Molecular Genetics in Epstein–Barr Virus-Associated Malignancies
Abstract
:1. Introduction
2. Material and Methods
3. Genomic Structure
4. Infection Cycle
5. EBV Associated Human Diseases
6. Infectious Mononucleosis (IM)
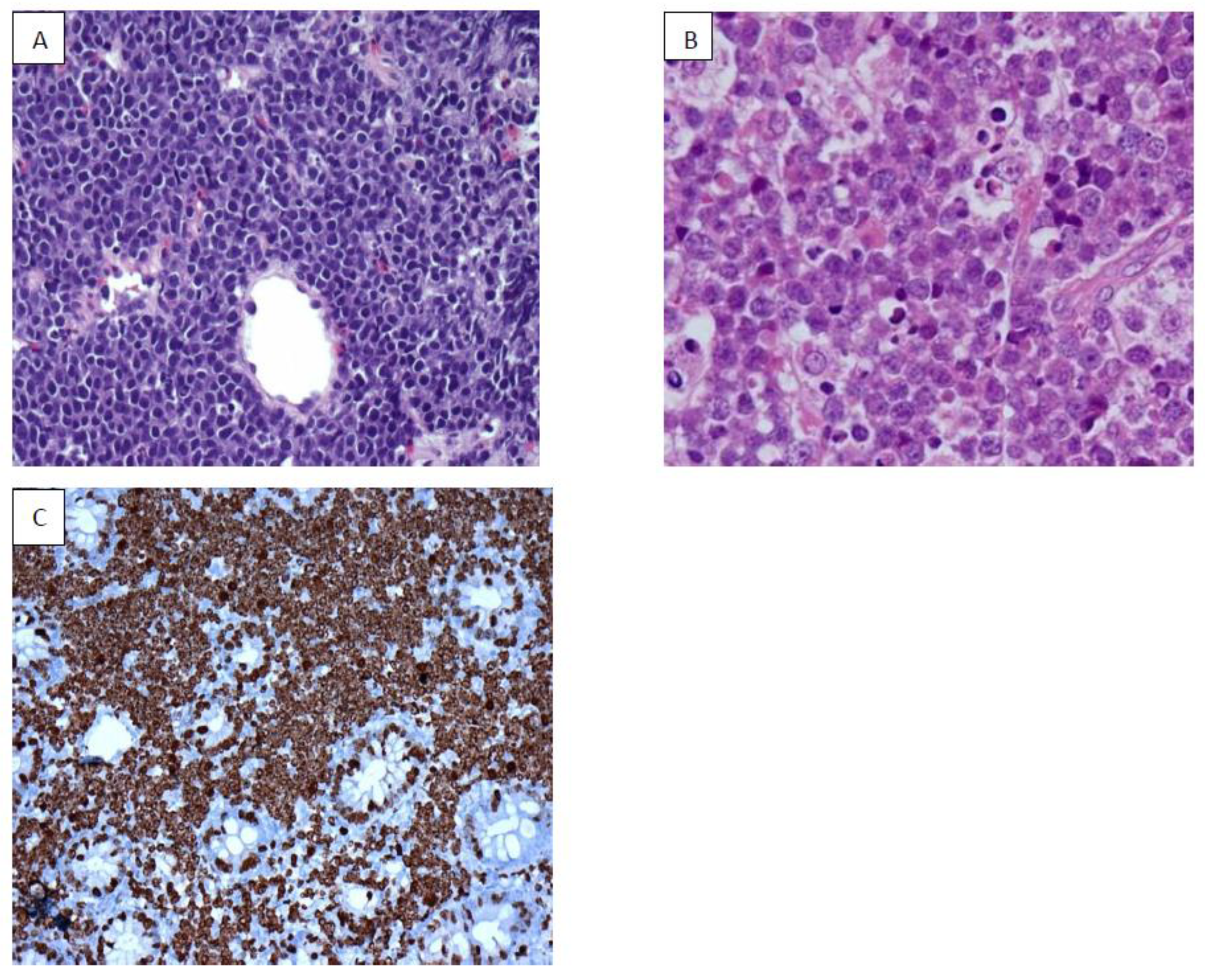
7. Burkitt Lymphoma (BL)
8. Hodgkin’s Lymphoma (HL)
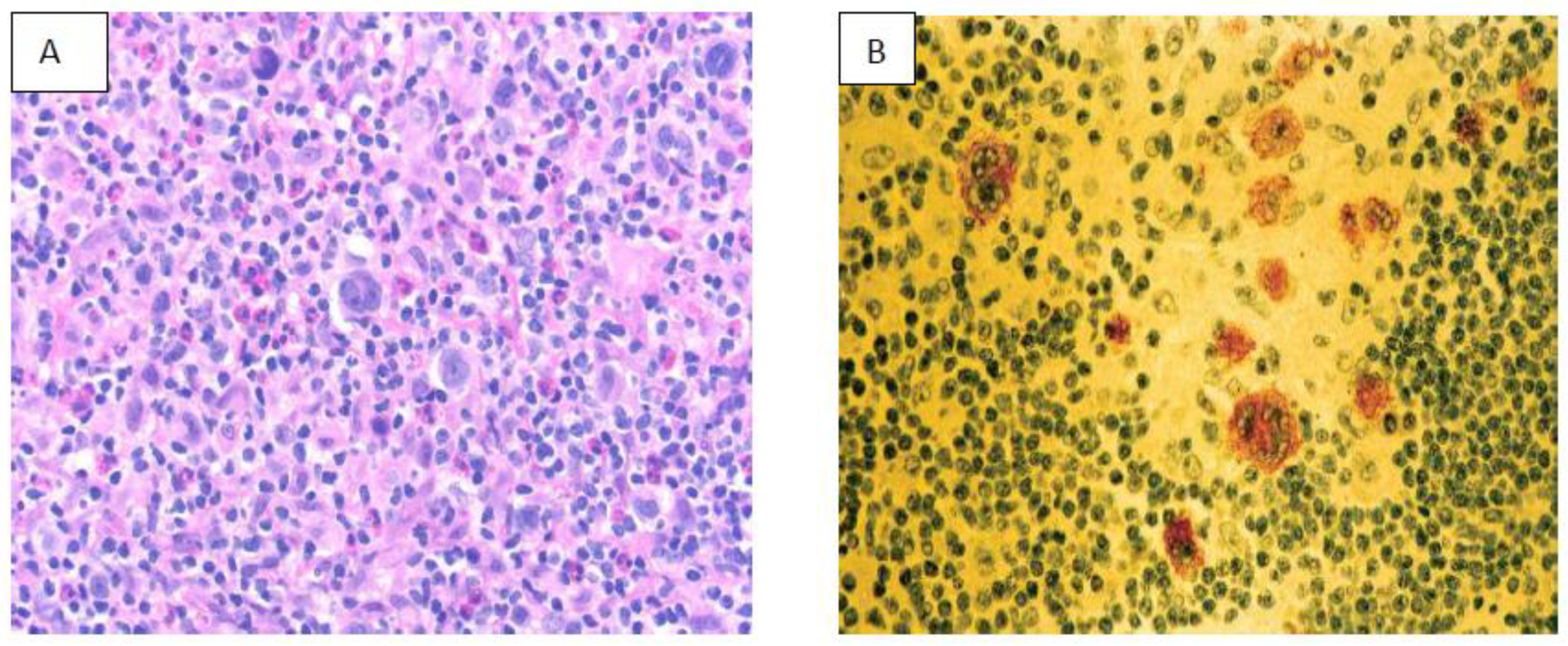
9. NK/T Cell Lymphoma (NKTCL)
10. Nasopharyngeal Carcinoma (NPC)
11. EBV-Associated Gastric Ca (EBVaGCs)
12. EBV Related Breast Cancer (EBVrBCa)
13. Genetic Targeted Pharmacological Strategies in EBV Associated Malignancies
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hjalgrim, H.; Friborg, J.; Melbye, M. The Epidemiology of EBV and its Association with Malignant Disease. In Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis; Arvin, A., Campadelli-Fiume, G., Mocarski, E., Moore, S.P., Roizman, B., Whitley, R., Yamanishi, K., Eds.; Cambridge University Press: Cambridge, UK, 2007; Chapter 53. Available online: https://www.ncbi.nlm.nih.gov/books/NBK47424/ (accessed on 22 May 2021).
- Abdel-Hamid, M.; Chen, J.-J.; Constantine, N.; Massoud, M.; Raab-Traub, N. EBV strain variation: Geographical distribution and relation to disease state. Virology 1992, 190, 168–175. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Biological Agents. In (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 100B.) EPSTEIN-BARR VIRUS; International Agency for Research on Cancer: Lyon, France, 2012. Available online: https://www.ncbi.nlm.nih.gov/books/NBK304353/ (accessed on 22 May 2021).
- Raab-Traub, N. EBV-induced oncogenesis. In Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis; Arvin, A., Campadelli-Fiume, G., Mocarski, E., Moore, S.P., Roizman, B., Whitley, R., Yamanishi, K., Eds.; Cambridge University Press: Cambridge, UK, 2007; Chapter 55. Available online: https://www.ncbi.nlm.nih.gov/books/NBK47429/ (accessed on 22 May 2021).
- Keating, S.; Prince, S.; Jones, M.; Rowe, M. The Lytic Cycle of Epstein-Barr Virus Is Associated with Decreased Expression of Cell Surface Major Histocompatibility Complex Class I and Class II Molecules. J. Virol. 2002, 76, 8179–8188. [Google Scholar] [CrossRef] [Green Version]
- Shannon-Lowe, C.; Rickinson, A.B.; Bell, A.I. Epstein–Barr virus-associated lymphomas. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160271. [Google Scholar] [CrossRef]
- Münz, M. Dendritic cells during Epstein Barr virus infection. Front. Microbiol. 2014, 5, 308. [Google Scholar] [CrossRef]
- Young, L.S.; Arrand, J.R.; Murray, P.G. EBV gene expression and regulation. In Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis; Arvin, A., Campadelli-Fiume, G., Mocarski, E., Moore, S.P., Roizman, B., Whitley, R., Yamanishi, K., Eds.; Cambridge University Press: Cambridge, UK, 2007; Chapter 27. Available online: https://www.ncbi.nlm.nih.gov/books/NBK47431/ (accessed on 22 May 2021).
- Sunagawa, K.; Hishima, T.; Fukumoto, H.; Hasegawa, H.; Katano, H. Conserved sequences of BART and BHRF regions encoding viral microRNAs in Epstein-Barr virus-associated lymphoma. BMC Res. Notes 2017, 10, 279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, E.R.; Lamers, S.L.; Henderson, J.L.; Melnikov, A.; Somasundaran, M.; Garber, M.; Selin, L.; Nusbaum, C.; Luzuriaga, K. Early Epstein-Barr Virus Genomic Diversity and Convergence toward the B95.8 Genome in Primary Infection. J. Virol. 2018, 92, e01466-17. [Google Scholar] [CrossRef] [Green Version]
- Lei, H.; Li, T.; Hung, G.-C.; Li, B.; Tsai, S.; Lo, S.-C. Identification and characterization of EBV genomes in spontaneously immortalized human peripheral blood B lymphocytes by NGS technology. BMC Genom. 2013, 14, 804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwok, H.; Tong, A.H.Y.; Lin, C.H.; Lok, S.; Farrell, P.; Kwong, D.L.W.; Chiang, A.K.S. Genomic Sequencing and Comparative Analysis of Epstein-Barr Virus Genome Isolated from Primary Nasopharyngeal Carcinoma Biopsy. PLoS ONE 2012, 7, e36939. [Google Scholar] [CrossRef] [Green Version]
- Abate, F.; Ambrosio, M.R.; Mundo, L.; Laginestra, M.A.; Fuligni, F.; Rossi, M.; Zairis, S.; Gazaneo, S.; De Falco, G.; Lazzi, S.; et al. Distinct Viral and Mutational Spectrum of Endemic Burkitt Lymphoma. PLoS Pathog. 2015, 11, e1005158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wohlford, E.M.; Baresel, P.C.; Wilmore, J.R.; Mortelliti, A.J.; Coleman, C.B.; Rochford, R. Changes in Tonsil B Cell Phenotypes and EBV Receptor Expression in Children under 5-Years-Old. Cytom. Part B Clin. Cytom. 2018, 94, 291–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jog, N.; McClain, M.T.; Heinlen, L.D.; Gross, T.; Towner, R.; Guthridge, J.M.; Axtell, R.C.; Pardo, G.; Harley, J.B.; James, J.A. Epstein Barr virus nuclear antigen 1 (EBNA-1) peptides recognized by adult multiple sclerosis patient sera induce neurologic symptoms in a murine model. J. Autoimmun. 2020, 106, 102332. [Google Scholar] [CrossRef] [PubMed]
- Frappier, L. The Epstein-Barr Virus EBNA1 Protein. Scientifica 2012, 2012, 438204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De França, T.R.T.; Carvalho, A.D.A.T.; Gomes, V.B.; Gueiros, L.A.; Porter, S.R.; Leão, J.C. Salivary shedding of Epstein–Barr virus and cytomegalovirus in people infected or not by human immunodeficiency virus 1. Clin. Oral Investig. 2011, 16, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Houen, G.; Trier, N.H. Epstein-Barr Virus and Systemic Autoimmune Diseases. Front. Immunol. 2021, 11, 7380. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.-H. EBV and human cancer. Exp. Mol. Med. 2015, 47, e130. [Google Scholar] [CrossRef] [Green Version]
- Balfour, H.H., Jr.; Dunmire, S.K.; Hogquist, K.A. Infectious mononucleosis. Clin. Transl. Immunol. 2015, 4, e33. [Google Scholar] [CrossRef]
- Aslan, N.; Watkin, L.B.; Gil, A.; Mishra, R.; Clark, F.G.; Welsh, R.M.; Ghersi, D.; Luzuriaga, K.; Selin, L.K. Severity of Acute Infectious Mononucleosis Correlates with Cross-Reactive Influenza CD8 T-Cell Receptor Repertoires. MBio 2017, 8, e01841-17. [Google Scholar] [CrossRef] [Green Version]
- Gequelin, L.C.F.; Riediger, I.N.; Nakatani, S.M.; Biondo, A.W.; Bonfim, C.M. Epstein-Barr virus: General factors, virus-related diseases and measurement of viral load after transplant. Rev Bras Hematol Hemoter. Rev. Bras. Hematol. Hemoter. 2011, 33, 383–388. [Google Scholar] [CrossRef]
- Houldcroft, C.J.; Kellam, P. Host genetics of Epstein–Barr virus infection, latency and disease. Rev. Med. Virol. 2015, 25, 71–84. [Google Scholar] [CrossRef] [Green Version]
- Li, F.-Y.; Chaigne-Delalande, B.; Kanellopoulou, C.; Davis, J.C.; Matthews, H.F.; Douek, D.C.; Cohen, J.I.; Uzel, G.; Su, H.C.; Lenardo, M.J. Second messenger role for Mg2+ revealed by human T-cell immunodeficiency. Nature 2011, 475, 471–476. [Google Scholar] [CrossRef]
- Fugl, A.; Andersen, C.L. Epstein-Barr virus and its association with disease—A review of relevance to general practice. BMC Fam. Pract. 2019, 20, 62. [Google Scholar] [CrossRef] [PubMed]
- Marshall-Andon, T.; Heinz, P. How to use … the Monospot and other heterophile antibody tests. Arch. Dis. Child. Educ. Pract. Ed. 2017, 102, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Sohn, M.J.; Cho, J.M.; Moon, J.S.; Ko, J.S.; Yang, H.R. EBV VCA IgM and cytomegalovirus IgM dual positivity is a false positive finding related to age and hepatic involvement of primary Epstein–Barr virus infection in children. Medicine 2018, 97, e12380. [Google Scholar] [CrossRef] [PubMed]
- Stanfield, B.A.; Luftig, M.A. Recent advances in understanding Epstein-Barr virus. F1000Research 2017, 6, 386. [Google Scholar] [CrossRef]
- Hämmerl, L.; Colombet, M.; Rochford, R.; Ogwang, D.M.; Parkin, D.M. The burden of Burkitt lymphoma in Africa. Infect. Agents Cancer 2019, 14, 17. [Google Scholar] [CrossRef] [Green Version]
- Kalisz, K.; Alessandrino, F.; Beck, R.; Smith, D.; Kikano, E.; Ramaiya, N.H.; Tirumani, S.H. An update on Burkitt lymphoma: A review of pathogenesis and multimodality imaging assessment of disease presentation, treatment response, and recurrence. Insights Imaging 2019, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Bouska, A.; Bi, C.; Lone, W.; Zhang, W.; Kedwaii, A.; Heavican, T.; Lachel, C.M.; Yu, J.; Ferro, R.; Eldorghamy, N.; et al. Adult high-grade B-cell lymphoma with Burkitt lymphoma signature: Genomic features and potential therapeutic targets. Blood 2017, 130, 1819–1831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Umakanthan, S.; Bukelo, M.M. Concise genetic profile of lung carcinoma. Postgrad Med. J. 2021. [Google Scholar] [CrossRef]
- Yu, L.; Yu, T.-T.; Young, K.H. Cross-talk between Myc and p53 in B-cell lymphomas. Chronic Dis. Transl. Med. 2019, 5, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.-S.; Ye, H.; Du, M.-Q.; Lu, C.-L.; Dogan, A.; Hsieh, P.-P.; Huang, W.-T.; Jung, Y.-C. Histopathology and Immunohistochemistry in Distinguishing Burkitt Lymphoma from Diffuse Large B-Cell Lymphoma with Very High Proliferation Index and with or without a Starry-Sky Pattern: A comparative study with EBER and FISH. Am. J. Clin. Pathol. 2007, 128, 558–564. [Google Scholar] [CrossRef]
- Oosten, L.E.M.; Chamuleau, M.E.D.; Thielen, F.W.; De Wreede, L.C.; Siemes, C.; Doorduijn, J.K.; Smeekes, O.S.; Kersten, M.J.; Hardi, L.; Baars, J.W.; et al. Treatment of sporadic Burkitt lymphoma in adults, a retrospective comparison of four treatment regimens. Ann. Hematol. 2018, 97, 255–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitz, R.; Young, R.M.; Ceribelli, M.; Jhavar, S.; Xiao, W.; Zhang, M.; Wright, G.L.; Shaffer, A.L.; Hodson, D.J.; Buras, E.; et al. Burkitt lymphoma pathogenesis and therapeutic targets from structural and functional genomics. Nat. Cell Biol. 2012, 490, 116–120. [Google Scholar] [CrossRef]
- Stone, M.J. Thomas Hodgkin: Medical Immortal and Uncompromising Idealist. In Baylor University Medical Center Proceedings; Informa UK Limited: London, UK, 2005; Volume 18, pp. 368–375. [Google Scholar] [CrossRef] [Green Version]
- Salati, M.; Cesaretti, M.; Macchia, M.; El Mistiri, M.; Federico, M. Epidemiological Overview of Hodgkin Lymphoma across the Mediterranean Basin. Mediterr. J. Hematol. Infect. Dis. 2014, 6, e2014048. [Google Scholar] [CrossRef] [Green Version]
- Massini, G.; Siemer, D.; Hohaus, S. EBV in Hodgkin Lymphoma. Mediterr. J. Hematol. Infect. Dis. 2009, 1, e2009013. [Google Scholar] [CrossRef]
- Arruga, F.; Vaisitti, T.; Deaglio, S. The NOTCH Pathway and Its Mutations in Mature B Cell Malignancies. Front. Oncol. 2018, 8, 550. [Google Scholar] [CrossRef] [Green Version]
- Vrzalikova, K.; Sunmonu, T.; Reynolds, G.; Murray, P. Contribution of Epstein–Barr Virus Latent Proteins to the Pathogenesis of Classical Hodgkin Lymphoma. Pathogens 2018, 7, 59. [Google Scholar] [CrossRef] [Green Version]
- Tan, G.W.; Visser, L.; Tan, L.P.; Berg, A.V.D.; Diepstra, A. The Microenvironment in Epstein–Barr Virus-Associated Malignancies. Pathogens 2018, 7, 40. [Google Scholar] [CrossRef] [Green Version]
- Schmitz, R.; Hansmann, M.-L.; Bohle, V.; Martin-Subero, J.I.; Hartmann, S.; Mechtersheimer, G.; Klapper, W.; Vater, I.; Giefing, M.; Gesk, S.; et al. TNFAIP3 (A20) is a tumor suppressor gene in Hodgkin lymphoma and primary mediastinal B cell lymphoma. J. Exp. Med. 2009, 206, 981–989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carbone, A.; Vaccher, E.; Gloghini, A.; Pantanowitz, L.; Abayomi, A.; De Paoli, P.; Franceschi, S. Diagnosis and management of lymphomas and other cancers in HIV-infected patients. Nat. Rev. Clin. Oncol. 2014, 11, 223–238. [Google Scholar] [CrossRef]
- Lima, M. Aggressive mature natural killer cell neoplasms: From epidemiology to diagnosis. Orphanet J. Rare Dis. 2013, 8, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avilés, A. Nasal NK/T-Cell Lymphoma. A Comparative Analysis of a Mexican Population with the other Populations of Latin-America. Mediterr. J. Hematol. Infect. Dis. 2015, 7, e2015052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortés, J.R.; Palomero, T. Biology and Molecular Pathogenesis of Mature T-Cell Lymphomas. Cold Spring Harb. Perspect. Med. 2021, 11, a035402. [Google Scholar] [CrossRef] [PubMed]
- Ayee, R.; Ofori, M.E.O.; Wright, E.; Quaye, O. Epstein Barr Virus Associated Lymphomas and Epithelia Cancers in Humans. J. Cancer 2020, 11, 1737–1750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gantuz, M.; Lorenzetti, M.A.; Chabay, P.A.; Preciado, M.V. A novel recombinant variant of latent membrane protein 1 from Epstein Barr virus in Argentina denotes phylogeographical association. PLoS ONE 2017, 12, e0174221. [Google Scholar] [CrossRef] [PubMed]
- Parri, E.; Kuusanmäki, H.; Bulanova, D.; Mustjoki, S.; Wennerberg, K. Selective drug combination vulnerabilities in STAT3- and TP53-mutant malignant NK cells. Blood Adv. 2021, 5, 1862–1875. [Google Scholar] [CrossRef]
- Al Shawabkeh, M.A.; Al Sulaiti, M.; Al Sa’Ey, H.; Ganesan, S. Nasal Type Extranodal Natural Killer/T (NK/T) Cell Lymphoma Presenting as Periorbital Cellulitis: A Case Report. Am. J. Case Rep. 2016, 17, 934–938. [Google Scholar] [CrossRef] [Green Version]
- Chan, S.-C.; Yeh, C.-H.; Chang, J.; Chang, K.-P.; Wang, J.-H.; Ng, S.-H. Combing MRI Perfusion and 18F-FDG PET/CT Metabolic Biomarkers Helps Predict Survival in Advanced Nasopharyngeal Carcinoma: A Prospective Multimodal Imaging Study. Cancers 2021, 13, 1550. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Miyazaki, K. Current treatment approaches for NK/T-cell lymphoma. J. Clin. Exp. Hematop. 2017, 57, 98–108. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Yuan, B.; Wu, H.; Chu, H.; Liu, Y.; Wu, S.; Li, H.; Lu, H.; Chen, H. Comparative Study of L-Asparaginase-Based LOP Regimen Over CHOP Regimen before Radiotherapy for Stage IIE Extranodal Nasal Type NK/T Cell Lymphoma: A Study of 2 Centers. Clin. Lymphoma Myeloma Leuk. 2017, 17, 152–158. [Google Scholar] [CrossRef]
- Xue, E.A.W.; Zhang, M. Updating targets for natural killer/T-cell lymphoma immunotherapy. Cancer Biol. Med. 2021, 18, 52–62. [Google Scholar] [CrossRef]
- Chang, E.T.; Adami, H.-O. The Enigmatic Epidemiology of Nasopharyngeal Carcinoma. Cancer Epidemiol. Biomark. Prev. 2006, 15, 1765–1777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inha, S.; Gajra, A. Nasopharyngeal Cancer. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Fernandes, Q.; Merhi, M.; Raza, A.; Inchakalody, V.P.; Abdelouahab, N.; Gul, A.R.Z.; Uddin, S.; Dermime, S. Role of Epstein–Barr Virus in the Pathogenesis of Head and Neck Cancers and Its Potential as an Immunotherapeutic Target. Front. Oncol. 2018, 8, 257. [Google Scholar] [CrossRef] [PubMed]
- Banko, A.V.; Lazarevic, I.; Folic, M.M.; Djukic, V.B.; Cirkovic, A.M.; Karalic, D.Z.; Cupic, M.; Jovanovic, T.P. Characterization of the Variability of Epstein-Barr Virus Genes in Nasopharyngeal Biopsies: Potential Predictors for Carcinoma Progression. PLoS ONE 2016, 11, e0153498. [Google Scholar] [CrossRef] [PubMed]
- Smatti, M.K.; Al-Sadeq, D.W.; Ali, N.H.; Pintus, G.; Abou-Saleh, H.; Nasrallah, G.K. Epstein–Barr Virus Epidemiology, Serology, and Genetic Variability of LMP-1 Oncogene Among Healthy Population: An Update. Front. Oncol. 2018, 8, 211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsao, S.W.; Tsang, C.M.; Lo, K.W. Epstein–Barr virus infection and nasopharyngeal carcinoma. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160270. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Guo, T.; Wang, T.; Zhen, Y.; Ma, X.; Wang, Y.; Zhang, Z.-X.; Cai, J.-P.; Mao, W.; Zhu, F.-M.; et al. HLA-B*07, HLA-DRB1*07, HLA-DRB1*12, and HLA-C*03:02 Strongly Associate with BMI: Data from 1.3 Million Healthy Chinese Adults. Diabetes 2018, 67, 861–871. [Google Scholar] [CrossRef] [Green Version]
- Lung, R.W.-M.; Hau, P.-M.; Yu, K.H.-O.; Yip, K.Y.; Tong, J.H.-M.; Chak, W.-P.; Chan, A.W.-H.; Lam, K.-H.; Lo, A.K.-F.; Tin, E.K.-Y.; et al. EBV-encoded miRNAs target ATM-mediated response in nasopharyngeal carcinoma. J. Pathol. 2018, 244, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Li, C.; Pan, L. Nasopharyngeal carcinoma: A review of current updates. Exp. Ther. Med. 2018, 15, 3687–3692. [Google Scholar] [CrossRef] [Green Version]
- Yu, X.; Li, F.; Cheng, W.; Wu, B.; Fang, H.; Xia, F.; Gong, Y.; Yu, W.; Liao, P.; Cao, Y.; et al. Efficacy of Chemiluminescence Immunoassays on VCA-IgA and EBNA1-IgA Antibodies of Epstein-Barr Virus in Diagnosing Nasopharyngeal Carcinoma. J. Cancer 2020, 11, 7176–7183. [Google Scholar] [CrossRef] [PubMed]
- Iizasa, H.; Nanbo, A.; Nishikawa, J.; Jinushi, M.; Yoshiyama, H. Epstein-Barr Virus (EBV)-associated Gastric Carcinoma. Viruses 2012, 4, 3420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nogueira, C.; Mota, M.; Gradiz, R.; Cipriano, M.A.; Caramelo, F.; Cruz, H.; Alarcão, A.; E Sousa, F.C.; Oliveira, F.; Martinho, F.; et al. Prevalence and characteristics of Epstein–Barr virus-associated gastric carcinomas in Portugal. Infect. Agents Cancer 2017, 12, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anyndita, N.V.M.; Dluha, N.; Rifa’I, M.; Himmah, K.; Wahyuningsih, M.D. Widodo Designing and overproducing a tandem epitope of gp350/220 that shows a potential to become an EBV vaccine. Heliyon 2018, 4, e00564. [Google Scholar] [CrossRef] [Green Version]
- Stanland, L.J.; Luftig, M.A. The Role of EBV-Induced Hypermethylation in Gastric Cancer Tumorigenesis. Viruses 2020, 12, 1222. [Google Scholar] [CrossRef]
- Takahashi-Kanemitsu, A.; Knight, C.T.; Hatakeyama, M. Molecular anatomy and pathogenic actions of Helicobacter pylori CagA that underpin gastric carcinogenesis. Cell. Mol. Immunol. 2020, 17, 50–63. [Google Scholar] [CrossRef] [Green Version]
- Nishikawa, J.; Iizasa, H.; Yoshiyama, H.; Shimokuri, K.; Kobayashi, Y.; Sasaki, S.; Nakamura, M.; Yanai, H.; Sakai, K.; Suehiro, Y.; et al. Clinical Importance of Epstein–Barr Virus-Associated Gastric Cancer. Cancers 2018, 10, 167. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Okabe, A.; Usui, G.; Fukuyo, M.; Matsusaka, K.; Rahmutulla, B.; Mano, Y.; Hoshii, T.; Funata, S.; Hiura, N.; et al. Activation of EHF via STAT3 phosphorylation by LMP2A in Epstein-Barr virus–positive gastric cancer. Cancer Sci. 2021. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, L.; Yang, Y.; Luo, B. Screening and identification of key genes in EBV-associated gastric carcinoma based on bioinformatics analysis. Pathol. Res. Pract. 2021, 222, 153439. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.-L.; Liu, H. Gastric adenocarcinoma mimicking a submucosal tumor: A case report. World J. Clin. Cases 2019, 7, 3138–3144. [Google Scholar] [CrossRef]
- Cho, J.; Kang, M.-S.; Kim, K.-M. Epstein-Barr Virus-Associated Gastric Carcinoma and Specific Features of the Accompanying Immune Response. J. Gastric Cancer 2016, 16, 1–7. [Google Scholar] [CrossRef] [Green Version]
- De Re, V.; Brisotto, G.; Repetto, O.; De Zorzi, M.; Caggiari, L.; Zanussi, S.; Alessandrini, L.; Canzonieri, V.; Miolo, G.; Puglisi, F.; et al. Overview of Epstein–Barr-Virus-Associated Gastric Cancer Correlated with Prognostic Classification and Development of Therapeutic Options. Int. J. Mol. Sci. 2020, 21, 9400. [Google Scholar] [CrossRef]
- Naseem, M.; Barzi, A.; Brezden-Masley, C.; Puccini, A.; Berger, M.D.; Tokunaga, R.; Battaglin, F.; Soni, S.; McSkane, M.; Zhang, W.; et al. Outlooks on Epstein-Barr virus associated gastric cancer. Cancer Treat. Rev. 2018, 66, 15–22. [Google Scholar] [CrossRef]
- Ginsburg, O.; Bray, F.; Coleman, M.P.; Vanderpuye, V.; Eniu, A.; Kotha, S.R.; Sarker, M.; Huong, T.T.; Allemani, C.; Dvaladze, A.; et al. The global burden of women’s cancers: A grand challenge in global health. Lancet 2017, 389, 847–860. [Google Scholar] [CrossRef]
- Godet, I.; Gilkes, D.M. BRCA1 and BRCA2 mutations and treatment strategies for breast cancer. Integr. Cancer Sci. Ther. 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Lawson, J.S.; Heng, B. Viruses and Breast Cancer. Cancers 2010, 2, 752. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Y.; Wang, H.-B.; Zhang, A.; Chen, M.-L.; Fang, Z.-X.; Dong, X.-D.; Li, S.-B.; Du, Y.; Xiong, D.; et al. Ephrin receptor A2 is an epithelial cell receptor for Epstein–Barr virus entry. Nat. Microbiol. 2018, 3, 164–171. [Google Scholar] [CrossRef]
- Hammerl, D.; Smid, M.; Timmermans, A.; Sleijfer, S.; Martens, J.; Debets, R. Breast cancer genomics and immuno-oncological markers to guide immune therapies. Semin. Cancer Biol. 2018, 52 Pt 2, 178–188. [Google Scholar] [CrossRef]
- Hu, H.; Luo, M.-L.; Desmedt, C.; Nabavi, S.; Yadegarynia, S.; Hong, A.; Konstantinopoulos, P.A.; Gabrielson, E.; Hines-Boykin, R.; Pihan, G.; et al. Epstein–Barr Virus Infection of Mammary Epithelial Cells Promotes Malignant Transformation. EBioMedicine 2016, 9, 148–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suppan, C.; Posch, F.; Mueller, H.; Mischitz, N.; Steiner, D.; Klocker, E.; Setaffy, L.; Bargfrieder, U.; Hammer, R.; Hauser, H.; et al. Patterns of Recurrence after Neoadjuvant Therapy in Early Breast Cancer, according to the Residual Cancer Burden Index and Reductions in Neoadjuvant Treatment Intensity. Cancers 2021, 13, 2492. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Sato, Y.; Al Masud, H.M.A.; Takayama, M.; Matsuda, H.; Hara, Y.; Yanagi, Y.; Yoshida, M.; Goshima, F.; Murata, T.; et al. Antitumor activity of cyclin-dependent kinase inhibitor alsterpaullone in Epstein-Barr virus-associated lymphoproliferative disorders. Cancer Sci. 2020, 111, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Shi, F.; Li, Y.; Li, W.; Yu, X.; Zhao, L.; Zhou, M.; Hu, J.; Luo, X.; Tang, M.; et al. Drp1-dependent remodeling of mitochondrial morphology triggered by EBV-LMP1 increases cisplatin resistance. Signal Transduct. Target. Ther. 2020, 5, 56. [Google Scholar] [CrossRef]
- Pei, Y.; Wong, J.H.Y.; Robertson, E.S. Targeted Therapies for Epstein-Barr Virus-Associated Lymphomas. Cancers 2020, 12, 2565. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; He, C.; Mao, Z.-R. Epstein-Barr virus interactions with the Bcl-2 protein family and apoptosis in human tumor cells. J. Zhejiang Univ. Sci. B 2013, 14, 8–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sejic, N.; George, L.C.; Tierney, R.J.; Chang, C.; Kondrashova, O.; MacKinnon, R.N.; Lan, P.; Bell, A.I.; Lessene, G.; Long, H.M.; et al. BCL-XL inhibition by BH3-mimetic drugs induces apoptosis in models of Epstein-Barr virus–associated T/NK-cell lymphoma. Blood Adv. 2020, 4, 4775–4787. [Google Scholar] [PubMed]
- Burslem, G.M.; Crews, C.M. Small-Molecule Modulation of Protein Homeostasis. Chem. Rev. 2017, 117, 11269–11301. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.C.; Crews, C.M. Induced protein degradation: An emerging drug discovery paradigm. Nat. Rev. Drug Discov. 2017, 16, 101–114. [Google Scholar] [CrossRef] [Green Version]
- Anastasiadou, E.; Stroopinsky, D.; Alimperti, S.; Jiao, A.L.; Pyzer, A.R.; Cippitelli, C.; Pepe, G.; Severa, M.; Rosenblatt, J.; Etna, M.P.; et al. Epstein−Barr virus-encoded EBNA2 alters immune checkpoint PD-L1 expression by downregulating miR-34a in B-cell lymphomas. Leukemia 2019, 33, 132–147. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umakanthan, S.; Bukelo, M.M. Molecular Genetics in Epstein–Barr Virus-Associated Malignancies. Life 2021, 11, 593. https://doi.org/10.3390/life11070593
Umakanthan S, Bukelo MM. Molecular Genetics in Epstein–Barr Virus-Associated Malignancies. Life. 2021; 11(7):593. https://doi.org/10.3390/life11070593
Chicago/Turabian StyleUmakanthan, Srikanth, and Maryann M Bukelo. 2021. "Molecular Genetics in Epstein–Barr Virus-Associated Malignancies" Life 11, no. 7: 593. https://doi.org/10.3390/life11070593
APA StyleUmakanthan, S., & Bukelo, M. M. (2021). Molecular Genetics in Epstein–Barr Virus-Associated Malignancies. Life, 11(7), 593. https://doi.org/10.3390/life11070593





