Autophagy Pathways in the Genesis of Plasmodium-Derived Microvesicles: A Double-Edged Sword?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice and Parasites
2.2. Confocal and Transmission Electron Microscopy
2.3. Quantification of Gene Expression
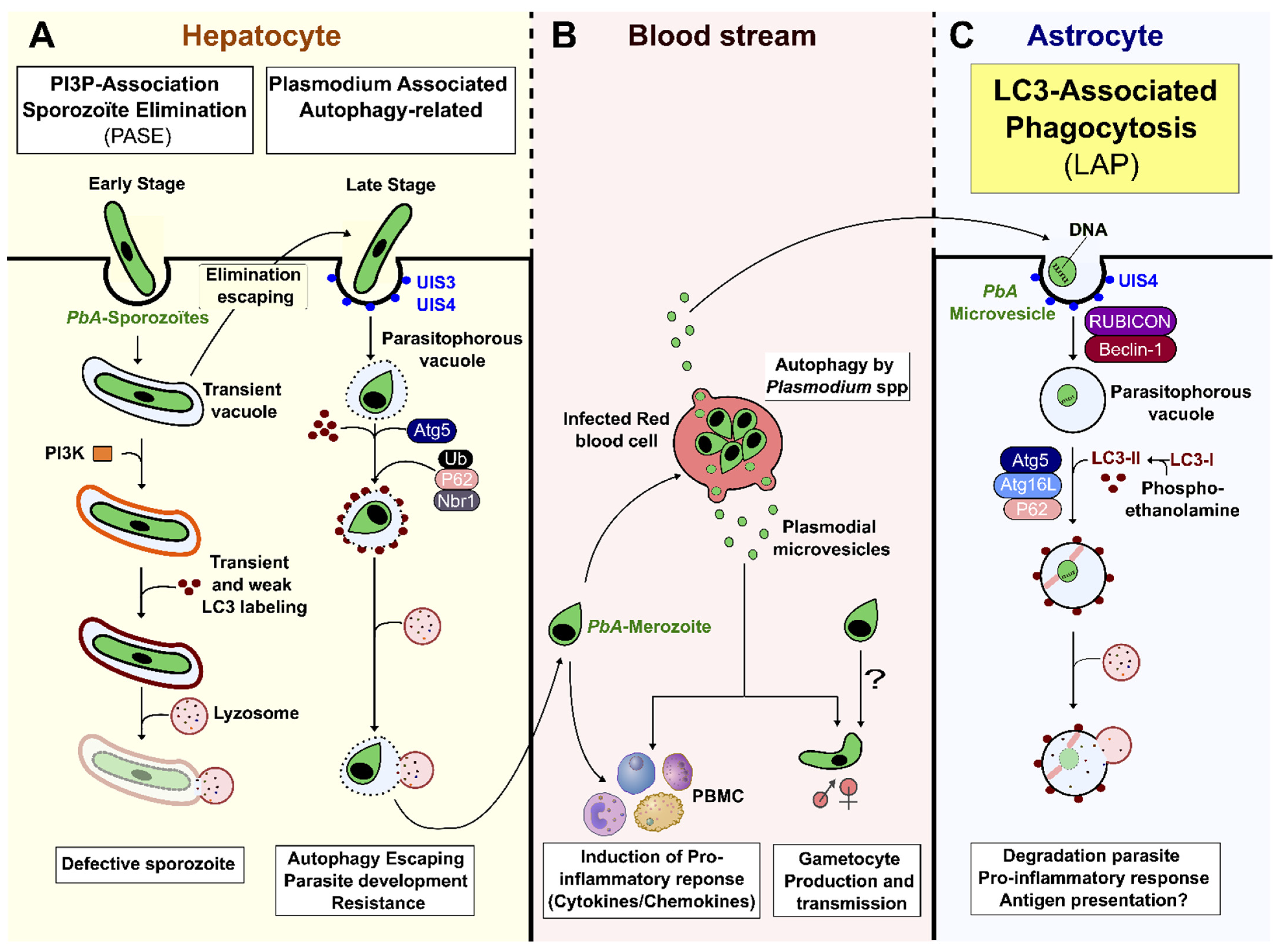
3. Results
3.1. Host Autophagy Pathways in Malaria
3.2. pMVs and Autophagy-Related Responses during Pre-Erythrocytic Development
3.3. Autophagy and pMV Crosstalk during Blood-Stage Infection
3.4. Role of Autophagy-Dependent pMVs in CM
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- World Health Organization. World Malaria Report 2020: 20 Years of Global Progress and Challenges; World Health Organization: Geneva, Switzerland, 2020; ISBN 978-92-4-001579-1. [Google Scholar]
- Frischknecht, F.; Matuschewski, K. Plasmodium Sporozoite Biology. Cold Spring Harb. Perspect. Med. 2017, 7, a025478. [Google Scholar] [CrossRef] [Green Version]
- Shears, M.J.; Sekhar Nirujogi, R.; Swearingen, K.E.; Renuse, S.; Mishra, S.; Jaipal Reddy, P.; Moritz, R.L.; Pandey, A.; Sinnis, P. Proteomic Analysis of Plasmodium Merosomes: The Link between Liver and Blood Stages in Malaria. J. Proteome Res. 2019, 18, 3404–3418. [Google Scholar] [CrossRef] [PubMed]
- Franke-Fayard, B.; Fonager, J.; Braks, A.; Khan, S.M.; Janse, C.J. Sequestration and Tissue Accumulation of Human Malaria Parasites: Can We Learn Anything from Rodent Models of Malaria? PLoS Pathog. 2010, 6, e1001032. [Google Scholar] [CrossRef] [Green Version]
- Lüder, C.G.K.; Stanway, R.R.; Chaussepied, M.; Langsley, G.; Heussler, V.T. Intracellular Survival of Apicomplexan Parasites and Host Cell Modification. Int. J. Parasitol. 2009, 39, 163–173. [Google Scholar] [CrossRef]
- Nyboer, B.; Heiss, K.; Mueller, A.-K.; Ingmundson, A. The Plasmodium Liver-Stage Parasitophorous Vacuole: A Front-Line of Communication between Parasite and Host. Int. J. Med. Microbiol. 2018, 308, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Deretic, V.; Levine, B. Autophagy, Immunity, and Microbial Adaptations. Cell Host Microbe 2009, 5, 527–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubinsztein, D.C.; Bento, C.F.; Deretic, V. Therapeutic Targeting of Autophagy in Neurodegenerative and Infectious Diseases. J. Exp. Med. 2015, 212, 979–990. [Google Scholar] [CrossRef]
- Mizushima, N.; Yoshimori, T.; Ohsumi, Y. The Role of Atg Proteins in Autophagosome Formation. Annu. Rev. Cell Dev. Biol. 2011, 27, 107–132. [Google Scholar] [CrossRef]
- Dikic, I.; Elazar, Z. Mechanism and Medical Implications of Mammalian Autophagy. Nat. Rev. Mol. Cell Biol. 2018, 19, 349–364. [Google Scholar] [CrossRef]
- He, C.; Klionsky, D.J. Regulation Mechanisms and Signaling Pathways of Autophagy. Annu. Rev. Genet. 2009, 43, 67–93. [Google Scholar] [CrossRef] [Green Version]
- Levine, B.; Mizushima, N.; Virgin, H.W. Autophagy in Immunity and Inflammation. Nature 2011, 469, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Deretic, V.; Saitoh, T.; Akira, S. Autophagy in Infection, Inflammation and Immunity. Nat. Rev. Immunol. 2013, 13, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, N.G.; Cheng, L.; Eriksson, E.M. The Role of Extracellular Vesicles in Malaria Biology and Pathogenesis. Malar. J. 2017, 16, 245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Z.; Wang, L.; Li, J.; Wang, L.; Wu, Z.; Sun, X. Extracellular Vesicle-Mediated Communication Within Host-Parasite Interactions. Front. Immunol. 2019, 9, 3066. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular Vesicles: Exosomes, Microvesicles, and Friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell. Mol. Neurobiol. 2016, 36, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Debs, S.; Cohen, A.; Hosseini-Beheshti, E.; Chimini, G.; Hunt, N.H.; Grau, G.E.R. Interplay of Extracellular Vesicles and Other Players in Cerebral Malaria Pathogenesis. Biochim. Biophys. Acta BBA-Gen. Subj. 2019, 1863, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Mantel, P.-Y.; Hoang, A.N.; Goldowitz, I.; Potashnikova, D.; Hamza, B.; Vorobjev, I.; Ghiran, I.; Toner, M.; Irimia, D.; Ivanov, A.R.; et al. Malaria-Infected Erythrocyte-Derived Microvesicles Mediate Cellular Communication within the Parasite Population and with the Host Immune System. Cell Host Microbe 2013, 13, 521–534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Combes, V.; Coltel, N.; Faille, D.; Wassmer, S.C.; Grau, G.E. Cerebral Malaria: Role of Microparticles and Platelets in Alterations of the Blood–Brain Barrier. Int. J. Parasitol. 2006, 36, 541–546. [Google Scholar] [CrossRef]
- Faille, D.; Combes, V.; Mitchell, A.J.; Fontaine, A.; Juhan-Vague, I.; Alessi, M.-C.; Chimini, G.; Fusaï, T.; Grau, G.E. Platelet Microparticles: A New Player in Malaria Parasite Cytoadherence to Human Brain Endothelium. FASEB J. 2009, 23, 3449–3458. [Google Scholar] [CrossRef] [PubMed]
- Combes, V.; Coltel, N.; Alibert, M.; van Eck, M.; Raymond, C.; Juhan-Vague, I.; Grau, G.E.; Chimini, G. ABCA1 Gene Deletion Protects against Cerebral Malaria. Am. J. Pathol. 2005, 166, 295–302. [Google Scholar] [CrossRef]
- El-Assaad, F.; Wheway, J.; Hunt, N.H.; Grau, G.E.R.; Combes, V. Production, Fate and Pathogenicity of Plasma Microparticles in Murine Cerebral Malaria. PLoS Pathog. 2014, 10, e1003839. [Google Scholar] [CrossRef] [Green Version]
- Babatunde, K.A.; Yesodha Subramanian, B.; Ahouidi, A.D.; Martinez Murillo, P.; Walch, M.; Mantel, P.-Y. Role of Extracellular Vesicles in Cellular Cross Talk in Malaria. Front. Immunol. 2020, 11, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexopoulou, L.; Holt, A.C.; Medzhitov, R.; Flavell, R.A. Recognition of Double-Stranded RNA and Activation of NF-ΚB by Toll-like Receptor 3. Nature 2001, 413, 732–738. [Google Scholar] [CrossRef]
- Keswani, T.; Roland, J.; Herbert, F.; Delcroix-Genete, D.; Bauderlique-Le Roy, H.; Gaayeb, L.; Cazenave, P.-A.; Pied, S. Expression of CD300lf by Microglia Contributes to Resistance to Cerebral Malaria by Impeding the Neuroinflammation. Genes Immun. 2020, 21, 45–62. [Google Scholar] [CrossRef]
- Leleu, I.; Genete, D.; Desnoulez, S.S.; Saidi, N.; Brodin, P.; Lafont, F.; Tomavo, S.; Pied, S. A Noncanonical Autophagy Is Involved in the Transfer of Plasmodium-Microvesicles to Astrocytes. Autophagy 2021, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Coppens, I. Metamorphoses of Malaria: The Role of Autophagy in Parasite Differentiation. Essays Biochem. 2011, 51, 127–136. [Google Scholar] [CrossRef]
- Tsukada, M.; Ohsumi, Y. Isolation and Characterization of Autophagy-Defective Mutants of Saccharomyces cerevisiae. FEBS Lett. 1993, 333, 169–174. [Google Scholar] [CrossRef] [Green Version]
- Hara, T.; Takamura, A.; Kishi, C.; Iemura, S.; Natsume, T.; Guan, J.-L.; Mizushima, N. FIP200, a ULK-Interacting Protein, Is Required for Autophagosome Formation in Mammalian Cells. J. Cell Biol. 2008, 181, 497–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, S.; Arasteh, M.; Jefferson, M.; Pearson, T.; Wang, Y.; Zhang, W.; Bicsak, B.; Divekar, D.; Powell, P.P.; Naumann, R.; et al. The ATG5-Binding and Coiled Coil Domains of ATG16L1 Maintain Autophagy and Tissue Homeostasis in Mice Independently of the WD Domain Required for LC3-Associated Phagocytosis. Autophagy 2019, 15, 599–612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mota, M.M. Migration of Plasmodium Sporozoites Through Cells Before Infection. Science 2001, 291, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Loubens, M.; Vincensini, L.; Fernandes, P.; Briquet, S.; Marinach, C.; Silvie, O. Plasmodium Sporozoites on the Move: Switching from Cell Traversal to Productive Invasion of Hepatocytes. Mol. Microbiol. 2021, 115, 870–881. [Google Scholar] [CrossRef] [PubMed]
- Latré de Laté, P.; Pineda, M.; Harnett, M.; Harnett, W.; Besteiro, S.; Langsley, G. Apicomplexan Autophagy and Modulation of Autophagy in Parasite-Infected Host Cells. Biomed. J. 2017, 40, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Agop-Nersesian, C.; De Niz, M.; Niklaus, L.; Prado, M.; Eickel, N.; Heussler, V.T. Shedding of Host Autophagic Proteins from the Parasitophorous Vacuolar Membrane of Plasmodium Berghei. Sci. Rep. 2017, 7, 2191. [Google Scholar] [CrossRef] [Green Version]
- Wacker, R.; Eickel, N.; Schmuckli-Maurer, J.; Annoura, T.; Niklaus, L.; Khan, S.M.; Guan, J.-L.; Heussler, V.T. LC3-Association with the Parasitophorous Vacuole Membrane of Plasmodium Berghei Liver Stages Follows a Noncanonical Autophagy Pathway. Cell. Microbiol. 2017, 19, e12754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Real, E.; Rodrigues, L.; Cabal, G.G.; Enguita, F.J.; Mancio-Silva, L.; Mello-Vieira, J.; Beatty, W.; Vera, I.M.; Zuzarte-Luís, V.; Figueira, T.N.; et al. Plasmodium UIS3 Sequesters Host LC3 to Avoid Elimination by Autophagy in Hepatocytes. Nat. Microbiol. 2018, 3, 17–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niklaus, L.; Agop-Nersesian, C.; Schmuckli-Maurer, J.; Wacker, R.; Grünig, V.; Heussler, V.T. Deciphering Host Lysosome-Mediated Elimination of Plasmodium Berghei Liver Stage Parasites. Sci. Rep. 2019, 9, 7967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bindschedler, A.; Wacker, R.; Egli, J.; Eickel, N.; Schmuckli-Maurer, J.; Franke-Fayard, B.M.; Janse, C.J.; Heussler, V.T. Plasmodium Berghei Sporozoites in Nonreplicative Vacuole Are Eliminated by a PI3P-mediated Autophagy-independent Pathway. Cell. Microbiol. 2021, 23, e13271. [Google Scholar] [CrossRef] [PubMed]
- Prado, M.; Eickel, N.; De Niz, M.; Heitmann, A.; Agop-Nersesian, C.; Wacker, R.; Schmuckli-Maurer, J.; Caldelari, R.; Janse, C.J.; Khan, S.M.; et al. Long-Term Live Imaging Reveals Cytosolic Immune Responses of Host Hepatocytes against Plasmodium Infection and Parasite Escape Mechanisms. Autophagy 2015, 11, 1561–1579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmuckli-Maurer, J.; Reber, V.; Wacker, R.; Bindschedler, A.; Zakher, A.; Heussler, V.T. Inverted Recruitment of Autophagy Proteins to the Plasmodium Berghei Parasitophorous Vacuole Membrane. PLoS ONE 2017, 12, e0183797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boonhok, R.; Rachaphaew, N.; Duangmanee, A.; Chobson, P.; Pattaradilokrat, S.; Utaisincharoen, P.; Sattabongkot, J.; Ponpuak, M. LAP-like Process as an Immune Mechanism Downstream of IFN-γ in Control of the Human Malaria Plasmodium vivax Liver Stage. Proc. Natl. Acad. Sci. USA 2016, 113, E3519–E3528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, Z.; Klionsky, D.J. Plasmodium Protein UIS3 Protects the Parasite from Autophagy Clearance. Autophagy 2018, 14, 1291–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopes da Silva, M.; Thieleke-Matos, C.; Cabrita-Santos, L.; Ramalho, J.S.; Wavre-Shapton, S.T.; Futter, C.E.; Barral, D.C.; Seabra, M.C. The Host Endocytic Pathway Is Essential for Plasmodium Berghei Late Liver Stage Development: Plasmodium Interaction with Host Endocytic Pathway. Traffic 2012, 13, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Thieleke-Matos, C.; Lopes da Silva, M.; Cabrita-Santos, L.; Portal, M.D.; Rodrigues, I.P.; Zuzarte-Luis, V.; Ramalho, J.S.; Futter, C.E.; Mota, M.M.; Barral, D.C.; et al. Host Cell Autophagy Contributes to Plasmodium Liver Development. Cell. Microbiol. 2016, 18, 437–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graewe, S.; Rankin, K.E.; Lehmann, C.; Deschermeier, C.; Hecht, L.; Froehlke, U.; Stanway, R.R.; Heussler, V. Hostile Takeover by Plasmodium: Reorganization of Parasite and Host Cell Membranes during Liver Stage Egress. PLoS Pathog. 2011, 7, e1002224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burda, P.-C.; Caldelari, R.; Heussler, V.T. Manipulation of the Host Cell Membrane during Plasmodium Liver Stage Egress. mBio 2017, 8, e00139-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prudêncio, M.; Rodriguez, A.; Mota, M.M. The Silent Path to Thousands of Merozoites: The Plasmodium Liver Stage. Nat. Rev. Microbiol. 2006, 4, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A. Manipulation of Host Hepatocytes by the Malaria Parasite for Delivery into Liver Sinusoids. Science 2006, 313, 1287–1290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cowman, A.F.; Crabb, B.S. Invasion of Red Blood Cells by Malaria Parasites. Cell 2006, 124, 755–766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cervantes, S.; Bunnik, E.M.; Saraf, A.; Conner, C.M.; Escalante, A.; Sardiu, M.E.; Ponts, N.; Prudhomme, J.; Florens, L.; Le Roch, K.G. The Multifunctional Autophagy Pathway in the Human Malaria Parasite, Plasmodium falciparum. Autophagy 2014, 10, 80–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomlins, A.M.; Ben-Rached, F.; Williams, R.A.; Proto, W.R.; Coppens, I.; Ruch, U.; Gilberger, T.W.; Coombs, G.H.; Mottram, J.C.; Müller, S.; et al. Plasmodium Falciparum ATG8 Implicated in Both Autophagy and Apicoplast Formation. Autophagy 2013, 9, 1540–1552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, P.G.T.; Griffin, J.T.; Cairns, M.; Rogerson, S.J.; van Eijk, A.M.; ter Kuile, F.; Ghani, A.C. A Model of Parity-Dependent Immunity to Placental Malaria. Nat. Commun. 2013, 4, 1609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Couper, K.N.; Barnes, T.; Hafalla, J.C.R.; Combes, V.; Ryffel, B.; Secher, T.; Grau, G.E.; Riley, E.M.; de Souza, J.B. Parasite-Derived Plasma Microparticles Contribute Significantly to Malaria Infection-Induced Inflammation through Potent Macrophage Stimulation. PLoS Pathog. 2010, 6, e1000744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toda, H.; Diaz-Varela, M.; Segui-Barber, J.; Roobsoong, W.; Baro, B.; Garcia-Silva, S.; Galiano, A.; Gualdrón-López, M.; Almeida, A.C.G.; Brito, M.A.M.; et al. Plasma-Derived Extracellular Vesicles from Plasmodium Vivax Patients Signal Spleen Fibroblasts via NF-KB Facilitating Parasite Cytoadherence. Nat. Commun. 2020, 11, 2761. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.; Chew, M.; Hou, J.; Lai, F.; Leopold, S.J.; Loo, H.L.; Ghose, A.; Dutta, A.K.; Chen, Q.; Ooi, E.E.; et al. Microvesicles from Malaria-Infected Red Blood Cells Activate Natural Killer Cells via MDA5 Pathway. PLoS Pathog. 2018, 14, e1007298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ketprasit, N.; Cheng, I.S.; Deutsch, F.; Tran, N.; Imwong, M.; Combes, V.; Palasuwan, D. The Characterization of Extracellular Vesicles-Derived MicroRNAs in Thai Malaria Patients. Malar. J. 2020, 19, 285. [Google Scholar] [CrossRef] [PubMed]
- Regev-Rudzki, N.; Wilson, D.W.; Carvalho, T.G.; Sisquella, X.; Coleman, B.M.; Rug, M.; Bursac, D.; Angrisano, F.; Gee, M.; Hill, A.F.; et al. Cell-Cell Communication between Malaria-Infected Red Blood Cells via Exosome-like Vesicles. Cell 2013, 153, 1120–1133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wheway, J.; Obeid, S.; Couraud, P.-O.; Combes, V.; Grau, G.E.R. The Brain Microvascular Endothelium Supports T Cell Proliferation and Has Potential for Alloantigen Presentation. PLoS ONE 2013, 8, e52586. [Google Scholar] [CrossRef]
- Wheway, J.; Latham, S.L.; Combes, V.; Grau, G.E.R. Endothelial Microparticles Interact with and Support the Proliferation of T Cells. J. Immunol. 2014, 193, 3378–3387. [Google Scholar] [CrossRef] [Green Version]
- Bagot, S.; Nogueira, F.; Collette, A.; do Rosario, V.; Lemonier, F.; Cazenave, P.-A.; Pied, S. Comparative Study of Brain CD8+ T Cells Induced by Sporozoites and Those Induced by Blood-Stage Plasmodium Berghei ANKA Involved in the Development of Cerebral Malaria. Infect. Immun. 2004, 72, 2817–2826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collette, A.; Bagot, S.; Ferrandiz, M.E.; Cazenave, P.-A.; Six, A.; Pied, S. A Profound Alteration of Blood TCRB Repertoire Allows Prediction of Cerebral Malaria. J. Immunol. 2004, 173, 4568–4575. [Google Scholar] [CrossRef] [PubMed]
- Baptista, F.G.; Pamplona, A.; Pena, A.C.; Mota, M.M.; Pied, S.; Vigário, A.M. Accumulation of Plasmodium berghei -Infected Red Blood Cells in the Brain Is Crucial for the Development of Cerebral Malaria in Mice. Infect. Immun. 2010, 78, 4033–4039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vigário, A.M.; Gorgette, O.; Dujardin, H.C.; Cruz, T.; Cazenave, P.-A.; Six, A.; Bandeira, A.; Pied, S. Regulatory CD4+CD25+ Foxp3+ T Cells Expand during Experimental Plasmodium Infection but Do Not Prevent Cerebral Malaria. Int. J. Parasitol. 2007, 37, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Shaw, T.N.; Stewart-Hutchinson, P.J.; Strangward, P.; Dandamudi, D.B.; Coles, J.A.; Villegas-Mendez, A.; Gallego-Delgado, J.; van Rooijen, N.; Zindy, E.; Rodriguez, A.; et al. Perivascular Arrest of CD8+ T Cells Is a Signature of Experimental Cerebral Malaria. PLoS Pathog. 2015, 11, e1005210. [Google Scholar] [CrossRef] [PubMed]
- Dalko, E.; Genete, D.; Auger, F.; Dovergne, C.; Lambert, C.; Herbert, F.; Cazenave, P.-A.; Roland, J.; Pied, S. Heme Dampens T-Cell Sequestration by Modulating Glial Cell Responses during Rodent Cerebral Malaria. Brain. Behav. Immun. 2016, 58, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.K.; Dalko, E.; Delcroix-Genete, D.; Herbert, F.; Cazenave, P.-A.; Pied, S. Uptake of Parasite-Derived Vesicles by Astrocytes and Microglial Phagocytosis of Infected Erythrocytes May Drive Neuroinflammation in Cerebral Malaria: Plasmodium Interaction with Glial Cells. Glia 2017, 65, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Strangward, P.; Haley, M.J.; Shaw, T.N.; Schwartz, J.-M.; Greig, R.; Mironov, A.; de Souza, J.B.; Cruickshank, S.M.; Craig, A.G.; Milner, D.A.; et al. A Quantitative Brain Map of Experimental Cerebral Malaria Pathology. PLoS Pathog. 2017, 13, e1006267. [Google Scholar] [CrossRef] [PubMed]
- Norden, D.M.; Trojanowski, P.J.; Villanueva, E.; Navarro, E.; Godbout, J.P. Sequential Activation of Microglia and Astrocyte Cytokine Expression Precedes Increased Iba-1 or GFAP Immunoreactivity Following Systemic Immune Challenge: Iba1 and GFAP Are Unreliable Activation Markers. Glia 2016, 64, 300–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liddelow, S.A.; Marsh, S.E.; Stevens, B. Microglia and Astrocytes in Disease: Dynamic Duo or Partners in Crime? Trends Immunol. 2020, 41, 820–835. [Google Scholar] [CrossRef] [PubMed]
- Belnoue, E.; Kayibanda, M.; Vigario, A.M.; Deschemin, J.-C.; van Rooijen, N.; Viguier, M.; Snounou, G.; Rénia, L. On the Pathogenic Role of Brain-Sequestered Aβ CD8 + T Cells in Experimental Cerebral Malaria. J. Immunol. 2002, 169, 6369–6375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schofield, L.; Grau, G.E. Immunological Processes in Malaria Pathogenesis. Nat. Rev. Immunol. 2005, 5, 722–735. [Google Scholar] [CrossRef] [PubMed]
- Farina, C.; Aloisi, F.; Meinl, E. Astrocytes Are Active Players in Cerebral Innate Immunity. Trends Immunol. 2007, 28, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Huda, S.; Sinha Babu, S.P. Toll-like Receptor Polymorphism in Host Immune Response to Infectious Diseases: A Review. Scand. J. Immunol. 2019, 90, e12771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Acioglu, C.; Heary, R.F.; Elkabes, S. Role of Astroglial Toll-like Receptors (TLRs) in Central Nervous System Infections, Injury and Neurodegenerative Diseases. Brain. Behav. Immun. 2021, 91, 740–755. [Google Scholar] [CrossRef] [PubMed]
- Keswani, T.; Delcroix-Genete, D.; Herbert, F.; Leleu, I.; Lambert, C.; Draheim, M.; Salome-Desnoulez, S.; Saliou, J.M.; Cazenave, P.-A.; Silvie, O.; et al. Plasmodium Yoelii Uses a TLR3-Dependent Pathway to Achieve Mammalian Host Parasitism. J. Immunol. 2020, 205, 3071–3082. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.M.; Collier, L.A.; Winford, E.D.; Leonardo, C.C.; Ajmo, C.T.; Foran, E.A.; Kopper, T.J.; Gensel, J.C.; Pennypacker, K.R. Leukemia Inhibitory Factor Modulates the Peripheral Immune Response in a Rat Model of Emergent Large Vessel Occlusion. J. Neuroinflamm. 2018, 15, 288. [Google Scholar] [CrossRef] [PubMed] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leleu, I.; Alloo, J.; Cazenave, P.-A.; Roland, J.; Pied, S. Autophagy Pathways in the Genesis of Plasmodium-Derived Microvesicles: A Double-Edged Sword? Life 2022, 12, 415. https://doi.org/10.3390/life12030415
Leleu I, Alloo J, Cazenave P-A, Roland J, Pied S. Autophagy Pathways in the Genesis of Plasmodium-Derived Microvesicles: A Double-Edged Sword? Life. 2022; 12(3):415. https://doi.org/10.3390/life12030415
Chicago/Turabian StyleLeleu, Inès, Jeremy Alloo, Pierre-André Cazenave, Jacques Roland, and Sylviane Pied. 2022. "Autophagy Pathways in the Genesis of Plasmodium-Derived Microvesicles: A Double-Edged Sword?" Life 12, no. 3: 415. https://doi.org/10.3390/life12030415
APA StyleLeleu, I., Alloo, J., Cazenave, P.-A., Roland, J., & Pied, S. (2022). Autophagy Pathways in the Genesis of Plasmodium-Derived Microvesicles: A Double-Edged Sword? Life, 12(3), 415. https://doi.org/10.3390/life12030415





