Abstract
The Lovozero Alkaline Massif intruded through the Archean granite-gneiss and Devonian volcaniclastic rocks ca. 360 Ma ago and formed a large laccolith-type body. The lower part of the massif (the Layered complex) is composed of regularly repeating rhythms: melanocratic nepheline syenite (lujavrite, at the top), leucocratic nepheline syenite (foyaite), foidolite (urtite). The upper part of the massif (the Eudialyte complex) is indistinctly layered, and lujavrite enriched with eudialyte-group minerals (EGM) prevails there. In this article, we present the results of a study of the chemical composition and petrography of more than 400 samples of the EGM from the main types of rock of the Lovozero massif. In all types of rock, the EGM form at the late magmatic stage later than alkaline clinopyroxenes and amphiboles or simultaneously with it. When the crystallization of pyroxenes and EGM is simultaneous, the content of ferrous iron in the EGM composition increases. The Mn/Fe ratio in the EGM increases during fractional crystallization from lujavrite to foyaite and urtite. The same process leads to an increase in the modal content of EGM in the foyaite of the Layered complex and to the appearance of primary minerals of the lovozerite group in the foyaite of the Eudialyte complex.
1. Introduction
Zirconosilicate of sodium, calcium, and iron, “eudialyte” was described more than 200 years ago in samples from the Ilimaussaq alkaline massif in Greenland [1]. Yet, even now this mineral, more precisely a group of minerals (eudialyte-group minerals, or EGM), is being actively studied and is a subject of debate. Such interest is caused not only by the unusual crystal structure of eudialyte-group minerals [2], but also the ability to use it to extract Zr and rare earth elements (REE) [3,4,5]. Currently, vast amounts of eudialyte mineral deposits have been reported, some of which are located in Pajarito (New Mexico, USA) [6], Lovozero (Kola Peninsula, Russia) [7], Ilimaussaq (South Greenland) [8], Mont Saint-Hilaire (Canada) [9], and Norra Kärr (Sweden) [10].
The chemical composition and crystal structure of eudialyte-group minerals are sensitive to even minor changes in the geochemical media during magmatic crystallization and subsequent ion-exchange processes. Therefore, the EGM are widely used to reconstruct the conditions of mineral crystallization [11,12,13,14,15,16,17]. The EGM are characteristic minerals of highly evolved, peralkaline (molar (Na + K)/Al > 1) magmatic rocks [8,18,19,20,21]. In syenitic systems, the EGM are indicator minerals of so-called agpaitic rocks [8,18,22,23]. Although such rocks are neglegible in volume of the earth’s crust, it is important to study conditions of their formation for mineralogy and material science, since agpaitic rocks concentrate high field strength elements (HFSE) and are famous for their exceptional variety of titanium- and zirconosilicates.
The EGM are extremely widespread in the Lovozero alkaline massif (Kola Peninsula, Russia). They are found in all types of rock, and often occur as rock-forming minerals. The EGM crystallized at all stages of the Lovozero massif evolution, recording a constant change in the composition of the melt/solution, temperature, and fO2. This led to a large diversity of EGM, and many new minerals of this group were discovered in the Lovozero massif [24,25,26,27]. Due to the long crystallization period and sensitivity to external conditions, the EGM are excellent petrogenetic indicators. This article is based on the results of studying the chemical composition, occurrence, paragenetic associations, and secondary transformations of the EGM from the main types of the Lovozero massif rocks. We emphasize that the aim of the present paper is not to treat crystallographic aspects, nor to put further constraints on possible site occupancies of the EGM structure. Instead, our results place constraints on the possibility of using eudialyte as a petrogenic indicator.
2. Geological Setting
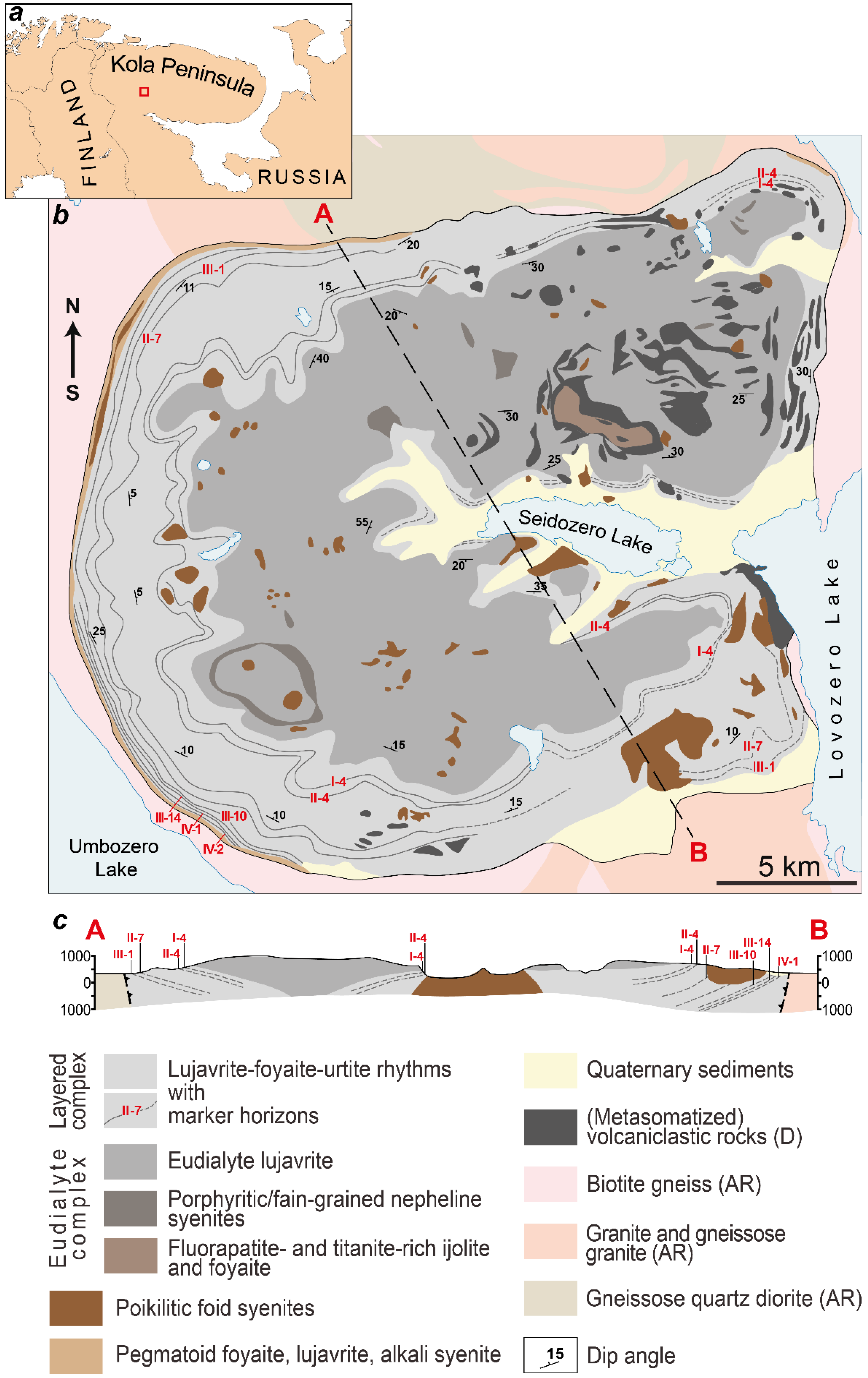
The Lovozero layered pluton (Figure 1) is located in the Archean granite and gneissose granite, has a form of laccolith, and covers an area of 650 km2. Its age is 370 ± 7 Ma (whole rock Rb–Sr isochron [28]). Pluton consists of two macro units: the Eudialyte complex (at the top) and the Layered complex [29,30]. The Layered complex of the Lovozero massif consists of the subhorizontal layers (or rhythms). The bottom of each rhythm is composed of urtite. Higher in the section, the content of alkali feldspar increases, and urtite gradually turns into foyaite (leucocratic nepheline syenite). At the top of the rhythm, the content of mafic minerals, such as pyroxene and amphiboles, increases, and foyaite gradually passes into lujavrite (trachytoid melanocratic nepheline syenite). Sometimes, there is no urtite in the rhythm and such a rhythm consists only of a foyaite-lujavrite sequence. The contact between underlying lujavrite and overlying urtite (or foyaite) is sharp. The rhythms are well-traced horizontally, and urtite (so-called “marker horizons”) has its own numbers consisting of Roman and Arabic numerals (Figure 1). The Eudialyte complex consists of lujavrite with lenses of foyaite, porphyritic/fine-grained nepheline syenites, and urtite.
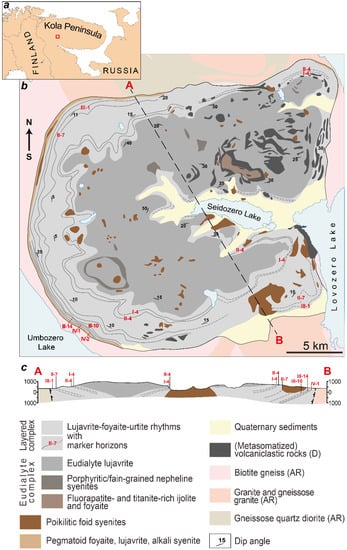
Figure 1.
(a) location of the Lovozero massif within the Kola Peninsula (red square); (b) geological scheme of the Lovozero massif and (c) cross-section along the line A–B [30].
Among the rocks of the Layered and Eudialyte complexes, xenoliths of volcaniclastic rocks are widespread. There are both unchanged xenoliths, which consist of overlapping olivine basalt, basalt tuff, tufite, sandstone, quartzite, and intensely metasomatized xenoliths. Poikilitic nepheline and sodalite-nepheline syenite (foid syenites) form lenses and rounded bodies among rocks of the Layered and Eudialyte complexes. Almost all pegmatites and hydrothermal veins of the Lovozero massif containing various rare-metal minerals are associated with these rocks. Pegmatites, the mineral composition of which is usually less diverse and corresponds to the composition of the host rocks, are common throughout the massif.
3. Materials and Methods
We used samples of eudialyte-containing rocks (487 samples) collected in the Lovozero massif during many years of its research at the GI KSC RAS. These samples were taken both from the surface of the massif and the drill cores (Mt. Alluaiv) and from an underground mine (Mt. Karnasurt and Mt. Kedykvyrpakhk). The 487 thin polished sections were analyzed using the scanning electron microscope LEO-1450 (Carl Zeiss Microscopy, Oberkochen, Germany) with the energy-dispersive system Röntek and Quantax 200 (from 2012) to obtain BSE (back scattered electron) images and pre-analyze all detected minerals. The chemical composition of minerals was analyzed with the Cameca MS-46 electron microprobe (Cameca, Gennevilliers, France) operating in the WDS-mode at 22 kV with a beam diameter of 10 µm, beam current of 20–40 nA, and counting times of 20 s (for a peak) and 2 × 10 s (for background before and after the peak), with 5–10 counts for every element in each point. The analytical precision (reproducibility) of mineral analyses is 0.2–0.05 wt.% (2 standard deviations) for the major element and ca. 0.01 wt.% for impurities. The standards used, the detection limits, and the analytical accuracy values are given in Supplementary Table S1. The systematic errors are within the random errors. Statistical analyses are carried out using the STATISTICA 13 [31]. For the statistics, resulting values of the analyses below the limit of accuracy (see Supplementary Table S1) are considered ten times lower than the limit. The EGM contain an extensive set of elements, and a factor analysis is the most suitable way to analyze the interrelations between the values of a large number of variables [32]. Factor analysis is a statistical method used to describe variability among observed, correlated variables in terms of a lower number of unobserved variables called factors. This method allows for describing an object comprehensively and at the same time compactly. Mineral abbreviations mentioned in this article are shown in Table 1.

Table 1.
Mineral abbreviation.
4. Results
4.1. Petrography
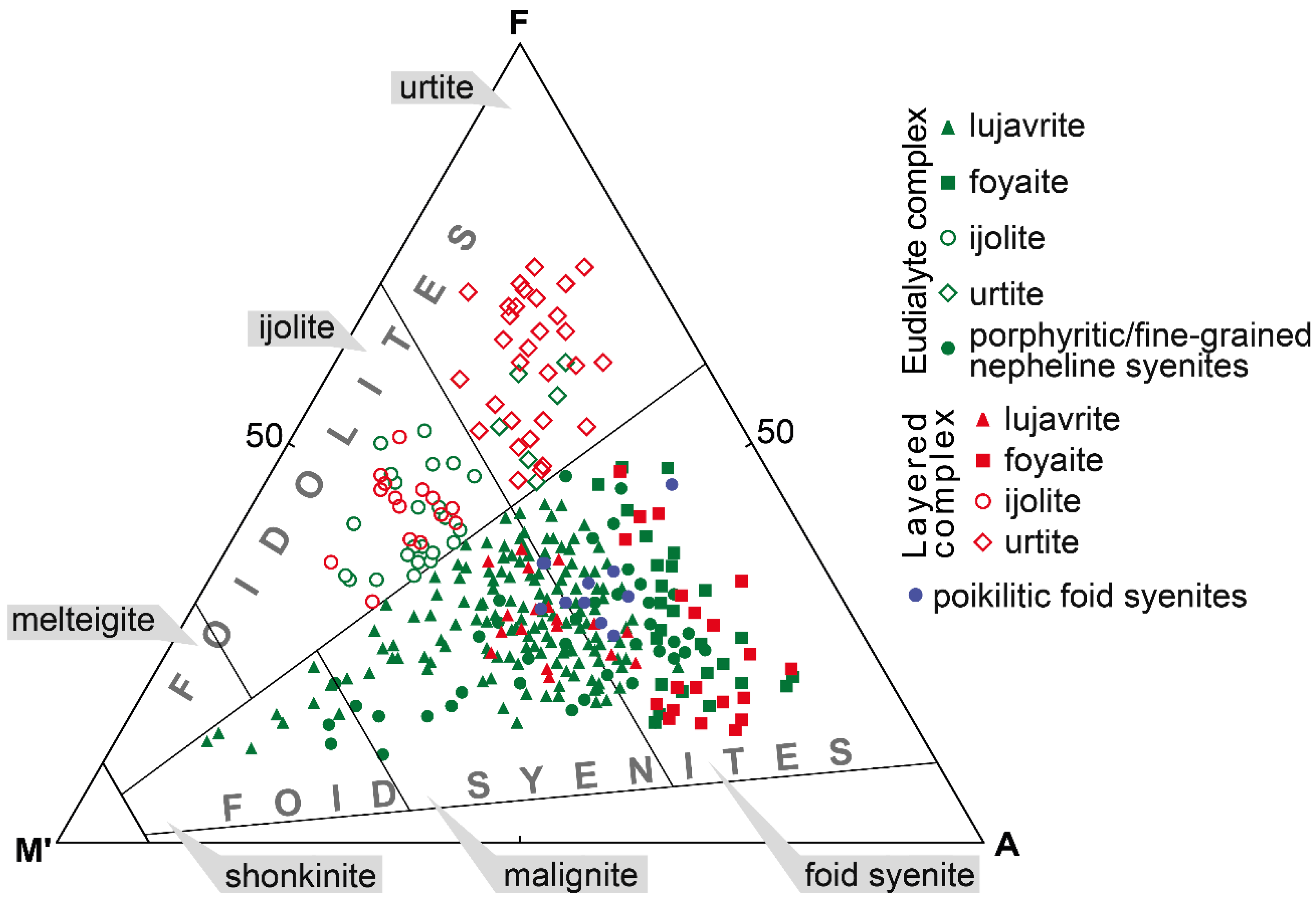
The Lovozero massif is composed of alkaline rocks, which are either foid syenites or foidolites. According to International Union of Geological Sciences (IUGS), these rocks are modally defined in the QAPF fields 11 and 15 [33]. Depending on the content of mafic minerals, nepheline syenites are additionally subdivided into shonkinite (M’ = 60–90 modal %, where M’ is defined as a sum of mafic minerals minus muscovite, apatite, primary carbonates), malignite (M’ = 30–60) and nepheline syenite (M’ < 30), and foidolites are subdivided into melteigite (M’ = 70–90), ijolite (M’ = 30–70), and urtite (M’ < 30) (Figure 2). Note that names of rocks for the Lovozero massif, as well as for the Ilimaussaq massif, are outdated, but suitable for use, e.g., lujavrite and foyaite, are widely used. Figure 2 shows the correspondence of the rock names used in this article to the nomenclature of the IUGS.
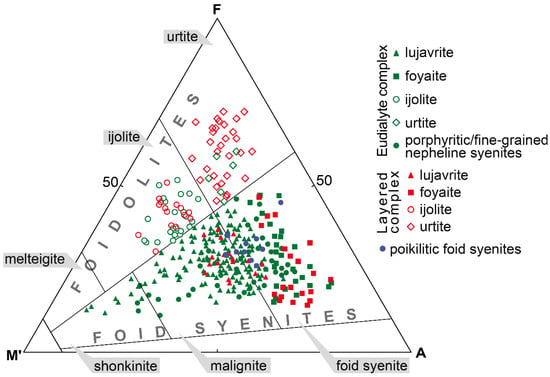
Figure 2.
Modal composition and rock names of the alkaline rocks of the Lovozero massif. A—alkali feldspar, F—foid, M’—color index (sum of mafic minerals minus muscovite, apatite, primary carbonates). The AFM’ triangle fields contain rock names accepted by the IUGS.
4.1.1. Rocks of the Layered Complex
Urtite (leucocratic foidolite, Figure 2) is a massive coarse- or medium-grained rock. Nepheline occurs as euhedral to subhedral phenocrysts and microcline forms up to 1.5 cm large euhedral to subhedral laths. Aegirine-(augite) forms large (up to 2 cm across) poikilitic crystals (Figure 3a), and aggregates of its small needle-like crystals in interstice of nepheline and feldspar are less common. In these aggregates, amphiboles are always in close intergrowth with clinopyroxenes, but their content is much lower than the one of clinopyroxenes and rarely reaches 5% of the rock volume. Accessory minerals of urtite are sodalite, fluorapatite, loparite-(Ce), and EGM. All of them are unevenly distributed in urtite. Fluorapatite and loparite-(Ce) are mainly in the lower parts of the urtite horizons, sodalite forms local clusters. The EGM content in urtite reaches 20%, and its average content is 2.4 modal % [29]. The EGM occur exclusively as anhedral grains in interstices of rock-forming minerals (Figure 3a), sometimes they form poikilitic crystals with inclusions of nepheline, aegirine, fluorapatite (Figure 3b). Poikilitic or skeletal crystals of loparite-(Ce) frequently enclose the EGM grains in mono- and polymineralic inclusions.

Figure 3.
BSE–images. Morphology of the EGM from the Layered complex of the Lovozero massif. In all types of rocks, the anhedral EGM grains fill the interstices of rock-forming minerals. (a) Urtite LV-01-54 (Mt. Kitknyun); (b) urtite LV-IV-2-2 (Mt. Kedykvyrpakhk); (c) ijolite LV-III-2-2 (Mt. Kedykvyrpakhk); (d) foyaite LV-307-2 (Mt. Alluaiv); (e) lujavrite LV-316-1 (Mt. Alluaiv); and (f) lujavrite LV-02-25 (Mt. Parguaiv).
Ijolite (mesocratic foidolite, Figure 2) differs from urtite by a higher content of mafic minerals (clinopyroxenes, amphiboles, and loparite-(Ce)). Parallel oriented long-prismatic crystals of amphibole and clinopyroxene are grouped into “streams”, and the EGM are located inside these “streams” (Figure 3c).
Foyaite (leucocratic nepheline syenite, Figure 2) is a massive to trachitoid coarse- to medium-grained rock. Rock-forming minerals are nepheline, microcline, aegirine-(augite), and amphiboles (arfvedsonite-magnesioarfvedsonite series). Foyaite differs from urtite only by a higher content of alkali feldspar. The accessory minerals of foyaite are the EGM, sodalite, lamprophyllite, murmanite, and lomonosovite. In foyaite, as well as in urtite, the EGM are widespread and sometimes become rock-forming (the content reaches 25%). The average content of the EGM in foyaite is 2.7 modal %. The EGM form anhedral grains in the interstices of rock-forming minerals (Figure 3d), as well as poikilitic crystals with inclusions, mainly nepheline.
Lujavrite (meso- or melanocratic nepheline syenite, Figure 2) consists of microcline, nepheline, clinopyroxenes (aegirine, aegirine-augite), and amphiboles (magnesioarfvedsonite, arfvedsonite) and has a trachytoid texture. Long prismatic crystals of clinopyroxenes and amphiboles are grouped into a communicating system of “streams”, as in ijolite. Such “streams” of mafic minerals flow around alkali feldspar and nepheline crystals. Typical accessory minerals of lujavrite are sodalite, loparite-(Ce), the EGM, lamprophyllite, and baritolamprophyllite. Sodalite forms euhedral or round crystals, (barito)lamprophyllite form poikilitic crystals, and loparite-(Ce) is usually present as skeletal twins. The EGM are in the interstices of clinopyroxenes and amphiboles, inside the “streams” (Figure 3e). The small EGM grains are often located inside skeletal crystals of loparite-(Ce) (Figure 3f). Overall, the EGM content in lujavrite is lower than in urtite and foyaite (average 2%, maximum 10% [29]).
4.1.2. Rocks of the Eudialyte Complex
Eudialyte lujavrite (Mesocratic or melanocratic nepheline syenite enriched in EGM, Figure 4a) prevail in the Eudialyte complex. In terms of mineral composition and texture, these rocks are similar to the lujavrite of the Layered complex, but there are also differences. First, the content of EGM in local areas can reach 90%. Secondly, the “streams” of mafic minerals often have an internal structure. The marginal parts of the “streams” consist of long prismatic clinopyroxene crystals, and the axial zones are composed of larger anhedral amphibole grains. The EGM form round or oval grains, and are situated in the axial parts of such “streams”. The marginal parts (up to 200 µm) of the EGM grains are saturated with inclusions of aegirine and, in smaller quantities, magnesioarfvedsonite (Figure 4b). In addition, the content of sodalite in eudialyte lujavrite is higher than in lujavrite of the Layered complex and reaches 20%.

Figure 4.
Diversity in morphology of the EGM from the Eudialyte complex of the Lovozero massif. (a) Euhedral grains of EGM in eudialyte lujavrite LV-44-6 (Mt. Alluaiv, photo of a polished thin section in transmitted light); BSE-images: (b) euhedral grains of EGM in eudialyte lujavrite LV-29-37 (Mt. Alluaiv); (c) anhedral grains of EGM in foyaite LV-33-35 (Mt. Alluaiv); (d) rounded grains of EGM in porphyritic nepheline syenite LV-191 (Mt. Kuivchorr); (e) rounded grains of EGM in fine-grained nepheline syenite LV-154-280 (Mt. Alluaiv). Morphology of other EGM rocks of the Lovozero massif: (f) anhedral grains of EGM in sodalite syenite LV-222-448 (Mt. Alluaiv); (g) anhedral grains of EGM in metasomatized volcaniclastic rock LV-180b (Mt. Kuivchorr); and (h) parakeldyshite surrounded by EGM in metasomatized volcaniclastic rock LV-152-9 (Mt. Appuaiv).
4.1.3. Poikilitic Foid Syenites
In these rocks, subhedral grains of sodalite, vishnevite-cancrinite, and nepheline are enclosed in large (up to 15 cm across) alkali feldspar crystals. Foids do not occur simultaneously, but form two associations: sodalite (predominant) + nepheline and vishnevite-cancrinite (predominant) + nepheline. Outside feldspar, interstices of feldspathoids are filled with long prismatic crystals of aegirine-(augite). The EGM are usually located among aegirine and form anhedral grains, poikilitic crystals (Figure 4f), and thin discontinuous rims around feldspathoid grains enclosed in microcline crystals. The EGM content does not exceed 5% of the rock volume.
4.1.4. Metasomatized Volcaniclastic Rock
The EGM were also formed during metasomatic changes (fenitization) of volcaniclastic rocks. As a result of this process, fine- or medium-grained rocks with a metasomatic texture consisting mainly of albite, nepheline, microcline, aegirine, and magnesioarfvedsonite are formed. Enrichment in titanite, fluorapatite, and ilmenite up to the formation of apatite-titanite ores is typical. The EGM form poikilitic crystals or clusters of small grains (Figure 4g). Segregations, in which the EGM grow around parakeldyshite grains in association with titanite, ilmenite, pyrophanite, fluorapatite, and lamprophyllite, are also common (Figure 4h).
4.2. Crystal Structure and Chemical Composition of the EGM
After [34], the general EGM formula can be written as:
where
N(1)3N(2)3N(3)3N(4)3N(5)3M(1)6M(2)3-6M(3)M(4)Z3[Si24O72]O’4-6X2,
N(1–5) = Na, H3O+, K, Sr, REE, Y, Ba, Mn, Ca;
M(1) = Ca, Mn, REE, Na, Sr, Fe;
M(2) = Fe, Mn, Na, Zr, Ta, Ti, K, Ba, H3O+;
M(3) and M(4) = Si, Nb, Ti, W, Na;
Z = Zr, Ti, Nb;
O’ = O, (OH), H2O;
X = Cl, F, H2O, OH, CO3 and SO4.
As a rule, one letter corresponds not to one site in the EGM structure, but to a group of closely spaced sites. Each letter corresponds to a region in the unit cell, which can be populated by different components that differ in size, coordination number, and/or charge [35].
In 1971, Golyshev with co-authors [36] and Giuseppetti with colleagues [37] independently solved the crystal structure of eudialyte. The EGM crystallize in the Rm, R3m or R3 space groups. The eudialyte sensu stricto has Rm symmetry, whereas substitutions like Ca + Fe2+ + Si → REE + Mn + Nb lead to decreasing symmetry [34,35]. In terms of modular crystallography, the crystal structure of EGM can be described as a stacking of complex ZTMT modules perpendicular to [001] with tq = 1/2a + 1/3c [38]. Each module contains four Z, T, M, and T layers based on T-, Z-, and M-polyhedra. Two adjacent tetrahedral T-layers has the same topology and consist of [Si3O9]6− and [Si9O27]18− rings, which are connected by shared vertexes with the N(4) distorted octahedra [36,37]. A nine-membered ring may be centered by an additional M(4B) tetrahedra occupied by Si or be vacant. Ordering of the additional Si-centered tetrahedra is a case of doubling c parameter ~60Å for the EGM [39]. The Z-layers are sandwiched between two T-layers and composed by the ZrO6 octahedra and N(1A, B) polyhedra. The M-layer is composed by octahedral six- and nine-membered rings based on edge-shared M1O6 or alternating M1O6 and M2O6 octahedra (square pyramid or square). One half of the nine-membered rings are usually centered by an additional M(4A) octahedra. Two different types of M-layer were observed in several “megaeudialytes” [40,41]. The Cl‒ and OH‒ anions are located in cavities of the M layer, and may reach 2 apfu. The eudialyte MT-framework usually contains a significant number of splitted and low-occupied sites {N(1A,B), M(2A,B), and M(4A,B)}.
The EGM structure contains numerous extra-framework N sites ordinary populated by Na, K, which can be easily exchanged by Ba2+, Sr2+, Pb2+, H3O+ H2O, CO32−, and SO42− via complex substitutions under hydrothermal conditions [2,42]. Various sites in the crystal structure can accommodate a third of the periodic table [2]. Consequently, the eudialyte group includes a wide range of minerals of varying compositions and space groups [43].
To calculate the EGM formula, the sum of (Si + Zr + Ti + Nb + Al + Hf + W + Ta) cations was normalized to 29 atoms per formula unit (apfu) and portions of all other cations resulted from this standardization [34,44]. Some of the calculation results are presented in Table 2 and Table 3.

Table 2.
Representative microprobe analyses of the EGM from the Layered complex, wt.%.

Table 3.
Representative microprobe analyses of the EGM, wt.%.
These calculations showed that Zr pfu exceeds 3 (up to 17.14 wt.% ZrO2 or 4.17 Zr pfu) in 70% of the analyses, i.e., these samples contain zirconium, which occupies not only the Z position. An excess number of Zr atoms was first revealed in the Giuseppetti eudialyte [37]. These excess Zr atoms are located at the M(3) and M(4) sites with an octahedral coordination. Subsequently, excess Zr atoms were found at the M(2) sites in the planar square in hyperzirconium eudialyte [45,46] and at sites with both fourfold and fivefold coordinations in hyperzirconium sulfate eudialyte [47]. A study on the crystal structure of five eudialyte samples (LV-154-0, LV-117-76, LV-117-111, LV-153-1, and LV-157-178) from drill cores located at Mt. Kedykvyrpakhk (Lovozero massif) showed that the excess of zirconium is located at the position M(2B) [48].
Calculation for 29 atoms (Si + Zr + Ti + Nb + Al + Hf + W + Ta) excludes the possibility of zirconium entering into positions that are not taken into account as the basis for calculating the formula. Therefore, it is not suitable for calculation of the formulae of the most EGM samples from the Lovozero massif. Following [35], the formula coefficients were calculated either (1) by the content of the unit cell or (2) by the number of Si atoms estimated by the IR spectra.
Table 4 shows two options for calculating the formula of the raslakite. This is a eudialyte group mineral discovered by Chukanov et al. [24] in 2003 in agpaitic pegmatite of the Lovozero massif. Raslakite contains an excess of zirconium, which occupies the the M(2) site. For calculation, we used the composition and structural data for the raslakite sample from the article of Chukanov et al. [24]. Table 4 shows the calculation based on Si = 25.40 (according to structural data) and (Si + Zr + Ti + Nb + Al + Hf + W + Ta) = 29 and the results of these calculations differ significantly from each other. Thus, the calculation of the formulas of the minerals of the eudialyte group without taking into account structural data may lead to incorrect results.

Table 4.
The chemical composition and calculation of the formula of raslakite [24].
Regardless of the calculation method, the coefficients in the formula of any mineral are obtained by normalizing the atomic amounts of cations and anions. Therefore, the ratio of the formula coefficients of any elements and the ratio of atomic quantities of the same elements are equal. So, in raslakite (Table 4), the ratio Mnatomic amount/Featomic amount (0.0283/0.0699 = 0.40) is equal to the ratio Mnpfu/Fepfu (0.88/2.17 = 0.40 or 0.85/2.10 = 0.40).
In this article, to compare the chemical composition of the EGM from different rocks of the Lovozero massif, we used the ratios of atomic amounts of elements. The results of the calculation of atomic amounts for the EGM samples used in this work are given in Supplementary Table S2. In this calculation, iron is divalent. R.K. Rastsvetaeva [2] notes that “Iron ions in the majority of eudialytes are divalent; however, eudialytes formed in an oxidizing medium can contain trivalent iron ions”. The reduced conditions during crystallization of the Lovozero pluton are recorded by a methane-dominated magmatic fluid phase [49,50] and by compositions of pyroxenes [51]. Previous Mössbauer spectroscopy studies indicate that the Fe3+/ΣFe of all natural eudialytes reported in the literary sources is <0.2, except for those that are significantly hydrated [52].
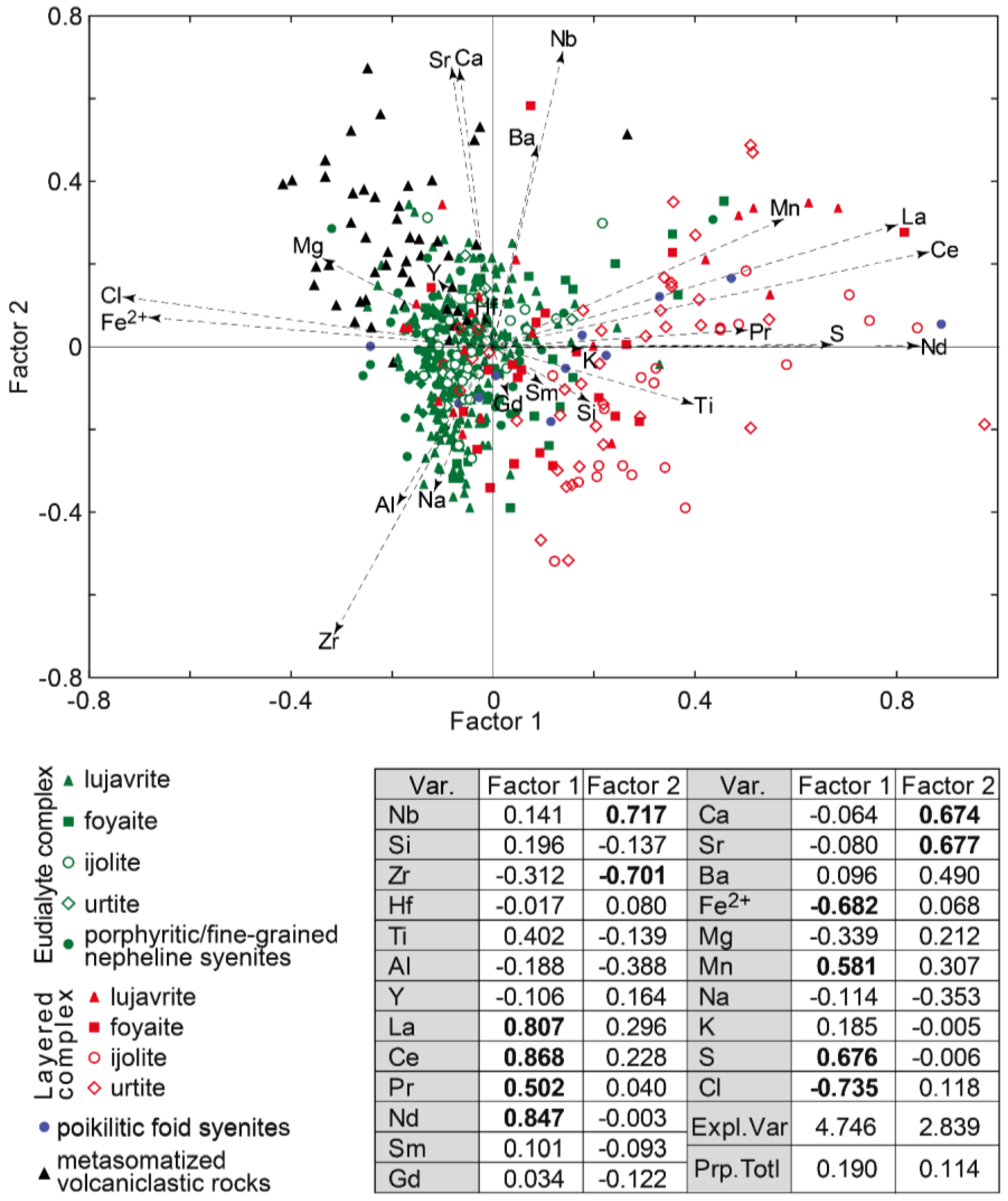
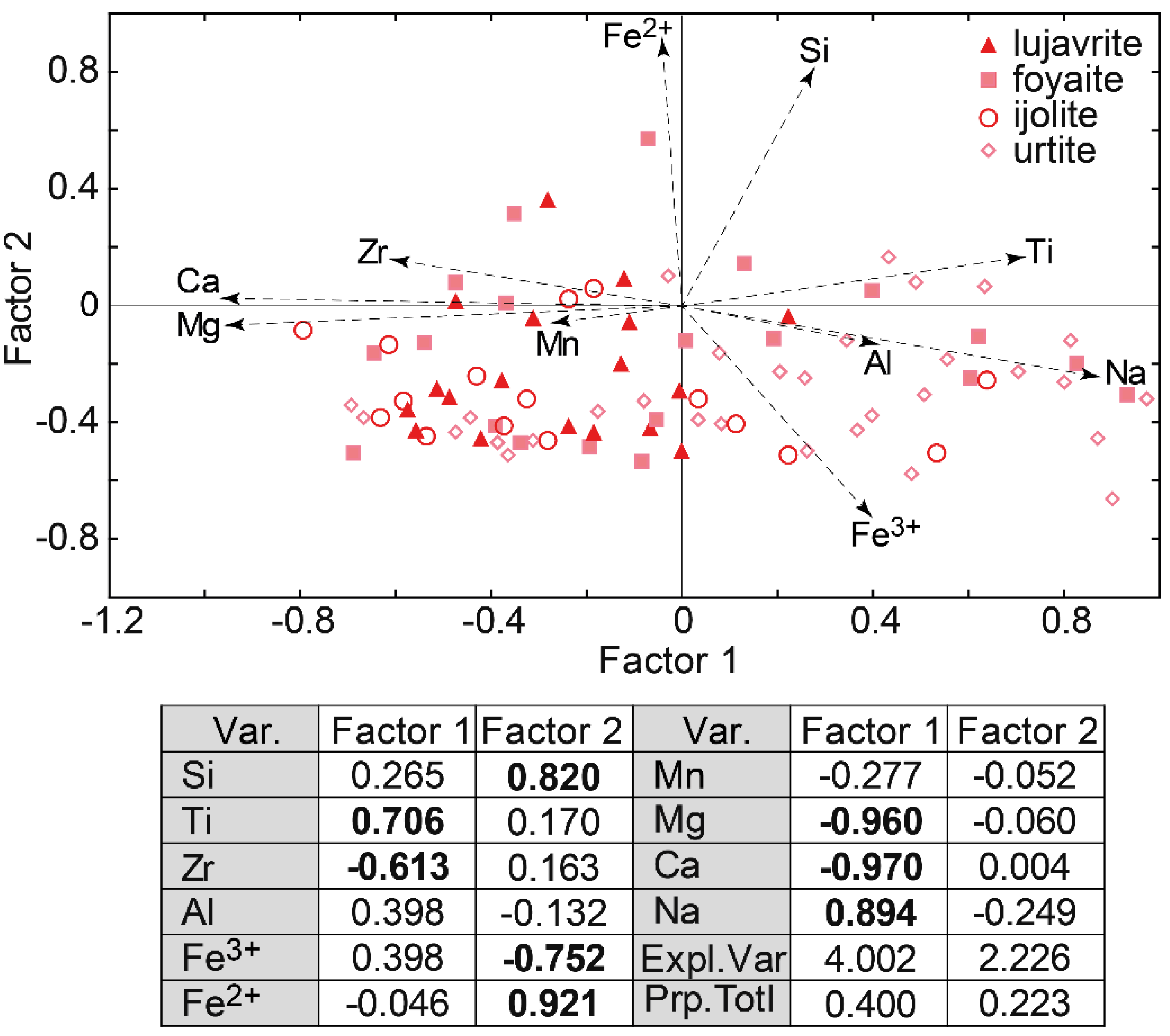
Factor analysis of data on the EGM composition showed that they are divided into three groups (Figure 5). The first group includes the EGM enriched in Fe2+, Mg, Cl, as well as in Zr. These are the EGM from rocks of the Eudialyte complex, mainly melanocratic (eudialyte lujavrite and ijolite). Among the rocks of the Eudialyte complex, the maximum contents of the elements listed above are typical of porphyritic/fine-grained nepheline syenite, and the minimum—for foyaite.
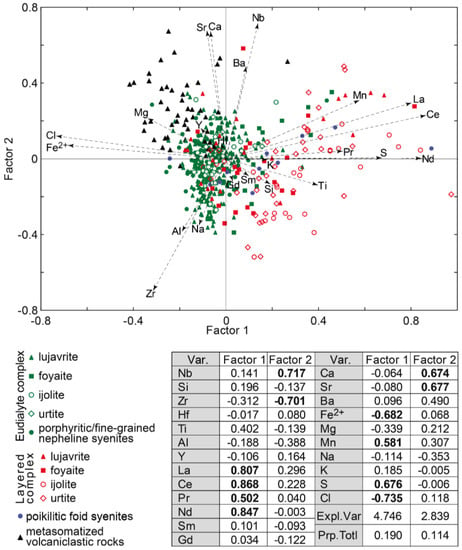
Figure 5.
Results of factor analyses of data on the composition (in atomic amount) of the EGM from the Lovozero massif. Var.—variables; Expl.var.—Explained variance; Prp.totl—proportion of total variance. Factor loadings > |0.5| are shown in bold.
The second group includes the EGM rich in REE, Mn, Ti, and S. This group includes the EGM from rocks of the Layered complex. The highest concentrations of manganese and associated elements are typical of the leucocratic rocks enriched in nepheline, i.e., urtite, as well as ijolite. The third group includes metasomatized volcaniclastic rocks. The eudialyte-group minerals in these rocks contain relatively high concentrations of Ca, Sr, Ba, Nb, and, in addition, are enriched in divalent iron, magnesium, and chlorine. Among the EGM from poikilitic foid syenites, the whole spectrum of compositions is presented, from enriched Fe2+, Mg, and Cl to those containing maximum concentrations of REE, Mn, and Ti.
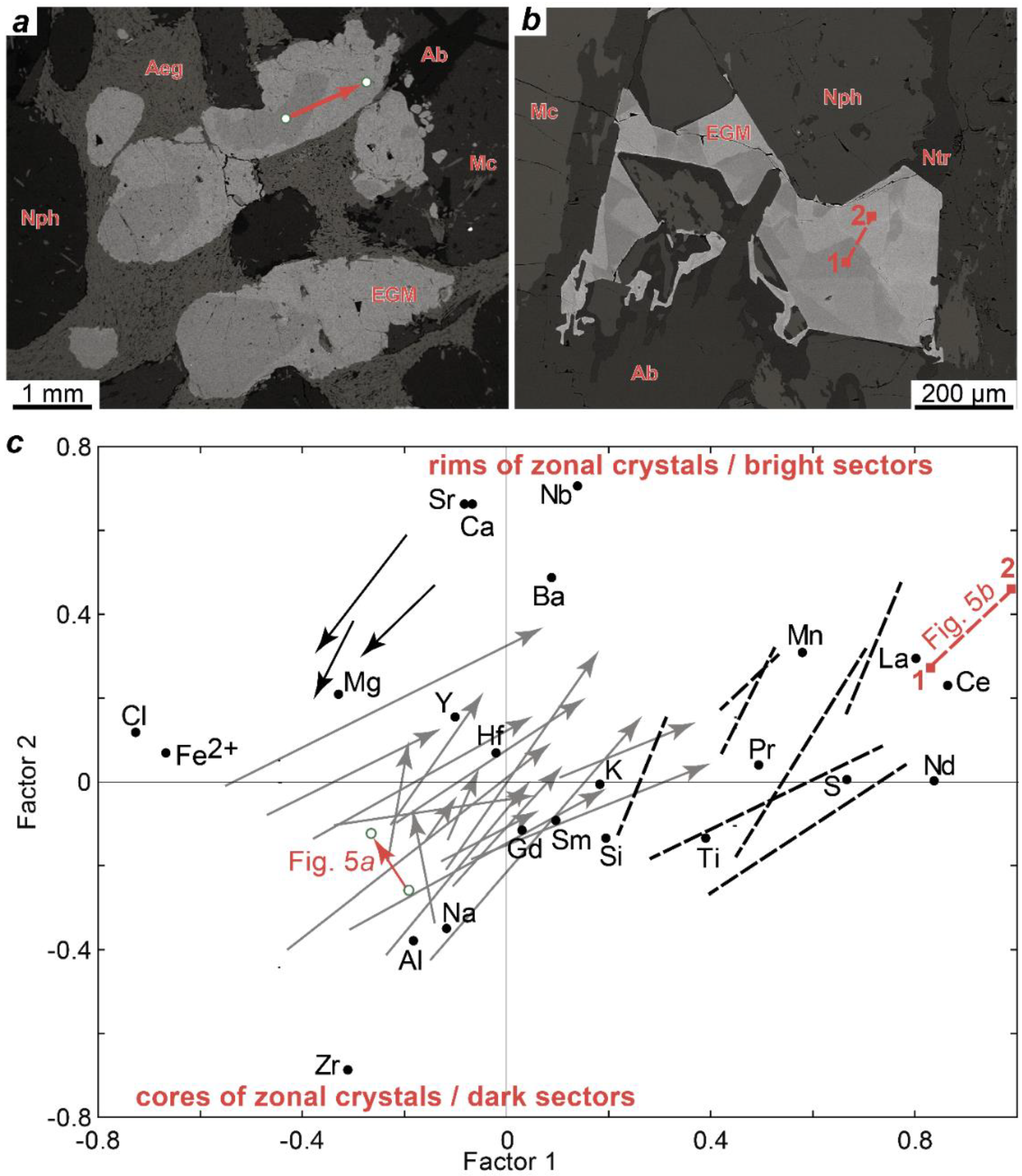
The Factor 2 is associated with the presence of zones and sectors of different composition in the EGM grains. Oscillatory zoning is typical of the rounded or oval EGM grains from the meso- and melanocratic rocks of the Eudialyte complex (lujavrite and ijolite). Two zones are usually most clearly distinguished: the core enriched in zirconium, aluminum, and sodium, and the rim with a high content of Ca, Sr, Ba, and Nb. Figure 6a shows the oscillatory zonal EGM grains from the ijolite of the Eudialyte complex, and Figure 6c shows the points corresponding to the composition of the core and rim of the grain in the coordinates of the Factor 1 vs Factor 2 (see Figure 5). The width of the outer zone is variable, and the widest sections of this zone in neighboring grains are often oriented in the same direction (Figure 6a). The sector zoning is typical of the anhedral EGM grains located in interstices of rock-forming minerals. This is most clearly observed in leucocratic rocks, namely in foyaite, and urtite from the Layered and Eudialyte complexes. One example of sector zoning is shown in Figure 6b, and the corresponding points are shown in Figure 6c.
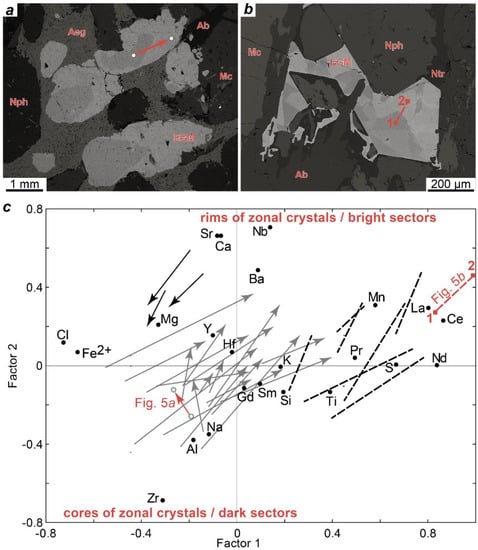
Figure 6.
(a) BSE-image of the oscillatory zonal EGM grains from the ijolite LV-222-151; (b) BSE-image of the sector-zoned EGM grains from the foyaite LV-IV-3-5; (c) position of the points shown in Figure 6a (red arrow) and Figure 6b (red dotted line) in the coordinates of the Factor 1 vs Factor 2 (see the results of factor analysis in Figure 5). The gray arrows show some examples of the oscillatory zoning (the arrow is directed from the core to the rim) in the EGM from magmatic rocks. The black arrows show some examples of the oscillatory zoning (the arrow is directed from the core to the rim) in the EGM from metasomatized volcaniclastic rocks. Black dotted lines show some examples of the sector zoning in the EGM.
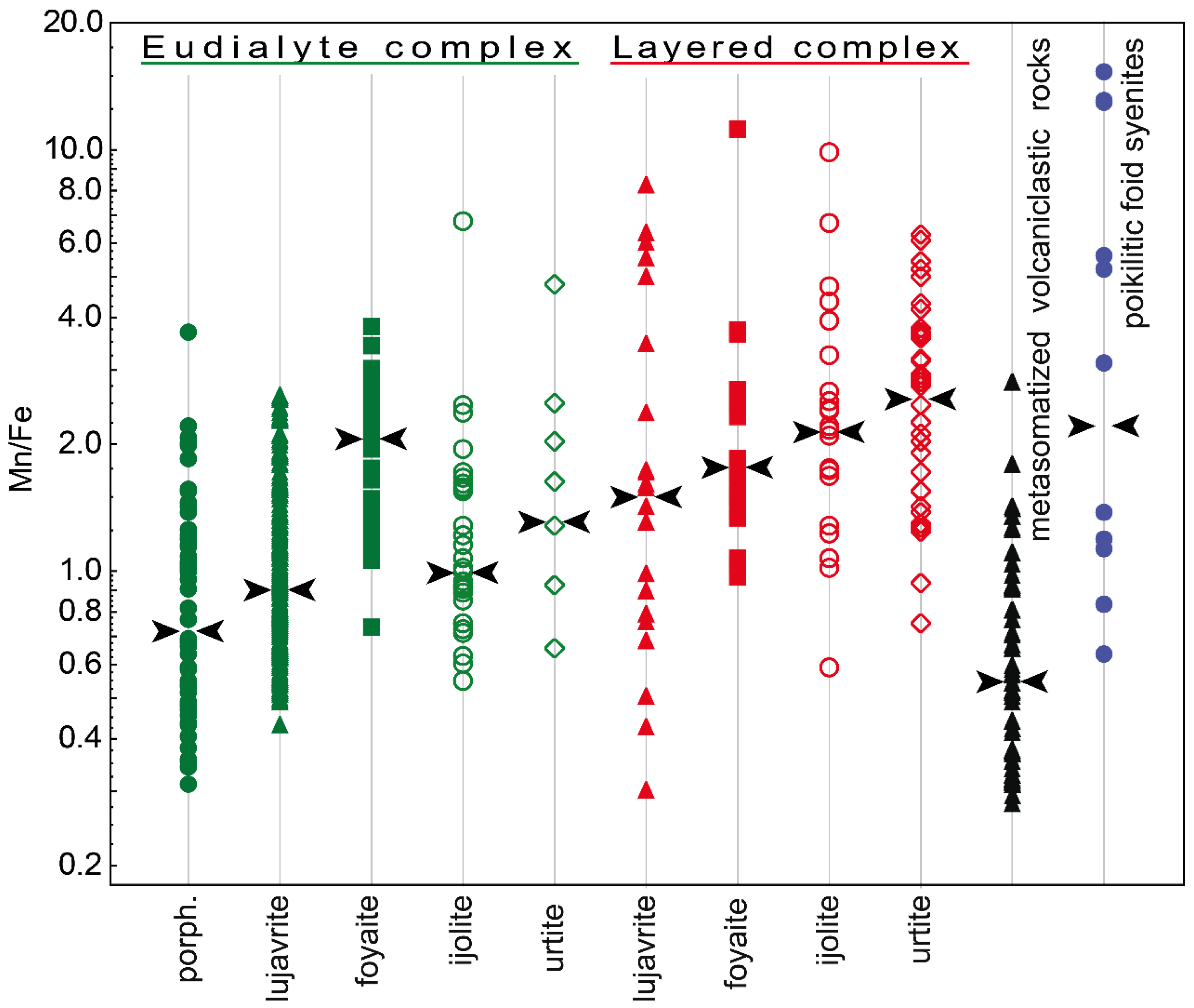
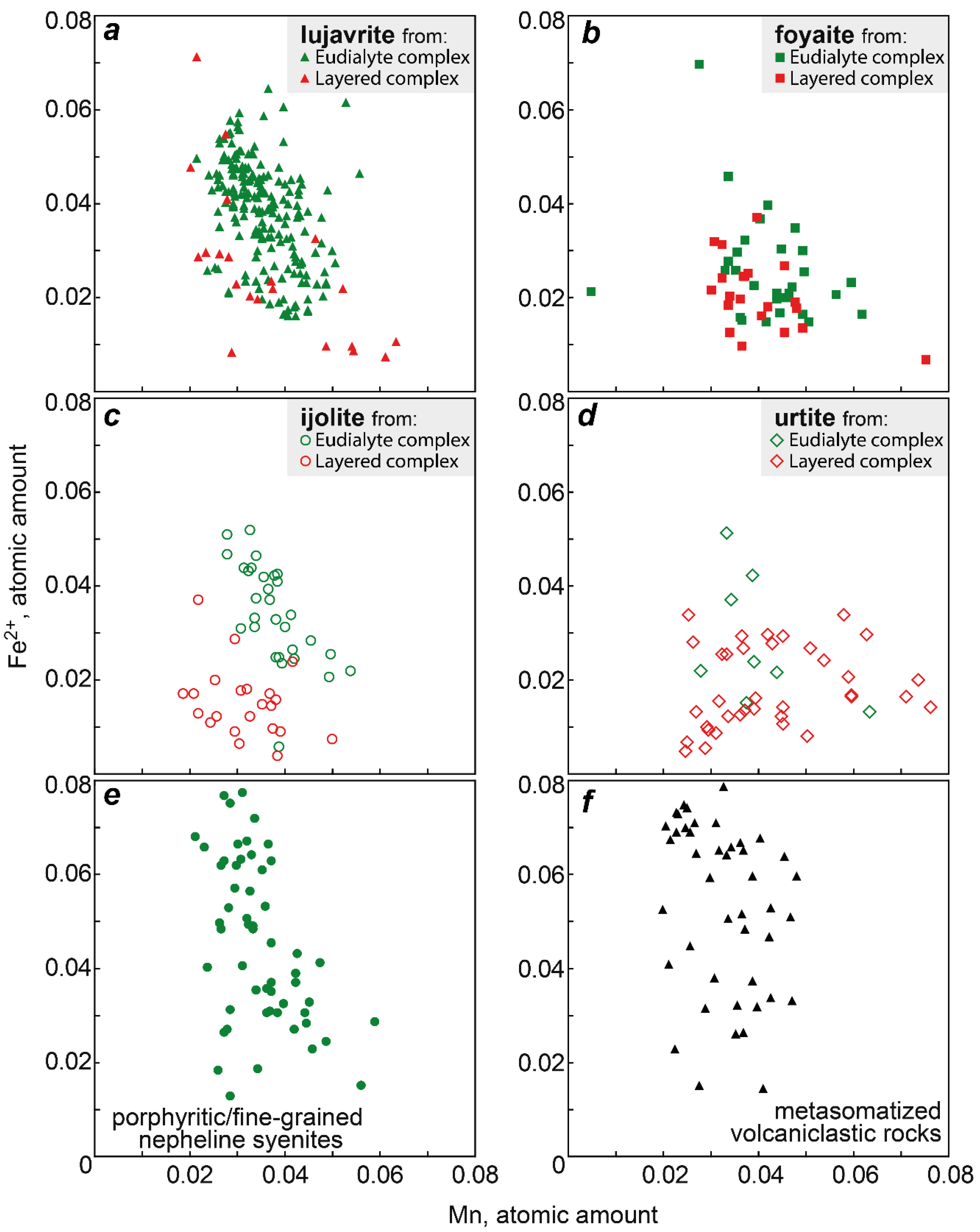
Figure 7 shows values of the Mn/Fe ratio for all types of the Lovozero massif rocks. Among the rocks of the Layered complex, the maximum Mn/Fe value is typical of urtite. Upwards the rhythm section, i.e., in the series urtite → (ijolite) → foyaite → lujavrite, this ratio is gradually decreasing. The same decrease occurs in the rocks of the Eudialyte complex, except for an increase in the Mn/Fe median value in foyaite. Yet, the Mn/Fe values in the rocks of the Eudialyte complex are generally lower. The median values of Mn/Fe in the series from urtite to lujavrite in the Layered complex decrease from 2.77 to 1.50, and in the similar series of rocks of the Eudialyte complex, from 1.27 to 0.90. The minimum Mn/Fe median ratio is in porphyritic/fine-grained nepheline syenites. Lesser values were recorded only for the EGM from metasomatized volcaniclastic rocks. The median value of Mn/Fe in poikilitic foid syenites is low and comparable with urtite and foyaite of the Layered complex. The Mn/Fe ratio decreases from the Layered complex to the Eudialyte complex mainly due to an increase in the amount of iron (Figure 8). Fe and Mn do not follow a strictly negative 1:1 correlation, and the decrease of Fe and the increase of Mn occur to variable degrees.
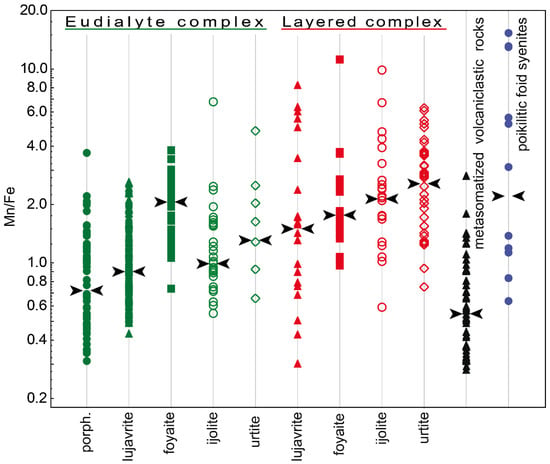
Figure 7.
The Mn/Fe ratio in the EGM from rocks of the Lovozero massif. Black arrows show the median contents. porph.—porphyritic/fine-grained nepheline syenites.
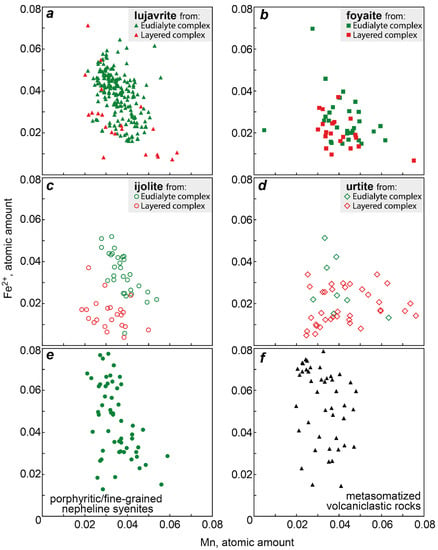
Figure 8.
Atomic amount Fe2+ vs atomic amount Mn in the EGM from different rocks of the Lovozero massif. (a) Fe2+ vs Mn in the EGM from lujavrite; (b) Fe2+ vs Mn in the EGM from foyaite; (c) Fe2+ vs Mn in the EGM from ijolite; (d) Fe2+ vs Mn in the EGM from urtite; (e) Fe2+ vs Mn in the EGM from porphyritic/fine-grained nepheline syenites; (f) Fe2+ vs Mn in the EGM from metasomatized volcaniclastic rocks.
4.3. Secondary Substitutions of the EGM
The EGM of the Lovozero massif rocks often undergo various secondary changes. Common products of the EGM change are the lovozerite-group minerals (Figure 9). They form porous rims around the EGM grains and sometimes complete pseudomorphoses. Exceedingly small grains of lueschite or rhabdophan-(Ce) are found in intergrowths with minerals of the lovozerite group. Lueschite and rhabdophan-(Ce) often fill thin cracks (Figure 9b–d). Minerals of the lovozerite group, which are formed as a result of the EGM substitution, usually contain an admixture of rare earth elements. This (in addition to intergrowths with lueschite or rhabdophan-(Ce)) distinguishes them from the primary lovozerite-group minerals, which are found in local areas in foyaite and porphyritic/fine-grained nepheline syenites of the Eudialyte complex. The EGM from melanocratic rocks (lujavrite and ijolite) are replaced by litvinskite, sometimes in association with tisinalite (Figure 9a,b, Table 5), and the EGM from leucocratic rocks (urtite and foyaite) are usually replaced by kapustinite (Figure 9c,d, Table 5). Moreover, the EGM grains at the periphery are often surrounded by a loose aggregate consisting of small grains of undiagnosed phases, including Na, Zr, Ti, Nb, Si, Mn, and REE in different ratios. Small grains of natrolite, rhabdophan-(Ce), fluorite, barite, pyrite, lueshite, loparite-(Ce), rutile, and manganese oxides are mixed with these phases. An important fact is that even with the complete destruction or replacement of the EGM, the natrolitization of adjacent nepheline and albite is weakly occurred. At the same time, EGM is completely preserved unchanged in intensely natrolitized rocks, e.g., in urtite.


Table 5.
Representative microprobe analyses of the lovozerite-group minerals, wt.%.
4.4. Associated Minerals: Clinopyroxenes
In the meso- and melanocratic rocks (lujavrite and ijolite) of the Layered and Eudialyte complexes of the Lovozero massif, the minerals of the eudialyte group are in close association with pyroxenes (aegirine and aegirine-augite). For example, in eudialyte lujavrite, the length of common boundaries between the EGM and clinopyroxene grains significantly exceeds the one between the EGM and leucocratic minerals (9:1 ratio) [53]. Therefore, the composition of coexisting pyroxenes is important for establishing crystallization conditions for the minerals of the eudialyte group.
The composition of clinopyroxenes from the rocks of the Eudialyte complex is presented by us in the article [53]. In these rocks, clinopyroxenes are presented by aegirine and aegirine-augite with high content of Ti (up to 0.16 apfu), Zr (up to 0.05 apfu), Al (up to 0.05 apfu), and Mn (up to 0.04 apfu). The main isomorphic scheme in clinopyroxene from the Eudialyte complex is as follows [53]:
Na+ + Ti4+ + (Al, Fe)3+ ⇄ Ca2+ + Mg2+ + Zr4+
Representative analyses of clinopyroxenes from the Layered complex are given in Table 6. Complete data on the composition of clinopyroxenes from rocks of the Layered complex are given in Supplementary Table S3. According to the results of factor analysis (Figure 10), the main scheme of isomorphism in clinopyroxenes from rocks of the Layered complex is the same as in pyroxenes of the Eudialyte complex (Factor 1). However, the clinopyroxenes from the Layered complex differ in the content of Ca, Mg, Zr, and Mn. A higher content of these elements is in clinopyroxenes from lujavrite and ijolite. The content of sodium and ferric iron is greater in clinopyroxenes from urtite (Figure 10).

Table 6.
Representative microprobe analyses of clinopyroxenes from the Layered complex, wt.%.
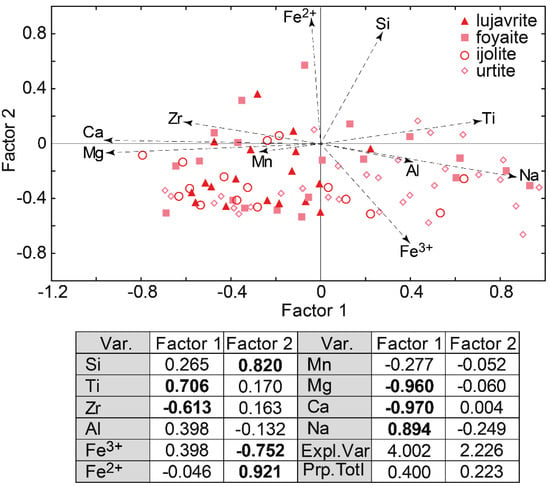
Figure 10.
Results of factor analyses of data on the composition (in apfu) of clinopyroxenes from the Layered complex of the Lovozero massif. Var.—variables; Expl.var.—Explained variance; Prp.totl—proportion of total variance. Factor loadings > |0.5| are shown in bold.
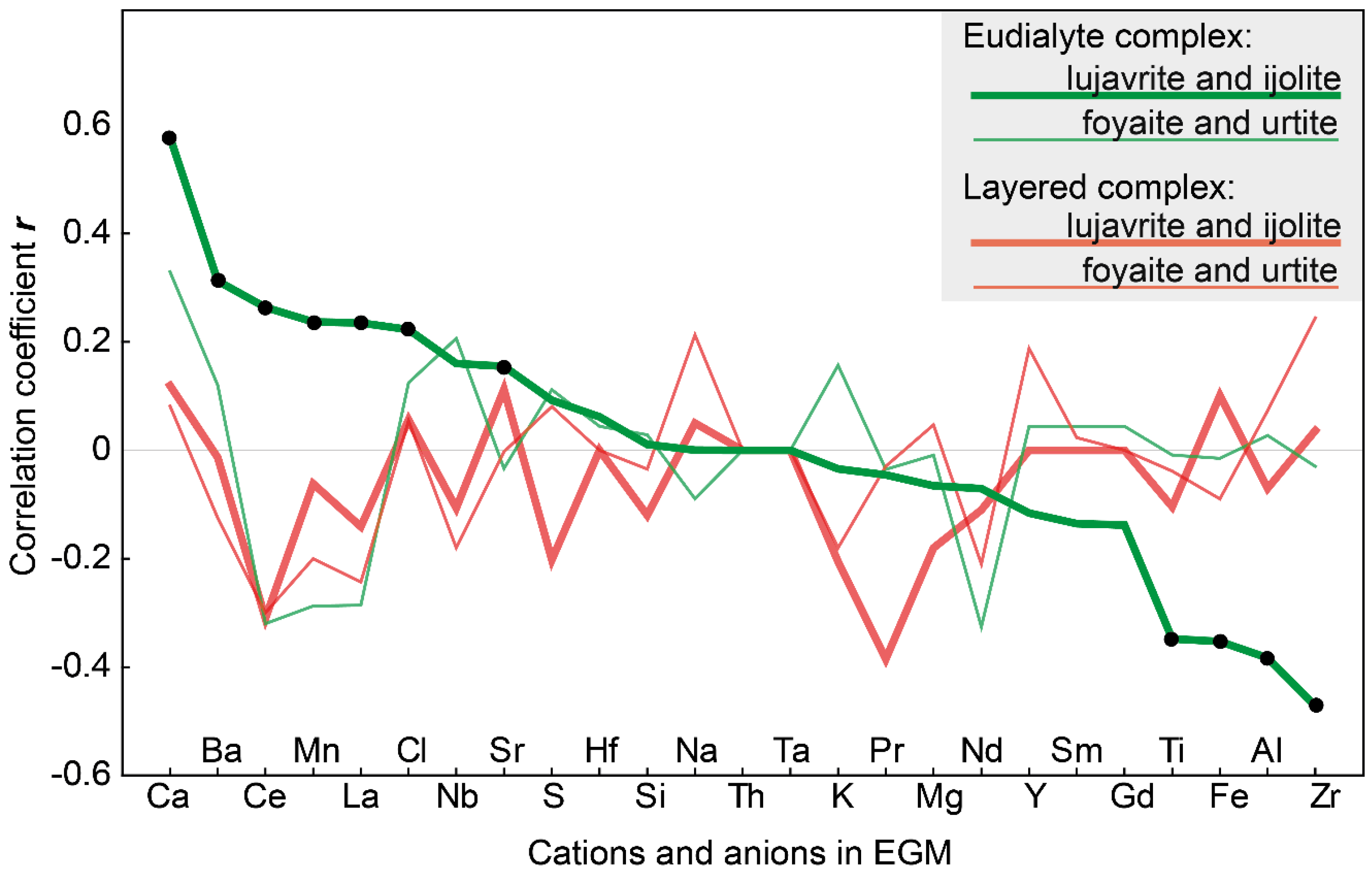
Figure 11 shows the correlation coefficients (r) between the magnesium content in clinopyroxenes (in apfu) and the contents of different cations and anions (in atomic amounts) in the coexisting EGM. It turned out that in the melanocratic rocks (eudialyte lujavrite and ijolite) of the Eudialyte complex, the compositions of pyroxenes and EGM are interconnected. Significant correlations (p ≤ 0.05) are between the magnesium content in pyroxenes (MgPx) and the content of 11 elements in the EGM. As the content of Mg (as well as Ca and Zr in accordance with isomorphic scheme) in clinopyroxene increases, the content of Mn, Ca, Sr, Ba, La, Ce, and Cl increases in the composition of the coexisting EGM, while the content of Fe2+, Zr, Ti, and Al decreases. The EGM grains in these rocks are oscillatory zonal, and Figure 11 shows the correlations between the composition of the cores of the EGM grains and the composition of pyroxenes. There are no significant correlations between the composition of the rim of the EGM grains and the composition of coexisting clinopyroxenes.
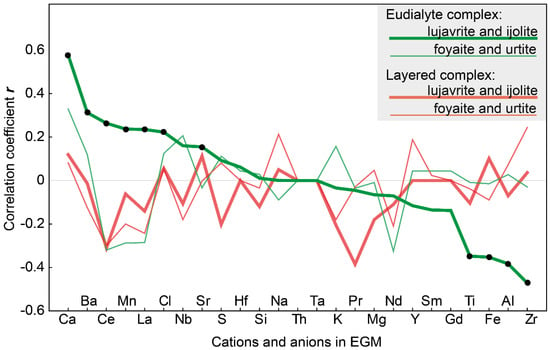
Figure 11.
Correlation coefficient (r) between the contents of Mg in clinopyroxene and elements in coexisting eudialyte-group minerals. Black dots indicate significant (p ≤ 0.05) correlation coefficients. The cations and anions in the EGM are placed in accordance with a decrease in the correlation coefficient between the composition of EGM and the MgPx from lujavrite and ijolite of the Eudialyte complex.
It is important to note that, in all types of rock of the layered complex and leucocratic rocks (foyaite, urtite) of the Eudialyte complex, there are no significant correlations between the compositions of pyroxenes and EGM (Figure 11).
5. Discussion
When a database consisting of 487 samples of the EGM from rocks of the Lovozero massif was formed, the problem of calculating the EGM formulas appeared. It turned out that the calculation method based on Si + Al + Ti + Zr + Nb + Hf + W + Ta = 29 [34] is not always suitable for the EGM from the Lovozero massif. In 30% of the analyses, the amount of zirconium does not exceed 3 Zr pfu, but, in the remaining 70%, there is excess zirconium (Table 2 and Table 3), which is included in the M(2B) site [35,47,48]. The basis Si + Al + Ti + Zr + Nb + Hf + W + Ta = 29 does not provide for the possibility of zirconium entering the M(2) position and cannot be used for the hyperzirconium samples. The apfu values obtained by this calculation method can differ significantly from the apfu calculated based on structural data (Table 3). Other methods for calculating the EGM formula are not suitable for large number of samples, since they require solving the crystal structure or other additional studies [35]. The rocks of the Lovozero massif were formed under narrow range of physical and chemical conditions, and even minor changes in the chemical composition of rock-forming minerals are important for establishing their genesis. That is why the best approach to studying the evolution of the Lovozero massif using indicator minerals is a statistical analysis of a large number of data [51,54].
The coefficients in the formula of any mineral are normalized values of atomic amounts of elements. To trace the patterns of changes in the composition of a mineral, e.g., the eudialyte group, it is possible to analyze changes in the ratios of atomic amounts. We suggest that this approach can be used for other minerals of complex composition.
The EGM are a characteristic accessory and sometimes rock-forming mineral in most types of rocks of the Lovozero alkaline massif. Petrographic data indicate that the EGM in lujavrite of the Layered complex crystallized in the late magmatic stage after alkaline clinopyroxenes and amphiboles and filled the interstices in the aggregate of their prismatic crystals (Figure 3e,f). It is important to note that Kogarko [55] proposed a gradual transition from melt to hydrothermal fluid in agpaitic systems. Therefore, in the studied rocks, it is impossible to accurately separate the late magmatic and hydrothermal mineral associations.
In foyaite and urtite, the content of mafic minerals decreases, clinopyroxenes, and amphiboles form large poikilitic crystals (Figure 3a), and the spatial connection between the EGM and mafic minerals disappears. During the transition from lujavrite to foyaite and urtite, the modal content of the EGM increases [29]. In foyaite and urtite, the EGM also crystallized at a late magmatic stage in the interstices of rock-forming minerals (Figure 3a,b,d). For example, in urtite, the EGM crystallized later than fluorapatite (Figure 3b), which is a late magmatic mineral [29].
Data on the composition of clinopyroxenes allowed us to additionally confirm petrographic conclusions about the relative crystallization time of the EGM. We suggested that, in rocks of the Layered complex, the eudialyte-group minerals crystallized later than clinopyroxenes because clinopyroxenes contain a significant amount of zirconium [56]. Following [56], zirconium is incorporated into the clinopyroxene structure, as an Na(Fe2+,Mg)0.5Zr0.5Si2O6 end-member, only when no other zirconium-bearing phase such as eudialyte is formed.
The question about the relative time of the EGM crystallization in meso- and melanocratic coarse-grained rocks (eudialyte lujavrite and ijolite) of the Eudialyte complex is difficult to solve only by petrographic observations. The EGM in these rocks form round or oval grains that are situated in the axial parts of aggregates (“streams”) of mafic minerals (Figure 4b). The rock-forming aegirine-(augite) in these rocks is also enriched in zirconium [49,51], but its content is lower than in the rocks of the Layered complex [49]. Larsen [57] suggested that “As long as eudialyte does not precipitate, Zr in the melt (and in the pyroxene) must rise with differentiation, but the onset of crystallization of eudialyte (with 12–15% ZrO2) in sufficient amounts will probably cause a fall in the Zr-content of both melt and pyroxenes”. However, rocks of the Eudialyte complex are more evolved then rocks of the Layered complex, and contain clinopyroxenes extremely enriched in the aegirine component [51]. After [58], Zr abundances are mainly controlled by the major element composition of the host clinopyroxene crystal, which, in turn, determines the crystal site parameters. Thus, the zirconium content in the clinopyroxenes of the Eudialyte complex is decreased due to the crystal-chemical effect. This decrease is apparently not related to the crystallization of EGM.
Yet, data on the composition of clinopyroxenes can still help to establish the time of eudialyte crystallization. In the eudialyte lujavrite and ijolite of the Eudialyte complex, the compositions of clinopyroxenes and EGM correlate with each other (Figure 11), and we assume that this relation is the result of simultaneous crystallization. The crystallization of EGM in these rocks was completed after the formation of clinopyroxenes. In the leucocratic rocks (foyaite and urtite) of the Eudialyte complex, there is no such relationship between the compositions of minerals.
In foyaite and urtite of the Eudialyte complex, the EGM crystallized at a late magmatic stage along with other zirconosilicates (minerals of the lovozerite group) in the interstices of rock-forming minerals. The texture of porphyritic/fine-grained nepheline syenites (Figure 4d,e) suggests that, in these rocks, the minerals of the eudialyte group crystallized simultaneously with clinopyroxenes. The composition of these rocks corresponds to the phonolitic eutectic, and they are one of the late and low-temperature igneous rocks of the Eudialyte complex [53]. Thus, in igneous rocks of different composition and texture, the minerals of the eudialyte group crystallized at a late magmatic stage, either immediately after clinopyroxenes (aegirine and aegirine-augite) or almost simultaneously with them.
The Mn/Fe ratio of eudialytes has been suggested as a monitor for magmatic evolution by various authors [13,59,60,61]. In general, the Mn/Fe ratio of both melt and EGM increase with fractionation. This ratio is controlled by (1) the fractionation stage and composition of the coexisting melt, (2) the partition coefficients of Fe and Mn for eudialyte-melt, and (3) co-crystallizing minerals [57]. The latter point implies that the EGM incorporate elements that are left after the crystallization of simultaneously and previously crystallizing Mn- and Fe-incorporating minerals. The Mn/Fe ratio in the EGM from all types of rocks varies very widely. In general, this ratio increases from lujavrite to urtite (i.e., from mela- and mesocratic to leucocratic rock) within both Eudialyte and Layered complexes. Moreover, the Mn/Fe ratio increases from the Eudialyte complex to the Layered complex (Figure 7), and this change occurs due to a decrease in the iron content (Figure 8).
A change in the Mn/Fe ratio from lujavrite to urtite of the Layered complex (Figure 7) indicates that lujavrite is the earliest rock of each rhythm, and urtite is the more evolved and latest one. The composition of clinopyroxenes can confirm it. Aegirine and aegirine-augite are found in all rocks of the Lovozero massif [51] and, therefore, are most suitable for tracing the physico-chemical evolution of the Lovozero magmas. The evolution from diopside-rich pyroxene compositions towards aegirine-rich pyroxene is typical of alkaline massifs worldwide [57]. The major difference between various massifs is the amount of Fe2+ enrichment relative to Na and Fe3+ enrichment during their evolution. Three evolutionary paths were documented: (1) from diopside to aegirine without significant Fe2+ enrichment (e.g., Katzenbuckel, Germany [62]), (2) from diopside via hedenbergite to aegirine (e.g., Ilímaussaq, Greenland [57,63]), and (3) intermediate paths (e.g., Lovozero [64]).
The clinopyroxenes from rocks of the Layered complex differ by the content of Ca, Mg (diopside end-member), and Na, Fe3+ (aegirine end-member). According to the results of factor analysis (Figure 10), the higher concentration of Ca, Mg (and Zr) is in clinopyroxenes from lujavrite. The content of sodium and ferric iron is greater in clinopyroxenes from urtite, and foyaite and ijolite are in an intermediate position. Thus, changes in the compositions of clinopyroxenes and EGM indicate that each rhythm of the Layered complex consists of less- (lujavrite) and more-evolved (urtite) rocks.
The Eudialyte complex was formed later than the Layered one. It is confirmed by geological observations [30], geochemical evidence (extremely high concentration of HFSE and volatile components, high (Na+K)/Al ratio [29]), as well as a change in the composition of rock-forming minerals. Kogarko and co-authors [51] described the composition of clinopyroxene throughout the whole vertical section (2.5 km thick) of the Lovozero massif. They found that pyroxenes from the rocks of the Eudialyte complex are more enriched in sodium and ferric iron compared to pyroxenes from the Layered complex. However, the Mn/Fe ratio in the EGM from rocks of the Eudialyte complex is significantly lower (Figure 7) due to the high iron content (Figure 8). The maximum iron contents and, accordingly, the minimum Mn/Fe values are typical of the EGM, which crystallized simultaneously with clinopyroxenes. These are eudialyte-group minerals from eudialyte lujavrite, ijolite, and porphyritic/fine-grained nepheline syenites. According to Markl and co-authors [65], crystallization of aegirine or arfvedsonite from a melt must involve the reduction of the melt according to the two following two reactions:
2Na2O + 4FeO + 8SiO2 + O2 (in the melt) = 4NaFeSi2O6 (aegirine)
3Na2O + 10FeO + 16SiO2 + O2 + H2O (in the melt) = 2Na3Fe2+4Fe3+Si8O22(OH)2 (arfvedsonite)
The crystallization of aegirine leads to an increase in the content of ferrous iron in the mineral-forming melt/solution, and this iron enters the EGM, which grow simultaneously with aegirine.
The increase of the EGM content in leucocratic rocks (foyaite, urtite) of the Layered complex and the appearance of primary minerals of the lovozerite group together with the EGM in the leucocratic rocks of the Eudialyte complex can be explained as follows. The lujavrite → foyaite → urtite fractionation path started with potassic alkali feldspar, clinopyroxene, and nepheline, whose removal drove the melts towards HFSE- and Na-rich compositions, then the EGM started to crystallize. A similar fractionation path occurs in the Na2O-Al2O3-Fe2O3-SiO2 system [66], where a melt of the “ijolite” type (approximately 50% of aegirine) evolves towards “nepheline syenite” (approximately 10% of aegirine). Following [66], after the crystallization of lujavrite, the residual foyaitic melt was significantly enriched in sodium disilicate. Thus, in the rhythm of the Layered complex, the higher EGM content in foyaite and urtite is a result of fractional crystallization.
It is important to note that the zirconosilicates of the eudialyte- and the lovozerite groups differ by sodium content and crystallize at different alkalinity. According to Khomyakov [67], for silicate minerals with the general formula AxMySipOq (A = Na, K, and other strong bases; M = Zr, Nb, Ti, Be, and other Al-substituting elements), the so-called alkalinity modulus can be calculated: Kalk = (x × 100)/(x + y + p). By the value of Kalk, the minerals and rocks, in which they are present, are subdivided into five groups, namely miaskitic (Kalk ≪ 15%), low agpaitic (Kalk = 15–25%), medium agpaitic (Kalk = 25–35%), highly agpaitic (Kalk = 35–40%), and hyperagpaitic (Kalk > 40%). Minerals of the lovozerite group are characterized by the highest alkalinity, and belong to the hyperagpaitic group, and the Kalk of the minerals of the eudialyte group is lower (highly agpaitic group). The melt evolutionary paths during crystallization of the Eudialyte and Layered complexes were similar, i.e., melanocratic melt evolved towards leucocratic (foyaitic) melt extremely enriched with sodium disilicate. However, during the crystallization of the rocks of the Eudialyte complex, the proportion of sodium in the residual melt was greater than during the crystallization of the rhythm (from lujavrite to foyaite and urtite) of the Layered complex. As a result, primary hyperagpaitic minerals (lovozerite, kapustinite, litvinskite) crystallized in the leucocratic rocks of the Eudialyte complex.
The EGM decomposition products are indicative of an increase in sodium concentration from early melanocratic rocks (lujavrite) to late foyaite and urtite. Substitution of the EGM by the lovozerite-group minerals occurs at the post-magmatic stage under the influence of residual solutions. The alkali concentration in these solutions determines which of the lovoserite-group minerals will be formed. In lujavrite and ijolite, eudialyte is replaced by litvinskite, and in foyaite and other leucocratic rocks, it is replaced by kapustinite. Late hydrothermal solutions may not affect eudialyte. Under their influence, only previously formed rims of the secondary lovozerite-group minerals are destroyed.
The eudialyte-group minerals are formed during the metasomatic alteration of volcanoclastic rocks. Unchanged, these rocks contain extremely low concentrations of ZrO2 (maximum 0.01 wt.% for basalt) and REE (Σ REE2O3 = 300 ppm in average for basalt), but are significantly enriched in CaO (10.18 wt.% in average for basalt), MgO (6.24 wt.% in average for basalt), and FeO (9.43 wt.% in average for basalt) [68]. The EGM from metasomatites are quite different in composition to the EGM from igneous rocks, since they are formed during the replacement of calcium-enriched rocks. The arrangement of zones in the oscillatory zoned EGM grains from metasomatites is opposite to the arrangement of zones in the magmatic EGM, and indicates an increase in the concentration of Zr, Al, Na, Fe2+, and Cl during metasomatism.
6. Conclusions
- The minerals of eudialyte group (EGM) are a characteristic accessory and sometimes rock-forming minerals of the Lovozero alkaline massif rocks. In all types of the rocks, they form at the late magmatic stage. In the rocks of the Layered complex, the EGM crystallize later than alkaline clinopyroxenes and amphiboles, but in the Eudialyte complex, crystallization of the EGM begins simultaneously with clinopyroxenes and ends after their formation.
- In the Layered complex, the maximum modal content of EGM is found in leucocratic rocks (foyaite and urtite). In the Eudialyte complex, the largest amount of EGM is concentrated in melanocratic rocks (lujavrite), while leucocratic rocks contain primary minerals of the lovozerite group. This distribution occurs due to fractional crystallization by the lujavrite → foyaite → urtite path, in which the sodium (and HFSE) concentration in the residual liquid progressively increases.
- Most of the EGM samples (70%) from the rocks of the Lovozero massif are hyperzirconium, i.e., their Zr content exceeds 3 apfu. Since the excess zirconium is included in the M(2B) position, the generally accepted formula calculation based on (Si + Zr + Ti + Nb + Al + Hf + W + Ta) = 29 cations cannot be applied. To trace changes in the composition of EGM during magmatic evolution, the ratio of the atomic amounts of cations can be used.
- In the lujavrite-foyaite-urtite series in both Layered and Eudialyte complexes, the Mn/Fe ratio in the EGM increases. However, the Mn/Fe ratio in the EGM from rocks of the Eudialyte complex is significantly lower due to the high iron content. The maximum iron contents and, accordingly, the minimum Mn/Fe values are typical of the EGM, which crystallized simultaneously with clinopyroxenes (aegirine and aegirine-augite). The reason for this is that, during the crystallization of alkali clinopyroxenes in the melt/solution, the concentration of ferrous iron sharply increases.
- At the post-magmatic stage, the EGM are replaced by minerals of the lovozerite group. Which mineral from this group will replace the EGM depends on the alkalinity of the residual solution. The EGM from melanocratic rocks (lujavrite and ijolite) are replaced by litvinskite, and the EGM from leucocratic rocks (urtite and foyaite) are usually replaced by kapustinite.
Supplementary Materials
The following materials are available online at https://www.mdpi.com/2075-163X/10/12/1070/s1, Table S1: Parameters of microprobe analyses, Table S2: Microprobe analyses of EGM, Table S3: Microprobe analyses of clinopyroxenes.
Author Contributions
J.A.M. designed the experiments, participated in field works, performed statistical investigations, and wrote the manuscript. T.L.P. participated in fieldworks, performed geostatistical investigation, drew maps, and reviewed the manuscript. Y.A.P. and A.V.B. took BSE-images and performed electron microscope investigations. V.N.Y. conceived of the work, participated in field works, and reviewed the manuscript. All authors discussed the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The Kola Science Center of Russian Academy of Sciences (0226-2019-0051) funded this research.
Acknowledgments
We are grateful to anonymous reviewers from MDPI who helped us improve the presentation of our results.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Stromeyer, F. Summary of meeting 16 December 1819. Göttingische Gelehrte Anz. 1819, 3, 1993–2000. [Google Scholar]
- Rastsvetaeva, R.K. Structural mineralogy of the eudialyte group: A review. Crystallogr. Rep. 2007, 52, 47–64. [Google Scholar] [CrossRef]
- Davris, P.; Stopic, S.; Balomenos, E.; Panias, D.; Paspaliaris, I.; Friedrich, B. Leaching of rare earth elements from eudialyte concentrate by suppressing silica gel formation. Miner. Eng. 2017, 108, 115–122. [Google Scholar] [CrossRef]
- Lebedev, V.N. Sulfuric Acid Technology for Processing of Eudialyte Concentrate. Russ. J. Appl. Chem. 2003, 76, 1559–1563. [Google Scholar] [CrossRef]
- Schønwandt, H.K.; Barnes, G.B.; Ulrich, T. A Description of the World-Class Rare Earth Element Deposit, Tanbreez, South Greenland. Rare Earths Ind. 2016, 73–85. [Google Scholar] [CrossRef]
- McLemore, V.; Reservation, M.I. Background and perspectives on the Pajarito Mountain yttrium-zirconium deposit, Mescalero Apache Indian Reseruation, Otero Gounty, New Mexico. Am. Mineral. 1990, 72, 801–811. [Google Scholar]
- Kogarko, L.N. Ore-forming potential of alkaline magmas. Lithos 1990, 26, 167–175. [Google Scholar] [CrossRef]
- Sørrensen, H. Agpaitic nepheline syenites: A potential source of rare elements. Appl. Geochem. 1992, 7, 417–427. [Google Scholar] [CrossRef]
- Schilling, J.; Marks, M.A.W.; Wenzel, T.; Vennemann, T.; Horváth, L.; Tarassoff, P.; Jacob, D.E.; Markl, G. The Magmatic to Hydrothermal Evolution of the Intrusive Mont Saint-Hilaire Complex: Insights into the Late-stage Evolution of Peralkaline Rocks. J. Pet. 2011, 52, 2147–2185. [Google Scholar] [CrossRef]
- Sjöqvist, A.S.; Cornell, D.; Andersen, T.B.; Erambert, M.; Ek, M.; Leijd, M. Three Compositional Varieties of Rare-Earth Element Ore: Eudialyte-Group Minerals from the Norra Kärr Alkaline Complex, Southern Sweden. Minerals 2013, 3, 94–120. [Google Scholar] [CrossRef]
- Olivo, G.R.; Williams-Jones, A.E. Hydrothermal REE-rich eudialyte from the Pilanesberg complex, South Africa. Can. Mineral. 1999, 37, 653–663. [Google Scholar]
- Mitchell, R.H.; Liferovich, R.P. Subsolidus deuteric/hydrothermal alteration of eudialyte in lujavrite from the Pilansberg alkaline complex, South Africa. Lithos 2006, 91, 352–372. [Google Scholar] [CrossRef]
- Schilling, J.; Marks, M.A.; Wenzel, T.; Märkl, G. Reconstruction of magmatic to subsolidus processes in an agpaitic system using eudialyte textures and composition: A case study from Tamazeght, Morocco. Can. Mineral. 2009, 47, 351–365. [Google Scholar] [CrossRef]
- Ratschbacher, B.C.; Marks, M.A.; Bons, P.D.; Wenzel, T.; Markl, G. Emplacement and geochemical evolution of highly evolved syenites investigated by a combined structural and geochemical field study: The lujavrites of the Ilímaussaq complex, SW Greenland. Lithos 2015, 231, 62–76. [Google Scholar] [CrossRef]
- Atanasova, P.; Marks, M.A.W.; Heinig, T.; Krause, J.; Gutzmer, J.; Markl, G. Distinguishing Magmatic and Metamorphic Processes in Peralkaline Rocks of the Norra Kärr Complex (Southern Sweden) Using Textural and Compositional Variations of Clinopyroxene and Eudialyte-group Minerals. J. Pet. 2017, 58, 361–384. [Google Scholar] [CrossRef]
- Borst, A.M.; Friis, H.; Andersen, T.; Nielsen, T.F.D.; Waight, T.E.; Smit, M.A. Zirconosilicates in the kakortokites of the Ilímaussaq complex, South Greenland: Implications for fluid evolution and high-field-strength and rare-earth element mineralization in agpaitic systems. Mineral. Mag. 2016, 80, 5–30. [Google Scholar] [CrossRef]
- Borst, A.M.; Friis, H.; Nielsen, T.F.D.; Waight, T.E. Bulk and Mush Melt Evolution in Agpaitic Intrusions: Insights from Compositional Zoning in Eudialyte, Ilímaussaq Complex, South Greenland. J. Pet. 2018, 59, 589–612. [Google Scholar] [CrossRef]
- Sørensen, H. The agpaitic rocks—An overview. Mineral. Mag. 1997, 61, 485–498. [Google Scholar] [CrossRef]
- Kogarko, L.N.; Lazutkina, L.N.; Romanchev, B.P. The origin of eudialyte mineralization. Geochem. Int. 1982, 19, 128–145. [Google Scholar]
- Estrade, G.; Salvi, S.; Béziat, D.; Estrade, G. Crystallization and destabilization of eudialyte-group minerals in peralkaline granite and pegmatite: A case study from the Ambohimirahavavy complex, Madagascar. Mineral. Mag. 2018, 82, 375–399. [Google Scholar] [CrossRef]
- Harris, C.; Rickard, R.S. Rare-earth-rich eudialyte and dalyite from a peralkaline granite dyke at Straumsvola, Dronning Maud Land, Antarctica. Can. Mineral. 1987, 25, 755–762. [Google Scholar]
- Marks, M.A.; Hettmann, K.; Schilling, J.; Frost, B.R.; Markl, G. The Mineralogical Diversity of Alkaline Igneous Rocks: Critical Factors for the Transition from Miaskitic to Agpaitic Phase Assemblages. J. Pet. 2011, 52, 439–455. [Google Scholar] [CrossRef]
- Marks, M.A.; Markl, G. A global review on agpaitic rocks. Earth Sci. Rev. 2017, 173, 229–258. [Google Scholar] [CrossRef]
- Chukanov, N.V.; Pekov, I.V.; Zadov, A.E.; Korovushkin, V.V.; Ekimenkova, I.A.; Rastsvetaeva, R.K. Ikranite (Na, H3O)15 (Ca, Mn, REE)6 Fe23+ Zr3 (□, Zr)(□, Si) Si24 O66 (O, OH)6 Cl· nH2O and raslakite Na15 Ca3 Fe3 (Na, Zr)3 Zr3 (Si, Nb)(Si25 O73)(OH, H2O)3 (Cl, OH)—New eudialyte-group minerals from the Lovozero Massif. ZRMO 2003, 5, 22–33. [Google Scholar]
- Khomyakov, A.P.; Nechelyustov, G.N.; Rastsvetaeva, R.K. Dualite, Na30 (Ca,Na,Ce,Sr)12 (Na,Mn,Fe,Ti)6 Zr3 Ti3 Mn Si51 O144 (OH, H2O, Cl)9, a new zircono-titanosilicate with a modular eudialyte-like structure from the Lovozero alkaline Pluton, Kola Peninsula, Russia. Geol. Ore Depos. 2008, 50, 574–582. [Google Scholar] [CrossRef]
- Khomyakov, A.P.; Nechelyustov, G.N.; Rastsvetaeva, R.K. Voronkovite, Na15 (Na,Ca,Ce)3 (Mn,Ca)3 Fe3 Zr3 Si26 O72 (OH,O)4 Cl · H2O, a new mineral species of the eudialyte group from the Lovozero alkaline pluton, Kola Peninsula, Russia. Geol. Ore Depos. 2009, 51, 750–756. [Google Scholar] [CrossRef]
- Chukanov, N.V.; Aksenov, S.M.; Pekov, I.V.; Belakovskiy, D.I.; Vozchikova, S.A.; Britvin, S.N. Sergevanite, Na15 (Ca3Mn3) (Na2Fe) Zr3Si26O72 (OH)3 · H2O, a new eudialyte-group mineral from the Lovozero alkaline massif, Kola Peninsula. Can. Mineral. 2020, 58, 421–436. [Google Scholar] [CrossRef]
- Kramm, U.; Kogarko, L. Nd and Sr isotope signatures of the Khibina and Lovozero agpaitic centres, Kola Alkaline province, Russia. Lithos 1994, 32, 225–242. [Google Scholar] [CrossRef]
- Gerasimovsky, V.I.; Volkov, V.P.; Kogarko, L.N.; Polyakov, A.I.; Saprykina, T.V.; Balashov, Y.A. Geochemistry of the Lovozero Alkaline Massif; Nauka: Moscow, Russia, 1966. (In Russian) [Google Scholar]
- Bussen, I.V.; Sakharov, A.S. Petrology of the Lovozero Alkaline Massif; Nauka: Leningrad, Russia, 1972. (In Russian) [Google Scholar]
- StatSoft Inc. Statistica 13. Available online: www.statsoft.ru (accessed on 19 June 2018).
- Reyment, R.A.; Jvreskog, K.G. Applied Factor Analysis in the Natural Sciences; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Le Maitre, R.W. (Ed.) Igneous Rocks: A Classification and Glossary of Terms: Recommendations of the International Union of Geological Sciences Subcommission on the Systematics of Igneous Rocks; Cambridge University Press: Cambridge, UK, 2005; ISBN 9780521662154. [Google Scholar]
- Johnsen, O.; Grice, J.D. The crystal chemistry of the eudialyte group. Can. Mineral. 1999, 37, 865–891. [Google Scholar]
- Rastsvetaeva, R.K.; Chukanov, N.V.; Aksenov, S.M. Minerals of the Eudialyte Group: Crystal Chemistry, Properties, Genesis; NSU: Nizhny Novgorod, Russia, 2012. (In Russian) [Google Scholar]
- Golyshev, V.M.; Simonov, V.I.; Belov, N.V. About the crystal structure of eudialyte. Crystallography 1971, 16, 93–98. [Google Scholar]
- Giuseppetti, G.; Mazzi, F.; Tadini, C. The crystal structure of eudialyte. Mineral. Pet. 1971, 16, 105–127. [Google Scholar] [CrossRef]
- Ferraris, G.; Makovicky, E.; Merlino, S. Crystallography of Modular Materials; Oxford University Press: Oxford, UK, 2004. [Google Scholar] [CrossRef]
- Khomyakov, A.P.; Nechelyustov, G.N.; Rastsvetaeva, R.K. Alluaivite, Na19 (Ca,Mn)6 (Ti,Nb)3 Si26 O74 Cl · H2O, a new titanosilicate with a eudialyte-like structure. Zap. VMO 1990, 119, 117–120. (In Russian) [Google Scholar]
- Khomyakov, A.P.; Nechelyustov, G.N.; Rastvetaeva, R.K. Labyrinthite (Na,K,Sr)35 Ca12 Fe3 Zr6 Ti Si51 O144 (O,OH,H2O)9 Cl3, a new mineral with the modular eudialyte-like structure from Khibiny alkaline massif, Kola Peninsula. Zap. RMO 2006, 135, 38–48. (In Russian) [Google Scholar]
- Khomyakov, A.P.; Nechelyustov, G.N.; Arakcheeva, A.V. Rastsvetaevite, Na27 K8 Ca12 Fe3 Zr6 Si4 [Si3O9]4 [Si9O27]4 (O,OH,H2O)6 Cl2, a new mineral with a modular eudialyte-like structure and crystal-chemical systematics of the eudialyte group. Zap. RMO 2006, 135, 49–65. (In Russian) [Google Scholar]
- Osipov, A.S.; Antonov, A.A.; Panikorovskii, T.L.; Zolotarev, A.A. Hydrated CO3-Bearing Analog of Manganoeudialyte from Alkali Pegmatites of the Konder Pluton, Khabarovsk Krai. Geol. Ore Depos. 2018, 60, 726–735. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Chukanov, N.V. Classification of eudialyte-group minerals. Geol. Ore Depos. 2012, 54, 487–497. [Google Scholar] [CrossRef]
- Pfaff, K.; Wenzel, T.; Schilling, J.; Marks, M.A.; Markl, G. A fast and easy-to-use approach to cation site assignment for eudialyte-group minerals. Neues Jahrb. Mineral. Abh. J. Mineral. Geochem. 2010, 187, 69–81. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Khomyakov, A.P. Crystal structure of a hyperzirconium analogue of eudialyte. Crystallogr. Rep. 2000, 45, 219–221. [Google Scholar] [CrossRef]
- Aksenov, S.M.; Rastsvetaeva, R.K. Crystal-structure refinement of zirconium-rich eudialyte and its place among calcium-poor eudialyte-group minerals. Crystallogr. Rep. 2013, 58, 671–677. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Chukanov, N.V.; Verin, I.A. Crystal structure of hyperzirconium sulfate analogue of eudialyte. Dokl. Earth Sci. 2006, 409, 985–989. [Google Scholar] [CrossRef]
- Panikorovskii, T.L.; Mikhailova, J.A.; Pakhomovsky, Y.A.; Ivanyuk, G.Y. Crystal Chemistry of the Eudialyte Group Minerals from the Lovozero Eudialyte Complex, Kola Peninsula, Russia. Magmat. Earth Relat. Strateg. Met. Depos. 2019, 36, 221–223. [Google Scholar]
- Nivin, V. Free hydrogen-hydrocarbon gases from the Lovozero loparite deposit (Kola Peninsula, NW Russia). Appl. Geochem. 2016, 74, 44–55. [Google Scholar] [CrossRef]
- Nivin, V. Variations in the composition and origin of hydrocarbon gases from inclusions in minerals of the Khibiny and Lovozero plutons, Kola Peninsula, Russia. Geol. Ore Depos. 2011, 53, 699–707. [Google Scholar] [CrossRef]
- Kogarko, L.N.; Williams, C.T.; Woolley, A.R. Compositional evolution and cryptic variation in pyroxenes of the peralkaline Lovozero intrusion, Kola Peninsula, Russia. Mineral. Mag. 2006, 70, 347–359. [Google Scholar] [CrossRef]
- Khomyakov, A.P.; Korovushkin, V.V.; Perfiliev, Y.D.; Cherepanov, V.M. Location, valence states, and oxidation mechanisms of iron in eudialyte-group minerals from Mössbauer spectroscopy. Phys. Chem. Mineral. 2010, 37, 543–554. [Google Scholar] [CrossRef]
- Mikhailova, J.A.; Ivanyuk, G.Y.; Kalashnikov, A.O.; Pakhomovsky, Y.A.; Bazai, A.; Yakovenchuk, V.N. Petrogenesis of the Eudialyte Complex of the Lovozero Alkaline Massif (Kola Peninsula, Russia). Minerals 2019, 9, 581. [Google Scholar] [CrossRef]
- Williams, C.T.; Kogarko, L.N.; Woolley, A.R. Chemical evolution and petrogenetic implications of loparite in the layered, agpaitic Lovozero complex, Kola Peninsula, Russia. Mineral. Pet. 2002, 74, 1–24. [Google Scholar] [CrossRef]
- Kogarko, L.N. Problems of the Genesis of the Agpaitic Magmas; Nauka: Moscow, Russia, 1977. (In Russian) [Google Scholar]
- Jones, A.P.; Peckett, A. Zirconium-bearing aegirines from Motzfeldt, South Greenland. Contrib. Mineral. Pet. 1981, 75, 251–255. [Google Scholar] [CrossRef]
- Larsen, L.M. Clinopyroxenes and Coexisting Mafic Minerals from the Alkaline Ilimaussaq Intrusion, South Greenland. J. Pet. 1976, 17, 258–290. [Google Scholar] [CrossRef]
- Marks, M.; Halama, R.; Wenzel, T.; Markl, G. Trace element variations in clinopyroxene and amphibole from alkaline to peralkaline syenites and granites: Implications for mineral–melt trace-element partitioning. Chem. Geol. 2004, 211, 185–215. [Google Scholar] [CrossRef]
- Johnsen, O.; Gault, R.A. Chemical variation in eudialyte. Neues Jahrb. Mineral. 1997, 171, 215–237. [Google Scholar]
- Schilling, J.; Wu, F.-Y.; McCammon, C.; Wenzel, T.; Marks, M.A.W.; Pfaff, K.; Jacob, D.E.; Markl, G. The compositional variability of eudialyte-group minerals. Mineral. Mag. 2011, 75, 87–115. [Google Scholar] [CrossRef]
- Pfaff, K.; Krumrei, T.; Marks, M.; Wenzel, T.; Rudolf, T.; Markl, G. Chemical and physical evolution of the ‘lower layered sequence’ from the nepheline syenitic Ilímaussaq intrusion, South Greenland: Implications for the origin of magmatic layering in peralkaline felsic liquids. Lithos 2008, 106, 280–296. [Google Scholar] [CrossRef]
- Mann, U.; Marks, M.; Markl, G. Influence of oxygen fugacity on mineral compositions in peralkaline melts: The Katzenbuckel volcano, Southwest Germany. Lithos 2006, 91, 262–285. [Google Scholar] [CrossRef]
- Marks, M.; Markl, G. Fractionation and Assimilation Processes in the Alkaline Augite Syenite Unit of the Ilímaussaq Intrusion, South Greenland, as Deduced from Phase Equilibria. J. Pet. 2001, 42, 1947–1969. [Google Scholar] [CrossRef]
- Korobeinikov, A.N.; Laajoki, K. Petrological aspects of the evolution of clinopyroxene composition in the intrusive rocks of the Lovozero alkaline massif. Geochem. Int. 1994, 31, 69–76. [Google Scholar]
- Markl, G.; Marks, M.A.; Frost, B.R. On the Controls of Oxygen Fugacity in the Generation and Crystallization of Peralkaline Melts. J. Pet. 2010, 51, 1831–1847. [Google Scholar] [CrossRef]
- Bailey, D.K.; Schairer, J.F. The System Na2O-Al2O3-Fe2O3-SiO2 at 1 atmosphere, and the petrogenesis of alkaline rocks. J. Petrol. 1966, 7, 114–170. [Google Scholar] [CrossRef]
- Khomyakov, A.P. Mineralogy of Hyperagpaitic Alkaline Rocks; Oxford University Press: Oxford, MS, USA, 1995. [Google Scholar]
- Mikhailova, J.A.; Men’Shikov, Y.P.; Pakhomovskii, Y.A.; Yakovenchuk, V.N.; Ivanyuk, G.Y. Trap formation of the Kola peninsula. Petrology 2011, 19, 87–101. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).