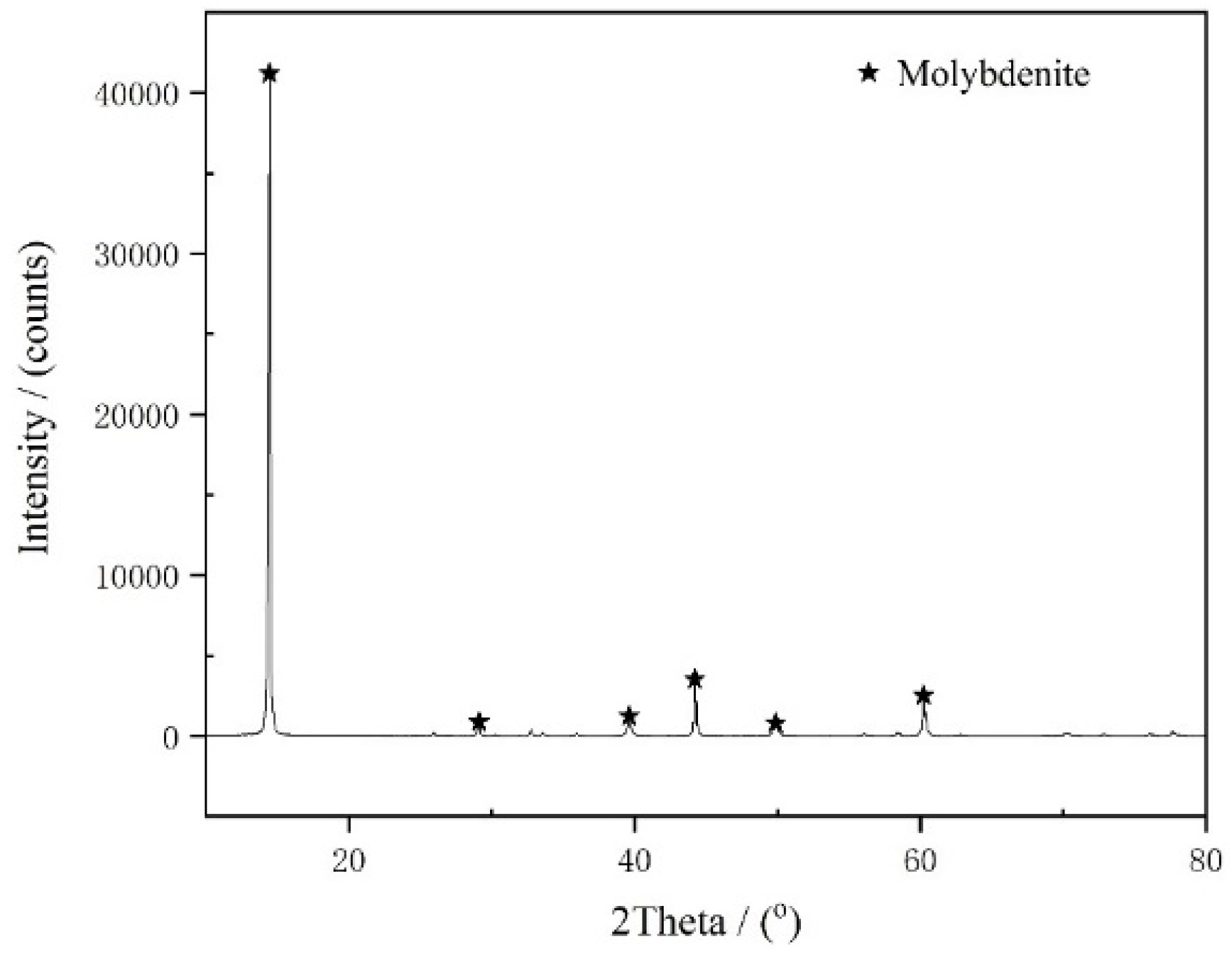
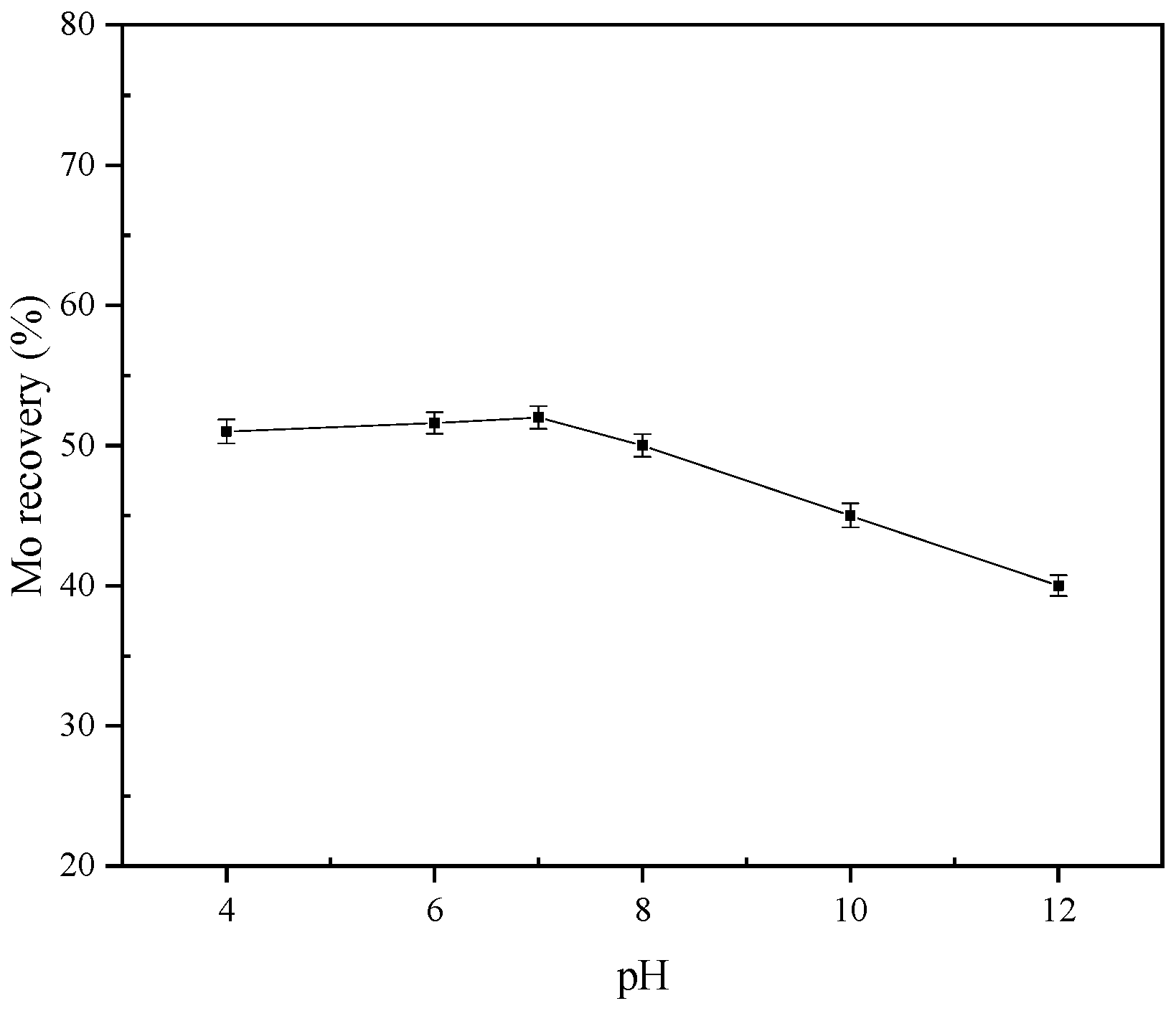
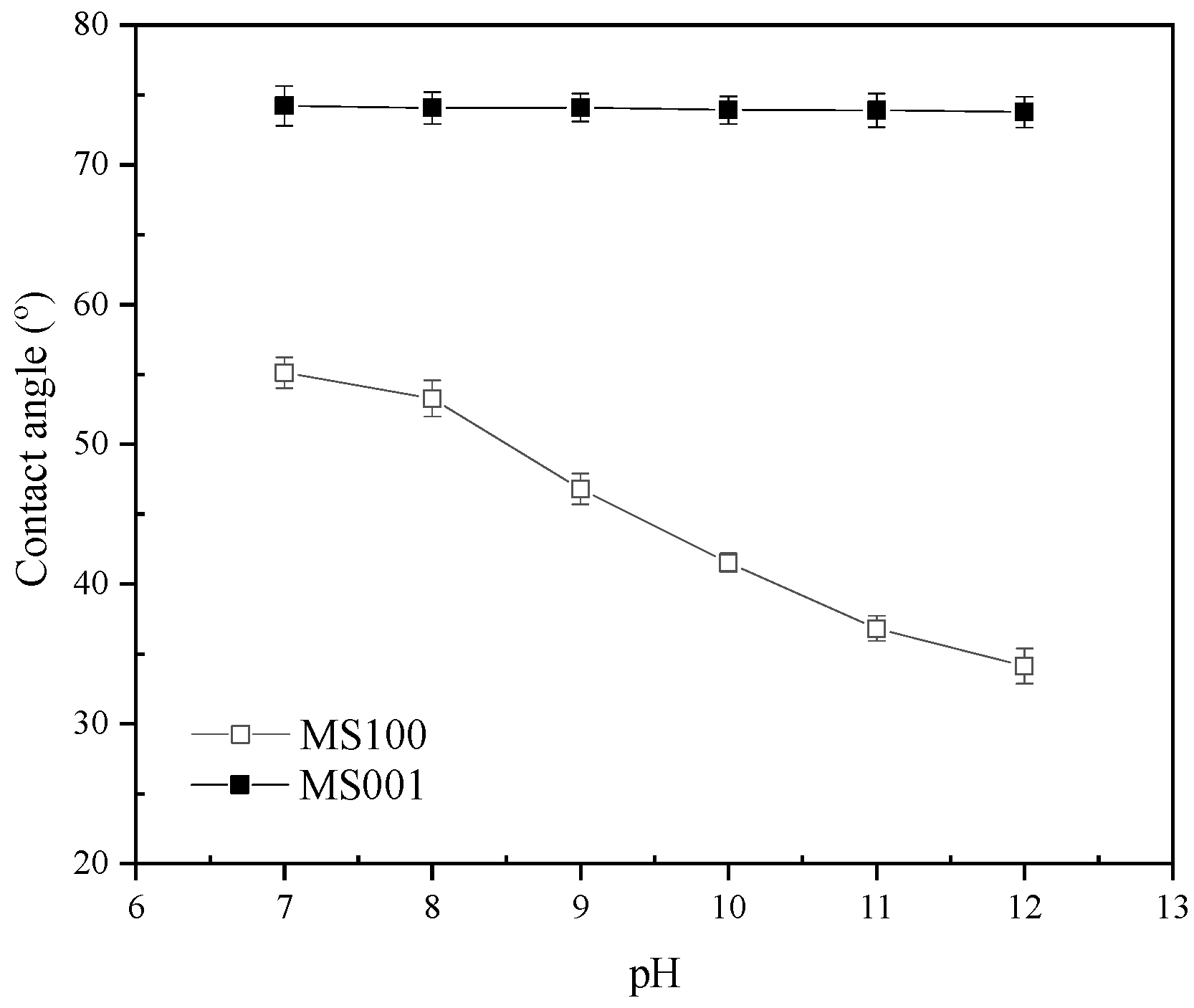
To further study the inhibiting mechanism of high pH on molybdenite flotation, DFT was used to analyze the interaction of water (H2O) and hydroxide/hydrogen ions (OH−/H+) with the molybdenite surface at the atomic and electronic levels.
3.4.2. Electronic Properties of H2O and OH−/H+ at MS001/MS100
The electronic properties of H2O and OH−/H+ at MS001/MS100 can reveal their adsorption mechanism on molybdenite surfaces at the molecular and electronic levels. It mainly includes the bond lengths, Mulliken bond population, differential density, density of states, and Mulliken charge population.
- (1)
Analysis of Water Bonding on the Surface of Molybdenite
The stable adsorption configurations of H
2O in MS001/MS100 are shown in
Figure 7 and
Figure 8, respectively. To quantitatively characterize the adsorption configuration of H
2O on the MS001/MS100, the Mulliken bond populations and their corresponding bond lengths after stable adsorption of H
2O on the MS001/MS100 are listed in
Table 2.
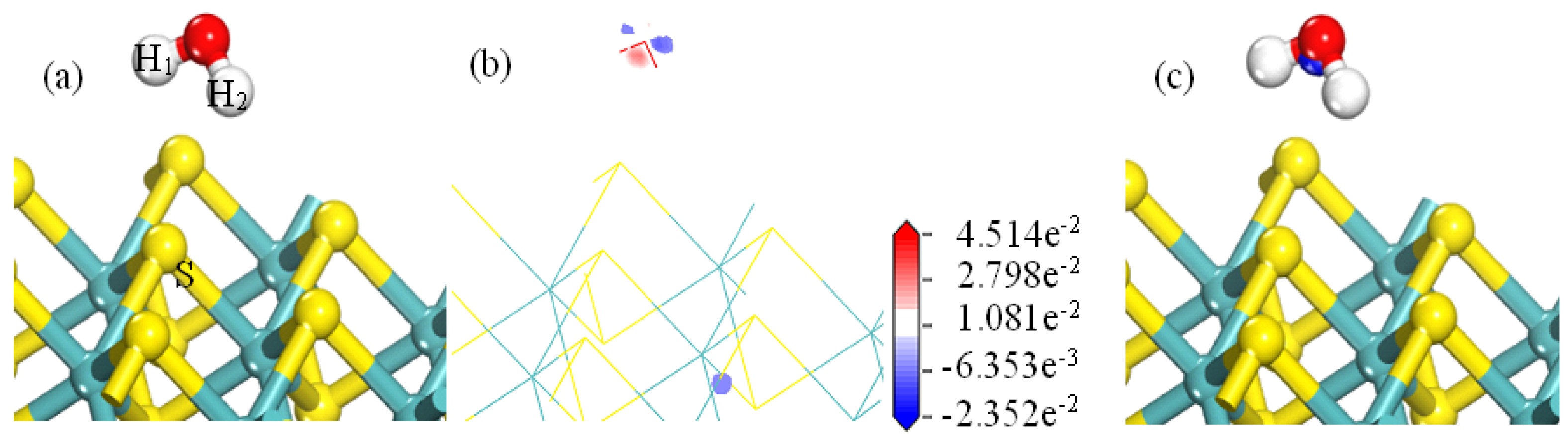
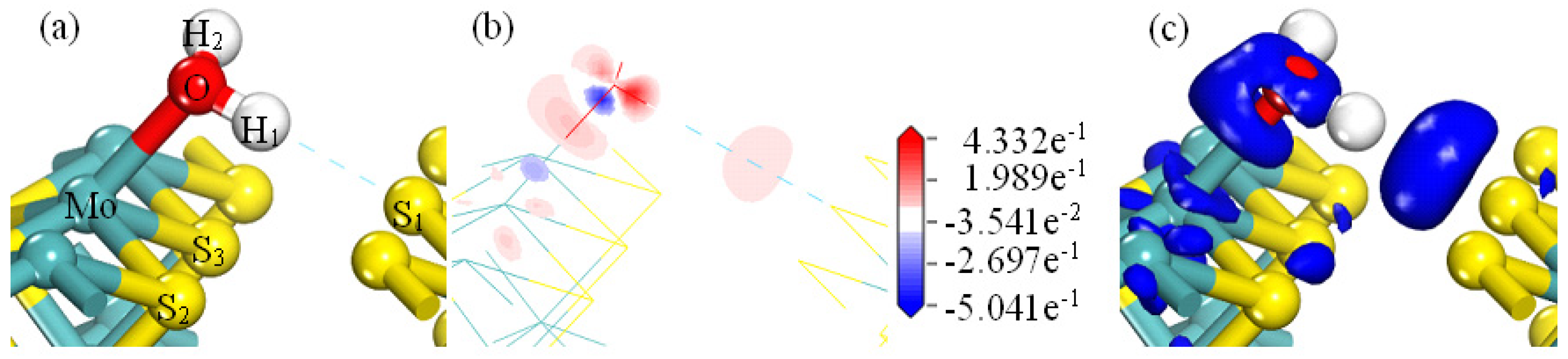
As shown in
Figure 7a, when the H
2O is adsorbed on the MS001, the water molecules are far away from the MS001. As shown in
Figure 7b, there is essentially no electron cloud density between the hydrogen atom and the S atom of MS001.
Figure 7c shows that there are essentially no electrons around the S atom of MS001. There is only weak electron aggregation between the O and H atoms in the water molecule. Combining the results in
Table 2 shows that the H atoms of the water molecules are weakly adsorbed to the S atoms on MS001. The corresponding bond lengths are 2.94672 nm and 2.93816 nm, respectively. The corresponding Mulliken bond population is zero. This is consistent with the results for the adsorption energy of water with the MS001 in
Table 1 (∆E = −8.61 kJ.mol
−1).
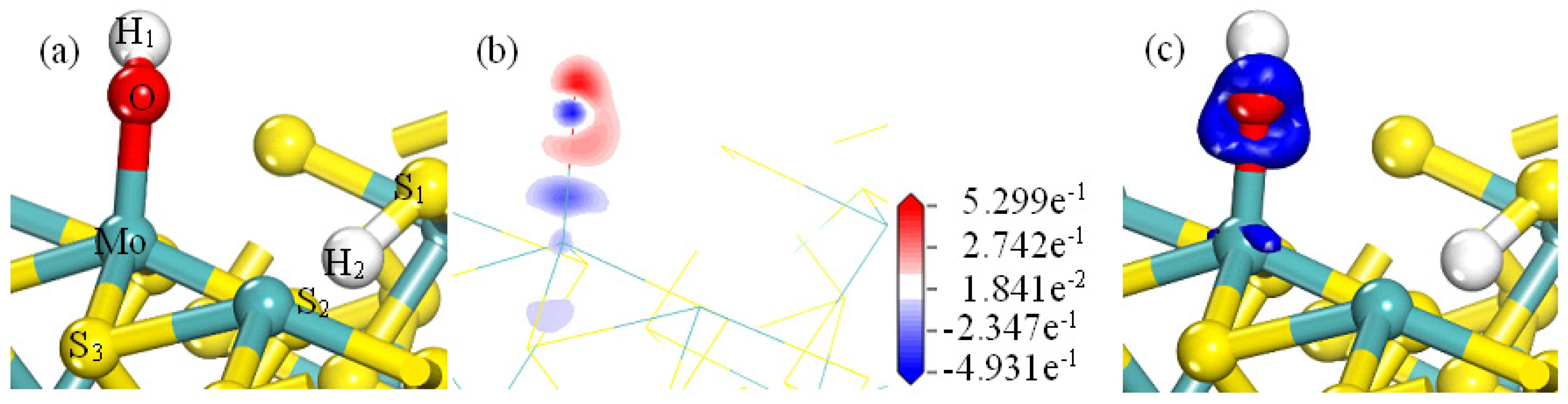
Figure 8a shows that when water molecules are adsorbed on the MS100, the water molecules are essentially located between the Mo-plane and the S-plane of MS100. Combining the results in
Table 2 shows that the H atoms of H
2O are located almost between the Mo and S surfaces of the MS100. The O atom of H
2O may be bonded to the Mo atom on MS100. H
1 forms a strong hydrogen bonding interaction with S
1 (with a sizeable covalent population). The corresponding bond lengths are 2.25993 nm and 2.34405 nm. The corresponding Mulliken bond population value is 0.10 and 0.09, respectively.
As shown in
Figure 8b, there is a high electron cloud density between the O and H atoms of the water molecule and the Mo atoms of the MS100. The corresponding population values are −0.16 and −0.17. A high charge density also exists between the H
1 atom and the S
1 atom.
Figure 8c shows that the electron cloud is located between the water molecules and the MS100. The electron aggregation occurs between the Mo atoms of MS100 and the O atoms in the water and between the hydrogen bonds formed by the H
1 and S
1 atoms. These indicate that the O/H atoms of the water molecule interact with the Mo/S atoms of MS100, respectively.
- (2)
Analysis of OH−/H+ Bonding on the MS100
The results of
Table 1 show that the interaction energies of OH
−/H
+ with MS001 are 288.30kJ·mol
−1. This indicates that OH
−/H
+ exhibits no adsorption on MS001. Therefore, only OH
−/H
+ bonding on the MS100 needs to be analyzed.
The Mulliken bond populations and their corresponding bond lengths after stable adsorption of OH
−/H
+ on the MS100 are listed in
Table 3 and
Figure 9.
Figure 9a shows that OH
− and H
+ can adsorb to the MS100 by bonding to the Mo and S atoms on MS100, respectively.
Figure 9b shows a clear charge transfer when the O atom of OH
− is bonded to the Mo atom on the MS100. The Mo atom loses its charge point, and the O atom in OH
− gains electrons. It is evident from
Figure 9c that the O atom in OH
− is surrounded by a cloud of electrons. The electrostatic adsorption between the OH
− and Mo atoms of MS100 enhances the adsorption of OH
− to the Mo-plane of MS100. The S atoms of MS100 exhibit a negative charge, which favors the adsorption of H
+ or cations on the S-plane of MS100. The results in
Table 3 further show that the O atom in OH
− can strongly bond with the Mo atom of the Mo-plane, with a population value of 0.44 and bond lengths of 1.90191 nm. The H
2 atom can also strongly bond with the S
1 atom of the S-plane, with a population value of 0.72 and bond lengths of 1.36465 nm. These indicate that strong adsorption of OH
−/H
+ occurs with MS100. This is consistent with the calculation results of adsorption energy (∆E(100)-OH
−/H
+) = −302.44 kJ·mol
−1, showing the strong hydrophilicity of MS100.
- (3)
State Density and Charge Transfer of H2O Adsorption Process
To study the variation of density of states and charge transfer during the adsorption of H
2O on the MS001/MS100, the S atom on MS001 and the H atom in H
2O molecules were selected as the research objects. The S atom on the S-plane of MS100 (MS100-S
1 in the differential density map) and the H atom in the water molecule (H
1) were selected as the research objects (bond population 0.09, interatom distance 2.34405 nm) to analyze the changes in state density and bonding before and after adsorption.
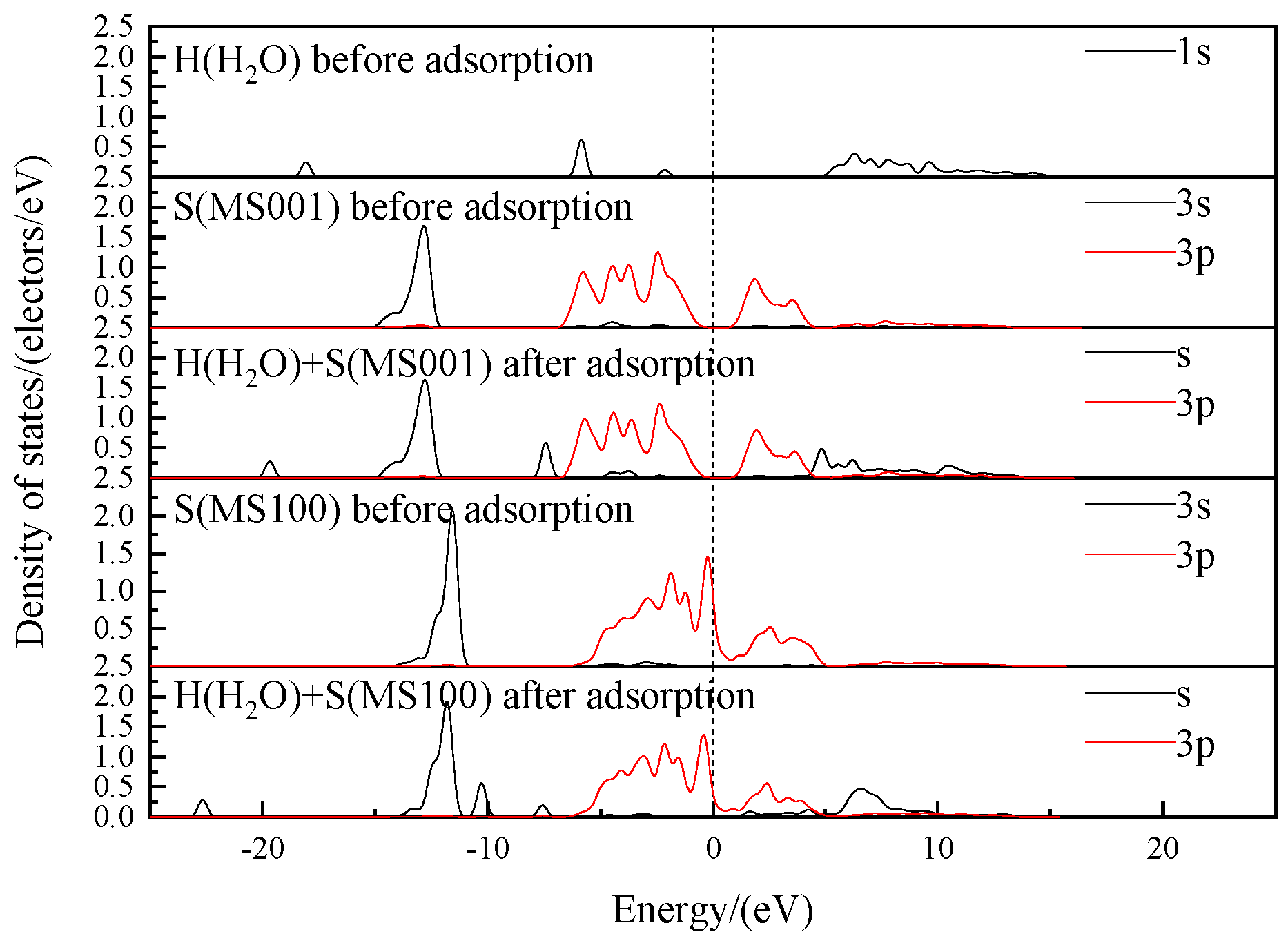
Figure 10 shows the density of states before and after the interaction of the H atoms in water with the S atoms on the molybdenite surface.
As shown in
Figure 10, after the adsorption of water molecules on MS100, the H atoms interact with the S atoms, causing a decrease in the density of electronic states near their Fermi energy levels. The overall density of states moves towards lower energy levels. This indicates that the energy of the electron orbitals decreases after the adsorption of water molecules on the MS100. Then, the system becomes stable. Meanwhile, between −7.5 and −7.1 eV are the bonding interactions between the H and S atoms, and between 5 and 8 are their antibonding interactions. Both of these interactions are weak, but the bonding interaction is stronger than the antibonding interaction. Therefore, the interaction between water molecules and molybdenite is in the form of a bonding interaction, with a bonding population of 0.09.
Table 4 and
Table 5 show the Mulliken charges of the bonding atoms involved before and after water adsorption on the MS001/MS100, respectively.
As shown in
Table 4, when the water molecules are adsorbed on the MS001, the density of states of the S atoms on the MS001 remains essentially unchanged. There is no significant charge transfer from the MS001. Combined with the results in
Figure 10, there is no charge transfer occurring in the vicinity of the O atom in the water molecule by aggregating only a small number of electrons. This suggests that water molecules do not adsorb stably or weakly on the MS001.
Table 5 shows that the 1s orbital population of the H
1 atom in the water molecule increases by 0.1, and the positive charge decreases by 0.1 e. The 3s orbital population of the S
1 atom on the S-plane of MS100 decreases by 0.02, and the 3d orbital population increases by 0.1, increasing the total charge by 0.08 e. This indicates that the H
1 atom loses electrons, and the S
1 atom gains electrons. The result is consistent with the results of
Figure 10. These indicate that water molecules can undergo stable adsorption on the MS100.
- (4)
State Density and Charge Transfer of OH− Adsorption Process
The results of
Table 1,
Figure 8, and
Figure 9 show that OH
−/H
+ exhibits no adsorption on MS001. Therefore, only the adsorption of OH
− on MS100 needs to be investigated. The O atom in OH
− and the Mo atom on the Mo-plane of MS100 were selected to analyze the density of states of the atoms before and after adsorption. The results are shown in
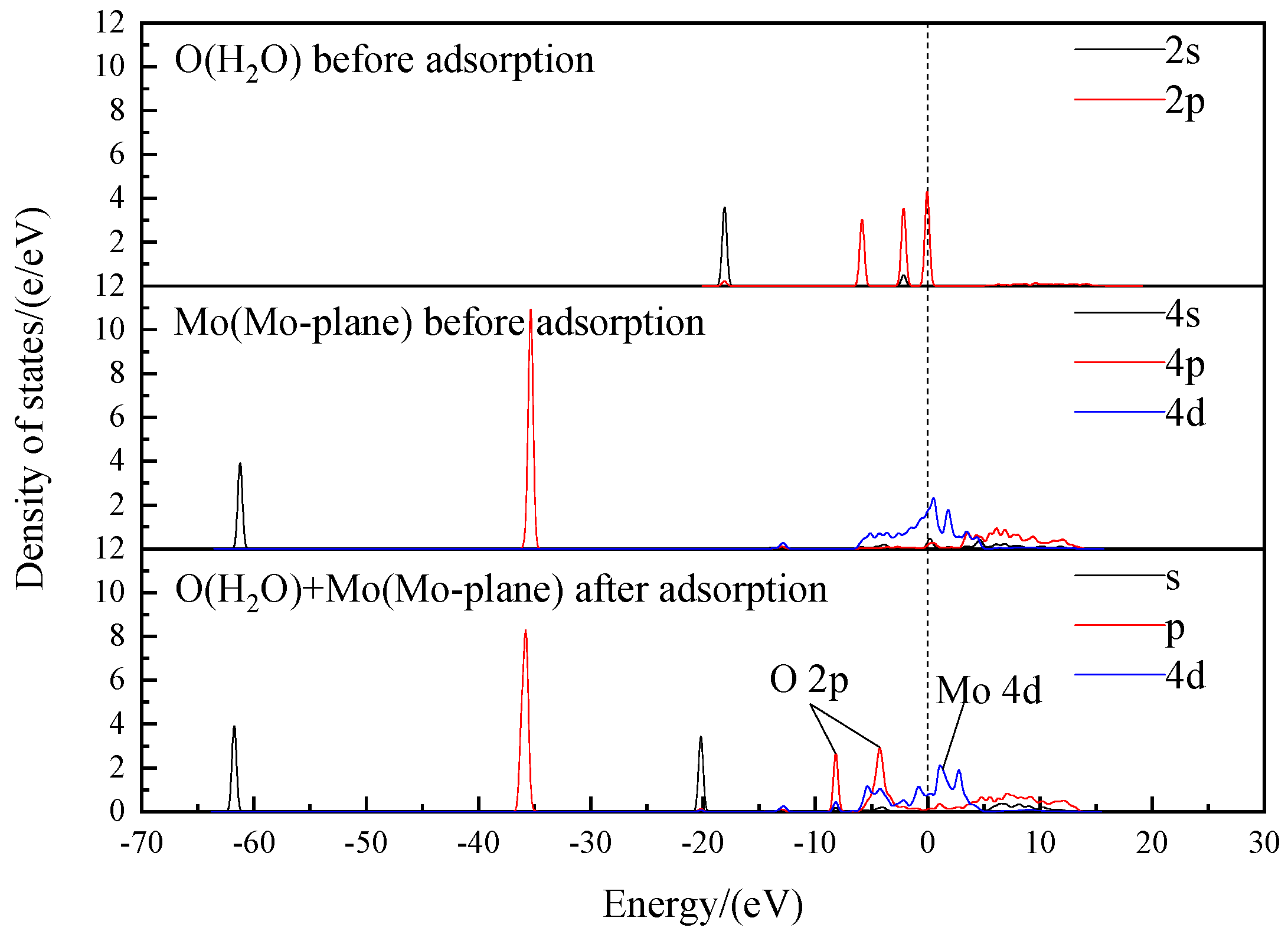
Figure 11.
As shown in
Figure 11, both the 4d orbital of the Mo atom and the 2p orbital of the O atom have a high density of states near the Fermi energy level. This indicates that both are highly active. After the interaction between the Mo atom and the O atom, the density of states of the 2p orbital of the O atom decreases and moves to the lower energy level, while the density of states of the 4p and 4d orbitals of the Mo atom decreases and the 4d orbital moves to the higher energy level, resulting in increased antibonding. The intervals of −8.6 to −7.5 eV/−6.1 to −2.5 eV are the bonding interactions between the 2p orbital of the O atom and the 4s/4d orbitals of the Mo atom, respectively.
The OH− ions can adsorb onto the Mo-plane of MS100 by chemisorption and form a hydroxy-molybdenum compound with the Mo atoms. It will cause a drop in the potential of the MS100. This result is consistent with the effect of OH− ion concentration on the zeta potential of molybdenite particles.
To obtain the charge transfer of atoms during the adsorption of OH
− on MS100, the Mulliken charge population of the directly bonded atoms was analyzed. The results of the analytical calculations are shown in
Table 6.
As shown in
Table 6, the 1s state of the H atom in OH
− gains 0.1e, and the 2p state of the O atom loses 0.25e. The 4s state of the Mo atom loses 0.01e; the 4d state loses 0.3e; the 4p state gains 0.06e; the Mo atom loses a total of 0.25e. This indicates that in the process of OH
− adsorption on MS100, in addition to the formation of hydrogen bonds between the H atoms and the S atoms of MS100, there may also be electrostatic adsorption when the OH
− is near the Mo-plane of MS100.