1. Introduction
Among the winter season cereals, wheat (
Triticum aestivum) is considered to have great economic importance, with large productive capacity [
1]. Wheat is the third most produced crop, with over 600 million tons produced globally per year [
2]. However, despite the technological advances available in plant nutrition, soil management, and machinery, Brazil needs to import about 50% of all wheat consumed in the country [
3].
The use of biological techniques such as plant growth-promoting bacteria (PGPB) can represent a sustainable alternative for cereal growth in tropical areas [
4,
5,
6,
7]. Several PGPB genera show associations with different species of agricultural importance, such as
Azospirillum,
Arthobacter,
Azobacter,
Bacillus,
Bradyrhizobium,
Burkholderia,
Clostridium,
Gluconacetobacter,
Herbaspirillum,
Pseudomonas,
Rhizobium, and
Streptomyces [
8]. The use of these PGPB is growing, particularly in Latin America, for different crops [
4,
9].
Azospirillum is considered one of the most studied plant growth-promoting genera [
10]. An analysis of field trials that were conducted worldwide for over 20 years, where various non-legume crops were inoculated with
Azospirillum spp. under different weather and soil conditions, concluded that crop yield can increase up to 30% [
11].
These bacteria can stimulate plant growth via a series of mechanisms, including the production of phytohormones, such as indole-3-acetic acid (IAA), gibberellins, cytokinins, and salicylic acid [
12,
13], as well as biological nitrogen fixation (BNF) [
14], and an increase in nutrient availability [
6] and nitric oxide production [
15]. In addition,
Azospirillum spp. were reported to reduce biotic and abiotic stresses, increase proline content in shoots and roots, improve water potential and apoplast water content, increase cell wall elasticity and chlorophyll content, increase photoprotection pigments, and improve stomatal conductance [
16].
Azospirillum spp. were related to an increase in plant resistance to pathogens, inhibiting the development of bacterial diseases [
17,
18]. The level of inoculation response was described as wide ranging, as determined by plant–inoculant–environment interactions [
19].
Seed inoculation is the most commonly used technique for delivery of PGPB to cropped plants. However, it is possible to achieve the same or greater responses with other inoculation forms, such as groove or leaf inoculation [
20]. Also, more technological inputs were introduced in seed treatment, such as fungicides, insecticides, micronutrients, and biostimulants, which may cause harmful effects on soil microbiota, reducing inoculation efficiency [
11,
21]. Therefore, studies with inoculation forms in tropical conditions should be performed, since very few field studies were conducted under Brazilian conditions, and new reports can be largely applicable to other important producing countries in South America and Africa [
11].
Another strategy related to improved plant growth is the use of Si-based fertilizers [
22,
23,
24]. Some crops (e.g., sugarcane (
Saccharum officinarum L.), rice (
Oryza sativa L.), wheat, and maize (
Zea mays L.) can remove Si quickly and in great quantity [
25]. In recent years, the number of studies on Si increased substantially in many crops, especially grasses, due to the beneficial effects of Si application on plant resistance to abiotic and biotic stresses and on crop yields [
26]. Silicon application is related to a reduction in insect and pathogen occurrences [
27], salt stress and drought [
28], and the negative effect of heavy rain and winds [
29], common in Savannah conditions. Silicon can also provide several benefits in plant growth and plant architecture, such as erectness and photosynthetic rate [
30,
31]; it also results in decreased transpiration rate [
32,
33], as well as improvements in pest and diseases control [
34] and water economy [
26]. Calcium and magnesium silicate can also increase base saturation and soluble levels of P, Ca, Mg, and Si, thus decreasing the phytotoxic effect of Fe, Mn, Zn, Al, and Cd [
35,
36] and correcting soil acidity [
37].
Despite the several benefits of Ca and Mg silicate application as an Si source and inoculation with Azospirillum brasilense, an increase in wheat grain yield is not always the case. Further research with inoculation forms associated with Si use are needed to determine how to maximize its benefits on growth promotion, wheat development, and yield. In addition, studies are needed to determine if Si utilization has any positive or negative interactions when wheat is inoculated with A. brasilense. This study was based on the hypothesis of a synergistic effect between A. brasilense inoculation forms and Si utilization, providing greater wheat development and yield. The objective of this study was to evaluate the effect of A. brasilense inoculation forms and Ca and Mg silicate application as an Si source on the nutritional and productive properties and wheat grain yield in Savannah conditions.
2. Materials and Methods
2.1. Field Site Description
The study was conducted under field conditions in Selvíria (Savannah region), state of Mato Grosso do Sul, Brazil (20°22′ south (S) and 51°22′ west (W), 335 m above sea level (a.s.l.)), during the crop years of 2016 and 2017. The soil was classified as clayey Oxisol (Rhodic Hapludox) according to the Soil Survey Staff [
38] (soil texture: 471 g∙kg
−1 sand, 90 g∙kg
−1 silt, and 439 g∙kg
−1 clay at a depth of 0–0.20 m). The experimental area was cultivated with annual crops (cereal and legume crops) for over 30 years, with the last 13 years using a no-tillage system. The last crop sequence prior to wheat was maize in both years. The maximum and minimum temperatures and the rainfall verified during the study are presented in
Figure 1.
2.2. Experimental Design and Treatments
The experimental design was a completely randomized block design with four replicates arranged in a 4 × 2 factorial scheme: four Azospirillum brasilense inoculation forms (control, seed, groove, and leaf inoculation), and two soil acidity corrective sources (Ca and Mg silicate as an Si source, with total Si = 10%, CaO = 25%, and MgO = 6%, with effective neutralizing power (ENP) = 88%, and dolomitic limestone with CaO = 28%, MgO = 20%, and ENP = 80%). The experimental plots were composed of twelve 5-m wheat rows spaced at a distance of 0.17 m, with the useful area of the plot being the central eight rows, with the exclusion of 0.5 m from each end.
2.3. Trial Establishment and Management
Soil chemical attributes were determined before the application of acidity corrective sources, according to the Raij et al. [
39] methodology. The total N was determined using the regular Kjeldahl method [
40]. Si was determined according to the methodology proposed by Korndörfer et al. [
41], in Ca chloride (0.01 mol∙L
−1) (
Table 1).
The desiccation of the experimental area was performed with herbicide application of 2,4-D (670 g∙ha−1 of the active ingredient (a.i.)) and glyphosate (1800 g∙ha−1 of the a.i.). The soil acidity corrective sources were applied 30 days before maize planting (predecessor crop, 2015 and 2016 crop seasons) without incorporation and as side dressing at doses of 1.76 mg∙ha−1 of silicate and 1.94 mg∙ha−1 of limestone. The Ca and Mg silicate and dolomitic limestone doses were calculated to increase the basis saturation to 80% based on the soil analysis. In both crop years, the mineral (nitrogen, phosphorus and potassium) NPK was applied as 275 kg∙ha−1 NPK for the 28 August 2016 formulation at planting, based on the soil analysis and wheat crop requirements.
The inoculation with Azospirillum brasilense strains Ab-V5 and Ab-V6 was performed at a dose of 300 mL of liquid inoculant per hectare (guarantee of 2 × 108 CFU (colony-forming units)∙mL−1) for all the inoculation forms. These are commercial strains used in Brazil with brand name AzoTotal®. The seed inoculation was realized one hour before planting the wheat crop and after seed treatment with fungicide and insecticide (the fungicides were thiophanate-methyl + pyraclostrobin (45 g + 5 g of a.i. per 100 kg of seed) and the insecticide fipronil (50 g of a.i. per 100 kg of seed) were used), when the seeds were completely dry. The groove inoculation was performed immediately after sowing the wheat, in the planting line (groove formed after wheat sowing). The inoculant mixed with water was applied using a manual sprayer delivering 300 L∙ha−1. The leaf inoculation was performed 10 days after wheat emergence (DAE) on all the wheat leaves. Also, the inoculant mixed with water was applied using a manual sprayer delivering 300 L∙ha−1. The control treatment did not receive A. brasilense inoculation.
The wheat cultivar used was CD 1104, and planting took place on 3 May 2016 and 10 May 2017, with a density of 70 seeds per meter. The seedling emergence occurred five days after sowing, on 8 May 2016 and 15 May 2017, respectively. Wheat crop was irrigated with supplementary irrigation using a center pivot sprinkling system, with a mean water depth of 14 mm and an irrigation interval of approximately 72 h. The herbicide metsulfuron-methyl (3 g∙ha−1 of a.i.) was applied for post-emergence weed control 20 days after emergence (DAE) of wheat in both seasons, on 28 May 2016 and 6 June 2017, respectively.
Nitrogen fertilizer (side dress application) was spread on the soil surface without incorporation by placing the fertilizer in the middle of the rows on 8 June 2016 and 15 June 2017, when the plants were in the tillering stage at a dose of 140 kg N∙ha−1 as a urea source, for all plots. After N fertilization, the area was irrigated (14 mm depth) at night to minimize losses by ammonia volatilization. The plants were harvested manually at 120 and 117 DAE on 8 September 2016 and 12 September 2017, respectively.
2.4. Measurements Collected
The following nutritional evaluations were performed: (a) leaf chlorophyll index (LCI), measured indirectly using a portable non-destructive chlorophyll meter ClorofiLOG
® model CFL-1030 [
42], when the plants were in the flowering stage. The readings were performed in 10 plants per plot in the flag leaf; (b) N and Si foliar concentration, collecting 30 flag leaves, in the flowering stage, according to the methodology described in Cantarella et al. [
43]; (c) N and Si concentration in root and shoot, in the full flowering of wheat plants, and with the values of root and shoot dry matter, N and Si accumulations were calculated. N determination followed the methodology described in Malavolta et al. [
44], and Si determination followed the methodology described by Silva [
45]; (d) silicon soil content, collecting four samples in each experimental plot, after wheat harvest and determined according to the methodology proposed by Korndörfer et al. [
41], in Ca chloride (0.01 mol∙L
−1).
The following productive component measurements were performed: (e) root and shoot dry matter at the flowering stage, collecting the plants in 0.17 m2 (1.0 m × 0.17 m) per plot and calculated as kg∙ha−1; (f) plant height, defined as the distance (cm) from the ground level to the apex of the spike, at harvest time; (g) number of spikes per meter, by counting spikes in 0.17 m2 (1.0 m × 0.17 m) per experimental unit, at harvest time. Ten spikes were collected at the harvest time for the following evaluations: (h) spike length (cm), distance from the apex to the base of the spike; (i) number of spikelets, by counting all spikelets with grains; (j) number of grains per spikelets, by counting the number of grains in each spikelet; (k) number of grains per spikes, by counting the number of grains in each spike in each plot, (l) hectoliter mass, corresponding to the mass of wheat grains in a 100-L container determined on a ¼ scale after adjusting the water content of the grains to 13% (wet basis); (m) mass of 1000 grains (g), determined using a precision scale (0.01 g), at 13% (wet basis); (n) harvest index (HI), calculated with the following equation: HI = (grain yield/grain yield + aerial part yield); and (o) grain yield, determined by the spikes contained in the useful area of each plot. After the mechanical track, the grains were quantified, and the data were transformed to kg−ha−1 and corrected to 13% moisture (wet basis).
2.5. Statistical Analysis
All data were initially tested for normality using the Shapiro and Wilk [
46] test, and it was observed that all data were distributed normally (
W ≥ 0.90). Data were submitted to analysis of variance (
F-test) using a factorial ANOVA, where the main factors considered were forms of inoculation and acidity corrective sources, and their interactions were considered fixed effects in the model. When appropriate, pairwise mean comparisons were made at
p ≤ 0.05 using the Tukey test with R software version 3.6.1 (Vienna, Austria) [
47].
3. Results
Statistical analysis showed that LCI was significantly affected by the interaction between acidity corrective sources (A) × forms of application (F) in 2016, in addition to the main effect of F and A in both years (
Table 2). Nitrogen foliar concentration was significantly affected by the main effects of A in both year and F in 2017 (
Table 2). Silicon foliar concentration was significantly affected by the main effects of A in both years (
Table 2). Nitrogen shoot accumulation was significantly affected by the main effects of F in 2017 (
Table 2). Silicon shoot accumulation was significantly affected by the main effect of A in both years (
Table 2). Nitrogen root accumulation was significantly affected by the main effects of F in 2017 (
Table 2). Silicon soil content was significantly affected by the main effect of A in both years (
Table 2). Root dry matter was significantly affected by the interaction between A × F, in addition to the main effect of F and A in 2017 (
Table 2). Shoot dry matter was significantly affected by the main effect of F in 2016 (
Table 2). Plant height was significantly affected by the main effect of F in 2017 (
Table 2). The number of spikes per meter was significantly affected by the main effect of A in 2016 (
Table 2). Spike length was significantly affected by the main effect of F in 2017 (
Table 2). The number of spikelets per spike was significantly affected by the interaction between F × A in 2016 (
Table 2). The number of grains per spikelets was significantly affected by the main effect of F and A in 2017 (
Table 2). The number of grains per spike was significantly affected by the main effect of A in 2016 and F in 2017 (
Table 2). Grain yield was significantly affected by the interaction between A × F in 2017, in addition to the main effect of F in both years and A in 2017 (
Table 2). Silicon root accumulation, hectoliter mass, mass of 1000 grains, and harvest index were not influenced by the interaction between F × A, nor by the main effects of F and A (
Table 2).
In 2016, seed and foliar application associated with limestone resulted in a greater LCI compared to control (
Table 3). However, control and seed application associated with silicate showed an increased LCI compared to groove application (
Table 3). Control plots associated with silicate resulted in a higher LCI compared to limestone application (
Table 3). However, groove and foliar application plots associated with limestone showed an increased LCI compared to silicate application (
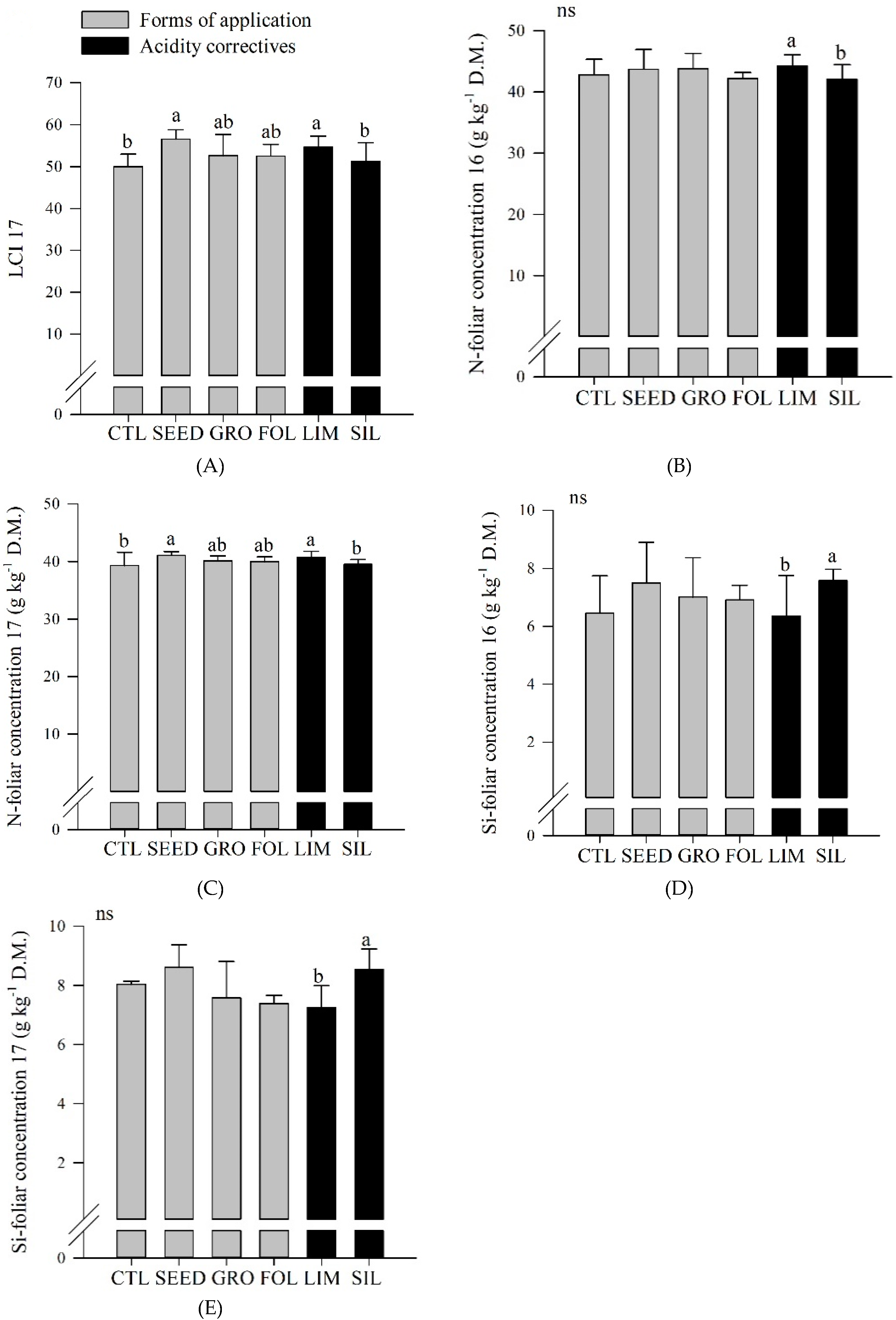
Table 3). In 2017, seed inoculated plots resulted in a greater LCI compared to control plots (
Figure 2A). Also, limestone resulted in an increased LCI compared to silicate application (
Figure 2A).
In both years, limestone resulted in a greater N foliar concentration compared to silicate application (
Figure 2B,C). In 2017, seed application resulted in an increased N foliar concentration compared to control (
Figure 2C). Also, in both years, silicate resulted in a greater Si foliar concentration compared to limestone application (
Figure 2D,E).
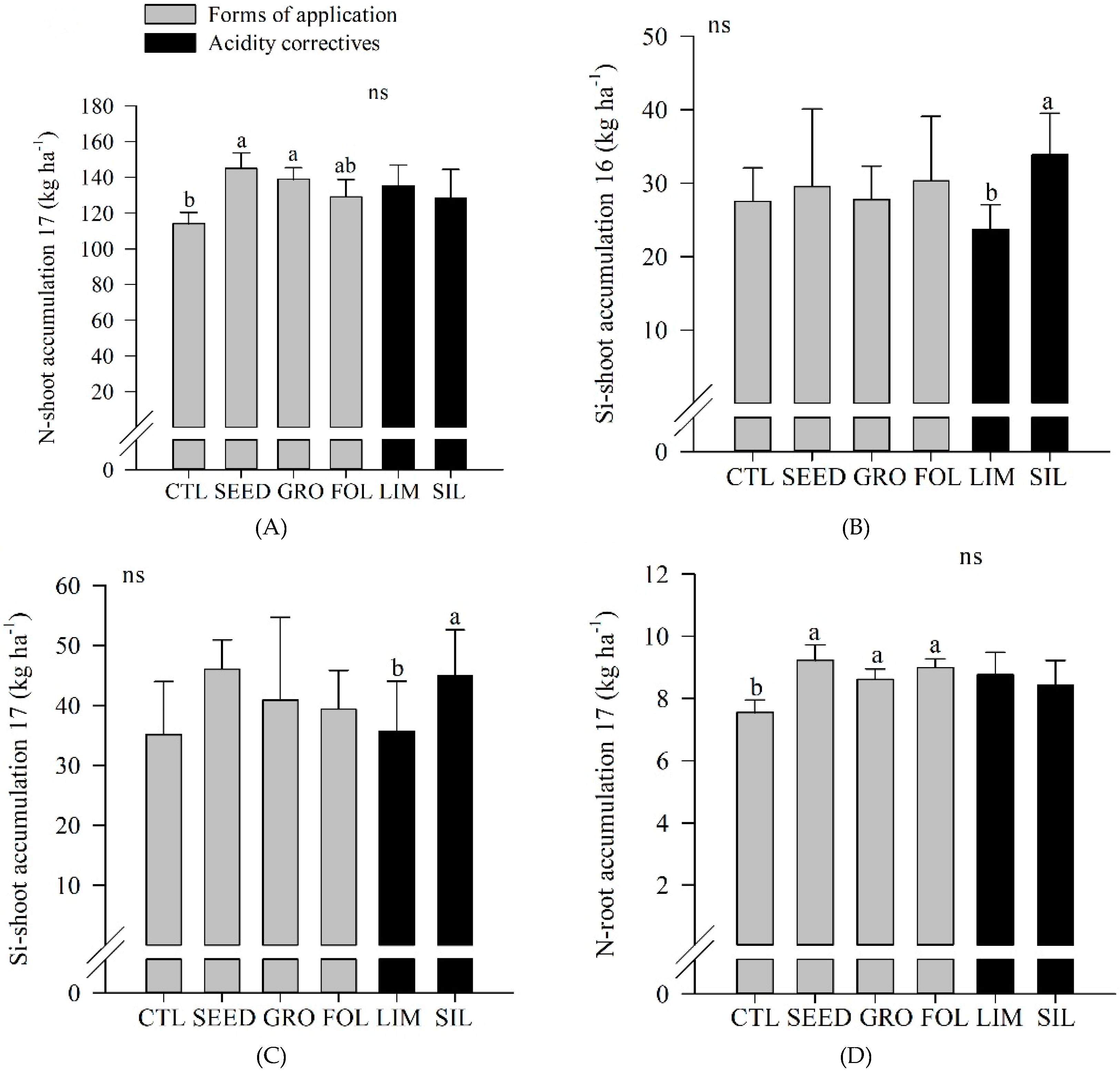
In 2017, seed application resulted in a greater N shoot accumulation compared to control (
Figure 3A). In both years, silicate resulted in a greater Si shoot accumulation compared to limestone application (
Figure 3B,C). In 2017, the control plot showed a lower N root accumulation compared to seed, groove, and foliar applications (
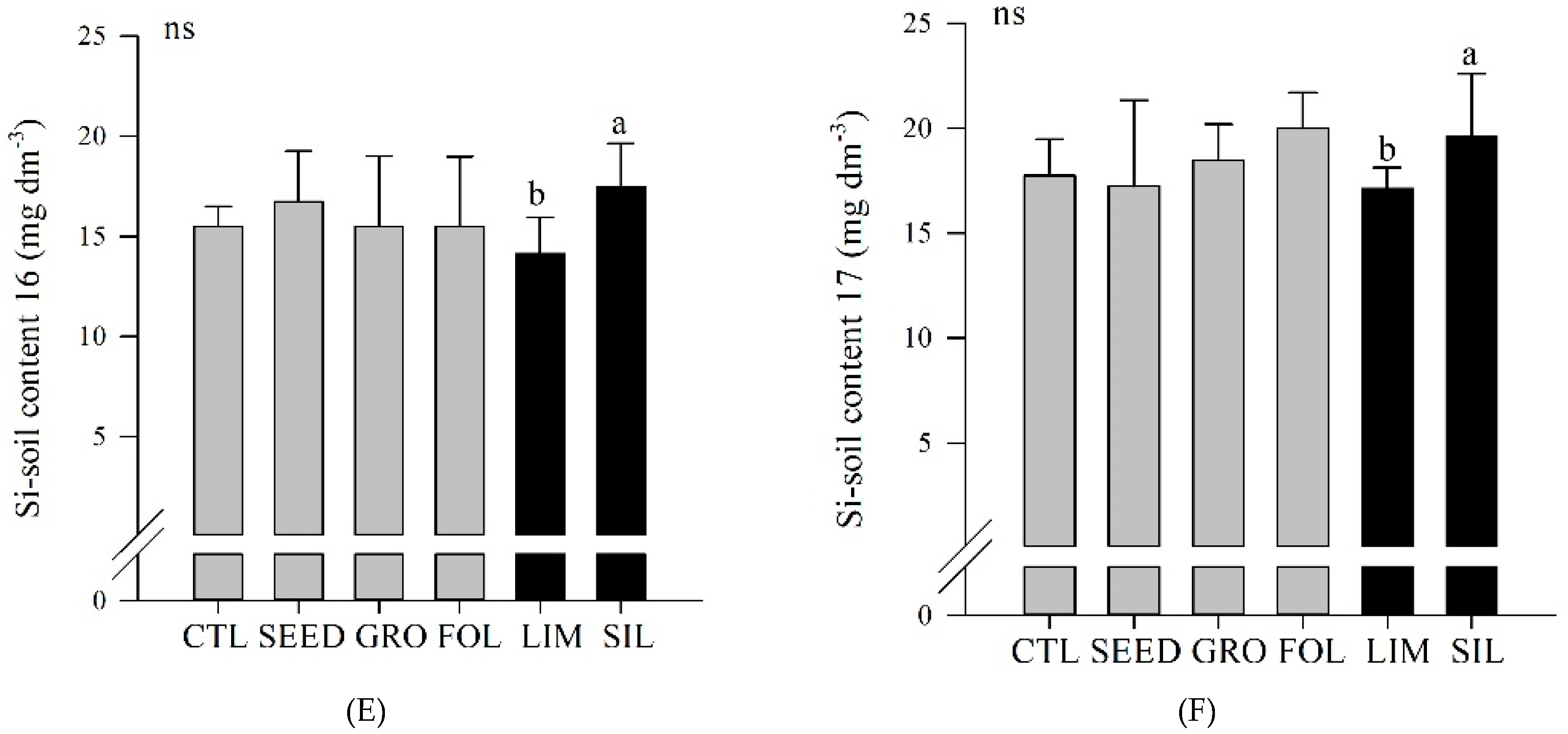
Figure 3D). Also, in both years, silicate resulted in an increased Si soil content compared to limestone application (
Figure 3E,F).
In 2017, groove application associated with limestone resulted in an increased root dry matter compared to control (
Table 4). However, seed and leaf applications associated with silicate resulted in a greater root dry matter compared to control (
Table 4). Control, and seed and leaf applications associated with limestone resulted in a higher root dry matter compared to silicate application (
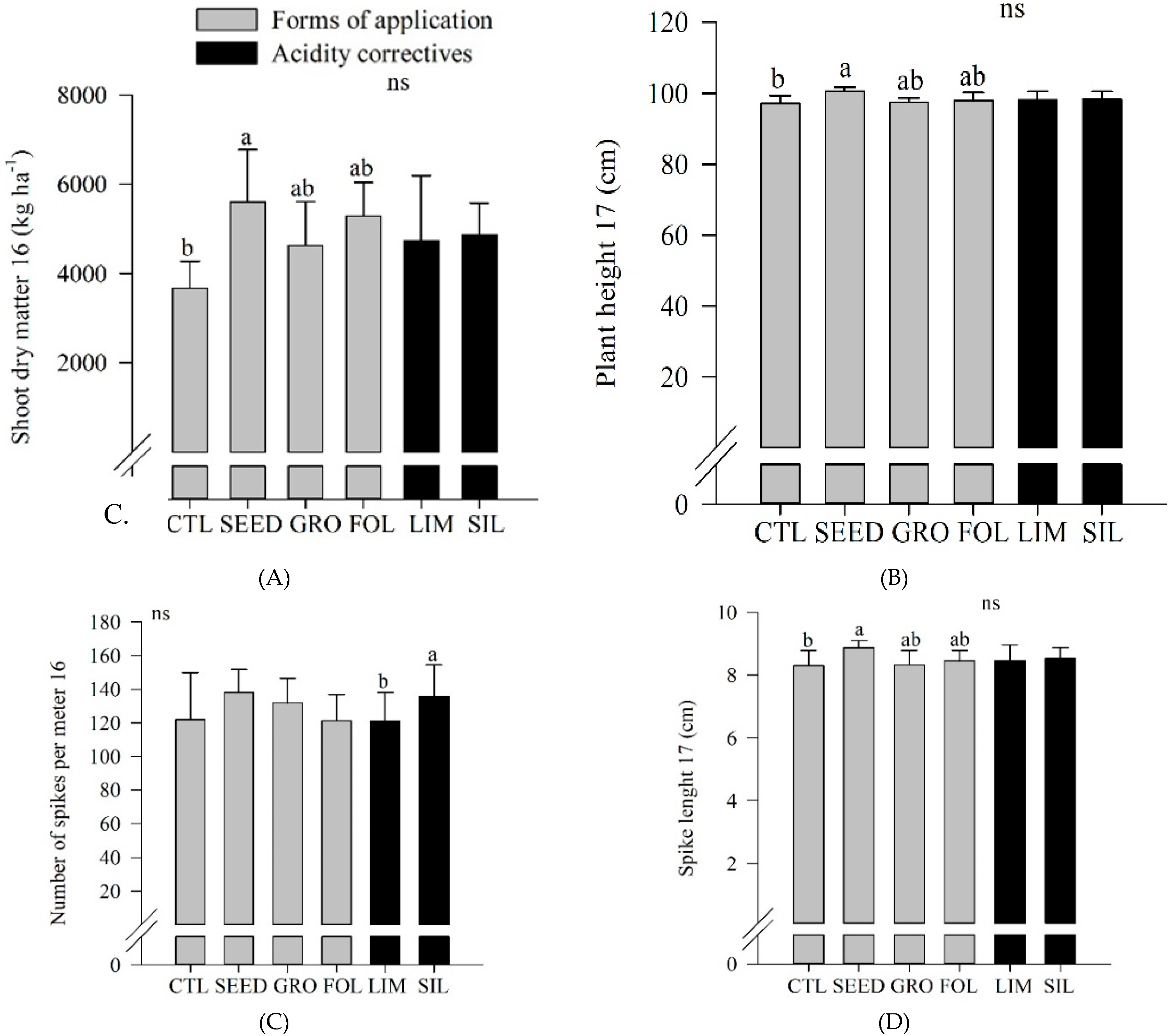
Figure 4A). In 2016, seed inoculated plots showed an increased shoot dry matter compared to control plots (
Figure 4A). Also, in 2017, seed inoculated plots showed an increased plant height compared to control plots (
Figure 4B). In 2016, silicate resulted in an increased number of spikes per meter compared to limestone application (
Figure 4C). In 2017, seed application resulted in an increased spike length compared to control (
Figure 4D).
In 2016, seed application associated with limestone resulted in an increased number of spikelets per spike compared to groove application (
Table 5). However, there was no difference between forms of inoculation when silicate was applied (
Table 5). Seed application associated with limestone resulted in an increased number of spikelets per spike compared to silicate application (
Table 5). In 2017, foliar application resulted in a lower number of grains per spikelets compared to control, and seed and groove applications (
Figure 5A). Silicate resulted in a greater number of grains per spikelet compared to limestone application (
Figure 5A). In 2016, limestone resulted in a greater number of grains per spike than silicate application (
Figure 5B). In 2017, seed application resulted in an increased number of grains per spike compared to foliar application (
Figure 5C).
In 2016, seed application resulted in an increased wheat grain yield compared to control (
Figure 5D). In 2017, seed application associated with limestone showed an increased grain yield compared to control and groove application (
Table 6). However, there was no difference between forms of inoculation when silicate was applied (
Table 6). Control and foliar application associated with silicate resulted in a higher grain yield compared to limestone application (
Table 6).
4. Discussion
Although the exact mechanisms underlying the PGPB effect on wheat development were not evaluated in the present study, it is very likely that the improvement in LCI and N uptake (N foliar concentration and N root and shoot accumulation), reflected in the improved root and shoot dry matter, plant height, spike length, and grain yield by wheat inoculated with
A. brasilense, is associated with its well-known ability to promote plant growth [
4,
13,
19,
48,
49]. Specifically, it was previously demonstrated in draft genome sequences that the strains Ab-V5 and Ab-V6 of
A. brasilense, which were tested in the present study, carry similar
nif and
fix genes that confer their ability to fix atmospheric N [
50]. Although the strains differ in their capacity to synthesize phytohormones [
13,
51,
52], both share the same genes related to the synthesis of auxins. One important feature of Ab-V5 and Ab-V6 is their capacity to induce genes related to the tolerance of biotic and abiotic stresses in plants [
13,
53], and the strains also carry several stress response genes, the majority of which are related to oxidative stresses. This growth promotion mechanisms might have improved the ability of the plants to more efficiently explore the soil, as indicated in previous studies using
A. brasilense [
4,
51,
52,
53,
54,
55].
Seed application was more effective in promoting wheat growth, development, and higher yield. The increase in wheat yield as a function of seed inoculation was, on average, equivalent to 26.7%. However, the increase in grain yield, independently of forms of inoculation, was equivalent to 16.1% compared to control. Similar results were reported in the literature where wheat yield was between 3.1% and 11.1% greater in inoculated plants compared with non-inoculated plants [
10,
56,
57,
58]. Fukami et al. [
11] reported that spray inoculation with
A. brasilense either on leaves or soil can increase plant growth and can replace 25% of N fertilization on grasses. Also, positive results with
A. brasilense inoculation in wheat shoot and root dry matter were reported with increases of 30% and 49% in shoot and root dry matter, respectively [
59]. Evidently, there is still great divergence in the use of
A. brasilense in wheat and other grasses due to the variable results with inoculation. However, it is important to highlight the importance of research on the subject and the potential of using this technology, mainly because it is easy to apply, with a low cost, and has great potential to promote plant growth.
In this study, most of the Si that was taken up was accumulated in shoots (34.6 and 3.3 kg∙ha
−1 in shoot and root, respectively), mainly in the foliar tissue, principally because the Si deposits occur via mass flow more frequently in tissues where water is lost in large amounts [
60] due to plant transpiration [
61]. The translocation rate varies among species, but once deposited in the cell wall, it becomes practically immobile in the plant tissue [
62]. Some grass species, such as wheat, can uptake and redistribute large amounts of Si in the shoot due to specific Si transporters [
63] which facilitate uptake and distribution of the element by the plant [
64,
65]. Although the N leaf concentration was higher than the Si concentration (an average of 41.6 g∙kg
−1 dry matter (D.M.) for N and 7.4 g∙kg
−1 D.M. for Si), it should be noted that the values that were obtained for Si in leaf tissue were high, at the same suitable concentration range for Ca (2.5–10 g∙kg
−1 D.M.), the third nutrient that is most absorbed by wheat for grain production, according to Cantarella et al. [
43]. Moreover, the average concentration of N in foliar tissue, the nutrient that is most absorbed by wheat, was above the suitable range (20–34 g∙kg
−1 D.M.) according to Cantarella et al. [
43], even in control plots (average of 41.1 g∙kg
−1 D.M.).
Silicon application decreased the photosynthetic capacity with a lower LCI and N foliar concentration; however, it provided a greater number of spikelets per meter and grains per spikelet. A higher Si accumulation can increase the leaf tissue density since this element can be incorporated in cell walls and increase the cell density in the mesophyll, affecting the gas diffusion inside the leaf and, thus, the photosynthetic potential [
66]. However, wheat can be favored by this change in the mesophyll structure/anatomy; although it could decrease the diffusion rate of CO
2 inside the leaf, the water vapor output rate would also decrease [
66]. In addition, greater Si availability can result in higher cellulose content in photosynthetic tissues, such as leaves, and lower cellulose content in tissues with a stabilization function, such as culm [
67]. Therefore, Si could benefit wheat foliar architecture, promote erectness, improve leaf angle and light interception, avoid excessive self-shading, reduce lodging, delay senescence, and increase the structural rigidity of the tissues [
37,
68,
69].
Silicon application had a slight effect on wheat development and grain yield. However, Ca and Mg silicate associated with foliar application and without
A. brasilense inoculation provided greater wheat grain yield in 2017. The Si benefits are more frequent in hyperaccumulator crops [
32,
33], which contain SiO
2 concentrations above 5% of shoot dry matter [
70,
71]. In addition, the Si-increased benefits and yields were associated with proline and total sugar accumulation under biotic and abiotic stress conditions [
26,
65,
72]. Silicon use was reported to increase rice [
73], maize [
74], sugar cane [
75], wheat [
37,
76], sorghum [
77], sunflower [
78], peanut, soybean, and common bean [
79] yields. As this field trial was carried out under irrigation, the stress caused by drought was not evident; moreover, sandy soils are more responsive than clay soils to silicate application [
80]. In addition, the increased grain yield is unlikely when the contents of Si available in soil are above than 10.0 mg∙kg
−1 [
37,
81]. In this study, the Si content after Si application was above this range (15.8 and 18.4 mg∙kg
−1 at a depth of 0–0.20 m, in 2015 and 2016, respectively). The use of silicate did not negatively affect the crop development or
A. brasilense inoculation and numerically provided an increase in grain yield compared to limestone application in 12.2%; being a source of Si, the silicate can neutralize the acidity and toxic Al in tropical soils [
35,
36,
37]. In addition, intensive cropping systems remove large Si amounts from the soil [
27]; thus, without adequate Si recycling and uptake by plants, the decrease in Si availability would negatively impact cropping systems if not properly restored [
62,
82].
5. Conclusions
Inoculation with A. brasilense increases N uptake and promotes better development and greater wheat grain yield, independently of inoculation forms. However, seed inoculation is more effective than other inoculation forms in promoting increased wheat growth, development, and yield with greater LCI, N foliar concentration, N shoot and root accumulation, shoot and root dry matter, plant height, spike length, and grain yield.
Calcium and magnesium silicate application as an Si source, associated with foliar inoculation or without A. brasilense inoculation, can increase wheat grain yield. Also, Si application can increase the number of spikes per meter and the number of grains per spikelet. However, this technique reduces LCI and N foliar concentration, and does not promote a sufficient increase in plant growth and development to provide greater wheat development. However, Si did not negatively affect inoculation and N uptake. Therefore, new studies are necessary to better understand Si use associated with growth-promoting bacteria to improve development and yield in crops with the potential for Si accumulation, such as wheat.