Abstract
The mineral surface structure and ions’ interaction were of significant interest to understanding mineral dissolution and reaction. In this study, X-ray photoemission spectroscopy combined with ICP emission spectrometer was used to investigate the influence of the leaching reaction conditions of 8 M dilute sulfuric acid and 12 M concentrated sulfuric acid on the surface chemical composition, chemical (valence) state and ion distribution of Columbite-(Fe) (FeNb2O6). The binding energy of the cations (Fe, Nb) bonding with different anions (O2−, SO42−) and the ratio of Fe3+/Fe2+ oxidation–reduction provided direct understanding of Fe and Nb releasing from the mineral surface during leaching. The results showed that the binding energy of the Nb5+-O bond was much smaller than that of Nb5+-SO4, and the binding energy decreased in sequence as Nb5+-O < Fe2+-O < Fe3+-O and increased in sequence as Fe3+-SO4 < Fe2+-SO4 < Nb5+-SO4. The mineral surface reaction during the leaching could be expressed with the formula: Fe-O + H2SO4 → Fe-SO4 + H2O, Nb-O + H2SO4 → Nb-SO4 + H2O. The results also revealed that Nb dissolution from Columbite-(Fe) occurred more easily compared to Fe. Nb dissolution from the mineral was owed to the content of H+ in solution, and increasing the H+ concentration could promote the dissolution. For Fe dissolution from the mineral, the oxidation potential could play an effective role in enhancement dissolution.
1. Introduction
Nb and Ta were often labeled as strategic raw materials which were considered critical materials utilized widely in the steel, electronic, high-tech industries [1] and the defense, nuclear and chemical industries [2,3]. The columbite minerals ((Fe, Mn) (Ta, Nb)2O6) [4] group was the most common Nb-Ta species compound with other metal elements, which mainly included the members of columbite-(Fe) (FeNb2O6) and columbite-(Mn) (MnNb2O6) [5].
In industry, the main method for the extraction of niobium from columbite-(Fe) was the concentrated hydrofluoric acid (HF) leaching process; associated with this technology was the evaporation loss of HF. Rodriguez et al. [6] studied the extraction of niobium and tantalum from ferrocolumbite by hydrofluoric acid pressure leaching, which showed that, at the maximum extraction of Nb 90%, it achieved a HF concentration of 9% v/v, reaction temperature of 220 °C. Majima et al. [7] studied the dissolution of columbite and tantalite in acidic fluoride media; the results showed that the presence of both H+ and F in the leaching solution is necessary for the fast dissolution of columbite and tantalite. This method was also widely used for the decomposition of columbite–tantalite minerals and brought the environmental and operation problems [8]. The sulfuric acid process [9,10] was mainly used for composite ore which was easy to decompose. It could comprehensively recover valuable metal components in ore and had a high metal leaching conversion rate. Adding sulfuric acid on the basis of hydrofluoric acid could effectively inhibit the harmful effects of HF. Studies by He et al. [11] showed that sulfuric acid could reduce the partial pressure of HF, reduce volatilization loss, and in this way reduce the total acid consumption. Zhu et al. [12] pointed out that the sulfate in the extracts could be removed together with fluoride ions by precipitation with alkaline reagents (such as lime). In addition, the mixture of HF-H2SO4 also promoted the dissolution of Ta/Nb better than the HF acid leaching alone [13]. Sulfuric acid was attracted as an economic method to leach the low-grade columbite-tantalum bearing mineral; sulfuric acid (60%) was used to decompose manganotantalite at 200 °C and 1.5 MPa for 2 h, and only 8.5% and 9.9% tantalum and niobium were leached, respectively [14]. Nitric acid was added to increase the oxidation potential during leaching which could improve the leaching extent obviously [15]. In our previous work, concentrated sulfuric acid was used to decompose columbite minerals, and the result indicated that there was nearly 10% niobium still in residue [16]. How to improve the niobium dissolving from the mineral was a key mission for metallurgical researchers. The information about columbite-(Fe) mineral surface structure, reaction and dissolution occurring in sulfuric acid leaching systems was essential to promote niobium leaching efficiency.
The mineral/aqueous interfaces’ structure and ions’ interaction were of significant interest to understanding the mineral dissolution and reaction. Researchers made great efforts to modify the columbite-(Fe) mineral surface structure which could improve the utilization of the limited niobium resources and niobium leaching from the mineral [17,18]. Yang et al. [19] studied the interfacial structure change and selective dissolution of the columbite-(Fe) mineral during HF acid leaching; the results showed that under this acid leaching condition, the leaching rate of Fe and Nb elements increased with the increase in the HF concentration, and Nb atoms were easier than Fe atoms to dissolve. However, there was less knowledge about dissolution based on the surface structure and surface reaction during leaching, and on the bond breakage and metals dissolution sequence occurring at the columbite surface. The aim of this paper was to study the columbite-(Fe) mineral surface structure and structural control reaction and dissolution in sulfuric acid leaching, and to improve the knowledge of governing columbite/fluid interface leaching. The X-ray spectroscopic method was carried out to investigate the columbite-(Fe) mineral surface structure and surface charge state, and to explore the mineral surface atomic binding bond. The bond breakage order and bond binding sequence were estimated selectively to the leaching reactivity. The surface structure and structure-sensitive reaction in highly acid systems were explored. The mechanism for the leaching reaction sequence and selective dissolution was investigated.
2. Materials and Methods
2.1. Material Preparation
Columbite-(Fe) samples were prepared by solid-state reaction with stoichiometric amounts of Nb2O5 and FeC2O4•2H2O of high purity (>99.9%). FeC2O4•2H2O could decompose to form FeO to provide divalent iron ions. The reaction equation was expressed as FeC2O4•2H2O + Nb2O5 → FeNb2O6 + CO↑ + CO2↑ + H2O↑. The particle size of Nb2O5 and FeC2O4•2H2O was uniform and mixed according to the molar ratio of columbite-(Fe). The mixtures were pressed, and a solid-state reaction at a vertical high-temperature tube furnace was conducted. The process was divided into three steps: firstly, the mixed blocks were heated up to 175 °C; the ferrous oxalate dihydrate was dehydrated, then heated up to 235 °C and kept at this temperature to make sure that FeC2O4 decomposed into FeO completely; finally, the sample was heated to 1100 °C and kept for 15 h and cooled with an Ar atmosphere to room temperature. The synthesized niobium iron phase is the FeNb2O6 phase, and the lattice parameters of the synthesized mineral phase of FeNb2O6 are consistent with those of the natural mineral phase [20].
2.2. Leaching Experiment and Surface Structure Characterization
According to the concentration limit of sulfuric acid, 8 M dilute sulfuric acid and 12 M concentrated sulfuric acid were selected for acid leaching of Ferrocolumbite. The leaching experiment conducted at 350 °C for 3 h in an air atmosphere. The minerals after the experiment (also called residues) were collected through a filter and cleaned with deionized water. The ferrocolumbite mineral and residues were analyzed with X-ray photoelectronic spectroscopy (XPS) and XRD. The leaching contents of Fe and Nb in the leaching filtrate were ascertained by ICP.
2.3. XPS Surface Analysis Technique
XPS was intrinsically surface sensitive which provided a 5-nm-thick surface layer structure and the chemical condition of its atoms [21], and enabled the study of mechanisms of the chemical and structural transformation of material surfaces under energy treatments [22]. The X-ray emission spectrum was recorded in a constant radiation transmission mode of 10 eV, and the basic elements in columbite mineral such as Nb, Fe, O, S, etc. were recorded before and after the leaching reaction. The internal electronic energy spectrum of the basic elements Nb 3d, Fe 2p, O 1s, S 2p, C 1s was recorded at 40 eV. The bond energy scale (Ebond) was corrected by the basic level Au 4f5/2 (Ebond = 83.96 eV), Ag 3d5/2 (Ebond = 368.21 eV) and the main standard sample (gold, silver, copper) with a precision accuracy of ±0.03 eV. The charge of elements was estimated by the spectrum of C 1s (284.8 eV). Overall, elements Nb 3d, Fe 2p, O 1s, S 2p, C 1s had photoelectron intensity and the signal originating from columbite-(Fe) and the residue, and both were well fit using the Lorentz-Gaussian peak with sub-peak regions. The background parameters were optimized, and the number of bands for the experimental spectral description was minimized. The peak position error was ±0.01 eV. The element content and ratio on the surface was determined with the expression as follows:
where I—the peak intensity, n—the concentration of the element to be tested in the sample, and S—the sensitivity factor.
I = n × S
3. Results and Discussion
During the Columbite-(Fe) leaching process, FeNb2O6 reacted with the concentrated sulfuric acid solution. In the presence of H+, the bond Fe-O, Nb-O in the ferrocolumbite surface could break, and the -SO4 bond would replace O in the bond Fe-O, Nb-O and form the bond of Fe-SO4, Nb-SO4. The bind energy of every binding bond in the surface mineral could be defined with the photoelectron spectra. The content of elements with different atomic charge states could be determined by combining the atomic chemical state spectra peak with its surface phase composition. The change in the surface layer depends on the different acid leaching conditions.
3.1. Ion Spatial Distributions in Columbite Surface
The columbite family of compounds was more representative of the AB2O6 series in which the A position was occupied mostly by Fe2+ and other bivalent ions such as Mn2+, Ti2+, and the B position mostly by Nb5+ and Ta5+. The cell unit of columbite-(Fe) (FeNb2O6) was with A = Fe and B = Nb, and the chemical bond in the mineral was basically Fe-O and Nb-O. Ferrocolumbite for XPS measurements was that the close proximity of the Fe 2p and Nb 3d orbitals meant that they were collected simultaneously in the same kinetic energy window using a single photon energy at the same depth in the solution. There was, therefore, no need to normalize to photon flux or to the transmission function of the hemispherical energy analyzer. The XPS experiment focused on ion spatial distributions. XPS photoelectron spectra of Fe 2p, O 1s and Nb 3d were collected from the surface of original FeNb2O6, and C 1s was standard sample. The Fe 2p spectra are composed of a narrow low-spin Fe2+ peak at about 709.8 eV (above 40% of total iron), and the Fe3+ peak at about 711.0 eV. The Nb 3d spectra was 207.0 eV. The H2SO4 leaching residues’ XPS spectra showed that the spectra peak positions of Fe 2p, O 1s, S 2p and Nb 3d were as a function of kinetic energy and provided further information about the amount of oxygen that decreased and the amount of Fe and Nb that increased.
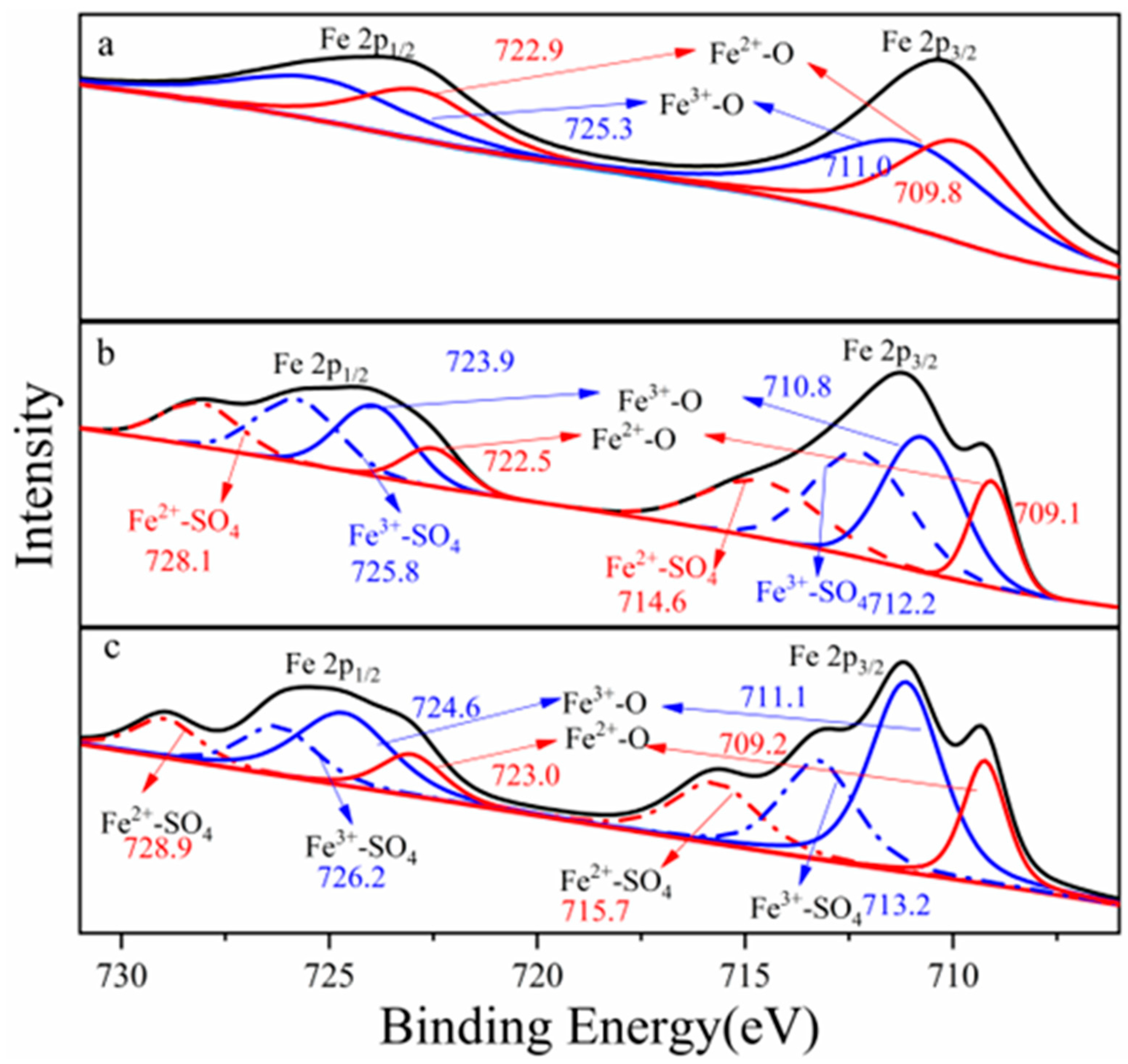
Figure 1 showed Fe 2p photoelectron spectra collected from columbite-(Fe) and leaching residues. The spectra showed that the ions’ distribution and the ions’ interaction binding energy changes were also provided from the spectra. Each spectrum was fit with two peaks from low to high energy. The Fe 2p photoelectron spectra recorded two peaks which meant that Fe 2p1/2 and Fe 2p3/2 orbitals exhibited two double-peaks, respectively, and the integrated peak area of Fe 2p3/2 orbitals contributed to more than that of Fe 2p1/2. Fe 2p photoelectron spectra were fitted with two components, the Fe3+ and Fe2+. The Fe 2p3/2 orbitals appeared with an electron binding energy of 709.8 eV which corresponded to the maximum Fe2+-O. The Fe 2p3/2 orbitals appeared with an electron binding energy of 711.0 eV which corresponded to the maximum Fe3+-O. Every element ion state in the columbite-(Fe) surface was estimated with this element chemical state spectrum peak intensity and its corresponding sensitivity factor. The sensitivity factor of the Fe 2p orbital was 2.957.
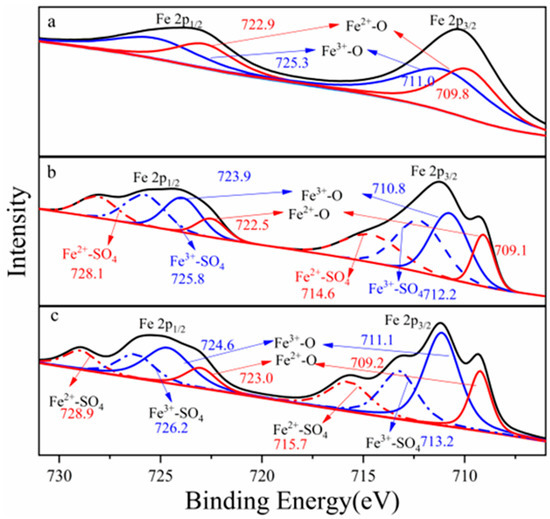
Figure 1.
Electron binding energy of Fe 2p, (a) ferrocolumbite, (b) 8 M H2SO4 acid leaching residue, (c) 12 M H2SO4 acid leaching residue.
X-ray photoelectron spectra were collected with Fe 2p3/2 spectra at photoelectron kinetic energies (pKEs) of 709.6, 710.8, 714.6 and 712.2 eV for the 8 M H2SO4 acid dissolution residues. The Fe3/2 spectra of 8 M H2SO4 acid residue recorded two Fe-O electron binding energy states which corresponded to the Fe3+-O peak maximum value of 710.8 eV and Fe2+-O peak value of 709.6 eV. The binding energy of Fe3+-O was 1.2 eV larger than that of Fe2+-O which meant ferric iron in the columbite-(Fe) mineral was more difficult to break and for dissolution. The Fe3/2 spectra peaks were also assigned to the surface species Fe2+-SO4 (714.6.1 eV) and Fe3+-SO4 (712.2 eV). The binding energy of Fe2+-SO4 was 2.4 eV larger than that of Fe3+-SO4 which meant the Fe2+ more easily formed the structure of Fe2+-SO4. We could deduce that the columbite-(Fe) mineral with more ferrous was a much easier structure to break and leach. The Fe 2p orbital spectrum recorded from the 12 M H2SO4 leaching residue surface photoelectron kinetic energies of 711.1, 709.2, 715.7 and 713.2 eV. The spectra were fitted with four ions’ bond components, Fe3+-O, Fe2+-O, Fe2+-SO4 and Fe3+-SO4. The binding energy of Fe3+-O was 1.9 eV larger than that of Fe2+-O which meant ferric iron in the columbite-(Fe) was more difficult to break and for dissolution. The binding energy of Fe2+-SO4 was 2.5 eV larger than that of Fe3+-SO4 which meant the Fe2+ more easily formed the structure of Fe2+-SO4.
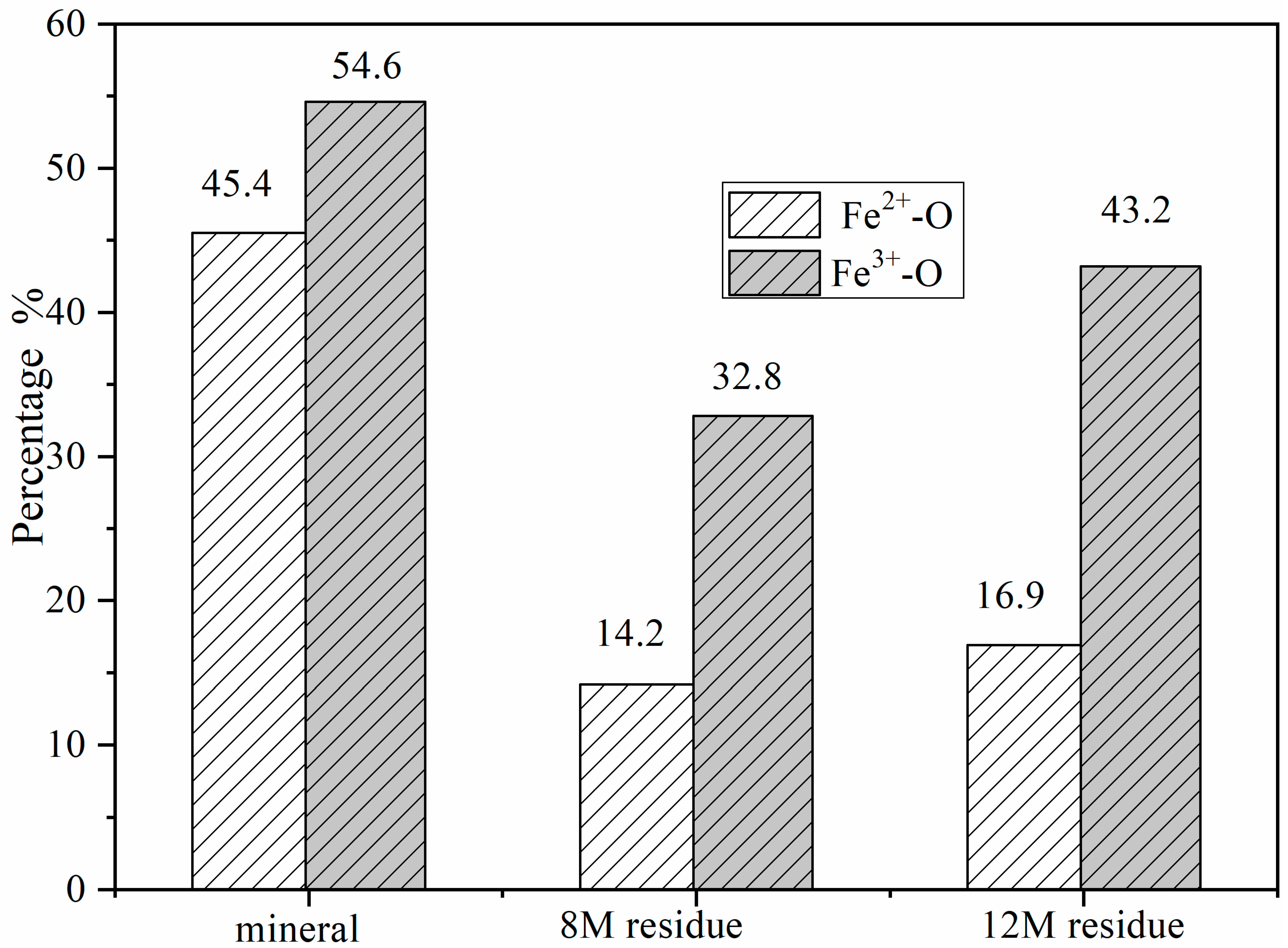
The Fe3+/Fe2+ ratio across the columbite-(Fe) surface was significant for understanding how the oxide-ion current couples with the pronounced oxidation state changes at the surface, providing insight into the behavior of the columbite-(Fe) solution. The area under the peaks indicates the relative contents of Fe2+ and Fe3+. Figure 2 shows the change in Fe2+/Fe3+ ratios at different minerals and residues. The contents of the Fe2+-O and Fe3+-O in the columbite-(Fe) surface were 45.4% and 54.6%, respectively, depending on the corresponding peak intensity of Fe2+-O and Fe3+-O. Fe-SO4 was formed on the surface of residues after columbite-(Fe) leaching. The peak of Fe3+-SO4 and Fe3+-O was split from spectra of Fe3+, and the peak of Fe2+-SO4 and Fe2+-O was split from spectra of Fe2+. The XPS-derived amount of Fe2+ and Fe3+ on the residues’ surface for both the 8 and 12 M H2SO4 acid leaching medium is shown in Figure 3. The results of XPS analysis revealed that the amounts of Fe3+-O in the 8 M leaching residue surface was 32.8%, and Fe2+-O content was 14.2%. The contents of Fe2+-SO4 in the 8 M leaching residue surface was 21.1%, and that of Fe3+-SO4 on the residue surface was 31.9%. The amounts of Fe3+-O on the residue surface was increased from 32.8% to 43.2% after 12 M H2SO4 acid leaching, and Fe2+-O content was 16.9%. The contents of Fe2+-SO4 on the 12 M residue surface was 16.6%, and that of Fe3+-SO4 on the residue surface was 23.3%.
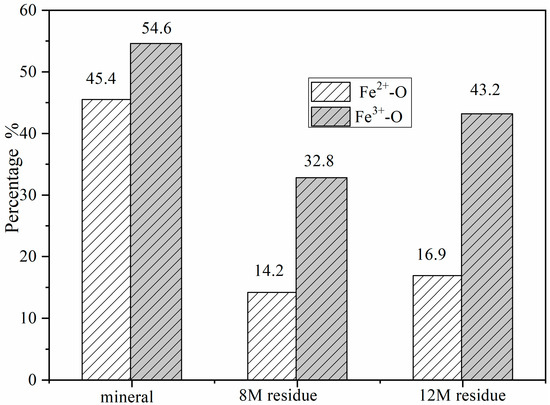
Figure 2.
The ratio of Fe2+/Fe3+ on columbite-(Fe) and residues surface.
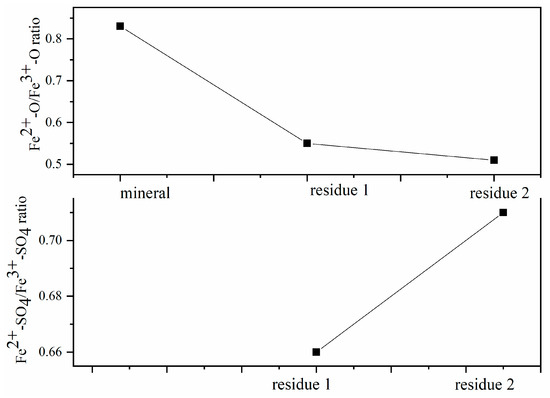
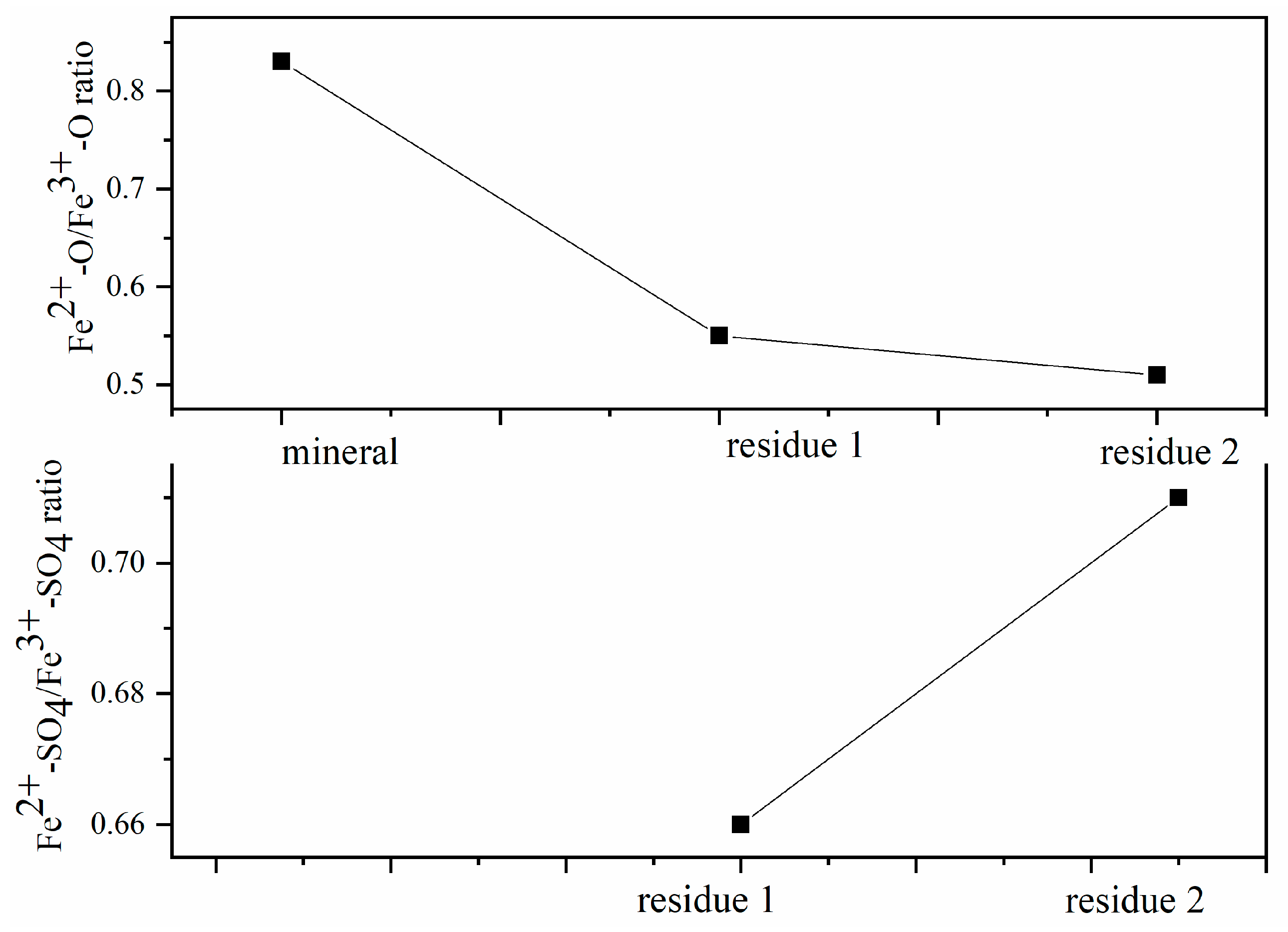
Figure 3.
Fe2+-O/Fe3+-O ratio and Fe2+-SO4/Fe3+-SO4 ratio on residues surface.
Changes in the Fe2+/Fe3+ ratios in the active regions are a consequence of oxidation dissolution on columbite-(Fe) surface shifts caused by the interaction between the surface chemical ion and the concentration of the H2SO4 acid leaching medium. The Fe2+/Fe3+ ratio on the mineral surface measured from XPS was 0.83, data which demonstrated the ability of XPS to resolve redox-active regions spatially under operating conditions by oxidation leaching. The ratio Fe2+/Fe3+ that shifted on the surface suggested that the reacted rates changed with leaching contents by the surface reaction and electron transport. Figure 3 shows the Fe2+-O/Fe3+-O ratio and Fe2+-SO4/Fe3+-SO4 ratio of columbite-(Fe) and residues. The ratio of Fe2+-O/Fe3+-O on the 8 M H2SO4 acid leaching residue surface was 0.55 which was 0.28 lower than the ratio of the initial mineral. The Fe2+-O on the residue surface decreasing meant Fe2+-O was broken and dissolved into the acid medium. The ratio of Fe2+-SO4/Fe3+-SO4 on the 8 M H2SO4 acid leaching residue surface was 0.66. The ratio of Fe2+-O/Fe3+-O on the 12 M H2SO4 acid leaching residue was 0.50, which was 0.33 lower than that of the initial mineral, and the ratio of Fe2+-SO4/Fe3+-SO4 on the surface was 0.71. The decreasing Fe2+-O/Fe3+-O ratio deduced that this increase in Fe2+ content meant the improvement in mineral surface dissolution. It could be due to the ability of the Fe2+ ions to react with the H2SO4 acid more effectively than with the Fe3+ ions.
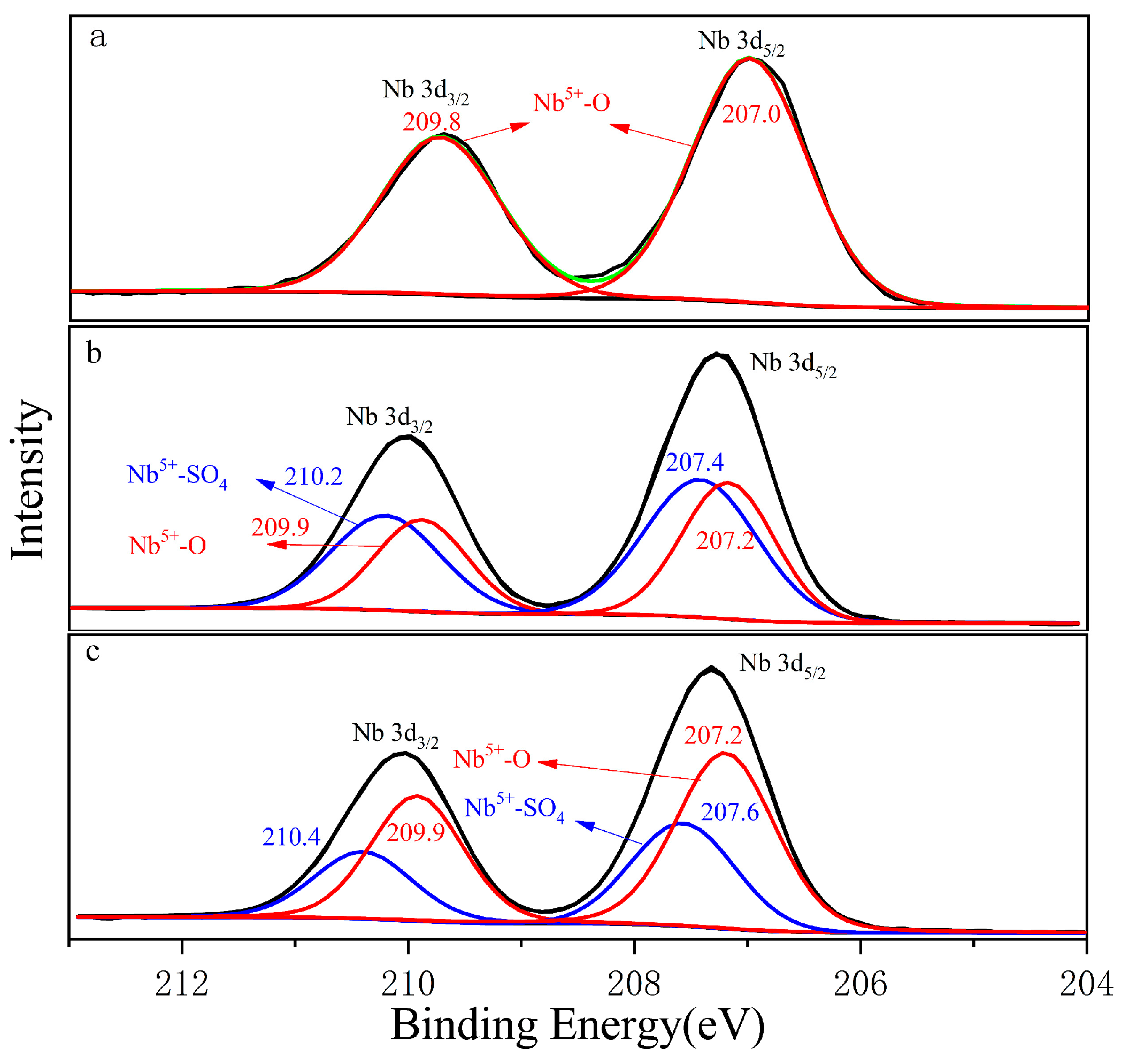
Figure 4a shows the Nb 3d orbital photoelectron spectrum collected from the columbite-(Fe) surface. The green curve was the total peak after sub-peak fitting. The Nb 3d orbitals exhibited double peaks corresponding to Nb 3d3/2 and Nb 3d5/2 double peaks which appeared at 209.8 eV and 207.0 eV, respectively. Nb 3d5/2 made an obviously main contribution. Figure 4b shows the Nb 3d orbital photoelectron spectrum of the 8 M H2SO4 acid leaching residue. The Nb 3d5/2 orbitals corresponding to the peak binding energy of Nb5+-SO4 was 207.4 eV, which was 0.2 eV larger than that of Nb5+-O with 207.2 eV. Figure 4c shows the Nb 3d orbital photoelectron spectrum of the 12 M H2SO4 acid leaching residue, and the binding energy was 207.2 eV and 207.6 eV corresponding to Nb5+-O and Nb5+-SO4, respectively. Nb5+-SO4 was formed during the H2SO4 acid leaching process; the main peak of Nb5+-SO4 on the residue surface was shifted from 207.4 eV for 8 M H2SO4 to 207.6 eV for the 12 M H2SO4 acid content, which meant O in Nb-O was easily replaceable with the -SO4 bond with the concentrated H2SO4 acid increasing.
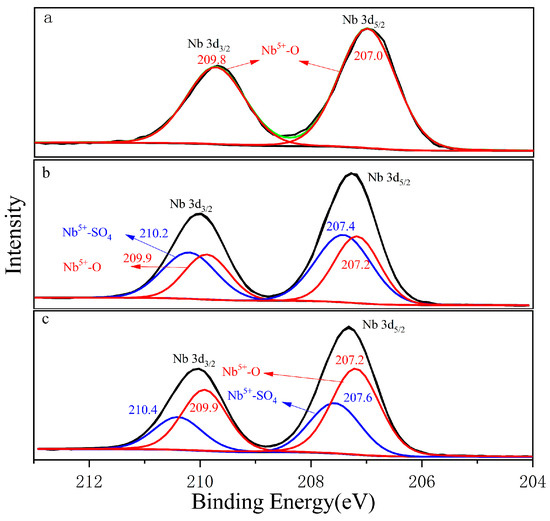
Figure 4.
The photoelectron spectrum of Nb, (a) ferrocolumbite; (b) 8 M H2SO4 leaching residue; (c) 12 M H2SO4 leaching residue.
3.2. Columbite-(Fe) Surface Structure-Sensitive of Selective Reaction
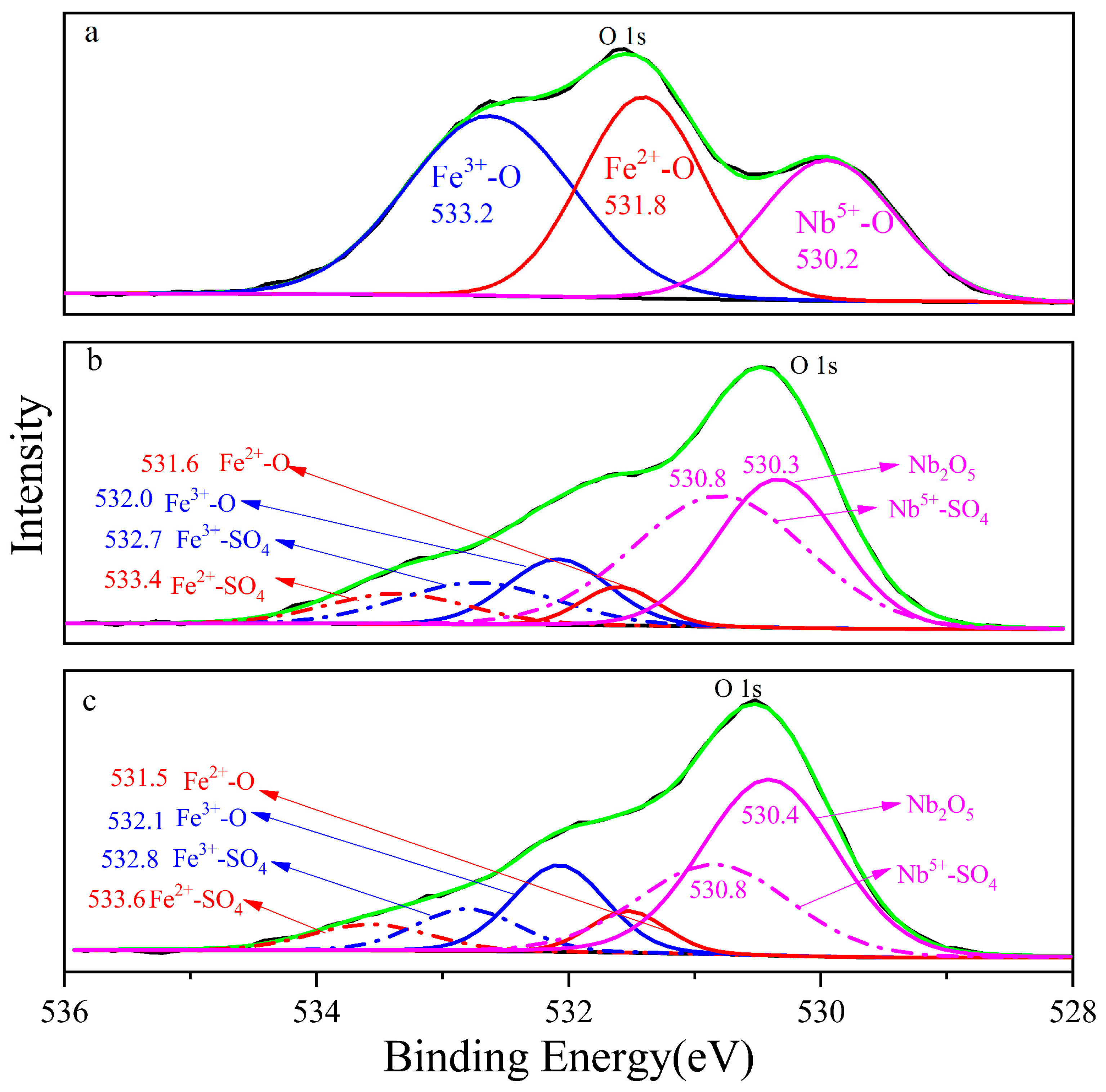
The photoelectron spectrum of columbite-(Fe) fitted O 1s orbital with three components is shown in Figure 5, and the three chemical elements’ state combined with oxygen, namely, Fe3+-O, Fe2+-O and Nb5+-O, with the corresponding electron binding energy decreased in proper sequence, namely, 533.2 eV, 531.8 eV and 530.2 eV, respectively. The binding energy of the bond structure of Fe2+-O on the 8 M residue was 533.4 eV, which was shifted 0.2 eV compared to columbite-(Fe). Fe3+-O and Nb5+-O and the corresponding electronic binding energy were 532.7 eV and 530.4 eV in sequence. The increased three photoelectron spectra meant the different valence in the 8 M residue. The binding energy of Fe2+-O, Fe3+-O and Nb5+-O on the 12 M H2SO4 acid leaching residue surface was 533.6 eV, 532.9 eV and 530.8 eV, respectively. Fe-O shifted to 0.2 eV increasing and Nb-O shifted 0.4 eV increasing from the 8 M residue to the 12 M one.
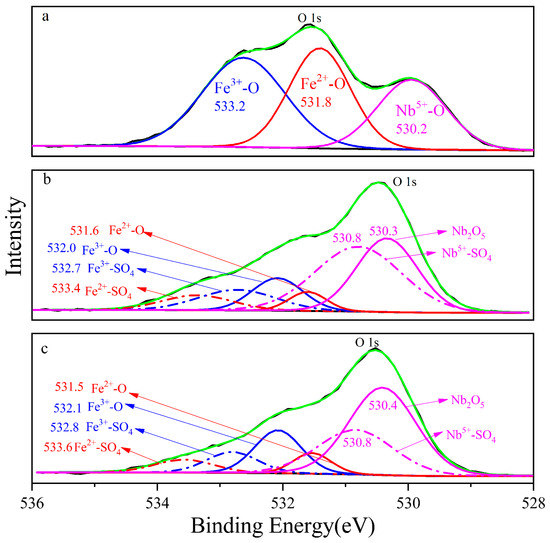
Figure 5.
The photoelectron spectrum of O 1s, (a) columbite-(Fe); (b) 8 M H2SO4 acid leaching residue; (c) 12 M H2SO4 acid leaching residue.
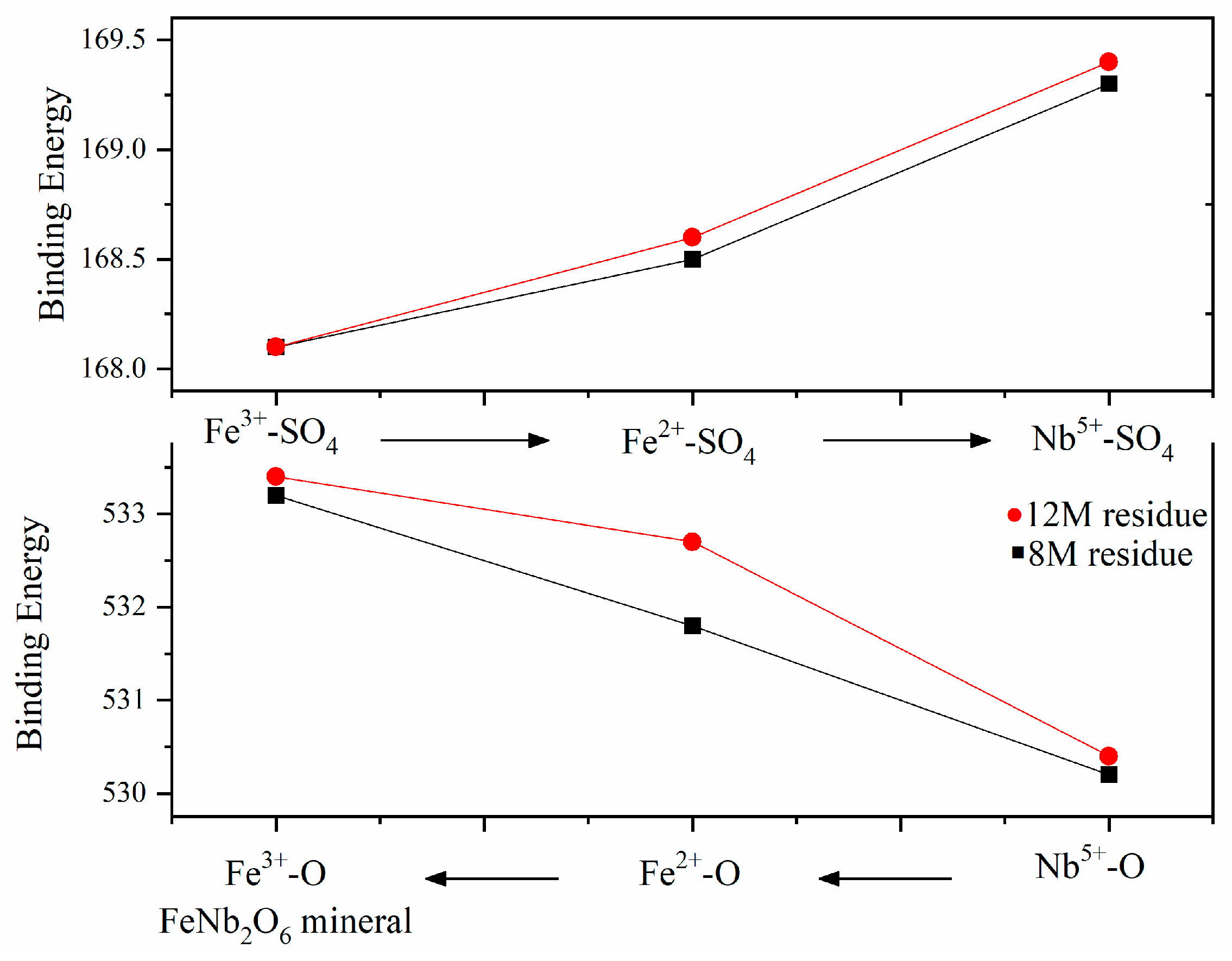
The comparison of metal ions’ binding energy on the surface indicated that the structure varied with the H2SO4 concentration, and structure-sensitive chemical reaction and dissolution were in sequence. As shown in Figure 6, during acid leaching, Nb-O was preferentially broken and dissolved into leaching liquor, while Fe3+-O was the most stable, which meant Fe3+ was the most difficult to dissolve from the columbite-(Fe) mineral. The chemical leaching reaction and dissolution with H2SO4 were under the sequence Nb5+-O < Fe2+-O < Fe3+-O.
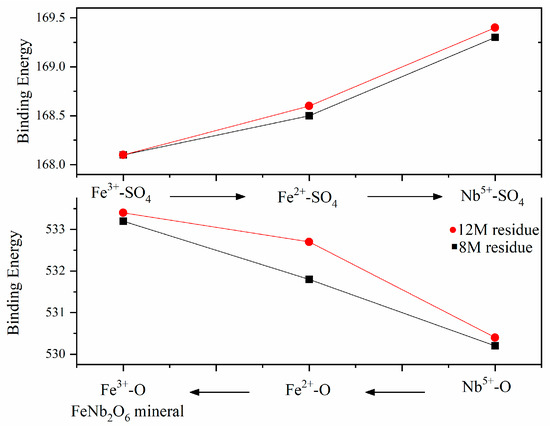
Figure 6.
The binding energy sequence of each bond.
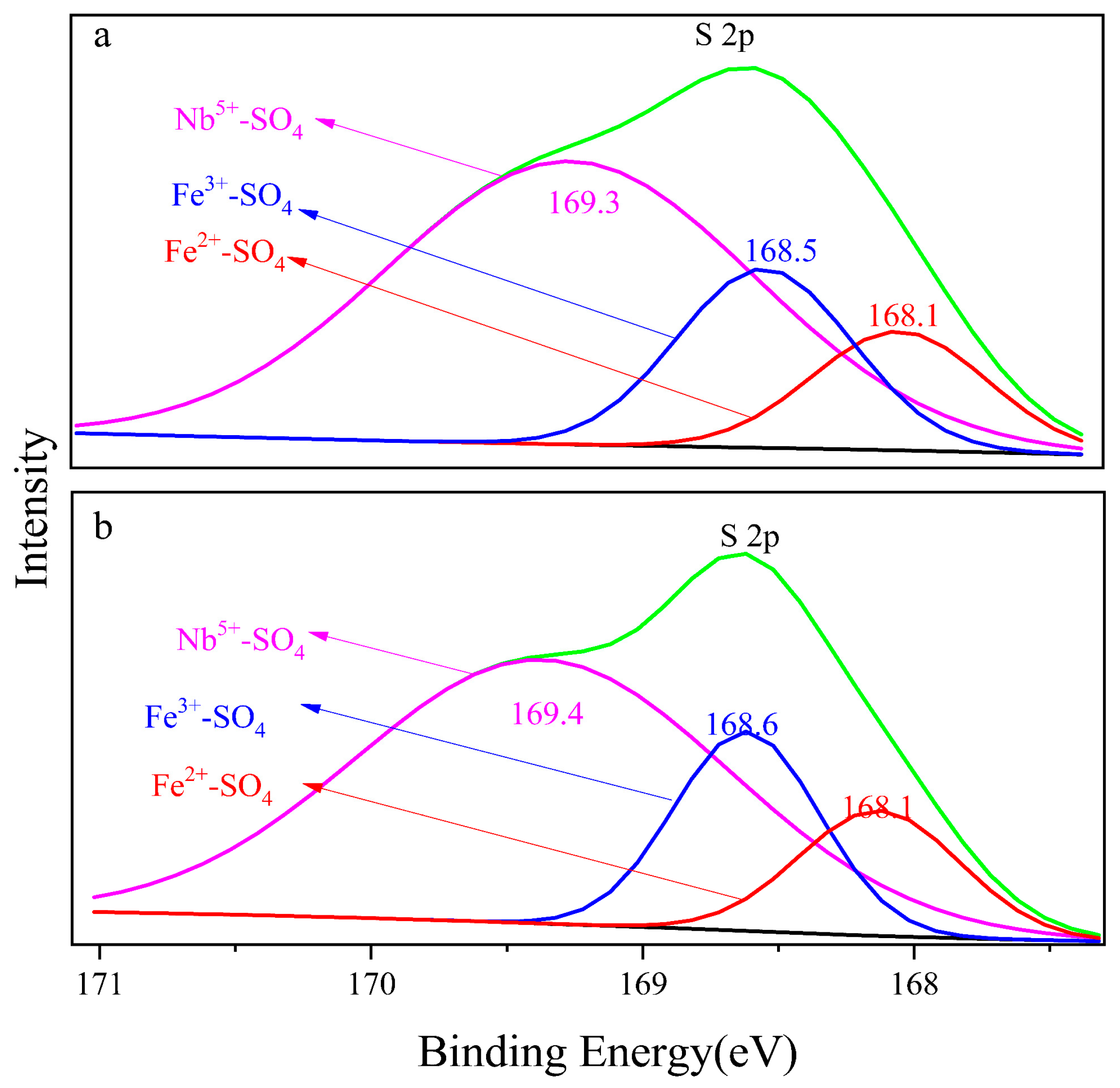
Figure 7a,b show the S 2p orbital photoelectron spectra of the 8 M H2SO4 and 12 M H2SO4 leaching residue, respectively. Naked Fe3+, Fe2+ and Nb5+ in the mineral surface contacted with SO42− and formed bonds. The binding energy of Fe3+-SO4, Fe2+-SO4 and Nb5+-SO4 on the 8 M H2SO4 leaching residue surface was 168.1 eV, 168.5 eV and 169.3 eV, respectively, and that on the surface of the 12 M H2SO4 leaching residue was 168.1 eV, 168.6 eV and 169.4 eV, respectively. The electron binding energy of Nb5+-SO4 was higher than that of Fe3+-SO4 and Fe2+-SO4, which meant the structure Nb5+-SO4 was the most stable. The formation of Nb5+-SO4 would promote the breakdown of Nb5+-O on the mineral surface during H2SO4 leaching. The binding energy of Fe3+-SO4 was 0.4 eV larger than that of Fe2+-SO4. The metal ion pairing SO42− in sequence was Fe3+-SO4 < Fe2+-SO4 < Nb5+-SO4 as shown in Figure 7.
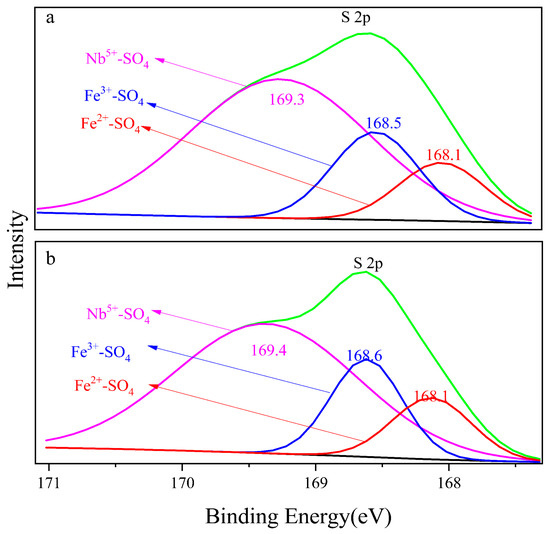
Figure 7.
Electron binding energy of S 2p, (a) 8 M H2SO4 acid leaching residue; (b) 12 M H2SO4 acid leaching residue.
The binding energy of Fe-SO4 increased compared with Fe-O, and the binding energy of Nb-SO4 compared with Nb-O was a parallel trend. The bond strength was Fe3+-O, Fe2+-O and Nb5+-O in sequence which meant the Nb5+-O bond broke preferentially to the Fe3+-O bond. The strength of the corresponding electronic binding energy of Fe2+-SO4, Fe3+-SO4 and Nb5+-SO4 increased in sequence, and Fe2+-SO4 was most stable. The contents of the Fe3+-SO4 on the 8 M H2SO4 acid leaching residue surface was 31.9% and on the 12 M H2SO4 acid leaching residue was a corresponding 23.3%. The contents of the Fe2+-SO4 of 21.1% on the 8 M leaching residue surface declined to 16.6% on the 12 M H2SO4 residue. The results indicated that the dissolution of Fe decreased as the acid medium content increased.
The surface of columbite-(Fe) was dissolved with H2SO4, and the binding energy of Fe-SO4 was relatively larger than that of Fe-O, which meant the Fe2+-O and the Fe3+-O in the surface were broken by H2SO4 during the acid leaching process, and a larger binding energy of Fe2+-SO4 and Fe3+-SO4 formed. The bind structure change during the leaching could be expressed with the formula: Fe-O + H2SO4 → Fe-SO4 + H2O. The binding energy of Nb-SO4 was relatively larger than that of Nb-O, which meant the more stable Nb-SO4 formed. The bind structure change during the leaching could be expressed with the formula: Nb-O + H2SO4 → Nb-SO4 + H2O.
Table 1 shows the leaching results of the ICP detection columbite-(Fe) mineral leached in different H2SO4 acid concentration solutions. The dissolution content of Nb and Fe improved as the H2SO4 acid concentration increased. The ratio of Nb/Fe in 8 M H2SO4 leaching liquor (dissolving ratio of Nb/Fe) was 2.29, and the ratio of Nb/Fe in the 12 M H2SO4 leaching liquor was 2.31, which was 0.29 and 0.31 increased compared with the columbite-(Fe) mineral with the Nb/Fe ratio of 2. The results revealed that Nb in the columbite mineral was more easily leached compared to Fe which was in good agreement with the result of photoelectron spectroscopy experiments.

Table 1.
ICP detection results of solution elements after different H2SO4 acid leaching (mg/L).
3.3. Surface Structure Control Reaction and Dissolution
For the dissolution of the Fe element, there are two hypothetical possibilities. One was that Fe2+-O in FeNb2O6 was first broken under the acidic action of H2SO4 to form Fe2+ dissolved in the solution, and then Fe2+ was oxidized Fe3+ by the H2SO4 molecule in the solution to promote dissolution. Another was that the H2SO4 molecule in the solution oxidized Fe2+-O to Fe3+-O firstly; then, Fe3+-O was broken into Fe3+ to dissolve into the solution under the acidic action of H2SO4.
The content of Fe2+-O on the 8 M H2SO4 leaching residue surface decreased by 31.2% and 28.5% on the 12 M leaching residue, compared to the content of Fe3+-O which decreased by 21.8% and 11.4%, respectively. The results showed that the mineral structure with ferrous iron was much more easily broken, and relevantly easily leached. The Fe2+/Fe3+ ratio of leaching residues was smaller than that of the initial mineral, which meant the content of Fe3+ on the residues’ surface increased. The reasons were that more Fe2+ was soluble in the acid leaching process, or a large amount of Fe3+ formed on surface. Therefore, it is necessary to analyze the percentage content of each component of Fe on the mineral surface. Irrespective of whether oxidation of the H2SO4 molecule causes a change in the valence state of Fe elements, the decreasing content of Fe2+-O and Fe3+-O should be equal to the generative Fe2+-SO4 and Fe3+-SO4 correspondingly. The content of Fe2+-SO4 and Fe3+-SO4 on the 8 M H2SO4 leaching residue surface increased by 21.9% (Fe2+-O decreased by 31.2) and 31.9% (Fe3+-O decreased by 21.8%). The content of Fe2+-SO4 and Fe3+-SO4 on the 12 M H2SO4 leaching residue surface increased by 16.6% and 23% while the content of Fe2+-O and Fe3+-O decreased by 16.5% and 11.4%, respectively. The contrast results indicated that there occurred an oxidation reaction during the leaching process. There was 10.1% Fe2+ oxidized to Fe3+ in 8 M H2SO4 and 11.9% in 12 M H2SO4.
The columbite-(Fe) mineral was leached with different H2SO4 acid concentrations; there was a large amount of Fe2+-SO4 existing on the residue surface. The dissolution of Fe from the mineral into the leaching liquor meant that Fe2+-O broke and released Fe2+ in the solution at first and then was oxidized to Fe3+. The redox reaction mechanism shows as follows:
Fe2+-O + H+ + SO4 → Fe2+-SO4 + H2O
Fe2+-SO4 − e → Fe2+-SO4
The results of the Nb dissolution from the mineral indicated that Nb5+-O was directly digested by H+, and the released Nb5+ complex with SO42− ion formed cation−anion pairs. The ratio of Nb5+-SO4 was 55.1% with Nb5+-O accounting for 44.9% on the 8 M H2SO4 acid leaching residue surface. The ratio of Nb5+-SO4 was 36.6% and Nb5+-O of 63.4% on the 12 M H2SO4 acid leaching residue surface. It was because the quantity of molecular H2SO4 in the bulk with concentrations of above 12 M lessened the ionization species of SO42− and HSO4−. The result indicated that the dissolution of Nb from the mineral owed to the content of H+ in the solution. In the 8 M sulfuric acid solution, there contained more species of H+, SO42−, which meant more Nb5+-SO4 was formed. The leaching reaction mechanism shows as follows:
Nb2+-O + H+ + SO4 → Nb5+-SO4 + H2O
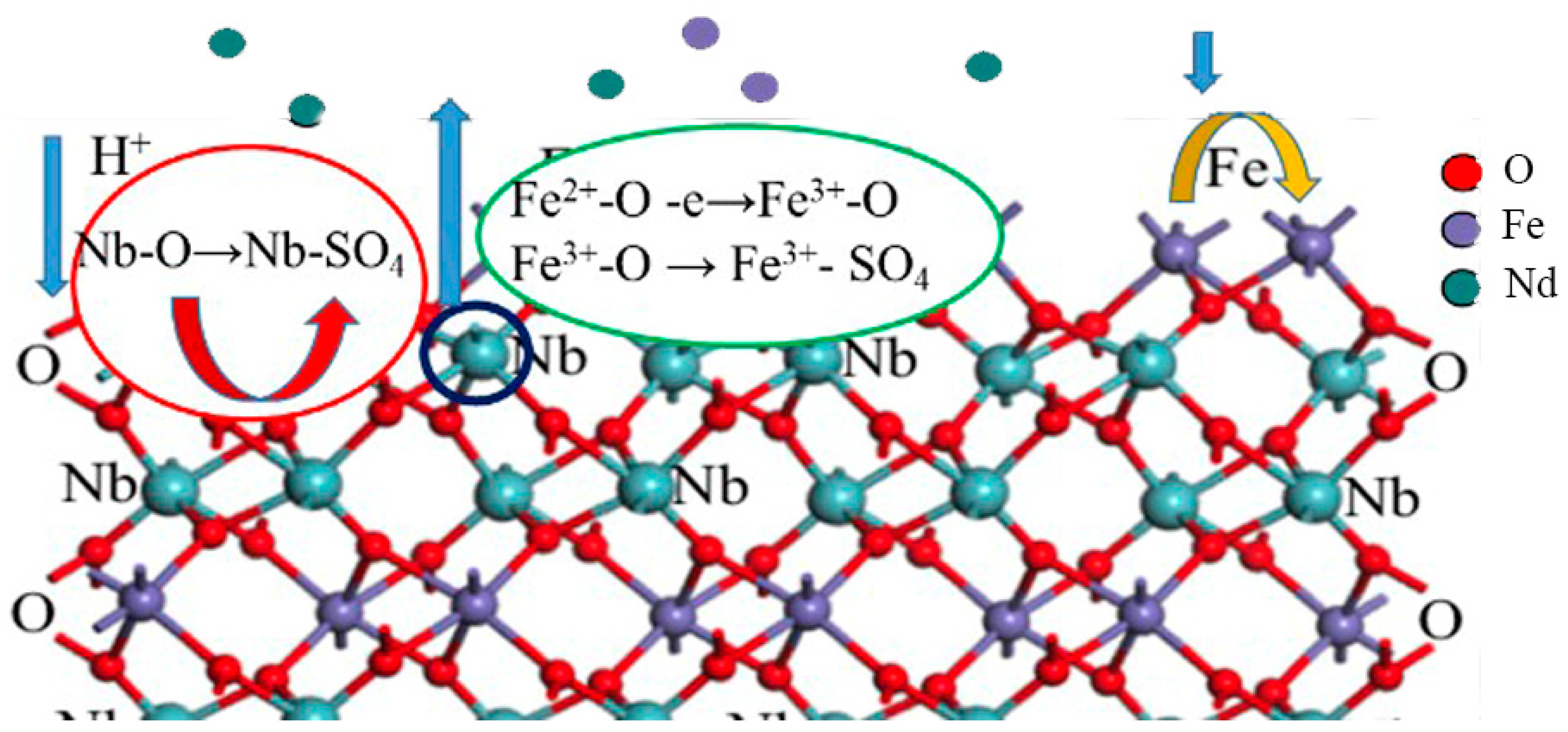
The extraction of niobium from the columbite-(Fe) mineral with the sulfuric acid solution occurred at the mineral surface. When the mineral surface was exposed to the acid medium ions, the cations could be dissolved and transferred from the mineral surface into the medium. Therefore, the quantity and distribution of ions on the new surface of the mineral was changed. The columbite-(Fe) mineral leached with H2SO4 acid was a reaction controlled by the surface structure. Figure 8 shows the surface dissolution of the columbite-(Fe) mechanism schematics. For the dissolution of the Nb element, the experimental results showed that increasing the H+ concentration could promote the dissolution. For the dissolution of the Fe element, the oxidation potential could be effective in the enhancement of dissolution. Some of the Fe2+ ions on the columbite-(Fe) surface converted to the Fe3+ ones through the concentrated sulfuric acid with oxidization. The greater tendency of Fe3+ ions was on the surface; less content of Fe dissolved in the acid medium.
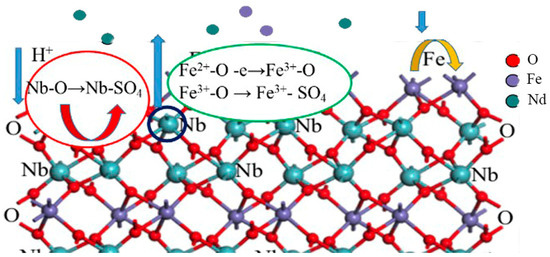
Figure 8.
Surface structure control reactivity and dissolution with H2SO4 acid leaching.
4. Conclusions
X-ray photoelectron spectroscopy combined with ICP was used to study the columbite surface chemical state and structure before and after different acid leaching processes. There are several conclusions as follows:
- The columbite-(Fe) surface electron binding energy of Nb5+-O was lower than that of Fe3+-O and Fe2+-O. The selective dissolution sequence was Nb5+-O < Fe2+-O < Fe3+-O. There formed a structure of Nb5+-SO4, Fe3+-SO4 and Fe2+-SO4 on the sulfuric acid residue surface. The electron binding energy of Nb5+-SO4 was higher than that of Fe3+-SO4 and Fe2+-SO4, which meant the structure Nb5+-SO4 was the most stable. The binding energy of Fe3+-SO4 was larger than that of Fe2+-SO4. The metal ion pairing SO4 in sequence was Fe3+-SO4 < Fe2+-SO4 < Nb5+-SO4. The electron binding energy of Me(Nb5+, Fe3+, Fe2+)-SO4 corresponding to each chemical state element was higher than the electron binding energy of Me(Nb5+, Fe3+, Fe2+)-O, which means the Me-O bond was broken, and a more stable Me-SO4 bond was formed.
- The ratio of Fe2+-O/Fe3+-O on the 8 M H2SO4 acid leaching residue surface was 0.55 which was 0.28 lower than the ratio of the initial mineral. The ratio of Fe2+-O/Fe3+-O on the 12 M H2SO4 acid leaching residue was 0.50, which was 0.33 lower than that of the initial mineral. The decreasing Fe2+-O/Fe3+-O ratio deduced that this increase in Fe2+ content meant the improvement in mineral surface dissolution. It could be due to the ability of the Fe2+ ions to react with the H2SO4 acid more effectively than with the Fe3+ ions.
- The leached and dissolution mechanism of columbite-(Fe) was that Nb5+-O was directly digested by H+, and the released Nb5+ complex with the SO42− ion formed cation−anion pairs. The result indicated that the dissolution of Nb from the mineral owed to the content of H+ in the solution, and increasing the H+ concentration could promote the dissolution. For the dissolution of the Fe element, the oxidation potential could be effective in the enhancement of dissolution.
Author Contributions
X.G.: Investigation, Writing—original draft, Visualization. Q.L.: Conceptualization, Methodology, Writing—review and editing. D.W.: Data curation. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China under Grant [No:51974185].
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, the writing of the manuscript; or the decision to publish the results.
References
- Malwina, N. Report on Critical Raw Materials for the EU; Report of the Ad Hoc Working Group on Defining Critical Raw Materials; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- Masahiko, B.; Tatsuya, K.; Ryosuke, O.S. Niobium powder synthesized by calciothermic reduction of niobium hydroxide for use in capacitors. J. Phys. Chem. Solids 2015, 78, 101–109. [Google Scholar] [CrossRef]
- Yuan, B.; Okabe, T.H. Niobium powder production by reducing electrochemically dissolved niobium ions in molten salt. J. Alloys Compd. 2008, 454, 185–193. [Google Scholar] [CrossRef]
- Santos, C.A.; Zawislak, L.I.; Kinast, E.J.; Antonietti, V.; Cunha, J.B. Crystal chemistry and structure of the orthorhombic (Fe,Mn,)(Ta,Na)2O6 family of compounds. Braz. J. Phys. 2001, 31, 616–631. [Google Scholar] [CrossRef]
- Tindle, A.G.; Breaks, F.W. Columbite-tantalite mineral chemistry from rare-element granitic pegmatites: Separation Lake area, N.W. Ontario, Canada. Miner. Petrol. 2000, 70, 165–198. [Google Scholar] [CrossRef]
- Rodriguez, M.H.; Rosales, G.D.; Pinna, E.G.; Suarez, D.S. Extraction of niobium and tantalum from ferrocolumbite by hydrofluoric acid pressure leaching. Hydrometallurgy 2015, 156, 17–20. [Google Scholar] [CrossRef]
- Majima, H.; Awakura, Y.; Mishima, M.; Hirato, T. Dissolution of columbite-tantalite in acidic fluoride media. Metall. Mater. Trans. B 1988, 19, 355–363. [Google Scholar] [CrossRef]
- Bludssus, W.; Eckert, J. Process for the Recovery and Separation of Tantalum and Niobium. U.S. Patent 5209910, 11 May 1993. [Google Scholar]
- Wu, B.; Shang, H.; Wen, J.K. Sulfuric acid leaching of low-grade refractory tantalum-niobium and associated rare earths minerals in Panxi area of China. Rare Met. 2015, 34, 202–206. [Google Scholar] [CrossRef]
- Bock, R. A Handbook of Decomposition Methods in Analytical Chemistry; Halsted Press: New York, NY, USA, 1979. [Google Scholar]
- He, J.; Zhang, Z.; Xu, Z. Hydrometallurgical extraction of tantalum and niobium in China. Rare Metal Mater. Eng. 1998, 27, 14–15. [Google Scholar]
- Zhu, Z.; Cheng, C.Y. Solvent extraction technology for the separation and purification of niobium and tantalum: A review. Hydrometallurgy 2011, 107, 1–12. [Google Scholar] [CrossRef]
- Shikika, A.; Sethurajan, M.; Muvundja, F.; Mugumaoderha, M.C.; Gaydardzhiev, S. A review on extractive metallurgy of tantalum and niobium. Hydrometallurgy 2020, 198, 105496. [Google Scholar] [CrossRef]
- Yang, X.L.; Huang, W.F.; Fang, Q.; Ouyang, H. Pressure leaching of manganotantalite by sulfuric acid using ammonium fluoride as an assistant reagent. Hydrometallurgy 2018, 175, 348–353. [Google Scholar] [CrossRef]
- Omneya, M.; Hussaini, E.l.; Mohamed, A.M. Sulfuric acid leaching of Kab Amiri niobium–tantalum bearing minerals, Central Eastern Desert, Egypt. Hydrometallurgy 2002, 64, 219–229. [Google Scholar] [CrossRef]
- Sun, Y.; He, Q.; Li, Q.J. Preparation of Nickel-Niobium alloy powder precursor from niobium ore. Non-Ferr. Met. (Smelt. Part) 2017, 2, 40–43. [Google Scholar] [CrossRef]
- Chanturiya, V.A.; Bunin, I.Z.; Ryazantseva, M.V.; Chanturiy, E.L. Effect of High-Vol-tage Nanosecond Pulses on the Physicochemical and Technological Properties of Rare-Metal Minerals. Bull. Rus. Academy Sci. Phys. 2018, 82, 781–785. [Google Scholar] [CrossRef]
- Chanturia, V.A.; Bunin, I.Z.; Ryazantseva, M.V.; Chanturia, E.L. Modification of Structural, Chemical and Process Properties of Rare Metal Minerals under Treatment by High-Voltage Nanosecond Pulses. J. Min. Sci. 2017, 53, 718–733. [Google Scholar] [CrossRef]
- Yang, F.X.; Li, Q.J.; Wang, D.; Zheng, S.B. Interfacial Structure Change and Selective Dissolution of Columbite–(Fe) Mineral during HF Acid Leaching. Minerals 2021, 11, 146. [Google Scholar] [CrossRef]
- Wang, D.; Li, Q.J.; Guo, X.; Zou, X.L. Study on dissolution mechanism of mineral/liquid interface during HF acid leaching niobium-tantalum ore. Miner. Eng. 2022, 188, 107835. [Google Scholar] [CrossRef]
- Chanturia, V.A.; Bunin, I.Z.; Ryazantseva, M.V.; Filippov, I.V.; Koporulina, E.V. Nan-osecond Electromagnetic Pulse Effect on Phase Composition of Pyrite and Arsenopyri-te Surfaces, Their Sorption and Flotation Properties. J. Min. Sci. 2011, 47, 506–513. [Google Scholar] [CrossRef]
- Zhang, C.J.; Grass, M.E.; McDaniel, A.H.; DeCaluwe, S.C.; Gabaly, F.E. Measuring fundamental properties in operating solid oxide electrochemical cells by using in situ X-ray photoelectron spectroscopy. Nat. Mater. 2010, 9, 944–949. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).