Human-Induced Sharp Salinity Changes in the World’s Largest Hypersaline Lagoon Bay Sivash (Crimea) and Their Effects on the Ecosystem
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Abiotic Parameters
3.2. Zooplankton
3.3. Zoobenthos
3.4. Zooplankton vs. Zoobenthos
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hart, B.T.; Lake, P.S.; Webb, J.A.; Grace, M.R. Ecological risk to aquatic systems from salinity increases. Aust. J. Bot. 2003, 51, 689–702. [Google Scholar] [CrossRef]
- Lin, Q.; Xu, L.; Hou, J.; Liu, Z.; Jeppesen, E.; Han, B.P. Responses of trophic structure and zooplankton community to salinity and temperature in Tibetan lakes: Implication for the effect of climate warming. Water Res. 2017, 124, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Golubkov, S.M.; Shadrin, N.V.; Golubkov, M.S.; Balushkina, E.V.; Litvinchuk, L.F. Food chains and their dynamics in ecosystems of shallow lakes with different water salinities. Russ. J. Ecol. 2018, 49, 442–448. [Google Scholar] [CrossRef]
- Shadrin, N.; Kolesnikova, E.; Revkova, T.; Latushkin, A.; Dyakov, C.; Anufriieva, E. Macrostructure of benthos along a salinity gradient: The case of Sivash Bay (the Sea of Azov), the largest hypersaline lagoon worldwide. J. Sea Res. 2019, 154, 101811. [Google Scholar] [CrossRef]
- Royer, T.C.; Grosch, C.E. Ocean warming and freshening in the northern Gulf of Alaska. Geophys. Res. Lett. 2006, 33, L16605. [Google Scholar] [CrossRef] [Green Version]
- Mao, D.; Wang, Z.; Yang, H.; Li, H.; Thompson, J.R.; Li, L.; Song, K.; Chen, B.; Gao, H.; Wu, J. Impacts of climate change on Tibetan lakes: Patterns and processes. Remote Sens. 2018, 10, 358. [Google Scholar] [CrossRef] [Green Version]
- Regan, H.C.; Holland, P.R.; Meredith, M.P.; Pike, J. Sources, variability and fate of freshwater in the Bellingshausen Sea, Antarctica. Deep Sea Res. Part I Oceanogr. Res. Pap. 2018, 133, 59–71. [Google Scholar] [CrossRef]
- Shadrin, N.V.; Simonov, V.G.; Anufriieva, E.V.; Popovichev, V.N.; Sirotina, N.O. Anthropogenic transformation of Kyzyl-Yar Lake in Crimea: Multiyear research findings. Arid Ecosyst. 2018, 8, 299–306. [Google Scholar] [CrossRef]
- Al-Dousari, A.E. Desalination leading to salinity variations in Kuwait marine waters. Am. J. Environ. Sci. 2009, 5, 451. [Google Scholar] [CrossRef] [Green Version]
- Dugan, H.A.; Bartlett, S.L.; Burke, S.M.; Doubek, J.P.; Krivak-Tetley, F.E.; Skaff, N.K.; Summers, J.C.; Farrell, K.J.; McCullough, I.M.; Morales-Williams, A.M.; et al. Salting our freshwater lakes. Proc. Natl Acad. Sci. USA 2017, 114, 4453–4458. [Google Scholar] [CrossRef] [Green Version]
- Kaushal, S.S.; Likens, G.E.; Pace, M.L.; Utz, R.M.; Haq, S.; Gorman, J.; Grese, M. Freshwater salinization syndrome on a continental scale. Proc. Natl. Acad. Sci. USA 2018, 115, E574–E583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shadrin, N.V.; Anufriieva, E.V.; Kipriyanova, L.M.; Kolesnikova, E.A.; Latushkin, A.A.; Romanov, R.E.; Sergeeva, N.G. The political decision caused the drastic ecosystem shift of the Sivash Bay (the Sea of Azov). Quat. Int. 2018, 475, 4–10. [Google Scholar] [CrossRef]
- Sharifinia, M.; Afshari Bahmanbeigloo, Z.; Smith, W.O., Jr.; Yap, C.K.; Keshavarzifard, M. Prevention is better than cure: Persian Gulf biodiversity vulnerability to the impacts of desalination plants. Glob. Chang. Biol. 2019, 25, 4022–4033. [Google Scholar] [CrossRef] [PubMed]
- Kjerfve, B. Coastal Lagoon Processes; Elsevier Science Publisher: Amsterdam, The Netherlands, 1994; 577p. [Google Scholar]
- Dyakov, N.N.; Belogudov, A.A.; Timoshenko, T.Y. Assessment of the components in the water balance of Bay Sivash Bay. Ecol. Saf. Coast. Shelf Zones Complex Manag. Shelf Res. 2013, 27, 439–445. (In Russian). [Google Scholar]
- Getmanenko, V.A.; Yanovsky, Y.G.; Grout, G.G. Impact of semi-mechanical trawling on the zoobenthos of the eastern Sivash (Azov Sea). Hydrobiol. J. 1996, 32, 54–60. (In Russian). [Google Scholar]
- Zagorodnyaya, Y.A. The taxonomic composition and quantitative characteristics of zooplankton in the eastern part of Gulf Sivash during summer 2004. Ecosyst. Res. Azov Sea Black Sea Casp. Sea 2006, 8, 103–114. (In Russian). [Google Scholar]
- Anufriieva, E.; Shadrin, N. The long-term changes in plankton composition: Is Bay Sivash transforming back into one of the world’s largest habitats of Artemia sp. (Crustacea, Anostraca)? Aquac. Res. 2020, 51, 341–350. [Google Scholar] [CrossRef]
- Shadrin, N.; Balycheva, D.; Anufriieva, E. Microphytobenthos in the hypersaline water bodies, the case of Bay Sivash (Crimea): Is salinity the main determinant of species composition? Water 2021, 13, 1542. [Google Scholar] [CrossRef]
- Sergeeva, N.G.; Shadrin, N.V.; Anufriieva, E.V. Long-term changes (1979–2015) in the nematode fauna in Sivash Bay (Sea of Azov), Russia, worldwide the largest hypersaline lagoon, during salinity transformations. Nematology 2019, 21, 337–347. [Google Scholar] [CrossRef]
- Müller, P.H.; Neuman, P.; Storm, R. Tafeln der Mathematischen Statistic; VEB Fachbuchverlag: Leipzig, Germany, 1979; 272p. [Google Scholar]
- Boulion, V.V.; Anokhin, L.E.; Arakelova, E.U. Primary production of the hypehaline lakes in Crimea. Trudy Zool. Inst. 1989, 205, 14–25. (In Russian). [Google Scholar]
- López-González, P.J.; Guerrero, F.; Castro, M.C. Seasonal fluctuations in the plankton community in a hypersaline temporary lake (Honda, southern Spain). Int. J. Salt Lake Res. 1997, 6, 353–371. [Google Scholar] [CrossRef]
- Schallenberg, M.; Burns, C.W. A temperate, tidal lake-wetland complex 2. Water quality and implications for zooplankton community structure. N. Z. J. Mar. Freshw. Res. 2003, 37, 429–447. [Google Scholar] [CrossRef]
- Abdo, M.H. Physico-chemical characteristics of Abu Za’baal ponds, Egypt. Egypt. J. Aquat. Res. 2005, 31, 1–15. [Google Scholar]
- Elloumi, J.; Carrias, J.F.; Ayadi, H.; Sime-Ngando, T.; Boukhris, M.; Bouaïn, A. Composition and distribution of planktonic ciliates from ponds of different salinity in the solar saltwork of Sfax, Tunisia. Estuar. Coast. Shelf Sci. 2006, 67, 21–29. [Google Scholar] [CrossRef]
- Telesh, I.V.; Khlebovich, V.V. Principal processes within the estuarine salinity gradient: A review. Mar. Pollut. Bull. 2010, 61, 149–155. [Google Scholar] [CrossRef]
- Ivanova, M.; Balushkina, E.; Basova, S. Structural functional reorganization of ecosystem of hyperhaline lake Saki (Crimea) at increased salinity. Russ. J. Aquat. Ecol. 1994, 3, 111–126. [Google Scholar]
- Abdullahi, A.S.; Underwood, G.J.; Gretz, M.R. Extracellular matrix assembly in diatoms (Bacillariophyceae). V. Environmental effects on polysaccharide synthesis in the model diatom, Phaeodactylum Tricornutum. J. Phycol. 2006, 42, 363–378. [Google Scholar] [CrossRef]
- Steele, D.J.; Franklin, D.J.; Underwood, G.J. Protection of cells from salinity stress by extracellular polymeric substances in diatom biofilms. Biofouling 2014, 30, 987–998. [Google Scholar] [CrossRef] [Green Version]
- Prazukin, A.V.; Anufriieva, E.V.; Shadrin, N.V. Cladophora mats in a Crimean hypersaline lake: Structure, dynamics, and inhabiting animals. J. Oceanol. Limnol. 2018, 36, 1930–1940. [Google Scholar] [CrossRef]
- Prazukin, A.; Shadrin, N.; Balycheva, D.; Firsov, Y.; Lee, R.; Anufriieva, E. Cladophora spp. (Chlorophyta) modulate environment and create a habitat for microalgae in hypersaline waters. Eur. J. Phycol. 2021, 56, 231–243. [Google Scholar] [CrossRef]
- Steinhorn, I. Evaporation estimate for the Dead Sea: Essential considerations for saline lakes. In The Dead Sea: The Lake and Its Setting; Niemi, T.M., Ben-Avraham, Z., Gat, J.R., Eds.; Oxford University Press: Oxford, UK, 1997; pp. 122–132. [Google Scholar]
- Sherwood, J.E.; Stagnitti, F.; Kokkinn, M.J.; Williams, W.D. A standard table for predicting equilibrium dissolved oxygen concentrations in salt lakes dominated by sodium chloride. Int. J. Salt Lake Res. 1992, 1, 1–6. [Google Scholar] [CrossRef]
- Naftz, D.; Angeroth, C.; Kenney, T.; Waddell, B.; Darnall, N.; Silva, S.; Perschon, C.; Whitehead, J. Anthropogenic influences on the input and biogeochemical cycling of nutrients and mercury in Great Salt Lake, Utah, USA. Appl. Geochem. 2008, 23, 1731–1744. [Google Scholar] [CrossRef]
- Zhang, D.; Lin, Q.; Xue, N.; Zhu, P.; Zhang, F.; Ge, C.; Ma, C.; Pan, X. Ca2+ complexation of dissolved organic matter in arid inland lakes is significantly affected by drastic seasonal change of salinity. Sci. Total Environ. 2019, 663, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Mirzoeva, N.; Shadrin, N.; Arkhipova, S.; Miroshnichenko, O.; Kravchenko, N.; Anufriieva, E. Does salinity affect the distribution of the artificial radionuclides 90Sr and 137Cs in water of the saline lakes? A case of the Crimean Peninsula. Water 2020, 12, 349. [Google Scholar] [CrossRef] [Green Version]
- Shadrin, N.; Stetsiuk, A.; Latushkin, A.; Anufriieva, E. Mercury in the world’s largest hypersaline lagoon Bay Sivash, the Sea of Azov. Environ. Sci. Pollut. Res. 2021, 28, 28704–28712. [Google Scholar] [CrossRef]
- Saccò, M.; White, N.E.; Harrod, C.; Salazar, G.; Aguilar, P.; Cubillos, C.F.; Meredith, K.; Baxter, B.K.; Oren, A.; Anufriieva, E.; et al. Allentoft, M.E. Salt to conserve: A review on the ecology and preservation of hypersaline ecosystems. Biol. Rev. 2021, 96, 2828–2850. [Google Scholar] [CrossRef]
- Wurtsbaugh, W.A.; Berry, T.S. Cascading effects of decreased salinity on the plankton chemistry, and physics of the Great Salt Lake (Utah). Can. J. Fish. Aquat. Sci. 1990, 47, 100–109. [Google Scholar] [CrossRef]
- Davis, J.A.; McGuire, M.; Halse, S.A.; Hamilton, D.; Horwitz, P.; McComb, A.J.; Froend, R.H.; Lyons, M.; Sim, L. What happens when you add salt: Predicting impacts of secondary salinisation on shallow aquatic ecosystems by using an alternative-states model. Aust. J. Bot. 2003, 51, 715–724. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Can Gammarus aequicauda (Amphipoda) suppress a population of Baeotendipes noctivagus (Chironomidae) in a hypersaline lake? A case of Lake Moynaki (Crimea). Aquac. Res. 2021, 52, 1705–1714. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Suppression of Artemia spp. (Crustacea, Anostraca) populations by predators in the Crimean hypersaline lakes: A review of the evidence. Int. Rev. Hydrobiol. 2019, 104, 5–13. [Google Scholar] [CrossRef] [Green Version]
- Jia, Q.; Anufriieva, E.; Liu, X.; Kong, F.; Shadrin, N. Intentional introduction of Artemia sinica (Anostraca) in the high-altitude Tibetan Lake Dangxiong Co: The new population and consequences for the environment and for humans. Chin. J. Oceanol. Limnol. 2015, 33, 1451–1460. [Google Scholar] [CrossRef]
- Shadrin, N.V. The alternative saline lake ecosystem states and adaptive environmental management. J. Oceanol. Limnol. 2018, 36, 2010–2017. [Google Scholar] [CrossRef]
- Scheffer, M. Alternative attractors of shallow lakes. Sci. World J. 2001, 1, 254–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Shabrawy, G.M.; Anufrieva, E.V.; Shadrin, N.V. Tintinnina (Ciliophora) and Foraminifera in plankton of hypersaline Lagoon Bardawil (Egypt): Spatial and temporal variability. Turk. J. Zool. 2018, 42, 218–229. [Google Scholar] [CrossRef] [Green Version]
- Anufriieva, E.V.; El-Shabrawy, G.M.; Shadrin, N.V. Copepoda in the shallow hypersaline Bardawil coastal lake (Egypt): Are there long-term changes in composition and abundance? Oceanol. Hydrobiol. Stud. 2018, 47, 219–229. [Google Scholar] [CrossRef]
- Pérez-Ruzafa, A.; De Pascalis, F.; Ghezzo, M.; Quispe-Becerra, J.I.; Hernández-García, R.; Munoz, I.; Vergara, C.; Pérez-Ruzafa, I.M.; Umgiesser, G.; Marcos, C. Connectivity between coastal lagoons and sea: Asymmetrical effects on assemblages’ and populations’ structure. Estuar. Coast. Shelf Sci. 2019, 216, 171–186. [Google Scholar] [CrossRef]
- Gera, A.; Kumaraswami, M.; Rao, V.R.; Vijay, A.; Pandiyarajan, R.S.; Ezhilarasan, P.; Murthy, M.R. The Pulicat, a distinctive shallow lagoon: Hypersalinity, thermodynamics and meromixis. Estuar. Coast. Shelf Sci. 2021, 252, 107292. [Google Scholar] [CrossRef]
- Case, T.J.; Sidell, R. Pattern and chance in the structure of model and natural communities. Evolution 1983, 1, 832–849. [Google Scholar] [CrossRef]
- Carranza, A.; Defeo, O.; Arim, M. Taxonomic relatedness and spatial structure of a shelf benthic gastropod assemblage. Divers. Distrib. 2011, 17, 25–34. [Google Scholar] [CrossRef]
- Boero, F.; Kraberg, A.C.; Krause, G.; Wiltshire, K.H. Time is an affliction: Why ecology cannot be as predictive as physics and why it needs time series. J. Sea Res. 2015, 101, 12–18. [Google Scholar] [CrossRef]
- Shadrin, N.V.; Belyakov, V.P.; Bazhora, A.I.; Anufriieva, E.V. The role of salinity as an environmental filtering factor in the determination of the Diptera taxonomic composition in the Crimean waters. Knowl. Manag. Aquat. Ecosyst. 2019, 420, 3. [Google Scholar] [CrossRef]
- Holling, C.S. Understanding the complexity of economic, ecological, and social system. Ecosystems 2001, 4, 390–405. [Google Scholar] [CrossRef]
- Grinchenko, A.B. History and dynamics of colonial settlements of Ciconiidae in the Eastern Crimea under the influence of anthropogenic succession at the Eastern Sivash and near Sivash area. Branta Trans. Azov-Black Sea Ornithol. Stn. 2004, 7, 61–81. (In Russian). [Google Scholar]
- Popenko, V.M.; Andryushchenko, Y.A. Nesting of Greater Flamingo (Phoenicopterus roseus) in Ukraine. Branta Trans. Azov-Black Sea Ornithol. Stn. 2017, 20, 220–223. (In Russian). [Google Scholar] [CrossRef] [Green Version]
- Hurbbert, S.H. Limnological studies of flamingo diets and distributions. Nat. Geogr. Soc. Res. Rep. 1982, 14, 351–356. [Google Scholar]
- Dejoux, C. Benthic invertebrates of some saline lakes of the Sud Lipez region, Bolivia. Hydrobiologia 1993, 267, 257–267. [Google Scholar] [CrossRef]
- Williams, W.D.; Carrick, T.R.; Bayly, I.A.; Green, J.; Herbst, D.B. Invertebrates in salt lakes of the Bolivian Altiplano. Int. J. Salt Lake Res. 1995, 4, 65–77. [Google Scholar] [CrossRef]
- Hurlbert, S.H.; Chang, C.Y. Ornitholimnology: Effects of grazing by the Andean flamingo (Phoenicoparrus andinus). Proc. Natl. Acad. Sci. USA 1983, 80, 4766–4769. [Google Scholar] [CrossRef] [Green Version]
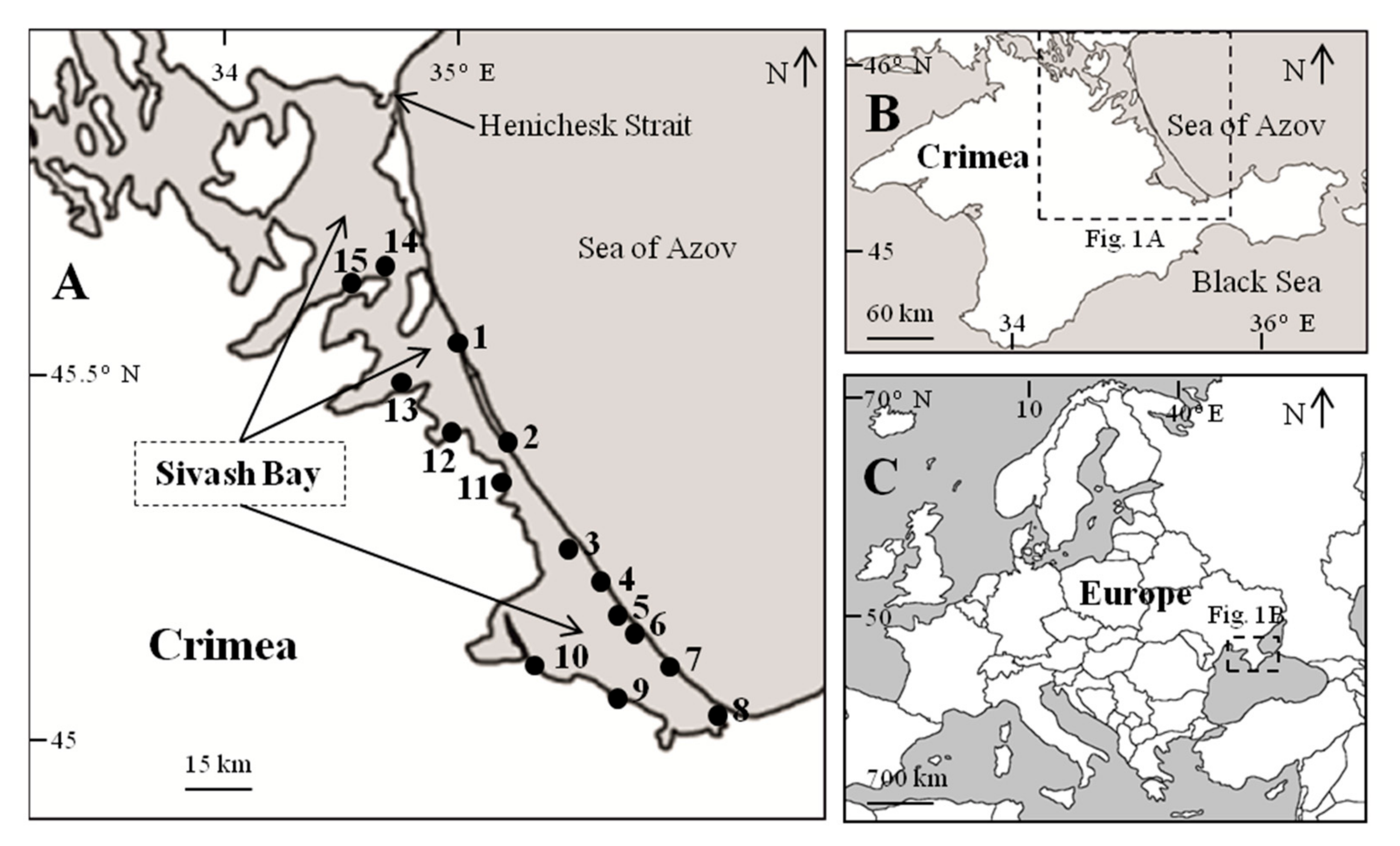







| The Station Number | Coordinates | The Station Number | Coordinates |
|---|---|---|---|
| 1 | 45°45′37.0″ N; 34°57′57.0″ E | 9 | 45°19′05.5″ N; 35°14′59.8″ E |
| 2 | 45°37′9.0″ N; 35°04′40.0″ E | 10 | 45°21′04.2″ N; 35°06′06.5″ E |
| 3 | 45°31′13.7″ N; 35°11′12.9″ E | 11 | 45°37′48.3″ N; 35°01′54.8″ E |
| 4 | 45°29′04.7″ N; 35°13′27.9″ E | 12 | 45°40′48.8″ N; 34°54′55.2″ E |
| 5 | 45°27′19.5″ N; 35°13′27.9″ E | 13 | 45°44′00.8″ N; 34°48′10.3″ E |
| 6 | 45°24′43.5″ N; 35°17′33.8″ E | 14 | 45°52′38.8″ N; 34°44′33.3″ E |
| 7 | 45°23′04.7″ N; 35°19′44.6″ E | 15 | 45°52′42.6″ N; 34°42′09.0″ E |
| 8 | 45°17′14.3″ N; 35°28′01.2″ E | – | – |
| Date | Temperature, °C | Salinity, g L−1 | TSS, mg L−1 | DOM, g L−1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Range | CV | Mean | Range | CV | Mean | Range | CV | Mean | Range | CV | |
| 2016, June | 32.50 | 31.00–34.00 | 0.090 | 51.00 | 24.00–70.00 | 0.270 | – | – | – | – | – | – |
| 2018, May | 21.80 | 19.50–26.00 | 0.103 | 64.00 | 30.00–82.00 | 0.292 | 13.64 | 2.37–24.76 | 0.632 | 3.57 | 2.66–4.16 | 0.154 |
| 2018, November | 11.90 | 10.00–14.00 | 0.116 | 74.00 | 30.00–100.00 | 0.315 | 13.98 | 1.31–70.16 | 1.335 | 9.31 | 2.97–42.14 | 1.158 |
| 2019, June | 28.00 | 26.00–32.00 | 0.056 | 73.00 | 35.00–100.00 | 0.241 | 13.79 | 2.45–26.66 | 0.623 | 7.95 | 2.98–15.35 | 0.617 |
| 2020, July | 26.70 | 21.00–34.00 | 0.124 | 87.00 | 41.00–128.00 | 0.245 | 18.00 * | 2.04–33.70 * | 0.603 * | – | – | – |
| 2020, September | 26.60 | 24.00–28.00 | 0.048 | 88.00 | 41.00–138.00 | 0.382 | – | – | – | – | – | – |
| 2020, December | 3.00 | 3.00–3.50 | 0.055 | 94.00 | 30.00–112.00 | 0.293 | – | – | – | – | – | – |
| Taxon | 2016 | 2018 | 2019 | 2020 | Max Salinity, g L−1 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| * June | June | November | June | July | September | December | ||||||||||||||
| Mean, Ind. m−2 | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | ||
| Artemia cysts | 260 | 0 | – | 0 | 120,060 | 1.948 | 100 | 17,846 | 1.158 | 100 | 11,763 | 2.146 | 100 | 43,211 | 1.252 | 100 | 276,039 | 1.327 | 100 | 138 |
| Artemia active stages | 0 | 0 | – | 0 | 4201 | 1.420 | 69 | 1058 | 2.821 | 22 | 51,323 | 1.527 | 86 | 4628 | 1.082 | 100 | 150 | 2.620 | 25 | 138 |
| Chironomidae larvae and pupae | 0 | 208 | 1.309 | 88 | 53 | 2.606 | 38 | 19 | 1.551 | 33 | 1441 | 1.830 | 100 | 114 | 0.972 | 100 | 430 | 1.388 | 75 | 138 |
| Calanoida | 1247 | 33 | 1.777 | 38 | 13 | 3.023 | 15 | 7 | 2.121 | 22 | 0 | – | 0 | 0 | – | 0 | 10 | 2.828 | 13 | 112 |
| Gastropoda | 0 | 37 | 1.732 | 13 | 3 | 2.663 | 15 | 261 | 1.469 | 67 | 711 | 3.557 | 43 | 38 | 0.992 | 80 | 25 | 2.828 | 25 | 112 |
| Bivalvia | 0 | 17 | 1.732 | 13 | 3 | 2.663 | 15 | 106 | 1.493 | 89 | 7 | 2.357 | 21 | 10 | 1.225 | 80 | 0 | – | 0 | 138 |
| Harpacticoida | 60 | 98 | 1.786 | 75 | 5 | 2.000 | 23 | 2870 | 2.468 | 100 | 2413 | 2.368 | 64 | 192 | 1.564 | 100 | 495 | 2.479 | 50 | 138 |
| Amphipoda | 107 | 0 | – | 0 | 8 | 2.748 | 23 | 41 | 1.400 | 44 | 1 | 3.742 | 7 | 0 | – | 0 | 0 | – | 0 | 95 |
| Ostracoda | 0 | 73 | 2.288 | 63 | 0 | – | 0 | 41 | 1.779 | 33 | 54 | 2.190 | 29 | 32 | 2.066 | 60 | 0 | – | 0 | 88 |
| Fish juveniles | 0 | 4 | 1.836 | 25 | 0 | – | 0 | 6 | 1.825 | 33 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 75 |
| Foraminifera | + | + | + | 50 | 0 | – | 0 | 0 | – | 0 | 1349 | 2.085 | 57 | 170 | 1.377 | 60 | 40 | 2.822 | 25 | 138 |
| Isopoda | 30 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 2 | 3.742 | 7 | 0 | – | 0 | 80 | 2.828 | 13 | 50 |
| Polychaeta | 13 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 45 |
| Araneae | 0 | 1 | 2.828 | 13 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 75 |
| Total zooplankton abundance (without Artemia cysts) | 1457 | 435 | 0.671 | 100 | 4286 | 1.417 | 100 | 4304 | 1.684 | 100 | 5991 | 1.774 | 100 | 5150 | 0.045 | 100 | 1154 | 1.152 | 100 | – |
| The number of macrotaxa | 6 | 8 | – | – | 7 | – | – | 9 | – | – | 9 | – | – | 7 | – | – | 7 | – | – | – |
| Share of Artemia (without cysts) in total zooplankton abundance, % | 0 | 0 | – | – | 74 | 0.559 | – | 12 | 2.590 | – | 53 | 0.762 | – | 60 | 0.861 | – | 10 | 2.428 | – | – |
| Taxon | 2018 | 2019 | 2020 | Max Salinity, g L−1 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| May–June * | November | June | July | December | ||||||||||||
| Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | Mean, Ind. m−2 | CV | FO, % | ||
| Gastropoda | 42 | 3.162 | 9 | 2000 | 1.641 | 58 | 1110 | 1.520 | 45 | 582 | 2.332 | 23 | 105 | 2.828 | 13 | 95 |
| Chironomidae larvae | 40,711 | 1.630 | 64 | 2842 | 2.046 | 58 | 2794 | 1.562 | 73 | 22,540 | 2.041 | 92 | 2894 | 1.558 | 63 | 128 |
| Polychaeta | 168 | 1.630 | 9 | 11,332 | 3.429 | 33 | 498 | 2.533 | 27 | 32 | 3.606 | 8 | 0 | – | 0 | 95 |
| Bivalvia | 0 | – | 0 | 35 | 3.464 | 8 | 0 | – | 0 | 32 | 3.606 | 8 | 0 | – | 0 | 87 |
| Amphipoda | 337 | 1.630 | 9 | 1438 | 3.193 | 25 | 612 | 2.308 | 18 | 0 | – | 0 | 0 | – | 0 | 90 |
| Isopoda | 0 | – | 0 | 0 | – | 0 | 77 | 3.317 | 9 | 0 | – | 0 | 0 | – | 0 | 50 |
| Oligochaeta | 0 | – | 0 | 737 | 3.464 | 8 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 38 |
| Artemia, without cysts | 0 | – | 0 | 5127 | 2.180 | 25 | 115 | 3.317 | 9 | 2245 | 1.549 | 54 | 789 | 2.829 | 13 | 128 |
| Acarina | 115 | – | 18 | 105 | 3.464 | 8 | 0 | – | 0 | 0 | – | 0 | 0 | – | 0 | 92 |
| Nematoda | 97,825 | 2.612 | 73 | 28,628 | 2.481 | 67 | 60,662 | 2.323 | 82 | 31,478 | 1.309 | 85 | 947 | 1.206 | 75 | 128 |
| Ostracoda | 1225 | 2.012 | 36 | 4736 | 1.425 | 83 | 3445 | 1.171 | 82 | 939 | 1.871 | 55 | 315 | 1.181 | 50 | 128 |
| Harpacticoida | 51,821 | 1.956 | 91 | 7824 | 2.035 | 75 | 25,030 | 1.831 | 73 | 8388 | 1.517 | 85 | 316 | 1.182 | 100 | 128 |
| Turbellaria | 82,133 | 2.457 | 45 | 3929 | 1.405 | 58 | 21,766 | 2.618 | 73 | 1360 | 1.036 | 69 | 1263 | 2.696 | 25 | 128 |
| Kinorhyncha | 0 | – | 0 | 0 | – | 0 | 306 | 1.953 | 27 | 0 | – | 0 | 0 | – | 0 | 70 |
| Ciliata | 22,925 | 1.598 | 45 | 30,242 | 1.956 | 75 | 651 | 1.621 | 45 | 6606 | 1.161 | 77 | 6631 | 0.880 | 88 | 128 |
| Foraminifera | 115 | 2.365 | 18 | 246 | 1.708 | 33 | 1225 | 2.462 | 36 | 324 | 1.925 | 31 | 157 | 1.984 | 25 | 128 |
| Allogromiidae | + | – | – | 211 | 2.892 | 17 | 3942 | 1.690 | 91 | 1425 | 2.776 | 23 | 158 | 2.828 | 13 | 128 |
| Gromia | + | – | – | 35 | 3.464 | 8 | 115 | 2.371 | 18 | 0 | – | 0 | 0 | – | 0 | 54 |
| Total zoobenthos abundance | 298,948 | 1.920 | 100 | 95,672 | 1.045 | 100 | 290,069 | 1.906 | 100 | 76,298 | 0.917 | 100 | 17,577 | 0.647 | 100 | – |
| The number of macrotaxa | 11 | – | – | 16 | – | – | 15 | – | – | 12 | – | – | 10 | – | – | – |
| Share of Chironomidae larvae in total zoobenthos abundance, % | 13.6 | 1.98 | – | 5.0 | 1.871 | – | 10.0 | 1.604 | – | 29.0 | 1.059 | – | 14.8 | 1.143 | – | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anufriieva, E.; Kolesnikova, E.; Revkova, T.; Latushkin, A.; Shadrin, N. Human-Induced Sharp Salinity Changes in the World’s Largest Hypersaline Lagoon Bay Sivash (Crimea) and Their Effects on the Ecosystem. Water 2022, 14, 403. https://doi.org/10.3390/w14030403
Anufriieva E, Kolesnikova E, Revkova T, Latushkin A, Shadrin N. Human-Induced Sharp Salinity Changes in the World’s Largest Hypersaline Lagoon Bay Sivash (Crimea) and Their Effects on the Ecosystem. Water. 2022; 14(3):403. https://doi.org/10.3390/w14030403
Chicago/Turabian StyleAnufriieva, Elena, Elena Kolesnikova, Tatiana Revkova, Alexander Latushkin, and Nickolai Shadrin. 2022. "Human-Induced Sharp Salinity Changes in the World’s Largest Hypersaline Lagoon Bay Sivash (Crimea) and Their Effects on the Ecosystem" Water 14, no. 3: 403. https://doi.org/10.3390/w14030403








