Serum OPG and RANKL Levels as Risk Factors for the Development of Cardiovascular Calcifications in End-Stage Renal Disease Patients in Hemodialysis
Abstract
1. Background
2. Methods
2.1. Patients and Study Design
2.2. Laboratory Measurements
2.3. Measurement of Osteoprotegerin and sRANKL
2.4. Clinical Variables
2.5. Vascular Calcifications
2.6. Statistical Analysis
3. Results
3.1. The Severity of Cardiovascular Calcifications and Correlations with Clinical and Laboratory Parameters
3.2. Multivariate Analysis
3.3. Correlations of Clinical Parameters and Initial Serum OPG Levels with the Progression of VCs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Foley, R.N.; Parfrey, P.S.; Sarnak, M.J. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am. J. Kidney Dis. 1998, 32, S112–S119. [Google Scholar] [CrossRef]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.-Y. Chronic Kidney Disease and the Risks of Death, Cardiovascular Events, and Hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Chen, N.X. Mechanisms of vascular calcification in chronic kidney disease. J. Am. Soc. Nephrol. 2008, 19, 213–216. [Google Scholar] [CrossRef]
- Paloian, N.J.; Giachelli, C.M. A current understanding of vascular calcification in CKD. Am. J. Physiol. Physiol. 2014, 307, F891–F900. [Google Scholar] [CrossRef]
- Schlieper, G.; Schurgers, L.; Brandenburg, V.; Reutelingsperger, C.; Floege, J. Vascular calcification in chronic kidney disease: An update. Nephrol. Dial. Transplant. 2015, 31, 31–39. [Google Scholar] [CrossRef]
- Mizobuchi, M.; Towler, D.; Slatopolsky, E. Vascular Calcification: The Killer of Patients with Chronic Kidney Disease. J. Am. Soc. Nephrol. 2009, 20, 1453–1464. [Google Scholar] [CrossRef]
- Hénaut, L.; Sanchez-Nino, M.D.; Castillo, G.A.-E.; Sanz, A.B.; Ortiz, A. Targeting local vascular and systemic consequences of inflammation on vascular and cardiac valve calcification. Expert Opin. Ther. Targets 2015, 20, 89–105. [Google Scholar] [CrossRef]
- Harper, E.; Forde, H.; Davenport, C.; Rochfort, K.D.; Smith, D.; Cummins, P.M. Vascular calcification in type-2 diabetes and cardiovascular disease: Intergrative roles for OPG, RANKL and TRAIL. Vascul. Pharmacol. 2016, 82, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Schoppet, M.; Shroff, R.C.; Hofbauer, L.C.; Shanahan, C.M. Exploring the biology of vascular calcification in chronic kidney disease: What’s circulating? Kidney Int. 2008, 73, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, Y.; Yu, C.-M.; Ji, Q.-W.; Cai, M.; Zhao, Y.-X.; Zhou, Y.-J. Current understanding of coronary artery calcification. J. Geriatr. Cardiol. 2015, 12, 668–675. [Google Scholar] [CrossRef]
- Evrard, S.; Delanaye, P.; Kamel, S.; Cristol, J.-P.; Cavalier, E.; Arnaud, J.; Zaoui, P.; Carlier, M.; Laville, M.; Fouque, D.; et al. Vascular calcification: From pathophysiology to biomarkers. Clin. Chim. Acta 2015, 438, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Znorko, B.; Oksztulska-Kolanek, E.; Michałowska, M.; Kamiński, T.; Pawlak, K. Does the OPG/RANKL system contribute to the bone-vascular axis in chronic kidney disease? A systematic review. Adv. Med. Sci. 2017, 62, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.C.; Choi, Y. Biology of the RANKL—RANK—OPG system in immunity, bone and beyond. Front. Immunol. 2014, 5, 511. [Google Scholar] [CrossRef]
- Collin-Osdoby, P. Regulation of Vascular Calcification by Osteoclast Regulatory Factors RANKL and Osteoprotegerin. Circ. Res. 2004, 95, 1046–1057. [Google Scholar] [CrossRef] [PubMed]
- Panizo, S.; Cardus, A.; Encinas, M.; Parisi, E.; Valcheva, P.; López-Ongil, S.; Coll, B.; Fernandez, E.; Valdivielso, J.M. RANKL Increases Vascular Smooth Muscle Cell Calcification Through a RANK-BMP4–Dependent Pathway. Circ. Res. 2009, 104, 1041–1048. [Google Scholar] [CrossRef]
- Baud’Huin, M.; Lamoureux, F.; Duplomb, L.; Rédini, F.; Heymann, D. RANKL, RANK, osteoprotegerin: Key partners of osteoimmunology and vascular diseases. Cell Mol. Life Sci. 2007, 64, 2334–2350. [Google Scholar] [CrossRef]
- Higgins, C.L.; Isbilir, S.; Basto, P.; Chen, I.Y.; Vaduganathan, M.; Vaduganathan, P.; Reardon, M.J.; Lawrie, G.; Peterson, L.; Morrisett, J.D. Distribution of Alkaline Phosphatase, Osteopontin, RANK Ligand and Osteoprotegerin in Calcified Human Carotid Atheroma. Protein J. 2015, 34, 315–328. [Google Scholar] [CrossRef]
- Forde, H.; Harper, E.; Davenport, C.; Rochfort, K.D.; Wallace, R.; Murphy, R.P.; Smith, D.; Cummins, P.M. The beneficial pleiotropic effects of tumour necrosis related factor apoptosis ligand (TRAIL) within the vasculature: A review of the literature. Atherosclerosis 2016, 247, 87–96. [Google Scholar] [CrossRef]
- Bucay, N.; Sarosi, I.; Dunstan, C.; Morony, S.; Tarpley, J.; Capparelli, C.; Scully, S.; Tan, H.L.; Xu, W.; Lacey, D.L.; et al. osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev. 1998, 12, 1260–1268. [Google Scholar] [CrossRef]
- Bennet, B.J.; Scatena, M.; Kirk, E.A.; Ratazzi, M.; Varon, R.M.; Averill, M.; Schwartz, S.M.; Giachelli, C.M.; Rosenfeld, M.E. Osteoprotegerin inactivation accelerates advanced atherosclerotic lesion progression and calcification in older ApoE -/- mice. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2117–2124. [Google Scholar] [CrossRef]
- Callegari, A.; Coons, M.L.; Ricks, J.L.; Rosenfeld, M.E.; Scatena, M. Increased calcification in osteoprotegerin-deficient smooth muscle cells: Dependence on receptor activator of NF-κB ligand and interleukin 6. J. Vasc. Res. 2014, 51, 118–131. [Google Scholar] [CrossRef]
- Bjerre, M. Osteoprotegerin (OPG) as a biomarker for diabetic cardiovascular complications. SpringerPLus 2013, 2, 658. [Google Scholar] [CrossRef]
- Vik, A.; Mathiesen, E.B.; Brox, J.; Wilsgaard, T.; Njølstad, I.; Jørgensen, L.; Hansen, J. Relation between serum osteoprotegerin and carotid intima media thickness in a general population—The Tromsø Study. J. Thromb. Haemost. 2010, 8, 2133–2139. [Google Scholar] [CrossRef] [PubMed]
- Kiechl, S.; Schett, G.; Wenning, G.; Redlich, K.; Oberhollenzer, M.; Mayr, A.; Santer, P.; Smolen, J.; Poewe, W.; Willeit, J. Osteoprotegerin Is a Risk Factor for Progressive Atherosclerosis and Cardiovascular Disease. Circulation 2004, 109, 2175–2180. [Google Scholar] [CrossRef] [PubMed]
- Collado, S.; Coll, E.; Nicolau, C.; Azqueta, M.; Pons, M.; Cruzado, J.M.; De La Torre, B.; Deulofeu, R.; Mojal, S.; Pascual, J.; et al. Serum osteoprotegerin in prevalent hemodialysis patients: Associations with mortality, atherosclerosis and cardiac function. BMC Nephrol. 2017, 18, 290. [Google Scholar] [CrossRef]
- Nascimento, M.; Hayashi, S.; Riella, M.; Lindholm, B. Elevated levels of plasma osteoprotegerin are associated with all-cause mortality risk and atherosclerosis in patients with stages 3 to 5 chronic kidney disease. Braz. J. Med. Biol. Res. 2014, 47, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, M.I.; Siriopol, D.; Saglam, M.; Ünal, H.U.; Karaman, M.; Gezer, M.; Kilinc, A.; Eyileten, T.; Guler, A.K.; Aydin, I.; et al. Osteoprotegerin in Chronic Kidney Disease: Associations with Vascular Damage and Cardiovascular Events. Calcif. Tissue Int. 2016, 99, 121–130. [Google Scholar] [CrossRef]
- Forde, H.; Davenport, C.; Harper, E.; Cummins, P.; Smith, D. The role of OPG/RANKL in the pathogenesis of diabetic cardiovascular disease. Cardiovasc. Endocrinol. Metab. 2018, 7, 28–33. [Google Scholar] [CrossRef]
- Adragao, T.; Pires, A.; Lucas, C.; Birne, R.; Magalhaes, L.; Gonçalves, M.; Negrao, A.P. A simple vascular calcification score predicts cardiovascular risk in haemodialysis patients. Nephrol. Dial. Transplant. 2004, 19, 1480–1488. [Google Scholar] [CrossRef]
- Wilson, P.W.F.; Kauppila, L.I.; O’Donnell, C.J.; Kiel, D.; Hannan, M.; Polak, J.M.; Cupples, L.A. Abdominal Aortic Calcific Deposits Are an Important Predictor of Vascular Morbidity and Mortality. Circulation 2001, 103, 1529–1534. [Google Scholar] [CrossRef]
- Klejna, K.; Naumnik, B.; Gasowska, K.; Myśliwiec, M. OPG/RANK/RANKL signaling system and its significance in nephrology. Folia Histochem. Cytobiol. 2009, 47, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Frazão, J.M. The bone-vessel axis in chronic kidney disease: An update on biochemical players and its future role in laboratory medicine. Clin. Chim. Acta 2020, 508, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Lysitska, A.; Galanis, N.; Skandalos, I.; Nikolaidou, C.; Briza, S.; Fylaktou, A.; Lioulios, G.; Mitsoglou, Z.; Papadopoulou, D.; Antoniadis, N.; et al. Histology and Immunohistochemistry of Radial Arteries Are Suggestive of an Interaction between Calcification and Early Atherosclerotic Lesions in Chronic Kidney Disease. Medicina 2021, 57, 1156. [Google Scholar] [CrossRef] [PubMed]
- Aaltonen, L.; Koivuviita, N.; Seppänen, M.; Kröger, H.; Tong, X.; Löyttyniemi, E.; Metsärinne, K. Association between bone mineral metabolism and vascular calcification in end-stage renal disease. BMC Nephrol. 2022, 23, 12. [Google Scholar] [CrossRef]
- Barreto, D.V.; Barreto Fde, C.; Carvalho, A.B.; Cuppari, L.; Draibe, S.A.; Dalboni, M.A.; Moyses, R.M.A.; Neves, K.R.; Jorgetti, V.; Miname, M.; et al. Association of changes in bone remodeling and coronary calcification in hemodialysis patients: A prospective study. Am. J. Kidney Dis. 2008, 52, 1139–1150. [Google Scholar] [CrossRef]
- Barreto, D.V.; Barreto, F.C.; Carvalho, A.B.; Cuppari, L.; Cendoroglo, M.; Draibe, S.A.; Moyses, R.M.A.; Neves, K.R.; Jorgetti, V.; Blair, A.; et al. Coronary calcification in hemodialysis patients: The contribution of traditional and uremia-related risk factors. Kidney Int. 2005, 67, 1576–1582. [Google Scholar] [CrossRef]
- Chen, H.-C.; Wang, W.-T.; Hsi, C.-N.; Chou, C.-Y.; Lin, H.-J.; Huang, C.-C.; Chang, C.-T. Abdominal aortic calcification score can predict future coronary artery disease in hemodialysis patients: A 5-year prospective cohort study. BMC Nephrol. 2018, 19, 313. [Google Scholar] [CrossRef]
- Morena, M.; Jaussent, I.; Dupuy, A.-M.; Bargnoux, A.-S.; Kuster, N.; Chenine, L.; Leray-Moragues, H.; Klouche, K.; Vernhet, H.; Canaud, B.; et al. Osteoprotegerin and sclerostin in chronic kidney disease prior to dialysis: Potential partners in vascular calcifications. Nephrol. Dial. Transplant. 2015, 30, 1345–1356. [Google Scholar] [CrossRef]
- Krzanowski, M.; Krzanowska, K.; Dumnicka, P.; Gajda, M.; Woziwodzka, K.; Fedak, D.; Grodzicki, T.; Litwin, J.A.; Sułowicz, W. Elevated Circulating Osteoprotegerin Levels in the Plasma of Hemodialyzed Patients with Severe Artery Calcification. Ther. Apher. Dial. 2018, 22, 519–529. [Google Scholar] [CrossRef]
- Morena, M.; Dupuy, A.-M.; Jaussent, I.; Vernhet, H.; Gahide, G.; Klouche, K.; Bargnoux, A.-S.; Delcourt, C.; Canaud, B.; Cristol, J.-P. A cut-off value of plasma osteoprotegerin level may predict the presence of coronary artery calcifications in chronic kidney disease patients. Nephrol. Dial. Transplant. 2009, 24, 3389–3397. [Google Scholar] [CrossRef]
- Avila, M.; Mora, C.; Zavala, M.; Prado, M.D.C.; Paniagua, R. Osteoprotegerin Is the Strongest Predictor for Progression of Arterial Calcification in Peritoneal Dialysis Patients. Am. J. Nephrol. 2017, 46, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and Cardiovascular Disease A Scientific Statement From the American Heart Association. Circulation 2021, 143, e984–e1010. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.C.; Dardari, Z.A.; Blaha, M.J.; White, S.; Shaw, L.J.; Rumberger, J.; Rozanski, A.; Berman, D.S.; Budoff, M.J.; Nasir, K.; et al. Association of Body Mass Index with Coronary Artery Calcium and Subsequent Cardiovascular Mortality. Circ. Cardiovasc. Imaging 2020, 13, e009495. [Google Scholar] [CrossRef]
- Lai, Y.; Lin, Y.; Wang, C.; Kuo, C.; Hsu, B. Positive Association of Serum Adipocyte Fatty Acid Binding Protein Level with Peripheral Artery Disease in Hemodialysis Patients. Ther. Apher. Dial. 2019, 24, 300–306. [Google Scholar] [CrossRef]
- Kurnatowska, I.; Grzelak, P.; Kaczmarska, M.; Stefańczyk, L.; Nowicki, M. Serum Osteoprotegerin is a Predictor of Progression of Atherosclerosis and Coronary Calcification in Hemodialysis Patients. Nephron Clin. Pract. 2010, 117, 297–304. [Google Scholar] [CrossRef]
- Ozkok, A.; Caliskan, Y.; Sakaci, T.; Erten, G.; Karahan, G.; Ozel, A.; Unsal, A.; Yildiz, A. Osteoprotegerin/RANKL Axis and Progression of Coronary Artery Calcification in Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2012, 7, 965–973. [Google Scholar] [CrossRef]
- Morony, S.; Tintut, Y.; Zhang, Z.; Cattley, R.; Van, G.; Dwyer, D.; Stolina, M.; Kostenuik, P.; Dimer, L. Osteoprotegerin inhibits vascular calcification without affecting atherosclerosis in ldlr (¯/¯) mice. Circulation 2008, 117, 411–420. [Google Scholar] [CrossRef]
- Mesquita, M.; Demulder, A.; Wolff, F.; Melot, C.; Damry, N.; Dratwa, M.; Bergmann, P. Osteoprotegerin and progression of coronary and aortic calcification in chroniv kindey disease. Transpl. Proc. 2010, 42, 3444–3449. [Google Scholar] [CrossRef]
- Ono, T.; Hayashi, M.; Sasaki, F.; Nakashima, T. RANKL biology: Bone metabolism, the immune system, and beyond. Inflamm. Regen. 2020, 40, 2. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Wang, M.; Gan, L.-Y.; Li, X. Relationship of sRANKL Level and Vascular Calcification Score to Cardiovascular Events in Maintenance Hemodialysis Patients. Blood Purif. 2009, 28, 342–345. [Google Scholar] [CrossRef]
- Lieb, W.; Gona, P.; Larson, M.G.; Massaro, J.M.; Lipinska, I.; Keaney, J.F., Jr.; Rong, J.; Corey, D.; Hoffmann, U.; Fox, C.S.; et al. Biomarkers of the osteoprotegerin pathway: Clinical correlates, subclinical disease, incident cardiovascular disease, and mortality. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1849–1854. [Google Scholar] [CrossRef] [PubMed]
- Shiota, J.; Izumi, N.; Kasahara, H.; Tagawa, H.; Chiba, T.; Nihei, T. Relationship between coronary and abdominal calcification score, serum osteoprotegerin (OPG), and serum tartrate—Resistant acid phosphatase (TRACP)—5b in pre-dialysis CKD patients. Nihon Jinzo Gakkai Shi. 2010, 52, 1022–1028. [Google Scholar] [PubMed]

| Age (yrs) (M ± SD) * | 57.2 ± 15.4 |
| Gender (N, %) * | |
| Male | 42 (52.5%) |
| Female | 38 (47.5%) |
| BMI (Kg/m2) (M ± SD) | 26.1 ± 4.9 |
| Dialysis parameters (M ± SD) | |
| Dialysis session (min) | 245 ± 13.5 |
| Time on dialysis (months) | 64.3 ± 58 |
| Primary cause of ESRD (N, %) | |
| Primary Glomerulonephritis | 23 (28.7%) |
| Polycystic Kidney disease | 11 (13.7%) |
| Diabetic nephropathy | 7 (8.8%) |
| Chronic Interstitial nephritis | 6 (7.5%) |
| Obstructive uropathy | 4 (5%) |
| Alport’s syndrome | 4 (5%) |
| Other reasons | 25 (31.3%) |
| Comorbid conditions (N, %) | |
| Smoking | 21 (26.3%) |
| Hypertension | 53 (66.3%) |
| Diabetes Mellitus (DM) | 9 (11.3%) |
| Coronary artery disease (CAD) | 11 (14.3%) |
| Atrial fibrillation (AF) | 16 (20.8%) |
| Characteristics, M± SD | |
|---|---|
| Serum urea (mg/dL) | 136 ± 22.5 |
| Serum creatinine (mg/dL) | 9 ± 2.6 |
| Hgb (g/dL) | 11.5 ± 1.15 |
| Serum Calcium (mg/dL) | 9.1 ± 0.54 |
| Serum Phosphorus (mg/dL) | 4.97 ± 0.98 |
| Serum Glucose (mg/dL) | 94 ± 19.4 |
| Serum Albumin (g/dL) | 4.6 ± 4.9 |
| Serum ALP (U/L) | 105 ± 77.5 |
| Serum AST (IU/L) | 14.3 ± 8.3 |
| Serum ALT (IU/L) | 13.8 ± 11.3 |
| Serum Triglyceride (mg/dL) | 152 ± 78.6 |
| Serum Cholesterol (mg/dL) | 157 ± 33 |
| Serum HDL (mg/dL) | 44.7 ± 15.1 |
| Serum LDL (mg/dL) | 84.8 ± 29 |
| Serum CRP (mg/L) | 6.7 ± 8.5 |
| Serum iPTH (pg/mL) | 396 ± 296 |
| Kt/V | 1.47 ± 0.27 |
| RAAS inhibitors | 14 (17.5%) * |
| b-blockers | 37 (48.7%) |
| CCBs | 24 (31.6%) |
| Statins | 29 (36.3%) |
| Anti-PLT agents | 42 (52.5%) |
| Sevelamer | 62 (77.5%) |
| Lanthanum Carbonate | 27 (33.8%) |
| Calcium Carbonate | 10 (12.5%) |
| VDR agonists | 47 (58.8%) |
| Cinacalcet | 27 (33.8%) |
| CCPBs | 17 (21.3%) |
| NCCPBs | 71 (88.8%) |
| Kauppila Group | Adragao Group | Calcscore | |
|---|---|---|---|
| Age | p < 0.001 | p < 0.001 | p < 0.001 |
| Hypertension | p = 0.006 | NS | p = 0.013 |
| DM | p =0.016 | p < 0.001 | p = 0.005 |
| CAD | p = 0.004 | p = 0.001 | p = 0.001 |
| CVD | p = 0.001 | p = 0.001 | p = 0.001 |
| Time on dialysis | NS | p = 0.036 | NS |
| Dialyzer | NS | NS | NS |
| Use of statins | p = 0.004 | p = 0.030 | p = 0.002 |
| Use of anti-PLTs | p = 0.007 | p = 0.040 | p = 0.002 |
| Use of ARBs | NS | NS | NS |
| Use of b-blockers | p = 0.039 | p = 0.038 | NS |
| Use of NCCPBs | NS | NS | NS |
| Serum Calcium (mg/dL) | NS | NS | NS |
| Serum Phosphorus (mg/dL) | NS | p = 0.028 | p = 0.045 |
| Serum iPTH (pg/mL) | NS | NS | NS |
| Serum urea (mg/dL) | NS | NS | NS |
| Serum creatinine (mg/dL) | NS | NS | NS |
| Serum glucose (mg/dL) | NS | NS | NS |
| CRP (mg/L) | NS | NS | NS |
| BMI (kg/m²) | NS | p = 0.001 | p = 0.036 |
| Univariate Analysis | |||
|---|---|---|---|
| OPG | RANKL | OPG/RANKL | |
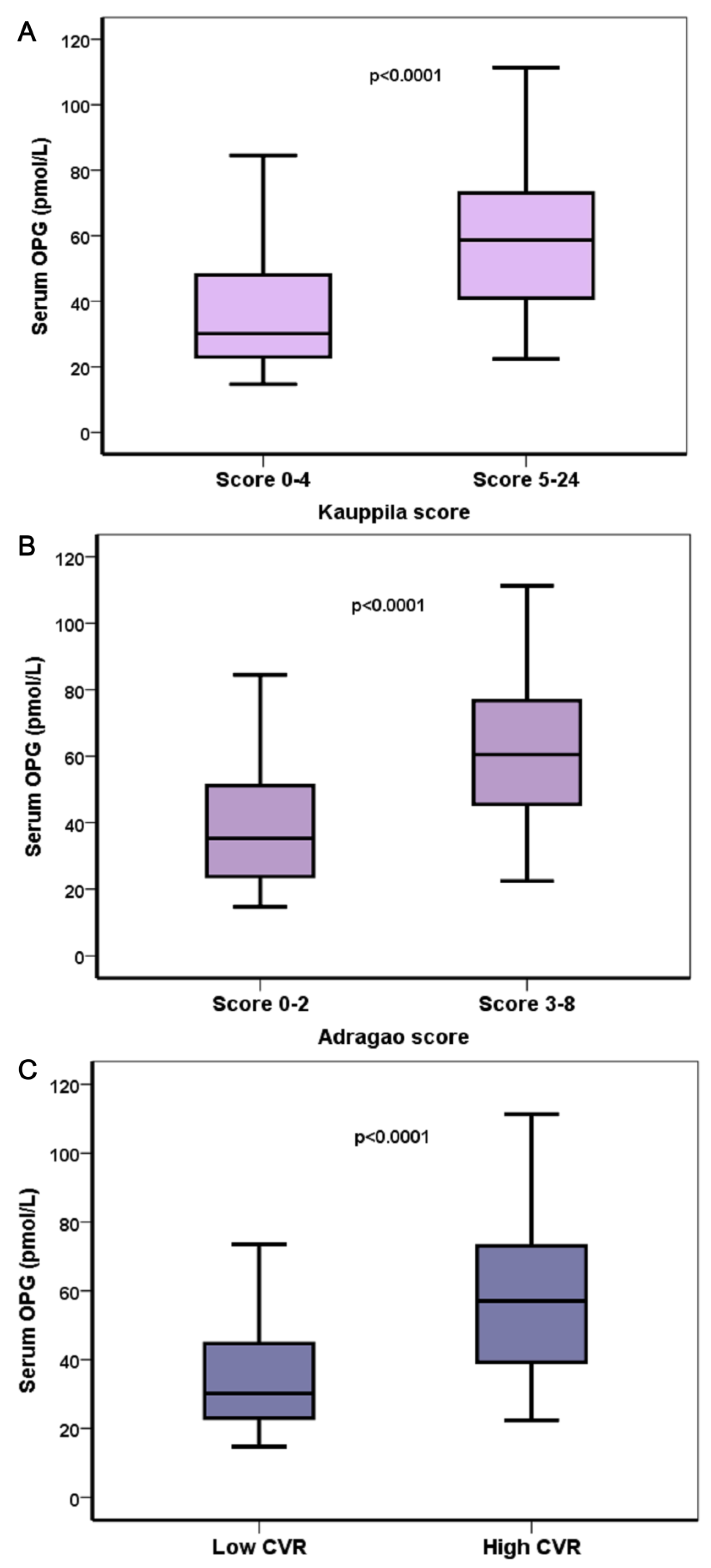
| Kauppila gr | p < 0.001 | NS | p = 0.022 |
| Adragao gr | p < 0.001 | NS | NS |
| CalcScore | p < 0.001 | NS | p = 0.014 |
| Kauppila gr | Adragao gr | |
|---|---|---|
| OPG levels | p = 0.020 | NS |
| Age | NS | NS |
| CVD | NS | NS |
| Diabetes mellitus | NS | NS |
| Hypertension | NS | Not included |
| BMI | Not included | p = 0.026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spartalis, M.; Kasimatis, E.; Liakou, E.; Sampani, E.; Lioulios, G.; Christodoulou, M.; Stai, S.; Moysidou, E.; Efstratiadis, G.; Papagianni, A. Serum OPG and RANKL Levels as Risk Factors for the Development of Cardiovascular Calcifications in End-Stage Renal Disease Patients in Hemodialysis. Life 2023, 13, 454. https://doi.org/10.3390/life13020454
Spartalis M, Kasimatis E, Liakou E, Sampani E, Lioulios G, Christodoulou M, Stai S, Moysidou E, Efstratiadis G, Papagianni A. Serum OPG and RANKL Levels as Risk Factors for the Development of Cardiovascular Calcifications in End-Stage Renal Disease Patients in Hemodialysis. Life. 2023; 13(2):454. https://doi.org/10.3390/life13020454
Chicago/Turabian StyleSpartalis, Michalis, Efstratios Kasimatis, Eleni Liakou, Erasmia Sampani, Georgios Lioulios, Michalis Christodoulou, Stamatia Stai, Eleni Moysidou, George Efstratiadis, and Aikaterini Papagianni. 2023. "Serum OPG and RANKL Levels as Risk Factors for the Development of Cardiovascular Calcifications in End-Stage Renal Disease Patients in Hemodialysis" Life 13, no. 2: 454. https://doi.org/10.3390/life13020454
APA StyleSpartalis, M., Kasimatis, E., Liakou, E., Sampani, E., Lioulios, G., Christodoulou, M., Stai, S., Moysidou, E., Efstratiadis, G., & Papagianni, A. (2023). Serum OPG and RANKL Levels as Risk Factors for the Development of Cardiovascular Calcifications in End-Stage Renal Disease Patients in Hemodialysis. Life, 13(2), 454. https://doi.org/10.3390/life13020454







