A Review of the Status, Effects, Prevention, and Remediation of Groundwater Contamination for Sustainable Environment
Abstract
:1. Introduction
2. Global Scenario
3. Freshwater Utilization
3.1. Agricultural
3.2. Industries
3.3. Domestic
4. Potential Sources of Contamination
4.1. Natural Process
4.2. Agricultural Practices
4.3. Hazardous Waste Disposal
4.4. Municipal Solid Waste
4.5. Extraction of Natural Gas
4.6. Mining and Quarrying
4.7. Climate Change
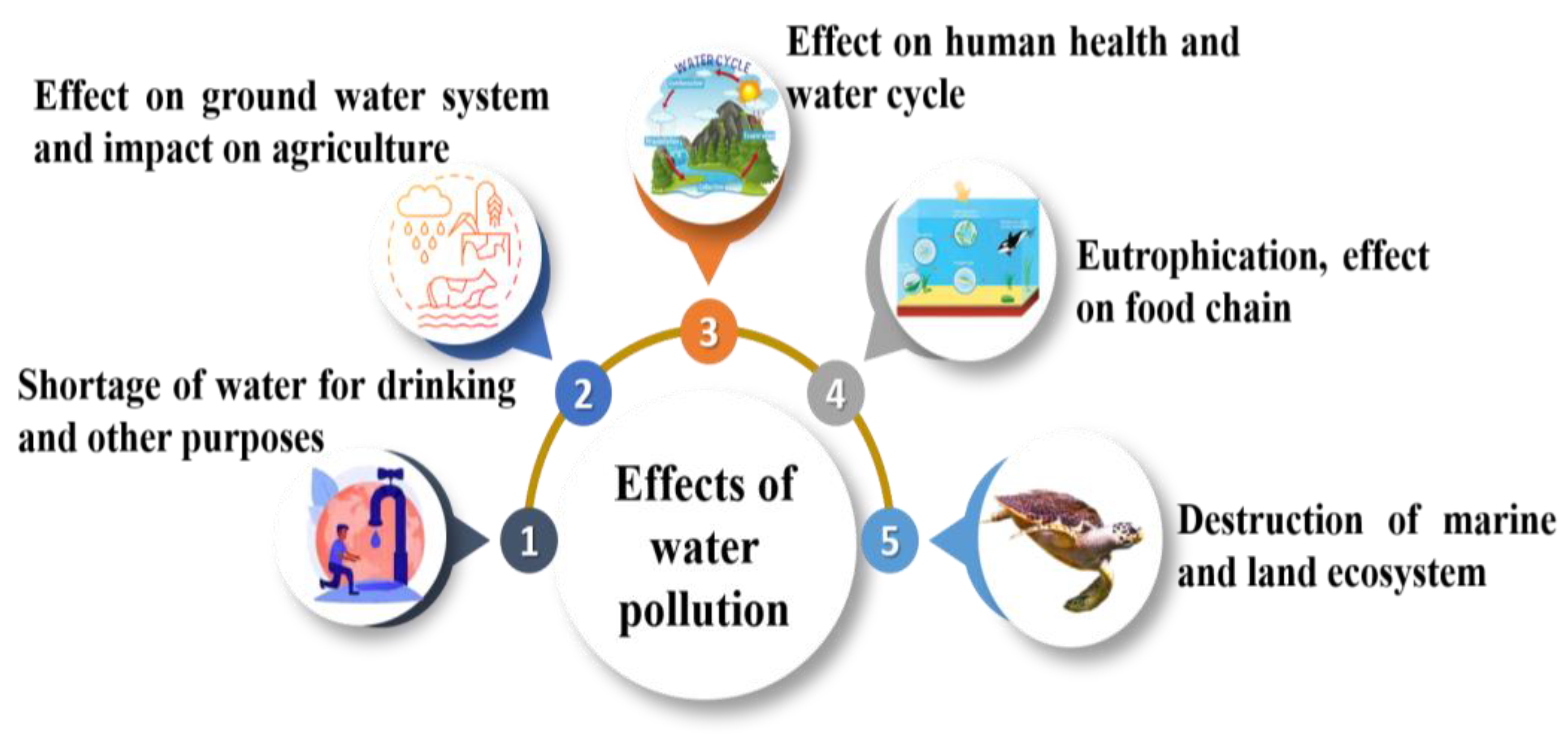
5. Consequences of Groundwater Contamination
5.1. Human Health
5.2. Economic Growth
5.3. Imbalance in Aquatic Ecosystem
5.4. Freshwater Scarcity
5.5. Vegetation
6. Prevention of Groundwater Pollution
7. Remediation Technique for Groundwater Pollution
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Okello, C.; Tomasello, B.; Greggio, N.; Wambiji, N.; Antonellini, M. Impact of Population Growth and Climate Change on the Freshwater Resources of Lamu Island, Kenya. Water 2015, 7, 1264–1290. [Google Scholar] [CrossRef]
- Cassardo, C.; Jones, J.A.A. Managing Water in a Changing World. Water 2011, 3, 618–628. [Google Scholar] [CrossRef]
- Kummu, M.; de Moel, H.; Ward, P.J.; Varis, O. How Close Do We Live to Water? A Global Analysis of Population to Freshwater Bodies. PLoS ONE 2011, 6, e20578. [Google Scholar] [CrossRef] [PubMed]
- Dinka, M.O.; Dinka, M.O. Safe Drinking Water: Concepts, Benefits, Principles and Standards. In Water Challenges of an Urbanizing World; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Ismail, Z.; Go, Y.I. Fog-to-Water for Water Scarcity in Climate-Change Hazards Hotspots: Pilot Study in Southeast Asia. Glob. Chall. 2021, 5, 2000036. [Google Scholar] [CrossRef] [PubMed]
- Damkjaer, S.; Taylor, R. The Measurement of Water Scarcity: Defining a Meaningful Indicator. Ambio 2017, 46, 513. [Google Scholar] [CrossRef] [PubMed]
- Adimalla, N.; Dhakate, R.; Kasarla, A.; Taloor, A.K. Appraisal of Groundwater Quality for Drinking and Irrigation Purposes in Central Telangana, India. Groundw. Sustain. Dev. 2020, 10, 100334. [Google Scholar] [CrossRef]
- Li, P.; Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K. Sources and Consequences of Groundwater Contamination. Arch. Environ. Contam. Toxicol. 2021, 80, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lenin Sundar, M.; Ragunath, S.; Hemalatha, J.; Vivek, S.; Mohanraj, M.; Sampathkumar, V.; Mohammed Siraj Ansari, A.; Parthiban, V.; Manoj, S. Simulation of Ground Water Quality for Noyyal River Basin of Coimbatore City, Tamilnadu Using MODFLOW. Chemosphere 2022, 306, 135649. [Google Scholar] [CrossRef] [PubMed]
- Carrard, N.; Foster, T.; Willetts, J. Groundwater as a Source of Drinking Water in Southeast Asia and the Pacific: A Multi-Country Review of Current Reliance and Resource Concerns. Water 2019, 11, 1605. [Google Scholar] [CrossRef]
- Swain, S.; Taloor, A.K.; Dhal, L.; Sahoo, S.; Al-Ansari, N. Impact of Climate Change on Groundwater Hydrology: A Comprehensive Review and Current Status of the Indian Hydrogeology. Appl. Water Sci. 2022, 12, 120. [Google Scholar] [CrossRef]
- Akhtar, N.; Syakir Ishak, M.I.; Bhawani, S.A.; Umar, K. Various Natural and Anthropogenic Factors Responsible for Water Quality Degradation: A Review. Water 2021, 13, 2660. [Google Scholar] [CrossRef]
- Roy, T.; Dey, T.K.; Jamal, M. Microplastic/Nanoplastic Toxicity in Plants: An Imminent Concern. Environ. Monit. Assess. 2022, 195, 27. [Google Scholar] [CrossRef]
- Rodríguez-Cruz, S.; Marín-Benito, J.M.; Peña, A.; Antonio Rodríguez-Liébana, J.; Delgado-Moreno, L. Interactions of Microplastics with Pesticides in Soils and Their Ecotoxicological Implications. Agronomy 2023, 13, 701. [Google Scholar] [CrossRef]
- Subaramaniyam, U.; Allimuthu, R.S.; Vappu, S.; Ramalingam, D.; Balan, R.; Paital, B.; Panda, N.; Rath, P.K.; Ramalingam, N.; Sahoo, D.K. Effects of Microplastics, Pesticides and Nano-Materials on Fish Health, Oxidative Stress and Antioxidant Defense Mechanism. Front. Physiol. 2023, 14, 1217666. [Google Scholar] [CrossRef] [PubMed]
- Ullah, Z.; Rashid, A.; Ghani, J.; Nawab, J.; Zeng, X.C.; Shah, M.; Alrefaei, A.F.; Kamel, M.; Aleya, L.; Abdel-Daim, M.M.; et al. Groundwater Contamination through Potentially Harmful Metals and Its Implications in Groundwater Management. Front. Environ. Sci. 2022, 10, 1021596. [Google Scholar] [CrossRef]
- Wang, H.; Liu, S.; Du, S.; Wang, H.; Liu, S.; Du, S. The Investigation and Assessment on Groundwater Organic Pollution. Org. Pollut. Monit. Risk Treat. 2013, 4, 87–110. [Google Scholar] [CrossRef]
- Riebe, C.S.; Hahm, W.J.; Brantley, S.L. Controls on Deep Critical Zone Architecture: A Historical Review and Four Testable Hypotheses. Earth Surf. Process. Landforms 2017, 42, 128–156. [Google Scholar] [CrossRef]
- Wymore, A.S.; Ward, A.S.; Wohl, E.; Harvey, J.W. Viewing River Corridors through the Lens of Critical Zone Science. Front. Water 2023, 5, 1147561. [Google Scholar] [CrossRef]
- Triassi, M.; Cerino, P.; Montuori, P.; Pizzolante, A.; Trama, U.; Nicodemo, F.; D’Auria, J.L.; de Vita, S.; de Rosa, E.; Limone, A. Heavy Metals in Groundwater of Southern Italy: Occurrence and Potential Adverse Effects on the Environment and Human Health. Int. J. Environ. Res. Public Health 2023, 20, 1693. [Google Scholar] [CrossRef]
- Brindha, K.; Paul, R.; Walter, J.; Tan, M.L.; Singh, M.K. Trace Metals Contamination in Groundwater and Implications on Human Health: Comprehensive Assessment Using Hydrogeochemical and Geostatistical Methods. Environ. Geochem. Health 2020, 42, 3819. [Google Scholar] [CrossRef]
- Shaji, E.; Santosh, M.; Sarath, K.V.; Prakash, P.; Deepchand, V.; Divya, B.V. Arsenic Contamination of Groundwater: A Global Synopsis with Focus on the Indian Peninsula. Geosci. Front. 2021, 12, 101079. [Google Scholar] [CrossRef]
- Subramanian, K.; Sreevidya, V.; Venkatasubramani, R.; Sivakumar, V. DRASTIC Model Developed with Lineament Density to Map Groundwater Susceptibility: A Case Study in Part of Coimbatore District, Tamilnadu, India. Environ. Dev. Sustain. 2023, 25, 10411–10423. [Google Scholar] [CrossRef]
- Meenakshi Balasubramanian, S.; Bhaskar, A.S.; Sivakumar, V. Block Level Assessment of Groundwater Potential Zones Using Hydrogeological and Remote Sensing and GIS Data’s: A Scientific Approach to Prevent Water Scarcity Problems. Desalination Water Treat. 2023, 297, 227–239. [Google Scholar] [CrossRef]
- El-taweel, R.M.; Mohamed, N.; Alrefaey, K.A.; Husien, S.; Abdel-Aziz, A.B.; Salim, A.I.; Mostafa, N.G.; Said, L.A.; Fahim, I.S.; Radwan, A.G. A Review of Coagulation Explaining Its Definition, Mechanism, Coagulant Types, and Optimization Models; RSM, and ANN. Curr. Res. Green Sustain. Chem. 2023, 6, 100358. [Google Scholar] [CrossRef]
- Pathak, V.M.; Verma, V.K.; Rawat, B.S.; Kaur, B.; Babu, N.; Sharma, A.; Dewali, S.; Yadav, M.; Kumari, R.; Singh, S.; et al. Current Status of Pesticide Effects on Environment, Human Health and It’s Eco-Friendly Management as Bioremediation: A Comprehensive Review. Front. Microbiol. 2022, 13, 962619. [Google Scholar] [CrossRef]
- Sharma, I.; Sharma, I. Bioremediation Techniques for Polluted Environment: Concept, Advantages, Limitations, and Prospects. In Trace Metals in the Environment—New Approaches and Recent Advances; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Alori, E.T.; Gabasawa, A.I.; Elenwo, C.E.; Agbeyegbe, O.O. Bioremediation Techniques as Affected by Limiting Factors in Soil Environment. Front. Soil Sci. 2022, 2, 937186. [Google Scholar] [CrossRef]
- Câmara, J.S.; Montesdeoca-Esponda, S.; Freitas, J.; Guedes-Alonso, R.; Sosa-Ferrera, Z.; Perestrelo, R. Emerging Contaminants in Seafront Zones. Environmental Impact and Analytical Approaches. Separations 2021, 8, 95. [Google Scholar] [CrossRef]
- Rout, P.R.; Zhang, T.C.; Bhunia, P.; Surampalli, R.Y. Treatment Technologies for Emerging Contaminants in Wastewater Treatment Plants: A Review. Sci. Total Environ. 2021, 753, 141990. [Google Scholar] [CrossRef]
- Valdez-Carrillo, M.; Abrell, L.; Ramírez-Hernández, J.; Reyes-López, J.A.; Carreón-Diazconti, C. Pharmaceuticals as Emerging Contaminants in the Aquatic Environment of Latin America: A Review. Environ. Sci. Pollut. Res. 2020, 27, 44863–44891. [Google Scholar] [CrossRef]
- Stefanarou, A.S.; Chrysikopoulos, C.V. Interaction of Titanium Dioxide with Formaldehyde in the Presence of Quartz Sand under Static and Dynamic Conditions. Water 2021, 13, 1420. [Google Scholar] [CrossRef]
- Fountouli, T.V.; Chrysikopoulos, C.V. Effect of Clay Colloid Particles on Formaldehyde Transport in Unsaturated Porous Media. Water 2020, 12, 3541. [Google Scholar] [CrossRef]
- Chrysikopoulos, C.V.; Fountouli, T.V. Cotransport of Titanium Dioxide Nanoparticles and Formaldehyde in Saturated and Unsaturated Columns Packed with Quartz Sand. Vadose Zone J. 2023, 22, e20175. [Google Scholar] [CrossRef]
- Georgopoulou, M.P.; Syngouna, V.I.; Chrysikopoulos, C.V. Influence of Graphene Oxide Nanoparticles on the Transport and Cotransport of Biocolloids in Saturated Porous Media. Colloids Surf. B Biointerfaces 2020, 189, 110841. [Google Scholar] [CrossRef] [PubMed]
- Syngouna, V.I.; Chrysikopoulos, C.V.; Kokkinos, P.; Tselepi, M.A.; Vantarakis, A. Cotransport of Human Adenoviruses with Clay Colloids and TiO2 Nanoparticles in Saturated Porous Media: Effect of Flow Velocity. Sci. Total Environ. 2017, 598, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Syngouna, V.I.; Chrysikopoulos, C.V. Cotransport of Clay Colloids and Viruses through Water-Saturated Vertically Oriented Columns Packed with Glass Beads: Gravity Effects. Sci. Total Environ. 2016, 545–546, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Syngouna, V.I.; Chrysikopoulos, C.V. Cotransport of Clay Colloids and Viruses in Water Saturated Porous Media. Colloids Surfaces A Physicochem. Eng. Asp. 2013, 416, 56–65. [Google Scholar] [CrossRef]
- Syngouna, V.I.; Chrysikopoulos, C.V. Transport of Biocolloids in Water Saturated Columns Packed with Sand: Effect of Grain Size and Pore Water Velocity. J. Contam. Hydrol. 2011, 126, 301–314. [Google Scholar] [CrossRef]
- Vasiliadou, I.A.; Chrysikopoulos, C.V. Cotransport of Pseudomonas Putida and Kaolinite Particles through Water-Saturated Columns Packed with Glass Beads. Water Resour. Res. 2011, 47, 2543. [Google Scholar] [CrossRef]
- Kumar, R.; Qureshi, M.; Vishwakarma, D.K.; Al-Ansari, N.; Kuriqi, A.; Elbeltagi, A.; Saraswat, A. A Review on Emerging Water Contaminants and the Application of Sustainable Removal Technologies. Case Stud. Chem. Environ. Eng. 2022, 6, 100219. [Google Scholar] [CrossRef]
- Islam, S.M.F.; Karim, Z.; Islam, S.M.F.; Karim, Z. World’s Demand for Food and Water: The Consequences of Climate Change. In Desalination—Challenges and Opportunities; IntechOpen: London, UK, 2019; pp. 1–27. [Google Scholar] [CrossRef]
- Kummu, M.; Guillaume, J.H.A.; de Moel, H.; Eisner, S.; Flörke, M.; Porkka, M.; Siebert, S.; Veldkamp, T.I.E.; Ward, P.J. The World’s Road to Water Scarcity: Shortage and Stress in the 20th Century and Pathways towards Sustainability. Sci. Rep. 2016, 6, 38495. [Google Scholar] [CrossRef]
- He, C.; Liu, Z.; Wu, J.; Pan, X.; Fang, Z.; Li, J.; Bryan, B.A. Future Global Urban Water Scarcity and Potential Solutions. Nat. Commun. 2021, 12, 4667. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Water Use and Stress. Our World Data 2017. Available online: https://ourworldindata.org/water-use-stress (accessed on 23 August 2023).
- Rodríguez, C.; García, B.; Pinto, C.; Sánchez, R.; Serrano, J.; Leiva, E. Water Context in Latin America and the Caribbean: Distribution, Regulations and Prospects for Water Reuse and Reclamation. Water 2022, 14, 3589. [Google Scholar] [CrossRef]
- Odhiambo, G.O. Water Scarcity in the Arabian Peninsula and Socio-Economic Implications. Appl. Water Sci. 2017, 7, 2479–2492. [Google Scholar] [CrossRef]
- Tzanakakis, V.A.; Paranychianakis, N.V.; Angelakis, A.N. Water Supply and Water Scarcity. Water 2020, 12, 2347. [Google Scholar] [CrossRef]
- Tariq, M.A.U.R.; Alotaibi, R.; Weththasinghe, K.K.; Rajabi, Z. A Detailed Perspective of Water Resource Management in a Dry and Water Scarce Country: The Case in Kuwait. Front. Environ. Sci. 2022, 10, 1073834. [Google Scholar] [CrossRef]
- Senthil Kumar, P.; Yaashikaa, P.R. Introduction—Water. In Water in Textiles and Fashion. Consumption, Footprint, and Life Cycle Assessment; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–20. [Google Scholar] [CrossRef]
- Costa, D.; Zhang, H.; Levison, J. Impacts of Climate Change on Groundwater in the Great Lakes Basin: A Review. J. Great Lakes Res. 2021, 47, 1613–1625. [Google Scholar] [CrossRef]
- Schaible, G.D.; Aillery, M.P. Water Conservation in Irrigated Agriculture: Trends and Challenges in the Face of Emerging Demands; United States Department of Agriculture Economic Research Service: Washington, DC, USA, 2012. [Google Scholar]
- Gleick, P.H. The World’s Water; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Boretti, A.; Rosa, L. Reassessing the Projections of the World Water Development Report. NPJ Clean Water 2019, 2, 15. [Google Scholar] [CrossRef]
- Atimtay, A.T.; Sikdar, S.K. Security of Industrial Water Supply and Management; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Brook, B.W.; Alonso, A.; Meneley, D.A.; Misak, J.; Blees, T.; van Erp, J.B. Why Nuclear Energy Is Sustainable and Has to Be Part of the Energy Mix. Sustain. Mater. Technol. 2014, 1, 8–16. [Google Scholar] [CrossRef]
- Doungmanee, P. The Nexus of Agricultural Water Use and Economic Development Level. Kasetsart J. Soc. Sci. 2016, 37, 38–45. [Google Scholar] [CrossRef]
- Lin, L.; Yang, H.; Xu, X. Effects of Water Pollution on Human Health and Disease Heterogeneity: A Review. Front. Environ. Sci. 2022, 10, 880246. [Google Scholar] [CrossRef]
- Yang, Y.; Li, P.; Elumalai, V.; Ning, J.; Xu, F.; Mu, D. Groundwater Quality Assessment Using EWQI With Updated Water Quality Classification Criteria: A Case Study in and Around Zhouzhi County, Guanzhong Basin (China). Expo. Health 2022, 1–16. [Google Scholar] [CrossRef]
- Our World in Data. Natural Disasters. Available online: https://ourworldindata.org/natural-disasters (accessed on 12 August 2023).
- Babuji, P.; Thirumalaisamy, S.; Duraisamy, K.; Periyasamy, G. Human Health Risks Due to Exposure to Water Pollution: A Review. Water 2023, 15, 2532. [Google Scholar] [CrossRef]
- Hug, S.J.; Winkel, L.H.E.; Voegelin, A.; Berg, M.; Johnson, A.C. Arsenic and Other Geogenic Contaminants in Groundwater—A Global Challenge. Chimia 2020, 74, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Singh, R.; Arfin, T.; Neeti, K. Fluoride Contamination, Consequences and Removal Techniques in Water: A Review. Environ. Sci. Adv. 2022, 1, 620–661. [Google Scholar] [CrossRef]
- Ma, L.; Wu, J.; Abuduwaili, J.; Liu, W. Geochemical Responses to Anthropogenic and Natural Influences in Ebinur Lake Sediments of Arid Northwest China. PLoS ONE 2016, 11, e0155819. [Google Scholar] [CrossRef] [PubMed]
- Onipe, T.; Edokpayi, J.N.; Odiyo, J.O. Geochemical Characterization and Assessment of Fluoride Sources in Groundwater of Siloam Area, Limpopo Province, South Africa. Sci. Rep. 2021, 11, 14000. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, B.; Chand, S.; Chand, S.; Rout, P.R.; Naik, S.K. Emerging Groundwater Contaminants: A Comprehensive Review on Their Health Hazards and Remediation Technologies. Groundw. Sustain. Dev. 2023, 20, 100868. [Google Scholar] [CrossRef]
- Rehman, F.; Siddique, J.; Shahab, A.; Azeem, T.; Bangash, A.A.; Naseem, A.A.; Riaz, O.; ur Rehman, Q. Hydrochemical Appraisal of Fluoride Contamination in Groundwater and Human Health Risk Assessment at Isa Khel, Punjab, Pakistan. Environ. Technol. Innov. 2022, 27, 102445. [Google Scholar] [CrossRef]
- Priyanka, M.; Venkata, R.G.; Ratnakar, D. Groundwater Quality Appraisal and Its Hydrochemical Characterization in and around Iron Ore Mine, Chitradurga, Karnataka. Int. J. Hydrol. 2017, 1, 151–161. [Google Scholar] [CrossRef]
- Masuda, H. Arsenic Cycling in the Earth’s Crust and Hydrosphere: Interaction between Naturally Occurring Arsenic and Human Activities. Prog. Earth Planet. Sci. 2018, 5, 68. [Google Scholar] [CrossRef]
- Åhlgren, K.; Sjöberg, V.; Allard, B.; Bäckström, M. Groundwater Chemistry Affected by Trace Elements (As, Mo, Ni, U and V) from a Burning Alum Shale Waste Deposit, Kvarntorp, Sweden. Environ. Sci. Pollut. Res. 2021, 28, 30219–30241. [Google Scholar] [CrossRef]
- Gamage, A.; Gangahagedara, R.; Gamage, J.; Jayasinghe, N.; Kodikara, N.; Suraweera, P.; Merah, O. Role of Organic Farming for Achieving Sustainability in Agriculture. Farming Syst. 2023, 1, 100005. [Google Scholar] [CrossRef]
- Lee, E.; Rout, P.R.; Bae, J. The Applicability of Anaerobically Treated Domestic Wastewater as a Nutrient Medium in Hydroponic Lettuce Cultivation: Nitrogen Toxicity and Health Risk Assessment. Sci. Total Environ. 2021, 780, 146482. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, P.; Yang, N.; Yang, C.; Zhou, Y.; Li, J. Distribution, Sources and Main Controlling Factors of Nitrate in a Typical Intensive Agricultural Region, Northwestern China: Vertical Profile Perspectives. Environ. Res. 2023, 237, 116911. [Google Scholar] [CrossRef] [PubMed]
- Kundu, M.C.; Mandal, B.; Sarkar, D. Assessment of the Potential Hazards of Nitrate Contamination in Surface and Groundwater in a Heavily Fertilized and Intensively Cultivated District of India. Environ. Monit. Assess. 2008, 146, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Ayyasamy, P.M.; Rajakumar, S.; Sathishkumar, M.; Swaminathan, K.; Shanthi, K.; Lakshmanaperumalsamy, P.; Lee, S. Nitrate Removal from Synthetic Medium and Groundwater with Aquatic Macrophytes. Desalination 2009, 242, 286–296. [Google Scholar] [CrossRef]
- Rout, P.R.; Bhunia, P.; Dash, R.R. Modeling Isotherms, Kinetics and Understanding the Mechanism of Phosphate Adsorption onto a Solid Waste: Ground Burnt Patties. J. Environ. Chem. Eng. 2014, 2, 1331–1342. [Google Scholar] [CrossRef]
- Rout, P.R.; Dash, R.R.; Bhunia, P. Nutrient Removal from Binary Aqueous Phase by Dolochar: Highlighting Optimization, Single and Binary Adsorption Isotherms and Nutrient Release. Process Saf. Environ. Prot. 2016, 100, 91–107. [Google Scholar] [CrossRef]
- Rout, P.R.; Bhunia, P.; Dash, R.R. Evaluation of Kinetic and Statistical Models for Predicting Breakthrough Curves of Phosphate Removal Using Dolochar-Packed Columns. J. Water Process Eng. 2017, 17, 168–180. [Google Scholar] [CrossRef]
- Priyadarshini, A.; Sahoo, M.M.; Raut, P.R.; Mahanty, B.; Sahoo, N.K. Kinetic Modelling and Process Engineering of Phenolics Microbial and Enzymatic Biodegradation: A Current Outlook and Challenges. J. Water Process Eng. 2021, 44, 102421. [Google Scholar] [CrossRef]
- Wear, S.L.; Acuña, V.; McDonald, R.; Font, C. Sewage Pollution, Declining Ecosystem Health, and Cross-Sector Collaboration. Biol. Conserv. 2021, 255, 109010. [Google Scholar] [CrossRef]
- Guo, W.; Li, P.; Du, Q.; Zhou, Y.; Xu, D.; Zhang, Z. Hydrogeochemical Processes Regulating the Groundwater Geochemistry and Human Health Risk of Groundwater in the Rural Areas of the Wei River Basin, China. Expo. Health 2023, 1–16. [Google Scholar] [CrossRef]
- Hosseini Beinabaj, S.M.; Heydariyan, H.; Mohammad Aleii, H.; Hosseinzadeh, A. Concentration of Heavy Metals in Leachate, Soil, and Plants in Tehran’s Landfill: Investigation of the Effect of Landfill Age on the Intensity of Pollution. Heliyon 2023, 9, e13017. [Google Scholar] [CrossRef]
- Abd El-Salam, M.M.; Abu-Zuid, G.I. Impact of Landfill Leachate on the Groundwater Quality: A Case Study in Egypt. J. Adv. Res. 2015, 6, 579. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, K.K.; Barik, D. Health Hazards of Medical Waste and Its Disposal. Energy from Toxic Organic Waste for Heat and Power Generation; Woodhead Publishing: Sawston, UK, 2019; pp. 99–118. [Google Scholar] [CrossRef]
- Ravichandran, R.; Binukumar, J.; Sreeram, R.; Arunkumar, L. An Overview of Radioactive Waste Disposal Procedures of a Nuclear Medicine Department. J. Med. Phys. 2011, 36, 95. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Cui, M.; Ye, Z.; Guo, Q. Environmental Challenges from the Increasing Medical Waste since SARS Outbreak. J. Clean. Prod. 2021, 291, 125246. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, Mechanism and Health Effects of Some Heavy Metals. Interdiscip. Toxicol. 2014, 7, 60. [Google Scholar] [CrossRef]
- Dongre, R.S.; Dongre, R.S. Lead: Toxicological Profile, Pollution Aspects and Remedial Solutions. Lead Chem. 2020, 1–18. [Google Scholar] [CrossRef]
- IARC. Chromium and Chromium Compounds. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 1990. [Google Scholar]
- Sidhu, G.P.S.; Bali, A.S. Cd in the Environment: Uptake, Toxicity and Management. In Appraisal of Metal(loids) in the Ecosystem; Elsevier: Amsterdam, The Netherlands, 2022; pp. 283–300. [Google Scholar] [CrossRef]
- Wuana, R.A.; Okieimen, F.E. Heavy Metals in Contaminated Soils: A Review of Sources, Chemistry, Risks and Best Available Strategies for Remediation. ISRN Ecol. 2011, 2011, 402647. [Google Scholar] [CrossRef]
- Zhang, Z.; Su, Y.; Zhu, J.; Shi, J.; Huang, H.; Xie, B. Distribution and Removal Characteristics of Microplastics in Different Processes of the Leachate Treatment System. Waste Manag. 2021, 120, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Vaid, M.; Mehra, K.; Gupta, A. Microplastics as Contaminants in Indian Environment: A Review. Environ. Sci. Pollut. Res. 2021, 28, 68025–68052. [Google Scholar] [CrossRef]
- Rout, P.R.; Mohanty, A.; Aastha; Sharma, A.; Miglani, M.; Liu, D.; Varjani, S. Micro- and Nanoplastics Removal Mechanisms in Wastewater Treatment Plants: A Review. J. Hazard. Mater. Adv. 2022, 6, 100070. [Google Scholar] [CrossRef]
- Ren, Z.; Gui, X.; Xu, X.; Zhao, L.; Qiu, H.; Cao, X. Microplastics in the Soil-Groundwater Environment: Aging, Migration, and Co-Transport of Contaminants—A Critical Review. J. Hazard. Mater. 2021, 419, 126455. [Google Scholar] [CrossRef]
- Peysson, W.; Vulliet, E. Determination of 136 Pharmaceuticals and Hormones in Sewage Sludge Using Quick, Easy, Cheap, Effective, Rugged and Safe Extraction Followed by Analysis with Liquid Chromatography-Time-of-Flight-Mass Spectrometry. J. Chromatogr. A 2013, 1290, 46–61. [Google Scholar] [CrossRef]
- Slack, R.J.; Gronow, J.R.; Voulvoulis, N. Household Hazardous Waste in Municipal Landfills: Contaminants in Leachate. Sci. Total Environ. 2005, 337, 119–137. [Google Scholar] [CrossRef]
- Buerge, I.J.; Buser, H.R.; Kahle, M.; Müller, M.D.; Poiger, T. Ubiquitous Occurrence of the Artificial Sweetener Acesulfame in the Aquatic Environment: An Ideal Chemical Marker of Domestic Wastewater in Groundwater. Environ. Sci. Technol. 2009, 43, 4381–4385. [Google Scholar] [CrossRef]
- Woda, J.; Wen, T.; Oakley, D.; Yoxtheimer, D.; Engelder, T.; Clara Castro, M.; Brantley, S.L. Detecting and Explaining Why Aquifers Occasionally Become Degraded near Hydraulically Fractured Shale Gas Wells. Proc. Natl. Acad. Sci. USA 2018, 115, 12349–12358. [Google Scholar] [CrossRef]
- Vidic, R.D.; Brantley, S.L.; Vandenbossche, J.M.; Yoxtheimer, D.; Abad, J.D. Impact of Shale Gas Development on Regional Water Quality. Science 2013, 340, 1235009. [Google Scholar] [CrossRef]
- Colborn, T.; Kwiatkowski, C.; Schultz, K.; Bachran, M. Natural Gas Operations from a Public Health Perspective. Hum. Ecol. Risk Assess. Int. J. 2011, 17, 1039–1056. [Google Scholar] [CrossRef]
- Soni, A.K.; Soni, A.K. Mining of Minerals and Groundwater in India. In Groundwater-Resource Characterisation and Management Aspects; IntechOpen: Rijeka, Croatia, 2019. [Google Scholar] [CrossRef]
- Kausher, R.; Singh, R.; Sinha, A.K.; Sethy, S.N.; Kumar, S.; Pandey, S.; Ragab, A.E.; Mohamed, A. Assessing Impacts of Mining-Induced Land Use Changes on Groundwater and Surface Water Quality Using Isotopic and Hydrogeochemical Signatures. Sustainability 2023, 15, 11041. [Google Scholar] [CrossRef]
- Anekwe, I.M.S.; Isa, Y.M. Bioremediation of Acid Mine Drainage—Review. Alexandria Eng. J. 2023, 65, 1047–1075. [Google Scholar] [CrossRef]
- Prusty, P.; Farooq, S.H. Seawater Intrusion in the Coastal Aquifers of India—A Review. HydroResearch 2020, 3, 61–74. [Google Scholar] [CrossRef]
- Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K.; Shankar, K.; Yang, Q.; Jayasena, H.C. Coastal Groundwater Dynamics, Environmental Issues and Sustainability: A Synthesis. Mar. Pollut. Bull. 2023, 191, 114973. [Google Scholar] [CrossRef]
- Ullah, A.; Bano, A.; Khan, N. Climate Change and Salinity Effects on Crops and Chemical Communication Between Plants and Plant Growth-Promoting Microorganisms Under Stress. Front. Sustain. Food Syst. 2021, 5, 618092. [Google Scholar] [CrossRef]
- Abdalla, F. Ionic Ratios as Tracers to Assess Seawater Intrusion and to Identify Salinity Sources in Jazan Coastal Aquifer, Saudi Arabia. Arab. J. Geosci. 2016, 9, 40. [Google Scholar] [CrossRef]
- Suhartono, E.; Purwanto, P.; Suripin, S. Seawater Intrusion Modeling on Groundwater Confined Aquifer in Semarang. Procedia Environ. Sci. 2015, 23, 110–115. [Google Scholar] [CrossRef]
- Manivannan, V.; Elango, L. Seawater Intrusion and Submarine Groundwater Discharge along the Indian Coast. Environ. Sci. Pollut. Res. 2019, 26, 31592–31608. [Google Scholar] [CrossRef]
- Bierkens, M.F.P.; Wada, Y. Non-Renewable Groundwater Use and Groundwater Depletion: A Review. Environ. Res. Lett. 2019, 14, 063002. [Google Scholar] [CrossRef]
- Shivanna, K.R. Climate Change and Its Impact on Biodiversity and Human Welfare. Proc. Indian Natl. Sci. Acad. Part A Phys. Sci. 2022, 88, 160. [Google Scholar] [CrossRef]
- Ajani, A.; van der Geest, K. Climate Change in Rural Pakistan: Evidence and Experiences from a People-Centered Perspective. Sustain. Sci. 2021, 16, 1999–2011. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Clean Water and Sanitation. Our World Data 2021. Available online: https://ourworldindata.org/water-use-stress (accessed on 23 August 2023).
- Alam, S.M.K.; Li, P.; Fida, M. Groundwater Nitrate Pollution Due to Excessive Use of N-Fertilizers in Rural Areas of Bangladesh: Pollution Status, Health Risk, Source Contribution, and Future Impacts. Expo. Health 2023, 1–24. [Google Scholar] [CrossRef]
- Xu, D.; Li, P.; Chen, X.; Yang, S.; Zhang, P.; Guo, F. Major Ion Hydrogeochemistry and Health Risk of Groundwater Nitrate in Selected Rural Areas of the Guanzhong Basin, China. Hum. Ecol. Risk Assess. Int. J. 2023, 29, 701–727. [Google Scholar] [CrossRef]
- Ward, M.H.; Jones, R.R.; Brender, J.D.; de Kok, T.M.; Weyer, P.J.; Nolan, B.T.; Villanueva, C.M.; van Breda, S.G. Drinking Water Nitrate and Human Health: An Updated Review. Int. J. Environ. Res. Public Health 2018, 15, 1557. [Google Scholar] [CrossRef]
- Khalid, S.; Murtaza, B.; Shaheen, I.; Ahmad, I.; Ullah, M.I.; Abbas, T.; Rehman, F.; Ashraf, M.R.; Khalid, S.; Abbas, S.; et al. Assessment and Public Perception of Drinking Water Quality and Safety in District Vehari, Punjab, Pakistan. J. Clean. Prod. 2018, 181, 224–234. [Google Scholar] [CrossRef]
- Melanda, V.S.; Galiciolli, M.E.A.; Lima, L.S.; Figueiredo, B.C.; Oliveira, C.S. Impact of Pesticides on Cancer and Congenital Malformation: A Systematic Review. Toxics 2022, 10, 676. [Google Scholar] [CrossRef]
- Solanki, Y.S.; Agarwal, M.; Gupta, A.B.; Gupta, S.; Shukla, P. Fluoride Occurrences, Health Problems, Detection, and Remediation Methods for Drinking Water: A Comprehensive Review. Sci. Total Environ. 2022, 807, 150601. [Google Scholar] [CrossRef]
- Ghosh, G.C.; Khan, M.J.H.; Chakraborty, T.K.; Zaman, S.; Kabir, A.H.M.E.; Tanaka, H. Human Health Risk Assessment of Elevated and Variable Iron and Manganese Intake with Arsenic-Safe Groundwater in Jashore, Bangladesh. Sci. Rep. 2020, 10, 5206. [Google Scholar] [CrossRef]
- Aminur Rahman, M.; Abul Hashem, M.; Sohel Rana, M.; Rashidul Islam, M. Manganese in Potable Water of Nine Districts, Bangladesh: Human Health Risk. Environ. Sci. Pollut. Res. 2021, 28, 45663–45675. [Google Scholar] [CrossRef]
- Karunanidhi, D.; Aravinthasamy, P.; Subramani, T.; Jayasena, H.A.H.C. Perchlorate Contamination in Groundwater and Associated Health Risks from Fireworks Manufacturing Area (Sivakasi Region) of South India. Expo. Health 2022, 14, 359–373. [Google Scholar] [CrossRef]
- Sivakumar, V.; Chidambaram, S.M.; Velusamy, S.; Rathinavel, R.; Shanmugasundaram, D.K.; Sundararaj, P.; Shanmugamoorthy, M.; Thangavel, R.; Balu, K. An Integrated Approach for an Impact Assessment of the Tank Water and Groundwater Quality in Coimbatore Region of South India: Implication from Anthropogenic Activities. Environ. Monit. Assess. 2023, 195, 88. [Google Scholar] [CrossRef] [PubMed]
- Chakraborti, D.; Singh, S.K.; Rahman, M.M.; Dutta, R.N.; Mukherjee, S.C.; Pati, S.; Kar, P.B. Groundwater Arsenic Contamination in the Ganga River Basin: A Future Health Danger. Int. J. Environ. Res. Public Health 2018, 15, 180. [Google Scholar] [CrossRef]
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead Toxicity: A Review. Interdiscip. Toxicol. 2015, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Rahimzadeh, M.R.; Rahimzadeh, M.R.; Kazemi, S.; Moghadamnia, A.A. Cadmium Toxicity and Treatment: An Update. Casp. J. Intern. Med. 2017, 8, 135. [Google Scholar] [CrossRef]
- US EPA. The Effects: Economy. Available online: https://www.epa.gov/nutrientpollution/effects-economy (accessed on 16 August 2023).
- Red Tide Report (August 10, 2018). Available online: https://content.govdelivery.com/accounts/FLFFWCC/bulletins/20510d9 (accessed on 16 August 2023).
- Florida Disaster. Gov. Scott Directs Additional $4 Million for Counties Impacted by Red Tide. 18 September 2018. Available online: https://www.floridadisaster.org/news-media/news/20180918-gov.-scott-directs-additional-$4-million-for-counties-impacted-by-red-tide/ (accessed on 16 August 2023).
- Morgan, K.L.; Larkin, S.L.; Adams, C.M. Public Costs of Florida Red Tides: A Survey of Coastal Managers; University of Florida: Gainesville, FL, USA, 2008. [Google Scholar]
- Russ, J.; Zaveri, E.; Desbureaux, S.; Damania, R.; Rodella, A.S. The Impact of Water Quality on GDP Growth: Evidence from around the World. Water Secur. 2022, 17, 100130. [Google Scholar] [CrossRef]
- Learn Science at Scitable. Eutrophication: Causes, Consequences, and Controls in Aquatic Ecosystems. Available online: https://www.nature.com/scitable/knowledge/library/eutrophication-causes-consequences-and-controls-in-aquatic-102364466/ (accessed on 16 August 2023).
- Saito, M.; Magara, Y.; Wisjnuprapto. Study on Self-Purification Capacity for Organic Pollutants in Stagnant Water. Water Sci. Technol. 2002, 46, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Bashir, I.; Lone, F.A.; Bhat, R.A.; Mir, S.A.; Dar, Z.A.; Dar, S.A. Concerns and Threats of Contamination on Aquatic Ecosystems. In Bioremediation and Biotechnology: Sustainable Approaches to Pollution Degradation; Hakeem, K.R., Bhat, R.A., Qadri, H., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–26. ISBN 978-3-030-35691-0. [Google Scholar]
- Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K.; Yang, Q. Environmental Chemistry, Toxicity and Health Risk Assessment of Groundwater: Environmental Persistence and Management Strategies. Environ. Res. 2022, 214, 113884. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.U.; Qayoom, U. Implications of Sewage Discharge on Freshwater Ecosystems; China Agricultural University: Beijing, China, 2021. [Google Scholar]
- ScienceDirect Topics. Dissolved Oxygen—An Overview. Available online: https://www.sciencedirect.com/topics/earth-and-planetary-sciences/dissolved-oxygen (accessed on 16 August 2023).
- WWF. Water Scarcity|Threats. Available online: https://www.worldwildlife.org/threats/water-scarcity (accessed on 15 August 2023).
- Glanville, K.; Sheldon, F.; Butler, D.; Capon, S. Effects and Significance of Groundwater for Vegetation: A Systematic Review. Sci. Total Environ. 2023, 875, 162577. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Guan, T.; Zhou, J.; Cai, W.; Gao, N.; Du, H.; Jiang, L.; Lai, L.; Zheng, Y. Groundwater Depth and Soil Properties Are Associated with Variation in Vegetation of a Desert Riparian Ecosystem in an Arid Area of China. Forests 2018, 9, 34. [Google Scholar] [CrossRef]
- Wang, D.; Li, P.; He, X.; He, S. Exploring the Response of Shallow Groundwater to Precipitation in the Northern Piedmont of the Qinling Mountains, China. Urban Clim. 2023, 47, 101379. [Google Scholar] [CrossRef]
- Huang, F.; Zhang, Y.; Zhang, D.; Chen, X. Environmental Groundwater Depth for Groundwater-Dependent Terrestrial Ecosystems in Arid/Semiarid Regions: A Review. Int. J. Environ. Res. Public Health 2019, 16, 763. [Google Scholar] [CrossRef]
- Huang, F.; Zhang, D.; Chen, X. Vegetation Response to Groundwater Variation in Arid Environments: Visualization of Research Evolution, Synthesis of Response Types, and Estimation of Groundwater Threshold. Int. J. Environ. Res. Public Health 2019, 16, 1849. [Google Scholar] [CrossRef]
- Anandhi, A.; Karunanidhi, D.; Sankar, G.M.; Panda, S.; Kannan, N. A Framework for Sustainable Groundwater Management. Water 2022, 14, 3416. [Google Scholar] [CrossRef]
- Madsen, E.L. Determining in Situ Biodegradation. Environ. Sci. Technol. 1991, 25, 1662–1673. [Google Scholar] [CrossRef]
- ScienceDirect Topics. Wastewater Management—An Overview. Available online: https://www.sciencedirect.com/topics/earth-and-planetary-sciences/wastewater-management (accessed on 16 August 2023).
- Talabi, A.O.; Kayode, T.J.; Talabi, A.O.; Kayode, T.J. Groundwater Pollution and Remediation. J. Water Resour. Prot. 2019, 11, 1–19. [Google Scholar] [CrossRef]
- Baier, J.H.; Lykins, B.W.; Fronk, C.A.; Kramer, S.J. Using Reverse Osmosis to Remove Agricultural Chemicals from Groundwater. J. Am. Water Works Assoc. 1987, 79, 55–60. [Google Scholar] [CrossRef]
- Alfonso-Muniozguren, P.; Serna-Galvis, E.A.; Bussemaker, M.; Torres-Palma, R.A.; Lee, J. A Review on Pharmaceuticals Removal from Waters by Single and Combined Biological, Membrane Filtration and Ultrasound Systems. Ultrason. Sonochem. 2021, 76, 105656. [Google Scholar] [CrossRef]
- Badawi, A.K.; Emam, H.E.; Hamad, H.N.; Idrus, S. Recent Developments in the Application of Bio-Waste-Derived Adsorbents for the Removal of Methylene Blue from Wastewater: A Review. Polymers 2022, 14, 783. [Google Scholar] [CrossRef]
- Kataoka, R. Biodegradability and Biodegradation Pathways of Chlorinated Cyclodiene Insecticides by Soil Fungi. J. Pestic. Sci. 2018, 43, 314. [Google Scholar] [CrossRef]
- Azubuike, C.C.; Chikere, C.B.; Okpokwasili, G.C. Bioremediation Techniques–Classification Based on Site of Application: Principles, Advantages, Limitations and Prospects. World J. Microbiol. Biotechnol. 2016, 32, 180. [Google Scholar] [CrossRef]
- Sankar, K.; Karunanidhi, D.; Kalaivanan, K.; Subramani, T.; Shanthi, D.; Balamurugan, P. Integrated Hydrogeophysical and GIS Based Demarcation of Groundwater Potential and Vulnerability Zones in a Hard Rock and Sedimentary Terrain of Southern India. Chemosphere 2023, 316, 137305. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Li, P.; Wang, Y.; Du, Q. Integration of Hydrochemistry and Stable Isotopes for Assessing Groundwater Recharge and Evaporation in Pre- and Post-Rainy Seasons in Hua County, China. Nat. Resour. Res. 2023, 32, 1959–1973. [Google Scholar] [CrossRef]
- Sharma, A.; Varandani, P.D.N.S. Ground Water Remediation Technologies. Int. J. Eng. Res. Technol. 2013, 2, 1552–1559. [Google Scholar] [CrossRef]
- Li, P.; Sabarathinam, C.; Elumalai, V. Groundwater Pollution and Its Remediation for Sustainable Water Management. Chemosphere 2023, 329, 138621. [Google Scholar] [CrossRef] [PubMed]



| ECs in Groundwater | WHO Permissible Limits (µg/L) |
|---|---|
| 1,4-Dioxane | 50 |
| 17 β-estradiol | 1 |
| 2,4,6-Trichlorophenol | 200 |
| 2,4-D | 30 |
| Alachlor | 20 |
| Benzo (a) pyrene | 0.7 |
| Bisphenol-A | 0.1 |
| Bromate | 10 |
| Bromodichloromethane | 60 |
| Bromoform | 100 |
| Chlorate | 700 |
| Chlordane | 0.2 |
| Chloroform | 300 |
| Chlorpyriphos | 30 |
| DBCP | 1 |
| DDT | 1 |
| Dibromoacetonitrile | 70 |
| Dibromochloromethane | 20 |
| Dichloroacetate | 50 |
| Edetic acid | 600 |
| Endrin | 0.6 |
| Erythromycin | 0.103 |
| Lindane | 0.2 |
| Malathion | 900 |
| Mecoprop | 10 |
| N-Nitrosodimethylamine | 0.1 |
| Pentachlorophenol | 9 |
| Perchlorate | 70 |
| Styrene | 20 |
| Terbuthylazine | 7 |
| Tetrachloroethane | 40 |
| Toluene | 700 |
| Trichloroacetate | 200 |
| Vinyl chloride | 0.3 |
| Xylenes | 500 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravindiran, G.; Rajamanickam, S.; Sivarethinamohan, S.; Karupaiya Sathaiah, B.; Ravindran, G.; Muniasamy, S.K.; Hayder, G. A Review of the Status, Effects, Prevention, and Remediation of Groundwater Contamination for Sustainable Environment. Water 2023, 15, 3662. https://doi.org/10.3390/w15203662
Ravindiran G, Rajamanickam S, Sivarethinamohan S, Karupaiya Sathaiah B, Ravindran G, Muniasamy SK, Hayder G. A Review of the Status, Effects, Prevention, and Remediation of Groundwater Contamination for Sustainable Environment. Water. 2023; 15(20):3662. https://doi.org/10.3390/w15203662
Chicago/Turabian StyleRavindiran, Gokulan, Sivarethinamohan Rajamanickam, Sujatha Sivarethinamohan, Balamurugan Karupaiya Sathaiah, Gobinath Ravindran, Senthil Kumar Muniasamy, and Gasim Hayder. 2023. "A Review of the Status, Effects, Prevention, and Remediation of Groundwater Contamination for Sustainable Environment" Water 15, no. 20: 3662. https://doi.org/10.3390/w15203662
APA StyleRavindiran, G., Rajamanickam, S., Sivarethinamohan, S., Karupaiya Sathaiah, B., Ravindran, G., Muniasamy, S. K., & Hayder, G. (2023). A Review of the Status, Effects, Prevention, and Remediation of Groundwater Contamination for Sustainable Environment. Water, 15(20), 3662. https://doi.org/10.3390/w15203662











