1. Introduction
Chromium is a common pollutant in aquatic environments, primarily existing in two stable oxidation states: hexavalent chromium (Cr
6+) and trivalent chromium (Cr
3+) [
1,
2,
3]. Various industrial sectors, from leather production to metal refinement, frequently use compounds containing chromium, resulting in wastewater enriched with this harmful element [
2,
3]. Numerous studies reported the adverse effects of chromium entering the human body through respiratory pathways and the skin, highlighting its potent carcinogenic and mutagenic properties [
1,
3]. Notably, Cr
6+ exhibits significantly higher toxicity levels compared to Cr
3+, with an approximate 100 times increased solubility and tendency to accumulate in organs such as the kidneys, liver, and stomach [
3]. Additionally, exposure to chromium through inhalation, ingestion, dermal absorption, or contact can lead to various health issues, including dermatitis, hypersensitivity reactions, epistaxis, and irritations. Prolonged contact with Cr
6+ has been reported to correlate with weakened immune function, respiratory abnormalities, and an increased risk of lung carcinoma, ultimately resulting in mortality [
4,
5]. These potential threats to the environment and human health necessitate the removal of chromium from contaminated water sources. Resultantly, globally recognized health organizations such as the World Health Organization and US-EPA restrict the acceptable concentration of Cr
6+ in potable water to 0.05 mg/L and 0.1 mg/L, respectively [
3,
6].
Several chemical and physical techniques are available for removing metals, including chromium. These include but are not limited to solvent extraction, ion exchange, electrochemical reduction, chemical precipitation, reverse osmosis, and electrodialysis. However, it is worth noting that many of these methods are associated with significant operational expenses and consequently, they are not extensively utilized in practice [
5,
7,
8]. In contrast, adsorption has gained widespread recognition due to its various attributes, including simplicity, cost-effectiveness, and efficiency [
5,
7,
8,
9]. The utilization of biomaterials with chemical treatment in the process of adsorption presents a viable and efficient option for the removal of chromium owing to its economical operational expenses, effectiveness, and environmentally friendly operation [
5,
10,
11]. To optimize Cr
6+ extraction via biomaterial-based adsorbents, scholars have delved into interpreting the underlying mechanism. They reported the pivotal roles of electrostatic affinity and complexation with activated functional sites [
4,
12]. Nevertheless, numerous techniques encounter economic or technological constraints, such as inadequate elimination of chromium, the complicated configuration of the facility, high operations and maintenance expenses, and a substantial requirement for knowledge [
10,
13]. Certain procedures also produce hazardous secondary waste, such as precipitation, and chemical oxidation that involves the extensive use of acids/bases for pH adjustment and results in environmental contamination [
11,
14].
The utilization of bio-material-based adsorption is prevalent and holds significant potential to effectively eliminate chromium ions from contaminated water [
9,
14,
15,
16]. The bio-materials can be quaternized which involves a particular surface chemical treatment that introduces groups (R-NR’
3+) onto lignocellulosic fibers. Herein, R denotes structures such as cellulose, lignin or hemicellulose, while R’ signifies an alkyl group. This modification can be achieved through epoxy substitution, enhancing the fiber’s tendency for cationic or anionic materials via ion-exchange adsorption mechanisms [
9,
15,
17,
18]. Cellulose, a linear natural polymer, comprises β-D-glucopyranose monomers interconnected covalently through β-1,4-glycocidic bonds. Despite the presence of numerous hydroxyl groups within a cellulose molecule, reactions predominantly occur at the hydroxyl group located in the C-6 structure [
9,
19].
Numerous quaternizing agents are available, each preferred based on the desired reaction mechanism. Notably, N-(3-chloro-2-hydroxypropyl) trimethylammonium chloride, abbreviated as CHMAC and trimethylamine, is frequently employed in research [
9,
19]. Trimethylamine and N, N-dimethylformamide termed DMF, prerequisite pyridine can be utilized as a catalyst. Given the carcinogenic nature and the high cost of these chemicals, CHMAC is often favored. This is because its reactions necessitate only sodium or potassium hydroxide, a relatively economical and benign chemical [
7,
9,
19,
20,
21]. Typically, a quaternization reaction entails three stages: cellulose’s interaction with hydroxide, CHMAC’s epoxy generation, and the subsequent reaction between the epoxide and cellulose’s hydroxyl group, resulting in ether production [
21,
22]. Quaternized lignocellulosic fibers exhibit enhanced adsorptive capacity and affinity for anionic and cationic substrates. Such perfections can be attributed to the increased surface area, porosity, and chelating functional groups initiated by the quaternization process [
21,
22].
Date palm waste is abundantly available in many regions, particularly in Middle Eastern and Asian countries with extensive date cultivation [
23]. This widespread availability makes it a cost-effective and sustainable option. In addition, date palm waste exhibits unique properties that make it effective for metal ion removal. Its fibrous structure and high cellulose content provide an excellent matrix for metal ion adsorption. Additionally, functional groups like hydroxyl, carboxyl, and phenolic compounds in date palm waste enhance its capacity to chelate metal ions. Many studies examined date palm waste-based biochar or activated carbon to remove metals and showed superior efficacy. Interestingly, based on our knowledge, no study has explored chromium removal using chemically modified fibers of date palm waste.
This research uses the quaternization procedure to synthesize date palm waste (DPW) as a precursor for a biomaterial-based adsorbent, employing CHMAC as the quaternizing agent. This study examines parameters such as solution pH, contact duration, sorbent dosage, initial concentrations, column study, adsorption isotherms, and kinetics, along with their subsequent modeling, to assess the suitability of these parameters for chromium removal from aqueous solutions using QDPW. This presents a credible solution for addressing the Cr6+ issue, offering a cost-effective and ecologically sustainable alternative to conventional water treatment techniques.
2. Materials and Methods
2.1. Synthesis of Quaternized Date Palm Waste
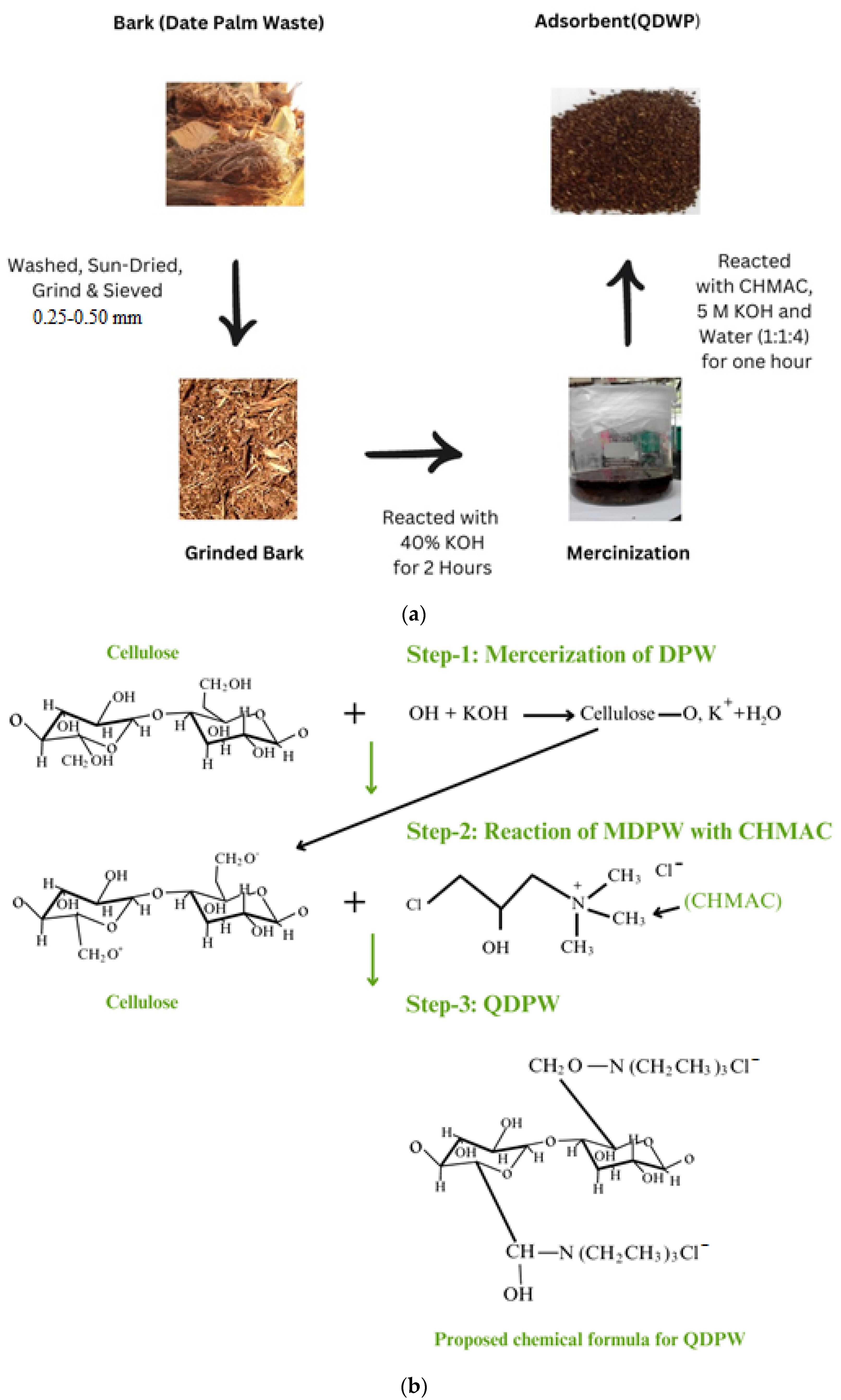
The date palm waste (DPW) bark is abundant in various regions, including Saudi Arabia. Initially, the DPW was washed with hot water, followed by rinsing with acetone to remove any remaining organic content. The washed DPW was then sun-dried for a period of 4–5 days [
18,
19]. Afterward, the unprocessed DPW underwent grinding and sieving, resulting in particle sizes ranging from 0.25 to 0.5 mm. The quaternization method employed for the synthesis of QDPW in which the fibrous DPW underwent mercerization using a 40 wt.% potassium hydroxide (KOH) (Aba Chemicals Corporation, Shanghai, China) solution for a duration of 2 h [
22]. The mercerized DPW was then rinsed with distilled water and dried for 24 h at a temperature of 60 °C in an oven.
During the process of quaternization, a stoichiometric balance was maintained for the reactants, consisting of 5 M KOH and N-(3-chloro-2-hydroxypropyl) trimethylammonium chloride (CHMAC) procured at 40% purity (Sigma, Taufkirchen, Germany). CHMAC was known as the limiting reactant in this synthesis. The weight-to-weight ratio for the reactants—KOH, CHMAC, and water was set at 1:1:4 for every gram of mercerized DPW. This mixture was sealed in an airtight container at room temperature for an hour. After the reaction, the fiber/granular material was rinsed with 0.5% acetic acid (Aba Chemicals Corporation, Shanghai, China) to halt the reaction. This was followed by multiple washes with distilled water until achieving a neutral pH. The QDPW samples were subsequently dried at 60 °C in the oven for 24 h and stored in a desiccator.
Figure 1a,b illustrate the methodology and proposed chain reaction to formulate QDPW, respectively.
Figure 1b also demonstrates the proposed chemical structure/formula of QDPW.
2.2. Characterization of DPW and QDPW
The characterization of DPW and QDPW was carried through various analytical techniques, including carbon, hydrogen, and nitrogen analysis (CHN analysis), scanning electron microscopy (SEM) equipped with energy dispersive X-ray analysis (EDX), Fourier transform infrared spectrometry (FTIR), Brunauer, Emmett, and Teller analysis (BET), thermogravimetric analysis (TGA), and X-ray diffraction (XRD).
The CHN analysis aimed to determine the proportion of nitrogen, hydrogen, and carbon present in the specimens. A sample with an approximate mass of 0.1 mg was weighed using a digital micro-balance and analyzed using LECO CHNS-932 (US) (EA Consumables, LLC, Marlton, NJ, USA).
SEM, coupled with an EDX accessory (Hitachi S-3400, Tokyo, Japan) was conducted at a voltage range of 20–30 kV. Before analysis, samples of DPW and QDPW were coated with a thin Au/Pd layer. Considering, the sample characteristics, EDX spectra were captured at magnifications ranging from 1000–5000×.
FTIR spectra were obtained using a Fourier-transform infrared spectrometer (Perkin Elmer, Waltham, MA, USA). The transmittance percentage was plotted against the wavenumber, spanning the 500 to 4000 cm−1 range, under ambient conditions. Analysis of the FTIR spectra facilitated the identification of functional groups in the samples.
The BET method was employed to analyze the surface area of DPW and QDPW. The BET surface area analyzer (Gemini analyzer, Micrometric, Sarasota, FL, USA) was used in this study. Determination of pore size (average), and average pore volume of the samples was observed using the Barrett, Johner, and Halenda (BJH) equation.
A thermogravimetric analyzer (Mettler Toledo, Columbus, OH, USA, TGA/SDTA851) was utilized in order to perform thermogravimetric analysis (TGA). About ten milligrams of the sample were utilized for the analysis, and it was heated at a rate of 10 °C per minute from 28 °C to 500 °C in a nitrogen atmosphere at a flow rate of twenty milliliters per minute. During the heating process, the weight loss was recorded for subsequent studies.
X-ray diffraction (XRD) analysis was performed to ascertain the structure of the substance and the percentage of crystallinity it possessed with help of Shimadzu XRD-6000 X-ray diffractometer (Kyoto, Japan). Nickel-filtered Cu Kα (λ = 1.542 Å) radiation with a wavelength of 1.542 Å was used for the X-ray beam, which was operated at 30 kV and 30 mA. The data obtained from diffraction were recorded at a rate of 2 degrees per minute, with scattering angles (2θ) ranging from 20 degrees to 90 degrees. The wood industry method was used to determine the proximate chemical composition of DPW and QDPW, including cellulose, hemicellulose, and lignin [
9,
24].
2.3. Preparation of Chromium Solution for Isotherms and Kinetics
A stock solution of chromium (Cr6+) was prepared by dissolving 2.8 g of potassium dichromate (K2Cr2O7) in one liter of distilled water. Subsequently, various concentrations were achieved through the process of diluting the stock solution with suitable amounts of distilled water. The Spectrophotometer (DR-900, Hach, Loveland, CO, USA) was used to verify the final concentrations. The batch studies were conducted employing 250-mL Erlenmeyer flasks. To ensure the prevention of leakage and evaporation, all flasks were appropriately sealed. The samples were mechanically agitated using a digital orbital shaker at a speed of 110 rpm (±2 rpm) and a temperature of 30 °C (±2 °C). The investigated adsorption parameters encompassed the starting pH, contact duration, QDPW doses, and initial chromium concentration. The column study and regeneration of the adsorbent was also conducted which is imperative for real-world applications.
2.4. Impact of pHpzc and pH on the Process
The pHpzc, also known as the isoelectric point, was observed by introducing 1 g/L of QDPW containing 0.01 M NaNO
3 using the drift method. The pH of the chromium solution was observed within the range of 2–9 by the addition of either 0.1 N NaOH or 0.1 N HCl. The flasks were carefully closed and agitated for a duration of three days at ambient temperature [
24]. The pH values of the chromium solutions were measured using a digital pH meter both before and after the shaking process.
The experimentation for evaluation of optimum pH for adsorption was also conducted at varying pH levels for chromium removal efficiency ranging from 2 to 9. The aqueous phase’s pH significantly influences chromium removal efficiency by bio-adsorbent [
11,
19,
25]. Biomass-derived bio-adsorbents encompass bio-macromolecules characterized by the presence of thiol, carboxyl groups, amino, and phenol. The bio-sorption process is dependent upon the deprotonation and protonation of the functional groups [
11,
14].
2.5. Impact of Dosage and Initial Concentration
The impact of different dosages of QDPW was examined in a series of batch experiments. Numerous quantities of QDPW (1–6 g/L) were introduced into 100 mL of chromium solutions with a 25 mg/L concentration. The pH of the solutions was adjusted to the optimal pH level followed by agitation process for a duration of 2 h.
The impact of the initial concentration for the adsorption of chromium onto QDPW was investigated by employing 100 mL of a chromium solution. The solution had varying initial chromium concentrations (25 mg/L, 50 mg/L, 75 mg/L, 100 mg/L, 125 mg/L, and 150 mg/L). These experiments were also performed in 250 mL Erlenmeyer flasks, and each flask was correctly sealed after adding 4 g/L of QDPW. The samples underwent agitation for a duration of 2 h.
2.6. Adsorption Dynamics of Chromium Removal on QDPW
2.6.1. Kinetic Modeling for Chromium Removal on QDPW
Kinetic studies in the realm of adsorption shed light on the rate of removal, the underlying mechanisms, and the determinants of the adsorption process. The pseudo-first- and pseudo-second-order kinetic models stand as fit models to summarize adsorption reactions. Lagergren reported the maiden model tracing the rate of adsorption encompassing the adsorption capacity [
9,
26]. Later on, Ho (2006) developed the adsorption of divalent cations onto peat, emphasizing the fundamental role of chemical bonds between the functional groups and cations on peat [
26]. His elucidation paved the path for what is known as the pseudo-second-order rate equation. This postulates that a proportionate relationship between the rate of occupation on adsorption sites and the square of the number of empty sites governs the kinetics of adsorption [
7,
9]. This frequently suggests the occurrence of chemisorption, which involves the interaction of valence forces through electron exchange or sharing between the adsorbent and adsorbate. Equations (1) and (2) manifest these two kinetic paradigms:
Here, qe is the quantity of adsorbate adsorbed at equilibrium (mg/g) and the variable “qt” represents the quantity of adsorbate that has been adsorbed at a certain time “t” in (mg/g). The symbol k1 signifies the rate constant related to the pseudo-first-order adsorption process, expressed in units of minutes to the power of a negative one.
In real-world adsorption circumstances, it is frequently observed that the second-order model may need to adequately account for all the intricacies, particularly when considering approaching equilibrium.
The symbol K2 represents the rate constant for pseudo-second-order adsorption, measured in grams per milligram per minute (g/mg·min).
In addition, the Elovich kinetic model is a mathematical model used to describe the kinetics of a chemical reaction. The Elovich equation (Equation (3)) is frequently employed in the characterization of chemisorption phenomena occurring between adsorbate and heterogeneous surfaces [
9,
25]. A higher α value indicates more rapid initial adsorption, which can be attributable to the greater abundance of active sites accessible at the onset of the process. It involves the determination of certain parameters that are essential for accurately representing the reaction.
The Weber–Morris equation suggests the prospect of diffusion governing the singular determinant of the adsorption rate. If the data unfolds a multi-linear trajectory, it signals the sorption being steered by multiple determinants (Equation (4)) [
9,
25].
2.6.2. Adsorption Isotherms
In addition, an adsorption isotherm offers a comprehensive view of the intricate linkage between the adsorbent and the adsorbate at equilibrium. This exploration unravels the essence of the adsorption sequence, whether it is a multilayer or singular layer adsorption, its optimum capacity, or the distinctions of its surface diversity. These revelations are instrumental in architecting an adept adsorption phenomenon. In this study, four distinguished isotherm paradigms, namely Langmuir, Freundlich, Redlich-Peterson, and Sips were employed to observe the data.
The Langmuir isotherm postulates the adsorption of a monolayer onto a surface that possesses a limited quantity of similar sites. Once a site has reached its capacity, no additional sorption can take place at that particular site [
7,
27]. The Langmuir equations can be expressed in a linearized form (Equations (5)–(7)).
Here, qe refers to the amount of solute adsorbed per unit weight of QDPW at equilibrium (mg/g), and Q0 is the maximum adsorption capacity corresponding to complete monolayer coverage on the surface (mg/g). In addition, KL denoted to Langmuir constant related to the energy of adsorption (L/mg). While Ce is the equilibrium concentration of the solute in the chromium solution (mg/L).
The Freundlich isotherm (Equation (8)) offers a narrative of multilayer adsorption on diversified patterns. If the value of “n” descends towards unity, the adsorption trajectory remains linear. A value less than one reflects a propensity towards chemisorption, while a value greater than 1 leans towards physisorption [
7,
15,
27].
Here, kF (mg/g) and n are Freundlich constants, indicating adsorption capacity and heterogeneity factor or adsorption intensity, respectively.
The Redlich–Peterson isotherm (Equation (9)) is a blend of the Langmuir and Freundlich isotherm principles [
9]. It offers a hybrid adsorption narrative that remains versatile across concentration gradients which is independent of the surface’s behavior.
The Sips isotherm is another hybrid model that at certain conditions, can be reduced to either the Langmuir or Freundlich isotherms [
1,
28]. It is given by (Equation (10)):
If β = 1, the Sips isotherm reduces to the Langmuir isotherm.
4. Conclusions
This research explored the potential applications of QDPW as a promising substitute adsorbent for chromium extraction from aqueous solutions. The empirical results proved the viability of QDPW as a potent adsorbent, particularly for removing hexavalent chromium from liquid mediums. At a pH level of 6.5, QDPW facilitated efficient chromium extraction. The removal efficiency (88%) with a 22.22 mg/g extraction rate was recorded with a 4 g/L dosage, making it the most economical option.
The characterization techniques, including FTIR, SEM/EDX, BET, XRD, and element analysis, showed drastic changes in the structural and surface properties of both DPW and QDPW fibers. In addition, differential thermal analysis (DTA) and thermogravimetric analysis (TGA) of DPW and QDPW revealed radical changes at various stages. In the analysis of the adsorption equilibrium data using various isotherm models, it was evident that the Langmuir, Redlich–Peterson, and Sips models provided the most accurate representation with a coefficient of determination (R2) of 0.989, 0.981, and 0.983, respectively. Based on the value of RMSE (0.6245), which is the lowest among all applied isotherm models, Redlich–Peterson best describes the phenomena. On the other hand, values of g (0.952) and B (0.931) in the Redlich–Peterson and Sips models, respectively, are close to unity, which showed dominance of monolayer adsorption.
Moreover, the kinetics associated with chromium adsorption onto QDPW were most consistent with the pseudo-second-order model’s correlation coefficient, nearing 0.999, signifying the robustness of the adsorption process. This high correlation intimates a dominant role of either an electrostatic attraction or ion exchange in chemisorption phenomena. In addition, the column study also revealed the adsorption capacity of 51.56 mg/g in a continuous flow system which showed its applicability on a commercial scale.