Efficacy of Selinexor in Relapsed/Refractory Multiple Myeloma (RRMM) Patients with del17p and Other High-Risk Abnormalities (A Retrospective Single-Center Study)
Abstract
:1. Introduction
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Razak, A.R.A.; Mau-Soerensen, M.; Gabrail, N.Y.; Gerecitano, J.F.; Shields, A.F.; Unger, T.J.; Saint-Martin, J.R.; Carlson, R.; Landesman, Y.; McCauley, D. First-in-class, first-in-human phase I study of selinexor, a selective inhibitor of nuclear export, in patients with advanced solid tumors. J. Clin. Oncol. 2016, 34, 4142. [Google Scholar] [CrossRef] [PubMed]
- Tai, Y.; Landesman, Y.; Acharya, C.; Calle, Y.; Zhong, M.; Cea, M.; Tannenbaum, D.; Cagnetta, A.; Reagan, M.; Munshi, A. CRM1 inhibition induces tumor cell cytotoxicity and impairs osteoclastogenesis in multiple myeloma: Molecular mechanisms and therapeutic implications. Leukemia 2014, 28, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Neupane, K.; Wahab, A.; Masood, A.; Faraz, T.; Bahram, S.; Ehsan, H.; Hannan, A.; Anwer, F. Profile and Management of Toxicity of Selinexor and Belantamab Mafodotin for the Treatment of Triple Class Refractory Multiple Myeloma. J. Blood Med. 2021, 12, 529–550. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Ishizawa, J.; Ruvolo, V.; Dilip, A.; Quintás-Cardama, A.; McDonnell, T.J.; Neelapu, S.S.; Kwak, L.W.; Shacham, S.; Kauffman, M. Induction of p53-mediated transcription and apoptosis by exportin-1 (XPO 1) inhibition in mantle cell lymphoma. Cancer Sci. 2014, 105, 795–801. [Google Scholar] [CrossRef]
- Schmidt, J.; Braggio, E.; Kortuem, K.; Egan, J.; Zhu, Y.; Xin, C.; Tiedemann, R.; Palmer, S.; Garbitt, V.; McCauley, D. Genome-wide studies in multiple myeloma identify XPO1/CRM1 as a critical target validated using the selective nuclear export inhibitor KPT-276. Leukemia 2013, 27, 2357–2365. [Google Scholar] [CrossRef]
- Fung, H.Y.J.; Chook, Y.M. (Eds.) Atomic basis of CRM1-cargo recognition, release and inhibition. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- van der Watt, P.J.; Leaner, V.D. The nuclear exporter, Crm1, is regulated by NFY and Sp1 in cancer cells and repressed by p53 in response to DNA damage. Biochim. Biophys. Acta BBA Gene Regul. Mech. 2011, 1809, 316–326. [Google Scholar] [CrossRef]
- Chen, C.; Siegel, D.; Gutierrez, M.; Jacoby, M.; Hofmeister, C.C.; Gabrail, N.; Baz, R.; Mau-Sorensen, M.; Berdeja, J.G.; Savona, M.; et al. Safety and efficacy of selinexor in relapsed or refractory multiple myeloma and Waldenstrom macroglobulinemia. Blood 2018, 131, 855–863. [Google Scholar] [CrossRef]
- Vousden, K.H.; Lu, X. Live or let die: The cell’s response to p53. Nat. Rev. Cancer 2002, 2, 594–604. [Google Scholar] [CrossRef]
- Mahfoudhi, E.; Lordier, L.; Marty, C.; Pan, J.; Roy, A.; Roy, L.; Rameau, P.; Abbes, S.; Debili, N.; Raslova, H. P53 activation inhibits all types of hematopoietic progenitors and all stages of megakaryopoiesis. Oncotarget 2016, 7, 31980. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Simpson, E.R.; Brown, K.A. p53: Protection against tumor growth beyond effects on cell cycle and apoptosis. Cancer Res. 2015, 75, 5001–5007. [Google Scholar] [CrossRef]
- Chin, M.; Sive, J.; Allen, C.; Roddie, C.; Chavda, S.; Smith, D.; Blombery, P.; Jones, K.; Ryland, G.; Popat, R. Prevalence and timing of TP53 mutations in del (17p) myeloma and effect on survival. Blood Cancer J. 2017, 7, e610. [Google Scholar] [CrossRef]
- Lionetti, M.; Barbieri, M.; Manzoni, M.; Fabris, S.; Bandini, C.; Todoerti, K.; Nozza, F.; Rossi, D.; Musto, P.; Baldini, L. Molecular spectrum of TP53 mutations in plasma cell dyscrasias by next generation sequencing: An Italian cohort study and overview of the literature. Oncotarget 2016, 7, 21353. [Google Scholar] [CrossRef]
- Lakshman, A.; Painuly, U.; Rajkumar, S.V.; Ketterling, R.P.; Kapoor, P.; Greipp, P.T.; Gertz, M.A.; Buadi, F.K.; Lacy, M.Q.; Dingli, D. Natural history of multiple myeloma with de novo del (17p). Blood Cancer J. 2019, 9, 32. [Google Scholar] [CrossRef]
- Bergsagel, P.L.; Mateos, M.-V.; Gutierrez, N.C.; Rajkumar, S.V.; San Miguel, J.F. Improving overall survival and overcoming adverse prognosis in the treatment of cytogenetically high-risk multiple myeloma. Blood J. Am. Soc. Hematol. 2013, 121, 884–892. [Google Scholar] [CrossRef]
- Jovanović, K.K.; Escure, G.; Demonchy, J.; Willaume, A.; Van de Wyngaert, Z.; Farhat, M.; Chauvet, P.; Facon, T.; Quesnel, B.; Manier, S. Deregulation and Targeting of TP53 Pathway in Multiple Myeloma. Front. Oncol. 2018, 8, 665. [Google Scholar] [CrossRef]
- Drach, J.; Ackermann, J.; Fritz, E.; Krömer, E.; Schuster, R.; Gisslinger, H.; DeSantis, M.; Zojer, N.; Fiegl, M.; Roka, S.; et al. Presence of a p53 Gene Deletion in Patients With Multiple Myeloma Predicts for Short Survival After Conventional-Dose Chemotherapy. Blood 1998, 92, 802–809. [Google Scholar] [CrossRef]
- Sonneveld, P.; Avet-Loiseau, H.; Lonial, S.; Usmani, S.; Siegel, D.; Anderson, K.C.; Chng, W.-J.; Moreau, P.; Attal, M.; Kyle, R.A. Treatment of multiple myeloma with high-risk cytogenetics: A consensus of the International Myeloma Working Group. Blood J. Am. Soc. Hematol. 2016, 127, 2955–2962. [Google Scholar] [CrossRef]
- Muz, B.; Azab, F.; de la Puente, P.; Landesman, Y.; Azab, A.K. Selinexor overcomes hypoxia-induced drug resistance in multiple myeloma. Transl. Oncol. 2017, 10, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Malandrakis, P.; Ntanasis-Stathopoulos, I.; Gavriatopoulou, M.; Terpos, E. Clinical utility of selinexor/dexamethasone in patients with relapsed or refractory multiple myeloma: A review of current evidence and patient selection. OncoTargets Ther. 2020, 13, 6405–6416. [Google Scholar] [CrossRef] [PubMed]
- Sellin, M.; Berg, S.; Hagen, P.; Zhang, J. The molecular mechanism and challenge of targeting XPO1 in treatment of relapsed and refractory myeloma. Transl. Oncol. 2022, 22, 101448. [Google Scholar] [CrossRef] [PubMed]
- Chari, A.; Vogl, D.T.; Gavriatopoulou, M.; Nooka, A.K.; Yee, A.J.; Huff, C.A.; Moreau, P.; Dingli, D.; Cole, C.; Lonial, S.; et al. Oral Selinexor–Dexamethasone for Triple-Class Refractory Multiple Myeloma. N. Engl. J. Med. 2019, 381, 727–738. [Google Scholar] [CrossRef]
- Grosicki, S.; Simonova, M.; Spicka, I.; Pour, L.; Kriachok, I.; Gavriatopoulou, M.; Pylypenko, H.; Auner, H.W.; Leleu, X.; Doronin, V. Once-per-week selinexor, bortezomib, and dexamethasone versus twice-per-week bortezomib and dexamethasone in patients with multiple myeloma (BOSTON): A randomised, open-label, phase 3 trial. Lancet 2020, 396, 1563–1573. [Google Scholar] [CrossRef] [PubMed]
- Avet-Loiseau, H.; Attal, M.; Moreau, P.; Charbonnel, C.; Garban, F.d.r.; Hulin, C.; Leyvraz, S.; Michallet, M.; Yakoub-Agha, I.; Garderet, L. Genetic abnormalities and survival in multiple myeloma: The experience of the Intergroupe Francophone du Myelome. Blood 2007, 109, 3489–3495. [Google Scholar] [CrossRef] [PubMed]
- Afrough, A.; Hashmi, H.; Hansen, D.K.; Sidana, S.; Ahn, C.; Dima, D.; Freeman, C.L.; Puglianini, O.A.C.; Kocoglu, M.H.; Atrash, S.; et al. Impact of bridging therapy (BT) on outcome of relapsed refractory multiple myeloma (RRMM) with Ide-cel CAR T-cell therapy: Real-world experience from the US myeloma CAR T consortium. J. Clin. Oncol. 2023, 41, 8013. [Google Scholar] [CrossRef]
- Van Oekelen, O.; Nath, K.; Mouhieddine, T.H.; Farzana, T.; Aleman, A.; Melnekoff, D.T.; Ghodke-Puranik, Y.; Shah, G.L.; Lesokhin, A.; Giralt, S.; et al. Interventions and outcomes of patients with multiple myeloma receiving salvage therapy after BCMA-directed CAR T therapy. Blood 2023, 141, 756–765. [Google Scholar] [CrossRef]
- Vogl, D.T.; Dingli, D.; Cornell, R.F.; Huff, C.A.; Jagannath, S.; Bhutani, D.; Zonder, J.; Baz, R.; Nooka, A.; Richter, J.; et al. Selective Inhibition of Nuclear Export With Oral Selinexor for Treatment of Relapsed or Refractory Multiple Myeloma. J. Clin. Oncol. 2018, 36, 859–866. [Google Scholar] [CrossRef]
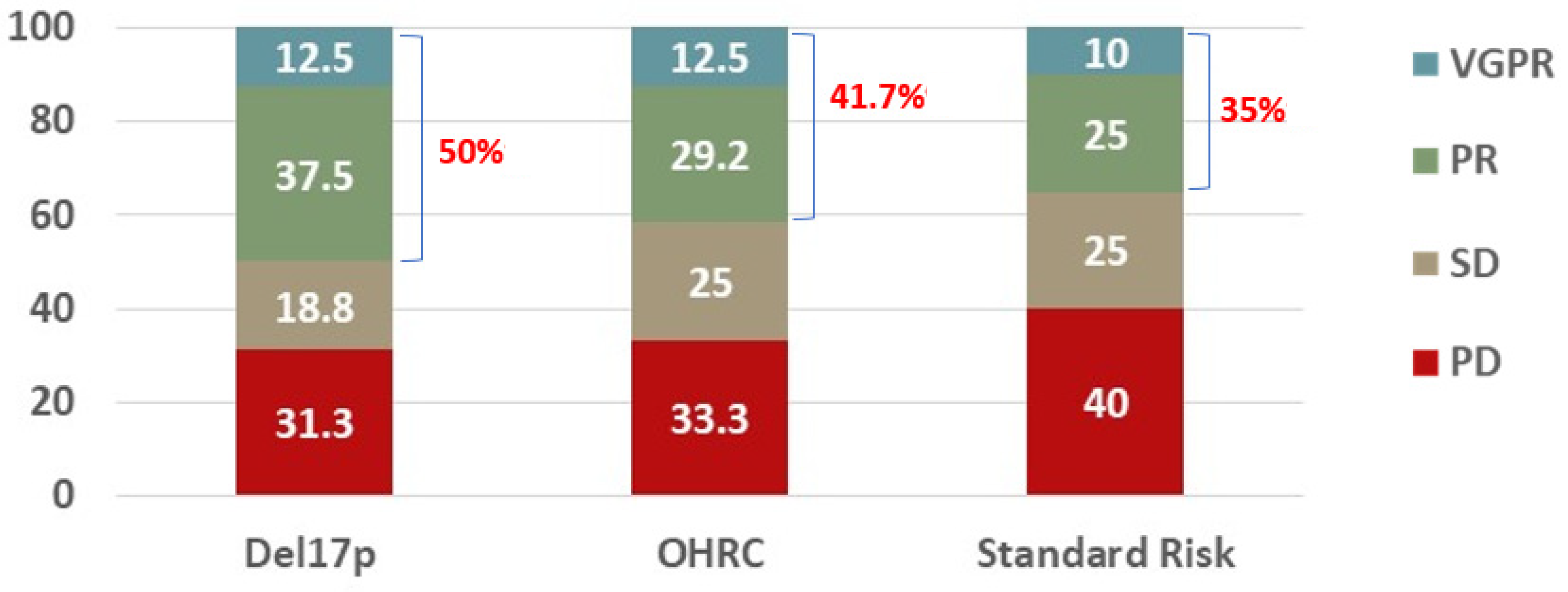

| del17p (N = 16) | OHRC (N = 24) | Standard Risk (N = 20) | ||||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Gender | ||||||
| Male | 10 | 62.5 | 15 | 62.5 | 13 | 65.0 |
| Female | 6 | 37.5 | 9 | 37.5 | 7 | 35.0 |
| Race | ||||||
| White | 13 | 81.3 | 14 | 58.3 | 16 | 80.0 |
| Black | 3 | 18.8 | 10 | 41.7 | 4 | 20.0 |
| Median Age in Years (Range) | 62.5 | 46–81 | 69 | 44–83 | 65.5 | 51–90 |
| High Risk Features | ||||||
| del17p | 16 | 100 | 0 | 0.0 | 0 | 0.0 |
| t(4;14) | 3 | 18.8 | 5 | 20.8 | 0 | 0.0 |
| t(14;16) | 0 | 0.0 | 3 | 12.5 | 0 | 0.0 |
| t(14;20) | 0 | 0.0 | 1 | 4.2 | 0 | 0.0 |
| Gain or Amp 1q21 | 10 | 62.5 | 22 | 91.7 | 0 | 0.0 |
| ISS at diagnosis | ||||||
| I | 1 | 6.3 | 2 | 8.3 | 4 | 20 |
| II | 6 | 37.5 | 10 | 41.7 | 8 | 40 |
| III | 6 | 37.5 | 11 | 45.8 | 2 | 10 |
| Unknown | 3 | 18.6 | 1 | 4.2 | 6 | 30 |
| R-ISS at diagnosis | ||||||
| I | 1 | 6.3 | 1 | 4.2 | 3 | 15 |
| II | 7 | 43.8 | 12 | 50.0 | 9 | 45 |
| III | 5 | 31.3 | 7 | 29.2 | 0 | 0 |
| Unknown | 3 | 18.8 | 4 | 16.7 | 8 | 40 |
| Median No. of prior lines of therapy (range) | 4 | 3–11 | 5 | 3–12 | 5 | 3–16 |
| Prior Transplant | 11 | 68.8 | 15 | 62.5 | 14 | 70.0 |
| Prior Lenalidomide | 15 | 93.8 | 22 | 91.7 | 20 | 100.0 |
| Refractory to Lenalidomide | 15 | 100.0 | 18 | 81.8 | 19 | 95.0 |
| Prior Bortezomib | 16 | 100.0 | 24 | 100.0 | 20 | 100.0 |
| Refractory to Bortezomib | 16 | 100.0 | 21 | 87.5 | 15 | 75.0 |
| Prior Carfilzomib | 15 | 93.8 | 21 | 87.5 | 18 | 90.0 |
| Refractory to Carfilzomib | 14 | 93.3 | 19 | 90.5 | 16 | 88.9 |
| Prior Pomalidomide | 16 | 100.0 | 21 | 87.5 | 19 | 95.0 |
| Refractory to Pomalidomide | 16 | 100.0 | 20 | 95.2 | 19 | 100.0 |
| Prior anti-CD38 antibody | 16 | 100.0 | 24 | 100.0 | 19 | 95.0 |
| Refractory to anti-CD38 antibody | 16 | 100.0 | 23 | 95.8 | 18 | 94.7 |
| Time since MM diagnosis, years | ||||||
| Median (Range) | 5.6 | 0.7–25.2 | 4.1 | 0.8–11.5 | 4.8 | 1.0–11.2 |
| Follow up, months | ||||||
| Median (Range) | 10.5 | 2.5–33.0 | 8.4 | 0.6–49.1 | 10.3 | 0.7–54.5 |
| del17p (N = 16) | OHRC (N = 24) | Standard Risk (N = 20) | ||||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Dara-Seli | 0 | 0.0 | 0 | 0.0 | 1 | 5.0 |
| Dara-Seli-dex | 2 | 12.5 | 6 | 25.0 | 2 | 10.0 |
| Seli-Kd | 5 | 31.3 | 7 | 29.2 | 10 | 50.0 |
| Seli-dex | 1 | 6.3 | 5 | 20.8 | 2 | 10.0 |
| Seli-pom | 0 | 0.0 | 1 | 4.2 | 0 | 0.0 |
| Seli-Pd | 5 | 31.3 | 2 | 8.3 | 3 | 15.0 |
| Seli-Vd | 3 | 18.8 | 3 | 12.5 | 2 | 10.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ehsan, H.; Robinson, M.; Voorhees, P.M.; Cassetta, K.; Borden, S.; Atrash, S.; Bhutani, M.; Varga, C.; Pineda-Roman, M.; Friend, R.; et al. Efficacy of Selinexor in Relapsed/Refractory Multiple Myeloma (RRMM) Patients with del17p and Other High-Risk Abnormalities (A Retrospective Single-Center Study). Life 2024, 14, 384. https://doi.org/10.3390/life14030384
Ehsan H, Robinson M, Voorhees PM, Cassetta K, Borden S, Atrash S, Bhutani M, Varga C, Pineda-Roman M, Friend R, et al. Efficacy of Selinexor in Relapsed/Refractory Multiple Myeloma (RRMM) Patients with del17p and Other High-Risk Abnormalities (A Retrospective Single-Center Study). Life. 2024; 14(3):384. https://doi.org/10.3390/life14030384
Chicago/Turabian StyleEhsan, Hamid, Myra Robinson, Peter M. Voorhees, Kristen Cassetta, Shanice Borden, Shebli Atrash, Manisha Bhutani, Cindy Varga, Mauricio Pineda-Roman, Reed Friend, and et al. 2024. "Efficacy of Selinexor in Relapsed/Refractory Multiple Myeloma (RRMM) Patients with del17p and Other High-Risk Abnormalities (A Retrospective Single-Center Study)" Life 14, no. 3: 384. https://doi.org/10.3390/life14030384







