Decomplexation of Ni-EDTA by Three-Dimensional Electro-Fenton
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Setup
2.3. Experimental Procedure
2.3.1. Degradation of Ni-EDTA by 3D-EF
2.3.2. The pH Window Was Widened in 3D-EF System
2.3.3. Parameter Optimization of Ni-EDTA Degradation by 3D-EF
2.3.4. The Effect of Coexisting Ions in 3D-EF System
2.4. Analytical and Statistical Methods
3. Results and Discussion
3.1. The Advantages of Ni-EDTA Decomplexation in 3D-EF System
3.2. pH Application Window of 3D-EF for Ni-EDTA Complex Breaking
| Anode | Cathode | Particle Electrode | Process Parameters | Efficiency | Reference |
|---|---|---|---|---|---|
| Ti/RuO2 | Ti/RuO2 | Activated carbon fiber loaded with MnOx | pH = 2 | Total organic carbon: 70% | [29] |
| SS316/β-PbO2 | Stainless-steel plate | Powder activated carbon/Fe3O4 | pH = 3 | 2,4-dichlorophenoxyacetic acid: 96.2% COD: 92.3% Total organic carbon: 86.5% | [30] |
| RuO2-IrO2-Ti | Activated carbon fiber | Cu-Fe/Sodium alginate Carbon | pH = 5.4 | Fulvic acid: 81.1% | [34] |
| Ti/RuO2 | Ti/RuO2 | GAC | pH = 5.56 | Amoxicillin: 98.98% Total organic carbon: 47.6% | [35] |
| Ti/RuO2-IrO2-Ta2O5 | Stainless-steel plate | AC@Ti-Cu-Ni-Zn-Sb-Mn | pH = 7 | P-aminophenol: 99.87% | [31] |
3.3. Influence Factors on Decomplexation of Ni-EDTA
3.3.1. Effect of Fe2+ Dosage
3.3.2. Effect of Particle Electrode Dosage
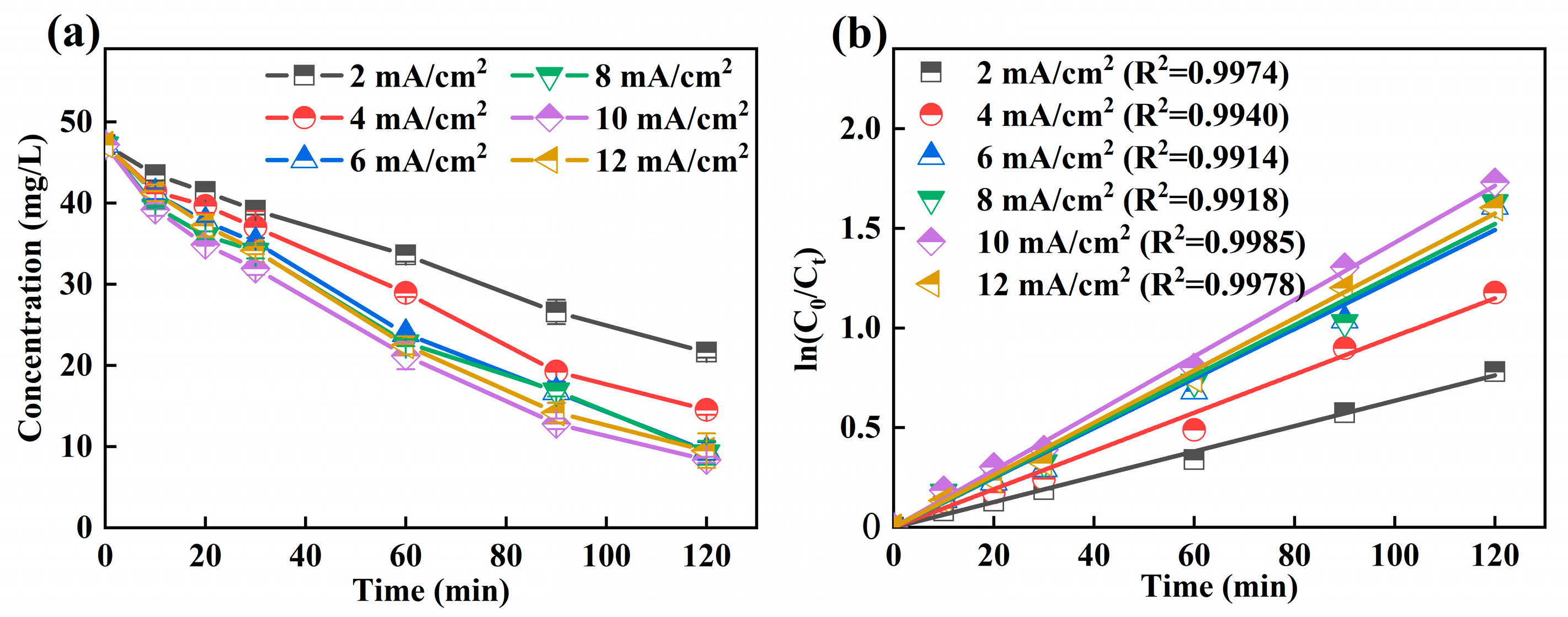
3.3.3. Effect of Current Density
3.4. Effect of Coexisting Ions on Ni-EDTA Removal
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, J.; Zhou, W.; Yang, L.; Chen, Y.; Hu, Y. Co-N-doped MoO2 modified carbon felt cathode for removal of EDTA-Ni in electro-Fenton process. Environ. Sci. Pollut. Res. 2018, 25, 22754–22765. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhou, F.; Bi, X.; Chen, D.; Huang, Z.; Li, Y.; Sun, S.; Liu, J. Decomposition of Nickel (Ⅱ)-ethylenediaminetetraacetic acid by Fenton-like reaction over oxygen vacancies-based Cu-Doped Fe3O4@ γ-Al2O3 catalyst: A synergy of oxidation and adsorption. Chemosphere 2019, 221, 563–572. [Google Scholar] [CrossRef]
- Liang, S.; Hu, X.; Xu, H.; Lei, Z.; Wei, C.; Feng, C. Mechanistic insight into the reaction pathway of peroxomonosulfate-initiated decomplexation of EDTA-NiII under alkaline conditions: Formation of high-valent Ni intermediate. Appl. Catal. B Environ. 2021, 296, 120375. [Google Scholar] [CrossRef]
- Cao, Y.; Qian, X.; Zhang, Y.; Qu, G.; Xia, T.; Guo, X.; Jia, H.; Wang, T. Decomplexation of EDTA-chelated copper and removal of copper ions by non-thermal plasma oxidation/alkaline precipitation. Chem. Eng. J. 2019, 362, 487–496. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, D.; Xue, M.; Song, R.; Zhang, Y.; Qu, G.; Wang, T. High-efficient decomplexation of Cu-EDTA and Cu removal by high-frequency non-thermal plasma oxidation/alkaline precipitation. Sep. Purif. Technol. 2021, 257, 117885. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Z.; Zhang, X.; Dong, W.; Cao, Z.; He, L.; Wang, X. Rapid decomplexation of Ni-EDTA by microwave-assisted Fenton reaction. Chem. Eng. J. 2020, 381, 122703. [Google Scholar] [CrossRef]
- Fei, L.; Ren, S.; Xijun, M.; Ali, N.; Jing, Z.; Yi, J.; Bilal, M. Efficient removal of EDTA-chelated Cu (II) by zero-valent iron and peroxydisulfate: Mutual activation process. Sep. Purif. Technol. 2021, 279, 119721. [Google Scholar] [CrossRef]
- Nguyen, M.K.; Pham, T.T.; Pham, H.G.; Hoang, B.L.; Nguyen, T.H.; Nguyen, T.H.; Tran, T.H.; Ngo, H.H. Fenton/ozone-based oxidation and coagulation processes for removing metals (Cu, Ni)-EDTA from plating wastewater. J. Water Process Eng. 2021, 39, 101836. [Google Scholar] [CrossRef]
- Nidheesh, P.; Zhou, M.; Oturan, M.A. An overview on the removal of synthetic dyes from water by electrochemical advanced oxidation processes. Chemosphere 2018, 197, 210–227. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, W.; Xu, L.; Zhou, C.; Sun, Y.; Niu, J. Treatment of Ni-EDTA containing wastewater by electrochemical degradation using Ti3+ self-doped TiO2 nanotube arrays anode. Chemosphere 2021, 278, 130465. [Google Scholar] [CrossRef]
- Sirés, I.; Arias, C.; Cabot, P.L.; Centellas, F.; Garrido, J.A.; Rodríguez, R.M.; Brillas, E. Degradation of clofibric acid in acidic aqueous medium by electro-Fenton and photoelectro-Fenton. Chemosphere 2007, 66, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Yao, B.; Luo, Z.; Yang, J.; Zhi, D.; Zhou, Y. FeII FeIII layered double hydroxide modified carbon felt cathode for removal of ciprofloxacin in electro-Fenton process. Environ. Res. 2021, 197, 111144. [Google Scholar] [CrossRef] [PubMed]
- Basturk, I.; Varank, G.; Murat-Hocaoglu, S.; Yazici-Guvenc, S.; Can-Güven, E.; Oktem-Olgun, E.E.; Canli, O. Simultaneous degradation of cephalexin, ciprofloxacin, and clarithromycin from medical laboratory wastewater by electro-Fenton process. J. Environ. Chem. Eng. 2021, 9, 104666. [Google Scholar] [CrossRef]
- Stupar, S.L.; Grgur, B.N.; Radišić, M.M.; Onjia, A.E.; Ivanković, N.D.; Tomašević, A.V.; Mijin, D.Ž. Oxidative degradation of Acid Blue 111 by electro-assisted Fenton process. J. Water Process Eng. 2020, 36, 101394. [Google Scholar] [CrossRef]
- Gamarra-Güere, C.D.; Dionisio, D.; Santos, G.O.S.; Lanza, M.R.V.; de Jesus Motheo, A. Application of Fenton, photo-Fenton and electro-Fenton processes for the methylparaben degradation: A comparative study. J. Environ. Chem. Eng. 2022, 10, 106992. [Google Scholar] [CrossRef]
- Li, H.; Yang, H.; Cheng, J.; Hu, C.; Yang, Z.; Wu, C. Three-dimensional particle electrode system treatment of organic wastewater: A general review based on patents. J. Clean. Prod. 2021, 308, 127324. [Google Scholar] [CrossRef]
- Lu, C.; Wei, G.; Ba, J.; Li, Q.; Zhang, L.; Li, Z.; Cen, L.; Ma, J.; Song, Y. Three-dimensional electro-Fenton degradation of ciprofloxacin catalyzed by CuO doped red mud particle electrodes: Electrodes preparation, kinetics and mechanism. J. Environ. Chem. Eng. 2022, 10, 107151. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Z.; Wu, P.; Duan, Y.; Zhou, L.; Lai, Y.; Wang, F.; Li, S. Three-dimensional heterogeneous Electro-Fenton system with a novel catalytic particle electrode for Bisphenol A removal. J. Hazard. Mater. 2020, 393, 120448. [Google Scholar] [CrossRef]
- Mohammadi, H.; Bina, B.; Ebrahimi, A. A novel three-dimensional electro-Fenton system and its application for degradation of anti-inflammatory pharmaceuticals: Modeling and degradation pathways. Process Saf. Environ. Prot. 2018, 117, 200–213. [Google Scholar] [CrossRef]
- Zhang, C.; Jiang, Y.; Li, Y.; Hu, Z.; Zhou, L.; Zhou, M. Three-dimensional electrochemical process for wastewater treatment: A general review. Chem. Eng. J. 2013, 228, 455–467. [Google Scholar] [CrossRef]
- Zhou, L.; Zhou, M.; Zhang, C.; Jiang, Y.; Bi, Z.; Yang, J. Electro-Fenton degradation of p-nitrophenol using the anodized graphite felts. Chem. Eng. J. 2013, 233, 185–192. [Google Scholar] [CrossRef]
- Chu, Y.; Su, H.; Lv, R.; Zhang, X. Enhanced electro-reduction of Fe3+ to Fe2+ by acidified carbon nanotube-modified graphite cathode and its application in a novel Fenton process for p-nitrophenol degradation. J. Water Process Eng. 2021, 40, 101912. [Google Scholar] [CrossRef]
- Qi, H.; Sun, X.; Sun, Z. Porous graphite felt electrode with catalytic defects for enhanced degradation of pollutants by electro-Fenton process. Chem. Eng. J. 2021, 403, 126270. [Google Scholar] [CrossRef]
- Song, D.; Li, J.; Wang, Z.; Zhao, C. Performance of graphite felt as anodes in the electro-Fenton oxidation systems: Changes in catalysis, conductivity and adsorption properties. Appl. Surf. Sci. 2020, 532, 147450. [Google Scholar] [CrossRef]
- Kuleyin, A.; Gök, A.; Akbal, F. Treatment of textile industry wastewater by electro-Fenton process using graphite electrodes in batch and continuous mode. J. Environ. Chem. Eng. 2021, 9, 104782. [Google Scholar] [CrossRef]
- Zhao, H.; Sun, Y.; Xu, L.-N.; Ni, J.-R. Removal of Acid Orange 7 in simulated wastewater using a three-dimensional electrode reactor: Removal mechanisms and dye degradation pathway. Chemosphere 2010, 78, 46–51. [Google Scholar] [CrossRef]
- Wang, L.; Hu, Y.; Li, P.; Zhang, Y.; Yan, Q.; Zhao, Y. Electrochemical treatment of industrial wastewater using a novel layer-upon-layer bipolar electrode system (nLBPEs). Chem. Eng. J. 2013, 215, 157–161. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Z.; Li, J.; Zhao, C.; He, X.; Yang, G. Fabrication of slag particle three-dimensional electrode system for methylene blue degradation: Characterization, performance and mechanism study. Chemosphere 2018, 213, 377–383. [Google Scholar] [CrossRef]
- Liu, W.; Duan, P.; Hu, X.; Gao, J.; Zhou, F. Fabrication of Efficient Nano-MnOx/ACF Particle Electrodes and their Application in the Electrooxidation of m-Cresol in the 3-D Electrode System. Ind. Eng. Chem. Res. 2019, 58, 22114–22123. [Google Scholar] [CrossRef]
- Dargahi, A.; Hasani, K.; Mokhtari, S.A.; Vosoughi, M.; Moradi, M.; Vaziri, Y. Highly effective degradation of in a three-dimensional sono-electro-Fenton (3D/SEF) system using powder activated carbon (PAC)/Fe3O4 as magnetic particle electrode. J. Environ. Chem. Eng. 2021, 9, 105889. [Google Scholar] [CrossRef]
- Wan, J.; Zhao, F.; Meng, Y.; Guo, M.; Tang, C.; Shi, Y.; Ke, Y.; Hu, R. Three-dimensional electrochemical degradation of p-aminophenol with efficient honeycomb block AC@Ti-Cu-Ni-Zn-Sb-Mn particle electrodes. Sep. Purif. Technol. 2021, 267, 118662. [Google Scholar] [CrossRef]
- Zheng, Y.; Qiu, S.; Deng, F.; Zhu, Y.; Li, G.; Ma, F. Three-dimensional electro-Fenton system with iron foam as particle electrode for folic acid wastewater pretreatment. Sep. Purif. Technol. 2019, 224, 463–474. [Google Scholar] [CrossRef]
- Guan, W.; Zhang, B.; Tian, S.; Zhao, X. The synergism between electro-Fenton and electrocoagulation process to remove Cu-EDTA. Appl. Catal. B Environ. 2018, 227, 252–257. [Google Scholar] [CrossRef]
- Li, M.; Qin, X.; Cui, J.; Guo, R.; Guo, C.; Wang, Z.; Li, T. Three-dimensional Electro-Fenton degradation for fulvic acids with Cu-Fe bimetallic aerogel-like carbon as particle electrode and catalyst: Electrode preparation, operation parameter optimization and mechanism. J. Environ. Chem. Eng. 2021, 9, 105573. [Google Scholar] [CrossRef]
- Shi, H.; Wang, Q.; Ni, J.; Xu, Y.; Song, N.; Gao, M. Highly efficient removal of amoxicillin from water by three-dimensional electrode system within granular activated carbon as particle electrode. J. Water Process Eng. 2020, 38, 101656. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, Z.; Wang, H.; Dong, W.; Wang, W. Sequential application of Fenton and ozone-based oxidation process for the abatement of Ni-EDTA containing nickel plating effluents. Chemosphere 2018, 202, 238–245. [Google Scholar] [CrossRef]
- Li, M.; Zeng, Z.; Li, Y.; Arowo, M.; Chen, J.; Meng, H.; Shao, L. Treatment of amoxicillin by O3/Fenton process in a rotating packed bed. J. Environ. Manag. 2015, 150, 404–411. [Google Scholar] [CrossRef]
- Li, J.; Ren, Y.; Lai, L.; Lai, B. Electrolysis assisted persulfate with annular iron sheet as anode for the enhanced degradation of 2,4-dinitrophenol in aqueous solution. J. Hazard. Mater. 2018, 344, 778–787. [Google Scholar] [CrossRef]
- Barhoumi, N.; Oturan, N.; Olvera-Vargas, H.; Brillas, E.; Gadri, A.; Ammar, S.; Oturan, M.A. Pyrite as a sustainable catalyst in electro-Fenton process for improving oxidation of sulfamethazine. Kinetics, mechanism and toxicity assessment. Water Res. 2016, 94, 52–61. [Google Scholar] [CrossRef]
- Zhang, B.; Hou, Y.; Yu, Z.; Liu, Y.; Huang, J.; Qian, L.; Xiong, J. Three-dimensional electro-Fenton degradation of Rhodamine B with efficient Fe-Cu/kaolin particle electrodes: Electrodes optimization, kinetics, influencing factors and mechanism. Sep. Purif. Technol. 2019, 210, 60–68. [Google Scholar] [CrossRef]
- Oturan, N.; Wu, J.; Zhang, H.; Sharma, V.K.; Oturan, M.A. Electrocatalytic destruction of the antibiotic tetracycline in aqueous medium by electrochemical advanced oxidation processes: Effect of electrode materials. Appl. Catal. B Environ. 2013, 140, 92–97. [Google Scholar] [CrossRef]
- Lan, Y.; Coetsier, C.; Causserand, C.; Serrano, K.G. On the role of salts for the treatment of wastewaters containing pharmaceuticals by electrochemical oxidation using a boron doped diamond anode. Electrochim. Acta 2017, 231, 309–318. [Google Scholar] [CrossRef] [Green Version]
- Ozturk, D.; Yilmaz, A.E. Treatment of slaughterhouse wastewater with the electrochemical oxidation process: Role of operating parameters on treatment efficiency and energy consumption. J. Water Process Eng. 2019, 31, 100834. [Google Scholar] [CrossRef]
- Jiang, Z.; Lv, L.; Zhang, W.; Du, Q.; Pan, B.; Yang, L.; Zhang, Q. Nitrate reduction using nanosized zero-valent iron supported by polystyrene resins: Role of surface functional groups. Water Res. 2011, 45, 2191–2198. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Zou, X.; Mao, J.; Wu, X. Decomposition of sulfadiazine in a sonochemical Fe0-catalyzed persulfate system: Parameters optimizing and interferences of wastewater matrix. Appl. Catal. B Environ. 2016, 185, 31–41. [Google Scholar] [CrossRef]
- Teng, X.; Li, J.; Wang, J.; Liu, J.; Ge, X.; Gu, T. Effective degradation of atrazine in wastewater by three-dimensional electrochemical system using fly ash-red mud particle electrode: Mechanism and pathway. Sep. Purif. Technol. 2021, 267, 118661. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, J.; Ma, Y.; Huang, X.; Yu, J.; Yu, F.; Gao, J. Decomplexation of Ni-EDTA by Three-Dimensional Electro-Fenton. Water 2022, 14, 1420. https://doi.org/10.3390/w14091420
Peng J, Ma Y, Huang X, Yu J, Yu F, Gao J. Decomplexation of Ni-EDTA by Three-Dimensional Electro-Fenton. Water. 2022; 14(9):1420. https://doi.org/10.3390/w14091420
Chicago/Turabian StylePeng, Juan, Yameng Ma, Xiao Huang, Jianghua Yu, Fengjiao Yu, and Jingsi Gao. 2022. "Decomplexation of Ni-EDTA by Three-Dimensional Electro-Fenton" Water 14, no. 9: 1420. https://doi.org/10.3390/w14091420
APA StylePeng, J., Ma, Y., Huang, X., Yu, J., Yu, F., & Gao, J. (2022). Decomplexation of Ni-EDTA by Three-Dimensional Electro-Fenton. Water, 14(9), 1420. https://doi.org/10.3390/w14091420






