Scholar]
- Niles, M.T.; Lubell, M.; Brown, M. How Limiting Factors Drive Agricultural Adaptation to Climate Change. Agric. Ecosyst. Environ. 2015, 200, 178–185. [Google Scholar] [CrossRef] [Green Version]
- USDA. Oilseeds: World Markets and Trade. In Circular Series FOP 06-17. Global Oilseed Consumption Continues to Grow Despite Slowing Trade and Production; USDA: Washington, DC, USA, 2019. [Google Scholar]
- Smolikova, G.; Dolgikh, E.; Vikhnina, M.; Frolov, A.; Medvedev, S. Genetic and Hormonal Regulation of Chlorophyll Degradation during Maturation of Seeds with Green Embryos. Int. J. Mol. Sci. 2017, 18, 1993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smolikova, G.; Shiroglazova, O.; Vinogradova, G.; Leppyanen, I.; Dinastiya, E.; Yakovleva, O.; Dolgikh, E.; Titova, G.; Frolov, A.; Medvedev, S. Comparative Analysis of the Plastid Conversion, Photochemical Activity and Chlorophyll Degradation in Developing Embryos of Green-Seeded and Yellow-Seeded Pea (Pisum Sativum) Cultivars. Funct. Plant Biol. 2020, 47, 409. [Google Scholar] [CrossRef]








| Statistics | Characters | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| tf (h) | tl (h) | T50 (h) | G (%) | CVt (%) | U (bit) | Z | Rate (Embryo Protrusion h−1) | (h−1) | ||||||||||||||
| ANOVA Assumptions | W (P) | 0.84 (0.01) | 0.97 (0.16) | 0.88 (0.01) | 0.94 (0.01) | 0.86 (0.01) | 0.98 (0.45) | 0.97 (0.16) | 0.87 (0.01) | 0.97 (0.16) | 0.97 (0.08) | |||||||||||
| 1F (P) | 2.50 (0.01) | 3.37 (0.05) | 5.50 (0.01) | 3.83 (0.01) | 6.30 (0.01) | 2.20 (0.02) | 5.10 (0.01) | 6.32 (0.01) | 3.42 (0.01) | 4.34 (0.01) | ||||||||||||
| Model | Source of Variation | DF | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F |
| Nested (Hierarchical) ANOVA | Genotype | 8 | 654.00 | 4.16 | 689.37 | 14.27 | 1404.67 | 5.85 | 438.12 | 4.61 | 800.96 | 6.90 | 27.82 | 1.05 | 0.14 | 1.49 | 0.004 | 1.21 ns | 800.96 | 6.90 | 2.0 × 10−5 | 10.90 |
| Greenish seeds (Genotype) | 9 | 794.67 | 5.05 ** | 511.62 | 10.59 ** | 944.00 | 3.93 ** | 294.95 | 2.82 ** | 1434.96 | 12.35 ** | 88.15 | 3.32 ** | 0.26 | 2.81 ** | 0.010 | 1.72 ns | 1434.96 | 12.35 ** | 1.2 × 10−5 | 6.53 ** | |
| Error | 54 | 157.33 | 98.31 | 240.00 | 104.71 | 116.15 | 26.57 | 0.09 | 0.0036 | 116.15 | 1.8 × 10−5 | |||||||||||
| CV (%) | 29.40 | 8.93 | 13.46 | 14.91 | 13.46 | 19.17 | 12.85 | 33.15 | 13.46 | 10.14 | ||||||||||||
| Genetic Parameters | h2 = | 0.76 | 0.93 | 0.83 | 0.96 | 0.86 | 0.05 | 0.33 | 0.17 | 0.86 | 0.91 | |||||||||||
| CVg (%) = | 21.32 | 13.27 | 12.10 | 23.50 | 13.34 | 1.69 | 3.67 | 6.15 | 13.34 | 13.02 | ||||||||||||
| CVg/CVe = | 0.72 | 1.48 | 0.90 | 2.04 | 0.99 | 0.09 | 0.28 | 0.19 | 0.99 | 1.28 | ||||||||||||
| r (%) = | 34.48 | 68.86 | 44.72 | 80.72 | 49.56 | 0.78, | 7.53 | 3.33 | 49.56 | 62.27 | ||||||||||||
| Statistics | Characters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ANOVA Assumptions | NS (%) | AS (%) | SD (%) | VS (%) | ||||||
| W (P) | 0.88 (0.01) | 0.92 (0.02) | 0.80 (0.01) | 0.91 (0.02) | ||||||
| 1F (P) | 6.65 (0.01) | 3.36 (0.01) | 9.48 (0.00) | 5.37 (0.01) | ||||||
| Model | Source of Variation | DF | MS | 2F | MS | 2F | MS | 2F | MS | 2F |
| Nested (Hierarchical) ANOVA | Genotype | 8 | 2446.04 | 6.16 | 805.73 | 8.53 ** | 695.16 | 3.08 | 2452.50 | 6.93 |
| Greenish seeds (Genotype) | 9 | 11,233.04 | 3.11 ** | 118.68 | 1.26 ns | 804.82 | 3.57 ** | 865.02 | 2.44 ** | |
| Error | 54 | 397.06 | 94.42 | 225.71 | 354.11 | |||||
| CV (%) | 30.03 | 52.22 | 99.97 | 31.98 | ||||||
| Genetic Parameters | h2 = | 0.93 | 0.98 | 0.98 | 0.96 | |||||
| CVg (%) = | 13.67 | 35.05 | 37.82 | 14.22 | ||||||
| CVg/CVe = | 1.26 | 2.73 | 2.31 | 1.82 | ||||||
| r (%) = | 61.48 | 88.18 | 84.26 | 76.86 | ||||||
| Statistics | Characters | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ANOVA Assumptions | RS (cm) | ShS (cm) | SS (cm) | RDrM (mg) | ShDrM (mg) | SDrM (mg) | ShS/RS | ShDrM/RDrM | ||||||||||
| W (P) | 0.80 (0.01) | 0.86 (0.02) | 0.75 (0.01) | 0.82 (0.01) | 0.98 (0.62) | 0.97 (0.11) | 0.90 (0.01) | 0.94 (0.01) | ||||||||||
| 1F (P) | 7.00 (0.02) | 6.08 (0.01) | 5.74 (0.01) | 1.37 (0.26) | 3.76 (0.02) | 3.91 (0.01) | 3.99 (0.01) | 2.90 (0.04) | ||||||||||
| Model | Source of Variation | DF | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F | MS | 2F |
| Nested (Hierarchical) ANOVA | Genotype | 8 | 153.58 | 1.18 ns | 53.17 | 4.28 ** | 132.58 | 2.11 ns | 2034.32 | 1.49 | 28,339.6 | 3.77 ** | 29,653.87 | 2.43 * | 0.29 | 3.56 * | 7.81 | 5.35 ** |
| Greenish seeds (Genotype) | 9 | 44.42 | 2.06 ns | 11.61 | 0.93 ns | 94.43 | 1.50 ns | 551.3 | 2.64 ** | 8763.30 | 1.17 ns | 23,026.20 | 1.89 ns | 0.04 | 1.61 ns | 2.35 | 1.61 ns | |
| Error | 54 | 12.43 | 12.43 | 62.94 | 1361.45 | 7471.66 | 12,187.95 | 0.08 | 1.46 | |||||||||
| CV (%) | 36.97 | 35.20 | 35.16 | 56.64 | 38.01 | 37.73 | 39.25 | 31.27 | ||||||||||
| Genetic Parameters | h2 = | 0.95 | 0.98 | 0.90 | 0.90 | 0.90 | 0.91 | 0.92 | 0.95 | |||||||||
| CVg (%) = | 61.69 | 82.80 | 24.92 | 23.74 | 26.85 | 26.54 | 44.58 | 29.53 | ||||||||||
| CVg/CVe = | 1.58 | 2.79 | 1.06 | 1.04 | 1.06 | 1.15 | 1.21 | 1.51 | ||||||||||
| r (%) = | 71.34 | 88.63 | 52.90 | 51.95 | 52.87 | 56.92 | 59.25 | 69.53 | ||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajala-Luccas, D.; Ribeiro-Oliveira, J.P.; Teixeira, R.N.; Ducatti, K.R.; França-Neto, J.B.; Hilhorst, H.W.M.; da Silva, E.A.A. The Seed–Seedling Transition in Commercial Soybean Cultivars with the Presence of Greenish Seeds in the Sample: A Perspective from Classical Genetic Parameters. Agronomy 2023, 13, 1966. https://doi.org/10.3390/agronomy13081966
Ajala-Luccas D, Ribeiro-Oliveira JP, Teixeira RN, Ducatti KR, França-Neto JB, Hilhorst HWM, da Silva EAA. The Seed–Seedling Transition in Commercial Soybean Cultivars with the Presence of Greenish Seeds in the Sample: A Perspective from Classical Genetic Parameters. Agronomy. 2023; 13(8):1966. https://doi.org/10.3390/agronomy13081966
Chicago/Turabian StyleAjala-Luccas, Daiani, João Paulo Ribeiro-Oliveira, Renake N. Teixeira, Karina Renostro Ducatti, J. B. França-Neto, Henk W. M. Hilhorst, and Edvaldo Aparecido Amaral da Silva. 2023. "The Seed–Seedling Transition in Commercial Soybean Cultivars with the Presence of Greenish Seeds in the Sample: A Perspective from Classical Genetic Parameters" Agronomy 13, no. 8: 1966. https://doi.org/10.3390/agronomy13081966






