Effects of Waterlogging Stress on Root Growth and Soil Nutrient Loss of Winter Wheat at Seedling Stage
Abstract
1. Introduction
2. Materials and Methods
2.1. Overview of the Experimental Area
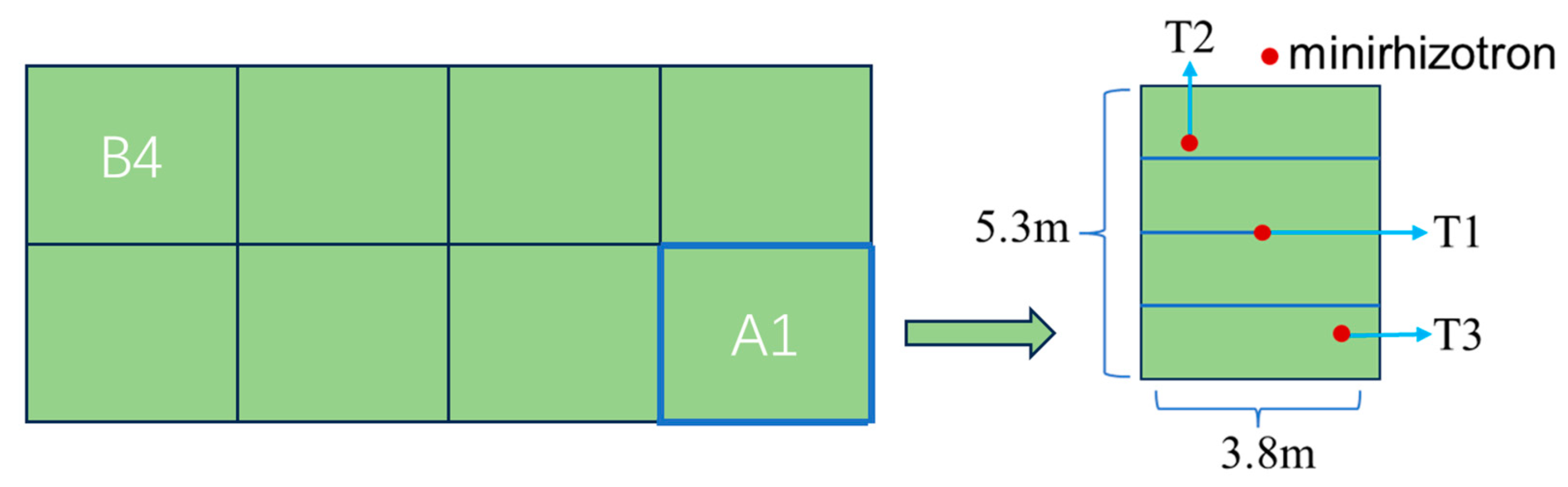
2.2. Configuration of Experimental Facilities
2.3. Experimental Plan Design
2.4. Test Sample Collection
2.5. Data Processing and Analysis
3. Results
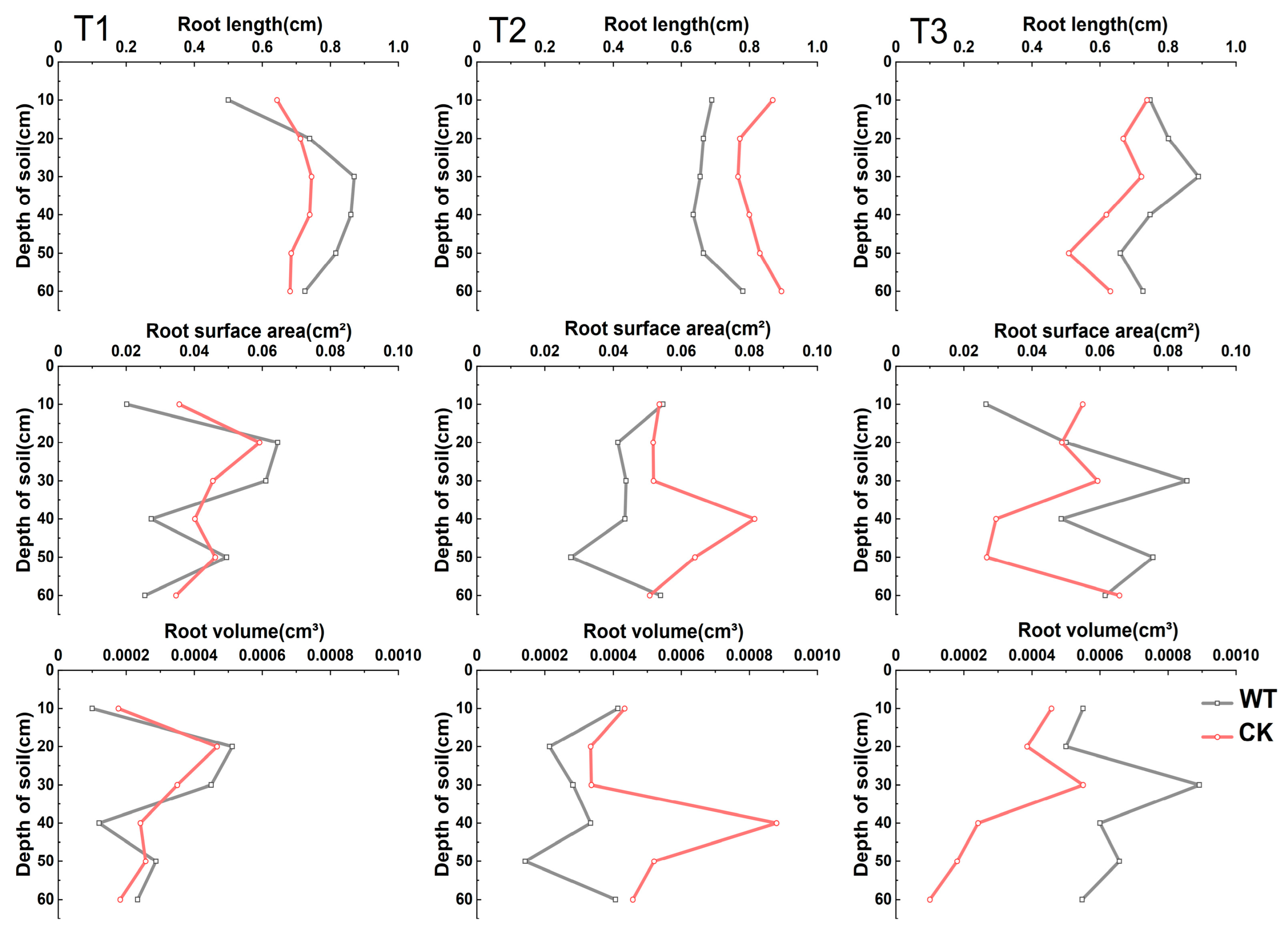
3.1. The Distribution Law of Winter Wheat Seedling Roots in Different Soil Layers
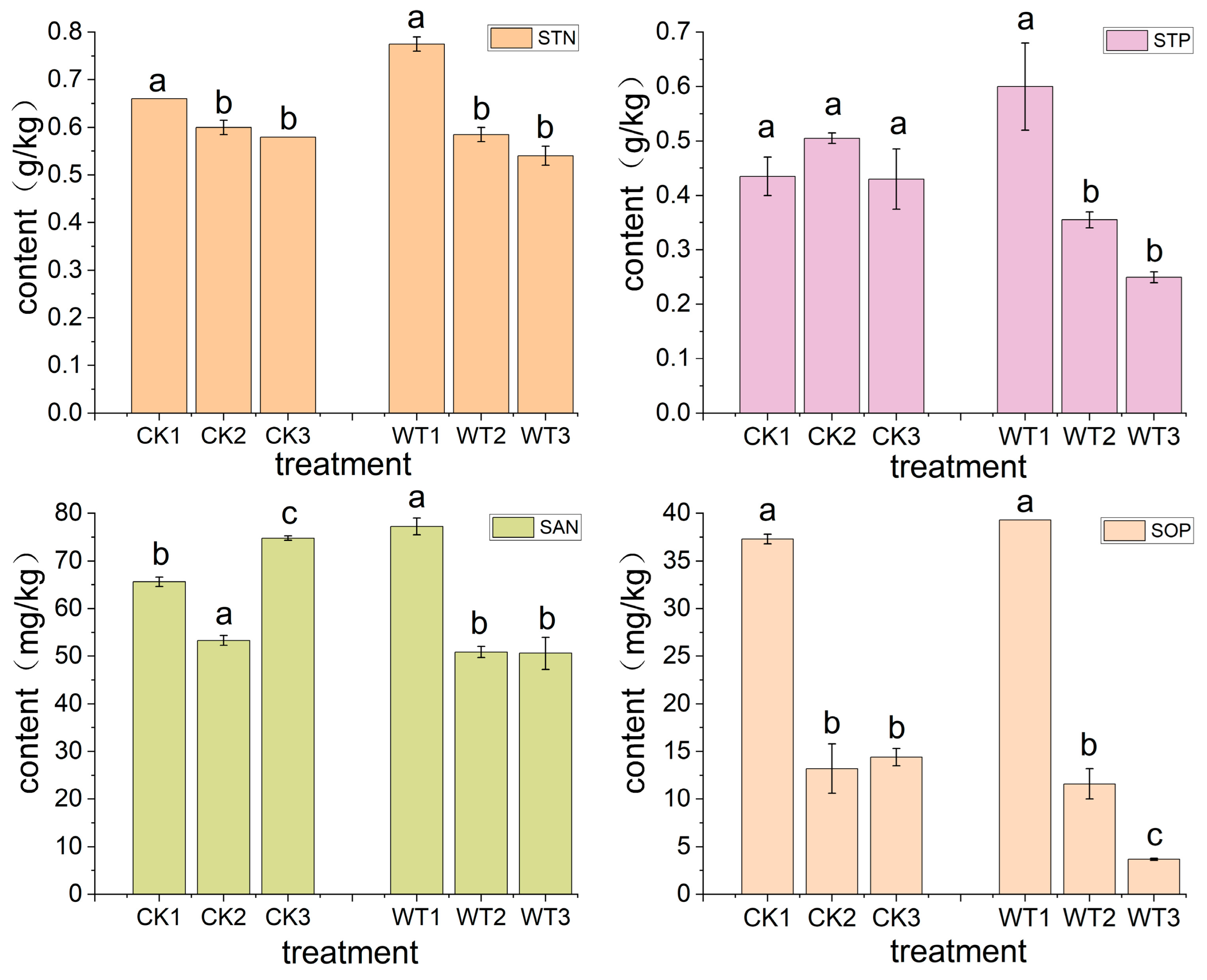
3.2. Variation Characteristics of Nitrogen and Phosphorus Content in Different Soil Layers
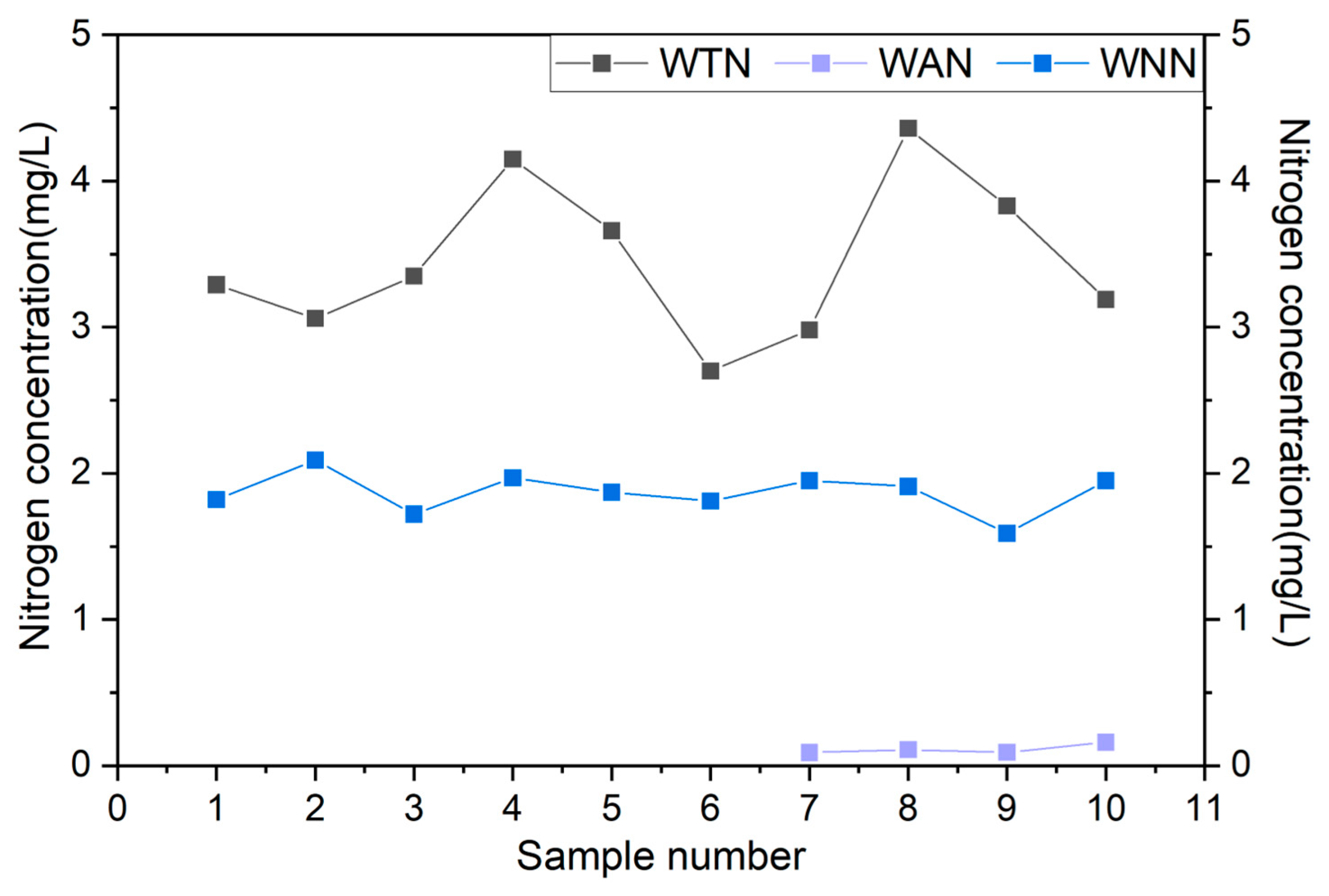
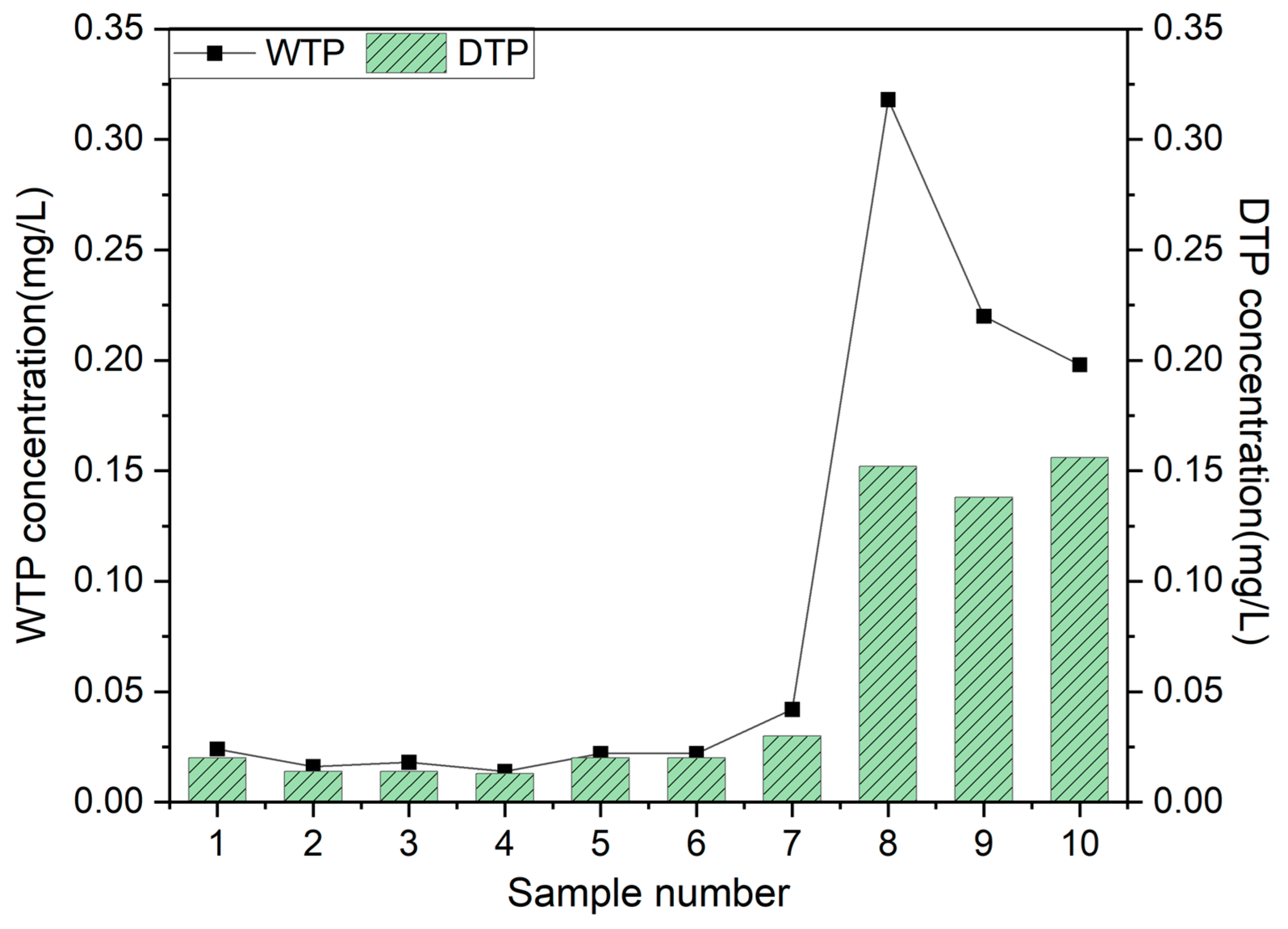
3.3. The Variation Law of Nitrogen and Phosphorus Loss Concentration in Runoff Water
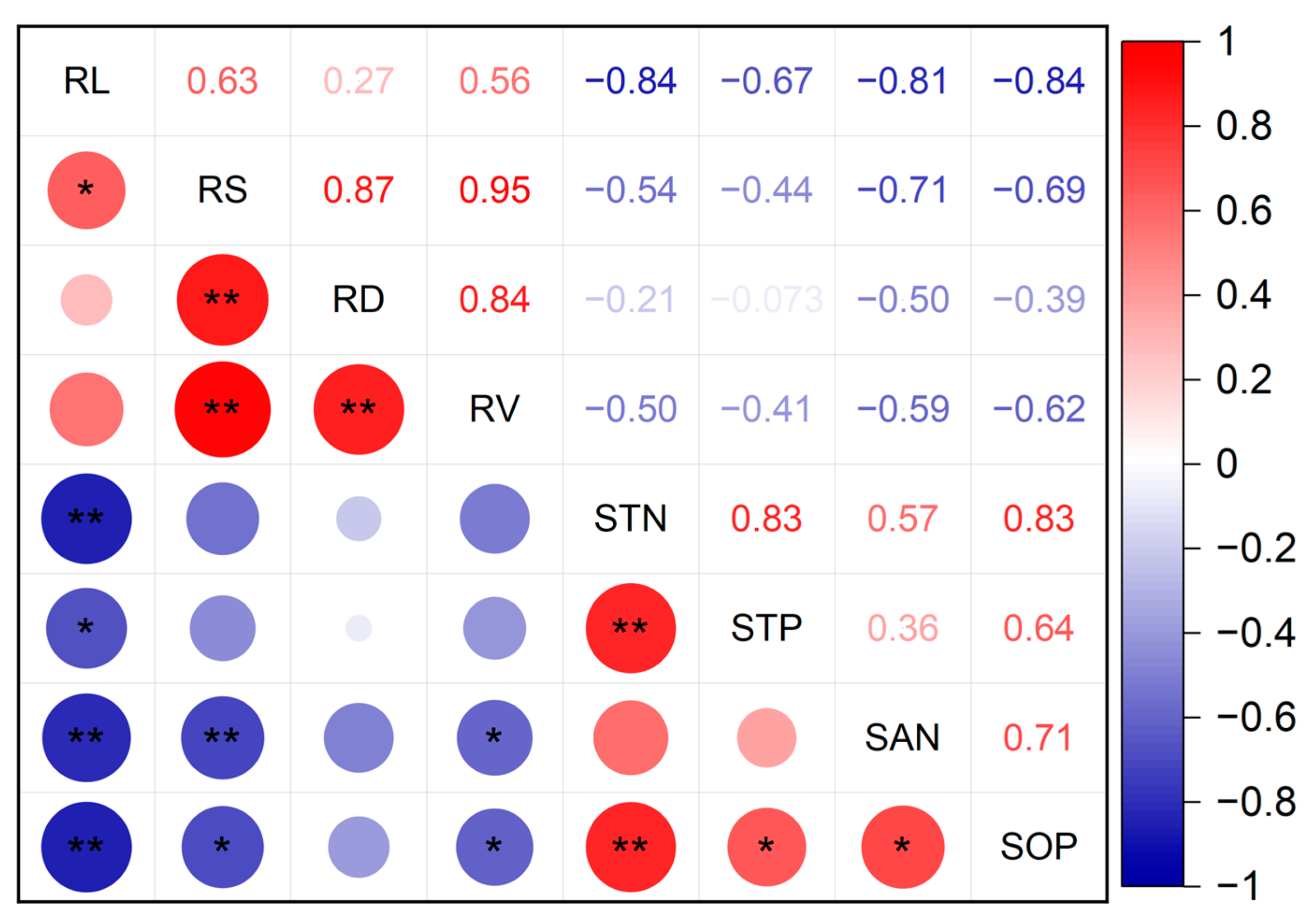
3.4. Correlation Analysis between Wheat Root Morphology and Soil Nutrients
3.5. Analysis of the Influence of Soil Nutrients and Runoff Water Quality
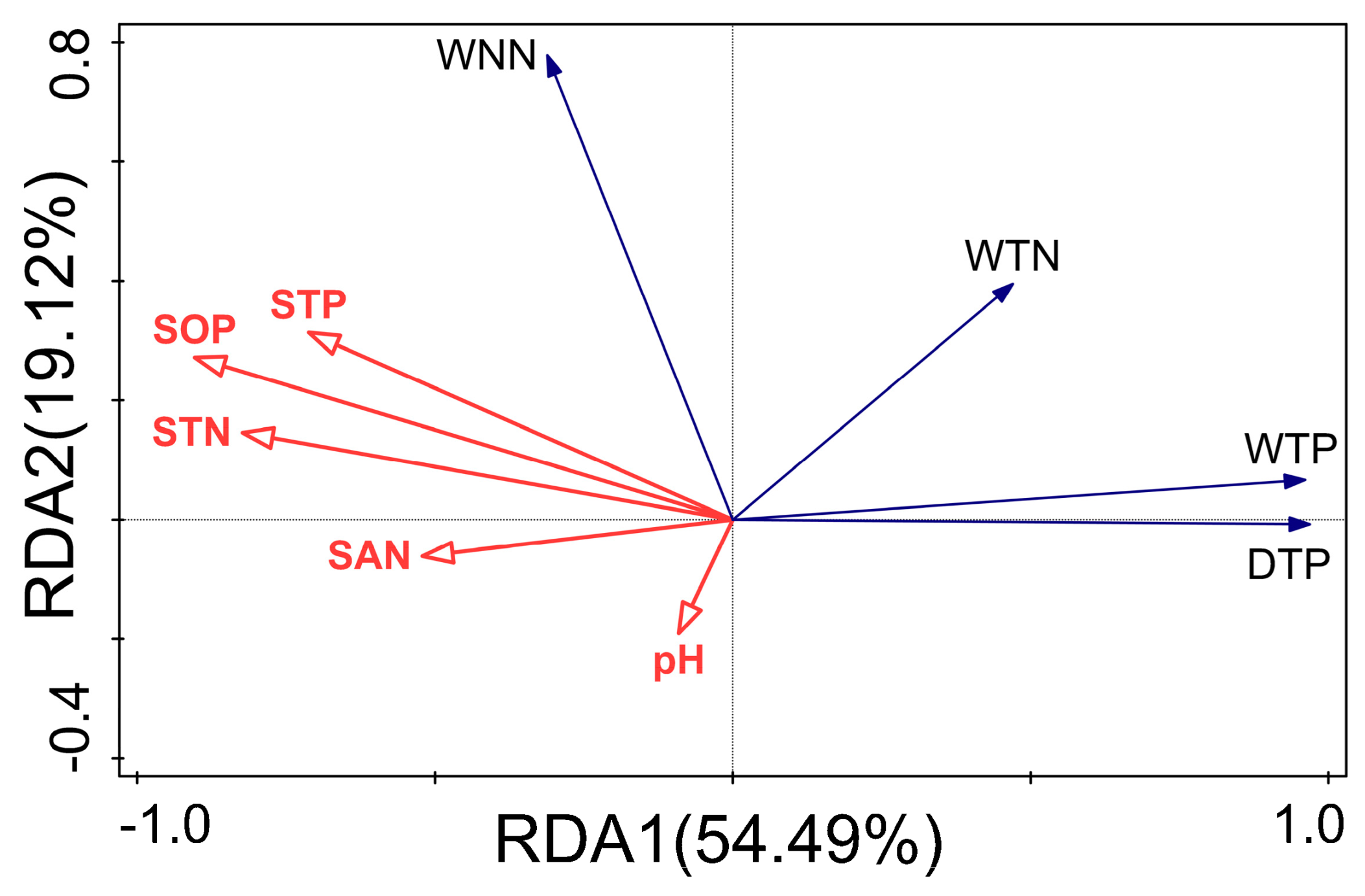
3.6. Analysis of the Influence of Runoff Water Quality and Wheat Root Morphology
4. Discussion
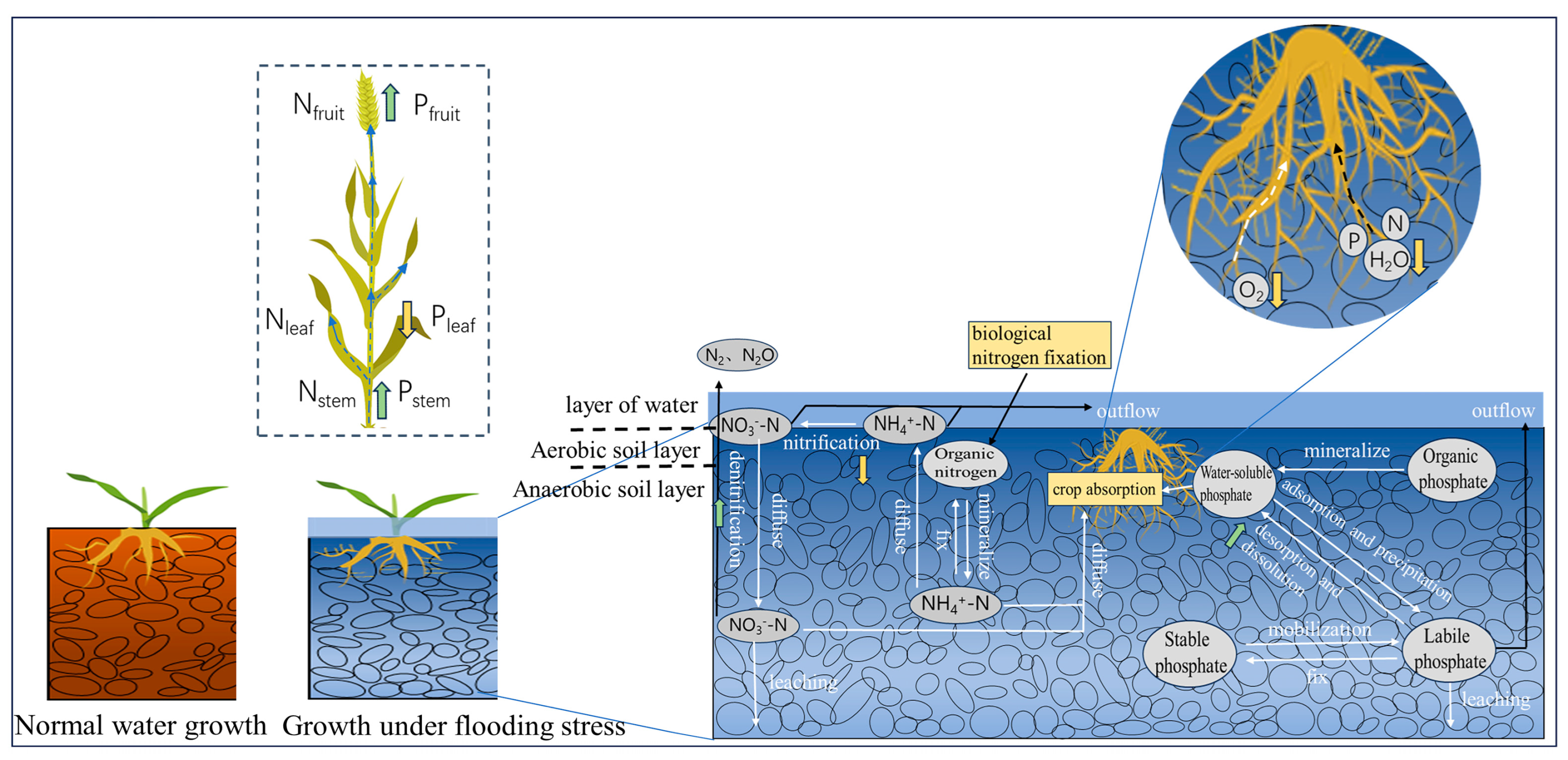
4.1. Effects of Waterlogging Stress on Root Morphology and Soil of Winter Wheat
4.2. Effects of Waterlogging Stress on Soil Nutrients and Runoff Water Quality
4.3. Effects of Runoff Water Quality on Root Growth of Winter Wheat under Waterlogging Stress
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- AghaKouchak, A.; Chiang, F.; Huning, L.S.; Love, C.A.; Mallakpour, I.; Mazdiyasni, O.; Moftakhari, H.; Papalexiou, S.M.; Ragno, E.; Sadegh, M. Climate extremes and compound hazards in a warming world. Annu. Rev. Earth Planet. Sci. 2020, 48, 519–548. [Google Scholar] [CrossRef]
- Manik, S.N.; Pengilley, G.; Dean, G.; Field, B.; Shabala, S.; Zhou, M. Soil and crop management practices to minimize the impact of waterlogging on crop productivity. Front. Plant Sci. 2019, 10, 140. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, Y.; Yang, B.; Ou, L. Research Progress in Waterlogging of Plant. Hubei Agric. Sci. 2015, 54, 4385–4389+4393. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, J.; Song, X.; Gao, P.; Wan, S.; Shi, L.; Wang, X.Z. Spatiotemporal Characteristics of Winter Wheat Waterlogging in the Middle and Lower Reaches of the Yangtze River, China. Adv. Meteorol. 2018, 2018, 3542103. [Google Scholar] [CrossRef]
- Furtak, K.; Wolińska, A. The impact of extreme weather events as a consequence of climate change on the soil moisture and on the quality of the soil environment and agriculture—A review. CATENA 2023, 231, 107378. [Google Scholar] [CrossRef]
- Liu, Z.; Li, G.; Zhang, H.; Zhang, Y.; Zhang, Y.; Duan, S.; Sheteiwy, M.S.A.; Zhang, H.; Shao, H.; Guo, X. TaHsfA2-1, a new gene for thermotolerance in wheat seedlings: Characterization and functional roles. J. Plant Physiol. 2020, 246, 153135. [Google Scholar] [CrossRef]
- Nguyen, L.; Osanai, Y.; Anderson, I.; Bange, M.; Braunack, M.; Tissue, D.T.; Singh, B.K. Impacts of waterlogging on soil nitrification and ammonia-oxidizing communities in farming system. Plant Soil 2018, 426, 299–311. [Google Scholar] [CrossRef]
- Bi, W.; Weng, B.; Yan, D.; Wang, M.; Wang, H.; Wang, J.; Yan, H. Effects of drought-flood abrupt alternation on phosphorus in summer maize farmland systems. Geoderma 2020, 363, 114147. [Google Scholar] [CrossRef]
- Yin, Y.; Chen, H.; Wang, G.; Xu, W.; Wang, S.; Yu, W. Characteristics of the precipitation concentration and their relationship with the precipitation structure: A case study in the Huai River basin, China. Atmos. Res. 2021, 253, 105484. [Google Scholar] [CrossRef]
- De-Campos, A.B.; Huang, C.-H.; Johnston, C.T. Biogeochemistry of terrestrial soils as influenced by short-term flooding. Biogeochemistry 2012, 111, 239–252. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, A.R.; Hill, P.W.; Chadwick, D.R.; Jones, D.L. Typology of extreme flood event leads to differential impacts on soil functioning. Soil Biol. Biochem. 2019, 129, 153–168. [Google Scholar] [CrossRef]
- Xu, Y.; Su, B.; Wang, H. Development of a Runoff Pollution Empirical Model and Pollution Machine Learning Models of the Paddy Field in the Taihu Lake Basin Based on the Paddy In Situ Observation Method. Water 2022, 14, 3277. [Google Scholar] [CrossRef]
- Yan, L.; Xue, L.; Petropoulos, E.; Qian, C.; Hou, P.; Xu, D.; Yang, L. Nutrient loss by runoff from rice-wheat rotation during the wheat season is dictated by rainfall duration. Environ. Pollut. 2021, 285, 117382. [Google Scholar] [CrossRef]
- Yeung, E.; Bailey-Serres, J.; Sasidharan, R. After the deluge: Plant revival post-flooding. Trends Plant Sci. 2019, 24, 443–454. [Google Scholar] [CrossRef]
- Kaur, G.; Singh, G.; Motavalli, P.P.; Nelson, K.A.; Orlowski, J.M.; Golden, B.R. Impacts and management strategies for crop production in waterlogged or flooded soils: A review. Agron. J. 2020, 112, 1475–1501. [Google Scholar] [CrossRef]
- Pan, J.; Sharif, R.; Xu, X.; Chen, X. Mechanisms of waterlogging tolerance in plants: Research progress and prospects. Front. Plant Sci. 2021, 11, 627331. [Google Scholar] [CrossRef]
- Feng, K.; Wang, X.; Zhou, Q.; Dai, T.; Cao, W.; Jiang, D.; Cai, J. Waterlogging Priming Enhances Hypoxia Stress Tolerance of Wheat Offspring Plants by Regulating Root Phenotypic and Physiological Adaption. Plants 2022, 11, 1969. [Google Scholar] [CrossRef]
- Mendoza, R.; Escudero, V. Plant growth, nutrient acquisition and mycorrhizal symbioses of a waterlogging tolerant legume (Lotus glaber Mill.) in a saline-sodic soil. Plant Soil 2005, 275, 305–315. [Google Scholar] [CrossRef]
- Yuan, L.; Chen, M.; Wang, L.; Sasidharan, R.; Voesenek, L.A.; Xiao, S. Multi-stress resilience in plants recovering from submergence. Plant Biotechnol. J. 2023, 21, 466–481. [Google Scholar] [CrossRef]
- Jiang, M.; Xuan, S.; Muneer, M.A.; Sun, B.; Shi, C.; Liu, F.; Wu, R. Response of dry matter partition and yield components to waterlogging and sunlight shortage in different growth stages of wheat. Nat. Hazards 2022, 110, 1133–1152. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, C.; Zhang, Z.; He, D. Effect of Waterlogging on the Growth and Nutrient Metabolism of the Root System of Winter Wheat. Acta Agronmica Sin. 2001, 27, 673–679. [Google Scholar]
- Ghobadi, M.E.; Ghobadi, M.; Zebarjadi, A. Effect of waterlogging at different growth stages on some morphological traits of wheat varieties. Int. J. Biometeorol. 2017, 61, 635–645. [Google Scholar] [CrossRef]
- Lv, Z.Y.; Qin, T.L.; Liu, S.S.; Nie, H.J.; Liu, F.; Wang, J.W. Porous-fiber module increases infiltration and reduces runoff. Agron. J. 2020, 112, 4420–4436. [Google Scholar] [CrossRef]
- Li, W.; Liu, S.; Qin, T.; Xiao, S.; Li, C.; Zhang, X.; Wang, K.; Abebe, S.A. Experiment Study of Porous Fiber Material on Infiltration and Runoff of Winter Wheat Farmland in Huaibei Plain, China. Front. Earth Sci. 2022, 10, 817084. [Google Scholar] [CrossRef]
- Administration, C.M. Specifications of Agrometeorological Observation-Winter Wheat; China Meteorological Press: Beijing, China, 2015; Volume QX/T 299-2015. [Google Scholar]
- Wang, X.; Zhao, C.; Zhao, M. Effects of different flooding duration on soil physical properties of upland rice. J. Hebei Agric. Univ. 2022, 45, 32–38. [Google Scholar] [CrossRef]
- Chen, Y.; Li, S.; Zhang, Y.; Li, T.; Ge, H.; Xia, S.; Gu, J.; Zhang, H.; Lü, B.; Wu, X.; et al. Rice root morphological and physiological traits interaction with rhizosphere soil and its effect on methane emissions in paddy fields. Soil Biol. Biochem. 2019, 129, 191–200. [Google Scholar] [CrossRef]
- Liu, K.; Li, T.T.; Chen, Y.; Huang, J.; Qiu, Y.Y.; Li, S.Y.; Wang, H.; Zhu, A.; Zhuo, X.X.; Yu, F.; et al. Effects of root morphology and physiology on the formation and regulation of large panicles in rice. Field Crops Res. 2020, 258, 107946. [Google Scholar] [CrossRef]
- Zúñiga-Feest, A.; Bustos-Salazar, A.; Alves, F.; Martinez, V.; Smith-Ramírez, C. Physiological and morphological responses to permanent and intermittent waterlogging in seedlings of four evergreen trees of temperate swamp forests. Tree Physiol. 2017, 37, 779–789. [Google Scholar] [CrossRef][Green Version]
- Qi, X.; Li, Q.; Ma, X.; Qian, C.; Wang, H.; Ren, N.; Shen, C.; Huang, S.; Xu, X.; Xu, Q. Waterlogging-induced adventitious root formation in cucumber is regulated by ethylene and auxin through reactive oxygen species signalling. Plant Cell Environ. 2019, 42, 1458–1470. [Google Scholar] [CrossRef]
- Eysholdt-Derzsò, E.; Sauter, M. Hypoxia and the group VII ethylene response transcription factor HRE2 promote adventitious root elongation in Arabidopsis. Plant Biol. 2019, 21, 103–108. [Google Scholar] [CrossRef]
- Visser, E.J.; Voesenek, L.A. Acclimation to soil flooding-sensing and signal-transduction. Plant Soil 2005, 274, 197–214. [Google Scholar] [CrossRef]
- Vidoz, M.L.; Loreti, E.; Mensuali, A.; Alpi, A.; Perata, P. Hormonal interplay during adventitious root formation in flooded tomato plants. Plant J. 2010, 63, 551–562. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, H.; Ju, C.; Xiong, Y.; Bian, J.; Zhao, B.; Yang, J. Changes in grain yield and root morphology and physiology of mid-season rice in the Yangtze River basin of China during the last 60 years. J. Agric. Sci. 2014, 6, 1. [Google Scholar] [CrossRef]
- Jackson, M.B. Ethylene and responses of plants to soil waterlogging and submergence. Annu. Rev. Plant Physiol. 1985, 36, 145–174. [Google Scholar] [CrossRef]
- Day, K.J.; John, E.A.; Hutchings, M.J. The effects of spatially heterogeneous nutrient supply on yield, intensity of competition and root placement patterns in Briza media and Festuca ovina. Funct. Ecol. 2003, 17, 454–463. [Google Scholar] [CrossRef]
- Herzog, M.; Striker, G.G.; Colmer, T.D.; Pedersen, O. Mechanisms of waterlogging tolerance in wheat—A review of root and shoot physiology. Plant Cell Environ. Sci. 2016, 39, 1068–1086. [Google Scholar] [CrossRef]
- Setter, T.L.; Waters, I.; Sharma, S.K.; Singh, K.N.; Kulshreshtha, N.; Yaduvanshi, N.P.; Ram, P.C.; Singh, B.N.; Rane, J.; McDonald, G.; et al. Review of wheat improvement for waterlogging tolerance in Australia and India: The importance of anaerobiosis and element toxicities associated with different soils. Ann. Bot. 2009, 103, 221–235. [Google Scholar] [CrossRef]
- Liesack, W.; Schnell, S.; Revsbech, N.P. Microbiology of flooded rice paddies. FEMS Microbiol. Rev. 2000, 24, 625–645. [Google Scholar] [CrossRef]
- Malik, A.I.; Colmer, T.D.; Lambers, H.; Schortemeyer, M. Changes in physiological and morphological traits of roots and shoots of wheat in response to different depths of waterlogging. Aust. J. Plant Physiol. 2001, 28, 1121–1131. [Google Scholar] [CrossRef]
- Pedersen, O.; Sauter, M.; Colmer, T.D.; Nakazono, M. Regulation of root adaptive anatomical and morphological traits during low soil oxygen. New Phytol. 2021, 229, 42–49. [Google Scholar] [CrossRef]
- Pang, J.; Zhou, M.; Mendham, N.; Shabala, S. Growth and physiological responses of six barley genotypes to waterlogging and subsequent recovery. Aust. J. Agric. Res. 2004, 55, 895–906. [Google Scholar] [CrossRef]
- Dresbøll, D.B.; Thorup-Kristensen, K.; McKenzie, B.M.; Dupuy, L.X.; Bengough, A.G. Timelapse scanning reveals spatial variation in tomato (Solanum lycopersicum L.) root elongation rates during partial waterlogging. Plant Soil 2013, 369, 467–477. [Google Scholar] [CrossRef]
- Desnos, T. Root branching responses to phosphate and nitrate. Curr. Opin. Plant Biol. 2008, 11, 82–87. [Google Scholar] [CrossRef]
- Paez-Garcia, A.; Motes, C.M.; Scheible, W.-R.; Chen, R.; Blancaflor, E.B.; Monteros, M.J. Root traits and phenotyping strategies for plant improvement. Plants 2015, 4, 334–355. [Google Scholar] [CrossRef]
- Rich, S.M.; Watt, M. Soil conditions and cereal root system architecture: Review and considerations for linking Darwin and Weaver. J. Exp. Bot. 2013, 64, 1193–1208. [Google Scholar] [CrossRef]
- Su, J.; Jing, G.; Jin, J.; Wei, L.; Liu, J. Identifying drivers of root community compositional changes in semiarid grassland on the loess plateau after long-term grazing exclusion. Ecol. Eng. 2017, 99, 13–21. [Google Scholar] [CrossRef]
- King, J.; Gay, A.; Sylvester-Bradley, R.; Bingham, I.; Foulkes, J.; Gregory, P.; Robinson, D. Modelling cereal root systems for water and nitrogen capture: Towards an economic optimum. Ann. Bot. 2003, 91, 383–390. [Google Scholar] [CrossRef]
- Comfort, S.; Malzer, G.; Busch, R. Nitrogen fertilization of spring wheat genotypes: Influence on root growth and soil water depletion. Agron. J. 1988, 80, 114–120. [Google Scholar] [CrossRef]
- Rubio, G.; Oesterheld, M.; Alvarez, C.R.; Lavado, R.S. Mechanisms for the increase in phosphorus uptake of waterlogged plants: Soil phosphorus availability, root morphology and uptake kinetics. Oecologia 1997, 112, 150–155. [Google Scholar] [CrossRef]
- Sauter, M. Root responses to flooding. Curr. Opin. Plant Biol. 2013, 16, 282–286. [Google Scholar] [CrossRef]
- Steffens, B.; Rasmussen, A. The physiology of adventitious roots. Plant Physiol. 2016, 170, 603–617. [Google Scholar] [CrossRef]
- Wu, B.; Zhang, H.; Wang, D. Timely supplemental irrigation changed nitrogen use of wheat by regulating root vertical distribution. J. Plant Nutr. Soil Sci. 2018, 181, 396–408. [Google Scholar] [CrossRef]
- Wang, S.; Yan, Y.; Fu, Z.; Chen, H. Rainfall-runoff characteristics and their threshold behaviors on a karst hillslope in a peak-cluster depression region. J. Hydrol. 2022, 605, 127370. [Google Scholar] [CrossRef]
- Pan, Y.; Koopmans, G.F.; Bonten, L.T.; Song, J.; Luo, Y.; Temminghoff, E.J.; Comans, R.N. Influence of pH on the redox chemistry of metal (hydr) oxides and organic matter in paddy soils. J. Soils Sediments 2014, 14, 1713–1726. [Google Scholar] [CrossRef]
- Jin, X.; Cui, Z.; Wang, S. Nitrogen mineralization processes of sediment and soil under continuously waterlogged incubation conditions. Chin. J. Soil Sci. 2006, 37, 909–915. [Google Scholar] [CrossRef]
- Yang, R.; Tong, J.; Li, J.; Wei, W. Numerical simulation of nitrogen loss in surface runoff and leaching from paddy field. J. Irrig. Drain Eng. 2018, 37, 63–69. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, M.; Liu, L. Effect of larmland drainage strategy on nitrogen loss. J. Irrig. Drain Eng. 2013, 32, 138–140. [Google Scholar]
- Unger, I.M.; Motavalli, P.P.; Muzika, R.-M. Changes in soil chemical properties with flooding: A field laboratory approach. Agric. Ecosyst. Environ. 2009, 131, 105–110. [Google Scholar] [CrossRef]
- Hemati Matin, N.; Jalali, M. The effect of waterlogging on electrochemical properties and soluble nutrients in paddy soils. Paddy Water Environ. 2017, 15, 443–455. [Google Scholar] [CrossRef]
- Li, X.; Zhang, W.; Liu, T.; Chen, L.; Chen, P.; Li, F. Changes in the composition and diversity of microbial communities during anaerobic nitrate reduction and Fe (II) oxidation at circumneutral pH in paddy soil. Soil Biol. Biochem. 2016, 94, 70–79. [Google Scholar] [CrossRef]
- Wang, R.; Min, J.; Kronzucker, H.J.; Li, Y.; Shi, W. N and P runoff losses in China’s vegetable production systems: Loss characteristics, impact, and management practices. Sci. Total Environ. 2019, 663, 971–979. [Google Scholar] [CrossRef]
- He, S.Q.; Gong, Y.B.; Zheng, Z.H.; Luo, Z.T.; Tan, B.; Zhang, Y.Q. Effects of rainfall intensities and slope gradients on nitrogen loss at the seedling stage of maize (Zea mays L.) in the purple soil regions of China. Int. J. Agric. Biol. Eng. 2022, 15, 142–148. [Google Scholar] [CrossRef]
- Ma, B.; Guan, R.; Liu, L.; Huang, Z.; Qi, S.; Xi, Z.; Zhao, Y.; Song, S.; Yang, H. Nitrogen loss in vegetable field under the simulated rainfall experiments in Hebei, China. Water 2021, 13, 552. [Google Scholar] [CrossRef]
- Reddy, K.R.; DeLaune, R.D.; Inglett, P.W. Biogeochemistry of wetlands: Science and applications. Soil Sci. Soc. Am. J. 2022, 73, 692. [Google Scholar] [CrossRef]
- Kinsman-Costello, L.E.; O’Brien, J.; Hamilton, S.K. Re-flooding a historically drained wetland leads to rapid sediment phosphorus release. Ecosystems 2014, 17, 641–656. [Google Scholar] [CrossRef]
- Maranguit, D.; Guillaume, T.; Kuzyakov, Y. Effects of flooding on phosphorus and iron mobilization in highly weathered soils under different land-use types: Short-term effects and mechanisms. CATENA 2017, 158, 161–170. [Google Scholar] [CrossRef]
- Xiao, M.; Miao, Z.; Li, Y. Changes of Root-Zone Soil Environment in Flooded Paddy Field under Controlled Drainage Conditions. Pol. J. Environ. Stud. 2017, 26, 881–892. [Google Scholar] [CrossRef]
- Geng, R. Nitrogen and Phosphorus Forms and Loss Risk in a Typical Artificial Woodland North of Huaibei Plain. Master’s Thesis, Hefei University of Technology, Hefei, China, 2017. [Google Scholar]
- Yang, J.; Tang, G.; Li, R.; Yuan, X. Loss of nitrogen and phosphorus from soil and surface water in flooded cropland. J. Irrig. Drain Eng. 2018, 37, 71–77. [Google Scholar] [CrossRef]
- Jiao, P.; Xu, D.; Wang, S.; Wang, Y. Nitrogen and Phosphorus Runoff Losses from Farmland Under Nature Rainfall. J. Agro-Environ. Sci. 2010, 29, 534–540. [Google Scholar] [CrossRef]
- Fan, C.; Zhang, Y.; Wang, J. Effect of pH on the ultraviolet spectra and fluorescence characteristics of dissolved organic matter in the process of straw humification. Spectrosc. Spect. Anal. 2015, 35, 1933–1937. [Google Scholar] [CrossRef]
- Vourlitis, G.L.; Hentz, C.S.; Pinto, O.B.; Carneiro, E.; de Souza Nogueira, J. Soil N, P, and C dynamics of upland and seasonally flooded forests of the Brazilian Pantanal. Glob. Ecol. Conserv. 2017, 12, 227–240. [Google Scholar] [CrossRef]
- Khan, S.U.; Hooda, P.S.; Blackwell, M.S.; Busquets, R. Effects of drying and simulated flooding on soil phosphorus dynamics from two contrasting UK grassland soils. Eur. J. Soil Sci. 2022, 73, e13196. [Google Scholar] [CrossRef]
- Li, W.; Liu, S.; Wang, Y.; Qin, T.; Zhang, X.; Li, C.; Feng, J. Experimental study of rock wool on the farmland soil erosion and crop growth of winter wheat and its comprehensive evaluation. Front. Environ. Sci. 2023, 10, 1090604. [Google Scholar] [CrossRef]
- Yang, J.; Liang, J.; Yang, G.; Feng, Y.; Ren, G.; Ren, C.; Han, X.; Wang, X. Characteristics of non-point source pollution under different land use types. Sustainability 2020, 12, 2012. [Google Scholar] [CrossRef]
- Li, S.; Wang, X.; Qiao, B.; Li, J.; Tu, J. First flush characteristics of rainfall runoff from a paddy field in the Taihu Lake watershed, China. Environ. Sci. Pollut. Res. 2017, 24, 8336–8351. [Google Scholar] [CrossRef]
- Rakotoson, T.; Amery, F.; Rabeharisoa, L.; Smolders, E. Soil flooding and rice straw addition can increase isotopic exchangeable phosphorus in P-deficient tropical soils. Soil Use Manag. 2014, 30, 189–197. [Google Scholar] [CrossRef]
- Malik, A.; Ailewe, T.; Erskine, W. Tolerance of three grain legume species to transient waterlogging. AoB Plants 2015, 7, plv040. [Google Scholar] [CrossRef]
- Gu, C.; Zhang, S.; Han, P.; Hu, X.; Xie, L.; Li, Y.; Brooks, M.; Liao, X.; Qin, L. Soil Enzyme Activity in Soils Subjected to Flooding and the Effect on Nitrogen and Phosphorus Uptake by Oilseed Rape. Front. Plant Sci. 2019, 10, 368. [Google Scholar] [CrossRef]



| STN(g/kg) | STP(g/kg) | SAN(mg/kg) | SOP(mg/kg) | SOM(g/kg) | pH | SP(%) | PP(%) | CP(%) | |
|---|---|---|---|---|---|---|---|---|---|
| 0–20 cm | 0.66 | 0.4 | 56.6 | 27.8 | 10.3 | 8.0 | 41.6 | 41.7 | 16.7 |
| 20–40 cm | 0.58 | 0.3 | 34.8 | 14.4 | 9.1 | 8.1 | 41.1 | 41.3 | 17.6 |
| 40–60 cm | 0.60 | 0.4 | 43.8 | 20.3 | 7.53 | 8.0 | 40.6 | 42.2 | 17.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, H.; Liu, S.; Song, Y.; Qin, T.; Xiao, S.; Li, W.; Xu, L.; Zhou, X. Effects of Waterlogging Stress on Root Growth and Soil Nutrient Loss of Winter Wheat at Seedling Stage. Agronomy 2024, 14, 1247. https://doi.org/10.3390/agronomy14061247
Luo H, Liu S, Song Y, Qin T, Xiao S, Li W, Xu L, Zhou X. Effects of Waterlogging Stress on Root Growth and Soil Nutrient Loss of Winter Wheat at Seedling Stage. Agronomy. 2024; 14(6):1247. https://doi.org/10.3390/agronomy14061247
Chicago/Turabian StyleLuo, Hao, Shanshan Liu, Yifan Song, Tianling Qin, Shangbin Xiao, Wei Li, Lulu Xu, and Xiaoxiang Zhou. 2024. "Effects of Waterlogging Stress on Root Growth and Soil Nutrient Loss of Winter Wheat at Seedling Stage" Agronomy 14, no. 6: 1247. https://doi.org/10.3390/agronomy14061247
APA StyleLuo, H., Liu, S., Song, Y., Qin, T., Xiao, S., Li, W., Xu, L., & Zhou, X. (2024). Effects of Waterlogging Stress on Root Growth and Soil Nutrient Loss of Winter Wheat at Seedling Stage. Agronomy, 14(6), 1247. https://doi.org/10.3390/agronomy14061247






