Magmatic-Hydrothermal Processes Associated with Rare Earth Element Enrichment in the Kangankunde Carbonatite Complex, Malawi
Abstract
:1. Introduction
2. Geology of the Kangankunde Carbonatite Complex
2.1. Geological Setting
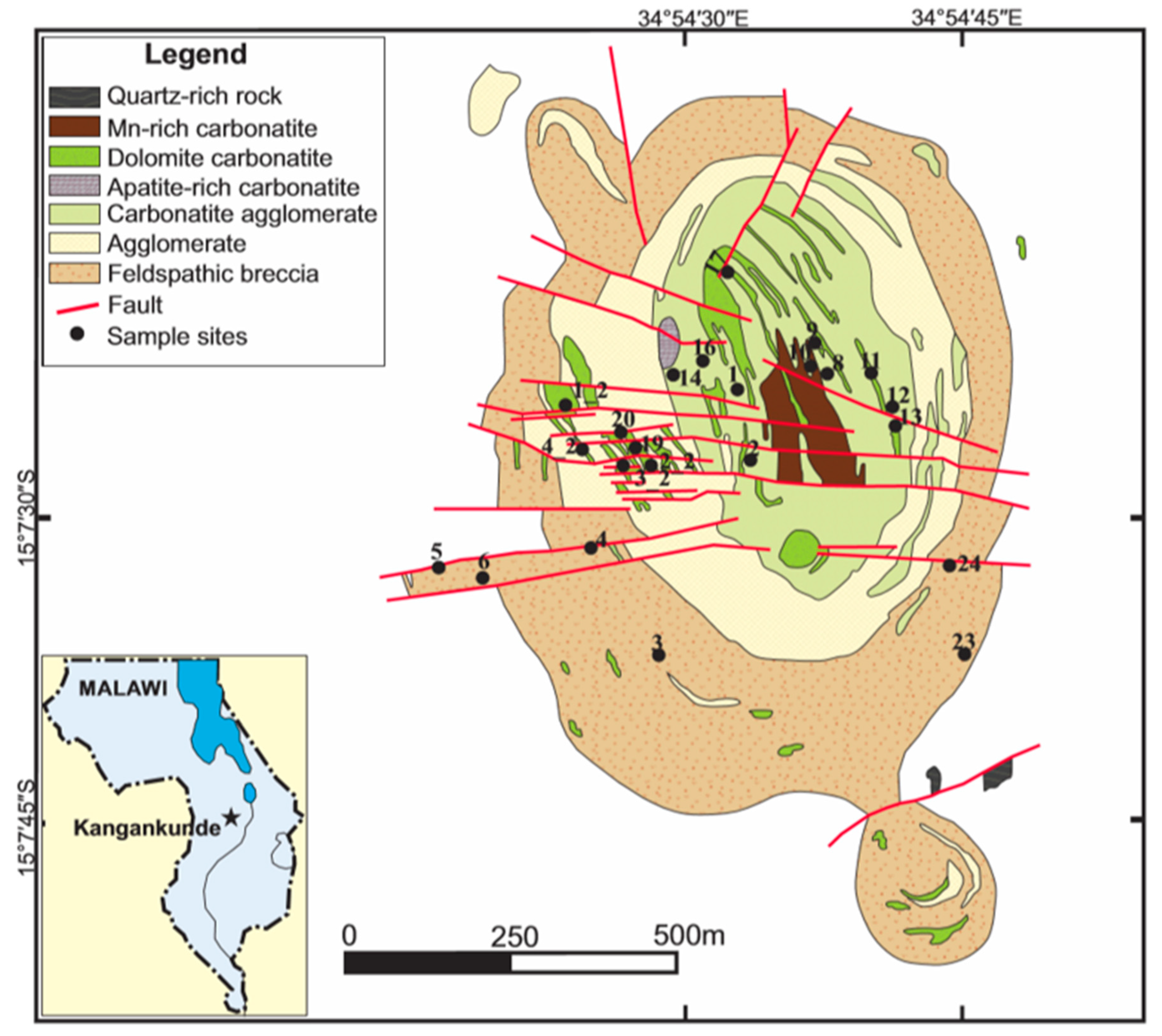
2.2. Local Geology
3. Methodology
4. Results
4.1. Whole-Rock Geochemistry
4.2. Petrography
4.3. Carbon and Oxygen Isotopes
4.4. Iron Isotopes
5. Discussion
5.1. Evolution of the Carbonatites
5.2. REE Mineralization in the Magmatic Stage
5.3. Magmatic–Hydrothermal Stages Associated with REE Enrichment
5.4. Low-Temperature Hydrothermal Overprint
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Major Element Compositions (wt. %) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Elements | Standards | Light Carbonatites | Dark Carbonatites | Apatite-Rich Carbonatites | Fenites | |||||||||||||
| JB-3 | JB-1b | JGb-1 | Ka14 | Ka20 | Ka1 | Ka4 | Ka12 | Ka17 | Ka16 | Ka19 | Ka3 | Ka8 | Ka10 | Ka11 | Ka9 | Ka6 | Ka23 | |
| SiO2 | 50.94 | 51.95 | 39.86 | 2.13 | 10.56 | 5.93 | 5.32 | 2.21 | 2.91 | 2.96 | 34.85 | 10.22 | 3.99 | 18.68 | 9.66 | 12.79 | 70.68 | 58.02 |
| TiO2 | 1.43 | 0.77 | 0.95 | b.d | 0.14 | 0.03 | 0.05 | b.d | 0.04 | 0.01 | 0.40 | 0.04 | b.d | 0.04 | 3.43 | 0.19 | 0.27 | 0.40 |
| Al2O3 | 18.20 | 14.41 | 17.35 | 0.05 | 0.59 | 0.85 | 0.52 | 0.55 | 0.47 | 0.05 | 10.19 | 1.08 | 0.14 | 0.53 | 1.62 | 1.39 | 12.39 | 13.52 |
| Fe2O3 | 7.47 | 9.03 | 14.28 | 6.67 | 11.61 | 28.92 | 25.61 | 23.25 | 10.60 | 16.02 | 9.99 | 19.23 | 13.75 | 16.14 | 28.38 | 21.78 | 3.90 | 10.20 |
| MnO | 0.18 | 0.15 | 0.18 | 2.31 | 3.49 | 6.58 | 6.97 | 5.54 | 3.82 | 5.08 | 0.62 | 4.18 | 5.15 | 4.83 | 1.44 | 3.18 | 0.11 | 0.45 |
| MgO | 5.14 | 8.28 | 7.34 | 18.09 | 16.50 | 18.40 | 6.02 | 24.29 | 20.56 | 24.15 | 8.64 | 4.05 | 13.79 | 5.82 | 12.57 | 12.19 | 0.19 | 1.57 |
| CaO | 10.78 | 9.65 | 11.40 | 34.08 | 32.68 | 26.99 | 13.61 | 35.88 | 33.12 | 38.39 | 11.86 | 44.17 | 35.52 | 21.21 | 31.38 | 28.90 | 0.26 | 3.98 |
| Na2O | 3.77 | 2.90 | 1.05 | 0.28 | 0.04 | 0.26 | 0.54 | 0.26 | 0.24 | 0.35 | 0.49 | 0.37 | 0.40 | 0.52 | 0.28 | 0.33 | 3.26 | 4.22 |
| K2O | 0.97 | 0.51 | 3.88 | 0.00 | 0.07 | 0.35 | 0.20 | 0.20 | 0.18 | 0.01 | 6.24 | 0.23 | 0.01 | 0.07 | 0.10 | 0.73 | 4.66 | 6.46 |
| P2O5 | 0.29 | 0.24 | 0.08 | 6.75 | 0.67 | 1.01 | 4.76 | 0.43 | 4.31 | 2.14 | 2.88 | 3.64 | 12.82 | 12.82 | 9.13 | 8.48 | 1.34 | 0.45 |
| SrO | 0.02 | 0.04 | 0.03 | 10.28 | 53.85 | 2.99 | 8.89 | 1.57 | 5.99 | 4.35 | 2.02 | 1.29 | 3.50 | 1.04 | 0.66 | 1.69 | 0.02 | 0.13 |
| BaO | 0.02 | 0.05 | 0.01 | 1.11 | 0.26 | 1.38 | 7.20 | 0.73 | 0.81 | 0.84 | 0.25 | 2.36 | 0.73 | 6.74 | 0.12 | 0.92 | 0.90 | 0.12 |
| TREE | 0.01 | 0.02 | 0.00 | 23.71 | 2.53 | 4.12 | 16.50 | 1.54 | 16.65 | 1.93 | 8.20 | 7.68 | 7.38 | 11.22 | 1.41 | 5.02 | 0.84 | 0.16 |
| LOI | - | - | - | 33.94 | 34.91 | 32.63 | 20.96 | 38.10 | 32.43 | 38.08 | 39.87 | 29.28 | 27.21 | 14.28 | 24.41 | 16.43 | 4.85 | 6.87 |
| Total | 99.22 | 97.99 | 96.53 | 94.06 | 78.89 | 97.08 | 86.61 | 96.67 | 92.90 | 96.67 | 94.36 | 95.65 | 95.45 | 97.28 | 99.39 | 95.10 | 98.54 | 99.74 |
| Trace Element Concentrations (ppm) | ||||||||||||||||||
| Sc | 31 | 27 | 28 | 27 | 293 | b.d | b.d | 5 | 32 | 7 | b.d | b.d | 15 | 51 | 3 | 15 | 19 | 36 |
| V | 379 | 220 | 622 | 4.01 | 51.8 | 199 | 1260 | 246 | 9 | 200 | 115 | 339 | 116 | 310 | 152 | 61.6 | 124 | 114 |
| Cr | 55 | 460 | 47 | 525 | 64 | 2 | 533 | 13 | 371 | 404 | 279 | b.d | 32 | b.d | 40 | 89 | 23 | 14 |
| Co | 36 | 39 | 57 | 18 | 21 | 17 | 12 | 12 | 13 | b.d | 15 | 10 | 15 | 23 | 32 | 0.5 | 2 | b.d |
| Ni | 36 | 143 | 18 | 235 | 276 | 140 | 300 | 110 | 161 | 195 | 128 | 146 | 109 | 115 | 84 | 116 | 11 | 15 |
| Cu | 191 | 36 | 45 | 444 | 374 | 432 | 394 | 286 | 220 | 203 | 141 | 243 | 247 | 228 | 191 | 153 | 84 | 25 |
| Zn | 158 | 77 | 114 | 2270 | 2060 | 3820 | 13,000 | 4920 | 2960 | 9280 | 2580 | 3880 | 7550 | 7110 | 658 | 4720 | 194 | 1190 |
| Rb | b.d | 298 | 153 | 709 | 2400 | 153 | 467 | 93 | 412 | 300 | 155 | 38 | 212 | 18 | b.d | 122 | 48 | 73 |
| Zr | 103 | 120 | 26 | 12,800 | b.d | 3470 | 13,300 | 1530 | 8220 | 6810 | 5180 | 2290 | 3610 | 770 | 1190 | 3060 | 215 | 18 |
| Nb | 2.5 | 6 | 4 | b.d | 3560 | 318 | 25 | b.d | 259 | 188 | 4700 | 1110 | b.d | 3 | 1650 | 647 | 83 | 170 |
| Pb | 6 | 5 | 2 | b.d | b.d | 9 | 1340 | 39 | b.d | 557 | 49 | 234 | 14 | 60 | 178 | b.d | 18 | 6 |
| Th | 4 | 10 | 2 | 73 | 925 | 6 | b.d | b.d | 136 | b.d | 468 | 73.1 | b.d | b.d | 113 | 105 | 13 | 22 |
| La | 10 | 42 | 4 | 113,000 | 12,400 | 14,000 | 45,200 | 4060 | 76,200 | 7510 | 37,700 | 29,400 | 27,400 | 32,900 | 4120 | 18,700 | 3080 | 399 |
| Ce | 24 | 734 | 9 | 58,500 | 6150 | 9980 | 39,900 | 3590 | 42,500 | 4720 | 20,500 | 20,300 | 18,000 | 29,300 | 3000 | 12,500 | 589 | 417 |
| Pr | 5 | 8 | 1 | 39,000 | 3930 | 8640 | 39,200 | 3560 | 28,800 | 3700 | 14,000 | 14,700 | 13,300 | 24,900 | 2730 | 10,200 | 2180 | 361 |
| Nd | 18 | 27 | 6 | 19,700 | 2020 | 5540 | 27,200 | 2570 | 14,500 | 2190 | 7260 | 8770 | 8490 | 16,900 | 1870 | 6080 | 1490 | 251 |
| Sm | 5 | 5 | 2 | 3600 | 423 | 1620 | 7910 | 869 | 2370 | 573 | 1370 | 1910 | 3000 | 4490 | 802 | 1490 | 456 | 83 |
| Eu | 1 | 1 | 0.6 | 1320 | 167 | 677 | 2880 | 351 | 878 | 249 | 499 | 886 | 1920 | 2100 | 453 | 567 | 261 | 50 |
| Gd | 5 | 5 | 2 | 1230 | 143 | 498 | 1860 | 230 | 879 | 205 | 429 | 493 | 1090 | 1030 | 404 | 418 | 146 | 32 |
| Tb | 0.9 | 0.8 | 0.3 | 306 | 45 | 167 | 504 | 72 | 255 | 1 | 110 | 174 | 320 | 291 | 235 | 133 | 63 | 18 |
| Dy | 5 | 4 | 2 | 89 | 21 | 50 | 167 | 29 | 92 | 36 | 37 | 81 | 106 | 117 | 150 | 58 | 36 | 11 |
| Y | 13 | 10 | 5 | 28 | 13 | 14 | 54 | 13 | 32 | 16 | 14 | 32 | 26 | 47 | 110 | 26 | 23 | 8 |
| Ho | 1. | 1 | 0.4 | 39 | 12 | 17 | 70 | 15 | 40 | 18 | 17 | 36 | 37 | 54 | 104 | 29 | 21 | 6 |
| Er | 3 | 2 | 1 | 19 | 8 | 8 | 34 | 9 | 18 | 9 | 9 | 19 | 17 | 29 | 74 | 15 | 16 | 3 |
| Tm | 0.5 | 0.4 | 0.3 | 11 | 6 | 5 | 17 | 7 | 9 | 4 | 4 | 12 | 12 | 19 | 57 | 9 | 15 | 4 |
| Yb | 3 | 2 | 1 | 6 | 4 | 3 | 9 | 6 | 4 | 3 | 2 | 9 | 7.8 | 13 | 40 | 5 | 13 | 3 |
| Lu | 0.5 | 0.4 | 0.2 | 5 | 4 | 3 | 7 | 6 | 3 | 2 | 2 | 9 | 7.5 | 12 | 32 | 5 | 13 | 4 |
| Element | 1 | 3 | 4 | 5 | 7 | 8 | 10 | 11 |
|---|---|---|---|---|---|---|---|---|
| P2O5 | 28.45 | 27.96 | 27.88 | 28.99 | 29.08 | 28.59 | 29.04 | 27.71 |
| La2O3 | 22.09 | 22.84 | 23.52 | 22.85 | 18.64 | 22.59 | 21.12 | 22.62 |
| Ce2O3 | 32.19 | 30.55 | 33.68 | 31.38 | 32.79 | 30.43 | 33.08 | 33.21 |
| Nd2O3 | 8.99 | 8.86 | 8.94 | 8.250 | 13.31 | 8.21 | 10.84 | 8.03 |
| Sm2O3 | 0.69 | 0.71 | 0.39 | 0.45 | 1.47 | 0.48 | 0.91 | 0.41 |
| Gd2O3 | 3.72 | 3.54 | 3.89 | 3.24 | 3.77 | 3.64 | 3.65 | 3.67 |
| Dy2O3 | 0.08 | 0.03 | 0.05 | 0.03 | 0.02 | 0.03 | 0.04 | 0.02 |
| Ho2O3 | 0.00 | 0.11 | 0.04 | 0.18 | 0.24 | 0.00 | 0.08 | 0.08 |
| Er2O3 | 0.00 | 0.00 | 0.00 | 0.15 | 0.08 | 0.04 | 0.00 | 0.03 |
| ThO2 | 0.16 | 0.09 | 0.19 | 0.00 | 0.22 | 0.20 | 0.00 | 0.12 |
| Pr2O3 | 0.02 | 0.00 | 0.02 | 0.01 | 0.00 | 0.03 | 0.01 | 0.04 |
| Y2O3 | 0.00 | 0.00 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| ZrO2 | 0.38 | 0.32 | 0.09 | 0.63 | 0.04 | 0.68 | 0.16 | 0.22 |
| CaO | 0.34 | 0.33 | 0.06 | 0.75 | 0.59 | 0.67 | 0.19 | 0.06 |
| Total | 97.11 | 95.36 | 98.76 | 96.93 | 100.25 | 95.59 | 99.12 | 96.23 |
| Carbonatite Sub-Group | Sample | δ13CVPDB (‰) | δ18OVSMOW (‰) |
|---|---|---|---|
| Light carbonatites | Ka20 | −3.00 | 13.61 |
| Ka14 | −2.28 | 13.65 | |
| Ka2 | −2.01 | 14.66 | |
| Dark carbonatites | Ka1 | −0.13 | 27.32 |
| Ka16 | −0.85 | 28.61 | |
| Ka12 | −0.03 | 27.25 | |
| Ka4 | −2.47 | 25.40 | |
| Ka13 | −2.47 | 26.65 | |
| Ka17 | −1.31 | 20.75 | |
| Apatite-rich carbonatites | Ka9 | −0.40 | 22.43 |
| Ka3 | −4.21 | 27.81 | |
| Ka8 | −0.91 | 22.91 | |
| Ka10 | −0.68 | 27.61 | |
| Ka11 | −2.82 | 17.31 |
| Carbonatite Subgroup | Sample | Main Fe bearing Mineral | Fe (wt. %) | Δ56Fe (‰) | 2σ |
|---|---|---|---|---|---|
| Light carbonatites | Ka20 | Ferroan Dolomite | 11.61 | −0.32 | 0.05 |
| Ka14 | Dolomite | 6.67 | −0.27 | 0.12 | |
| Ka2 | Dolomite | 3.86 | −0.38 | 0.09 | |
| Dark carbonatites | Ka12 | Dolomite | 23.25 | −0.31 | 0.06 |
| Ka4 | Ankerite | 25.61 | −0.33 | 0.05 | |
| Ka17 | Ankerite | 10.60 | −0.32 | 0.06 | |
| Ka4-2 | Ankerite | 9.86 | −0.25 | 0.05 | |
| Ka3-2 | Ankerite | 7.43 | −0.28 | 0.06 | |
| Apatite-rich carbonatites | Ka8 | Ferroan Dolomite | 13.75 | −0.33 | 0.07 |
| Ka10 | Ferroan Dolomite | 16.14 | −0.27 | 0.14 | |
| Fenites | Ka6 | Dolomite | 3.90 | −0.18 | 0.06 |
References
- Nassar, N.T.; Du, X.; Graedel, T.E. Criticality of the Rare Earth Elements. J. Ind. Ecol. 2015, 19, 1044–1054. [Google Scholar] [CrossRef]
- Chakhmouradian, A.R.; Wall, F. Rare earth elements: Minerals, mines, magnets (and more). Elements 2012, 8, 333–340. [Google Scholar] [CrossRef]
- Mariano, A.N. Economic geology of rare earth minerals. Rev. Mineral. 1989, 21, 309–337. [Google Scholar]
- Verplanck, P.L.; Mariano, A.N.; Mariano Anthony, J. Rare earth element ore geology of carbonatites. Rare Earth Elem. Crit. Elem. Ore Depos. 2016, 18, 5–32. [Google Scholar]
- Broom-Fendley, S.; Wall, F.; Spiro, B.; Ullmann, C.V. Deducing the source and composition of rare earth mineralising fluids in carbonatites: Insights from isotopic (C, O,87Sr/86Sr) data from Kangankunde, Malawi. Contrib. Mineral. Petrol. 2017, 172, 96. [Google Scholar] [CrossRef]
- Ruberti, E.; Enrich, G.E.R.; Gomes, C.B.; Comin-Chiaramonti, P. Hydrothermal REE fluorocarbonate mineralization at Barra do Itapirapuã, a multiple stockwork carbonate, southern Brazil. Can. Mineral. 2008, 46, 901–914. [Google Scholar] [CrossRef]
- Liu, Y. Geochemical and mineralogical characteristics of weathered ore in the Dalucao REE deposit, Mianning-Dechang REE Belt, western Sichuan Province, southwestern China. Ore Geol. Rev. 2015, 71, 437–456. [Google Scholar] [CrossRef]
- Broom-Fendley, S.; Styles, M.T.; Appleton, J.D.; Gunn, G.; Wall, F. Evidence for dissolution-reprecipitation of apatite and preferential LREE mobility in carbonatite-derived late-stage hydrothermal processes. Am. Mineral. 2016, 101, 596–611. [Google Scholar] [CrossRef] [Green Version]
- Ngwenya, B.T. Hydrothermal rare earth mineralisation in carbonatites of the Tundulu complex, Malawi: Processes at the fluid/rock interface. Geochim. Cosmochim. Acta 1994, 58, 2061–2072. [Google Scholar] [CrossRef]
- Johannes Giebel, R.; Gauert, C.D.K.; Marks, M.A.W.; Costin, G.; Markl, G. Multi-stage formation of REE minerals in the Palabora Carbonatite Complex, South Africa. Am. Mineral. 2017, 102, 1218–1233. [Google Scholar] [CrossRef]
- Witt, W.K.; Hammond, D.P.; Hughes, M. Geology of the Ngualla carbonatite complex, Tanzania, and origin of the Weathered Bastnaesite Zone REE ore. Ore Geol. Rev. 2019, 105, 28–54. [Google Scholar] [CrossRef]
- Hamilton, D.L.; Freestone, I.C.; Dawson, J.B.; Donaldson, C.H. Origin of carbonatites by liquid immiscibility. Nature 1979, 279, 52–54. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Viladkar, S.G.; Ripp, G.S.; Burtseva, M.V. Hydrothermal REE mineralization in the Amba Dongar carbonatite complex, Gujarat, India. Can. Mineral. 2009, 47, 1105–1116. [Google Scholar] [CrossRef]
- Moore, M.; Chakhmouradian, A.R.; Mariano, A.N.; Sidhu, R. Evolution of rare-earth mineralization in the Bear Lodge carbonatite, Wyoming: Mineralogical and isotopic evidence. Ore Geol. Rev. 2015, 64, 499–521. [Google Scholar] [CrossRef]
- Garson, M.S. Carbonatites of southern Malawi. Bull. Geol. Surv. Malawi 1965, 15, 1–128. [Google Scholar]
- Duraiswami, R.A.; Shaikh, T.N. Fluid-rock interaction in the Kangankunde Carbonatite Complex, Malawi: SEM based evidence for late stage pervasive hydrothermal mineralisation. Open Geosci. 2014, 6, 476–491. [Google Scholar] [CrossRef] [Green Version]
- Verplanck, P.L. The Role of Fluids in the Formation of Rare Earth Element Deposits. Procedia Earth Planet. Sci. 2017, 17, 758–761. [Google Scholar] [CrossRef]
- Zhou, B.; Li, Z.; Chen, C. Global Potential of Rare Earth Resources and Rare Earth Demand from Clean Technologies. Minerals 2017, 7, 203. [Google Scholar] [CrossRef]
- British Geological Survey. Mineral Potential of Malawi; Malawi SDNP: Blantyre, Malawi, 2009; pp. 1–8. [Google Scholar]
- Chen, W.; Honghui, H.; Bai, T.; Jiang, S. Geochemistry of Monazite within Carbonatite Related REE Deposits. Resources 2017, 6, 51. [Google Scholar] [CrossRef]
- Downes, P.J. Stable H–C–O isotope and trace element geochemistry of the Cummins Range Carbonatite Complex, Kimberley region, Western Australia: Implications for hydrothermal REE mineralization, carbonatite evolution and mantle source regions. Miner. Depos. 2014, 49, 905–932. [Google Scholar] [CrossRef]
- Li, X.C.; Zhou, M.F. Hydrothermal alteration of monazite-(Ce) and chevkinite-(Ce) from the Sin Quyen Fe-Cu-LREE-Au deposit, northwestern Vietnam. Am. Mineral. 2017, 102, 1525–1541. [Google Scholar] [CrossRef]
- Johnson, C.M.; Bell, K.; Beard, B.L.; Shultis, A.I. Iron isotope compositions of carbonatites record melt generation, crystallization, and late-stage volatile-transport processes. Mineral. Petrol. 2010, 98, 91–110. [Google Scholar] [CrossRef]
- Sun, J.; Zhu, X.; Chen, Y.; Fang, N. Iron isotopic constraints on the genesis of Bayan Obo ore deposit, Inner Mongolia, China. Precambrian Res. 2013, 235, 88–106. [Google Scholar] [CrossRef]
- Heimann, A.; Beard, B.L.; Johnson, C.M. The role of volatile exsolution and sub-solidus fluid/rock interactions in producing high 56Fe/54Fe ratios in siliceous igneous rocks. Geochim. Cosmochim. Acta 2008, 72, 4379–4396. [Google Scholar] [CrossRef]
- Schuessler, J.A.; Schoenberg, R.; Sigmarsson, O. Iron and lithium isotope systematics of the Hekla volcano, Iceland—Evidence for Fe isotope fractionation during magma differentiation. Chem. Geol. 2009, 258, 78–91. [Google Scholar] [CrossRef]
- Teng, F.; Dauphas, N.; Helz, R. Iron Isotope Fractionation Differentiation During Magmatic in Kilauea Iki Lava Lake. Science 2008, 320, 1620–1622. [Google Scholar] [CrossRef] [PubMed]
- Zambardi, T.; Lundstrom, C.C.; Li, X.; McCurry, M. Fe and Si isotope variations at Cedar Butte volcano; insight into magmatic differentiation. Earth Planet. Sci. Lett. 2014, 405, 169–179. [Google Scholar] [CrossRef]
- Garson, M.S.; Campbell, S.W. Carbonatite and agglomeratic vents in the western Shire Valley. Memoir. Geol. Surv. Malawi 1965, 3, 15–17. [Google Scholar]
- Woolley, A.R. Lithosphere metasomatism and the petrogenesis of the Chilwa Province of alkaline igneous rocks and carbonatites, Malawi. J. Afr. Earth Sci. 1987, 6, 891–898. [Google Scholar] [CrossRef]
- Breitenbach, S.F.M.; Bernasconi, S.M. Carbon and oxygen isotope analysis of small carbonate samples (20 to 100 μg) with a GasBench II preparation device. Rapid Commun. Mass Spectrom. 2011, 25, 1910–1914. [Google Scholar] [CrossRef]
- Kusaka, S.; Nakano, T. Carbon and oxygen isotope ratios and their temperature dependence in carbonate and tooth enamel using a GasBench II preparation device. Rapid Commun. Mass Spectrom. 2014, 28, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, T.; Makishima, A.; Nakamura, E. Evaluation of the coprecipitation of incompatible trace elements with fluoride during silicate rock dissolution by acid digestion. Chem. Geol. 1999, 157, 175–187. [Google Scholar] [CrossRef]
- Ito, A.; Otake, T.; Shin, K.C.; Ariffin, K.S.; Yeoh, F.Y.; Sato, T. Geochemical signatures and processes in a stream contaminated by heavy mineral processing near Ipoh city, Malaysia. Appl. Geochem. 2017, 82, 89–101. [Google Scholar] [CrossRef]
- Gittins, J.; Harmer, R.E. What is ferrocarbonatite? A revised classification. J. Afr. Earth Sci. 1997, 25, 159–168. [Google Scholar] [CrossRef]
- McDonough, W.; Sun, S. The composition of the earth. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
- Chai, L.; Navrotsky, A. Synthesis, characterization, and energetics of solid solution along the dolomite-ankerite join, and implications for the stability of ordered CaFe(CO3)2. Am. Mineral. 1996, 81, 1141–1147. [Google Scholar] [CrossRef]
- Beard, B.L.; Johnson, C.M. High precision iron isotope measurements of terrestrial and lunar materials. Geochim. Cosmochim. Acta 1999, 63, 1653–1660. [Google Scholar] [CrossRef]
- Shahar, A.; Young, E.D.; Manning, C.E. Equilibrium high-temperature Fe isotope fractionation between fayalite and magnetite: An experimental calibration. Earth Planet. Sci. Lett. 2008, 268, 330–338. [Google Scholar] [CrossRef] [Green Version]
- Yang, K.F.; Fan, H.R.; Santosh, M.; Hu, F.F.; Wang, K.Y. Mesoproterozoic carbonatitic magmatism in the Bayan Obo deposit, Inner Mongolia, North China: Constraints for the mechanism of super accumulation of rare earth elements. Ore Geol. Rev. 2011, 40, 122–131. [Google Scholar] [CrossRef]
- Hou, Z.; Liu, Y.; Tian, S.; Yang, Z.; Xie, Y. Formation of carbonatite-related giant rare-earth-element deposits by the recycling of marine sediments. Sci. Rep. 2015, 5, 10231. [Google Scholar] [CrossRef]
- Ling, M.X. Formation of the world’s largest REE deposit through protracted fluxing of carbonatite by subduction-derived fluids. Sci. Rep. 2013, 3, 1776. [Google Scholar] [CrossRef]
- Veksler, I.V.; Petibon, C.; Jenner, G.A.; Dorfman, A.M.; Dingwell, D.B. Trace element partitioning in immiscible silicate-carbonate liquid systems: An initial experimental study using a centrifuge autoclave. J. Petrol. 1998, 39, 2095–2104. [Google Scholar] [CrossRef]
- Wendlandt, R.F.; Harrison, W.J. Rare earth partitioning between immiscible carbonate and silicate liquids and CO2 vapor: Results and implications for the formation of light rare earth-enriched rocks. Contrib. Mineral. Petrol. 1979, 69, 409–419. [Google Scholar] [CrossRef]
- O’Nions, R.K.; Zhu, X.K. Monazite chemical composition: Some implications for monazite geochronology. Contrib. Mineral. Petrol. 1999, 137, 351–363. [Google Scholar]
- Guarino, V.; Wu, F.Y.; Melluso, L.; Gomes, C.B.; Tassinari, C.C.G.; Ruberti, E.; Brilli, M. U–Pb ages, geochemistry, C–O–Nd–Sr–Hf isotopes and petrogenesis of the Catalão II carbonatitic complex (Alto Paranaíba Igneous Province, Brazil): Implications for regional-scale heterogeneities in the Brazilian carbonatite associations. Int. J. Earth Sci. 2017, 106, 1963–1989. [Google Scholar] [CrossRef]
- Rubatto, D.; Williams, I.S.; Buick, I.S. Zircon and monazite response to prograde metamorphism in the Reynolds Range, central Australia. Contrib. Mineral. Petrol. 2001, 140, 458–468. [Google Scholar] [CrossRef]
- Zirner, A.L.K.; Marks, M.A.W.; Wenzel, T.; Jacob, D.E.; Markl, G. Rare earth elements in apatite as a monitor of magmatic and metasomatic processes: The Ilímaussaq complex, South Greenland. Lithos 2015, 228, 12–22. [Google Scholar] [CrossRef]
- Goswami-Banerjee, S.; Robyr, M. Pressure and temperature conditions for crystallization of metamorphic allanite and monazite in metapelites: A case study from the Miyar Valley (high Himalayan Crystalline of Zanskar, NW India). J. Metamorph. Geol. 2015, 33, 535–556. [Google Scholar] [CrossRef]
- Zaitsev, A.N. Rhombohedral carbonates from carbonatites of the Khibina Massif, Kola Peninsula, Russia. Can. Mineral. 1996, 34, 453–468. [Google Scholar]
- Morogan, V. Mass transfer and REE mobility during fenitization at Alnö, Sweden. Contrib. Mineral. Petrol. 1989, 103, 25–34. [Google Scholar] [CrossRef]
- Le Bas, M.J.; Xueming, Y.; Taylor, R.N.; Spiro, B.; Milton, J.A.; Peishan, Z. New evidence from a calcite-dolomite carbonatite dyke for the magmatic origin of the massive Bayan Obo ore-bearing dolomite marble, Inner Mongolia, China. Mineral. Petrol. 2007, 90, 223–248. [Google Scholar] [CrossRef]
- Cordeiro, P.F.; Brod, J.A.; Palmieri, M.; de Oliveira, C.G.; Barbosa, E.S.; Santos, R.V.; Gaspar, J.C.; Assis, L.C. The Catalão I niobium deposit, central Brazil: Resources, geology and pyrochlore chemistry. Ore Geol. Rev. 2011, 41, 112–121. [Google Scholar] [CrossRef]
- Cressey, G.; Wall, F.; Cressey, B.A. Differential REE uptake by sector growth of monazite. Mineral. Mag. 1999, 63, 813–828. [Google Scholar] [CrossRef]
- Poitrasson, F.; Freydier, R. Heavy iron isotope composition of granites determined by high resolution MC-ICP-MS. Chem. Geol. 2005, 222, 132–147. [Google Scholar] [CrossRef]
- Telus, M. Iron, zinc, magnesium and uranium isotopic fractionation during continental crust differentiation: The tale from migmatites, granitoids, and pegmatites. Geochim. Cosmochim. Acta 2012, 97, 247–265. [Google Scholar] [CrossRef]
- Welch, S.A.; Beard, B.L.; Johnson, C.M.; Braterman, P.S. Kinetic and equilibrium Fe isotope fractionation between aqueous Fe (II) and Fe (III). Geochem. Cosmochim. Acta. 2003, 67, 4231–4250. [Google Scholar] [CrossRef]
- Sossi, P.A.; Foden, J.D.; Halverson, G.P. Redox-controlled iron isotope fractionation during magmatic differentiation: An example from the Red Hill intrusion, S. Tasmania. Mineral. Petrol. 2012, 164, 757–772. [Google Scholar] [CrossRef]
- Dauphas, N. Magma redox and structural controls on iron isotope variations in Earth’s mantle and crust. Earth Planet. Sci. Lett. 2014, 398, 127–140. [Google Scholar] [CrossRef]
- Lundstrom, C. Hypothesis for the origin of convergent margin granitoids and Earth’s continental crust by thermal migration zone refining. Geochim. Cosmochim. Acta 2009, 73, 5709–5729. [Google Scholar] [CrossRef]
- Huang, F.; Chakraborty, P.; Lundstrom, C.C.; Holmden, C.; Glessner, J.J.G.; Kieffer, S.W.; Lesher, C.E. Isotope fractionation in silicate melts by thermal diffusion. Nature 2011, 464, 396–400. [Google Scholar] [CrossRef]
- Bilenker, L.D. Elucidating Igneous and Ore-Forming Processes with Iron Isotopes by Using Experimental and Field-Based Methods. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 2015. [Google Scholar]
- Schöpa, A.; Annen, C. The effects of magma flux variations on the formation and lifetime of large silicic magma chambers. J. Geophys. Res. Solid Earth 2013, 118, 926–942. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.P.; Genge, M.; Carmody, L. Carbonate Melts and Carbonatites. Rev. Mineral. Geochem. 2013, 75, 289–322. [Google Scholar] [CrossRef] [Green Version]
- Ray, J.S.; Shukla, A.D.; Dewangan, L.K. Carbon and oxygen isotopic compositions of Newania Dolomite Carbonatites, Rajasthan, India: Implications for source of carbonatites. Mineral. Petrol. 2010, 98, 269–282. [Google Scholar] [CrossRef]
- Ray, J.S.; Ramesh, R. Rayleigh fractionation of stable isotopes from a multicomponent source. Geochim. Cosmochim. Acta 2000, 64, 299–306. [Google Scholar] [CrossRef]









© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chikanda, F.; Otake, T.; Ohtomo, Y.; Ito, A.; Yokoyama, T.D.; Sato, T. Magmatic-Hydrothermal Processes Associated with Rare Earth Element Enrichment in the Kangankunde Carbonatite Complex, Malawi. Minerals 2019, 9, 442. https://doi.org/10.3390/min9070442
Chikanda F, Otake T, Ohtomo Y, Ito A, Yokoyama TD, Sato T. Magmatic-Hydrothermal Processes Associated with Rare Earth Element Enrichment in the Kangankunde Carbonatite Complex, Malawi. Minerals. 2019; 9(7):442. https://doi.org/10.3390/min9070442
Chicago/Turabian StyleChikanda, Frances, Tsubasa Otake, Yoko Ohtomo, Akane Ito, Takaomi D. Yokoyama, and Tsutomu Sato. 2019. "Magmatic-Hydrothermal Processes Associated with Rare Earth Element Enrichment in the Kangankunde Carbonatite Complex, Malawi" Minerals 9, no. 7: 442. https://doi.org/10.3390/min9070442






