Ocimum sanctum Alters the Lipid Landscape of the Brain Cortex and Plasma to Ameliorate the Effect of Photothrombotic Stroke in a Mouse Model
Abstract
:1. Introduction
2. Material and Methods
2.1. Animals
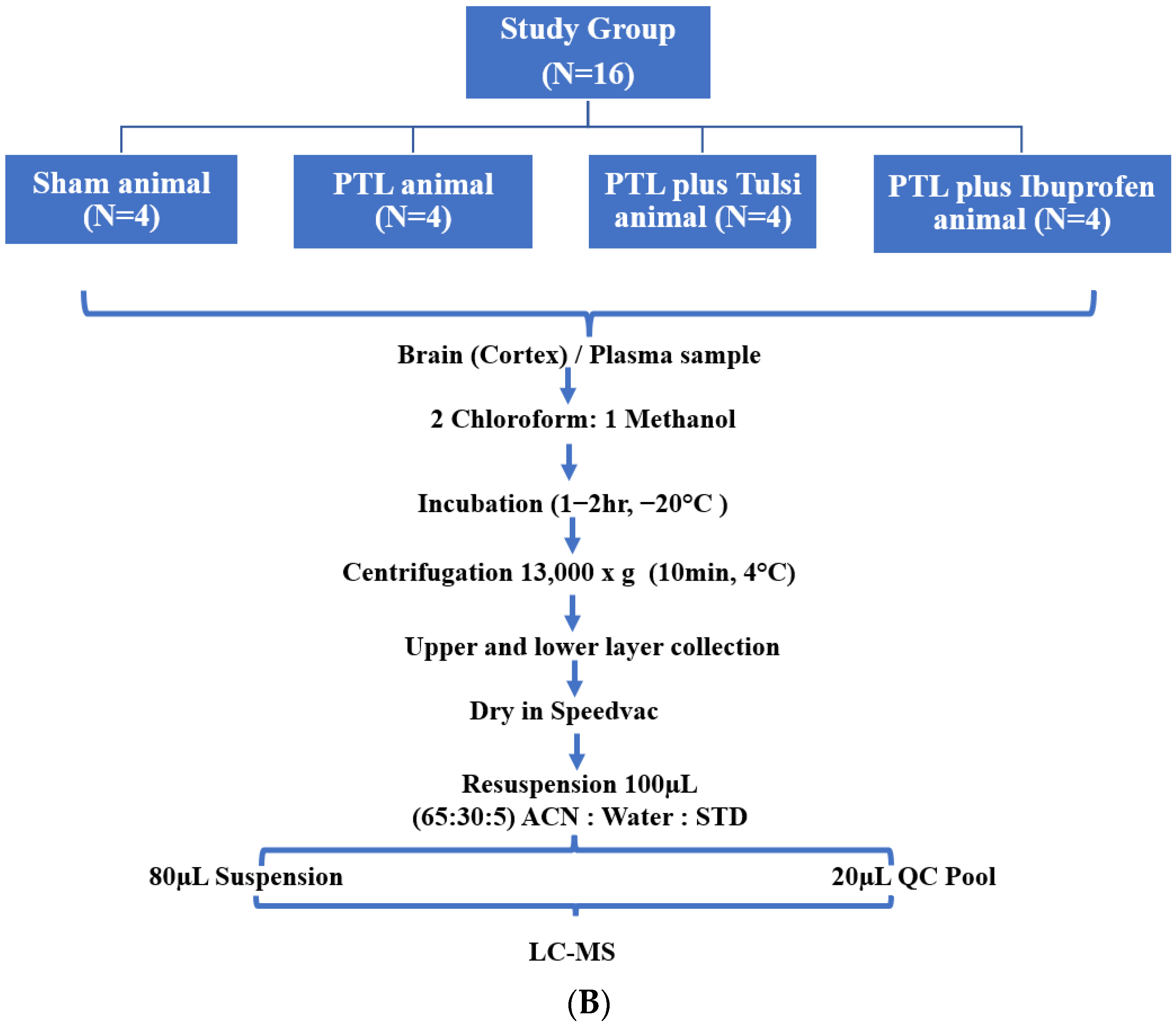
2.2. Experimental Groups
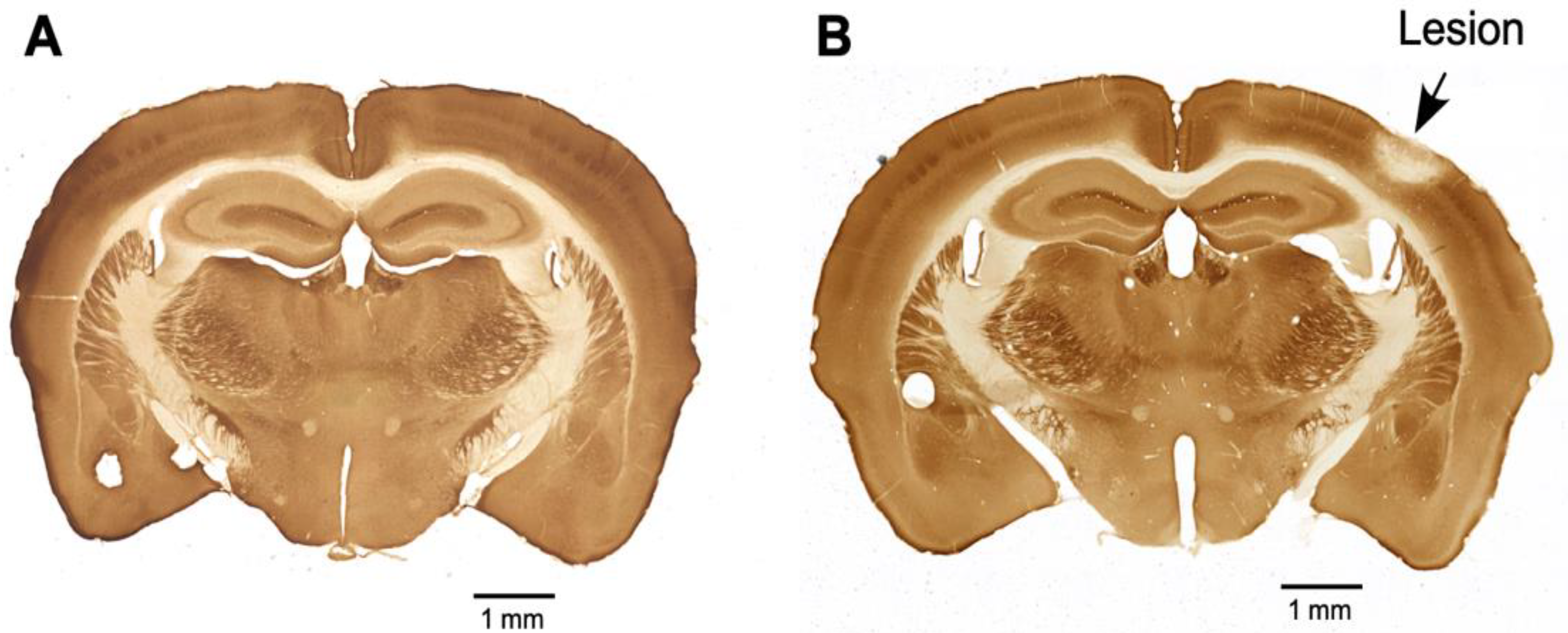
2.3. Photothrombotic Lesion (PTL)
2.4. Cytochrome Oxidase Reaction
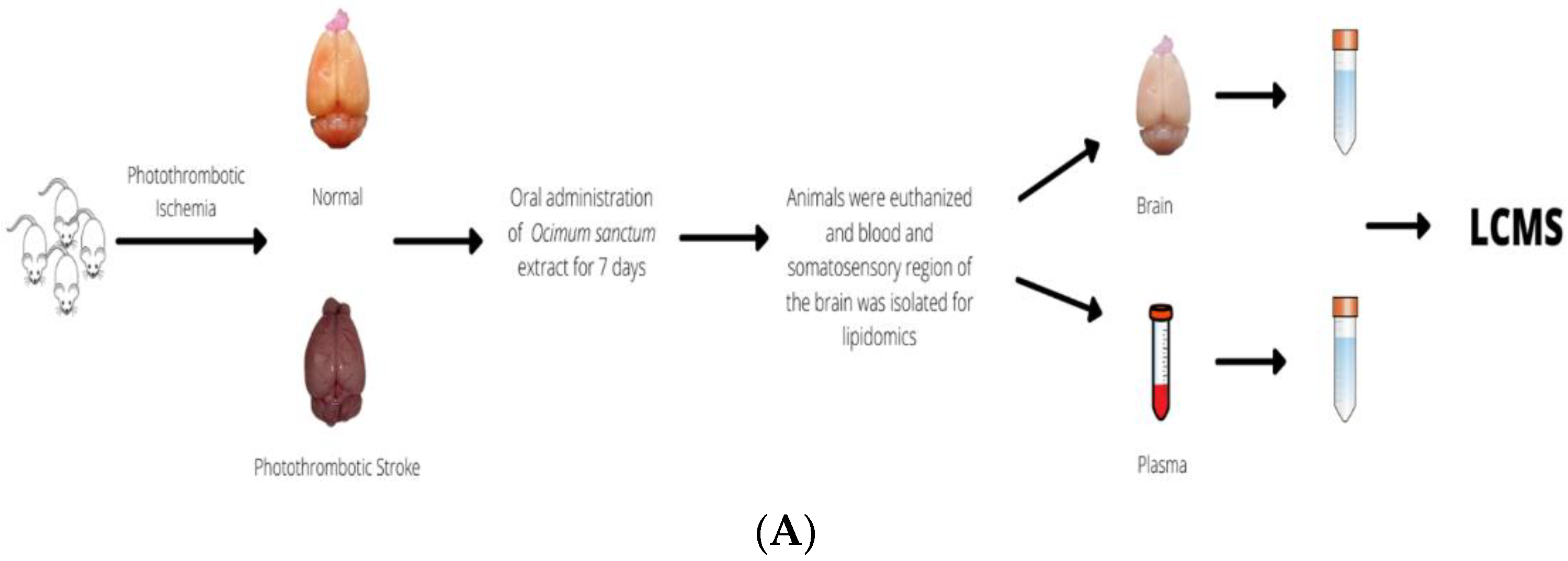
2.5. Sample Collection and Preparation
2.6. Mass Spectrometry
Mass Spectrometry for Metabolomics of Tulsi Extract
2.7. Software Analysis
2.8. Statistical Analysis
3. Results
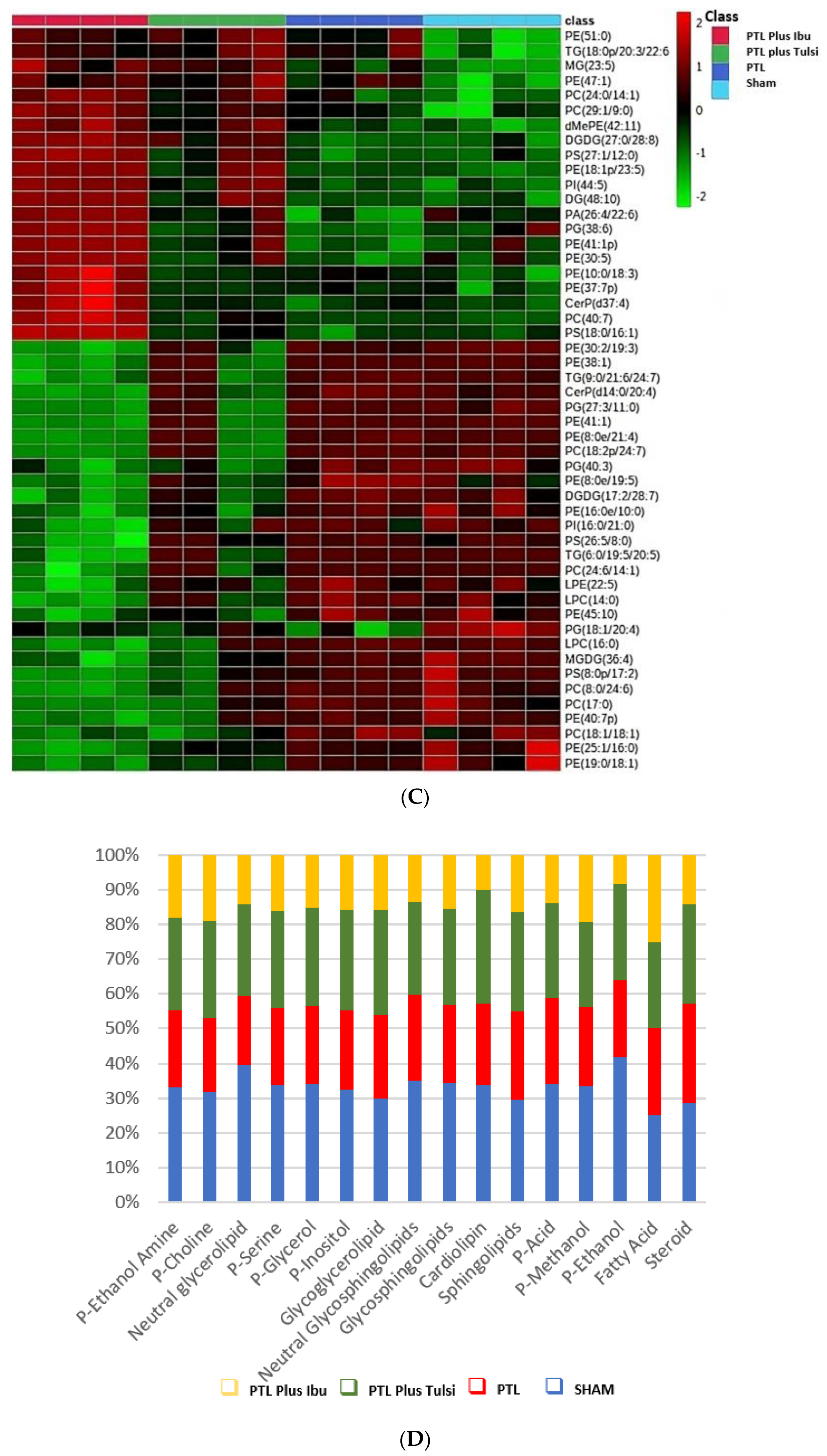
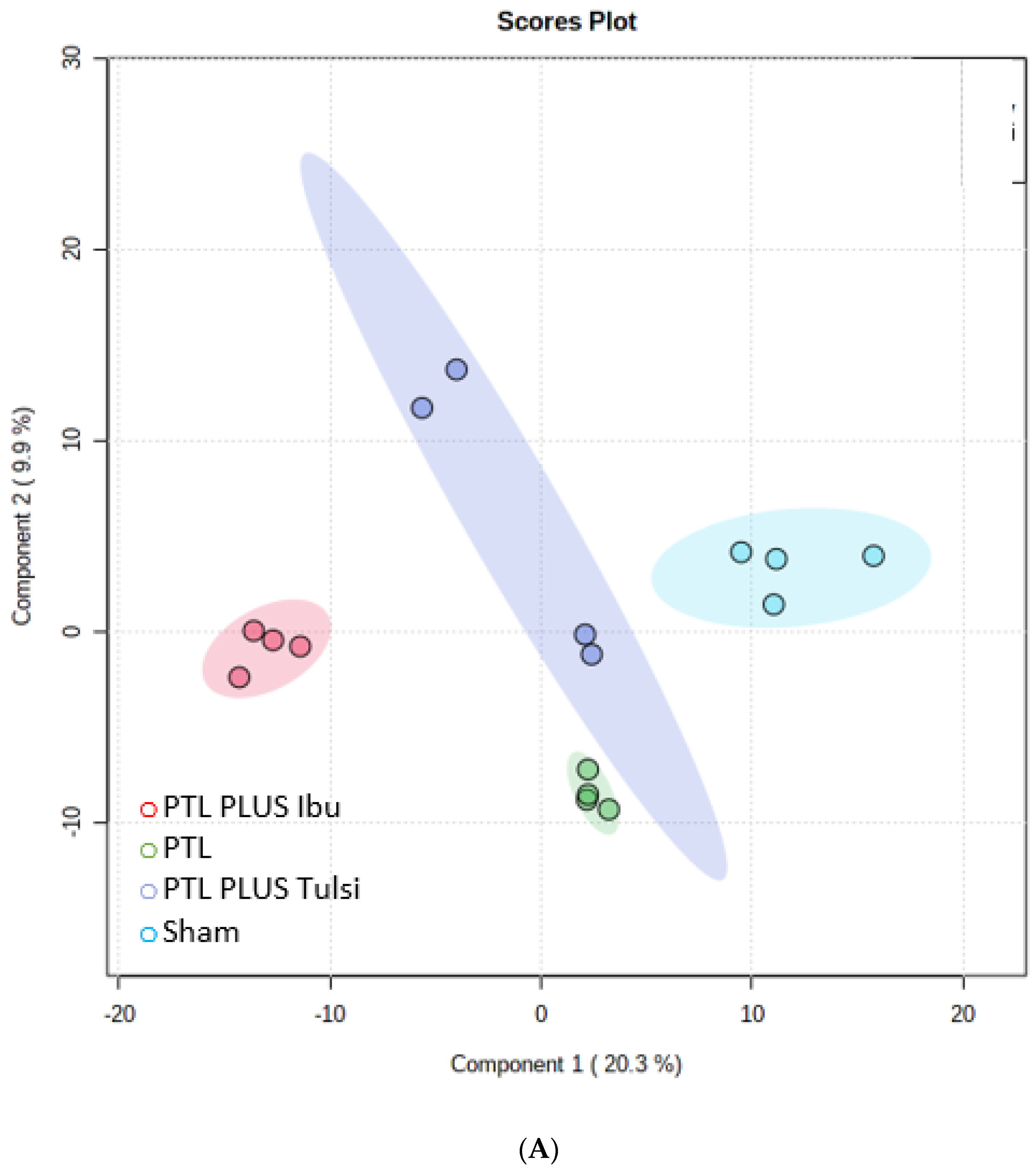
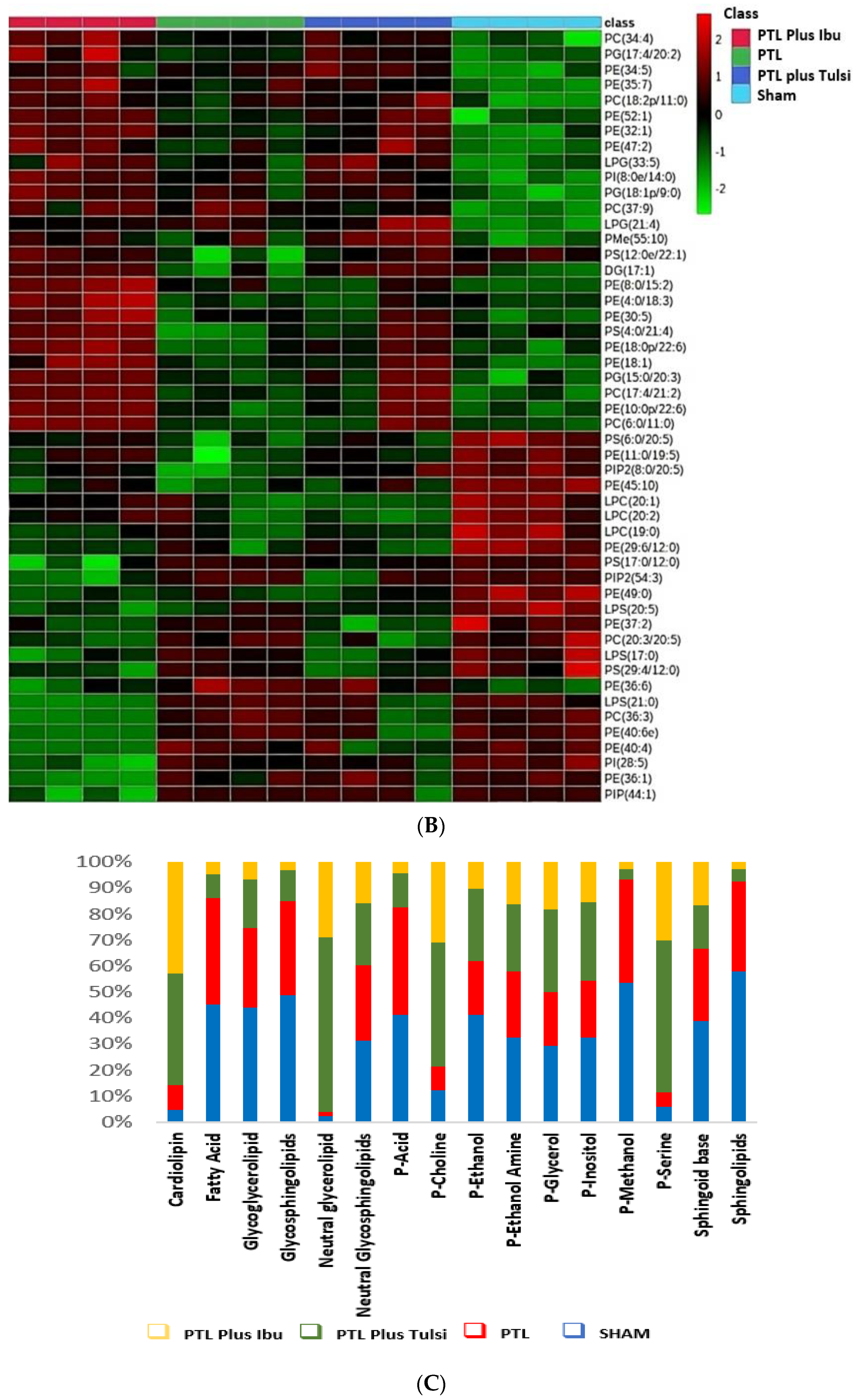
3.1. Tulsi Modulates Lipidomic Signature in the Lesioned Cortical Hemisphere of Mice with Photothrombotic Ischemic Stroke-like Lesion
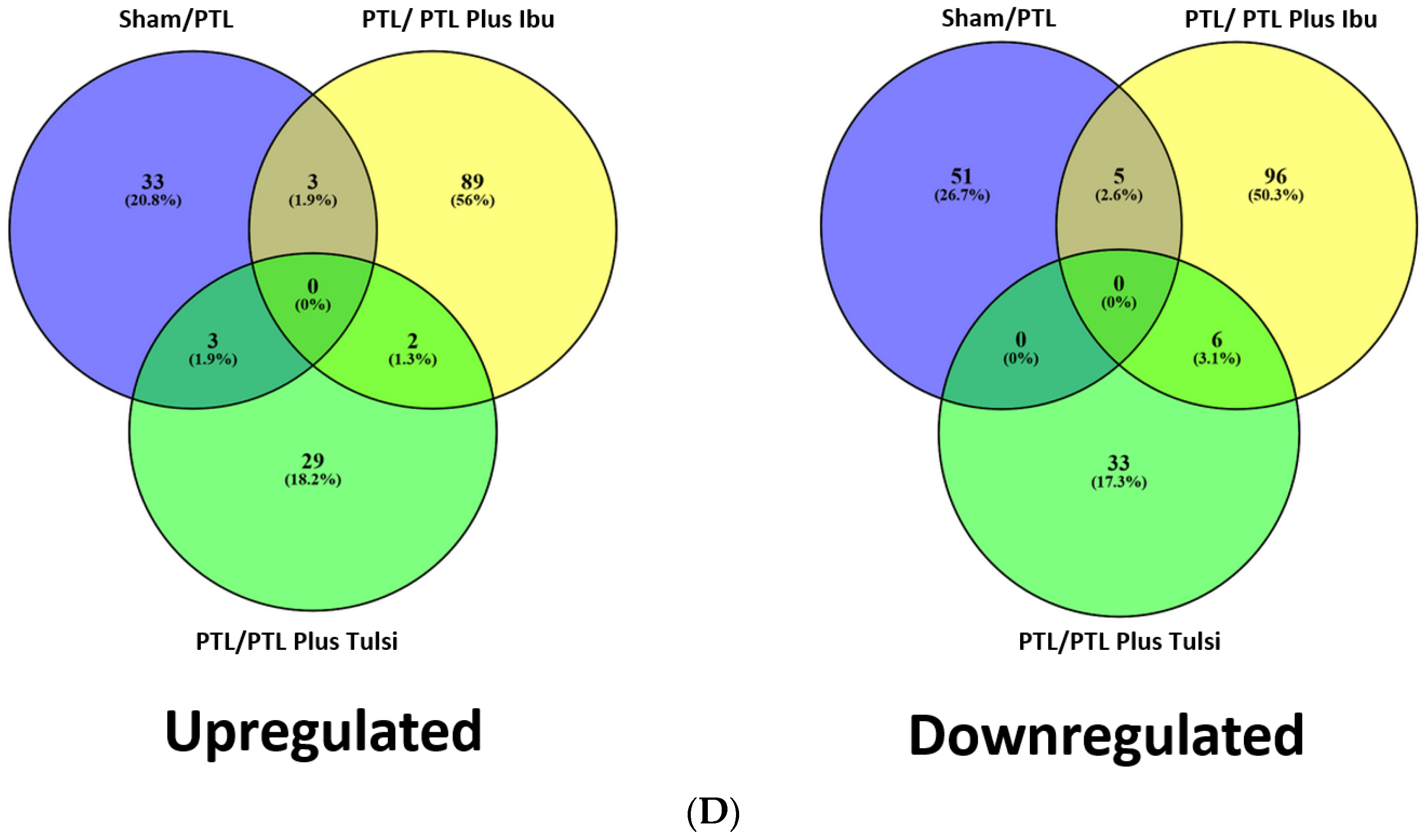
3.2. Comparison of Lipidome in Photothrombotic Ischemia-Induced Brain of Mice Treated with Tulsi vs. Ibuprofen
3.3. Effect of Tulsi in Modulating Lipidome Signature of Plasma in Mice with Photothrombotic Ischemic Lesion of the Cerebral Cortex
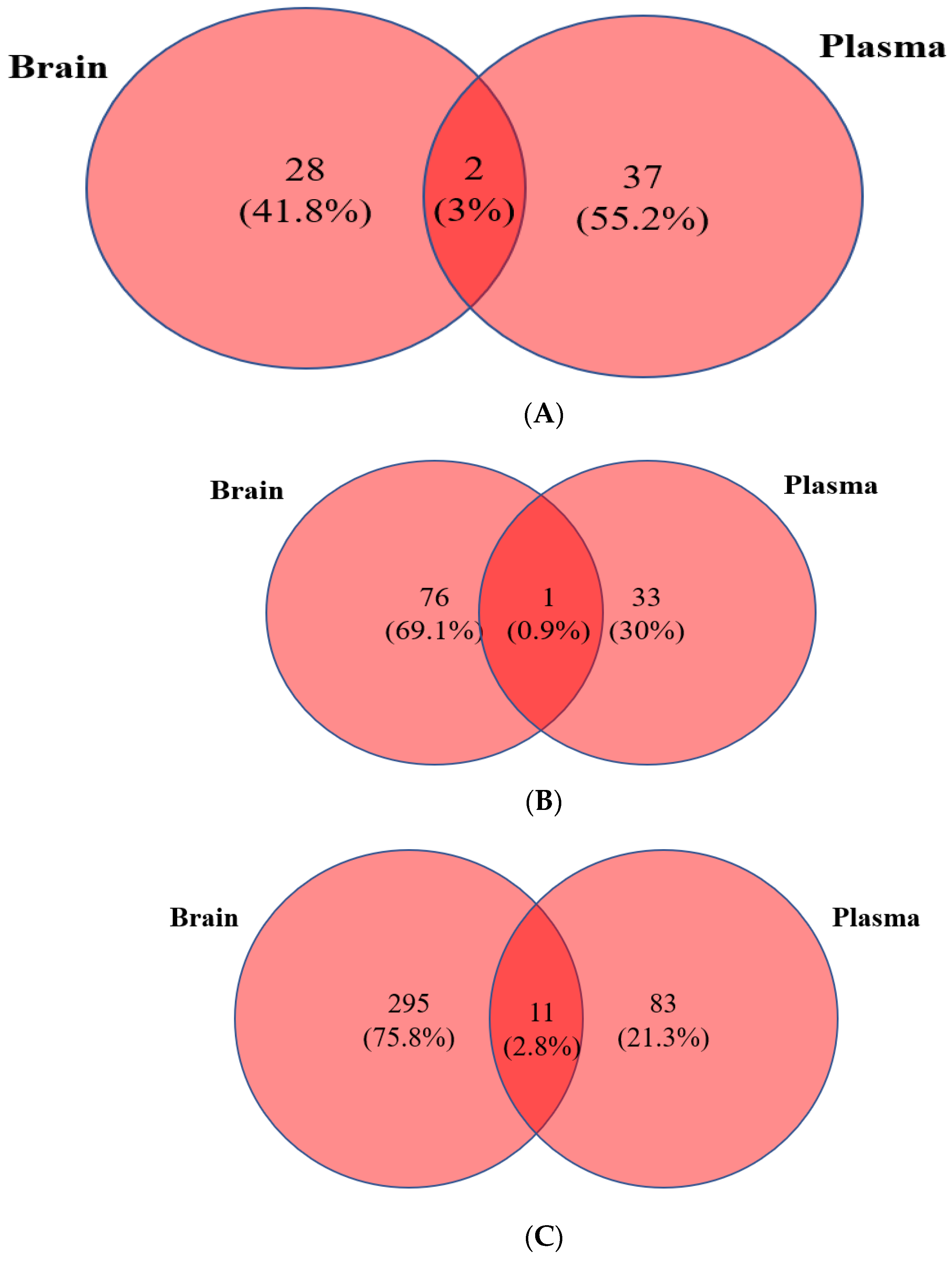
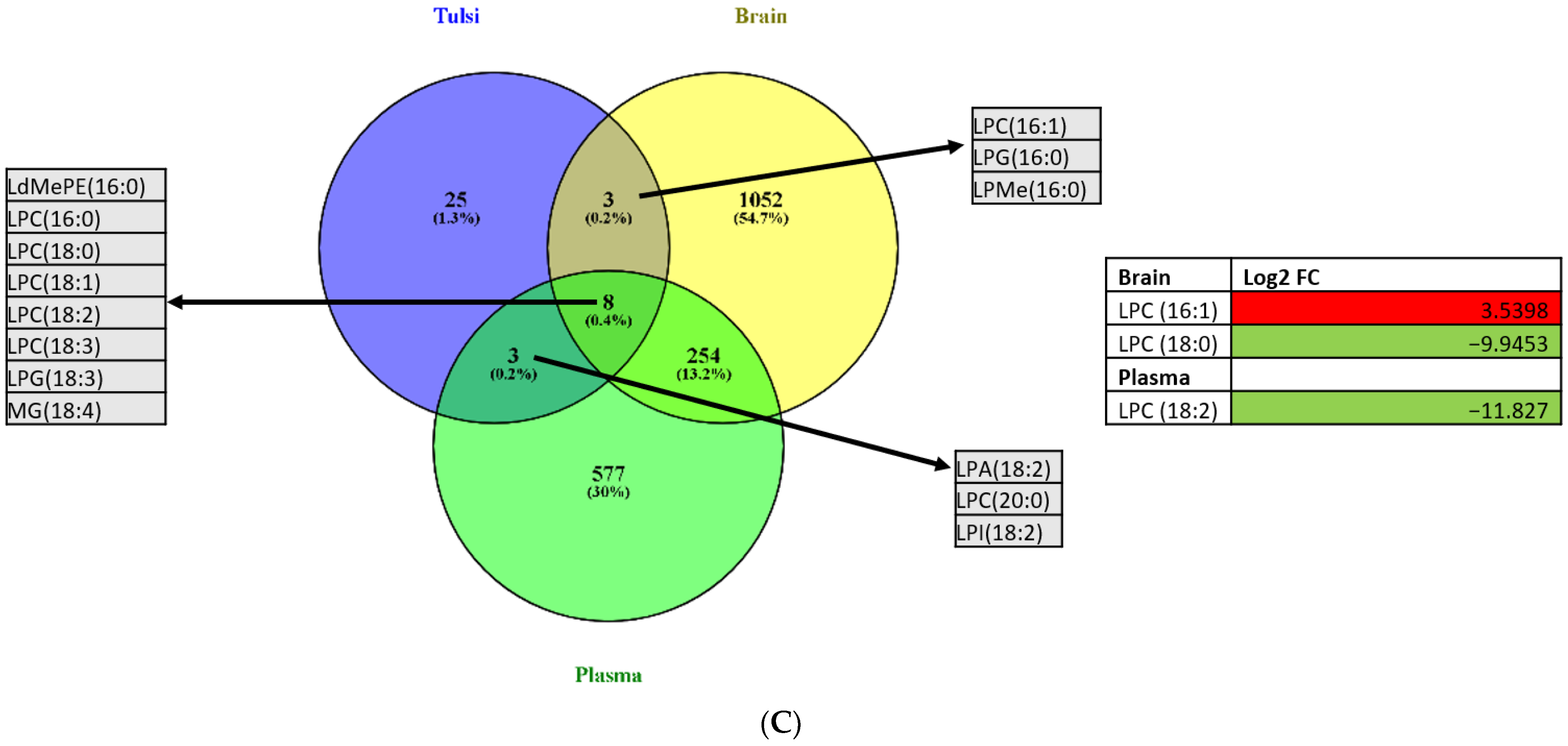
3.4. Integration of Brain and Plasma Lipidomic Analysis
3.5. Untargeted Lipidomic and Metabolomics of Ocimum sanctum
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; De Ferranti, S.; Després, J.P.; Fullerton, H.J.; Howard, V.J.; et al. Heart Disease and Stroke Statistics-2015 Update: A Report from the American Heart Association. Circulation 2015, 131, e29–e39. [Google Scholar] [CrossRef] [PubMed]
- Barber, P.A.; Demchuk, A.M.; Hirt, L.; Buchan, A.M. Biochemistry of Ischemic Stroke. Adv. Neurol. 2003, 92, 151–164. [Google Scholar] [PubMed]
- Jivad, N.; Rabiei, Z. A Review Study on Medicinal Plants Used in the Treatment of Learning and Memory Impairments. Asian Pac. J. Trop. Biomed. 2014, 4, 780–789. [Google Scholar] [CrossRef]
- Mackay, J.; Mensah, G. The Atlas of Heart Disease and Stroke; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Chang, Y.; Eom, S.; Kim, M.; Song, T.J. Medical Management of Dyslipidemia for Secondary Stroke Prevention: Narrative Review. Medicina 2023, 59, 776. [Google Scholar] [CrossRef]
- Chen, K.N.; He, L.; Zhong, L.M.; Ran, Y.Q.; Liu, Y. Meta-Analysis of Dyslipidemia Management for the Prevention of Ischemic Stroke Recurrence in China. Front. Neurol. 2020, 11, 1–8. [Google Scholar] [CrossRef]
- Lee, J.S.; Chang, P.Y.; Zhang, Y.; Kizer, J.R.; Best, L.G.; Howard, B.V. Triglyceride and HDL-C Dyslipidemia and Risks of Coronary Heart Disease and Ischemic Stroke by Glycemic Dysregulation Status: The Strong Heart Study. Diabetes Care 2017, 40, 529–537. [Google Scholar] [CrossRef]
- Yaghi, S.; Elkind, M.S.V. Lipids and Cerebrovascular Disease: Research and Practice. Stroke 2015, 46, 3322–3328. [Google Scholar] [CrossRef]
- Gu, X.; Li, Y.; Chen, S.; Yang, X.; Liu, F.; Li, Y.; Li, J.; Cao, J.; Liu, X.; Chen, J.; et al. Association of Lipids with Ischemic and Hemorrhagic Stroke a Prospective Cohort Study among 267,500 Chinese. Stroke 2019, 50, 3376–3384. [Google Scholar] [CrossRef]
- Kloska, A.; Malinowska, M.; Gabig-Cimińska, M.; Jakóbkiewicz-Banecka, J. Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke. Int. J. Mol. Sci. 2020, 21, 3618. [Google Scholar] [CrossRef]
- Liu, X.; Yan, L.; Xue, F. The Associations of Lipids and Lipid Ratios with Stroke: A Prospective Cohort Study. J. Clin. Hypertens. 2019, 21, 127–135. [Google Scholar] [CrossRef]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Imran, A.; Qasim, M.; Zafar, S.; Kamran, S.K.S.; Razzaq, A.; Aziz, N.; et al. Role of Cholesterol and Sphingolipids in Brain Development and Neurological Diseases. Lipids Health Dis. 2019, 18, 26. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Tie, C.; Wang, C.; Wu, C.; Zhang, J. Perturbed Lipidomic Profiles in Rats With Chronic Cerebral Ischemia Are Regulated by Xiao-Xu-Ming Decoction. Front. Pharmacol. 2019, 10, 264. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, S.; Liu, T.; Wu, J.; Zhang, H.; Sun, Z.; Liu, Z. Mass Spectrometry-Based Serum Lipidomics Strategy to Explore the Mechanism of: Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. Leaves in the Treatment of Ischemic Stroke. Food Funct. 2021, 12, 4519–4534. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Chen, M.; Luo, Y.; Wang, H.; Huang, H.; Peng, Z.; Li, M.; Fei, H.; Luo, W.; Yang, J. Lipidomic Profiling of Ipsilateral Brain and Plasma after Celastrol Post-Treatment in Transient Middle Cerebral Artery Occlusion Mice Model. Molecules 2021, 26, 4124. [Google Scholar] [CrossRef]
- Jaiswal, Y.S.; Williams, L.L. A Glimpse of Ayurveda—The Forgotten History and Principles of Indian Traditional Medicine. J. Tradit. Complement. Med. 2017, 7, 50–53. [Google Scholar] [CrossRef]
- Sharma, P.; Kumar, P.; Sharma, R.; Gupta, G.; Chaudhary, A. Immunomodulators: Role of Medicinal Plants in Immune System. Natl. J. Physiol. Pharm. Pharmacol. 2017, 7, 552–556. [Google Scholar] [CrossRef]
- Almatroodi, S.A.; Alsahli, M.A.; Almatroudi, A.; Rahmani, A.H. Ocimum Sanctum: Role in Diseases Management through Modulating Various Biological Activity. Pharmacogn. J. 2020, 12, 1198–1205. [Google Scholar] [CrossRef]
- Hanumanthaiah, P.; Panari, H.; Chebte, A.; Haile, A.; Belachew, G.T. Tulsi (Ocimum sanctum)—A Myriad Medicinal Plant, Secrets behind the Innumerable Benefits. Arab. J. Med. Aromat. Plants 2020, 6, 106–127. [Google Scholar] [CrossRef]
- Jamshidi, N.; Cohen, M.M. The Clinical Efficacy and Safety of Tulsi in Humans: A Systematic Review of the Literature. Evid.-Based Complement. Altern. Med. 2017, 2017, 9217567. [Google Scholar] [CrossRef]
- Cohen, M.M. Tulsi-Ocimum Sanctum: A Herb for All Reasons. J. Ayurveda Integr. Med. 2014, 5, 251–259. [Google Scholar] [CrossRef]
- Singh, N.; Hoette, Y.; Miller, D.R. Tulsi: The Mother Medicine of Nature; International Institute of Herbal Medicine: Lucknow, India, 2002; ISBN 8188007005. [Google Scholar]
- Mohan, L.; Amberkar, M.V.; Kumari, M. Ocimum sanctum Linn. (TULSI)—An Overview. Int. J. Pharm. Sci. Rev. 2011, 7, 51–53. [Google Scholar]
- Pattanayak, P.; Behera, P.; Das, D.; Panda, S.K. Ocimum sanctum Linn. A Reservoir Plant for Therapeutic Applications: An Overview. Pharmacogn. Rev. 2010, 4, 95. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Mirdha, B.R.; Mahapatra, S.C. The Science behind Sacredness of Tulsi (Ocimum sanctum Linn.). Indian J. Physiol. Pharmacol. 2009, 53, 291–306. [Google Scholar]
- Jaggi, R.K.; Madaan, R.; Singh, B. Anticonvulsant Potential of Holy Basil, Ocimum sanctum Linn., and Its Cultures. Indian J. Exp. Biol. 2003, 41, 1329–1333. [Google Scholar] [PubMed]
- Yanpallewar, S.U.; Rai, S.; Kumar, M.; Acharya, S.B. Evaluation of Antioxidant and Neuroprotective Effect of Ocimum sanctum on Transient Cerebral Ischemia and Long-Term Cerebral Hypoperfusion. Pharmacol. Biochem. Behav. 2004, 79, 155–164. [Google Scholar] [CrossRef]
- De Almeida, I.; Alviano, D.S.; Vieira, D.P.; Alves, P.B.; Blank, A.F.; Lopes, A.H.C.S.; Alviano, C.S.; Rosa, M.D.S.S. Antigiardial Activity of Ocimum basilicum Essential Oil. Parasitol. Res. 2007, 101, 443–452. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Sur, T.K.; Jana, U.; Debnath, P.K. Controlled Programmed Trial of Ocimum sanctum Leaf on Generalized Anxiety Disorders. Nepal Med. Coll. J. 2008, 10, 176–179. [Google Scholar]
- Baliga, M.S.; Jimmy, R.; Thilakchand, K.R.; Sunitha, V.; Bhat, N.R.; Saldanha, E.; Rao, S.; Rao, P.; Arora, R.; Palatty, P.L. Ocimum sanctum L (Holy Basil or Tulsi) and Its Phytochemicals in the Prevention and Treatment of Cancer. Nutr. Cancer 2013, 65, 26–35. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, M.M.; Raza, S.S.; Javed, H.; Ashafaq, M.; Islam, F.; Safhi, M.M.; Islam, F. Ocimum sanctum Attenuates Oxidative Damage and Neurological Deficits Following Focal Cerebral Ischemia/Reperfusion Injury in Rats. Neurol. Sci. 2012, 33, 1239–1247. [Google Scholar] [CrossRef]
- Kothari, S.K.; Bhattacharya, A.K.; Ramesh, S.; Garg, S.N.; Khanuja, S.P.S. Volatile Constituents in Oil from Different Plant Parts of Methyl Eugenol-Rich Ocimum tenuiflorum L.F. (Syn. O. Sanctum L.) Grown in South India. J. Essent. Oil Res. 2005, 17, 656–658. [Google Scholar] [CrossRef]
- Ahmad, A.; Abuzinadah, M.F.; Alkreathy, H.M.; Banaganapalli, B.; Mujeeb, M. Ursolic Acid Rich Ocimum sanctum L Leaf Extract Loaded Nanostructured Lipid Carriers Ameliorate Adjuvant Induced Arthritis in Rats by Inhibition of COX-1, COX-2, TNF-α and IL-1: Pharmacological and Docking Studies. PLoS ONE 2018, 13, e0193451. [Google Scholar] [CrossRef] [PubMed]
- Godhwani, J.L.; Vyas, D.S. Ocimum sanctuim: An Experimental Study Evaluating Its Anti-Inflammatory, Analgesic Andantipyretic Activity in Animals. J. Ethnopharmacol. 1987, 21, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Hannan, J.M.A.; Das, B.K.; Uddin, A.; Bhattacharjee, R.; Das, B.; Chowdury, H.S.; Mosaddek, A.S.M. Analgesic and Anti-Inflammatory Effects of Ocimum sanctum (Linn) in Laboratory Animals. Int. J. Pharm. Sci. Res. 2011, 2, 2121–2125. [Google Scholar]
- Yuniarti, W.M.; Krismaharani, N.; Ciptaningsih, P.; Celia, K.; Veteriananta, K.D.; Ma’ruf, A.; Lukiswanto, B.S. The Protective Effect of Ocimum sanctum Leaf Extract against Lead Acetate-Induced Nephrotoxicity and Hepatotoxicity in Mice (Mus Musculus). Vet. World 2021, 14, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Raina, P.; Chandrasekaran, C.V.; Deepak, M.; Agarwal, A.; Ruchika, K.G. Evaluation of Subacute Toxicity of Methanolic/Aqueous Preparation of Aerial Parts of O. Sanctum in Wistar Rats: Clinical, Haematological, Biochemical and Histopathological Studies. J. Ethnopharmacol. 2015, 175, 509–517. [Google Scholar] [CrossRef]
- Gautam, M.K.; Goel, R.K. Toxicological Study of Ocimum sanctum Linn Leaves: Hematological, Biochemical, and Histopathological Studies. J. Toxicol. 2014, 2014, 135654. [Google Scholar] [CrossRef]
- Das, S.; Das, S.; Das, M.K.; Basu, S.P. Evaluation of Anti-Inflammatory Effect of Calotropis Gigantea and Tridax Procumbens on Wistar Albino Rats. J. Pharm. Sci. Res. 2009, 1, 123–126. [Google Scholar]
- Baghdadi, H.H.; El-Demerdash, F.M.; Hussein, S.; Radwan, E.H. The Protective Effect of Coriandrum sativum L. Oil against Liver Toxicity Induced by Ibuprofen in Rats. J. Biosci. Appl. Res. 2016, 2, 197–202. [Google Scholar] [CrossRef]
- Watson, B.D.; Dietrich, W.D.; Busto, R.; Wachtel, M.S.; Ginsberg, M.D. Induction of Reproducible Brain Infarction by Photochemically Initiated Thrombosis. Ann. Neurol. 1985, 17, 497–504. [Google Scholar] [CrossRef]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates; Academic Press: Cambridge, MA, USA, 2001. [Google Scholar]
- Sharma, N.; Yadav, M.; Tripathi, G.; Mathew, B.; Bindal, V.; Falari, S.; Pamecha, V.; Maras, J.S. Bile Multi-Omics Analysis Classifies Lipid Species and Microbial Peptides Predictive of Carcinoma of Gallbladder. Hepatology 2022, 76, 920–935. [Google Scholar] [CrossRef]
- Pang, Z.; Chong, J.; Zhou, G.; de Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.-É.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the Gap between Raw Spectra and Functional Insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef]
- Chi, Y.; Ma, Q.; Ding, X.Q.; Qin, X.; Wang, C.; Zhang, J. Research on Protective Mechanism of Ibuprofen in Myocardial Ischemia-Reperfusion Injury in Rats through the PI3K/Akt/MTOR Signaling Pathway. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 4465–4473. [Google Scholar] [CrossRef] [PubMed]
- Dokmeci, D.; Kanter, M.; Inan, M.; Aydogdu, N.; Basaran, U.N.; Yalcin, O.; Turan, F.N. Protective Effects of Ibuprofen on Testicular Torsion/Detorsion-Induced Ischemia/Reperfusion Injury in Rats. Arch. Toxicol. 2007, 81, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Iwata, Y.; Nicole, O.; Zurakowski, D.; Okamura, T.; Jonas, R.A. Ibuprofen for Neuroprotection after Cerebral Ischemia. J. Thorac. Cardiovasc. Surg. 2010, 139, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Park, E.M.; Cho, B.P.; Volpe, B.T.; Cruz, M.O.; Joh, T.H.; Cho, S. Ibuprofen Protects Ischemia-Induced Neuronal Injury via up-Regulating Interleukin-1 Receptor Antagonist Expression. Neuroscience 2005, 132, 625–631. [Google Scholar] [CrossRef]
- Giridharan, V.V.; Thandavarayan, R.A.; Mani, V.; Ashok Dundapa, T.; Watanabe, K.; Konishi, T. Ocimum sanctum Linn. Leaf Extracts Inhibit Acetylcholinesterase and Improve Cognition in Rats with Experimentally Induced Dementia. J. Med. Food 2011, 14, 912–919. [Google Scholar] [CrossRef]
- Crack, P.J.; Taylor, J.M. Reactive Oxygen Species and the Modulation of Stroke. Free Radic. Biol. Med. 2005, 38, 1433–1444. [Google Scholar] [CrossRef]
- Saeed, S.A.; Shad, K.F.; Saleem, T.; Javed, F.; Khan, M.U. Some New Prospects in the Understanding of the Molecular Basis of the Pathogenesis of Stroke. Exp. Brain Res. 2007, 182, 1–10. [Google Scholar] [CrossRef]
- Kothari, A.; Sharma, S. Evaluation of Anti-Inflammatory Effect of Fresh Tulsi Leaves (Ocimum sanctum) against Different Mediators of Inflammation in Albino Rats. Int. J. Pharm. Sci. Rev. Res. 2012, 14, 119–123. [Google Scholar]
- Fernández, P.B.; Figueredo, Y.N.; Dominguez, C.C.; Hernández, I.C.; Sanabria, M.L.G.; González, R. Anti-Inflammatory Effect of Lyophilized Aqueous Extract of Ocinum tenuiflorum on Rats. Acta Farm Bonaer. 2004, 23, 92–97. [Google Scholar]
- Thakur, K.; Chem, K.P.-R.J. Undefined Anti-Inflammatory Activity of Extracted Eugenol from Ocimum sanctum L. Leaves. Citeseer 2009, 2, 472–474. [Google Scholar]
- Singh, S.; Majumdar, D.K. Evaluation of Antiinflammatory Activity of Fatty Acids of Ocimum sanctum Fixed Oil. Indian J. Exp. Biol. 1997, 35, 380–383. [Google Scholar] [PubMed]
- Singh, S. Comparative Evaluation of Antiinflammatory Potential of Fixed Oil of Different Species of Ocimum and Its Possible Mechanism of Action. Indian J. Exp. Biol. 1998, 36, 1028–1031. [Google Scholar] [PubMed]
- Klein, M.E.; Mauch, S.; Rieckmann, M.; Martínez, D.G.; Hause, G.; Noutsias, M.; Hofmann, U.; Lucas, H.; Meister, A.; Ramos, G.; et al. Phosphatidylserine (PS) and Phosphatidylglycerol (PG) Nanodispersions as Potential Anti-Inflammatory Therapeutics: Comparison of in Vitro Activity and Impact of Pegylation. Nanomed. Nanotechnol. Biol. Med. 2020, 23, 102096. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.E.; Rieckmann, M.; Sedding, D.; Hause, G.; Meister, A.; Mäder, K.; Lucas, H. Towards the Development of Long Circulating Phosphatidylserine (Ps)- and Phosphatidylglycerol (Pg)-enriched Anti-inflammatory Liposomes: Is Pegylation Effective? Pharmaceutics 2021, 13, 282. [Google Scholar] [CrossRef]
- Klein, M.E.; Rieckmann, M.; Lucas, H.; Meister, A.; Loppnow, H.; Mäder, K. Phosphatidylserine (PS) and Phosphatidylglycerol (PG) Enriched Mixed Micelles (MM): A New Nano-Drug Delivery System with Anti-Inflammatory Potential? Eur. J. Pharm. Sci. 2020, 152, 105451. [Google Scholar] [CrossRef]
- Treede, I.; Braun, A.; Sparla, R.; Kühnel, M.; Giese, T.; Turner, J.R.; Anes, E.; Kulaksiz, H.; Füllekrug, J.; Stremmel, W.; et al. Anti-Inflammatory Effects of Phosphatidylcholine. J. Biol. Chem. 2007, 282, 27155–27164. [Google Scholar] [CrossRef]
- Erõs, G.; Varga, G.; Váradi, R.; Czóbel, M.; Kaszaki, J.; Ghyczy, M.; Boros, M. Anti-Inflammatory Action of a Phosphatidylcholine, Phosphatidylethanolamine and N-Acylphosphatidylethanolamine-Enriched Diet in Carrageenan-Induced Pleurisy. Eur. Surg. Res. 2008, 42, 40–48. [Google Scholar] [CrossRef]
- Chen, L.; Beppu, F.; Takatani, N.; Miyashita, K.; Hosokawa, M. N-3 Polyunsaturated Fatty Acid-Enriched Phosphatidylglycerol Suppresses Inflammation in RAW264.7 Cells through Nrf2 Activation via Alteration of Fatty Acids in Cellular Phospholipids. Fish. Sci. 2021, 87, 727–737. [Google Scholar] [CrossRef]
- Ireland, R.; Schwarz, B.; Nardone, G.; Wehrly, T.D.; Broeckling, C.D.; Chiramel, A.I.; Best, S.M.; Bosio, C.M. Unique Francisella Phosphatidylethanolamine Acts as a Potent Anti-Inflammatory Lipid. J. Innate Immun. 2018, 10, 291–305. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, P.; Gong, F.; Tan, Y.; Han, J.; Tian, L.; Yan, J.; Li, K.; Xi, Z.; Liu, X. Altered Lipidomic Profiles in Lung and Serum of Rat after Sub-Chronic Exposure to Ozone. Sci. Total Environ. 2022, 806, 150630. [Google Scholar] [CrossRef]
- Ulivi, V.; Lenti, M.; Gentili, C.; Marcolongo, G.; Cancedda, R.; Descalzi Cancedda, F. Anti-Inflammatory Activity of Monogalactosyldiacylglycerol in Human Articular Cartilage in Vitro: Activation of an Anti-Inflammatory Cyclooxygenase-2 (COX-2) Pathway. Arthritis Res. Ther. 2011, 13, R92. [Google Scholar] [CrossRef] [PubMed]
- Bruno, A.; Rossi, C.; Marcolongo, G.; Di Lena, A.; Venzo, A.; Berrie, C.P.; Corda, D. Selective in Vivo Anti-Inflammatory Action of the Galactolipid Monogalactosyldiacylglycerol. Eur. J. Pharmacol. 2005, 524, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Zi, Y.; Yao, M.; Lu, Z.; Lu, F.; Bie, X.; Zhang, C.; Zhao, H. Glycoglycerolipids from the Leaves of Perilla frutescens (L.) Britton (Labiatae) and Their Anti-Inflammatory Activities in Lipopolysaccharide-Stimulated RAW264.7 Cells. Phytochemistry 2021, 184, 112679. [Google Scholar] [CrossRef] [PubMed]
- Leutou, A.S.; McCall, J.R.; York, R.; Govindapur, R.R.; Bourdelais, A.J. Anti-Inflammatory Activity of Glycolipids and a Polyunsaturated Fatty Acid Methyl Ester Isolated from the Marine Dinoflagellate Karenia Mikimotoi. Mar. Drugs 2020, 18, 138. [Google Scholar] [CrossRef]
- Maeda, N.; Kokai, Y.; Hada, T.; Yoshida, H.; Mizushina, Y. Oral Administration of Monogalactosyl Diacylglycerol from Spinach Inhibits Colon Tumor Growth in Mice. Exp. Ther. Med. 2013, 5, 17–22. [Google Scholar] [CrossRef]
- Murakami, C.; Kumagai, T.; Hada, T.; Kanekazu, U.; Nakazawa, S.; Kamisuki, S.; Maeda, N.; Xu, X.; Yoshida, H.; Sugawara, F.; et al. Effects of Glycolipids from Spinach on Mammalian DNA Polymerases. Biochem. Pharmacol. 2003, 65, 259–267. [Google Scholar] [CrossRef]
- Calzada, E.; Onguka, O.; Claypool, S.M. Phosphatidylethanolamine Metabolism in Health and Disease. Int. Rev. Cell Mol. Biol. 2016, 321, 29–88. [Google Scholar] [CrossRef]
- Akyol, S.; Ugur, Z.; Yilmaz, A.; Ustun, I.; Gorti, S.K.K.; Oh, K.; McGuinness, B.; Passmore, P.; Kehoe, P.G.; Maddens, M.E.; et al. Lipid Profiling of Alzheimer’s Disease Brain Highlights Enrichment in Glycerol(Phospho)Lipid, and Sphingolipid Metabolism. Cells 2021, 10, 2591. [Google Scholar] [CrossRef]
- Rao, A.M.; Hatcher, J.F.; Dempsey, R.J. Lipid Alterations in Transient Forebrain Ischemia: Possible New Mechanisms of CDP-Choline Neuroprotection. J. Neurochem. 2000, 75, 2528–2535. [Google Scholar] [CrossRef]
- Yang, Y.; Zhong, Q.; Zhang, H.; Mo, C.; Yao, J.; Huang, T.; Zhou, T.; Tan, W. Lipidomics Study of the Protective Effects of Isosteviol Sodium on Stroke Rats Using Ultra High-Performance Supercritical Fluid Chromatography Coupling with Ion-Trap and Time-of-Flight Tandem Mass Spectrometry. J. Pharm. Biomed. Anal. 2018, 157, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Glade, M.J.; Smith, K. Phosphatidylserine and the Human Brain. Nutrition 2015, 31, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Huang, B.X.; Spector, A.A. Phosphatidylserine in the Brain: Metabolism and Function. Prog. Lipid Res. 2014, 56, 1–18. [Google Scholar] [CrossRef]
- Dharsono, H.D.A.; Putri, S.A.; Kurnia, D.; Dudi, D.; Satari, M.H. Ocimum Species: A Review on Chemical Constituents and Antibacterial Activity. Molecules 2022, 27, 6350. [Google Scholar] [CrossRef] [PubMed]
- Ramaiah, M.; Prathi, A.; Singam, B.; Tulluru, G.; Tummala, L. A Review on Ocimum Species: Ocimum americanum L., Ocimum basilicum L., Ocimum gratissimum L. and Ocimum tenuiflorum L. Int. J. Res. Ayurveda Pharm. 2019, 10, 41–48. [Google Scholar] [CrossRef]
- Rahman, S.; Islam, R.; Kamruzzaman, M.; Alam, K.; Jamal, A.H.M. Ocimum sanctum L.: A Review of Phytochemical and Pharmacological Profile. Am. J. Drug Discov. Dev. 2011, 1, 1–15. [Google Scholar]
- Liu, J. Pharmacology of Oleanolic Acid and Ursolic Acid. J. Ethnopharmacol. 1995, 49, 57–68. [Google Scholar] [CrossRef]
- Kazmi, I.; Narooka, A.R.; Afzal, M.; Singh, R.; Al-Abbasi, F.A.; Ahmad, A.; Anwar, F. Anticancer Effect of Ursolic Acid Stearoyl Glucoside in Chemically Induced Hepatocellular Carcinoma. J. Physiol. Biochem. 2013, 69, 687–695. [Google Scholar] [CrossRef]
- Kazmi, I.; Afzal, M.; Gupta, G.; Anwar, F. Antiepileptic Potential of Ursolic Acid Stearoyl Glucoside by Gaba Receptor Stimulation. CNS Neurosci. Ther. 2012, 18, 799–800. [Google Scholar] [CrossRef]
- Kazmi, I.; Rahman, M.; Afzal, M.; Gupta, G.; Saleem, S.; Afzal, O.; Shaharyar, M.A.; Nautiyal, U.; Ahmed, S.; Anwar, F. Anti-Diabetic Potential of Ursolic Acid Stearoyl Glucoside: A New Triterpenic Gycosidic Ester from Lantana Camara. Fitoterapia 2012, 83, 142–146. [Google Scholar] [CrossRef]
- Kim, S.H.; Hong, J.H.; Lee, Y.C. Ursolic Acid, a Potential PPARγ Agonist, Suppresses Ovalbumin-Induced Airway Inflammation and Penh by down-Regulating IL-5, IL-13, and IL-17 in a Mouse Model of Allergic Asthma. Eur. J. Pharmacol. 2013, 701, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, S.; Azad, H.; Rathinavelu, A. Apoptosis Induction by Ocimum sanctum Extract in LNCaP Prostate Cancer Cells. J. Med. Food 2015, 18, 776–785. [Google Scholar] [CrossRef]
- Ding, H.; Wang, H.; Zhu, L.; Wei, W. Ursolic Acid Ameliorates Early Brain Injury After Experimental Traumatic Brain Injury in Mice by Activating the Nrf2 Pathway. Neurochem. Res. 2016, 42, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Su, J.; Wang, K.; Zhu, T.; Li, X. Ursolic Acid Reduces Oxidative Stress to Alleviate Early Brain Injury Following Experimental Subarachnoid Hemorrhage. Neurosci. Lett. 2014, 579, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Su, J.; Guo, B.; Zhu, T.; Wang, K.; Li, X. Ursolic Acid Alleviates Early Brain Injury after Experimental Subarachnoid Hemorrhage by Suppressing {TLR}4-Mediated Inflammatory Pathway. Int. Immunopharmacol. 2014, 23, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, L.; Deng, S.; Liu, F.; He, Z. Ursolic Acid Ameliorates Inflammation in Cerebral Ischemia and Reperfusion Injury Possibly via High Mobility Group Box 1/Toll-like Receptor 4/NFκB Pathway. Front. Neurol. 2018, 9, 253. [Google Scholar] [CrossRef]
- Papangelis, A.; Ulven, T. Synthesis of Lysophosphatidylcholine and Mixed Phosphatidylcholine. J. Org. Chem. 2022, 87, 8194–8197. [Google Scholar] [CrossRef]
- Whitehead, S.N.; Chan, K.H.N.; Gangaraju, S.; Slinn, J.; Li, J.; Hou, S.T. Imaging Mass Spectrometry Detection of Gangliosides Species in the Mouse Brain Following Transient Focal Cerebral Ischemia and Long-Term Recovery. PLoS ONE 2011, 6, e20808. [Google Scholar] [CrossRef]
- Jove, M.; Mauri-Capdevila, G.; Suarez, I.; Cambray, S.; Sanahuja, J.; Quilez, A.; Farre, J.; Benabdelhak, I.; Pamplona, R.; Portero-Otin, M.; et al. Metabolomics Predicts Stroke Recurrence after Transient Ischemic Attack. Neurology 2014, 84, 36–45. [Google Scholar] [CrossRef]
- Koizumi, S.; Yamamoto, S.; Hayasaka, T.; Konishi, Y.; Yamaguchi-Okada, M.; Goto-Inoue, N.; Sugiura, Y.; Setou, M.; Namba, H. Imaging Mass Spectrometry Revealed the Production of Lyso-Phosphatidylcholine in the Injured Ischemic Rat Brain. Neuroscience 2010, 168, 219–225. [Google Scholar] [CrossRef]
- Wang, H.Y.J.; Liu, C.B.; Wu, H.W.; Kuo, S. Direct Profiling of Phospholipids and Lysophospholipids in Rat Brain Sections after Ischemic Stroke. Rapid Commun. Mass Spectrom. 2010, 24, 2057–2064. [Google Scholar] [CrossRef] [PubMed]
- Takenouchi, T.; Sato, M.; Kitani, H. Lysophosphatidylcholine Potentiates Ca2+ Influx, Pore Formation and P44/42 MAP Kinase Phosphorylation Mediated by P2X7 Receptor Activation in Mouse Microglial Cells. J. Neurochem. 2007, 102, 1518–1532. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, I.; Sharma, N.; Velayudhan, R.; Fatima, Z.; Maras, J.S. Ocimum sanctum Alters the Lipid Landscape of the Brain Cortex and Plasma to Ameliorate the Effect of Photothrombotic Stroke in a Mouse Model. Life 2023, 13, 1877. https://doi.org/10.3390/life13091877
Yadav I, Sharma N, Velayudhan R, Fatima Z, Maras JS. Ocimum sanctum Alters the Lipid Landscape of the Brain Cortex and Plasma to Ameliorate the Effect of Photothrombotic Stroke in a Mouse Model. Life. 2023; 13(9):1877. https://doi.org/10.3390/life13091877
Chicago/Turabian StyleYadav, Inderjeet, Nupur Sharma, Rema Velayudhan, Zeeshan Fatima, and Jaswinder Singh Maras. 2023. "Ocimum sanctum Alters the Lipid Landscape of the Brain Cortex and Plasma to Ameliorate the Effect of Photothrombotic Stroke in a Mouse Model" Life 13, no. 9: 1877. https://doi.org/10.3390/life13091877






