Ploidy Status, Nuclear DNA Content and Start Codon Targeted (SCoT) Genetic Homogeneity Assessment in Digitalis purpurea L., Regenerated In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Seed Materials and Explant Preparation
2.2. Seed Germination and Culture Conditions
2.3. Callus Induction and Proliferation
2.4. Indirect Somatic Embryogenesis
2.5. Scanning Electron Microscopy (SEM)
2.6. Shoot Organogenesis via Direct and Indirect Method
2.7. Root Initiation and Acclimatization
2.8. Flow Cytometry
2C DNA content of standard (pg) × mean position of G0/G1 peak of sample
mean position of G0/G1 peak of standard
2.9. SCoT Marker Analysis
2.10. Statistical Analysis
3. Results
3.1. In Vitro Seed Germination
3.2. Callus Induction and Proliferation
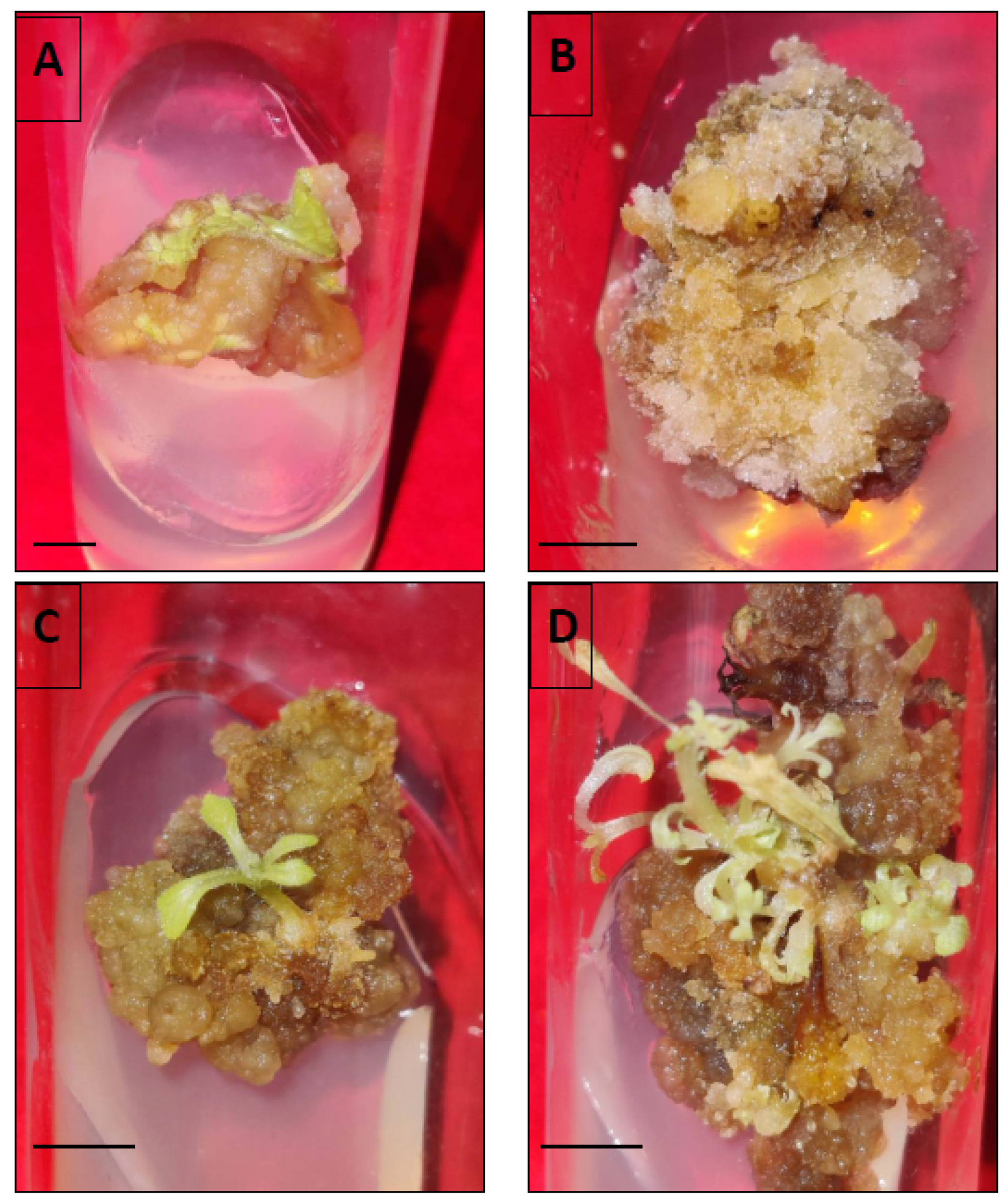
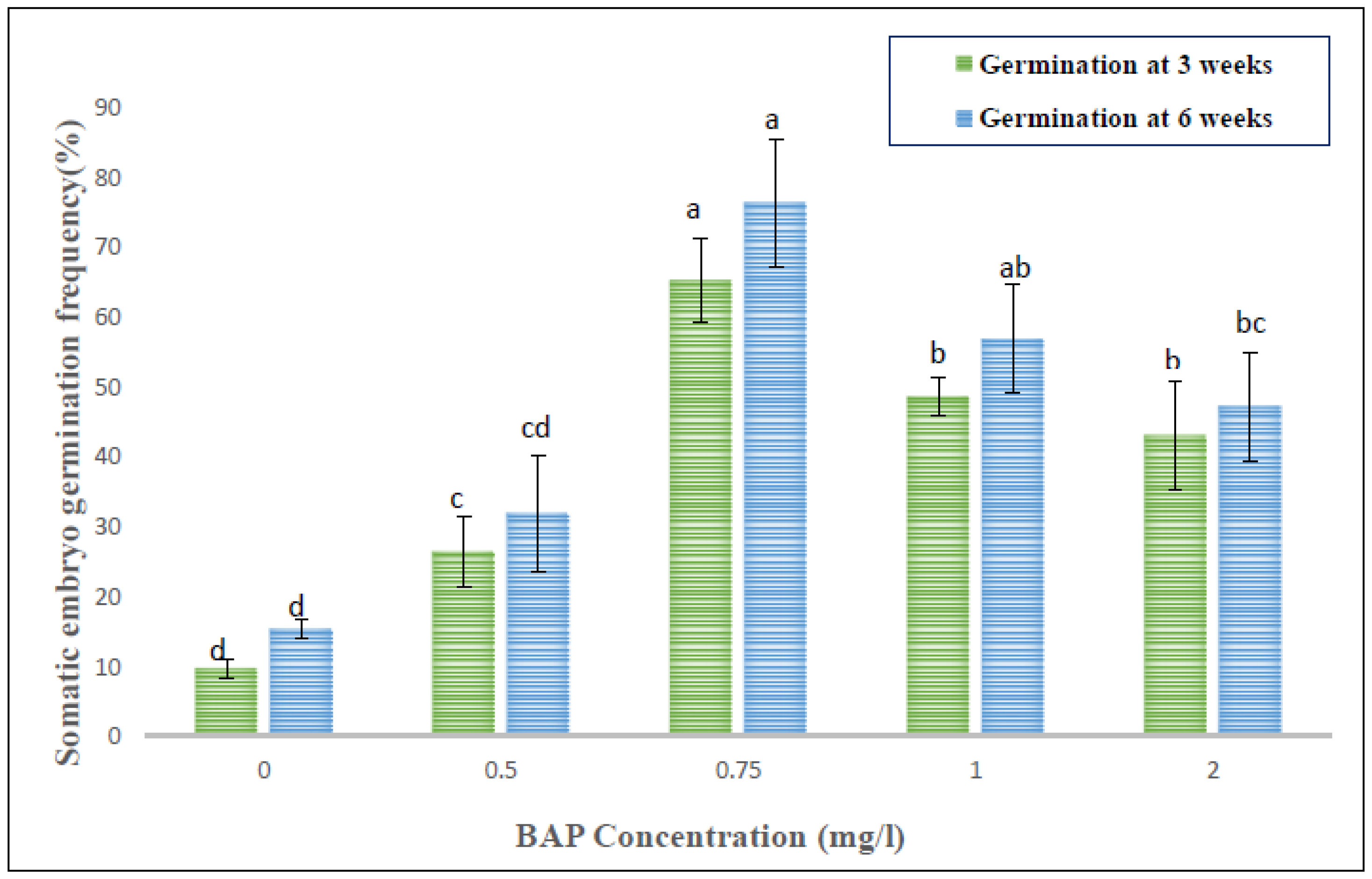
3.3. Somatic Embryo Formation

3.4. Scanning Electron Microscopy (SEM)
3.5. Shoot Organogenesis via Direct and Indirect Method
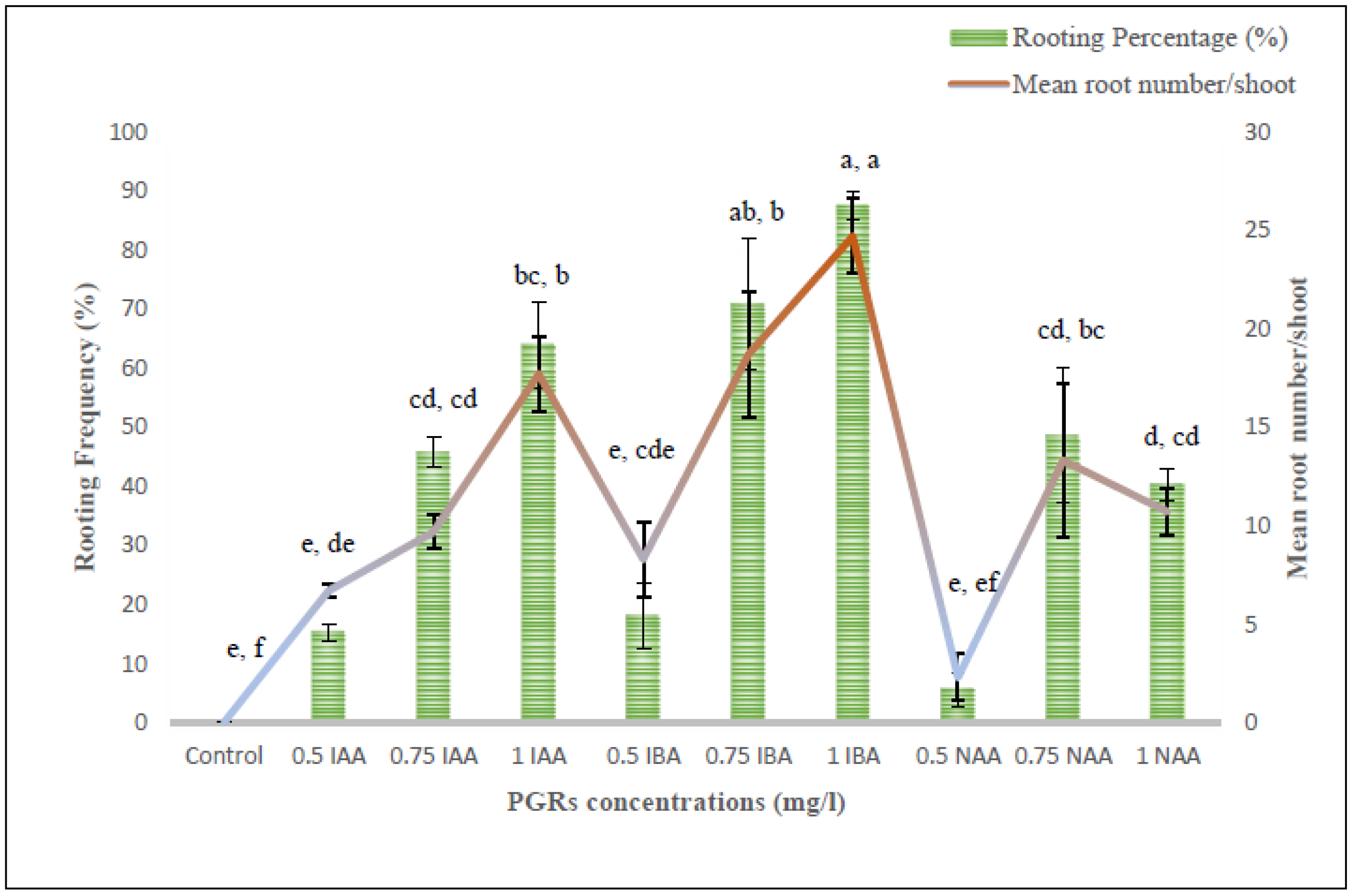
3.6. Rooting and Plantlets Acclimatization
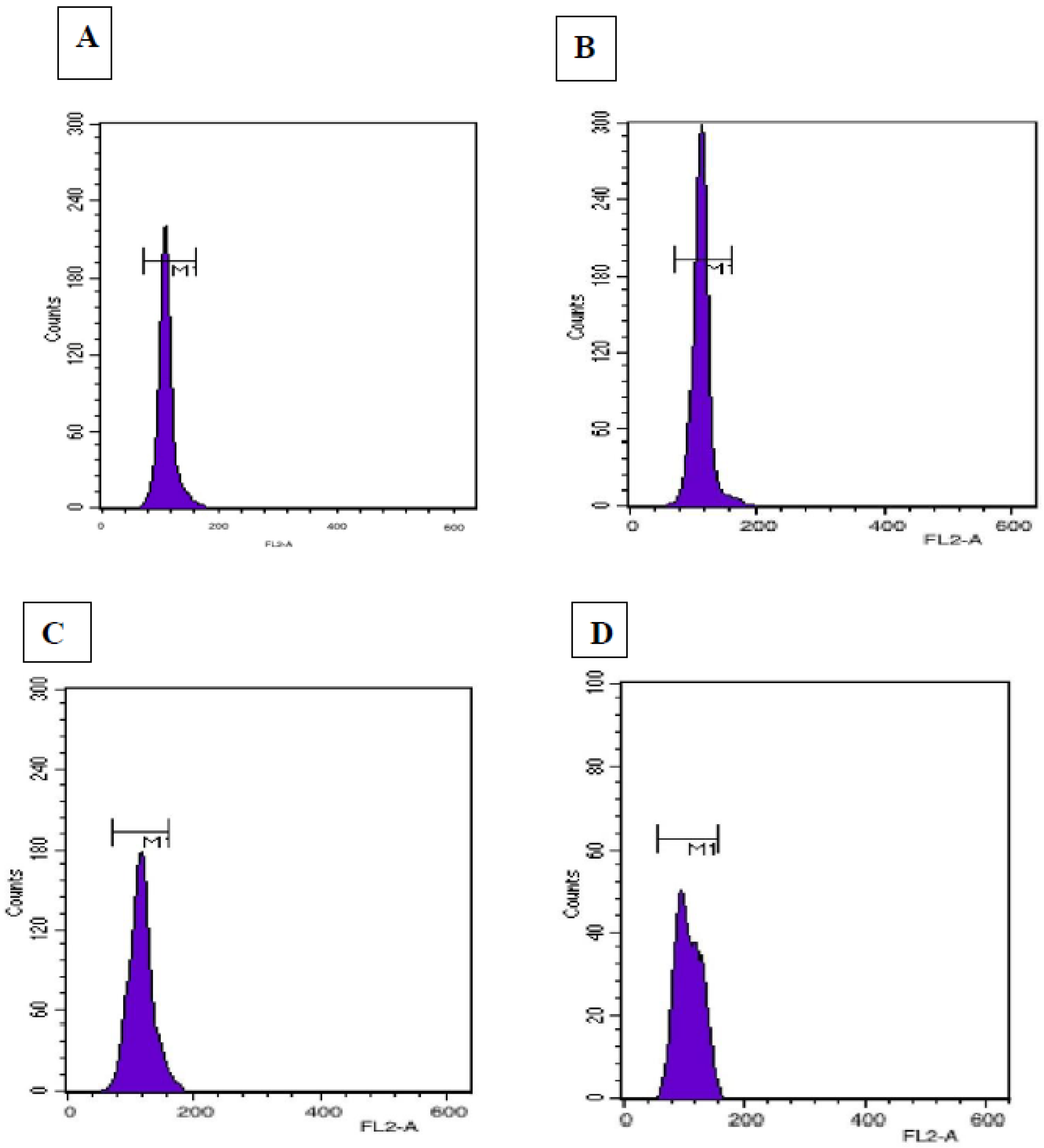
3.7. Flow Cytometric Analysis
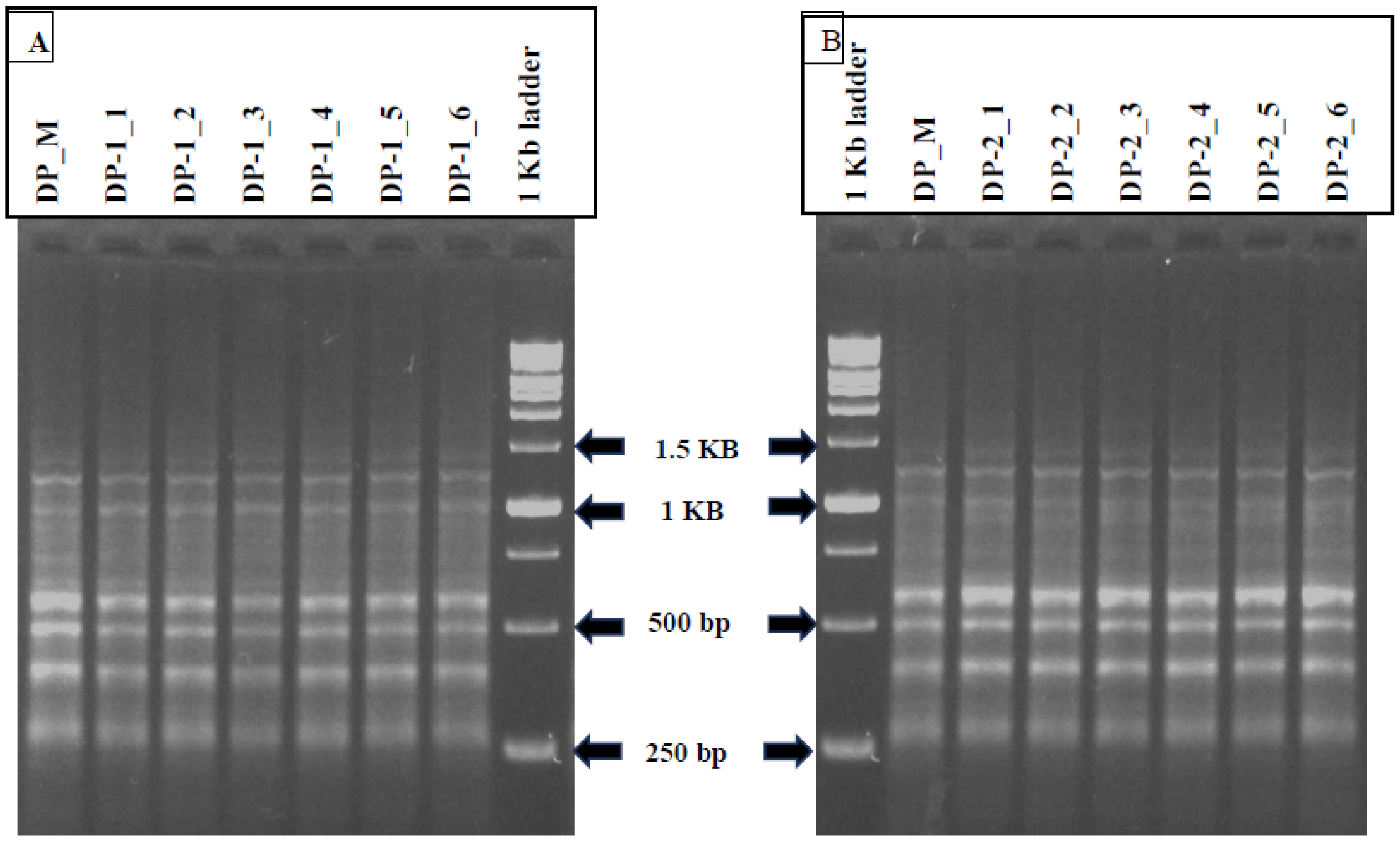
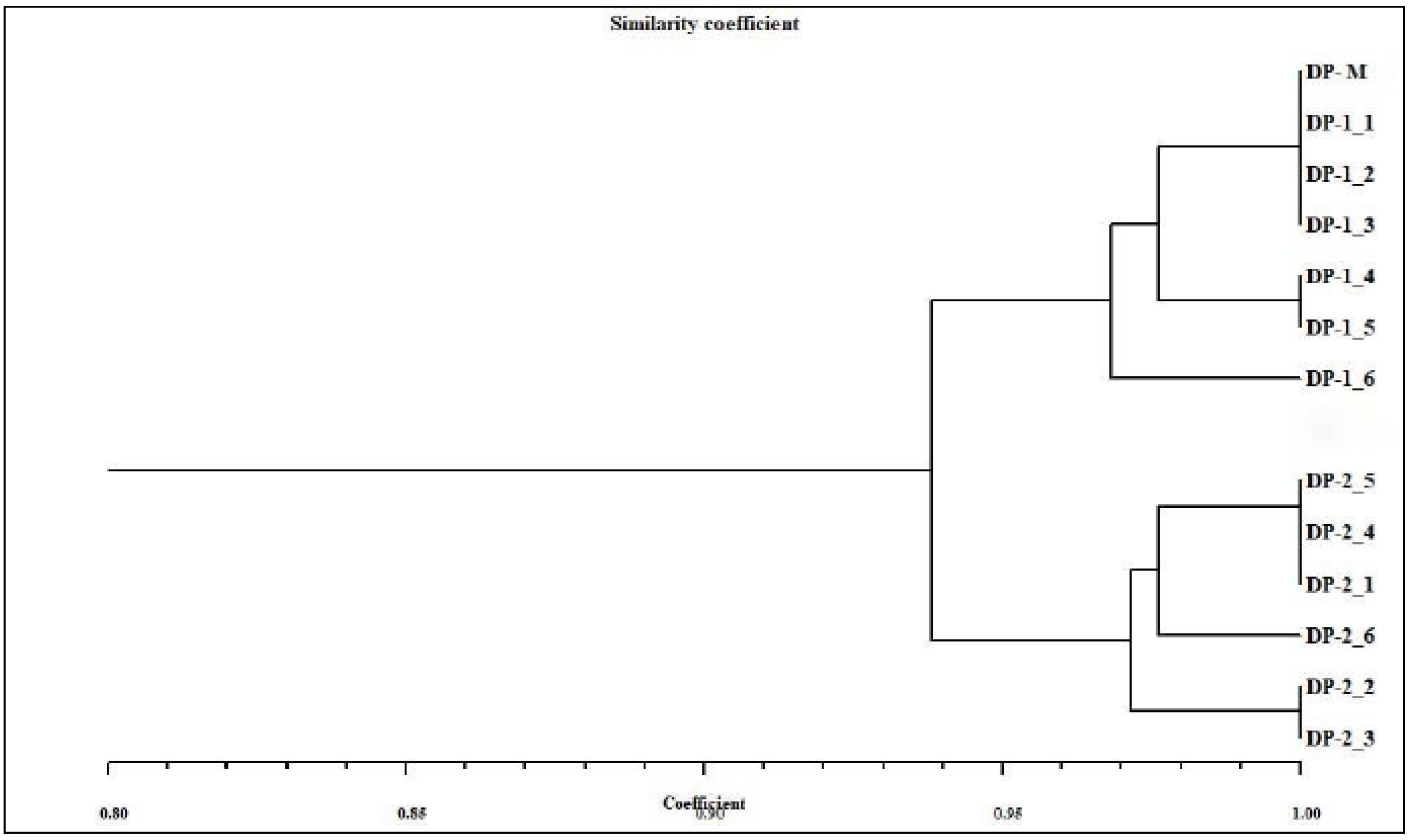
3.8. SCoTmarker Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PGRs | Plant Growth Regulators |
| MS | Murashige and Skoog |
| BAP | 6-benzylaminopurine |
| 2,4-D | 2,4- dichlorophenoxyacetic acid |
| NAA | α-Naphthaleneacetic acid |
| Kn | Kinetin |
| IAA | Indole-3-Acetic Acid |
| IBA | Indole-3-butyric Acid |
| SEM | Scanning Electron Microscopy |
| SCoT | Start Codon Targeted |
| DMRT | Duncan’s Multiple Range Test |
References
- Patil, J.G.; Ahire, M.L.; Nitnaware, K.M.; Panda, S.; Bhatt, V.P.; Kishor, P.B.K.; Nikam, T.D. In Vitro propagation and production of cardiotonic glycosides in shoot cultures of Digitalis purpurea L. by elicitation and precursor feeding. Appl. Microbiol. Biotechnol. 2013, 97, 2379–2393. [Google Scholar] [CrossRef]
- Wu, B.; Li, Y.; Yan, H.; Ma, Y.; Luo, H.; Yuan, L.; Chen, S.; Lu, S. Comprehensive transcriptome analysis reveals novel genes involved in cardiac glycoside biosynthesis and mlnc RNAs associated with secondary metabolism and stress response in Digitalis purpurea. BMC Genom. 2012, 13, 15. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Das, A.K.; Cingoz, G.S.; Gurel, E. In Vitro culture of Digitalis L. (Foxglove) and the production of cardenolides: An up-to-date review. Ind. Crops Prod. 2016, 94, 20–51. [Google Scholar] [CrossRef]
- Rad, M.M.; Abdossi, V.; Moradi, P.; Rakhshandehroo, F.; Mehrafarin, A. Phytochemical changes of Digitalis purpurea L. in response to polyamines and methyl jasmonate application in callus culture. J. Plant Biochem. Biotechnol. 2022, 31, 310–319. [Google Scholar] [CrossRef]
- Verma, S.K.; Sahin, G.; Yucesan, B.; Eker, I.; Sahbaz, N.; Gurel, S.; Gurel, E. Direct somatic embryogenesis from hypocotyl segments of Digitalis trojana Ivan and subsequent plant regeneration. Ind. Crops Prod. 2012, 40, 76–80. [Google Scholar] [CrossRef]
- Bhusare, B.P.; John, C.K.; Bhatt, V.P.; Nikam, T.D. In Vitro propagation of Digitalis lanata Ehrh. through direct shoot regeneration–A source of cardiotonic glycosides. Ind. Crops Prod. 2018, 121, 313–319. [Google Scholar] [CrossRef]
- Verma, S.K.; Das, A.K.; Gantait, S.; Gurel, S.; Gurel, E. Influence of auxin and its polar transport inhibitor on the development of somatic embryos in Digitalis trojana. 3 Biotech 2018, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Yücesan, B.; Demir-Ordu, Ö.; Cihangir, C.; Eker, İ.; Kreis, W.; Gürel, E. In Vitro regeneration and cardenolide determination of an endemic foxglove, Digitalis cariensis (Aegean Foxglove). Vitr. Cell. Dev. Biol. -Plant. 2015, 51, 438–444. [Google Scholar] [CrossRef]
- Pérez-Alonso, N.; Martín, R.; Capote, A.; Pérez, A.; Hernández-Díaz, E.K.; Rojas, L.; Jiménez, E.; Quiala, E.; Angenon, G.; Garcia-Gonzales, R.; et al. Efficient direct shoot organogenesis, genetic stability and secondary metabolite production of micropropagated Digitalis purpurea L. Ind. Crops Prod. 2018, 116, 259–266. [Google Scholar] [CrossRef]
- Syeed, R.; Mujib, A.; Malik, M.Q.; Gulzar, B.; Zafar, N.; Mamgain, J.; Ejaz, B. Direct somatic embryogenesis and flow cytometric assessment of ploidy stability in regenerants of Caladium × hortulanum ‘Fancy’. J. Appl. Genet. 2022, 63, 199–211. [Google Scholar] [CrossRef]
- Niazian, M. Application of genetics and biotechnology for improving medicinal plants. Planta 2019, 249, 953–973. [Google Scholar] [CrossRef] [PubMed]
- Gulzar, B.; Mujib, A.; Mushtaq, Z.; Malik, M.Q. Old Catharanthus roseus culture (14 years) produced somatic embryos and plants and showed normal genome size; demonstrated an increased antioxidant defense mechanism; and synthesized stress proteins as biochemical, proteomics, and flow-cytometry studies reveal. J. Appl. Genet. 2021, 62, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Mujib, A.; Ali, M.; Isah, T. Somatic embryo mediated mass production of Catharanthus roseus in culture vessel (bioreactor)–A comparative study. Saudi J. Biol. Sci. 2014, 21, 442–449. [Google Scholar] [CrossRef] [PubMed]
- García-Gonzáles, R.; Quiroz, K.; Carrasco, B.; Caligari, P. Plant tissue culture: Current status, opportunities and challenges. Cien. Inv. Agr. 2010, 37, 5–30. [Google Scholar] [CrossRef]
- Escobedo-GraciaMedrano, R.M.; Maldonado-Borges, J.I.; Burgos-Tan, M.J.; Valadez-González, N.; Ku-Cauich, J.R. Using flow cytometry and cytological analyses to assess the genetic stability of somatic embryo-derived plantlets from embryogenic Musa acuminata Colla (AA) ssp. malaccensis cell suspension cultures. Plant Cell Tiss. Organ Cult. 2014, 116, 175–185. [Google Scholar] [CrossRef]
- Kairuz, E.; Pérez-Alonso, N.; Angenon, G.; Jiménez, E.; Chong-Pérez, B. Shoot organogenesis, genetic stability, and secondary metabolite production of micropropagated Digitalis purpurea. In Plant Cell and Tissue Differentiation and Secondary Metabolites; Ramawat, K.G., Ekiert, H.M., Goyal, S., Eds.; Springer: Cham, Switzerland, 2021; pp. 169–186. [Google Scholar] [CrossRef]
- Faisal, M.; Alatar, A.A.; Hegazy, A.K.; Alharbi, S.A.; El-Sheikh, M.; Okla, M.K. Thidiazuron induced in Vitro multiplication of Menthaarvensis and evaluation of genetic stability by flow cytometry and molecular markers. Ind. Crops Prod. 2014, 62, 100–106. [Google Scholar] [CrossRef]
- Ali, M.; Mujib, A.; Tonk, D.; Zafar, N. Plant regeneration through somatic embryogenesis and genome size analysis of Coriandrum sativum L. Protoplasma 2017, 254, 343–352. [Google Scholar] [CrossRef]
- Zafar, N.; Mujib, A.; Ali, M.; Tonk, D.; Gulzar, B.; Malik, M.; Sayeed, R.; Mamgain, J. Genome size analysis of field grown and tissue culture regenerated Rauvolfia serpentina (L) by flow cytometry: Histology and scanning electron microscopic study for in vitro morphogenesis. Ind. Crops Prod. 2019, 128, 545–555. [Google Scholar] [CrossRef]
- Çördük, N.; Yücel, G.; Akıncı, N.; Tuna, M. Assessment of the genetic stability of indirect shoot organogenesis-derived plantlets of Digitalis trojana Ivanina by flow cytometry and cytological analyses. Tekirdağ Ziraat Fakültesi Derg. 2017, 14. [Google Scholar]
- Malik, M.Q.; Mujib, A.; Gulzar, B.; Zafar, N.; Syeed, R.; Mamgain, J.; Ejaz, B. Genome size analysis of field grown and somatic embryo regenerated plants in Allium sativum L. J. Appl. Genet. 2020, 61, 25–35. [Google Scholar] [CrossRef]
- Syeed, R.; Mujib, A.; Malik, M.Q.; Mamgain, J.; Ejaz, B.; Gulzar, B.; Zafar, N. Mass propagation through direct and indirect organogenesis in three species of genus Zephyranthes and ploidy assessment of regenerants through flow cytometry. Mol. Biol. Rep. 2021, 48, 513–526. [Google Scholar] [CrossRef] [PubMed]
- Mujib, A.; Malik, M.Q.; Bansal, Y.; Syeed, R.; Ejaz, B.; Mamgain, J. Somatic Embryogenesis in Catharanthus Roseus: Proteomics of embryogenic and non-embryogenic tissues; and genome size analysis of regenerated plant. In The Catharanthus Genome; Kole, C., Ed.; Springer: Cham, Switzerland, 2022; pp. 85–100. [Google Scholar] [CrossRef]
- Ajithan, C.; Vasudevan, V.; Sathish, D.; Sathish, S.; Krishnan, V.; Manickavasagam, M. The influential role of polyamines on the in vitro regeneration of pea (Pisum sativum L.) and genetic fidelity assessment by SCoT and RAPD markers. Plant Cell Tiss. Organ Cult. 2019, 139, 547–561. [Google Scholar] [CrossRef]
- Erişen, S.; Kurt-Gür, G.; Servi, H. In Vitro propagation of Salvia sclarea L. by meta-Topolin, and assessment of genetic stability and secondary metabolite profiling of micropropagated plants. Ind. Crops Prod. 2020, 157, 112892. [Google Scholar] [CrossRef]
- Ejaz, B.; Mujib, A.; Mamgain, J.; Malik, M.Q.; Syeed, R.; Gulzar, B.; Bansal, Y. Comprehensive in vitro regeneration study with SCoT marker assisted clonal stability assessment and flow cytometric genome size analysis of Carthamus tinctorius L.: An important medicinal plant. Plant Cell Tiss. Organ Cult. 2022, 148, 403–418. [Google Scholar] [CrossRef]
- Mamgain, J.; Mujib, A.; Ejaz, B.; Gulzar, B.; Malik, M.Q.; Syeed, R. Flow cytometry and start codon targeted (SCoT) genetic fidelity assessment of regenerated plantlets in Tylophora indica (Burm. f.) Merrill. Plant Cell Tiss. Organ Cult. 2022, 150, 129–140. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Galbraith, D.W. Simultaneous flow cytometric quantification of plant nuclear DNA contents over the full range of described angiosperm 2C values. Cytom Part A J. Int. Soc. Adv. Cytom. 2009, 75, 692–698. [Google Scholar] [CrossRef]
- Bhusare, B.P.; John, C.K.; Bhatt, V.P.; Nikam, T.D. Induction of somatic embryogenesis in leaf and root explants of Digitalis lanata Ehrh.: Direct and indirect method. S. Afr. J. Bot. 2020, 130, 356–365. [Google Scholar] [CrossRef]
- Doležel, J.; Greilhuber, J.; Suda, J. Estimation of nuclear DNA content in plants using flow cytometry. Nat. Protoc. 2007, 2, 2233–2244. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.J. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Rohlf, F.J. NTSYSpc: Numerical Taxonomy and Multivariate Analysis System, Version 2.02; Setauket: New York, NY, USA, 2000. [Google Scholar]
- Sneath, P.H.; Sokal, R.R. Numerical Taxonomy. The Principles and Practice of Numerical Classification; W. H. Freeman and Company: New York, NY, USA, 1973. [Google Scholar]
- Duncan, D.B. Multiple range and multiple F tests. Biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]
- Dolezel, J. Nuclear DNA content and genome size of trout and human. Cytom. Part A 2003, 51, 127–128. [Google Scholar]
- Verma, S.K.; Yucesan, B.; Sahin, G.; Gurel, E. Embryogenesis, plant regeneration and cardiac glycoside determination in Digitalis ferruginea subsp. ferruginea L. Plant Cell Tiss. Organ Cult. 2014, 119, 625–634. [Google Scholar] [CrossRef]
- Hsieh, Y.F.; Jain, M.; Wang, J.; Gallo, M. Direct organogenesis from cotyledonary node explants suitable for Agrobacterium-mediated transformation in peanut (Arachis hypogaea L.). Plant Cell Tiss. Organ Cult. 2017, 128, 161–175. [Google Scholar] [CrossRef]
- Hesami, M.; Naderi, R.; Yoosefzadeh-Najafabadi, M. Optimizing sterilization conditions and growth regulator effects on in vitro shoot regeneration through direct organogenesis in Chenopodium quinoa. BioTechnologia 2018, 99, 49–57. [Google Scholar] [CrossRef]
- Alttaher, A.G.A.; Yusof, Z.N.B.; Mahmood, M.; Shaharuddin, N.A. High-frequency induction of multiple shoots and plant regeneration from cotyledonary node explants of Tongkat Ali (Eurycoma longifolia Jack). Appl. Ecol. Environ. Res. 2020, 18, 6321–6333. [Google Scholar] [CrossRef]
- Sadhu, S.; Jogam, P.; Thampu, R.K.; Abbagani, S.; Penna, S.; Peddaboina, V. High efficiency plant regeneration and genetic fidelity of regenerants by SCoT and ISSR markers in chickpea (Cicer arietinum L.). Plant Cell Tiss. Organ Cult. 2020, 141, 465–477. [Google Scholar] [CrossRef]
- Fatima, Z.; Mujib, A.; Fatima, S.; Arshi, A.; Umar, S. Callus induction, biomass growth, and plant regeneration in Digitalis lanata Ehrh.: Influence of plant growth regulators and carbohydrates. Turk. J. Bot. 2009, 33, 393–405. [Google Scholar] [CrossRef]
- Yücesan, B.; Müller-Uri, F.; Kreis, W.; Gürel, E. Cardenolide estimation in callus-mediated regenerants of Digitalis lamarckii Ivanina (dwarf foxglove). Vitr. Cell. Dev. Biol.-Plant 2014, 50, 137–142. [Google Scholar] [CrossRef]
- Verma, S.K.; Sahin, G.; Gurel, E. Somatic embryogenesis, pigment accumulation, and synthetic seed production in Digitalis davisiana Heywood. Indian J. Exp. Biol. 2016, 54, 245–253. [Google Scholar] [PubMed]
- Mamgain, J.; Mujib, A.; Syeed, R.; Ejaz, B.; Malik, M.Q.; Bansal, Y. Genome size and gas chromatography-mass spectrometry (GC–MS) analysis of field-grown and in vitro regenerated Pluchea lanceolata plants. J. Appl. Genet. 2022, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Flores, R.; Maldaner, J.; Brondani, D.; Cezarotto, V.; Giacomelli, S.; Garlet, T.; Nicoloso, F. Direct and indirect regeneration from Pfaffia tuberosa (Spreng.) Hicken calli and its β-ecdysone production. Rev. Bras. Pl. Med. 2017, 19, 266–273. [Google Scholar]
- Hesami, M.; Daneshvar, M.H. Indirect Organogenesis through Seedling-Derived Leaf Segments of Ficus Religiosa—A Multipurpose Woody Medicinal Plant. J. Crop Sci. Biotechnol. 2018, 21, 129–136. [Google Scholar] [CrossRef]
- Banu, T.A.; Khan, S.; Goswami, B.; Afrin, S.; Habib, A.; Akter, S. Indirect organogenesis and somatic embryogenesis for regeneration of Rauvolfia serpentina L. from root explants. Bangladesh J. Bot. 2020, 49, 1021–1027. [Google Scholar] [CrossRef]
- Okello, D.; Yang, S.; Komakech, R.; Chung, Y.; Rahmat, E.; Gang, R.; Omujal, F.; Lamwaka, A.V.; Kang, Y. Indirect in vitro regeneration of the medicinal plant, Aspilia africana, and histological assessment at different developmental stages. Front. Plant Sci. 2021, 12, 2928. [Google Scholar] [CrossRef] [PubMed]
- Mani, T.; Senthil, K. Multiplication of Chrysanthemum through somatic embryogenesis. Asian J. Pharma Technol. 2011, 1, 13–16. [Google Scholar]
- Ouyang, Y.; Chen, Y.; Lü, J.; Teixeira da Silva, J.A.; Zhang, X.; Ma, G. Somatic embryogenesis and enhanced shoot organogenesis in Metabriggsia ovalifolia W. T. Wang. Sci. Rep. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- MusfirMehaboob, V.; Faizal, K.; Thilip, C.; Raja, P.; Thiagu, G.; Aslam, A.; Shajahan, A. Indirect somatic embryogenesis and Agrobacterium-mediated transient transformation of ginger (Zingiber officinale Rosc.) using leaf sheath explants. J. Hortic. Sci. Biotechnol. 2019, 94, 753–760. [Google Scholar] [CrossRef]
- Mujib, A.; Bansal, Y.; Malik, M.Q.; Syeed, R.; Mamgain, J.; Ejaz, B. Internal and external regulatory elements controlling somatic embryogenesis in Catharanthus: A model medicinal plant. In Somatic Embryogenesis; Ramírez-Mosqueda, M.A., Ed.; Humana: New York, NY, USA, 2022; Volume 2527, pp. 11–27. [Google Scholar] [CrossRef]
- Mazri, M.A.; Naciri, R.; Belkoura, I. Maturation and conversion of somatic embryos derived from seeds of Olive (Olea europaea L.) cv. Dahbia: Occurrence of secondary embryogenesis and adventitious bud formation. Plants 2020, 9, 1489. [Google Scholar] [CrossRef] [PubMed]
- Sinchana, N.S.; Kattimani, K.N.; Prabhuling, G.; Sudesh, K.; Jagadeesha, N. Standardization of tissue culture protocol for turmeric (Curcuma longa L.) Cv. Salem. Int. J. Chem. Stud. 2020, 8, 2721–2726. [Google Scholar]
- Jena, S.; Ray, A.; Sahoo, A.; Sahoo, S.; Dash, B.; Kar, B.; Nayak, S. Rapid plant regeneration in industrially important Curcuma zedoaria revealing genetic and biochemical fidelity of the regenerants. 3 Biotech 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chirumamilla, P.; Gopu, C.; Jogam, P.; Taduri, S. Highly efficient rapid micropropagation and assessment of genetic fidelity of regenerants by ISSR and SCoT markers of Solanum khasianum Clarke. Plant Cell Tiss. Organ Cult. 2021, 144, 397–407. [Google Scholar] [CrossRef]
- Jena, S.; Ray, A.; Sahoo, A.; Sahoo, S.; Kar, B.; Panda, P.C.; Nayak, S. High-frequency clonal propagation of Curcuma angustifolia ensuring genetic fidelity of micropropagated plants. Plant Cell Tiss. Organ Cult. 2018, 135, 473–486. [Google Scholar] [CrossRef]
- Raji, M.R.; Farajpour, M. Genetic fidelity of regenerated plants via shoot regeneration of muskmelon by inter simple sequence repeat and flow cytometry. J. Saudi Soc. Agric. Sci. 2020, 20, 88–93. [Google Scholar] [CrossRef]
- Ulvrova, T.; Vitamvas, J.; Cepkova, P.H.; Eliasova, K.; Janovska, D.; Bazant, V.; Viehmannova, I. Micropropagation of an ornamental shrub Disanthus cercidifolius Maxim. and assessment of genetic fidelity of regenerants using ISSR and flow cytometry. Plant Cell Tiss. Organ Cult. 2021, 144, 555–566. [Google Scholar] [CrossRef]
- Sathish, D.; Vasudevan, V.; Theboral, J.; Elayaraja, D.; Appunu, C.; Siva, R.; Manickavasagam, M. Efficient direct plant regeneration from immature leaf roll explants of sugarcane (Saccharum officinarum L.) using polyamines and assessment of genetic fidelity by SCoT markers. Vitr. Cell Dev. Biol. Plant 2018, 54, 399–412. [Google Scholar] [CrossRef]
- Sharma, U.; Rai, M.K.; Shekhawat, N.S.; Kataria, V. Genetic homogeneity revealed in micropropagated Bauhinia racemosa Lam. using gene targeted markers CBDP and SCoT. Physiol. Mol. Biol. Plants 2019, 25, 581–588. [Google Scholar] [CrossRef]
- Tikendra, L.; Potshangbam, A.M.; Dey, A.; Devi, T.R.; Sahoo, M.R.; Nongdam, P. RAPD, ISSR, and SCoT markers based genetic stability assessment of micropropagated Dendrobium fimbriatum Lindl. var. oculatum Hk. f.-an important endangered orchid. Physiol. Mol. Biol. Plants 2021, 27, 341–357. [Google Scholar] [CrossRef]
- Mood, K.; Jogam, P.; Sirikonda, A.; Shekhawat, M.S.; Rohela, G.K.; Manokari, M.; Allini, V.R. Micropropagation, morpho-anatomical characterization, and genetic stability studies in Lippia javanica (Burm. f.) Spreng: A multipurpose medicinal plant. Plant Cell Tiss. Organ Cult. 2022, 150, 427–437. [Google Scholar] [CrossRef]



| Medium | Germination Rate (%) |
|---|---|
| Half MS | 48.9 ± 4 b |
| Full MS | 62.2 ± 1.1 a |
| Full MS + 0.5 mg/L GA3 | 73.3 ± 3.8 a |
| PGRs | Concentration (mg/L) | Explants | ||
|---|---|---|---|---|
| Hypocotyl | Node | Leaf | ||
| Control | 0 | 0 g | 0 i | 0 i |
| BAP | 0.5 | 51.4 ± 10.8 c | 9.7 ± 2.8 h,i | 12.5 ± 2.4 f,g,h,i |
| 0.75 | 90.3 ± 1.4 a | 19.4 ± 3.7 g,h | 20.8 ± 4.2 e,f,g,h | |
| 1 | 58.3 ± 4.2 b,c | 11.1 ± 1.4 h,i | 6.9 ± 3.7 g,h,i | |
| 2 | 30.6 ± 2.8 d,e,f | 12.5 ± 2.4 h,i | 5.6 ± 1.4 h,i | |
| BAP + 2,4-D | 0.5 + 1.0 | 18.1 ± 6.1 f,g | 22.2 ± 3.7 g,h | 13.9 ± 3.7 f,g,h,i |
| 0.5 + 2.0 | 40.3 ± 10 c,d,e | 26.4 ± 3.7 f,g,h | 25 ± 6.4 d,e,f,g,h | |
| 1.0 + 0.5 | 45.8 ± 13.4 c,d | 36.1 ± 5 f,g | 29.2 ± 2.4 d,e,f | |
| 1.0 + 2.0 | 50 ± 10.5 c | 43.1 ± 5 e,f | 37.5 ± 4.8 c,d,e | |
| 2.0 + 0.5 | 0 ± 0 g | 84.7 ± 6.9 a | 77.8 ± 6.1 a | |
| 2.0 + 1.0 | 0 ± 0 g | 58.3 ± 4.2 c,d,e | 54.2 ± 10.5 b,c | |
| BAP + NAA | 0.5 + 1.0 | 25 ± 2.4 e,f | 54.2 ± 6.4 d,e | 26.4 ± 1.4 d,e,f,g |
| 0.5 + 2.0 | 70.8 ± 4.2 b | 56.9 ± 9.7 c,d,e | 43.1 ± 10.8 b,c,d | |
| 1.0 + 0.5 | 12.5 ± 2.4 f,g | 68.1 ± 7.3 a,b,c,d | 29.2 ± 4.8 d,e,f | |
| 1.0 + 2.0 | 0 ± 0 g | 65.3 ± 10.8 b,c,d | 59.7 ± 13.2 b | |
| 2.0 + 0.5 | 0 ± 0 g | 73.6 ± 7.3 a,b,c | 93.1 ± 1.4 a | |
| 2.0 + 1.0 | 0 ± 0 g | 81.9 ± 1.4 a,b | 84.7 ± 5 a | |
| PGRs | Concentration (mg/L) | Frequency (%) of Somatic Embryogenesis | Mean Number of SEs/Callus Mass (250 mg) |
|---|---|---|---|
| Control | 0 | 0 g | 0 f |
| BAP | 0.5 | 56.9 ± 7.3 b | 11.3 ± 0.9 a,b |
| 0.75 | 81.9 ± 3.7 a | 13.7 ± 2.3 a | |
| 1.0 | 45.8 ± 6.4 b,c | 8.3 ± 1.9 b,c | |
| BAP + 2,4-D | 0.5 + 1.0 | 18.1 ± 3.7 e,f | 2.3 ± 0.3 d,e,f |
| 0.75 + 0.5 | 9.7 ± 2.8 f,g | 0.7 ± 0.3 e,f | |
| 1.0 + 0.5 | 0 ± 0 g | 0 ± 0 f | |
| BAP + NAA | 0.5 + 1.0 | 34.7 ± 9.7 c,d | 5.7 ± 1.5 c,d |
| 0.75 + 0.5 | 55.6 ± 3.7 b | 11.7 ± 1.3 a,b | |
| 1.0 + 0.5 | 26.4 ± 3.7 d,e | 4.3 ± 0.9 d,e |
| PGRs | Concentration (mg/L) | Shoot Proliferation Frequency (%) | Mean No. of shoots/explant | Length of Shoots (cm) | |||
|---|---|---|---|---|---|---|---|
| Apical Bud | Cotyledonary Node | Apical Bud | Cotyledonary Node | Apical Bud | Cotyledonary Node | ||
| Control | 0 | 0 f | 0 e | 0 d | 0 e | 0 e | 0e |
| BAP | 0.5 | 45.8 ± 4.8 c,d | 18.1 ± 6.1 d | 4.7 ± 0.9 ab | 2.7 ± 0.3 bc | 3.7 ± 0.3 a | 2.1 ± 0.2 b,c |
| 0.75 | 77.8 ± 5.6 b | 81.9 ± 6.9 a | 5.3 ± 0.3 a | 4.7 ± 0.9 ab | 3.0 ± 0.5 a,b | 3.7 ± 0.2 a | |
| 1.0 | 93.1 ± 1.4 a | 87.5 ± 2.4 a | 5.3 ± 0.7 a | 5.7 ± 0.9 a | 3.8 ± 0.4 a | 4.1 ± 0.3 a | |
| 2.0 | 26.4 ± 1.4 e | 37.5 ± 2.4 c | 3.3 ± 0.3 bc | 3.7 ± 0.7 abc | 1.9 ± 0.1 c,d | 3.8 ± 0.2 a | |
| Kn | 0.5 | 11.1 ± 1.4 f | 8.3 ± 4.2 d,e | 2.0 ± 1.2 c | 0.3 ± 0.3 de | 1.2 ± 0.2 d | 1.2 ± 0.2 d |
| 0.75 | 58.3 ± 8.7 c | 44.4 ± 5 b,c | 4.3 ± 0.3 ab | 4.3 ± 0.9 abc | 2.2 ± 0.2 b,c | 2.3 ± 0.3 b,c | |
| 1.0 | 34.7 ± 5 d,e | 58.3 ± 4.2 b | 3.7 ± 0.3 abc | 5.0 ± 0.6 a | 3.4 ± 0.3 a | 2.7 ± 0.3 b | |
| 2.0 | 29.2 ± 2.4 e | 20.8 ± 6.4 d | 2.3 ± 0.3 c | 2.3 ± 0.9 cd | 2.0 ± 0.2 c,d | 2.0 ± 0.1 c | |
| PGRs | Concentration (mg/L) | Frequency of Organogenesis (%) | Mean No. of Shoots/Callus Mass | ||
|---|---|---|---|---|---|
| Leaf | Hypocotyl | Leaf | Hypocotyl | ||
| Control | 0 | 0 d | 0 c | 0 e | 0 b |
| BAP + 2,4-D | 0.5 +1.0 | 18.1 ± 6.1 c,d | 8.3 ± 4.2 c | 2.3 ± 0.3 d | 2.3 ± 1.2 a,b |
| 0.75 + 1.0 | 43.1 ± 10 b | 6.9 ± 1.4 c | 4.3 ± 0.7 c,d | 1.3 ± 0.3 a,b | |
| 1.0 + 0.5 | 56.9 ± 3.7 a,b | 22.2 ± 1.4 b | 7.3 ± 0.9 b | 4.3 ± 1.9 a | |
| 1.5 + 0.75 | 37.5 ± 4.8 b,c | 29.2 ± 2.4 b | 4.7 ± 0.9 c | 2.3 ± 0.9 a,b | |
| 2.0 + 0.5 | 70.8 ± 8.7 a | 40.3 ± 3.7 a | 10.3 ± 0.9 a | 3.7 ± 1.2 a,b | |
| Plant Sample Types | 2C DNA Content (pg *) | 1C DNA Content (pg) | Genome Size (Mbp) |
|---|---|---|---|
| In vivo (donor) plant | 3.1 ± 0.01 a | 1.55 ± 0.0 a | 3031.8 a |
| Somatic embryo regenerated plant | 3.2± 0.01 a | 1.6 ± 0.01 a | 3129.6 a |
| Callus-mediated plant | 3.0 ± 0.01 a | 1.5 ± 0.0 a | 2934 a |
| S. No | Primer Name | Primer Sequences (5′-3′) | %G/C | Tm (°C) | No. of Bands Amplified | Approximate Band Length (bp) | |
|---|---|---|---|---|---|---|---|
| DP_1 * | DP_2 * | ||||||
| 1 | SCoT3 | CAACAATGGCTACCACCG | 56 | 48 °C | 10 | 10 | 300–1400 |
| 2 | SCoT7 | CAACAATGGCTACCACGG | 56 | 48 °C | 5 | 5 | 300–1500 |
| 3 | SCoT14 | ACGACATGGCGACCACGC | 56 | 48 °C | 6 | 6 | 300–1250 |
| 4 | SCoT16 | ACCATGGCTACCACCGAC | 56 | 48 °C | 7 | 6 | 300–1400 |
| 5 | SCoT26 | ACCATGGCTACCACCGTC | 61 | 50 °C | 5 | 4 | 500–1500 |
| 6 | SCoT33 | CCATGGCTACCACCGCAG | 67 | 50 °C | 4 | 4 | 200–1600 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bansal, Y.; Mujib, A.; Siddiqui, Z.H.; Mamgain, J.; Syeed, R.; Ejaz, B. Ploidy Status, Nuclear DNA Content and Start Codon Targeted (SCoT) Genetic Homogeneity Assessment in Digitalis purpurea L., Regenerated In Vitro. Genes 2022, 13, 2335. https://doi.org/10.3390/genes13122335
Bansal Y, Mujib A, Siddiqui ZH, Mamgain J, Syeed R, Ejaz B. Ploidy Status, Nuclear DNA Content and Start Codon Targeted (SCoT) Genetic Homogeneity Assessment in Digitalis purpurea L., Regenerated In Vitro. Genes. 2022; 13(12):2335. https://doi.org/10.3390/genes13122335
Chicago/Turabian StyleBansal, Yashika, A. Mujib, Zahid H. Siddiqui, Jyoti Mamgain, Rukaya Syeed, and Bushra Ejaz. 2022. "Ploidy Status, Nuclear DNA Content and Start Codon Targeted (SCoT) Genetic Homogeneity Assessment in Digitalis purpurea L., Regenerated In Vitro" Genes 13, no. 12: 2335. https://doi.org/10.3390/genes13122335
APA StyleBansal, Y., Mujib, A., Siddiqui, Z. H., Mamgain, J., Syeed, R., & Ejaz, B. (2022). Ploidy Status, Nuclear DNA Content and Start Codon Targeted (SCoT) Genetic Homogeneity Assessment in Digitalis purpurea L., Regenerated In Vitro. Genes, 13(12), 2335. https://doi.org/10.3390/genes13122335