Ammonia Influences the Zooplankton Assemblage and Beta Diversity Patterns in Complicated Urban River Ecosystems
Abstract
1. Introduction
2. Materials
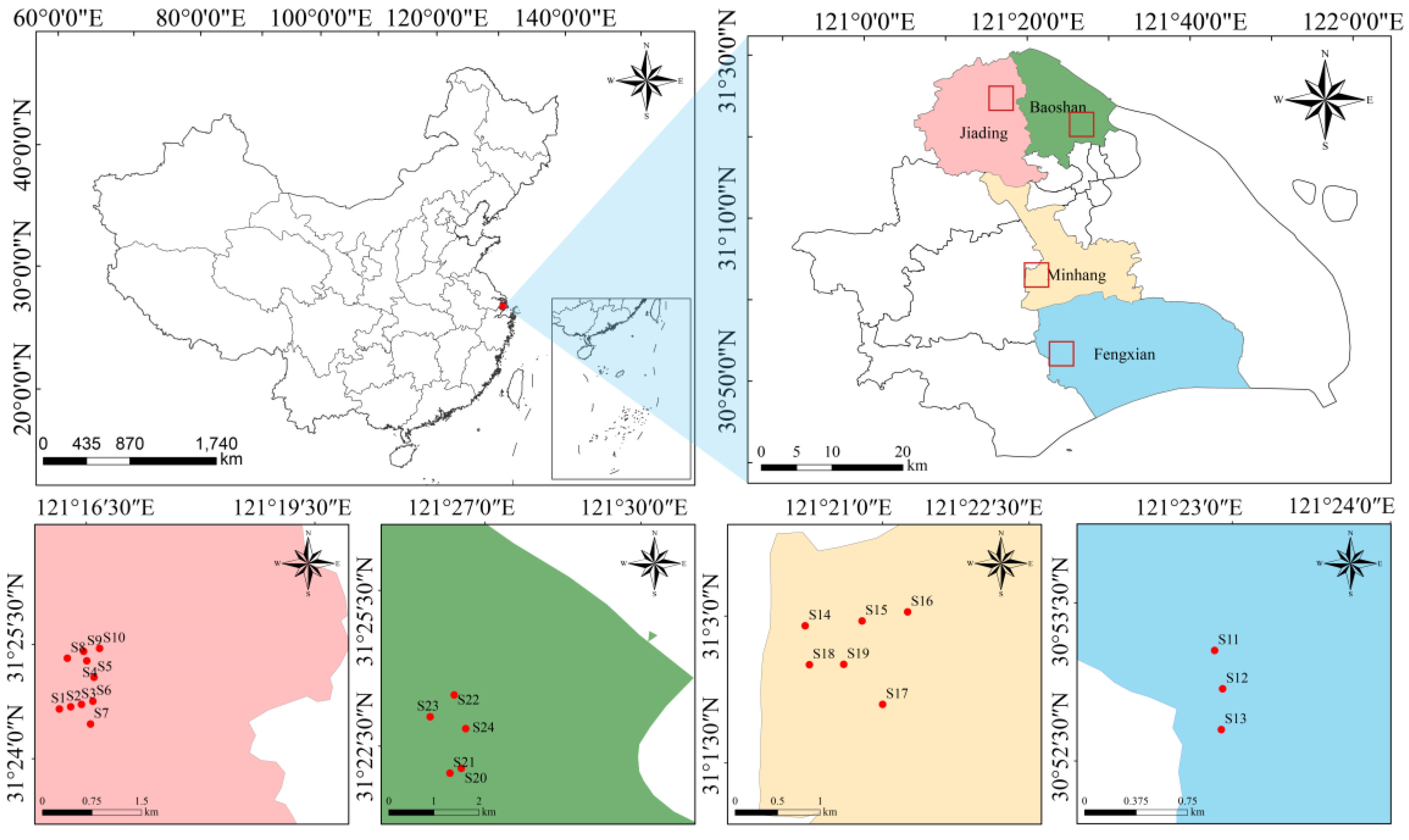
2.1. Study Region
2.2. Sample Collection and Laboratory Analyses
2.3. Experimental Design
2.4. Analytical Methods
3. Results
3.1. Zooplankton Assemblages in Urban Rivers of Shanghai City
3.2. Environmental Variables
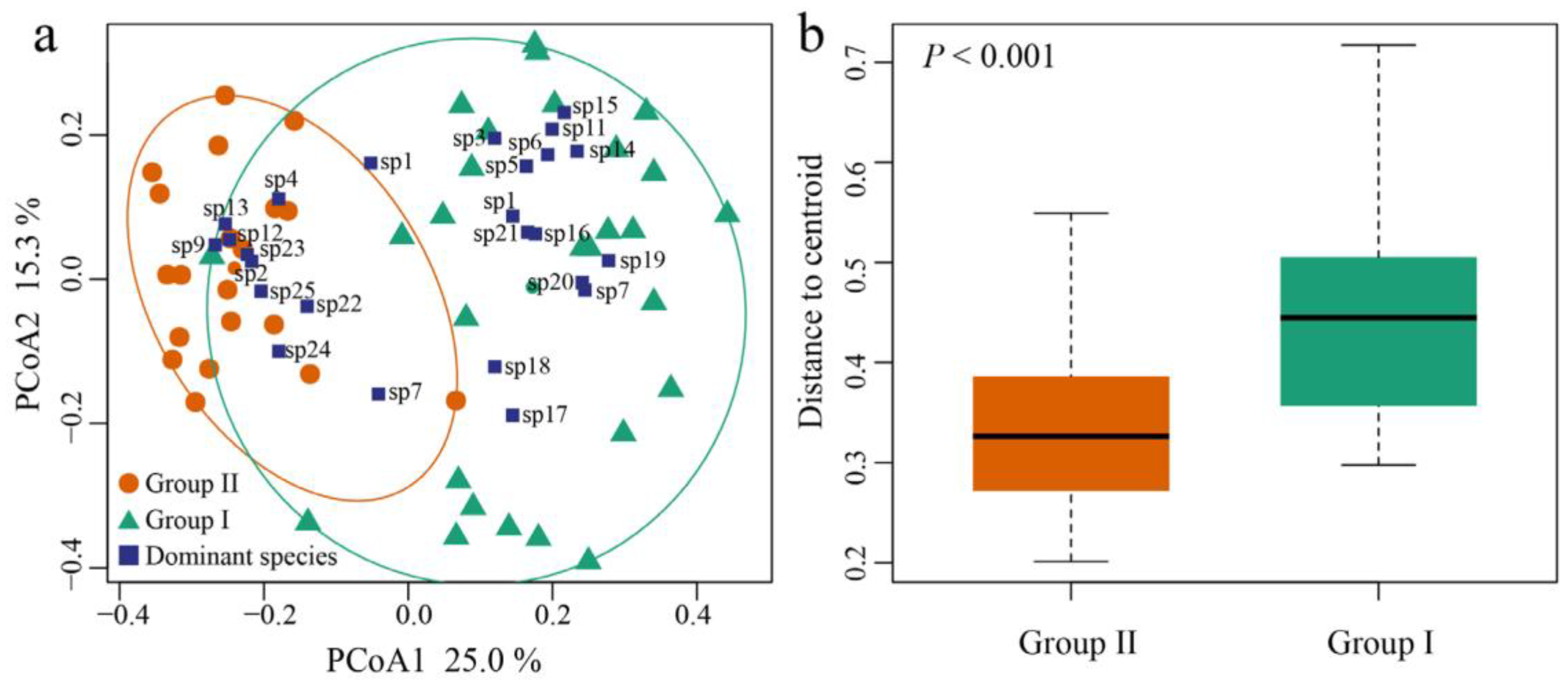
3.3. Beta Diversity of Zooplankton Assemblages
3.4. Environmental Factor Variations in the Nutrient Addition Experiment
3.5. Effect of Ammonia Nitrogen on the Structure and Beta Diversity of Zooplankton
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hooper, D.U.; Adair, E.C.; Cardinale, B.J.; Byrnes, J.; Hungate, B.A.; Matulich, K.L.; Gonzalez, A.; Duffy, J.E.; Gamfeldt, L.; O’Connor, M. A global synthesis reveals biodiversity loss as a major driver of ecosystem change. Nature 2012, 486, 105–108. [Google Scholar] [CrossRef]
- Baselga, A. Partitioning the turnover and nestedness components of beta diversity. Glob. Ecol. Biogeogr. 2010, 19, 134–143. [Google Scholar] [CrossRef]
- Buckley, L.B.; Jetz, W. Linking global turnover of species and environments. Proc. Natl. Acad. Sci. USA 2008, 105, 17836–17841. [Google Scholar] [CrossRef]
- Anderson, M.J.; Crist, T.O.; Chase, J.M.; Vellend, M.; Inouye, B.D.; Freestone, A.L.; Sanders, N.J.; Cornell, H.V.; Comita, L.S.; Davies, K.F.; et al. Navigating the multiple meanings of β diversity: A roadmap for the practicing ecologist. Ecol. Lett. 2011, 14, 19–28. [Google Scholar] [CrossRef]
- Mcknight, M.W.; White, P.S.; Mcdonald, R.I.; Lamoreux, J.F.; Stuart, S.N. Putting Beta-Diversity on the Map: Broad-Scale Congruence and Coincidence in the Extremes. PLoS Biol. 2007, 5, 2424–2432. [Google Scholar] [CrossRef]
- Koleff, P.; Lennon, J.J.; Gaston, K.J. Are there latitudinal gradients in species turnover? Glob. Ecol. Biogeogr. 2003, 12, 483–498. [Google Scholar] [CrossRef]
- Stegen, J.C.; Freestone, A.L.; Crist, T.O.; Anderson, M.J.; Vellend, M. Stochastic and deterministic drivers of spatial and temporal turnover in breeding bird communities. Glob. Ecol. Biogeogr. 2013, 22, 202–212. [Google Scholar] [CrossRef]
- Kraft, N.J.B.; Comita, L.S.; Chase, J.M.; Sanders, N.J.; Swenson, N.G.; Crist, T.O.; Stegen, J.C.; Vellend, M.; Boyle, B.; Anderson, M.J.; et al. Disentangling the Drivers of β Diversity Along Latitudinal and Elevational Gradients. Science 2011, 333, 1755–1758. [Google Scholar] [CrossRef]
- Baselga, A.; Gómez-Rodríguez, C.; Lobo, J.M. Historical Legacies in World Amphibian Diversity Revealed by the Turnover and Nestedness Components of Beta Diversity. PLoS ONE 2012, 7, e32341. [Google Scholar] [CrossRef]
- Carvalho, J.C.; Cardoso, P.; Gomes, P. Determining the relative roles of species replacement and species richness differences in generating beta-diversity patterns. Glob. Ecol. Biogeogr. 2012, 21, 760–771. [Google Scholar] [CrossRef]
- Wen, Z.Q.; Quan, Q.; Xia, L.; Ge, D.; Lv, X. Multiscale partitioning of small mammal β-diversity provides novel insights into the Quaternary faunal history of Qinghai–Tibetan Plateau and Hengduan Mountains. J. Biogeogr. 2016, 43, 1412–1424. [Google Scholar] [CrossRef]
- Heino, J.; Alahuhta, J.; Fattorini, S.; Schmera, D. Predicting beta diversity of terrestrial and aquatic beetles using ecogeographical variables: Insights from the replacement and richness difference components. J. Biogeogr. 2019, 46, 304–315. [Google Scholar] [CrossRef]
- Legendre, P. Interpreting the replacement and richness difference components of beta diversity. Glob. Ecol. Biogeogr. 2014, 23, 1324–1334. [Google Scholar] [CrossRef]
- Loick-Wilde, N.; Weber, S.C.; Conroy, B.J.; Capone, D.G.; Coles, V.J.; Medeiros, P.M.; Steinberg, D.K.; Montoya, J.P. Nitrogen sources and net growth efficiency of zooplankton in three Amazon River plume food webs. Limnol. Oceanogr. 2016, 61, 460–481. [Google Scholar] [CrossRef]
- Jagadeesan, L.; Jyothi Ba Bu, R.; Arunpandi, N.; Parthasarathi, S. Copepod grazing and their impact on phytoplankton standing stock and production in a tropical coastal water during the different seasons. Environ. Monit. Assess. 2017, 189, 105. [Google Scholar] [CrossRef]
- Simoes, N.R.; Dias, J.D.; Leal, C.M.; Braghin, L.D.S.M.; Lansac-Toha, F.A.; Bonecker, C.C. Floods control the influence of environmental gradients on the diversity of zooplankton communities in a neotropical floodplain. Aquat. Sci. 2013, 75, 607–617. [Google Scholar] [CrossRef]
- de Souza, C.A.; Beisner, B.E.; Velho, L.F.M.; de Carvalho, P.; Pineda, A.; Vieira, L.C.G. Impoundment, environmental variables and temporal scale predict zooplankton beta diversity patterns in an Amazonian river basin. Sci. Total Environ. 2021, 776, 145948. [Google Scholar] [CrossRef]
- Jeppesen, E.; Noges, P.; Davidson, T.A.; Haberman, J.; Noges, T.; Blank, K.; Lauridsen, T.L.; Sondergaard, M.; Sayer, C.; Laugaste, R.; et al. Zooplankton as indicators in lakes: A scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- de Melo, T.X.; Dias, J.D.; Simões, N.R.; Bonecker, C.C. Effects of nutrient enrichment on primary and secondary productivity in a subtropical floodplain system: An experimental approach. Hydrobiologia 2019, 827, 171–181. [Google Scholar] [CrossRef]
- Tavares, D.A.; Lambrecht, R.W.; de Almeida Castilho, M.C.; Henry, R.; Ferragut, C. Epipelon responses to N and P enrichment and the relationships with phytoplankton and zooplankton in a mesotrophic reservoir. Aquat. Ecol. 2019, 53, 303–314. [Google Scholar] [CrossRef]
- Santos, S.A.M.; dos Santos, T.R.; Furtado, M.S.R.; Henry, R.; Ferragut, C. Periphyton nutrient content, biomass and algal community on artificial substrate: Response to experimental nutrient enrichment and the effect of its interruption in a tropical reservoir. Limnology 2018, 19, 209–218. [Google Scholar] [CrossRef]
- Melo, A.S.; Schneck, F.; Hepp, L.U.; Simões, N.R.; Siqueira, T.; Bini, L.M. Focusing on variation: Methods and applications of the concept of beta diversity in aquatic ecosystems. Acta Limnol. Bras. 2011, 23, 318–331. [Google Scholar] [CrossRef]
- da Silva, N.J.; Lansac-Tôha, F.M.; Lansac-Tôha, F.A.; Sales, P.C.L.; de Ribamar Sousa Rocha, J. Beta diversity patterns in zooplankton assemblages from a semiarid river ecosystem. Int. Rev. Hydrobiol. 2021, 106, 29–40. [Google Scholar] [CrossRef]
- Bozelli, R.L.; Thomaz, S.M.; Padial, A.A.; Lopes, P.M.; Bini, L.M. Floods decrease zooplankton beta diversity and environmental heterogeneity in an Amazonian floodplain system. Hydrobiologia 2015, 753, 233–241. [Google Scholar] [CrossRef]
- Braghin, L.; Almeida, B.D.C.; Canella, T.F.; Garcia, B.; Bonecker, C.C. Effects of dams decrease zooplankton functional β-diversity in river-associated lakes. Freshw. Biol. 2018, 63, 721–730. [Google Scholar] [CrossRef]
- Tran Khac, V.; Hong, Y.; Plec, D.; Lemaire, B.J.; Dubois, P.; Saad, M.; Vinçon-Leite, B. An Automatic Monitoring System for High-Frequency Measuring and Real-Time Management of Cyanobacterial Blooms in Urban Water Bodies. Processes 2018, 6, 11. [Google Scholar] [CrossRef]
- Suthar, S.; Sharma, J.; Chabukdhara, M.; Nema, A.K. Water quality assessment of river Hindon at Ghaziabad, India: Impact of industrial and urban wastewater. Environ. Monit. Assess. 2009, 165, 103–112. [Google Scholar] [CrossRef]
- Pan, M.; Zhao, J.; Zhen, S.; Heng, S.; Wu, J. Effects of the combination of aeration and biofilm technology on transformation of nitrogen in black-odor river. Water Sci. Technol. 2016, 74, 655–662. [Google Scholar] [CrossRef]
- Xia, Y.; Wang, H.; He, X.; Yuan, W.; Zeng, Y.; Yan, H.; Zhang, X.; Tu, Y. Spatiotemporal heterogeneity of hydrodynamic forces and water quality in typical lakeside river networks in Taihu Basin. Lake Sci. 2021, 33, 1100–1111. [Google Scholar]
- Gao, Q.; He, G.; Fang, H.; Bai, S.; Huang, L. Numerical simulation of water age and its potential effects on the water quality in Xiangxi Bay of Three Gorges Reservoir. J. Hydrol. 2018, 566, 484–499. [Google Scholar] [CrossRef]
- He, X.; Wang, H.; Yan, H.; Ao, Y. Numerical simulation of microcystin distribution in Liangxi River, downstream of Taihu Lake. Water Environ. Res. 2021, 93, 1934–1943. [Google Scholar] [CrossRef] [PubMed]
- Clesceri, L.S.; Greenberg, A.E.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Zhang, Z.; Huang, X. Research Methods on Plankton in Freshwater; Science Press: Beijing, China, 1991. (In Chinese) [Google Scholar]
- Chiang, S.C.; Du, N. Fauna Sinica Crustacea: Freshwater Cladocera; Science Press: Beijing, China, 1979. (In Chinese) [Google Scholar]
- Li, H.; Wang, C.; Huang, X.; Hug, A. Spatial Assessment of Water Quality with Urbanization in 2007–2015, Shanghai, China. Remote Sens. 2018, 10, 1024. [Google Scholar] [CrossRef]
- De’ath, G. Multivariate Regression Trees: A New Technique for Modeling Species-Environment Relationships. Ecology 2002, 83, 1105–1117. [Google Scholar]
- De’ath, G. MVPART: Multivariate Partitioning. R Package Version 1.6–2. 2014. Available online: https://mran.microsoft.com/snapshot/2014-12-11/web/packages/mvpart/index.html (accessed on 2 April 2023).
- Oksanen, J. Vegan: Ecological Diversity. 2018. Available online: https://cran.r-project.org/web/packages/vegan/vignettes/diversity-vegan.pdf (accessed on 2 April 2023).
- Mcnaughton, S.J. Relationships among Functional Properties of Californian Grassland. Nature 1967, 216, 168–169. [Google Scholar] [CrossRef]
- Podani, J.; Schmera, D. A new conceptual and methodological framework for exploring and explaining pattern in presence-absence data. Oikos 2011, 120, 1625–1638. [Google Scholar] [CrossRef]
- Li, Y.; Xu, E.G.; Liu, W.; Chen, Y.; Liu, H.; Li, D.; Liu, Z.; Giesy, J.P.; Yu, H. Spatial and temporal ecological risk assessment of unionized ammonia nitrogen in Tai Lake, China (2004–2015). Ecotoxicol. Environ. Saf. 2017, 140, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ni, P.; Gao, Y.; Xiong, W.; Zhao, Y.; Zhan, A. Geographical distribution of zooplankton biodiversity in highly polluted running water ecosystems: Validation of fine-scale species sorting hypothesis. Ecol. Evol. 2018, 8, 4830–4840. [Google Scholar] [CrossRef]
- Du, C.; Yang, L.; Zhao, Y.; Wu, X.; Xu, M.; Wang, L.; Zhang, W. Temporal and spatial variation of zooplankton community structure and its relationship with environmental factors in Dianshan lake, shanghai. Environ. Sci. 2019, 40, 4513–4522. (In Chinese) [Google Scholar]
- Yang, J.; Zhang, X.; Xie, Y.; Song, C.; Sun, J.; Zhang, Y.; Giesy, J.P.; Yu, H. Ecogenomics of Zooplankton Community Reveals Ecological Threshold of Ammonia Nitrogen. Environ. Sci. Technol. 2017, 51, 3057–3064. [Google Scholar] [CrossRef]
- Trommer, G.; Poxleitner, M.; Stibor, H. Responses of lake phytoplankton communities to changing inorganic nitrogen supply forms. Aquat. Sci. 2020, 82, 22. [Google Scholar] [CrossRef]
- Li, C.; Feng, W.; Chen, H.; Li, X.; Song, F.; Guo, W.; Giesy, J.P.; Sun, F. Temporal variation in zooplankton and phytoplankton community species composition and the affecting factors in Lake Taihu-a large freshwater lake in China. Environ. Pollut. 2019, 245, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Šorf, M.; Davidson, T.A.; Brucet, S.; Menezes, R.F.; Søndergaard, M.; Lauridsen, T.L.; Landkildehus, F.; Liboriussen, L.; Jeppesen, E. Zooplankton response to climate warming: A mesocosm experiment at contrasting temperatures and nutrient levels. Hydrobiologia 2015, 742, 185–203. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, X.; Shen, G.; Zhu, Y.; Qian, X.; Zhang, X.; Zhang, W.; Hu, S.; Bai, Y. Spatial and Temporal Variation of Phytoplankton Community Structure and Its Influencing Factors in Shanghai River Channels. Environ. Sci. 2020, 41, 3621–3628. (In Chinese) [Google Scholar]
- Pan, Y.; Zhang, Y.; Sun, S. Phytoplankton–zooplankton dynamics vary with nutrients: A microcosm study with the cyanobacterium Coleofasciculus chthonoplastes and cladoceran Moina micrura. J. Plankton Res. 2014, 36, 1323–1332. [Google Scholar] [CrossRef]
- Mamani, A.; Koncurat, M.L.; Boveri, M. Combined effects of fish and macroinvertebrate predation on zooplankton in a littoral mesocosm experiment. Hydrobiologia 2019, 829, 19–29. [Google Scholar] [CrossRef]
- Akhurst, D.J.; Jones, G.B.; Clark, M.; Reichelt-Brushett, A. Effects of fish and macrophytes on phytoplankton and zooplankton community structure in a subtropical freshwater reservoir. Limnologica 2017, 62, 5–18. [Google Scholar] [CrossRef]
- Ersoy, Z.; Brucet, S.; Bartrons, M.; Mehner, T. Short-term fish predation destroys resilience of zooplankton communities and prevents recovery of phytoplankton control by zooplankton grazing. PLoS ONE 2019, 14, e0212351. [Google Scholar] [CrossRef]
- Kolarova, N.; Napiórkowski, P. How Do Specific Environmental Conditions in Canals Affect the Structure and Variability of the Zooplankton Community? Water 2022, 14, 979. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, L.; Riseng, C.; Wehrly, K.; Pan, Y.; Song, K.; Da, L.; Pang, W.; You, Q.; Tian, H. Factors determining zooplankton assemblage difference among a man-made lake, connecting canals, and the water-origin river. Ecol. Indic. 2018, 84, 488–496. [Google Scholar] [CrossRef]
- Pourriot, R.; Rougier, C.; Miquelis, A. Origin and development of river zooplankton: Example of the Marne. Hydrobiologia 1997, 345, 143–148. [Google Scholar] [CrossRef]
- Bum, B.K.; Pick, F.R. Factors regulating phytoplankton and zooplankton biomass in temperate rivers. Limnol. Oceanogr. 1996, 41, 1572–1577. [Google Scholar] [CrossRef]
- Krupa, E.; Romanova, S.; Berkinbaev, G.; Yakovleva, N.; Sadvakasov, E. Zooplankton as Indicator of the Ecological State of Protected Aquatic Ecosystems (Lake Borovoe, Burabay National Nature Park, Northern Kazakhstan). Water 2020, 12, 2580. [Google Scholar] [CrossRef]
- Rosińska, J.; Romanowicz-Brzozowska, W.; Kozak, A.; Gołdyn, R. Zooplankton changes during bottom-up and top-down control due to sustainable restoration in a shallow urban lake. Environ. Sci. Pollut. Res. 2019, 26, 19575–19587. [Google Scholar] [CrossRef] [PubMed]
- Pound, K.L.; Lawrence, G.B.; Passy, S.I. Beta diversity response to stress severity and heterogeneity in sensitive versus tolerant stream diatoms. Divers. Distrib. 2019, 25, 374–384. [Google Scholar] [CrossRef]
- Fu, H.; Yuan, G.; Jeppesen, E.; Ge, D.; Li, W.; Zou, D.; Huang, Z.; Wu, A.; Liu, Q. Local and regional drivers of turnover and nestedness components of species and functional beta diversity in lake macrophyte communities in China. Sci. Total Environ. 2019, 687, 206–217. [Google Scholar] [CrossRef]
- Rocha, M.P.; Bini, L.M.; Domisch, S.; Tolonen, K.T.; Jyrkankallio-Mikkola, J.; Soininen, J.; Hjort, J.; Heino, J. Local environment and space drive multiple facets of stream macroinvertebrate beta diversity. J. Biogeogr. 2018, 45, 2744–2754. [Google Scholar] [CrossRef]
- Pelaez, O.; Pavanelli, C.S. Environmental heterogeneity and dispersal limitation explain different aspects of beta-diversity in Neotropical fish assemblages. Freshw. Biol. 2019, 64, 497–505. [Google Scholar] [CrossRef]
- Picapedra, P.H.S.; Fernandes, C.; Lansac-Toha, F.A. Zooplankton community in the Upper Parnaiba River (Northeastern, Brazil). Braz. J. Biol. 2017, 77, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Gianuca, A.T.; Declerck, S.A.; Lemmens, P.; De Meester, L. Effects of dispersal and environmental heterogeneity on the replacement and nestedness components of β-diversity. Ecology 2017, 98, 525–533. [Google Scholar] [CrossRef]
- Thomaz, S.M.; Bini, L.M.; Bozelli, R.L. Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia 2007, 579, 1–13. [Google Scholar] [CrossRef]
- Zhao, K.; Song, K.; Pan, Y.; Wang, L.; Da, L.; Wang, Q. Metacommunity structure of zooplankton in river networks: Roles of environmental and spatial factors. Ecol. Indic. 2017, 73, 96–104. [Google Scholar] [CrossRef]
- Mimouni, E.; Pinel-Alloul, B.; Beisner, B.E.; Legendre, P. Summer assessment of zooplankton biodiversity and environmental control in urban waterbodies on the Island of Montréal. Ecosphere 2018, 9, e02277. [Google Scholar] [CrossRef]
- Lt, A.; Jh, B.; Baq, A.; Gam, A.; Op, A.; Brm, A.; Lcr, A.; Sj, A.; Lt, A.; Lfmva, C. Differently dispersing organism groups show contrasting beta diversity patterns in a dammed subtropical river basin. Sci. Total Environ. 2019, 691, 1271–1281. [Google Scholar]
- Jamoneau, A.; Passy, S.I.; Soininen, J.; Leboucher, T.; Tison-Rosebery, J. Beta diversity of diatom species and ecological guilds: Response to environmental and spatial mechanisms along the stream watercourse. Freshw. Biol. 2017, 63, 62–73. [Google Scholar] [CrossRef]
- Lopez, E.; Winemiller, K.; Villa-Navarro, F. Local environmental factors influence beta diversity patterns of tropical fish assemblages more than spatial factors. Ecology 2019, 101, e02940. [Google Scholar]
- Gutiérrez-Cánovas, C.; Millán, A.; Velasco, J.; Vaughan, I.P.; Ormerod, S.J. Contrasting effects of natural and anthropogenic stressors on beta diversity in river organisms. Glob. Ecol. Biogeogr. 2013, 22, 796–805. [Google Scholar] [CrossRef]



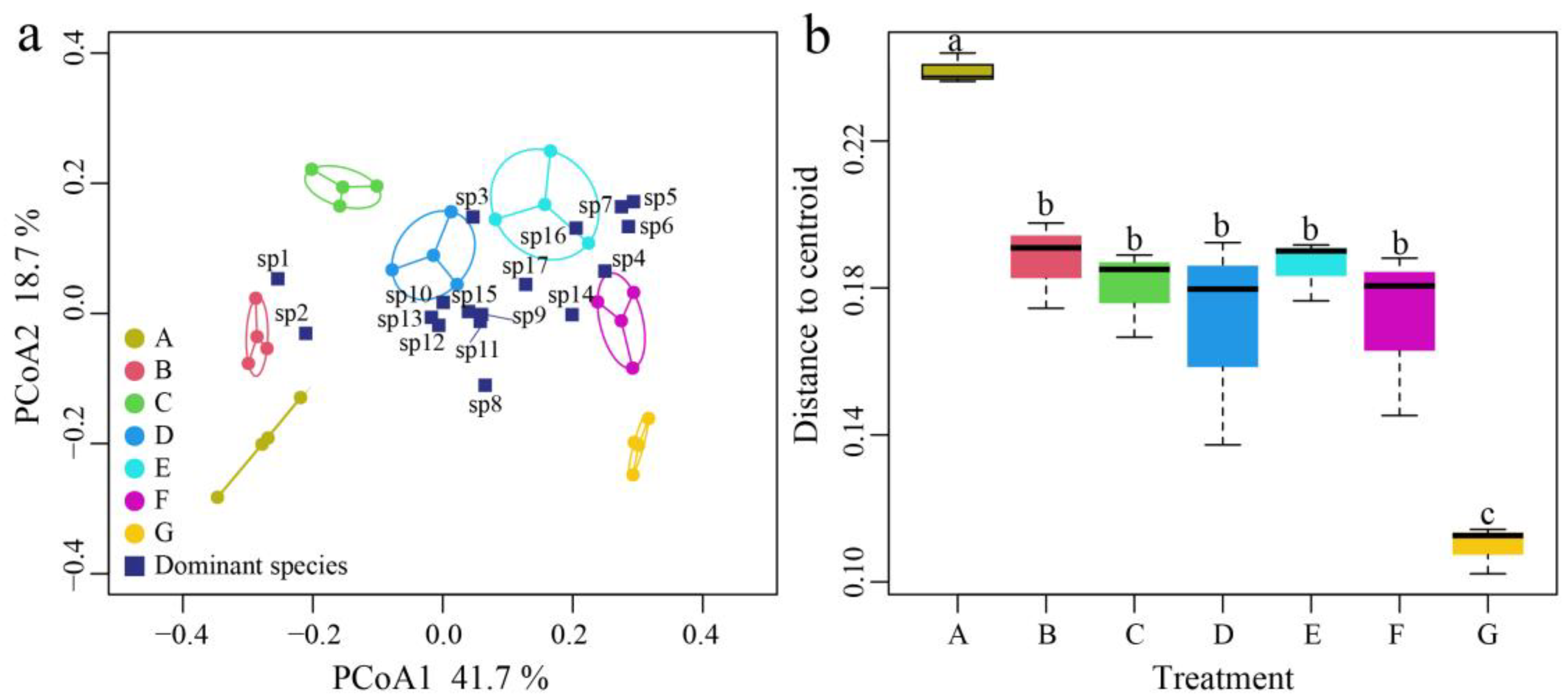
| Treatment Groups | A (Initial Value) | B | C | D | E | F | G |
|---|---|---|---|---|---|---|---|
| NH3-N concentration (mg N·L−1) | 0.2 | 0.6 | 1.0 | 1.8 | 2.6 | 3.4 | 5.0 |
| Beta Diversity | Group I (NH3-N < 1.03 mg·L−1) | Group II (NH3-N ≥ 1.03 mg·L−1) |
|---|---|---|
| Btotal * | 0.56 (0.04) | 0.41 (0.01) |
| Brepl * | 0.37 (0.03) | 0.24 (0.01) |
| Brich | 0.20 (0.01) | 0.16 (0.01) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, C.; Zhao, F.; Shang, G.; Wang, L.; Jeppesen, E.; Zhang, L.; Zhang, W.; Fang, X. Ammonia Influences the Zooplankton Assemblage and Beta Diversity Patterns in Complicated Urban River Ecosystems. Water 2023, 15, 1449. https://doi.org/10.3390/w15081449
Du C, Zhao F, Shang G, Wang L, Jeppesen E, Zhang L, Zhang W, Fang X. Ammonia Influences the Zooplankton Assemblage and Beta Diversity Patterns in Complicated Urban River Ecosystems. Water. 2023; 15(8):1449. https://doi.org/10.3390/w15081449
Chicago/Turabian StyleDu, Caili, Fengbin Zhao, Guangxia Shang, Liqing Wang, Erik Jeppesen, Lieyu Zhang, Wei Zhang, and Xin Fang. 2023. "Ammonia Influences the Zooplankton Assemblage and Beta Diversity Patterns in Complicated Urban River Ecosystems" Water 15, no. 8: 1449. https://doi.org/10.3390/w15081449
APA StyleDu, C., Zhao, F., Shang, G., Wang, L., Jeppesen, E., Zhang, L., Zhang, W., & Fang, X. (2023). Ammonia Influences the Zooplankton Assemblage and Beta Diversity Patterns in Complicated Urban River Ecosystems. Water, 15(8), 1449. https://doi.org/10.3390/w15081449








