The Application of Sediment Microbial Fuel Cells in Aquacultural Sediment Remediation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sediment and Water Sources
2.2. Construction and Operation of the SMFC
2.3. Analysis and Calculation
3. Analysis of Electricity Generation Performance
3.1. Voltage Analysis
3.2. Cell Internal Resistance and Power Density
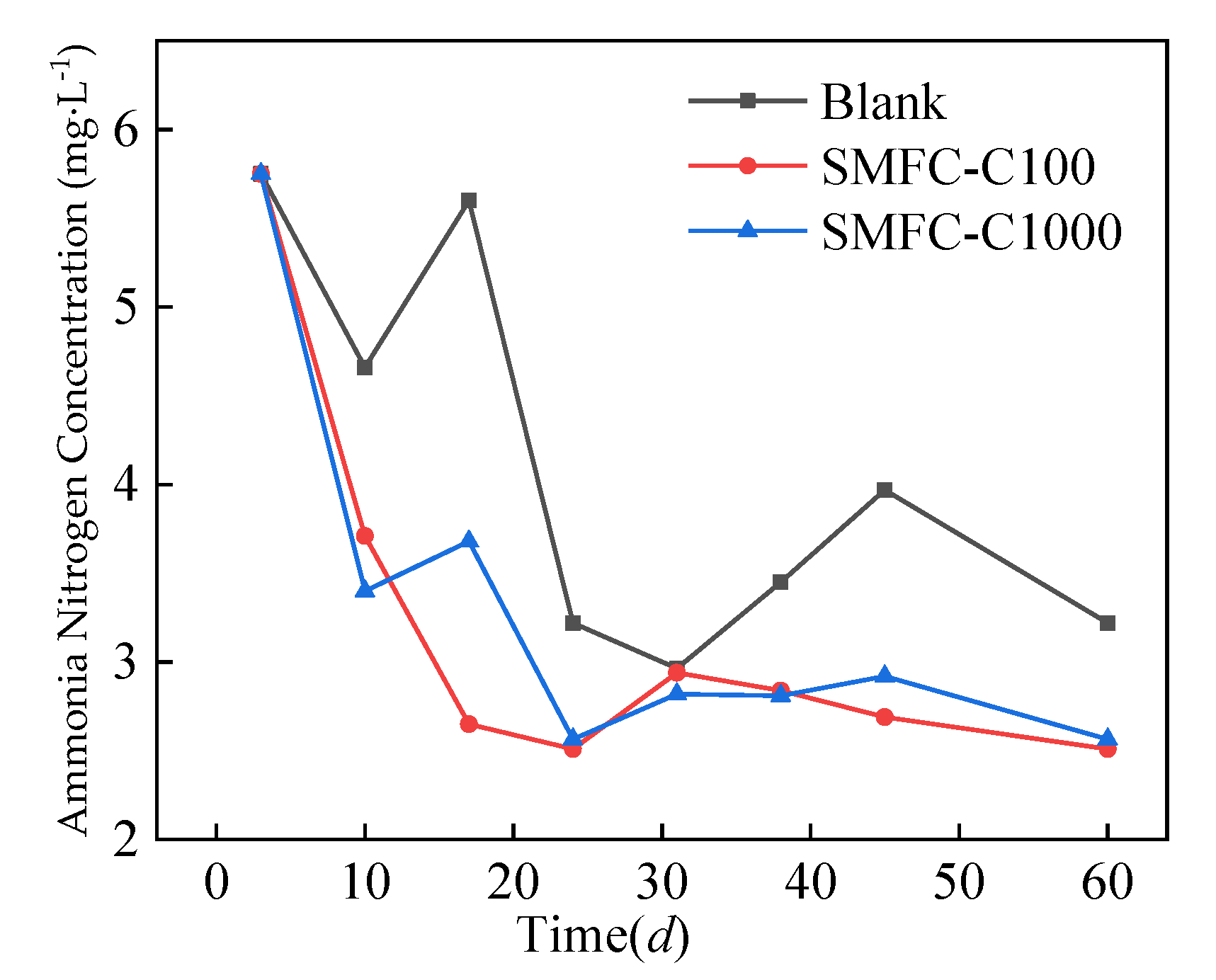
4. Analysis of Overlying Water Remediation
5. Analysis of Sediment Pollutant Removal
6. Conclusions
- (1)
- The average voltage of SMFC-A, which had a carbon fiber brush electrode, was the highest, and the average voltages of SMFCs with the other three electrode structures were relatively close. The power density was slightly higher for the SMFC using carbon fiber brushes vs. a graphite felt electrode, but the graphite felt material has a higher application value because of its lower cost. The SMFC using a carbon fiber cloth electrode had a relatively low power density, probably because of the smooth surface, which is unfavorable for microbial attachment.
- (2)
- The voltage and maximum output power of the SMFCs were significantly increased when the external resistance was 100 vs. 1000 Ω, and the electricity generation performance of the SMFC increased with increasing external resistance. However, an external resistance of 100 Ω led to a better effect on sediment remediation and water purification. At low resistance, more electrons are transferred to the cathode, forming a higher current, which produces a faster cathodic reaction and higher external electrical activity and thus accelerates the pollutant removal rate.
- (3)
- Compared with the open-circuit condition, introducing SMFCs accelerated the removal of pollutants from the overlying water. After 60 days of operation, the concentrations of COD, ammonia-N, TN, and TP in the overlying water of SMFC-C100 and SMFC-C1000 were lower than those of the open-circuit control group, and the removal effect on TP was the highest.
- (4)
- Compared with the open-circuit condition, introducing the SMFC promoted the degradation of organic matter and the fixation of P in sediments. After 2 months of operation, the TOC content in the sediments of SMFC-C100 and SMFC-C1000 decreased by 19.73% and 15.17%, respectively, which were significantly higher rates than that for the open-circuit control group. Compared with the initial state, the TP content in the sediments of SMFC-C100 and SMFC-C1000 increased by 9.49% and 2.83%, respectively, while that in the open-circuit control group decreased slightly. SMFCs increased the redox potential and thus the P adsorption capacity of sediments.
- (5)
- The SMFCs significantly inhibited water eutrophication and rapid algal blooms. After two months, green algal and brown algal blooms appeared in the two open-circuit control groups but not in the experimental groups with SMFCs. The average chlorophyll a content in the open-circuit control group was found to be significantly higher than that of the experimental groups with SMFCs, demonstrating that the SMFCs had a significant effect on the water quality of the overlying water.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, Y.; Lu, Z.; Lin, X.; Yang, Y.G.; Lu, Z.J.; Lin, X.K.; Xia, C.Y.; Sun, G.P.; Lian, Y.L.; Xu, M.Y. Enhancing the bioremediation by harvesting electricity from the heavily contaminated sediments. Bioresour. Technol. 2015, 79, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Li, H.N.; He, W.H.; Qu, Y.P.; Li, C.; Tian, Y.; Feng, Y.J. Pilot-scale benthic microbial electrochemical system (BMES) for the bioremediation of polluted river sediment. J. Power Sources 2017, 356, 430–437. [Google Scholar] [CrossRef]
- Hong, S.W.; Kim, H.S.; Chung, T.H. Alteration of sediment organic matter in sediment microbial fuel cells. Environ. Pollution 2010, 158, 185–191. [Google Scholar] [CrossRef]
- Hong, S.W.; Kim, H.J.; Choi, Y.S.; Chung, T.H. Field experiments on bioelectricity production from lake sediment using microbial fuel cell technology. Bull. Korean Chem. Soc. 2008, 29, 2189–2194. [Google Scholar]
- Yan, Z.S.; Song, N.; Cai, H.Y.; Tay, J.H.; Jiang, H.L. Enhanced degradation of phenanthrene and pyrene in freshwater sediments by combined employment of sediment microbial fuel cell and amorphous ferric hydroxide. J. Hazard. Mater. 2012, 199, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Li, X.J.; Wang, X.; Wan, L.L.; Zhang, Y.Y.; Li, N.; Li, D.S.; Zhou, Q.X. Enhanced biodegradation of aged petroleum hydrocarbons in soils by glucose addition in microbial fuel cells. J. Chem. Technol. Biotechnol. 2016, 91, 267–275. [Google Scholar] [CrossRef]
- Sherafatmand, M.; Ng, H.Y. Using sediment microbial fuel cells (SMFCs) for bioremediation of polycyclic aromatic hydrocarbons (PAHs). Bioresour. Technol. 2015, 195, 122–130. [Google Scholar] [CrossRef]
- Xu, X.; Zhao, Q.L.; Wu, M.S. Improved biodegradation of total organic carbon and polychlorinated biphenyls for electricity generation by sediment microbial fuel cell and surfactant addition. Rsc Adv. 2015, 5, 62534–62538. [Google Scholar] [CrossRef]
- Cao, X.; Song, H.L.; Yu, C.Y.; Li, X.N. Simultaneous degradation of toxic refractory organic pesticide and bioelectricity generation using a soil microbial fuel cell. Bioresour. Technol. 2015, 189, 87–93. [Google Scholar] [CrossRef]
- Ding, S.; Wang, Y.; Wang, D.; Li, Y.Y.; Gong, M.; Zhang, C. In situ, high-resolution evidence for iron-coupled mobilization of phosphorus in sediments. Sci. Rep. 2016, 6, 24341. [Google Scholar]
- Chen, M.S.; Ding, S.M.; Chen, X.; Sun, Q.; Fan, X.F.; Lin, J.; Ren, M.Y.; Yang, L.Y.; Zhang, C.S. Mechanisms driving phosphorus release during algal blooms based on hourly changes in iron and phosphorus concentrations in sediments. Water Res. 2018, 133, 153–164. [Google Scholar] [CrossRef]
- Li, R.H.; Cui, J.L.; Hu, J.H.; Wang, W.J.; Li, B.; Li, X.D.; Li, X.Y. Transformation of Fe-P complexes in bioreactors and P recovery from sludge: Investigation by XANES spectroscopy. Environ. Sci. Technol 2020, 54, 4641–4650. [Google Scholar] [CrossRef]
- Haxthausen, K.V.; Lu, X.Y.; Zhang, Y.F.; Gosewinkel, U.; Petersen, D.G.; Marzocchi, U.; Brock, A.L.; Trapp, S. Novel method to immobilize phosphate in lakes using sediment microbial fuel cells. Water Res. 2021, 198, 117108. [Google Scholar] [CrossRef]
- Feng, C.H.; Huang, L.Q.; Yu, H.; Yi, X.Y.; Wei, C.H. Simultaneous phenol removal, nitrification and denitrification using microbial fuel cell technology. Water Res. 2015, 76, 160–170. [Google Scholar] [CrossRef]
- Haochi, Z.; Dengfeng, H.; Shuai, Z.; Xian, C.; Hui, W.; Xianning, L. Aerobic Denitrification Is Enhanced Using Biocathode of SMFC in Low-Organic Matter Wastewater. Water 2021, 13, 3512. [Google Scholar]
- Sajana, T.K.; Ghangrekar, M.M.; Mitra, A. Application of sediment microbial fuel cell for in situ reclamation of aquaculture pond water quality. Aquac. Eng. 2013, 57, 101–107. [Google Scholar] [CrossRef]
- Sajana, T.K.; Ghangrekar, M.M.; Mitra, A. Effect of operating parameters on the performance of sediment microbial fuel cell treating aquaculture water. Aquac. Eng. 2014, 61, 17–26. [Google Scholar] [CrossRef]
- Sajana, T.K.; Ghangrekar, M.M.; Mitra, A. In Situ Bioremediation Using Sediment Microbial Fuel Cell. J. Hazard. Toxic Radioact. Waste 2017, 21, 04016022. [Google Scholar]
- Yu, B.; Feng, L.; He, Y.; Yang, L.; Xun, Y. Effects of anode materials on the performance and anode microbial community of soil microbial fuel cell. J. Hazard. Mater. 2020, 401, 123394. [Google Scholar] [CrossRef]
- Zhu, D.W.; Wang, D.B.; Song, T.S.; Guo, T.; Ouyang, P.K.; Wei, P.; Xie, J.J. Effect of carbon nanotube modified cathode by electrophoretic deposition method on the performance of sediment microbial fuel cells. Biotechnol. Lett. 2015, 37, 101–107. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhou, S.; Zhuang, L. A new approach to in situ sediment remediation based on air-cathode microbial fuel cells. J. Soils Sediments 2010, 10, 1427–1433. [Google Scholar] [CrossRef]
- Song, T.S.; Yan, Z.S.; Zhao, Z.W.; Jiang, H.L. Removal of organic matter in freshwater sediment by microbial fuel cells at various external resistances. J. Chem. Technol. Biotechnol. 2010, 11, 1489–1493. [Google Scholar] [CrossRef]
- Hong, S.W.; Chang, I.S.; Choi, Y.S.; Chung, T.H. Experimental evaluation of influential factors for electricity harvesting from sediment using microbial fuel cell. Bioresour. Technol. 2009, 100, 3029–3035. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Tian, J.; Feng, L. Remediation of PAH Polluted Soils Using a Soil Microbial Fuel Cell: Influence of Electrode Interval and Role of Microbial Community. J. Hazard. Mater. 2017, 336, 110–118. [Google Scholar] [CrossRef]
- Wu, Y.C.; Wu, H.J.; Fu, H.Y.; Dai, Z.N.; Wang, Z.J. Burial depth of anode affected the bacterial community structure of sediment microbial fuel cells. Environ. Eng. Res. 2019, 25, 871–877. [Google Scholar] [CrossRef]
- An, J.; Kim, B.; Nam, J.; Nam, J.; Ng, H.Y.; Chang, I.S. Comparison in performance of sediment microbial fuel cells according to depth of embedded anode. Bioresour. Technol. 2013, 127, 138–142. [Google Scholar] [CrossRef]
- Zhang, F.; Yu, S.S.; Li, J.; Li, W.W.; Yu, H.Q. Mechanisms behind the accelerated extracellular electron transfer in Geobacter sulfurreducens DL-1 by modifying gold electrode with self-assembled monolayers. Front. Environ. Sci. Eng. 2016, 10, 531–538. [Google Scholar] [CrossRef]
- Zhang, Y.; Angelidaki, I. Bioelectrode-based approach for enhancing nitrate and nitrite removal and electricity generation from eutrophic lakes. Water Res. 2012, 46, 6445–6453. [Google Scholar] [CrossRef]
- Saravanan, R.; Arun, A.; Venkatamohan, S.; Kandavelu, T. Membraneless dairy wastewatersediment interface for bioelectricity generation employing sediment microbial fuel cell (SMFC). Afr. J. Microbiol. Res. 2010, 4, 2640–2646. [Google Scholar]
- Zhao, Q.; Li, R.Y.; Ji, M.; Ren, Z.J. Organic content influences sediment microbial fuel cell performance and community structure. Bioresour. Technol. 2016, 220, 549–556. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, H.; Yang, Y.; Xu, M. Diffusion and filamentous bacteria jointly govern the spatiotemporal process of sulfide removal in sediment microbial fuel cells. Chem. Eng. J. 2021, 405, 126680. [Google Scholar] [CrossRef]
- Van Dael, T.; De Cooman, T.; Verbeeck, M.; Smolders, E. Sediment respiration contributes to phosphate release in lowland surface waters. Water Res. 2020, 168, 115168. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.C.; Liang, P.; Huang, X. Recent progress in electrodes for microbial fuel cells. Bioresour. Technol. 2011, 102, 9335–9344. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Meng, L.J.; Chen, G.; Xi, Y.; Jiang, N.; Song, J.L.; Zheng, S.Y.; Liu, Y.B.; Zhen, G.Y.; Huang, M.H. Application of advanced anodes in microbial fuel cells for power generation: A review. Chemosphere 2020, 248, 125985. [Google Scholar] [CrossRef]
- Zhang, J.F.; Shi, G.C.; Shi, Z.T.; Hu, J.l. Application of SMFC in Power Generation and Organic Matter Removal in Estuaries. J. Tianjin Univ. (Sci. Technol.) 2022, 55, 85–89. [Google Scholar]
- Chen, Y.; Chen, M.; Shen, N.; Zeng, R.J. H2 production by the thermoelectric microconverter coupled with microbial electrolysis cell. Int. J. Hydrogen Energy 2016, 41, 22760–22768. [Google Scholar] [CrossRef]
- Qi, C.; Zhang, L.M.; Fang, J.Q.; Lei, B.; Tang, X.C.; Huang, H.X.; Wang, Z.S.; Si, Z.J.; Wang, G.X. Benthic cyanobacterial detritus mats in lacustrine sediment: Characterization and odorant producing potential. Environ. Pollut. 2020, 256, 113453. [Google Scholar] [CrossRef]
- Song, N.; Jiang, H.; Yan, Z. Contrasting Effects of Sediment Microbial Fuel Cells (SMFCs) on the Degradation of Macrophyte Litter in Sediments from Different Areas of a Shallow Eutrophic Lake. Appl. Sci.-Basel 2019, 9, 9183703. [Google Scholar] [CrossRef]
- Pettersson, K.; Bostrom, B.; Jacobsen, O.-S. Phosphorus in Sediments-Speciation and Analysis. In Phosphorus in Freshwater Ecosystems; Springer: Berlin/Heidelberg, Germany, 1988; pp. 91–101. [Google Scholar]
- Xuan, W.; Yingying, Z.; Yun, C.; Nan, S.; Guoxiang, W.; Yan, Y. Realignment of phosphorus in lake sediment induced by sediment microbial fuel cells (SMFC). Chemosphere 2022, 291, 132927. [Google Scholar]
- Pasupuleti, S.B.; Srikanth, S.; Mohan, S.V.; Pant, D. Continuous mode operation of microbial fuel cell (MFC) stack with dual gas diffusion cathode design for the treatment of dark fermentation effluent. Int. J. Hydrog. Energy 2015, 40, 12424–12435. [Google Scholar] [CrossRef]
- Bruce, L.; Shaoan, C.; Valerie, W.; Garett, E. Graphite Fiber Brush Anodes for Increased Power Production in Air-Cathode Microbial Fuel Cells. Environ. Sci. Technol. 2007, 41, 3341–3346. [Google Scholar]
- Mccarthy, M.J.; James, R.T.; Chen, Y.W.; East, T.L.; Gardner, W.S. Nutrient ratios and phytoplankton community structure in the large, shallow, eutrophic, subtropical Lakes Okeechobee (Florida, USA) and Taihu (China). Limnology 2009, 10, 215–227. [Google Scholar] [CrossRef]
- Rismani-Yazdi, H.; Christy, A.D.; Carver, S.M.; Yu, Z.T.; Dehority, B.A.; Tuovinen, O.H. Effect of external resistance on bacterial diversity and metabolism in cellulose-fed microbial fuel cells. Bioresour. Technol. 2011, 102, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.M.; Zhang, W.; Wang, X.X.; Zhou, Y.Y.; Yang, H.; Ji, G.L. Phosphorus in Interstitial Water Induced by Redox Potential in Sediment of Dianchi Lake, China. Pedosphere 2007, 17, 739–746. [Google Scholar] [CrossRef]









| Reactor Type | Anode Material | Cathode Material | Anode Specification | Cathode Specification | External Resistance |
|---|---|---|---|---|---|
| SMFC-A | Carbon fiber brush | Carbon fiber brush | 3 × 10 × 15 cm (5) | 3 × 10 × 15 cm (5) | 100 Ω |
| SMFC-B | Carbon fiber brush | Graphite felt | 3 × 10 × 15 cm (5) | 10 × 10 × 0.2 cm (3) | 100 Ω |
| SMFC-C100 | Graphite felt | Graphite felt | 10 × 10 × 0.5 cm (3) | 10 × 10 × 0.2 cm (3) | 100 Ω |
| SMFC-C1000 | Graphite felt | Graphite felt | 10 × 10 × 0.5 mm (3) | 10 × 10 × 0.2 cm (3) | 1000 Ω |
| SMFC-D | Carbon fiber cloth | Graphite felt | 10 × 10 mm (6) | 10 × 10 × 0.2 cm (3) | 100 Ω |
| Blank | Control group with an open circuit | ||||
| Reactor Type Voltage (V) | Startup | 0–15 Day | 16–30 Day | 31–45 Day | 46–60 Day | Average |
|---|---|---|---|---|---|---|
| SMFC-A | 0.078 | 0.195 | 0.226 | 0.275 | 0.203 | 0.212 |
| SMFC-B | 0.020 | 0.208 | 0.206 | 0.265 | 0.172 | 0.194 |
| SMFC-C100 | 0.065 | 0.200 | 0.204 | 0.266 | 0.150 | 0.192 |
| SMFC-C1000 | 0.300 | 0.580 | 0.618 | 0.665 | 0.675 | 0.604 |
| SMFC-D | 0.020 | 0.145 | 0.236 | 0.250 | 0.193 | 0.199 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, J.; Sun, Z.; Zhang, J.; Ye, C. The Application of Sediment Microbial Fuel Cells in Aquacultural Sediment Remediation. Water 2022, 14, 2668. https://doi.org/10.3390/w14172668
Qi J, Sun Z, Zhang J, Ye C. The Application of Sediment Microbial Fuel Cells in Aquacultural Sediment Remediation. Water. 2022; 14(17):2668. https://doi.org/10.3390/w14172668
Chicago/Turabian StyleQi, Jiarui, Zhuteng Sun, Jinfeng Zhang, and Chen Ye. 2022. "The Application of Sediment Microbial Fuel Cells in Aquacultural Sediment Remediation" Water 14, no. 17: 2668. https://doi.org/10.3390/w14172668
APA StyleQi, J., Sun, Z., Zhang, J., & Ye, C. (2022). The Application of Sediment Microbial Fuel Cells in Aquacultural Sediment Remediation. Water, 14(17), 2668. https://doi.org/10.3390/w14172668





