1. Introduction
The results of studies reported in the literature show a fairly common presence of pharmaceuticals in drinking water [
1,
2,
3,
4,
5,
6]. Classical water treatment techniques are generally inadequate for the removal of pharmaceuticals. Combined treatment techniques, such as advanced oxidation processes (AOPs) and membrane separation (in particular nanofiltration), are technologies for possibly complete removal of these pollutants. However, the oxidation products may go through the membrane and influence the retention degree of pollutants [
7].
Salicylic acid and its derivatives are often used in the pharmaceutical, cosmetic, and food industries for food preservation. Salicylic acid, as a key ingredient in many skin care products, is regularly detected in wastewater and surface waters, being the so-called pharmaceutical pollution [
8,
9]. Its concentration in the tested samples taken from different sources ranges from a few to even 977 ng/L [
10,
11]. This aromatic organic compound is toxic to organisms that live in water. Salicylic acid is a product of the decomposition of aspirin (acetylsalicylic acid), one of the most widely used medicines in the world. At ambient temperature, acetylsalicylic acid is slowly hydrolyzed to salicylic acid and acetic acid [
12,
13].
Therefore, it is very important to develop appropriate methods to remove salicylic acid from the environment and to degrade this dangerous, biologically active compound. Particularly promising are advanced oxidation and membrane filtration. The nanofiltration process enables effective separation of dissolved low molecular weight substances, such as divalent inorganic salts and organic molecules. The aim of the process is to separate hazardous contaminants on the membrane, that is, to obtain a clear filtrate and concentrate containing the retained substances. The methods of advanced oxidation are used to chemically modify oxidized molecules. The integration of both methods allows us to use their positive features while reducing disadvantages.
Advanced oxidation processes provide chemical modification of hazardous substances to non-toxic forms, while membrane filtration ensures removal of substrates and their oxidation products from water. As a result of combining both methods, a pure filtrate and concentrate containing oxidized products of hazardous materials are obtained.
Salicylic acid (SA) is characterized by high reaction rate with OH radical (5 × 10
9 M
−1 s
−1). Reaction products are stable, which makes it possible to perform their qualitative analysis easily [
14]. Main products resulting from the reaction of salicylic acid with hydroxyl radicals are 2,3-dihydroxybenzoic acid (2,3-DHBA), 2,5-dihydroxybenzoic acid (2,5-DHBA), and catechol [
9,
15]. Karnik [
15] successfully determined two products of the oxidation of salicylic acid with ozone, that is, 2,3-DHBA and 2,5-DHBA, with 2,3-DHBA being the dominant reaction product. Chen et al. [
9] obtained a similar result in the process of salicylic acid oxidation with Fenton reagent and UV/H
2O
2. The main product was 2,3-DHBA, while 2,5-DHBA and catechol were produced in small amounts.
Filtration processes are widely used in many areas of life, in the food and pharmaceutical industries, as well as in environmental protection [
3,
16,
17,
18,
19]. However, the cost of these processes is high which is related, among other things, to the high sensitivity of membranes to pollutants.
The effectiveness of retention of a given compound in the membrane filtration process depends on many factors, for example, chemical structure and membrane properties, structure, charge and molecular weight of the compound being removed, and its affinity for the membrane. In the research dedicated to the removal of pharmaceuticals in the membrane filtration process, three mechanisms of their removal have been identified [
20], that is, retention of molecules due to their size—the molecule diameter must be significantly larger than the membrane pore diameter, hydrophobic interaction (adsorption of particles on the membrane), and electrostatic repulsion.
The initial adsorption efficiency gradually decreases and then stabilizes, when the process reaches equilibrium state [
1]. After reaching the equilibrium state, adsorption can have a disadvantageous effect on retention because it has been shown that adsorbed compounds can dissolve in the active membrane layers, then diffuse through the polymer and finally desorb to the membrane layer on the filtrate side [
21]. In addition, desorption of adsorbed compounds to the membrane from the filtrate side may also occur if the compound concentration in the feed water is lower than the equilibrium concentration [
22].
Comerton et al. [
1] studied the effect of various factors on the adsorption of chemical compounds on a polymer membrane. They found that there was a correlation between the solubility of a given compound and its adsorption on the membrane. Adsorption would increase with decreasing water solubility and increasing hydrophobicity of the tested compound. Karnik et al. [
13] showed that the use of membrane filtration at pH from 2.5 to 3 did not cause a significant decrease in the concentration of salicylic acid. This suggests that salicylic acid is not adsorbed on the membrane surface at low pH (2.5–3). In the initial stage, the concentration of salicylic acid in the filtrate decreased, then after reaching the minimum, it grew slightly. This may be due to the fact that after this time, the available sorption sites on the surface and/or within the membrane have become saturated.
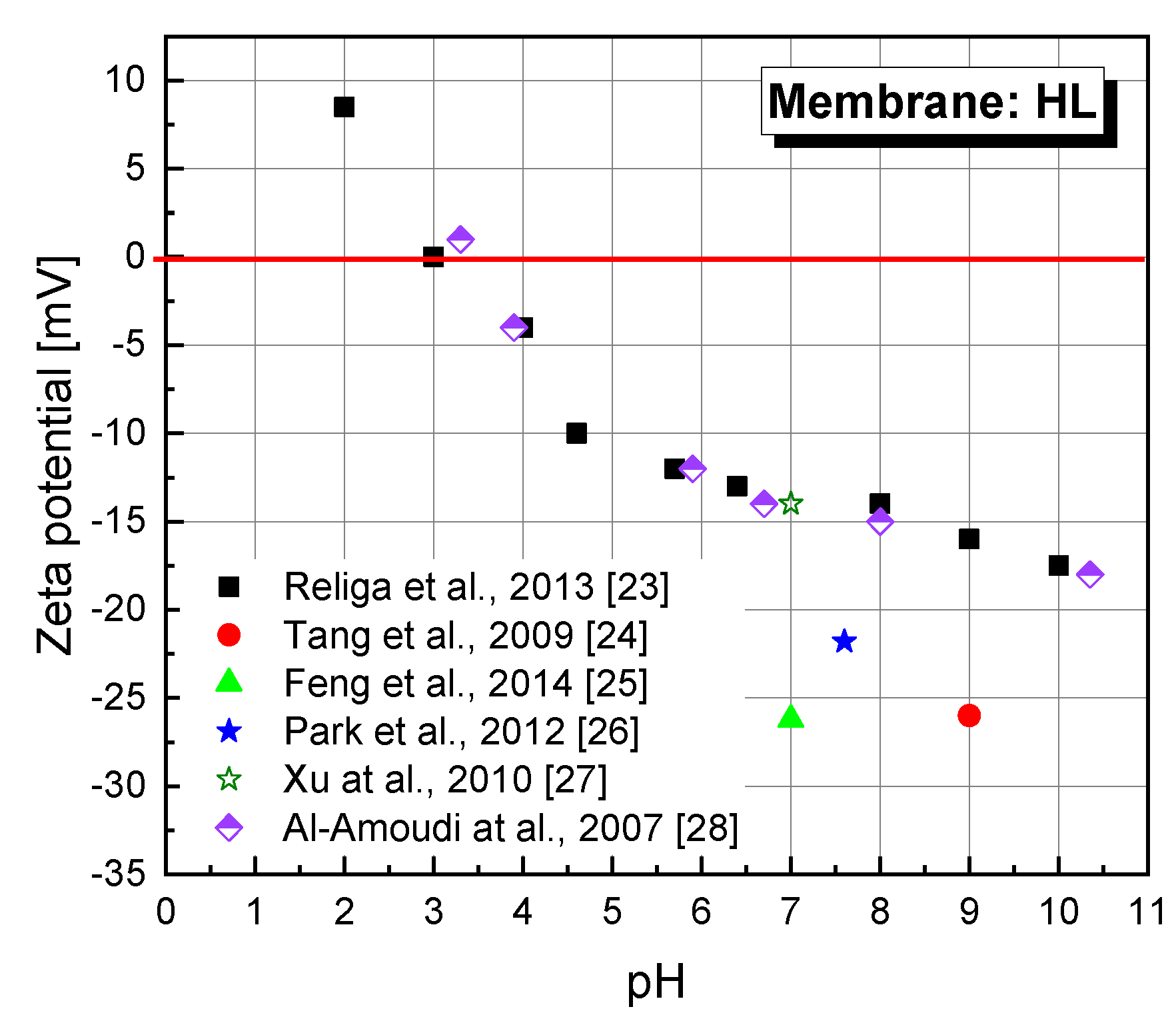
An important factor affecting the efficiency and separation mechanism is the charge of the membrane surface. The tests have shown that thin-film polymer composite membranes are negatively charged at neutral pH due to deprotonation of acidic functional groups on the membrane surface [
23,
24,
25,
26,
27,
28] (
Figure 1).
Acidic functional groups are deliberately introduced during the preparation of the membrane in order to increase its selectivity and permeability. Filtration efficiency is usually lower for positively charged (basic) particles of medicines, while the highest efficiency is obtained for pharmaceuticals that are negatively charged (acidic) [
20].
Another membrane property that has a significant impact on the mechanism of separation of organic compounds is its hydrophilicity/hydrophobicity. A parameter that makes it possible to determine the degree of hydrophobicity of a given surface is its contact angle. In the literature, the HL membrane is considered to be one of the most hydrophilic [
24,
29,
30,
31]. In most works, the contact angle, determined by the embedded drop method with the use of pure water as a medium, for this membrane ranges from approx. 27 to approx. 36° [
24,
25,
26,
31]. However, there are reports that the HL membrane is much more hydrophobic. Al-Amoudi et al. [
32] stated that their HL membrane had a contact angle of 56.7°. Park et al. [
29] in their earlier work determined the contact angle for the DESAL HL membrane as equal to 50.9°.
Integration of advanced methods, such as nanofiltration/reverse osmosis and advanced chemical oxidation using ozonation, O
3/UV, H
2O
2/UV, and O
3/H
2O
2, are currently the latest research direction, allowing for a complete solution to the problem of biologically active substances commonly found in waters around the world. The great majority of works are focused on achieving the most effective removal of initial compounds using the methods of advanced oxidation and membrane techniques (nanofiltration/reverse osmosis) without a deeper analysis of what is happening to their degradation products [
33,
34,
35,
36,
37,
38,
39]. The influence of oxidation products on the performance and yield of the membranes has not been studied so far. Therefore, the aim of this work was to determine the impact of oxidation products of salicylic acid on the effectiveness of its further removal in the membrane filtration process.
3. Results and Discussion
3.1. UV/H2O2 Oxidation
First, experiments with UV/H
2O
2 oxidation of selected compounds were carried out.
Figure 2 shows the dependence of the concentration of salicylic acid (SA). UV radiation alone caused a slight decrease in the concentration of salicylic acid constituting 10% of the initial value. Hydrogen peroxide at a concentration of 2.86 × 10
−3 mol/L introduced into the system caused considerable acceleration of salicylic acid decomposition. Increasing the dose of hydrogen peroxide to 7.15 × 10
−3 mol/L resulted in further acceleration of salicylic acid decomposition. After 30 min, an 80% reduction of the tested compound was obtained, whereas, after 60 min, salicylic acid remained in trace amounts.
On the basis of the HPLC analysis, the concentrations of oxidation products of salicylic acid, subjected to UV/H
2O
2, were identified and determined. These products were further subjected to nanofiltration. The tests were performed at a dose of H
2O
2 equal to 2.86 × 10
−3 mol/L after 60 min of exposure of salicylic acid to oxidizing agents. Three intermediates were identified whose concentrations are summarized in
Table 3. As a result of the reaction of 75% salicylic acid, 14.8 mg/L of 2,3 dihydroxybenzoic acid (2,3-DHBA), 5.1 mg/L of catechol, and trace amounts of 2,5-dihydroxybenzoic acid (2,5-DHBA) were obtained.
Figure 3 shows structural formulas of salicylic acid and identified products of its oxidation.
The results are consistent with the results published by other researchers [
9,
13]. Based on the analysis of the concentration of the resulting products and the degree of salicylic acid conversion, it was estimated that oxidation products, such as 2,3 DHBA, 2,5-DHBA, and catechol, constitute about 40% of all oxidation products.
3.2. Nanofiltration
The nanofiltration process was carried out using two polyamide-based polymer membranes: HL and TS80.
Figure 4A shows retention coefficients of salicylic acid for varying degrees of concentration of the initial solution (white bar) on the HL membrane. The measurements were taken at the beginning of the process (zero degree concentration), obtaining 500 mL of filtrate from 3000 mL of the initial bath (concentration degree 1/6), and analogously—obtaining 1000 mL of filtrate (concentration degree 1/3) and 1500 mL of filtrate (concentration degree 1/2).
Figure 4B,C show the concentration of salicylic acid in the concentrate and filtrate, respectively, depending on the degree of initial bath concentration.
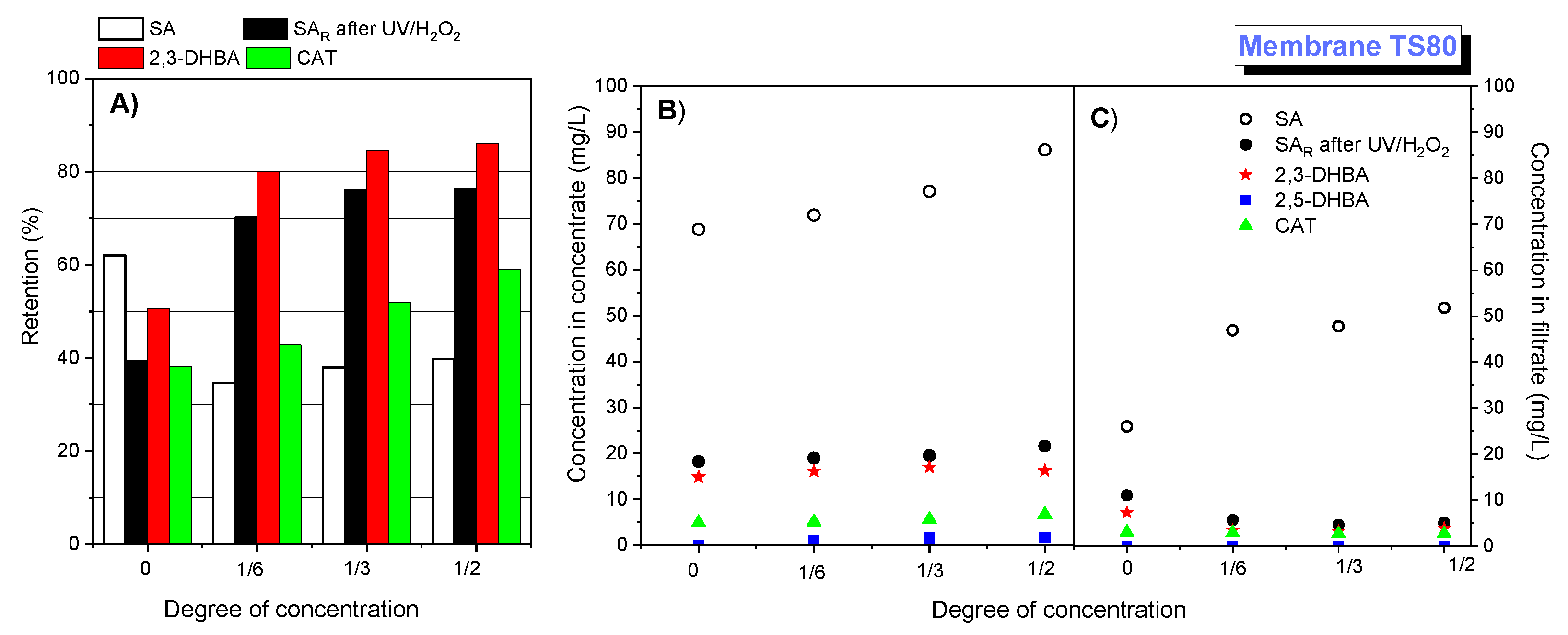
Figure 5 presents the results obtained with the TS80 membrane.
Salicylic acid retention in the initial reaction step was 37.6% and gradually decreased to 22.8%. Salicylic acid is characterized by a relatively low molecular weight (138.12 g/mol). The molecule of this acid does not contain additional spatial substituents besides the carbonyl group. The HL membrane is characterized by MWCO (Molecular weight cut-off) in the range of 150–300 (Da). It should be noted that the tests were carried out at native pH of the solution, that is, pH = 3.5. The dissociation constant of salicylic acid is pK
a = 2.97. It is known from the literature that HL membrane at this pH has a zeta potential close to zero [
23,
24,
25,
26,
27,
28] (
Figure 1). However, the results of some studies indicate that the DESAL HL membrane can be negatively charged at pH 3 (ca. −10 mV, measurements with 10 mM KCl, [
45]). The low molar mass of salicylic acid which is lower than the MWCO and unfavorable conditions for electrostatic repulsion cause low retention of salicylic acid. One of the phenomena that seem to be important in the first stage of the filtration is the adsorption of salicylic acid molecules on the membrane. For the adsorption process, there is a gradual decrease in the filtration efficiency because molecule retention on the membrane by sorption is a short-time incident. Molecules move through the membrane along its pores and pass into the filtrate. Probably the sorption process can take place until active areas of the membrane surface are saturated and then, after 1/3 degree of concentration, the retention percentage is constant on both membranes (
Figure 4A and
Figure 5A).
Figure 6 shows the dependence of the filtrate flux on filtration time. In the case of the HL membrane, a very small filtrate flux decrease was observed, approx. 5% at pH = 3.5.
In the case of the TS80 membrane, the salicylic acid separation efficiency was evidently higher.
Figure 5A shows the retention of salicylic acid (white bar) depending on the degree of initial solution concentration.
Figure 5B,C show the concentration of salicylic acid in the concentrate and filtrate, respectively, depending on the degree of concentration. The salicylic acid retention in the initial stage was 62.2% and gradually decreased to 39.8%. Higher retention may result from the fact that the TS80 membrane is more compact (MWCO 150 Da) and rougher (RMS (Root Mean Squared roughness) 89 nm) than the HL membrane [
1]. Higher filtration efficiency may result from the fact that the TS80 membrane is negatively charged at pH 3 [
45]. For the TS80 membrane, the filtrate flux was lower than for the HL membrane (
Figure 6). It was observed that at pH 3.5, the filtrate flux decline was slightly higher than for the HL membrane, amounting to about 11%.
Figure 7 presents SEM micrographs with a magnification of 20,000× prepared using an electron scanning microscope. On the basis of microscopic images, it can be concluded that the TS80 membrane has more undulated and uneven surface than the HL membrane. The HL membrane is characterized by a smooth surface, as evidenced by the micrograph shown in
Figure 7A. Its RMS roughness has been determined by other researchers as about 10 nm [
30,
43]. It should be taken into account that retention of oxidation products on the TS80 membrane, in particular, 2,3 DHBA, is much higher than for pure SA, in contrast to the HL membrane. This phenomenon is discussed further.
The HL and TS80 membranes were characterized by similar values of the contact angle. The contact angle for the HL membrane was 59.0° (for water), while for the TS80 membrane was 53.4° (for water). Both membranes can be classified as hydrophilic.
3.3. Retention of Oxidation Products
As a result of the investigation with the use of the HL membrane for nanofiltration of oxidized salicylic acid solutions, it was found that one of the oxidation products, namely 2,3-dihydroxybenzoic acid, showed similar retention coefficients to salicylic acid, while the other product, catechol, showed very low retention ability on the membrane (
Figure 4A). Probably this was due to the fact that the structure of 2,3-dihydroxybenzoic acid was not different from that of salicylic acid molecules, with the exception of the attached –OH group in the ortho position, while in catechol molecules, the −COOH group was replaced by –OH during SA oxidation. Similar results were obtained for the TS80 membrane, for which an analogous comparison of results is shown in
Figure 5A. Also, in the case of this membrane, it was observed that the main oxidation product, 2,3-DHBA, had similar retention coefficients as SA, while the efficiency of separation of catechol molecules on the TS80 membrane was much worse than that of the above-mentioned compounds.
This is important from the point of view of environmental protection. Using integrated methods of advanced oxidation and nanofiltration processes for removing dangerous pollutants from wastewater, it should be noted that despite a significant decomposition of the starting compound, we can allow the emission oxidation products whose chemical structure and low molecular weight contribute to the low efficiency of separation on the membrane. Although catechol is a biodegradable compound, it has been shown that there is a risk that some more dangerous oxidation products may penetrate the permeate in water treatment processes. Therefore, such research should be carried out, and potential risks resulting from the integration of advanced oxidation and membrane techniques should be analyzed.
3.4. The Influence of Oxidation Products on the Retention of Residual Salicylic Acid
In the case of the HL membrane, it was observed that, at the beginning of the filtration process, there was a slight increase in the retention of undecomposed salicylic acid in the reaction mixture compared to the retention of pure salicylic acid solution. After concentrating the solution to 1/6 of the initial volume, a slight but permanent deterioration of the separation efficiency of the membrane for this acid was observed. A small but noticeable decrease of salicylic acid retention in the reaction mixture was reported compared to the pure salicylic acid solution.
In the case of the TS80 membrane, a marked improvement in the filtration efficiency of salicylic acid was observed in the presence of its oxidation products (
Figure 5). During the concentration of the initial solution, retention of salicylic acid and its oxidation products clearly increased. The TS80 membrane is more compact (lower MWCO value) and rougher than the HL membrane, which can contribute to more efficient retention of oxidation products. This may be particularly important for low molecular weight oxidation products, for example, glyoxylic, maleic, fumaric, malic, oxalic, and tartronic acids, which on passing through the membrane can be blocked inside the membrane pores, gradually contributing to improved separation of the tested compounds on the membrane.
In the next stage of research, an experiment was carried out by adding 2,3-DHBA at a concentration of 3.24 × 10
−4 mol/L (50 mg/L) to SA solution and catechol at a concentration of 3.24 × 10
−4 mol/L (35.72 mg/L) to the other SA solution. The solutions prepared in this way were nanofiltered on the HL membrane, and then calculated retention coefficients of salicylic acid were compared with the values obtained for pure SA solution. The results are shown in
Figure 8.
If results for the HL membrane at pH = 3.5 are considered, it is found that 2,3-dihydroxybenzoic acid had a negative impact on the retention of salicylic acid, which is evidenced by a slight decrease in the retention coefficient of this acid in the presence of 2,3-DHBA (
Figure 8A). Despite the low molecular weight of salicylic acid which is lower than the MWCO, apart from the adsorption phenomenon, the size exclusion mechanism and the interference effect of oxidation products may affect the SA separation process. 2,3-DHBA can be a competitive compound to the active sites of the membrane, causing weaker adsorption of SA on it. The tests showed that the retention coefficients of 2,3-DHBA in the above experiment were close to the coefficients for salicylic acid and ranged from 20 to 28% depending on the concentration of the initial solution (results not shown in the Figures).
In the case of the TS80 membrane, the negative effect of 2,3-DHBA is visible only in the first stage of the process (
Figure 8B). In the final stage, with a concentration of 1:2, the presence of both oxidation products improved SA retention coefficients. This may be due to the fact that in the initial stage, the adsorption process dominates, while during the concentration, the separation through spatial blocking and electrostatic repulsion becomes dominant. In the case of TS80 membrane, at pH 3, the membrane surface is charged negatively (ca. −20 mV, measurement with 10mM KCl, [
45]), and consequently, the effect of electrostatic interaction should be taken into account. The results are consistent with
Figure 5. During concentrate concentration, higher SA retention coefficients were obtained compared to pure SA solution.
If the results for pH = 8.0 are considered, it is found that 2,3-DHBA did not affect the salicylic acid separation process (
Figure 8C,D). This may be due to the fact that at pH = 8, the mechanism of separation by electrostatic repulsion dominates. With this mechanism, the oxidation products do not compete with the starting substrate.
It was observed that in the case of catechol, there was a tendency to improve the SA retention coefficient regardless of the type of membrane and pH. This may be due to the fact that catechol is blocked inside the pores of the membrane, reducing the voids, while not blocking the active sites on the membrane surface.
Table 4 summarizes the pH and conductivity values of the concentrate and filtrate during the filtration process on the HL membrane. The conductivity of the SA solution concentrates ranged from 153 to 165 μS/cm. The presence of 2,3-DHBA caused an increase in the conductivity of the SA solution ranging from 208 to 251 μS/cm. 2,3-DHBA had a slightly higher acidity (pK
a = 2.91) than SA due to the presence of an additional OH group in the aromatic ring. The presence of two acids in the solution caused weaker dissociation (the conductivity of the SA + 2,3-DHBA mixture was lower than the sum of the values obtained for pure solutions of these acids). The poorer dissociation of SA could, to a certain extent, influence the effectiveness of its separation on the membrane. The presence of catechol had no effect on the conductivity of concentrates and filtrates. This was due to the low dissociation of catechol (pK
a = 9.45). Solutions containing catechol had a slightly higher pH.
Piperazine-based polyamide membranes, which include the HL membrane, have a charge on their surface as a result of the presence of amino and carboxyl groups. Typical membranes of this type have an isoelectric point within pH 3–4 [
23,
28]. At lower pH, amine groups are activated (ammonium ions are formed), which causes a positive charge on the membrane surface. At pH above 3–4, carboxyl groups of the polyamide are dissociated, resulting in a negative charge. Zeta potential studies revealed that the membrane used for SA filtration showed a significant decrease in the negative charge on the membrane surface (at pH 5.2, zeta potential was −2.5 mV, at pH 7.9, it was −7.0 mV) as compared to the pure HL membrane (
Figure 1). The presence of 2,3-DHBA in the SA solution caused a further shift towards positive values of the zeta potential. This may be due to the fact that the acidity of carboxyl groups of SA and 2,3-DHBA is greater than that of carboxyl groups on the membrane surface. In the case of a system in which a stronger and weaker acid is present, the H
+ ions combine with the weak acid residues and neutralize the negative charge. This could explain a decrease in the amount of negative charge of the HL membrane used for the filtration of SA and SA + 2,3-DHBA. The membrane used for filtration of the SA + catechol mixture has very similar characteristics of the dependence of zeta potential on pH as for the pure membrane, only to a small extent are the values shifted towards positive values, suggesting that the presence of catechol neutralizes the effect of salicylic acid on the membrane surface. One of the probable reasons may be the formation of hydrogen bonds between the molecules of SA and catechol, resulting in better separation of SA during filtration. Further research is necessary in this respect because, at pH = 3.5, catechol interacts poorly with the membrane surface passing through it virtually unaffected. It can be assumed that at pH = 8, catechol may improve SA retention because its presence contributes to the increase in hydrophilicity of the membrane (
Table 5), thereby increasing the driving force of electrostatic repulsion.
3.5. Determination of Contact Angles and Free Surface Energy
The contact angle and free surface energy (FSE) were determined for the HL and TS80 membranes. For the HL membrane, extended tests, including measurement of the above-mentioned parameters for the pure membrane and membranes used, for the filtration of aqueous solutions with various chemical composition were performed (
Table 5). The tests also included the membrane used to filter the mixture obtained by the oxidation of salicylic acid with UV/H
2O
2. For the TS80 membrane, the tests included measurement of the contact angle and free surface energy for the pure membrane and the membrane used for the filtration of salicylic acid (
Table 6).
The pure HL membrane is a relatively hydrophilic membrane, and the determined contact angle for this membrane is 59.0°. Adsorption of salicylic acid molecules on the membrane surface resulted in an increase in the contact angle to 75.9°. The increase of the contact angle indicates a decrease in the hydrophilic properties of the membrane surface. A similar effect was obtained for the TS80 membrane, although the increase in the contact angle was not so significant. The contact angle increased from 53.4° (for pure membrane) to 59.7°. At the same time, a slight decrease in free surface energy was observed. In the case of the HL membrane, the free surface energy decreased from 43.3 mJ/m2 to 42.5 mJ/m2. In the case of the TS80 membrane, the value of FSE decreased from 48.3 mJ/m2 to 44.5 mJ/m2.
The polar, that is, acid-base component γ
SAB of FSE for the pure TS80 membrane was over six times higher than for the pure HL membrane. It should be remembered that the individual components of FSE depend on interactions at the liquid-solid interface. The polar component depends on the appearing interactions which have mainly chemical nature, that is, hydrogen, acceptor-donor, acid-base, and inductive bonds [
46]. The dispersion component, on the other hand, depends on the dispersion of a given liquid and is closely related to the adsorption of liquid on the solid surface [
47]. Considering the above, it can be assumed that the TS80 membrane was covered with a layer rich in polar groups. It was observed that after filtration of salicylic acid, the HL membrane showed a much higher value of the acid-base component than the value for the pure HL membrane, while in the case of the TS80 membrane, the effect was reversed. Further research is necessary in order to thoroughly explain this phenomenon. The use of the van Oss-Good model for HL membrane has shown that the values of the base component γ
S- varied from 4.32 to 6.43 [
48]. The highest value was obtained for HL membranes after filtration of oxidized salicylic acid. The lowest values were obtained for the membrane after SA (4.32) and the SA + 2,3-DHBA mixture (4.33) filtration. The value of the acid component γ
S+ ranged from −0.87 to 1.33. The lowest values were obtained for the membrane after SA (−0.87) and the SA + 2,3-DHBA mixture (−0.27) filtration, while the highest values were obtained for the clean membrane (1.25) and after filtration of the SA + Catechol mixture (1.33).
In the case of the HL membrane, which was used to filter the oxidized salicylic acid solution, a significant decrease in the contact angle was reported (to 48.6°). At the same time, a considerable decrease in the value of free surface energy to 37.0 mJ/m2 was observed. The acid-base component γSAB of FSE was zero mJ/m2. The dispersion (non-polar) component γLW has also been reduced. This may indicate damage to the top layer of the membrane due to too long exposure to hydrogen peroxide. This sample, before the measurements, was kept in the refrigerator at 4 °C for several days. Additional studies have shown that a relatively short exposure to hydrogen peroxide during nanofiltration (2–3 h) did not significantly affect the membrane properties and its surface morphology. This seems to be confirmed by the measurements of contact angle and FSE for the HL membrane used for filtration of water with the addition of hydrogen peroxide (0.324 mL/L), which were made immediately after the filtration process. All parameters remained without major changes, apart from the acid-base component γSAB of free surface energy, which increased to 4.1 mJ/m2.
After filtration of the mixture of salicylic acid and 2,3-DHBA, the HL membrane showed a markedly increased hydrophobicity (contact angle 66.4°) compared to the hydrophobicity of the pure membrane, but lower than the membrane used for the filtration of pure salicylic acid. The acid-base component γSAB of free surface energy also slightly increased. On the other hand, the addition of catechol to the salicylic acid solution caused a slight decrease in the contact angle (55.1°) of the HL membrane during filtration. A decrease in the polar component of FSE to 0.3 mJ/m2 was also observed. The determined values of contact angles correlated well with solubility (S) of the tested compounds. With increasing solubility of the filtered compound (SSA < S2,3-DHBA < SCAT), the contact angle of the membrane in use was decreasing.
4. Summary
Experiments with the removal of salicylic acid by means of UV/H2O2 and nanofiltration have been carried out. From the point of view of oxidation products, two basic conclusions were drawn: i) 2,3-dihydroxybenzoic acid, whose structure practically did not differ from the salicylic acid molecule except for the attached −OH group in the ortho position, showed similar retention coefficients as salicylic acid; ii) catechol, whose molecules were deprived of −COOH substituent but enriched with an additional −OH group, showed very little ability of being retained on the membrane. 2,3-dihydroxybenzoic acid has a negative impact on retention degree of SA during nanofiltration. Catechol, another oxidation product of SA, improves SA retention. Probably in an alkaline environment, catechol improves SA retention because its presence contributes to the increase in hydrophilicity of the membrane, thereby increasing the driving force of electrostatic repulsion.
The example of catechol showed that the use of integrated methods of removing hazardous compounds from wastewater, in which advanced oxidation and nanofiltration processes are applied, may contribute to the emission of secondary pollutants in the form of oxidation products, whose chemical structure and low molecular weight contribute to low efficiency of the separation on a membrane. Although catechol belongs to biodegradable products, it has been shown, in this example, that there is a potential risk of emission of toxic oxidation products which constitute a serious environmental hazard. Therefore, it is necessary to carry out research in this area and analyze threats resulting from the integration of advanced oxidation methods and membrane filtration for wastewater purification and water treatment.