Impact of the Controlled Dump of Fez City (Morocco): Evaluation of Metallic Trace Elements Contamination in the Sediments
Abstract
:1. Introduction
2. Study Area and Sampling
- Leachate sampling
- Sediments sampling
3. Materials and Methods
- Leachate analysis
- Sediment analysis
4. Results and Discussion
4.1. Leachate
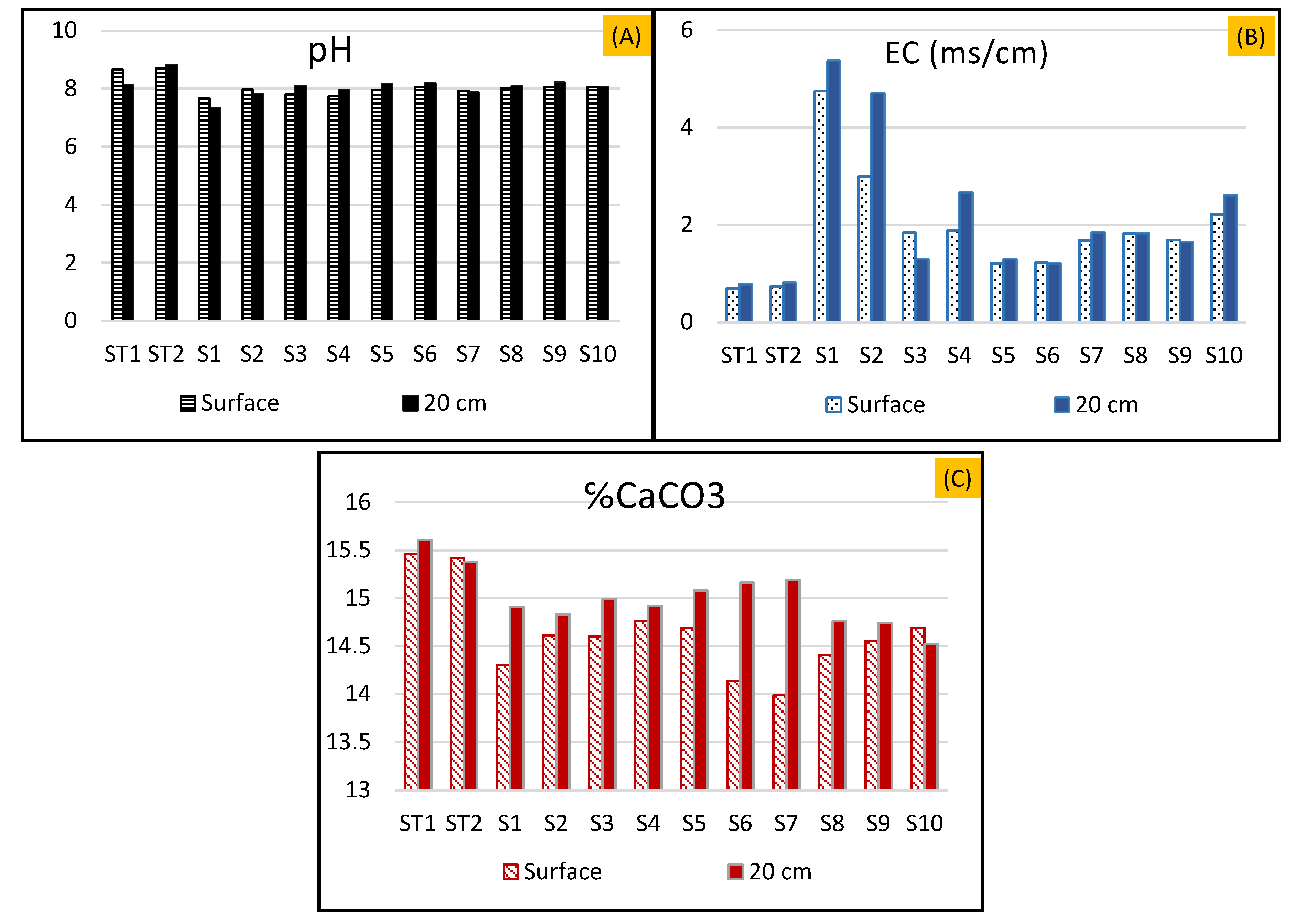
4.2. Sediments Downstream of the Dump
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smahi, D.; Fekri, A.; El Hammoumi, W. Environmental Impact of Casablanca Landfill on Groundwater Quality, Morocco. Int. J. Geosci. 2013, 4, 202–211. [Google Scholar] [CrossRef] [Green Version]
- Ben Abou, M.; El Haji, M.; Zemzami, M.; Fadil, F. Impact des lixiviats de la décharge sauvage de la ville de Taza sur les ressources hydriques (Maroc). Afr. Sci. 2014, 10, 171–180. [Google Scholar]
- Soudi, B.; Benaabou, R. Gestion des déchets ménagers dans les petites et moyennes communes au Maroc. Terre et Vie 2002; N° 53 et 54. Available online: http://terrevie.ovh.org/dechetpdf (accessed on 23 January 2023).
- Barakat, A.; El Baghdadi, M.; Rais, J.; Nadem, S. Assessment of Heavy Metal in Surface Sediments of Day River at Beni-Mellal Region, Morocco. Res. J. Environ. Earth Sci. 2012, 4, 797–806. [Google Scholar]
- Bodjona, M.B.; Kili, K.A.; Tchegueni, S.; Kennou, B.; Tchangbedji, G.; El Meray, M. Evaluation de la quantité des métaux lourds dans la décharge d’Agoè (LoméTogo): Cas du plomb, cadmium, cuivre, nickel et zinc. Int. J. Biol. Chem. Sci. 2012, 6, 1368–1380. [Google Scholar]
- Legret, M.; Divet, L.; Juste, C. Migration et spéciation des métaux lourds dans un sol soumis a des épandages de boues de station d’épuration à très forte charge en Cd et Ni. Water Ressour. 1988, 22, 953–959. [Google Scholar] [CrossRef]
- Assouguem, A.; Kara, M.; Mechchate, H.; Al-Mekhlafi, F.A.; Nasr, F.; Farah, A.; Lazraq, A. Evaluation of the Impact of Different Management Methods on Tetranychus urticae (Acari: Tetranychidae) and Their Predators in Citrus Orchards. Plants 2022, 11, 623. [Google Scholar] [CrossRef] [PubMed]
- Aun, D.L.; Christensen, T.H. Speciation of Heavy Metals in Landfill Leachate: A Review. Waste Manag. Res. 2004, 22, 3–23. [Google Scholar]
- Harris, A.; Xanthos, S.J.; Galiotos, J.K.; Douvris, C. Investigation of the metal content of sediments around the historically polluted Potomac River basin in Washington, D.C.; United States by inductively coupled plasma-optical emission spectroscopy (ICP-OES). Microchem. J. 2018, 142, 140–143. [Google Scholar] [CrossRef]
- Cambier, P.; Schvartz, C.; Van Oort, F. Contaminations Métalliques des Agrosystèmes et Ecosystèmes Péri-Industriels; Collection Update Sciences & Technologies; Editions Quae: Montpellier, France, 2009. [Google Scholar]
- Sirven, B.J. Détection de Métaux Lourds Dans les Sols par Spectroscopie D’émission sur Plasma Induit Par Laser (LIBS). Ph.D. Thesis, Université Sciences et Technologies-Bordeaux I, Bordeaux, France, 2006; 252p. [Google Scholar]
- Degryse, F.; Smolders, E.; Parker, D.R. Partitioning of metals (Cd, Co, Cu, Ni, Pb, Zn) in soils: Concepts, methodologies, prediction and applications—A review. Eur. J. Soil Sci. 2009, 60, 590–612. [Google Scholar] [CrossRef] [Green Version]
- Alamgir, M. The Effects of Soil Properties to the Extent of Soil Contamination with Metals. In Environmental Remediation Technologies for Metal-Contaminated Soils; Springer: Berlin/Heidelberg, Germany, 2016; pp. 1–19. [Google Scholar]
- Merzouki, M.; Bernet, N.; Delgenes, J.P.; Moletta, R.; Benlemlih, M. Effect of operating parameters on anoxic biological phosphorus removal in anaerobic–anoxic sequencing batch reactor. Environ. Technol. 2001, 22, 397–408. [Google Scholar] [CrossRef]
- El Fadel, H.; Merzouki, M.; Faouzi, M.; Chaouch, M.; Benlemlih, M. Traitement biologique et chimique des lixiviats de la décharge publique contrôlée de la ville de Fès au Maroc. Tech. Sci. Méthodes 2012, 6, 39–51. [Google Scholar] [CrossRef]
- El Mrabet, I.; Benzina, M.; Valdes, H.; Zaitan, H. Treatment of landfill leachates from Fez city (Morocco) using a sequence of aerobic and Fenton processes. Sci. Afr. 2020, 8, e00434. [Google Scholar] [CrossRef]
- Fedorak, P.M.; Hrudey, S.E. Anaerobic biodegradation of monochlorophenols. Water Sci. Technol. 1987, 19, 65–76. [Google Scholar]
- Ozanne, F. Les lixiviats de décharges: Le point des connaissances en 1990. Tech. Sci. Méthodes—L’Eau 1990, 5, 289–312. [Google Scholar]
- Assouguem, A.; Kara, M.; Mechchate, H.; Korkmaz, Y.B.; Benmessaoud, S.; Ramzi, A.; Abdullah, K.R.; Noman, O.M.; Farah, A.; Lazraq, A. Current Situation of Tetranychus urticae (Acari: Tetranychidae) in Northern Africa: The Sustainable Control Methods and Priorities for Future Research. Sustainability 2022, 14, 2395. [Google Scholar] [CrossRef]
- Grundfelt, B.; Hoglund, L.O. Some views on effect of different types of barriers in waste deposits Groundwater Quality. Rémédiation and Protection. In Proceedings of the Prague Conference, International Association of Hydrological Sciences (IAHS), Oxfordshire, UK, 1995; pp. 347–354. [Google Scholar]
- Khattabi, H.; Lotfi, A.; Mania, J. Evaluation de l’impact des lixiviats d’une décharge d’ordures ménagères sur la qualité physico-chimique et bactériologique des eaux d’un ruisseau de Franche-Comté. Déchets Sci. Et Tech. 2002, 24, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Chofgi, A.; Younsi, A.; El Lhadi, K.; Mania, J.; Veron, A. Impact Environnemental D’une Décharge Publique non Contrôlé sur la Population et les Eaux Souterraines: Cas d’El Jadida, Maroc; Secrétariat International Francophone pour L’évolution Environnementale: Quebec City, QC, Canada, 2006; pp. 12–16. [Google Scholar]
- Mejbri, R.; Matejka, G.; Lafrance, P.; et Mazet, M. Fractionnement et caractérisation de la matière organique des lixiviats de décharges d’ordures ménagères. Rev. Des Sci. De L’eau/J. Water Sci. 1995, 8, 217–236. [Google Scholar] [CrossRef] [Green Version]
- Assouguem, A.; Farah, A.; Ullah, R.; Korkmaz, Y.B.; Almeer, R.; Sayed, A.A.; Najda, A.; Lazraq, A. Evaluation of the Varietal Impact of Two Citrus Species on Fluctuations of Tetranychus urticae (Acari: Tetranychidae) and Beneficial Phytoseiid Mites. Sustainability 2022, 14, 3088. [Google Scholar] [CrossRef]
- Naifar, E.M. La Situation Actuelle de L’élimination des Ordures Ménagères de L’agglomération de Rabat. Etude de L’impact des Rejets Liquides de la Décharge d’Akrach sur L’environnement. Ph.D. Thesis, Faculté des sciences, Université Mohammed V, Rabat, Morocco, 1996. [Google Scholar]
- Amhoud, S. Apports de la Géologie et de L’hydrogéologie à L’étude de L’impact de la Décharge D’oued Akrech sur les Ressources en Eau. Ph.D. Thesis, Mohammed V University, Rabat, Morocco, 1997. 3éme cycle. [Google Scholar]
- Jensen, M.P.; Turner, J.A.; Romano, J.M.; Fisher, L.D. Comparative reliability and validity of chronic pain intensity measures. Pain 1999, 83, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Baun, A.; Jensen, S.D.; Bjerg, L.; Christensen, T.H.; Nyholm, N. Toxicity of organic chemical pollution in groundwater down gradient of a landfill (Grindsted, Denmark). Environ. Technol. 2000, 34, 1647–1652. [Google Scholar] [CrossRef]
- Chofqi, A. Mise en Evidence des Mecanismes de Contamination des Eaux Souterraines Par Les Lixiviats D’une Decharge Incontrolee (El Jadida, Maroc): Geologie, Hydrogeologie, Geo-Electrique, Geochimie et Epidemiologie. Ph.D. Thesis, Université d’El Jadida, El Jadida, Morocco, 2004. [Google Scholar]
- Bennama, T.; Younsi, A.; Zoubir, D.; Debab, A. Caractérisation et traitement physico-chimique des lixiviats de la décharge publique d’El-Kerma (Algérie) par adsorption en discontinu sur de la sciure de bois naturelle et activée chimiquement. Water Qual. Res. J. Can. 2010, 45, 81–90. [Google Scholar] [CrossRef]
- Kerbachi, R.; Belkacemi, M. Caractérisation et évolution des lixiviats de la décharge de Oued-Smar à Alger. T.S.M.-L’eau 1994, 11, 615–618. [Google Scholar]
- Rodier, J.; Legube, B.; Merlet, N. Et coll. L’analyse de L’eau; Eaux Naturelles; Eaux. 2009. Edition 9, Dunod, Paris, France. Available online: http://excerpts.numilog.com/books/9782100754120.pdf (accessed on 23 January 2023).
- OFEFP. Directives Pour le Prélèvement D’échantillons de Sols et Analyses de Substances Polluantes; OCFIM: Berne, Switzerland, 1989; 24p. [Google Scholar]
- Baize, D. Teneurs Totales en Elements Traces Metalliques Dans Les Sols (France); INRA: Paris, France, 1997; 408p. [Google Scholar]
- Association Française, de Normalisation. Documentation: Recueil de Normes Françaises, 3rd ed.; Bulletin des Bibliothèques de France (BBF): Paris, France, 1987; pp. 386–388. [Google Scholar]
- Mathieu, C.; Pieltain, F. Analyse Chimique des Sols: Méthode Choisies; Editions Tecet Doc; Lavoisier: Paris, France, 2003; 214p. [Google Scholar]
- AFNOR. Qualité des sols—Recueil des Normes; Agence Française de Normalisation: Saint-Denis, France, 1994. [Google Scholar]
- Hoening, M.; Dupire, S.; Wollast, R. L’atomisation Electrothermique en Spectrométrie D’absorption Atomique et Son Application Dans les Etudes de L’environnement; Tec et Doc: Paris, France, 1979. [Google Scholar]
- Tomlinson, D.L.; Wilson, J.G.; Harris, C.R.; Jeffrey, D.W. Problems in the assessments of heavy metal levels in estuaries and formation of a pollution index. Helgoländer Meeresunters. 1980, 33, 566–575. [Google Scholar] [CrossRef] [Green Version]
- Navarro, A.; Bernard, D.; Millot, N. Les problèmes de pollution par les lixiviats de décharge. Tech. Sci. Et Méthodes-L’Eau 1988, 3, 541–545. [Google Scholar]
- Leclerc, G.; Bonneau, A. La lixiviation: Source, composition et atténuation. Eau Du Québec 1989, 15, 37–45. [Google Scholar]
- Matejka, G.; Rinke, M.; Mejbri, R.; Bril, H. Pollution engendrée par une décharge d’ordures ménagères: Bilan hydrique et caractérisation. Environ. Technol. 1994, 15, 313–322. [Google Scholar] [CrossRef]
- Keenan, J.D.; Steiner, R.L.; Fungaroli, A.A. Chemical-Physical Leachate Treatment. J. Environ. Eng. 1983, 109, 1371–1384. [Google Scholar] [CrossRef]
- Parveaud, M. Le traitement des lixiviats par osmose inverse. L’eau L’industrie Les Nuis. 1993, 162, 48–50. [Google Scholar]
- Renou, S.; Poulain, S.; Gagnaire, J.; Cadarache, D.; Marrot, B.; Moulin, P. Lixiviat de centre de stockage: Déchet généré par des déchets. L’eau L’industrie Les Nuis. 2008, 310, 37–43. [Google Scholar]
- Tchobanoglous, G.; Theisen, H.; Vigil, S. Integrated Solid Waste Management; McGraw-Hill International Edition: New York, NY, USA, 1993; p. 978. [Google Scholar]
- Kjeldsen, P.; Barlaz, M.A.; Rooker, A.P.; Baum, A.; Ledin, A.; Christensen, T.A. present and long-term composition of MSW landfill leachate. Crit. Rev. Environ. Sci. Technol. 2002, 32, 297–336. [Google Scholar] [CrossRef]
- Chian, E. Stability of organic matter in landfill leachates. Water Ressour. 1977, 11, 225–232. [Google Scholar] [CrossRef]
- Amokrane, A. Epuration des Lixiviats de Décharges. Prétraitement par Coagulation-Fl Oculation. Traitement par Osmose Inverse. Post-Traitement par Incinération. Ph.D. Thesis, INSA de Lyon, Villeurbanne, France, 1994; 286p. [Google Scholar]
- Vilomet, J.D. Evaluation du Risque lié à une Décharge D’ordures Ménagères: Suivi de la Qualité d’un Aquifère au Moyen des Isotopes Stables du Plomb et du Strontium. Corrélation Avec des Polluants Spécifiques des Lixiviats. Ph.D. Thesis, Université d’Aix-Marseille III, Marseille, France, 2000; 187p. [Google Scholar]
- Hakou, R.; Wahbi, M.; Bachnou, M.; Elamari, K.; Hanich, L.; Hibti, M. Impact de la décharge publique de Marrakech (Maroc) sur les ressources en eau. Bull. Eng. Geol. Environ. 2001, 60, 325–336. [Google Scholar] [CrossRef]
- Kurniawan, T.A.; Lo, W.; Chan, G.Y.S. Physico-Chemical Treatments for Removal of Recalcitrant Contaminants from Landfill Leachate. J. Hazard. Mater. 2006, 129, 80–100. [Google Scholar] [CrossRef]
- Thomas, H.C.; Peter, K.; Poul, L.B.; Dorthe, L.J.; Jette, B.C.; Anders, B.; Hans-Jørgen, A.; Gorm, H. Biogeochemistry of landfill leachate plumes. Appl. Geochem. 2001, 16, 659–718. [Google Scholar]
- Aluko, O.; Sridhar, M.K.C.; Oluwande, P.A. Characterization of leachates from a municipal solid waste landfi ll site in Ibadan, Nigeria. J. Environ. Health Res. 2003, 2, 32–37. [Google Scholar]
- El M’ssari, N. Elaboration d’une Méthodologie de Caractérisation des Ordures Ménagères et du Compost de L’usine de Salé. Ph.D. Thesis, Faculté des Sciences, Université Moulay Ismail, Meknès, Morocco, 1993. [Google Scholar]
- Hakanson, L. An ecological risk index for aquatic pollution control.a sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Rubio, B.; Nombela, M.A.; Vilas, F.J.M.P.B. Geochemistry of major and trace elements in sediments of the Ria de Vigo (NW Spain): An assessment of metal pollution. Mar. Pollut. Bull. 2000, 40, 968–980. [Google Scholar] [CrossRef]
- Derwichi, E.; Benziane, Z.; Benaabidate, L.; Lahrach, A.; Boushaba, A. Chrome, Plomb,Zinc, Cuivre et Nickel dans le bassin versant du Sebou: Sources d’apport et impact sur la qualité des eaux de surface. Notes Mém. Serv. Géol. Maroc. 2012, 575, 84–87. [Google Scholar]
- Perrin, J.L.; Raïs, N.; Chahinian, N.; Moulin, P.; Ijjaali, M. Water quality assessment of highly polluted rivers in a semi-arid Mediterranean zone Oued Fez and Sebou River (Morocco). J. Hydrol. 2014, 510, 26–34. [Google Scholar] [CrossRef]
- El Hajjami, S.; Abriak, N.; Souabi, S.; El Alami, M. Study of metallic contamination of Oued Sebou sediments, Morocco. Environ. Technol. Innov. 2021, 23, 101680. [Google Scholar] [CrossRef]
- Amri, N.; Benslimane, M.; Zaoui, H.; Hamedoun, M.; Outiti, B. Evaluation of the heavy metals accumulate in samples of the sediments, soils and plants by ICP-OES with the average sebou. Moroc. J. Condens. Matter 2007, 8, 43–52. [Google Scholar]











| Range Value | Average ± SD Value | General Limit Values for Discharges to Surface Water and Groundwater | |
|---|---|---|---|
| pH | 6.91–8 | 7.7 ± 0.3 | 5.5–9.5 |
| Temperature | 19–23 | 21 ± 1.60 | - |
| Electrical conductivity (ms/cm) | 24–105.1 | 64.55 ± 33.15 | 2.7 |
| Suspended solids (TSS) (mg/L) | 314–1022 | 740 ± 234 | 100 |
| Chemical Oxygen Demand (COD) (mg d’O2/L) | 430.7–7962 | 5540.35 ± 1872.67 | 500 |
| BOD5 (mg d’O2/L) | 274–530.7 | 370.7 ± 115.4 | 100 |
| DBO5/COD (mg d’O2/L) | 0.058–0.096 | 0.084 ± 0.012 | - |
| NH4 (mg/L) | 50–135 | 79.4 ± 16.78 | - |
| NO3− (mg/L) | 13–46 | 29.78 ± 10.89 | - |
| NO2− (mg/L) | 0.376–3.01 | 1.78 ± 0.45 | - |
| PO4 (mg/L) | 5–12 | 8.51 ± 1.34 | 15 |
| SO4 (mg/L) | 1036.07–4567 | 2322.14 ± 1117.07 | 600 |
| Total Kjeldahl Nitrogen (TKN) (mg/L) | 1440–1955 | 1514 ± 98.70 | - |
| As | Cr | Cu | Ni | Zn | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range Value | Average ± SD | Range Value | Average ± SD | Range Value | Average ± SD | Range Value | Average ± SD | Range Value | Average ± SD | |
| BJ | 0.14–0.21 | 0.18 ± 0.03 | 4.07–5.78 | 4.9 ± 0.60 | 0.02–0.15 | 0.07 ± 0.06 | 0.35–0.64 | 0.480 ± 0.10 | 1.73–2.48 | 2.160 ± 0.31 |
| B1 | 0.09–0.23 | 0.18 ± 0.05 | 0.87–1.97 | 1.68 ± 0.40 | 0.03–0.12 | 0.07 ± 0.03 | 0.3–0.6 | 0.532 ± 0.12 | 0.24–0.72 | 0.492 ± 0.16 |
| B2 | 0.1–0.22 | 0.19 ± 0.04 | 0.77–1.69 | 1.4 ± 0.33 | 0.05–0.06 | 0.05 ± 0.01 | 0.50–0.6 | 0.498 ± 0.12 | 0.29–0.41 | 0.315 ± 0.09 |
| BCr | 0.44- 0.56 | 0.49 ± 0.04 | 83.13–93.63 | 88.29 ± 3.51 | 0.01 | 0.01 ± 0.00 | 0.11–0.13 | 0.123 ± 0.01 | 0.10–0.11 | 0.108 ± 0.00 |
| CFi | PLI | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cd | Cr | Cu | Ni | Pb | Zn | Fe | |||
| Surface (0 cm) | S1 | 1.29 | 2.07 | 6.28 | 9.53 | 1.31 | 4.4 | 0.97 | 2.64 |
| S2 | 1.27 | 1.85 | 6.46 | 9.74 | 1.27 | 4.7 | 0.99 | 2.63 | |
| S3 | 1.28 | 1.59 | 6.79 | 9.8 | 1.43 | 4.95 | 1.04 | 2.68 | |
| S4 | 1.29 | 1.66 | 7.01 | 9.7 | 1.51 | 5 | 1.02 | 2.73 | |
| S5 | 1.29 | 1.88 | 6.59 | 9.38 | 1.69 | 4.57 | 1.04 | 2.76 | |
| S6 | 1.28 | 1.89 | 6.39 | 9.27 | 1.58 | 4.51 | 1 | 2.69 | |
| S7 | 1.29 | 1.83 | 6.31 | 9.16 | 1.61 | 4.77 | 0.98 | 2.69 | |
| S8 | 1.29 | 1.86 | 6.17 | 9.01 | 1.55 | 4.56 | 0.92 | 2.63 | |
| S9 | 1.31 | 1.84 | 6.72 | 9.2 | 1.59 | 4.61 | 0.91 | 2.68 | |
| S10 | 1.31 | 1.91 | 6.68 | 9.36 | 2.47 | 4.55 | 0.98 | 2.9 | |
| 20 cm | S1′ | 1.31 | 1.82 | 6.47 | 9.44 | 3.08 | 4.34 | 0.92 | 2.92 |
| S2′ | 1.32 | 1.61 | 6.41 | 9.19 | 1.89 | 4.41 | 0.96 | 2.68 | |
| S3′ | 1.28 | 1.52 | 7.02 | 9.57 | 1.29 | 5.54 | 1.05 | 2.67 | |
| S4′ | 1.26 | 1.6 | 7.11 | 9.76 | 2.17 | 5.63 | 1.02 | 2.91 | |
| S5′ | 1.27 | 1.96 | 6.4 | 9.35 | 2.13 | 4.69 | 1.03 | 2.85 | |
| S6′ | 1.27 | 1.91 | 6.42 | 9.32 | 1.99 | 4.78 | 1 | 2.81 | |
| S7′ | 1.28 | 1.73 | 6.11 | 9.03 | 3.13 | 4.59 | 0.97 | 2.89 | |
| S8′ | 1.3 | 1.82 | 6.14 | 9.16 | 1.73 | 4.55 | 0.9 | 2.66 | |
| S9′ | 1.31 | 1.87 | 6.44 | 9.44 | 1.38 | 4.77 | 0.88 | 2.62 | |
| S10′ | 1.33 | 1.89 | 6.11 | 9.69 | 1.43 | 4.81 | 0.96 | 2.42 | |
| Cd | Cr | Cu | Ni | Pb | Zn | Fe | CaCO3% | Al | K | Mg | Mn | Na | Si | pH | CE | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cd | 0.92 | 0.96 | 0.97 | 0.52 | 0.94 | 0.21 | −0.16 | 0.29 | 0.45 | 0.82 | 0.78 | 0.38 | 0.94 | −0.60 | 0.39 | |
| Cr | 0.92 | 0.88 | 0.91 | 0.48 | 0.85 | 0.17 | −0.23 | 0.40 | 0.48 | 0.82 | 0.77 | 0.42 | 0.93 | −0.50 | 0.33 | |
| Cu | 0.96 | 0.88 | 0.99 | 0.51 | 0.99 | 0.37 | −0.05 | 0.30 | 0.48 | 0.80 | 0.81 | 0.36 | 0.96 | −0.58 | 0.36 | |
| Ni | 0.97 | 0.91 | 0.99 | 0.51 | 0.98 | 0.35 | −0.09 | 0.28 | 0.48 | 0.83 | 0.82 | 0.39 | 0.96 | −0.58 | 0.38 | |
| Pb | 0.52 | 0.48 | 0.51 | 0.51 | 0.47 | 0.09 | 0.07 | 0.52 | 0.46 | 0.63 | 0.56 | 0.42 | 0.58 | −0.50 | 0.35 | |
| Zn | 0.94 | 0.85 | 0.99 | 0.98 | 0.47 | 0.41 | −0.06 | 0.24 | 0.46 | 0.76 | 0.82 | 0.30 | 0.94 | −0.53 | 0.30 | |
| Fe | 0.21 | 0.17 | 0.37 | 0.35 | 0.09 | 0.41 | 0.40 | −0.05 | −0.22 | 0.05 | 0.31 | 0.14 | 0.38 | −0.16 | 0.12 | |
| CaCO3% | −0.16 | −0.23 | −0.05 | −0.09 | 0.07 | −0.06 | 0.40 | 0.24 | 0.03 | −0.18 | −0.02 | −0.21 | −0.14 | 0.09 | 0.22 | |
| Al | 0.29 | 0.40 | 0.30 | 0.28 | 0.52 | 0.24 | −0.05 | 0.24 | 0.73 | 0.31 | 0.54 | −0.02 | 0.39 | −0.17 | 0.14 | |
| K | 0.45 | 0.48 | 0.48 | 0.48 | 0.46 | 0.46 | −0.22 | 0.03 | 0.73 | 0.51 | 0.70 | 0.05 | 0.44 | −0.07 | 0.05 | |
| Mg | 0.82 | 0.82 | 0.80 | 0.83 | 0.63 | 0.76 | 0.05 | −0.18 | 0.31 | 0.51 | 0.67 | 0.53 | 0.80 | −0.66 | 0.49 | |
| Mn | 0.78 | 0.77 | 0.81 | 0.82 | 0.56 | 0.82 | 0.31 | −0.02 | 0.54 | 0.70 | 0.67 | 0.23 | 0.80 | −0.32 | 0.21 | |
| Na | 0.38 | 0.42 | 0.36 | 0.39 | 0.42 | 0.30 | 0.14 | −0.21 | −0.02 | 0.05 | 0.53 | 0.23 | 0.39 | −0.56 | 0.60 | |
| Si | 0.94 | 0.93 | 0.96 | 0.96 | 0.58 | 0.94 | 0.38 | −0.14 | 0.39 | 0.44 | 0.80 | 0.80 | 0.39 | −0.57 | 0.34 | |
| pH | −0.60 | −0.50 | −0.58 | −0.58 | −0.50 | −0.53 | −0.16 | 0.09 | −0.17 | −0.07 | −0.66 | −0.32 | −0.56 | −0.57 | −0.67 | |
| CE | 0.39 | 0.33 | 0.36 | 0.38 | 0.35 | 0.30 | 0.12 | 0.22 | 0.14 | 0.05 | 0.49 | 0.21 | 0.60 | 0.34 | −0.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahouach, Y.; Baali, A.; Boushaba, A.; Hakam, O.; Azennoud, K.; Lyazidi, A.; Benmessaoud, S.; Assouguem, A.; Kara, M.; Alsaigh, M.A.; et al. Impact of the Controlled Dump of Fez City (Morocco): Evaluation of Metallic Trace Elements Contamination in the Sediments. Water 2023, 15, 1209. https://doi.org/10.3390/w15061209
Ahouach Y, Baali A, Boushaba A, Hakam O, Azennoud K, Lyazidi A, Benmessaoud S, Assouguem A, Kara M, Alsaigh MA, et al. Impact of the Controlled Dump of Fez City (Morocco): Evaluation of Metallic Trace Elements Contamination in the Sediments. Water. 2023; 15(6):1209. https://doi.org/10.3390/w15061209
Chicago/Turabian StyleAhouach, Youssra, Abdennasser Baali, Abdellah Boushaba, Oualid Hakam, Khalil Azennoud, Aziza Lyazidi, Safaa Benmessaoud, Amine Assouguem, Mohammed Kara, Mona Abdullah Alsaigh, and et al. 2023. "Impact of the Controlled Dump of Fez City (Morocco): Evaluation of Metallic Trace Elements Contamination in the Sediments" Water 15, no. 6: 1209. https://doi.org/10.3390/w15061209









