Neurotoxicity of Some Environmental Pollutants to Zebrafish
Abstract
:1. Introduction
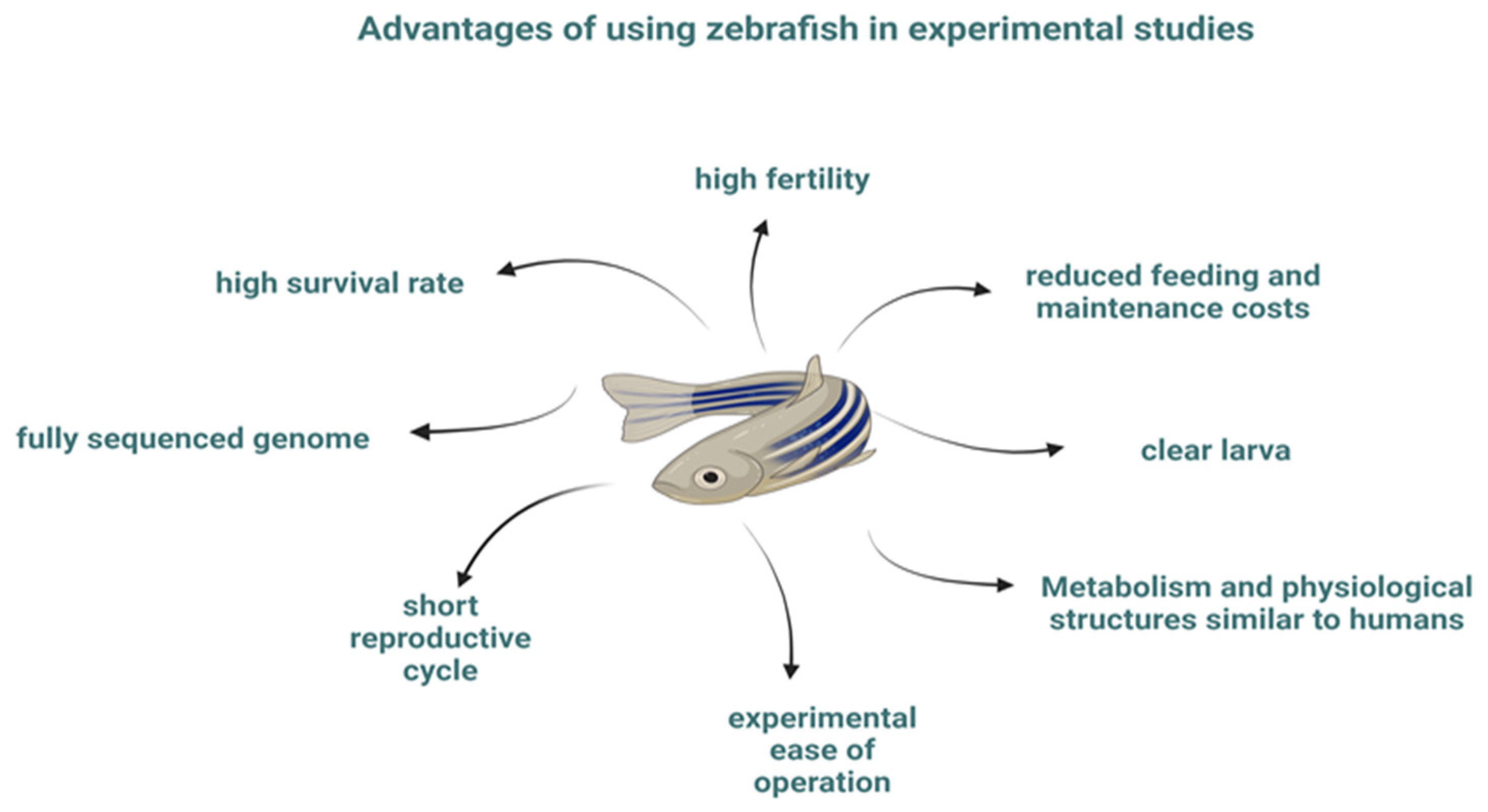
2. Nervous System of Zebrafish
3. Effects of Different-Sized Plastics on Zebrafish
3.1. Effects of Exposure to Nanoplastics in Zebrafish
3.2. Effects of Exposure to Microplastics in Zebrafish
- Ingestion and accumulation: Zebrafish, like many aquatic organisms, can ingest microplastics and nanoplastics either directly or indirectly through the food chain. Once ingested, these particles can accumulate in various tissues, including the brain and nervous system.
- Physical damage: Microplastics and nanoplastics can cause physical damage to the nervous system of zebrafish. These particles can disrupt neuronal connections, interfere with synaptic transmission, and induce inflammation in brain tissues. Accumulation of plastic particles in neuronal tissues can lead to structural abnormalities and impaired neuronal function.
- Leaching of chemicals: Nanoplastics and microplastics can release adsorbed chemical additives and pollutants into the environment, including the water column and sediments. These chemicals include neurotoxic substances such as plasticizers, flame retardants, and persistent organic pollutants (POPs). Once released, these neurotoxic chemicals can be absorbed by zebrafish and affect the function of their nervous system.
- Oxidative stress and neuroinflammation: Exposure to microplastics and nanoplastics can induce oxidative stress and neuroinflammation in zebrafish. The presence of plastic particles in neuronal tissues can trigger the production of ROS and inflammatory mediators, leading to cell damage and dysfunction within the nervous system. Oxidative stress and neuroinflammation can disrupt neuronal signaling pathways and contribute to neurobehavioral abnormalities in zebrafish.
- Behavioral and cognitive effects: Neurotoxic effects induced by microplastics and nanoplastics can manifest as altered behavior and cognitive function in zebrafish. Studies have shown that exposure to plastic particles can affect locomotor activity, learning and memory, social behavior, and predator avoidance responses in zebrafish. These behavioral changes can result from direct neurotoxicity or the secondary effects of neuronal damage and dysfunction caused by plastic exposure.
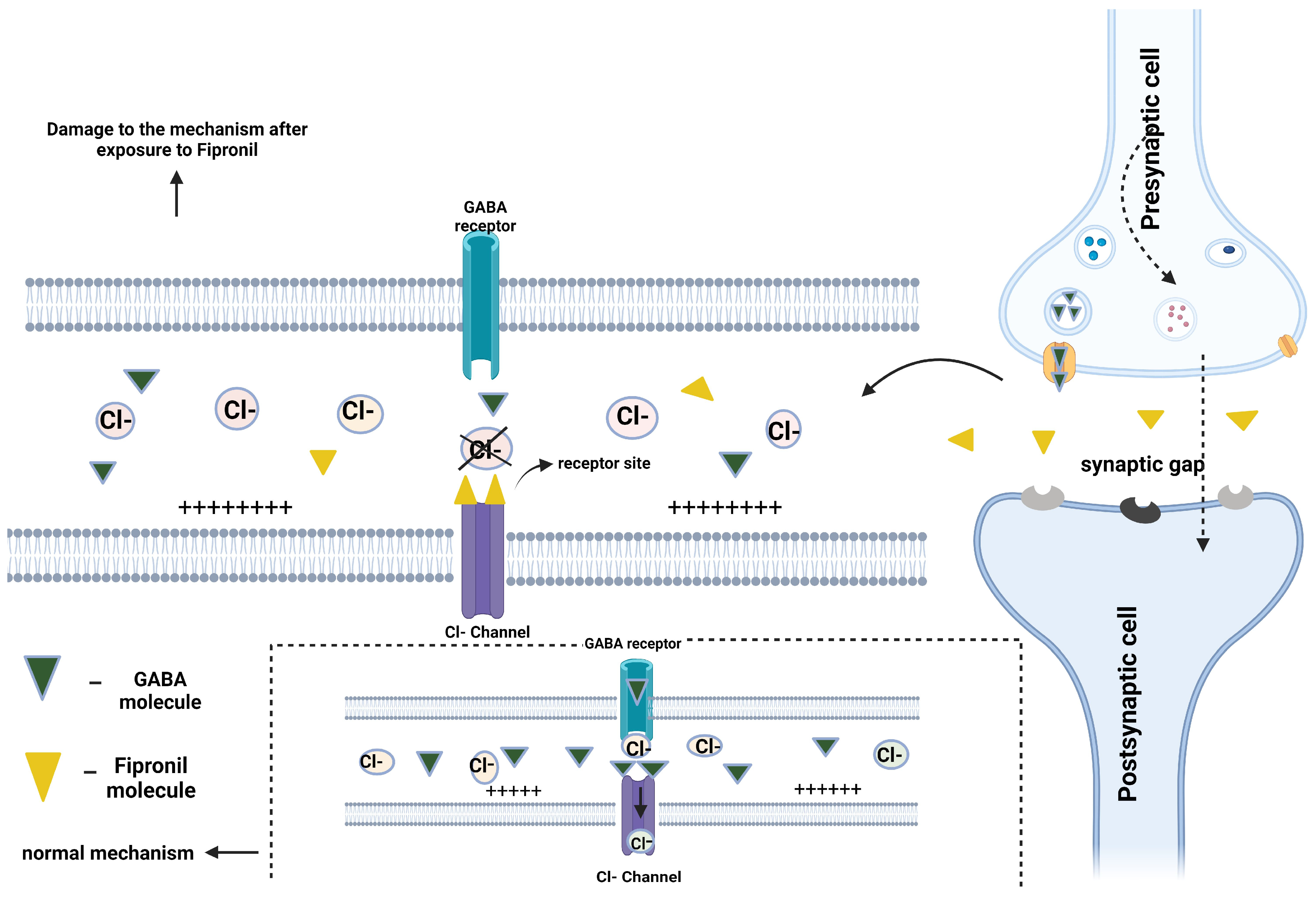
4. Neurotoxicity of Fipronil to Zebrafish
- Increased SOD levels.
- Increased lipid peroxidation (GPx) levels.
- Increased malondialdehyde (MDA) levels.
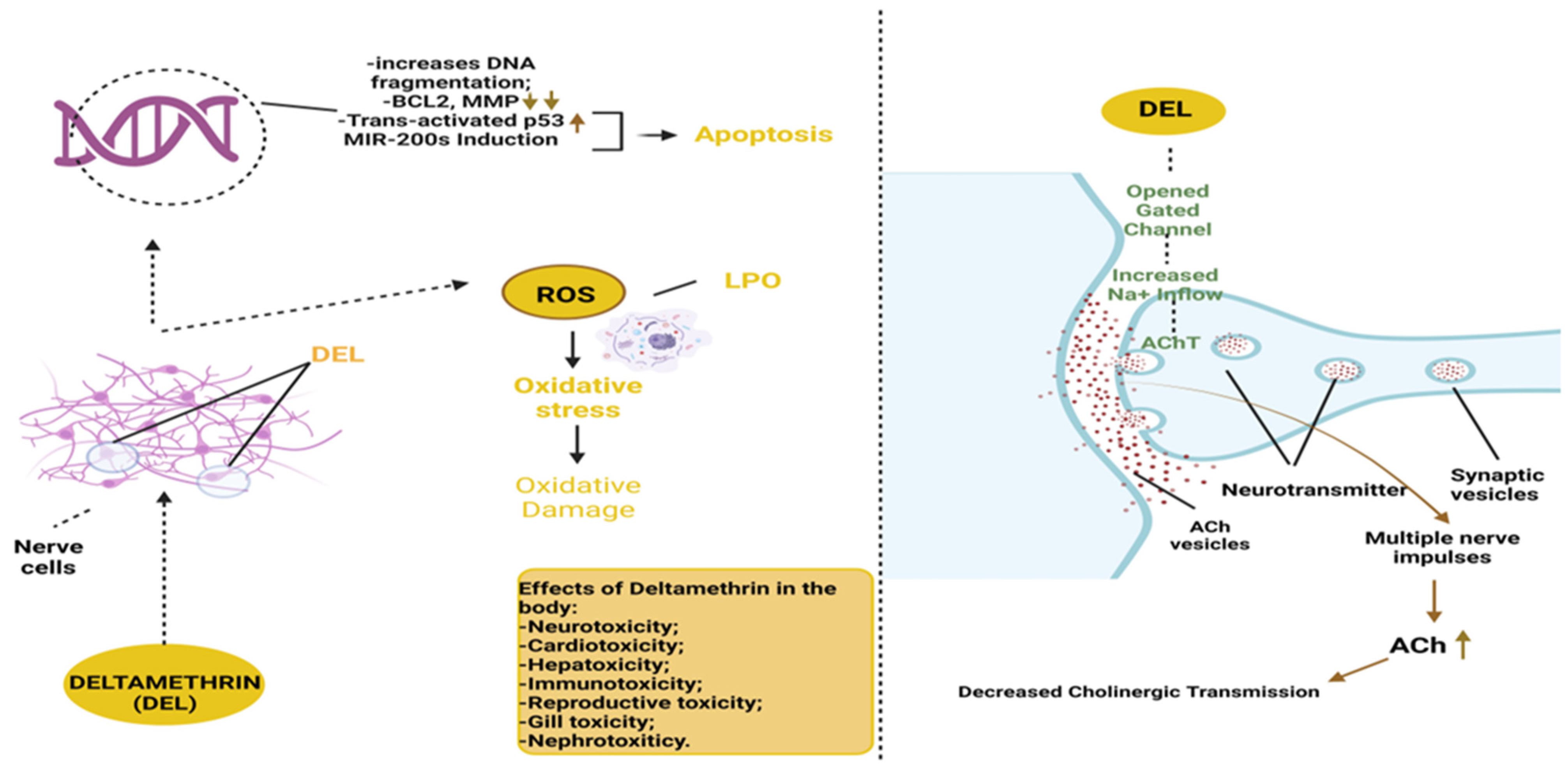
5. Influence of Deltamethrin on Oxidative Stress Parameters and Behavioral Variables in Zebrafish
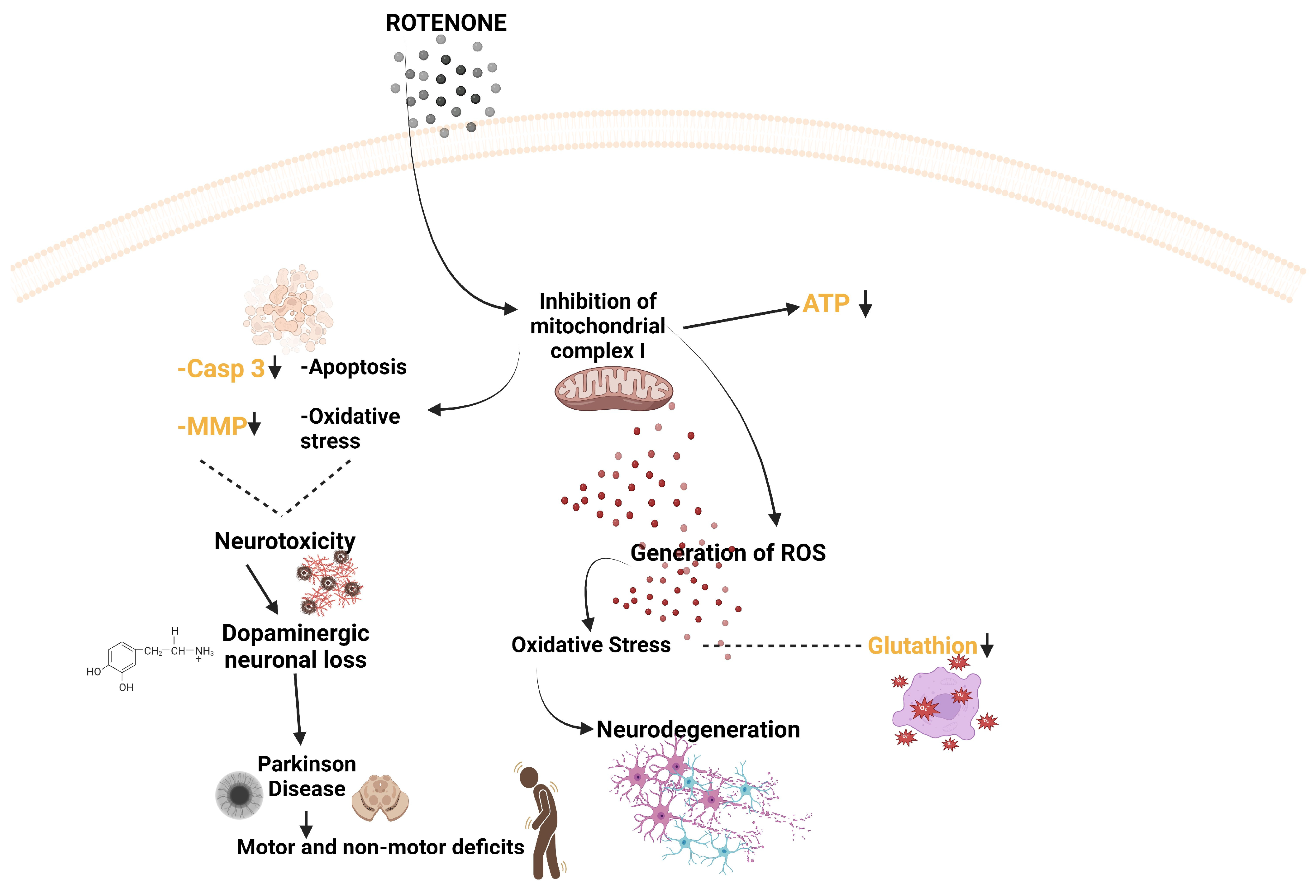
6. Evaluation of Neurotoxicity after Exposure of Zebrafish to Different Doses of Rotenone
7. Conclusions
- Co-occurrence and sorption: Microplastics and nanoplastics can serve as carriers of or sorbents for pesticides in aquatic environments. Pesticides can adsorb onto the surface of plastic particles, leading to their accumulation and persistence in the water column, sediments, and biota. This co-occurrence increases the exposure of aquatic organisms, including zebrafish, to both plastic pollution and pesticide contamination.
- Bioaccumulation and trophic transfer: Microplastics, nanoplastics, and pesticides can bioaccumulate and trophically transfer in aquatic food webs. Zebrafish can ingest plastic particles and pesticides directly or indirectly through their diet, leading to the accumulation of these contaminants in their tissues over time. Bioaccumulation and trophic transfer can amplify the concentrations of microplastics, nanoplastics, and pesticides at higher trophic levels, including in zebrafish predators, further exacerbating their ecological impact.
- Synergistic effects and toxicity: Microplastics, nanoplastics, and pesticides can have synergistic or additive effects on zebrafish and other aquatic organisms. Combined exposure to plastic particles and pesticides can increase the toxicity of individual contaminants, leading to greater adverse effects on the health and physiology of zebrafish. Synergistic effects can arise from interactions between plastic-induced stress responses, such as inflammation and oxidative stress, and pesticide-induced neurotoxicity, developmental toxicity, or endocrine disruption in zebrafish.
Author Contributions
Funding
Conflicts of Interest
References
- Fang, F. Phylogenetic Analysis of the Asian Cyprinid Genus Danio (Teleostei, Cyprinidae). Copeia 2003, 2003, 714–728. [Google Scholar] [CrossRef]
- Horzmann, K.A.; Freeman, J.L. Making Waves: New Developments in Toxicology with the Zebrafish. Toxicol. Sci. 2018, 163, 5–12. [Google Scholar] [CrossRef]
- Bauer, B.; Mally, A.; Liedtke, D. Zebrafish Embryos and Larvae as Alternative Animal Models for Toxicity Testing. Int. J. Mol. Sci. 2021, 22, 13417. [Google Scholar] [CrossRef]
- Batel, A.; Borchert, F.; Reinwald, H.; Erdinger, L.; Braunbeck, T. Microplastic Accumulation Patterns and Transfer of Benzo[a]Pyrene to Adult Zebrafish (Danio rerio) Gills and Zebrafish Embryos. Environ. Pollut. 2018, 235, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.A.; Trevisan, R.; Massarsky, A.; Kozal, J.S.; Levin, E.D.; Di Giulio, R.T. Maternal Transfer of Nanoplastics to Offspring in Zebrafish (Danio rerio): A Case Study with Nanopolystyrene. Sci. Total Environ. 2018, 643, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Brugman, S. The Zebrafish as a Model to Study Intestinal Inflammation. Dev. Comp. Immunol. 2016, 64, 82–92. [Google Scholar] [CrossRef]
- Gao, Y.; Jin, Q.; Gao, C.; Chen, Y.; Sun, Z.; Guo, G.; Peng, J. Unraveling Differential Transcriptomes and Cell Types in Zebrafish Larvae Intestine and Liver. Cells 2022, 11, 3290. [Google Scholar] [CrossRef] [PubMed]
- Lei, P.; Zhang, W.; Ma, J.; Xia, Y.; Yu, H.; Du, J.; Fang, Y.; Wang, L.; Zhang, K.; Jin, L.; et al. Advances in the Utilization of Zebrafish for Assessing and Understanding the Mechanisms of Nano-/Microparticles Toxicity in Water. Toxics 2023, 11, 380. [Google Scholar] [CrossRef]
- Grinblat, Y.; Gamse, J.; Patel, M.; Sive, H. Determination of the Zebrafish Forebrain: Induction and Patterning. Development 1998, 125, 4403–4416. [Google Scholar] [CrossRef]
- Hauptmann, G.; Söll, I.; Gerster, T. The Early Embryonic Zebrafish Forebrain Is Subdivided into Molecularly Distinct Transverse and Longitudinal Domains. Brain Res. Bull. 2002, 57, 371–375. [Google Scholar] [CrossRef]
- Kaslin, J.; Ganz, J. Chapter 18—Zebrafish Nervous Systems. In The Zebrafish in Biomedical Research; Cartner, S.C., Eisen, J.S., Farmer, S.C., Guillemin, K.J., Kent, M.L., Sanders, G.E., Eds.; American College of Laboratory Animal Medicine; Academic Press: Cambridge, MA, USA, 2020; pp. 181–189. [Google Scholar] [CrossRef]
- Orger, M.B.; de Polavieja, G.G. Zebrafish Behavior: Opportunities and Challenges. Annu. Rev. Neurosci. 2017, 40, 125–147. [Google Scholar] [CrossRef] [PubMed]
- Northcutt, R.G. The Forebrain of Gnathostomes: In Search of a Morphotype. Brain Behav. Evol. 1995, 46, 275–318. [Google Scholar] [CrossRef] [PubMed]
- Ganz, J.; Kaslin, J.; Hochmann, S.; Freudenreich, D.; Brand, M. Heterogeneity and Fgf Dependence of Adult Neural Progenitors in the Zebrafish Telencephalon. Glia 2010, 58, 1345–1363. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, L.A.; Hofmann, H.A. The Vertebrate Mesolimbic Reward System and Social Behavior Network: A Comparative Synthesis. J. Comp. Neurol. 2011, 519, 3599–3639. [Google Scholar] [CrossRef] [PubMed]
- Diotel, N.; Lübke, L.; Strähle, U.; Rastegar, S. Common and Distinct Features of Adult Neurogenesis and Regeneration in the Telencephalon of Zebrafish and Mammals. Front. Neurosci. 2020, 14, 568930. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, H.; Bianco, I.H.; Hamaoka, T.; Miyashita, T.; Uemura, O.; Concha, M.L.; Russell, C.; Wilson, S.W.; Okamoto, H. Laterotopic Representation of Left-Right Information onto the Dorso-Ventral Axis of a Zebrafish Midbrain Target Nucleus. Curr. Biol. 2005, 15, 238–243. [Google Scholar] [CrossRef]
- Gamse, J.T.; Kuan, Y.-S.; Macurak, M.; Brösamle, C.; Thisse, B.; Thisse, C.; Halpern, M.E. Directional Asymmetry of the Zebrafish Epithalamus Guides Dorsoventral Innervation of the Midbrain Target. Development 2005, 132, 4869–4881. [Google Scholar] [CrossRef]
- Amo, R.; Aizawa, H.; Takahoko, M.; Kobayashi, M.; Takahashi, R.; Aoki, T.; Okamoto, H. Identification of the Zebrafish Ventral Habenula As a Homolog of the Mammalian Lateral Habenula. J. Neurosci. 2010, 30, 1566–1574. [Google Scholar] [CrossRef]
- Mueller, T. What Is the Thalamus in Zebrafish? Front. Neurosci. 2012, 6, 64. [Google Scholar] [CrossRef]
- Gutnick, A.; Levkowitz, G. The Neurohypophysis: Fishing for New Insights. J. Neuroendocr. 2012, 24, 973–974. [Google Scholar] [CrossRef]
- Xie, Y.; Dorsky, R.I. Development of the Hypothalamus: Conservation, Modification and Innovation. Development 2017, 144, 1588–1599. [Google Scholar] [CrossRef] [PubMed]
- Menke, A.L.; Spitsbergen, J.M.; Wolterbeek, A.P.M.; Woutersen, R.A. Normal Anatomy and Histology of the Adult Zebrafish. Toxicol. Pathol. 2011, 39, 759–775. [Google Scholar] [CrossRef] [PubMed]
- Marachlian, E.; Avitan, L.; Goodhill, G.J.; Sumbre, G. Principles of Functional Circuit Connectivity: Insights From Spontaneous Activity in the Zebrafish Optic Tectum. Front. Neural Circuits 2018, 12, 46. [Google Scholar] [CrossRef] [PubMed]
- Kaslin, J.; Kroehne, V.; Benato, F.; Argenton, F.; Brand, M. Development and Specification of Cerebellar Stem and Progenitor Cells in Zebrafish: From Embryo to Adult. Neural Dev. 2013, 8, 9. [Google Scholar] [CrossRef]
- Hibi, M.; Shimizu, T. Development of the Cerebellum and Cerebellar Neural Circuits. Dev. Neurobiol. 2012, 72, 282–301. [Google Scholar] [CrossRef] [PubMed]
- Teng, L.; Labosky, P.A. The Contribution of the Neural Crest to the Vertebrate Body—Madame Curie Bioscience Database—NCBI Bookshelf. Available online: https://www.ncbi.nlm.nih.gov/books/NBK6098/ (accessed on 7 April 2024).
- Djenoune, L.; Wyart, C. Light on a Sensory Interface Linking the Cerebrospinal Fluid to Motor Circuits in Vertebrates. J. Neurogenet. 2017, 31, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Berg, E.M.; Björnfors, E.R.; Pallucchi, I.; Picton, L.D.; El Manira, A. Principles Governing Locomotion in Vertebrates: Lessons From Zebrafish. Front. Neural Circuits 2018, 12, 73. [Google Scholar] [CrossRef] [PubMed]
- Adolf, B.; Chapouton, P.; Lam, C.S.; Topp, S.; Tannhäuser, B.; Strähle, U.; Götz, M.; Bally-Cuif, L. Conserved and Acquired Features of Adult Neurogenesis in the Zebrafish Telencephalon. Dev. Biol. 2006, 295, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, B.W.; Douek, A.M.; Loosli, F.; Kaslin, J. A Whole Brain Staining, Embedding, and Clearing Pipeline for Adult Zebrafish to Visualize Cell Proliferation and Morphology in 3-Dimensions. Front. Neurosci. 2017, 11, 750. [Google Scholar] [CrossRef]
- Li, Y.; Chen, T.; Miao, X.; Yi, X.; Wang, X.; Zhao, H.; Lee, S.M.-Y.; Zheng, Y. Zebrafish: A promising in vivo model for assessing the delivery of natural products, fluorescence dyes and drugs across the blood-brain barrier. Pharmacol. Res. 2017, 125, 246–257. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Stewart, A.M.; Gerlai, R. Zebrafish as an Emerging Model for Studying Complex Brain Disorders. Trends Pharmacol. Sci. 2014, 35, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Leng, Z.; Padhan, R.K.; Sreeram, A. Production of a Sustainable Paving Material through Chemical Recycling of Waste PET into Crumb Rubber Modified Asphalt. J. Clean. Prod. 2018, 180, 682–688. [Google Scholar] [CrossRef]
- Abdurahman, A.; Cui, K.; Wu, J.; Li, S.; Gao, R.; Dai, J.; Liang, W.; Zeng, F. Adsorption of Dissolved Organic Matter (DOM) on Polystyrene Microplastics in Aquatic Environments: Kinetic, Isotherm and Site Energy Distribution Analysis. Ecotoxicol. Environ. Saf. 2020, 198, 110658. [Google Scholar] [CrossRef]
- Zhu, X.; Wang, C.; Duan, X.; Liang, B.; Genbo Xu, E.; Huang, Z. Micro- and Nanoplastics: A New Cardiovascular Risk Factor? Environ. Int. 2023, 171, 107662. [Google Scholar] [CrossRef] [PubMed]
- Environments. A Review about Microalgae Wastewater Treatment for Bioremediation and Biomass Production—A New Challenge for Europe. Available online: https://www.mdpi.com/2076-3298/8/12/136 (accessed on 8 April 2024).
- Catone, C.M.; Ripa, M.; Geremia, E.; Ulgiati, S. Bio-Products from Algae-Based Biorefinery on Wastewater: A Review. J. Environ. Manag. 2021, 293, 112792. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Besseling, E.; Shim, W.J. Nanoplastics in the Aquatic Environment. In Marine Anthropogene Litter; Springer: Berlin/Heidelberg, Germany, 2015; pp. 325–340. [Google Scholar] [CrossRef]
- Lacerda, A.L.D.F.; Rodrigues, L.D.S.; van Sebille, E.; Rodrigues, F.L.; Ribeiro, L.; Secchi, E.R.; Kessler, F.; Proietti, M.C. Plastics in Sea Surface Waters around the Antarctic Peninsula. Sci. Rep. 2019, 9, 3977. [Google Scholar] [CrossRef] [PubMed]
- Alfaro-Núñez, A.; Astorga, D.; Cáceres-Farías, L.; Bastidas, L.; Soto Villegas, C.; Macay, K.; Christensen, J.H. Microplastic Pollution in Seawater and Marine Organisms across the Tropical Eastern Pacific and Galápagos. Sci. Rep. 2021, 11, 6424. [Google Scholar] [CrossRef]
- Hou, R.; Xu, Y.; Wang, Z. Review of OPFRs in Animals and Humans: Absorption, Bioaccumulation, Metabolism, and Internal Exposure Research. Chemosphere 2016, 153, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Kovalakova, P.; Cizmas, L.; McDonald, T.J.; Marsalek, B.; Feng, M.; Sharma, V.K. Occurrence and Toxicity of Antibiotics in the Aquatic Environment: A Review. Chemosphere 2020, 251, 126351. [Google Scholar] [CrossRef]
- Elizalde-Velázquez, G.A.; Gómez-Oliván, L.M. Microplastics in Aquatic Environments: A Review on Occurrence, Distribution, Toxic Effects, and Implications for Human Health. Sci. Total Environ. 2021, 780, 146551. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Hemavathy, R.V.; Jeevanantham, S.; Harikumar, P.; Priyanka, G.; Devakirubai, D.R.A. A Comprehensive Review on Sources, Analysis and Toxicity of Environmental Pollutants and Its Removal Methods from Water Environment. Sci. Total Environ. 2022, 812, 152456. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Sosa-Hernández, J.E.; Raza, A.; Nabeel, F.; Iqbal, H.M.N. Biosorption: An Interplay between Marine Algae and Potentially Toxic Elements—A Review. Mar. Drugs 2018, 16, 65. [Google Scholar] [CrossRef]
- Kang, H.-M.; Byeon, E.; Jeong, H.; Kim, M.-S.; Chen, Q.; Lee, J.-S. Different Effects of Nano- and Microplastics on Oxidative Status and Gut Microbiota in the Marine Medaka Oryzias Melastigma. J. Hazard. Mater. 2021, 405, 124207. [Google Scholar] [CrossRef] [PubMed]
- Vethaak, A.D.; Legler, J. Microplastics and Human Health. Science 2021, 371, 672–674. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Yu, H.; Yi, J.; Lei, P.; He, J.; Ruan, J.; Xu, P.; Tao, R.; Jin, L.; Wu, W.; et al. Behavioral Studies of Zebrafish Reveal a New Perspective on the Reproductive Toxicity of Micro- and Nanoplastics. Toxics 2024, 12, 178. [Google Scholar] [CrossRef]
- Chen, Q.; Yin, D.; Jia, Y.; Schiwy, S.; Legradi, J.; Yang, S.; Hollert, H. Enhanced Uptake of BPA in the Presence of Nanoplastics Can Lead to Neurotoxic Effects in Adult Zebrafish. Sci. Total Environ. 2017, 609, 1312–1321. [Google Scholar] [CrossRef] [PubMed]
- Gigault, J.; Halle, A.T.; Baudrimont, M.; Pascal, P.-Y.; Gauffre, F.; Phi, T.-L.; El Hadri, H.; Grassl, B.; Reynaud, S. Current Opinion: What Is A Nanoplastic? Environ. Pollut. 2018, 235, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Cho, H.-J.; Kim, E.; Huh, Y.H.; Kim, H.-J.; Kim, B.; Kang, T.; Lee, J.-S.; Jeong, J. Correction: Bioaccumulation of Polystyrene Nanoplastics and Their Effect on the Toxicity of Au Ions in Zebrafish Embryos. Nanoscale 2019, 11, 3396. [Google Scholar] [CrossRef] [PubMed]
- Kahlon, S.K.; Sharma, G.; Julka, J.M.; Kumar, A.; Sharma, S.; Stadler, F.J. Impact of Heavy Metals and Nanoparticles on Aquatic Biota. Environ. Chem. Lett. 2018, 3, 919–946. [Google Scholar] [CrossRef]
- Turan, N.B.; Erkan, H.S.; Engin, G.O.; Bilgili, M.S. Nanoparticles in the Aquatic Environment: Usage, Properties, Transformation and Toxicity—A Review. Process Saf. Environ. Prot. 2019, 130, 238–249. [Google Scholar] [CrossRef]
- Nowack, B.; Bucheli, T.D. Occurrence, Behavior and Effects of Nanoparticles in the Environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, K.; Jocic, S.; Doverbratt, I.; Hansson, L.A. Nanoplastics in the Aquatic Environment. In Microplastic Contamination in Aquatic Environments; Elsevier: Amsterdam, The Netherlands, 2018; pp. 379–399. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the Marine Environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Sharma, N. Mechanistic Implications of Plastic Degradation. Polym. Degrad. Stab. 2008, 93, 561–584. [Google Scholar] [CrossRef]
- Zettler, E.R.; Mincer, T.J.; Amaral-Zettler, L.A. Life in the “Plastisphere”: Microbial Communities on Plastic Marine Debris. Environ. Sci. Technol. 2013, 47, 7137–7146. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, C.; Von Goetz, N.; Scheringer, M.; Wormuth, M.; Hungerbühler, K. Potential Exposure of German Consumers to Engineered Nanoparticles in Cosmetics and Personal Care Products. Nanotoxicology 2011, 5, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Chatterjee, S. Microplastic Pollution, a Threat to Marine Ecosystem and Human Health: A Short Review. Environ. Sci. Pollut. Res. Int. 2017, 24, 21530–21547. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kuo, Y.-Y.; Gerecke, A.C.; Wang, J. Co-Release of Hexabromocyclododecane (HBCD) and Nano- and Microparticles from Thermal Cutting of Polystyrene Foams. Environ. Sci. Technol. 2012, 46, 10990–10996. [Google Scholar] [CrossRef] [PubMed]
- Stephens, B.; Azimi, P.; Orch, Z.; Ramos, T. Ultrafine Particle Emissions from Desktop 3D Printers. Atmos. Environ. 2013, 79, 334–339. [Google Scholar] [CrossRef]
- Karlsson, H.L.; Gustafsson, J.; Cronholm, P.; Möller, L. Size-Dependent Toxicity of Metal Oxide Particles--a Comparison between Nano- and Micrometer Size. Toxicol. Lett. 2009, 188, 112–118. [Google Scholar] [CrossRef]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Bottero, J.-Y.; Lowry, G.V.; Jolivet, J.-P.; Wiesner, M.R. Towards a Definition of Inorganic Nanoparticles from an Environmental, Health and Safety Perspective. Nat. Nanotechnol. 2009, 4, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Strungaru, S.-A.; Jijie, R.; Nicoara, M.; Plavan, G.; Faggio, C. Micro- (Nano) Plastics in Freshwater Ecosystems: Abundance, Toxicological Impact and Quantification Methodology. TrAC Trends Anal. Chem. 2019, 110, 116–128. [Google Scholar] [CrossRef]
- Trevisan, R.; Voy, C.; Chen, S.; Di Giulio, R.T. Nanoplastics Decrease the Toxicity of a Complex PAH Mixture but Impair Mitochondrial Energy Production in Developing Zebrafish. Environ. Sci. Technol. 2019, 53, 8405–8415. [Google Scholar] [CrossRef] [PubMed]
- Barreto, Â.; Santos, J.; Amorim, M.; Maria, V. Polystyrene Nanoplastics Can Alter the Toxicological Effects of Simvastatin on Danio rerio. Toxics 2021, 9, 44. [Google Scholar] [CrossRef] [PubMed]
- Lomonaco, T.; Manco, E.; Corti, A.; La Nasa, J.; Ghimenti, S.; Biagini, D.; Di Francesco, F.; Modugno, F.; Ceccarini, A.; Fuoco, R.; et al. Release of Harmful Volatile Organic Compounds (VOCs) from Photo-Degraded Plastic Debris: A Neglected Source of Environmental Pollution. J. Hazard. Mater. 2020, 394, 122596. [Google Scholar] [CrossRef] [PubMed]
- Brandts, I.; Teles, M.; Gonçalves, A.P.; Barreto, A.; Franco-Martinez, L.; Tvarijonaviciute, A.; Martins, M.A.; Soares, A.M.V.M.; Tort, L.; Oliveira, M. Effects of Nanoplastics on Mytilus Galloprovincialis after Individual and Combined Exposure with Carbamazepine. Sci. Total Environ. 2018, 643, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Parenti, C.C.; Ghilardi, A.; Della Torre, C.; Magni, S.; Del Giacco, L.; Binelli, A. Evaluation of the Infiltration of Polystyrene Nanobeads in Zebrafish Embryo Tissues after Short-Term Exposure and the Related Biochemical and Behavioural Effects. Environ. Pollut. 2019, 254 Pt A, 112947. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Goodhead, R.; Moger, J.; Galloway, T.S. Microplastic Ingestion by Zooplankton. Environ. Sci. Technol. 2013, 47, 6646–6655. [Google Scholar] [CrossRef]
- Mazurais, D.; Ernande, B.; Quazuguel, P.; Severe, A.; Huelvan, C.; Madec, L.; Mouchel, O.; Soudant, P.; Robbens, J.; Huvet, A.; et al. Evaluation of the impact of polyethylene microbeads ingestion in European sea bass (Dicentrarchus labrax) larvae. Mar. Environ. Res. 2015, 112, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Karami, A.; Groman, D.B.; Wilson, S.P.; Ismail, P.; Neela, V.K. Biomarker Responses in Zebrafish (Danio rerio) Larvae Exposed to Pristine Low-Density Polyethylene Fragments. Environ. Pollut. 2017, 223, 466–475. [Google Scholar] [CrossRef]
- Santos, D.; Félix, L.; Luzio, A.; Parra, S.; Bellas, J.; Monteiro, S.M. Single and Combined Acute and Subchronic Toxic Effects of Microplastics and Copper in Zebrafish (Danio rerio) Early Life Stages. Chemosphere 2021, 277, 130262. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Li, Y.; Gao, J.; Cao, R.; Shang, E.; Zhang, W. ROS-Mediated Photoaging Pathways of Nano- and Micro-Plastic Particles under UV Irradiation. Water Res. 2022, 216, 118320. [Google Scholar] [CrossRef] [PubMed]
- Van Pomeren, M.; Brun, N.R.; Peijnenburg, W.J.G.M.; Vijver, M.G. Exploring Uptake and Biodistribution of Polystyrene (Nano)Particles in Zebrafish Embryos at Different Developmental Stages. Aquat. Toxicol. 2017, 190, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Sökmen, T.Ö.; Sulukan, E.; Türkoğlu, M.; Baran, A.; Özkaraca, M.; Ceyhun, S.B. Polystyrene Nanoplastics (20 Nm) Are Able to Bioaccumulate and Cause Oxidative DNA Damages in the Brain Tissue of Zebrafish Embryo (Danio rerio). Neurotoxicology 2020, 77, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Sarasamma, S.; Audira, G.; Siregar, P.; Malhotra, N.; Lai, Y.-H.; Liang, S.-T.; Chen, J.-R.; Chen, K.H.-C.; Hsiao, C.-D. Nanoplastics Cause Neurobehavioral Impairments, Reproductive and Oxidative Damages, and Biomarker Responses in Zebrafish: Throwing up Alarms of Wide Spread Health Risk of Exposure. Int. J. Mol. Sci. 2020, 21, 1410. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Zhang, Y.; Deng, Y.; Jiang, W.; Zhao, Y.; Geng, J.; Ding, L.; Ren, H. Uptake and Accumulation of Polystyrene Microplastics in Zebrafish (Danio rerio) and Toxic Effects in Liver. Environ. Sci. Technol. 2016, 50, 4054–4060. [Google Scholar] [CrossRef] [PubMed]
- Teng, L.; Zhu, Y.; Li, H.; Song, X.; Shi, L. The Phytotoxicity of Microplastics to the Photosynthetic Performance and Transcriptome Profiling of Nicotiana tabacum Seedlings. Ecotoxicol. Environ. Saf. 2022, 231, 113155. [Google Scholar] [CrossRef]
- Chen, L.; Yu, K.; Huang, C.; Yu, L.; Zhu, B.; Lam, P.K.S.; Lam, J.C.W.; Zhou, B. Prenatal Transfer of Polybrominated Diphenyl Ethers (PBDEs) Results in Developmental Neurotoxicity in Zebrafish Larvae. Environ. Sci. Technol. 2012, 46, 9727–9734. [Google Scholar] [CrossRef] [PubMed]
- Environments |Free Full-Text|. The Impact of Micro- and Nanoplastics on Aquatic Organisms: Mechanisms of Oxidative Stress and Implications for Human Health—A Review. Available online: https://www.mdpi.com/2076-3298/10/9/161 (accessed on 8 April 2024).
- Barboza, L.G.A.; Vieira, L.R.; Branco, V.; Carvalho, C.; Guilhermino, L. Microplastics Increase Mercury Bioconcentration in Gills and Bioaccumulation in the Liver, and Cause Oxidative Stress and Damage in Dicentrarchus labrax Juveniles. Sci. Rep. 2018, 8, 15655. [Google Scholar] [CrossRef]
- Savuca, A.; Curpan, A.; Hritcu, L.; Ciobîcă, A.; Plavan, G.; Nicoara, M. Preliminary Study on the Behavioral Response of Zebrafish to the Presence of Methionine and Polypropylene Residues in Water. Analele Universității Dunărea de Jos din Galați 2022, 45, 89–96. [Google Scholar] [CrossRef]
- Frias, J.P.G.L.; Nash, R. Microplastics: Finding a Consensus on the Definition. Mar. Pollut. Bull. 2019, 138, 145–147. [Google Scholar] [CrossRef]
- Cowger, W.; Gray, A.; Christiansen, S.H.; DeFrond, H.; Deshpande, A.D.; Hemabessiere, L.; Lee, E.; Mill, L.; Munno, K.; Ossmann, B.E.; et al. Critical Review of Processing and Classification Techniques for Images and Spectra in Microplastic. Res. Appl. Spectrosc. 2020, 74, 989–1010. [Google Scholar] [CrossRef]
- Burns, E.E.; Boxall, A.B.A. Microplastics in the Aquatic Environment: Evidence for or against Adverse Impacts and Major Knowledge Gaps. Environ. Toxicol. Chem. 2018, 37, 2776–2796. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef]
- Lassen, C.; Hansen, S.F.; Magnusson, K.; Hartmann, N.B.; Rehne Jensen, P.; Nielsen, T.G.; Brinch, A.; Microplastics: Occurrence, Effects and Sources of Releases to the Environment in Denmark. Danish Environmental Protection Agency. 2015. Available online: http://mst.dk/service/publikationer/publikationsarkiv/2015/nov/rapport-ommikroplast (accessed on 8 April 2024).
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.G.; McGonigle, D.; Russell, A.E. Lost at Sea: Where Is All the Plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Besseling, E.; Quik, J.T.K.; Sun, M.; Koelmans, A.A. Fate of Nano- and Microplastic in Freshwater Systems: A Modeling Study. Environ. Pollut. 2017, 220, 540–548. [Google Scholar] [CrossRef]
- Jeong, C.-B.; Won, E.-J.; Kang, H.-M.; Lee, M.-C.; Hwang, D.-S.; Hwang, U.-K.; Zhou, B.; Souissi, S.; Lee, S.-J.; Lee, J.-S. Microplastic Size-Dependent Toxicity, Oxidative Stress Induction, and p-JNK and p-P38 Activation in the Monogonont Rotifer (Brachionus koreanus). Environ. Sci. Technol. 2016, 50, 8849–8857. [Google Scholar] [CrossRef]
- Duis, K.; Coors, A. Microplastics in the aquatic and terrestrial environment: Sources (with a specific focus on personal care products), fate and effects. Environ. Sci. Eur. 2016, 28, 2. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, L.C.M.; van der Zwet, J.; Damsteeg, J.-W.; Slat, B.; Andrady, A.; Reisser, J. River Plastic Emissions to the World’s Oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef]
- Song, Y.K.; Hong, S.H.; Jang, M.; Han, G.M.; Jung, S.W.; Shim, W.J. Combined Effects of UV Exposure Duration and Mechanical Abrasion on Microplastic Fragmentation by Polymer Type. Environ. Sci. Technol. 2017, 51, 4368–4376. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Paul Chen, J. Microplastics in Freshwater Systems: A Review on Occurrence, Environmental Effects, and Methods for Microplastics Detection. Water Res. 2018, 137, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Pothiraj, C.; Gokul, T.A.; Kumar, K.; Ramasubramanian, A.; Palanichamy, A.; Venkatachalam, K.; Pastorino, P.; Barcelò, D.; Balaji, P.; Faggio, C. Vulnerability of Microplastics on Marine Environment: A Review. Ecol. Indic. 2023, 155, 111058. [Google Scholar] [CrossRef]
- Issac, M.N.; Kandasubramanian, B. Effect of Microplastics in Water and Aquatic Systems. Environ. Sci. Pollut. Res. Int. 2021, 28, 19544–19562. [Google Scholar] [CrossRef] [PubMed]
- Fadare, O.O.; Wan, B.; Guo, L.-H.; Zhao, L. Microplastics from Consumer Plastic Food Containers: Are We Consuming It? Chemosphere 2020, 253, 126787. [Google Scholar] [CrossRef] [PubMed]
- Wastewater Treatment Plant Effluent as a Source of Microplastics: Review of the Fate, Chemical Interactions and Potential Risks to Aquatic Organisms. Available online: https://pubmed.ncbi.nlm.nih.gov/27858783/ (accessed on 8 April 2024).
- Deng, J.; Guo, P.; Zhang, X.; Su, H.; Zhang, Y.; Wu, Y.; Li, Y. Microplastics and Accumulated Heavy Metals in Restored Mangrove Wetland Surface Sediments at Jinjiang Estuary (Fujian, China). Mar. Pollut. Bull. 2020, 159, 111482. [Google Scholar] [CrossRef] [PubMed]
- Muting, Y.; Huayue, N.; Kaihang, X.; Yuhui, H.; Yingtong, H.; Yumei, H.; Jun, W. Microplastic abundance, distribution and composition in the Pearl River along Guangzhou city and Pearl River estuary, China. Chemosphere 2019, 217, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Eo, S.; Hong, S.H.; Song, Y.K.; Lee, J.; Lee, J.; Shim, W.J. Abundance, Composition, and Distribution of Microplastics Larger than 20 μm in Sand Beaches of South Korea. Environ. Pollut. 2018, 238, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, X.; Huang, W.; Li, J.; Wang, C.; Zhang, D.; Zhang, C. Microplastic Pollution in Deep-Sea Sediments and Organisms of the Western Pacific Ocean. Environ. Pollut. 2020, 259, 113948. [Google Scholar] [CrossRef]
- Xie, X.; Deng, T.; Duan, J.; Xie, J.; Yuan, J.; Chen, M. Exposure to Polystyrene Microplastics Causes Reproductive Toxicity through Oxidative Stress and Activation of the P38 MAPK Signaling Pathway. Ecotoxicol. Environ. Saf. 2020, 190, 110133. [Google Scholar] [CrossRef]
- Harmon, S. The Effects of Microplastic Pollution on Aquatic Organisms. In Microplastic Contamination in Aquatic Environments; Elsevier: Amsterdam, The Netherlands, 2018; pp. 249–270. [Google Scholar] [CrossRef]
- Carlos de Sá, L.; Luís, L.G.; Guilhermino, L. Effects of microplastics on juveniles of the common goby (Pomatoschistus microps): Confusion with prey, reduction of the predatory performance and efficiency, and possible influence of developmental conditions. Environ. Pollut. 2015, 196, 359–362. [Google Scholar] [CrossRef]
- Xu, S.; Ma, J.; Ji, R.; Pan, K.; Miao, A.J. Microplastics in aquatic environments: Occurrence, accumulation, and biological effects. Sci. Total Environ. 2020, 703, 134699. [Google Scholar] [CrossRef] [PubMed]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and Importance of Microplastics in the Marine Environment: A Review of the Sources, Fate, Effects, and Potential Solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Okoffo, E.D.; Ribeiro, F.; O’Brien, J.W.; O’Brien, S.; Tscharke, B.J.; Gallen, M.; Samanipour, S.; Mueller, J.F.; Thomas, K.V. Identification and quantification of selected plastics in biosolids by pressurized liquid extraction combined with double-shot pyrolysis gas chromatography–mass spectrometry. Sci. Total Environ. 2020, 715, 136924. [Google Scholar] [CrossRef] [PubMed]
- Kalčíková, G.; Alič, B.; Skalar, T.; Bundschuh, M.; Gotvajn, A.Ž. Wastewater Treatment Plant Effluents as Source of Cosmetic Polyethylene Microbeads to Freshwater. Chemosphere 2017, 188, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Napper, I.E.; Thompson, R.C. Release of Synthetic Microplastic Plastic Fibres from Domestic Washing Machines: Effects of Fabric Type and Washing Conditions. Mar. Pollut. Bull. 2016, 112, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Li, W.C.; Tse, H.F.; Fok, L. Plastic Waste in the Marine Environment: A Review of Sources, Occurrence and Effects. Sci. Total Environ. 2016, 566–567, 333–349. [Google Scholar] [CrossRef] [PubMed]
- Sources of Microplastic-Pollution to the Marine Environment—Miljødirektoratet. Available online: https://www.miljodirektoratet.no/publikasjoner/2015/februar/sources-of-microplastic-pollution-to-the-marine-environment/ (accessed on 8 April 2024).
- Setälä, O.; Fleming-Lehtinen, V.; Lehtiniemi, M. Ingestion and Transfer of Microplastics in the Planktonic Food Web. Environ. Pollut. 2014, 185, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Desforges, J.-P.W.; Galbraith, M.; Ross, P.S. Ingestion of Microplastics by Zooplankton in the Northeast Pacific Ocean. Arch. Environ. Contam. Toxicol. 2015, 69, 320–330. [Google Scholar] [CrossRef]
- Walkinshaw, C.; Lindeque, P.K.; Thompson, R.; Tolhurst, T.; Cole, M. Microplastics and seafood: Lower trophic organisms at highest risk of contamination. Ecotoxicol. Environ. Saf. 2020, 190, 110066. [Google Scholar] [CrossRef]
- Marn, N.; Jusup, M.; Kooijman, S.A.L.M.; Klanjscek, T. Quantifying Impacts of Plastic Debris on Marine Wildlife Identifies Ecological Breakpoints. Ecol. Lett. 2020, 23, 1479–1487. [Google Scholar] [CrossRef]
- Driedger, A.G.J.; Dürr, H.H.; Mitchell, K.; Van Cappellen, P. Plastic Debris in the Laurentian Great Lakes: A Review. J. Great Lakes Res. 2015, 41, 9–19. [Google Scholar] [CrossRef]
- Saha, M.; Naik, A.; Desai, A.; Nanajkar, M.; Rathore, C.; Kumar, M.; Gupta, P. Microplastics in Seafood as an Emerging Threat to Marine Environment: A Case Study in Goa, West Coast of India. Chemosphere 2021, 270, 129359. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Svelato, A.; Santacroce, C.; Catalano, P.; Notarstefano, V.; Carnevali, O.; Papa, F.; Rongioletti, M.C.A.; Baiocco, F.; Draghi, S.; et al. Plasticenta: First Evidence of Microplastics in Human Placenta. Environ. Int. 2021, 146, 106274. [Google Scholar] [CrossRef]
- Cózar, A.; Echevarría, F.; González-Gordillo, J.I.; Irigoien, X.; Ubeda, B.; Hernández-León, S.; Palma, A.T.; Navarro, S.; García-de-Lomas, J.; Ruiz, A.; et al. Plastic Debris in the Open Ocean. Proc. Natl. Acad. Sci. USA 2014, 111, 10239–10244. [Google Scholar] [CrossRef]
- Wright, S.L.; Rowe, D.; Thompson, R.C.; Galloway, T.S. Microplastic Ingestion Decreases Energy Reserves in Marine Worms. Curr. Biol. 2013, 23, R1031–R1033. [Google Scholar] [CrossRef] [PubMed]
- Gonte, R.; Balasubramanian, K. Chemically Modified Polymer Beads for Sorption of Gold from Waste Gold Solution. J. Hazard. Mater. 2012, 217–218, 447–451. [Google Scholar] [CrossRef]
- Gore, P.M.; Kandasubramanian, B. Heterogeneous Wettable Cotton Based Superhydrophobic Janus Biofabric Engineered with PLA/Functionalized-Organoclay Microfibers for Efficient Oil–Water Separation. J. Mater. Chem. A 2018, 6, 7457–7479. [Google Scholar] [CrossRef]
- Thakur, K.; Kandasubramanian, B. Graphene and Graphene Oxide-Based Composites for Removal of Organic Pollutants: A Review. J. Chem. Eng. Data 2019, 64, 833–867. [Google Scholar] [CrossRef]
- Rajhans, A.; Gore, P.M.; Siddique, S.K.; Kandasubramanian, B. Ion-imprinted nanofibers of PVDF/1-butyl-3-methylimidazolium tetrafluoroborate for dynamic recovery of europium (III) ions from mimicked effluent. J. Environ. Chem. Eng. 2019, 7, 103068. [Google Scholar] [CrossRef]
- Campanale, C.; Massarelli, C.; Savino, I.; Locaputo, V.; Uricchio, V.F. A Detailed Review Study on Potential Effects of Microplastics and Additives of Concern on Human Health. Int. J. Environ. Res. Public. Health 2020, 17, 1212. [Google Scholar] [CrossRef]
- Shiry, N.; Darvishi, P.; Gholamhossieni, A.; Pastorino, P.; Faggio, C. Exploring the Combined Interplays: Effects of Cypermethrin and Microplastic Exposure on the Survival and Antioxidant Physiology of Astacus leptodactylus. J. Contam. Hydrol. 2023, 259, 104257. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ren, X.; Rene, E.R.; Wang, Z.; Zhou, L.; Zhang, Z.; Wang, Q. The Degradation Performance of Different Microplastics and Their Effect on Microbial Community during Composting Process. Bioresour. Technol. 2021, 332, 125133. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-C.; Chen, M.-Y.; Fang, C.; Zheng, R.-H.; Jiang, Y.-L.; Zhang, Y.-S.; Wang, K.-J.; Bailey, C.; Segner, H.; Bo, J. Microplastics Negatively Impact Embryogenesis and Modulate the Immune Response of the Marine Medaka Oryzias Melastigma. Mar. Pollut. Bull. 2020, 158, 111349. [Google Scholar] [CrossRef]
- Yang, S.; Li, M.; Kong, R.Y.C.; Li, L.; Li, R.; Chen, J.; Lai, K.P. Reproductive Toxicity of Micro- and Nanoplastics. Environ. Int. 2023, 177, 108002. [Google Scholar] [CrossRef] [PubMed]
- Hodkovicova, N.; Hollerova, A.; Svobodova, Z.; Faldyna, M.; Faggio, C. Effects of Plastic Particles on Aquatic Invertebrates and Fish—A Review. Environ. Toxicol. Pharmacol. 2022, 96, 104013. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.; Dudas, S.E. Microplastic Ingestion by Wild and Cultured Manila Clams (Venerupis philippinarum) from Baynes Sound, British Columbia. Arch. Environ. Contam. Toxicol. 2016, 71, 147–156. [Google Scholar] [CrossRef]
- Pedersen, A.F.; Gopalakrishnan, K.; Boegehold, A.G.; Peraino, N.J.; Westrick, J.A.; Kashian, D.R. Microplastic Ingestion by Quagga Mussels, Dreissena Bugensis, and Its Effects on Physiological Processes. Environ. Pollut. 2020, 260, 113964. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Jürgens, M.D.; Lahive, E.; van Bodegom, P.M.; Vijver, M.G. The Influence of Exposure and Physiology on Microplastic Ingestion by the Freshwater Fish Rutilus Rutilus (Roach) in the River Thames, UK. Environ. Pollut. 2018, 236, 188–194. [Google Scholar] [CrossRef] [PubMed]
- O’Donovan, S.; Mestre, N.C.; Abel, S.; Fonseca, T.G.; Carteny, C.C.; Cormier, B.; Keiter, S.H.; Bebianno, M.J. Ecotoxicological Effects of Chemical Contaminants Adsorbed to Microplastics in the Clam Scrobicularia Plana. Front. Mar. Sci. 2018, 5, 143. [Google Scholar] [CrossRef]
- Savuca, A.; Nicoara, M.N.; Ciobica, A.; Gorgan, D.L.; Ureche, D.; Balmus, I.M. Current Aspects on the Plastic Nano- and Microparticles Toxicity in Zebrafish—Focus on the Correlation between Oxidative Stress Responses and Neurodevelopment. Animals 2023, 13, 1810. [Google Scholar] [CrossRef]
- Trestrail, C.; Nugegoda, D.; Shimeta, J. Invertebrate Responses to Microplastic Ingestion: Reviewing the Role of the Antioxidant System. Sci. Total Environ. 2020, 734, 138559. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Xu, K.; Zhang, B.; Ye, Y.; Zhang, Q.; Jiang, W. Cellular Internalization and Release of Polystyrene Microplastics and Nanoplastics. Sci. Total Environ. 2021, 779, 146523. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.Y.; Lee, T.H. Antioxidant enzymes as redox-based biomarkers: A brief review. BMB Rep. 2015, 48, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Ratan, R.R. The Chemical Biology of Ferroptosis in the Central Nervous System. Cell Chem. Biol. 2020, 27, 479–498. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.; Chen, H.; Liu, X.; Dang, Y.; Li, X.; Yu, Y.; Li, B.; Li, X.; Sun, Y.; Ding, P.; et al. UV-Aged Microplastics Induces Neurotoxicity by Affecting the Neurotransmission in Larval Zebrafish. Chemosphere 2023, 324, 138252. [Google Scholar] [CrossRef]
- Yu, H.; Chen, Q.; Qiu, W.; Ma, C.; Gao, Z.; Chu, W.; Shi, H. Concurrent Water- and Foodborne Exposure to Microplastics Leads to Differential Microplastic Ingestion and Neurotoxic Effects in Zebrafish. Water Res. 2022, 219, 118582. [Google Scholar] [CrossRef] [PubMed]
- Mak, C.W.; Ching-Fong Yeung, K.; Chan, K.M. Acute Toxic Effects of Polyethylene Microplastic on Adult Zebrafish. Ecotoxicol. Environ. Saf. 2019, 182, 109442. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liu, P.; Wu, X.; Shi, H.; Huang, H.; Wang, H.; Gao, S. Insight into Chain Scission and Release Profiles from Photodegradation of Polycarbonate Microplastics. Water Res. 2021, 195, 116980. [Google Scholar] [CrossRef] [PubMed]
- Umamaheswari, S.; Priyadarshinee, S.; Bhattacharjee, M.; Kadirvelu, K.; Ramesh, M. Exposure to Polystyrene Microplastics Induced Gene Modulated Biological Responses in Zebrafish (Danio rerio). Chemosphere 2021, 281, 128592. [Google Scholar] [CrossRef]
- Lee, Y.; Yoon, D.-S.; Lee, Y.; Kwak, J.I.; An, Y.-J.; Lee, J.-S.; Park, J.J.C. Combined Exposure to Microplastics and Zinc Produces Sex-Specific Responses in the Water Flea Daphnia magna. J. Hazard. Mater. 2021, 420, 126652. [Google Scholar] [CrossRef]
- Lu, K.; Qiao, R.; An, H.; Zhang, Y. Influence of Microplastics on the Accumulation and Chronic Toxic Effects of Cadmium in Zebrafish (Danio rerio). Chemosphere 2018, 202, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Brieño-Enríquez, M.A.; Larriba, E.; Del Mazo, J. Endocrine Disrupters, microRNAs, and Primordial Germ Cells: A Dangerous Cocktail. Fertil. Steril. 2016, 106, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Cormier, B.; Cachot, J.; Blanc, M.; Cabar, M.; Clérandeau, C.; Dubocq, F.; Le Bihanic, F.; Morin, B.; Zapata, S.; Bégout, M.-L.; et al. Environmental Microplastics Disrupt Swimming Activity in Acute Exposure in Danio rerio Larvae and Reduce Growth and Reproduction Success in Chronic Exposure in D. rerio and Oryzias melastigma. Environ. Pollut. 2022, 308, 119721. [Google Scholar] [CrossRef] [PubMed]
- Plavan, G.; Strungaru, S.A.; Nicoara, M.; Todirascu-Ciornea, E. Oxidative Stress and Changes in Swimming Performances at Zebrafish Model (Danio rerio H. 1822) Produced by Acute Exposure to Deltamethrin. J. Surv. Fish. Sci. 2019, 5, 121–137. [Google Scholar]
- Schäfer, R.B.; Von Der Ohe, P.C.; Rasmussen, J.; Kefford, B.J.; Beketov, M.A.; Schulz, R.; Liess, M. Thresholds for the Effects of Pesticides on Invertebrate Communities and Leaf Breakdown in Stream Ecosystems. Environ. Sci. Technol. 2012, 46, 5134–5142. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.C. Biomarkers in Toxicology; Academic Press: Cambridge, MA, USA, 2019; ISBN 978-0-12-404630-6. [Google Scholar]
- An Introduction to Insecticides (4th edition)|Radcliffe’s IPM World Textbook. Available online: https://ipmworld.umn.edu/ware-intro-insecticides (accessed on 8 April 2024).
- Robea, M.A.; Petrovici, A.; Ureche, D.; Nicoara, M.; Ciobica, A.S. Histopathological and Behavioral Impairments in Zebrafish (Danio rerio) Chronically Exposed to a Cocktail of Fipronil and Pyriproxyfen. Life 2023, 13, 1874. [Google Scholar] [CrossRef] [PubMed]
- PubChem. Fipronil. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/3352 (accessed on 8 April 2024).
- Gunasekara, A.; Truong, T.; Goh, K.; Spurlock, F.; Tjeerdema, R. Environmental Fate and Toxicology of Fipronil. J. Pestic. Sci. 2007, 32, 189–199. [Google Scholar] [CrossRef]
- Bloomquist, J.R. Chloride Channels as Tools for Developing Selective Insecticides. Arch. Insect Biochem. Physiol. 2003, 54, 145–156. [Google Scholar] [CrossRef]
- Narahashi, T. Neurophysiological Effects of Insecticides. Hayes Handb. Pestic. Toxicol. 2010, 1, 799–817. [Google Scholar] [CrossRef]
- Ghit, A.; Assal, D.; Al-Shami, A.S.; Hussein, D.E.E. GABAA receptors: Structure, function, pharmacology, and related disorders. J. Genet. Eng. Biotechnol. 2021, 19, 123. [Google Scholar] [CrossRef]
- Wu, C.-H.; Lu, C.-W.; Hsu, T.-H.; Wu, W.-J.; Wang, S.-E. Neurotoxicity of Fipronil Affects Sensory and Motor Systems in Zebrafish. Pestic. Biochem. Physiol. 2021, 177, 104896. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA), Conclusion on the peer review of the pesticide risk assessment for bees for the active substance fipronil. EFSA J. 2013, 11, 3158. [CrossRef]
- Chemical Pesticides Mode of Action and Toxicology|Jørgen Stenersen. Available online: https://www.taylorfrancis.com/books/mono/10.1201/9780203646830/chemical-pesticides-mode-action-toxicology-j%C3%B8rgen-stenersen (accessed on 8 April 2024).
- Page, S. Antiparasitic Drugs. In Small Animal Clinical Pharmacology; Elsevier Health Sciences: Amsterdam, The Netherlands, 2008; pp. 198–260. [Google Scholar] [CrossRef]
- Jeyaratnam, J. Acute pesticide poisoning: A major global health problem. World Health Stat. Q. 1990, 43, 139–144. [Google Scholar] [PubMed]
- Chodorowski, Z.; Anand, J.S. Accidental Dermal and Inhalation Exposure with Fipronil—A Case Report. J. Toxicol. Clin. Toxicol. 2004, 42, 189–190. [Google Scholar] [CrossRef]
- Bobe, A.; Meallier, P.; Cooper, J.F.; Coste, C.M. Kinetics and Mechanisms of Abiotic Degradation of Fipronil (Hydrolysis and Photolysis). J. Agric. Food Chem. 1998, 46, 2834–2839. [Google Scholar] [CrossRef]
- Mohamed, F.; Senarathna, L.; Percy, A.; Abeyewardene, M.; Eaglesham, G.; Cheng, R.; Azher, S.; Hittarage, A.; Dissanayake, W.; Sheriff, M.H.R.; et al. Acute Human Self-Poisoning with the N-Phenylpyrazole Insecticide Fipronil—A GABA A-Gated Chloride Channel Blocker. J. Toxicol. Clin. Toxicol. 2004, 42, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-Y.; Chen, W.-T.; Lee, H.-C.; Yang, P.-H.; Yang, H.-J.; Tsai, H.-J. The Transcription Factor Six1a Plays an Essential Role in the Craniofacial Myogenesis of Zebrafish. Dev. Biol. 2009, 331, 152–166. [Google Scholar] [CrossRef] [PubMed]
- Ying, G.-G.; Kookana, R.S. Sorption of Fipronil and Its Metabolites on Soils from South Australia. J. Environ. Sci. Health Part. B 2001, 36, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Ha, T. Fipronil in Surface Water: An Environmental Calamity Remaining Under Radar in the Netherlands. J. Ecol. Toxicol. 2018, 2, 111. [Google Scholar]
- Stehr, C.M.; Linbo, T.L.; Incardona, J.P.; Scholz, N.L. The Developmental Neurotoxicity of Fipronil: Notochord Degeneration and Locomotor Defects in Zebrafish Embryos and Larvae. Toxicol. Sci. 2006, 92, 270–278. [Google Scholar] [CrossRef]
- Sun, L.; Jin, R.; Peng, Z.; Zhou, Q.; Qian, H.; Fu, Z. Effects of Trilostane and Fipronil on the Reproductive Axis in an Early Life Stage of the Japanese Medaka (Oryzias latipes). Ecotoxicology 2014, 23, 1044–1054. [Google Scholar] [CrossRef] [PubMed]
- Beggel, S.; Werner, I.; Connon, R.E.; Geist, J.P. Impacts of the Phenylpyrazole Insecticide Fipronil on Larval Fish: Time-Series Gene Transcription Responses in Fathead Minnow (Pimephales promelas) Following Short-Term Exposure. Sci. Total Environ. 2012, 426, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Martínez, M.A.; Wu, Q.; Ares, I.; Martínez-Larrañaga, M.R.; Anadón, A.; Yuan, Z. Fipronil Insecticide Toxicology: Oxidative Stress and Metabolism. Crit. Rev. Toxicol. 2016, 46, 876–899. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Wang, C.; Wang, J.; Zhang, X.; Zhou, Z.; Zhao, M.; Lu, C. Fipronil-Induced Enantioselective Developmental Toxicity to Zebrafish Embryo-Larvae Involves Changes in DNA Methylation. Sci. Rep. 2017, 7, 2284. [Google Scholar] [CrossRef]
- Sullivan, J.J.; Goh, K.S. Environmental Fate and Properties of Pyriproxyfen. J. Pestic. Sci. 2008, 33, 339–350. [Google Scholar] [CrossRef]
- Devillers, J. Fate and Ecotoxicological Effects of Pyriproxyfen in Aquatic Ecosystems. Environ. Sci. Pollut. Res. Int. 2020, 27, 16052–16068. [Google Scholar] [CrossRef] [PubMed]
- Moura, L.; de Nadai, B.L.; Corbi, J.J. One Does Not Simply Apply Larvicides: Aedes Aegypti from Araraquara (Brazil) Has Reduced Susceptibility to Pyriproxyfen. Vet. Parasitol. Reg. Stud. Rep. 2023, 41, 100875. [Google Scholar] [CrossRef]
- Effects of Pyriproxyfen on Zebrafish Brain Mitochondria and Acetylcholinesterase. Available online: https://pubmed.ncbi.nlm.nih.gov/33297050/ (accessed on 8 April 2024).
- Park, H.; Lee, J.-Y.; Park, S.; Song, G.; Lim, W. Developmental Toxicity of Fipronil in Early Development of Zebrafish (Danio rerio) Larvae: Disrupted Vascular Formation with Angiogenic Failure and Inhibited Neurogenesis. J. Hazard. Mater. 2020, 385, 121531. [Google Scholar] [CrossRef] [PubMed]
- Gusso, D.; Reolon, G.K.; Gonzalez, J.B.; Altenhofen, S.; Kist, L.W.; Bogo, M.R.; Bonan, C.D. Pyriproxyfen Exposure Impairs Cognitive Parameters and Alters Cortisol Levels in Zebrafish. Front. Behav. Neurosci. 2020, 14, 103. [Google Scholar] [CrossRef]
- Maharajan, K.; Muthulakshmi, S.; Nataraj, B.; Ramesh, M.; Kadirvelu, K. Toxicity Assessment of Pyriproxyfen in Vertebrate Model Zebrafish Embryos (Danio rerio): A Multi Biomarker Study. Aquat. Toxicol. 2018, 196, 132–145. [Google Scholar] [CrossRef]
- Costa, L.G.; Giordano, G.; Guizzetti, M.; Vitalone, A. Neurotoxicity of pesticides: A brief review. Front. Biosci. 2008, 13, 1240–1249. [Google Scholar] [CrossRef]
- Richardson, R.; Tracey-White, D.; Webster, A.; Moosajee, M. The Zebrafish Eye-a Paradigm for Investigating Human Ocular Genetics. Eye 2017, 31, 68–86. [Google Scholar] [CrossRef] [PubMed]
- Ghaffar, A.; Hussain, R.; Abbas, G.; Kalim, M.; Khan, A.; Ferrando, S.; Gallus, L.; Ahmed, Z. Fipronil (Phenylpyrazole) Induces Hemato-Biochemical, Histological and Genetic Damage at Low Doses in Common carp, Cyprinus carpio (Linnaeus, 1758). Ecotoxicology 2018, 27, 1261–1271. [Google Scholar] [CrossRef]
- Xu, H.; Liu, X.; Jia, Y.; Dong, F.; Xu, J.; Wu, X.; Yang, Y.; Zheng, Y. Fipronil-induced toxic effects in zebrafish (Danio rerio) larvae by using digital gene expression profiling. Sci. Total Environ. 2018, 15, 550–559. [Google Scholar] [CrossRef]
- Abdel-Daim, M.M.; El-Ghoneimy, A. Synergistic Protective Effects of Ceftriaxone and Ascorbic Acid against Subacute Deltamethrin-Induced Nephrotoxicity in Rats. Ren. Fail. 2015, 37, 297–304. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, Q.; Pang, Y.; Song, X.; Zhou, N.; Wang, J.; He, L.; Lv, J.; Song, Y.; Cheng, Y.; et al. The protective effects of melatonin on oxidative damage and the immune system of the Chinese mitten crab (Eriocheir sinensis) exposed to deltamethrin. Sci. Total Environ. 2019, 25, 1426–1434. [Google Scholar] [CrossRef] [PubMed]
- Dubey, N.; Khan, A.M.; Raina, R. Sub-Acute Deltamethrin and Fluoride Toxicity Induced Hepatic Oxidative Stress and Biochemical Alterations in Rats. Bull. Environ. Contam. Toxicol. 2013, 91, 334–338. [Google Scholar] [CrossRef]
- He, F.; Wang, S.; Liu, L.; Chen, S.; Zhang, Z.; Sun, J. Clinical Manifestations and Diagnosis of Acute Pyrethroid Poisoning. Arch. Toxicol. 1989, 63, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, R.; Megha, P.; Sreedev, P. Organochlorine Pesticides, Their Toxic Effects on Living Organisms and Their Fate in the Environment. Interdiscip. Toxicol. 2016, 9, 90–100. [Google Scholar] [CrossRef]
- Matsuo, N. Discovery and Development of Pyrethroid Insecticides. Proc. Jpn. Acad. Ser. B 2019, 95, 378–400. [Google Scholar] [CrossRef]
- Chauhan, R.; Kumari, B.; Rana, M.K. Effect of Fruit and Vegetable Processing on Reduction of Synthetic Pyrethroid Residues. Rev. Environ. Contam. Toxicol. 2014, 229, 89–110. [Google Scholar] [CrossRef] [PubMed]
- Albaseer, S.S. Factors controlling the fate of pyrethroids residues during post-harvest processing of raw agricultural crops: An overview. Food Chem. 2019, 295, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Dubey, P.; Khare, R.L.; Lakra, D. A Unique Case of Intravenous Pyrethroid Administration with Poisoning. MRIMS J. Health Sci. 2023, 11, 216–218. [Google Scholar]
- Liu, Y.; Li, S.; Ni, Z.; Qu, M.; Zhong, D.; Ye, C.; Tang, F. Pesticides in Persimmons, Jujubes and Soil from China: Residue Levels, Risk Assessment and Relationship between Fruits and Soils. Sci. Total Environ. 2016, 542 Pt A, 620–628. [Google Scholar] [CrossRef]
- O’Neil, M.J.; Heckelman, P.E.; Koch, C.B.; Roman, K.J. (Eds.) The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th ed.; Merck and Co., Inc.: Hoboken, NJ, USA, 2006; ISBN 13-978-0-911910-001. [Google Scholar]
- Meister, R.T.; Sine, C. (Eds.) Crop Protection Handbook; Meister Publishing Company: Willoughby, OH, USA, 2014; Volume 100, ISBN 9781892829276. [Google Scholar]
- PubChem. Deltamethrin. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/40585 (accessed on 9 April 2024).
- Subacute Poisoning of Mice with Deltamethrin Produces Memory Impairment, Reduced Locomotor Activity, Liver Damage and Changes in Blood Morphology in the Mechanism of Oxidative Stress. Available online: https://pubmed.ncbi.nlm.nih.gov/25933966/ (accessed on 9 April 2024).
- Shakerkhatibi, M.; Mosaferi, M.; Asghari Jafarabadi, M.; Lotfi, E.; Belvasi, M. Pesticides Residue in Drinking Groundwater Resources of Rural Areas in the Northwest of Iran. Health Promot. Perspect. 2014, 4, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Allinson, G.; Zhang, P.; Bui, A.; Allinson, M.; Rose, G.; Marshall, S.; Pettigrove, V. Pesticide and Trace Metal Occurrence and Aquatic Benchmark Exceedances in Surface Waters and Sediments of Urban Wetlands and Retention Ponds in Melbourne, Australia. Environ. Sci. Pollut. Res. Int. 2015, 22, 10214–10226. [Google Scholar] [CrossRef] [PubMed]
- Feo, M.L.; Ginebreda, A.; Eljarrat, E.; Barceló, D. Presence of Pyrethroid Pesticides in Water and Sediments of Ebro River Delta. J. Hydrol. 2010, 393, 156–162. [Google Scholar] [CrossRef]
- Lao, W.; Tsukada, D.; Greenstein, D.J.; Bay, S.M.; Maruya, K.A. Analysis, Occurrence, and Toxic Potential of Pyrethroids, and Fipronil in Sediments from an Urban Estuary. Environ. Toxicol. Chem. 2010, 29, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Miao, W.; Jiang, Y.; Hong, Q.; Sheng, H.; Liu, P.; Huang, Y.; Cheng, J.; Pan, X.; Yu, Q.; Wu, Y.; et al. Systematic Evaluation of the Toxicological Effects of Deltamethrin Exposure in Zebrafish Larvae. Environ. Toxicol. Pharmacol. 2023, 100, 104155. [Google Scholar] [CrossRef]
- Yadav, R.; Shinde, N.; Patil, K.; Kote, A.; Kadam, P. Deltamethrin Toxicity: Impacts on Non-Target Organisms and the Environment. Environ. Ecol. 2023, 41, 2039–2043. [Google Scholar] [CrossRef]
- Ullah, S.; Li, Z.U.L.; Arifeen, M.Z.; Khan, S.U.; Fahad, S. Multiple biomarkers based appraisal of deltamethrin induced toxicity in silver carp (Hypophthalmichthys molitrix). Chemosphere 2019, 214, 519–533. [Google Scholar] [CrossRef] [PubMed]
- Barata, C.; Baird, D.J.; Nogueira, A.J.A.; Soares, A.M.V.M.; Riva, M.C. Toxicity of Binary Mixtures of Metals and Pyrethroid Insecticides to Daphnia magna Straus. Implications for Multi-Substance Risks Assessment. Aquat. Toxicol. 2006, 78, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Jijie, R.; Solcan, G.; Nicoara, M.; Micu, D.; Strungaru, S.-A. Antagonistic Effects in Zebrafish (Danio rerio) Behavior and Oxidative Stress Induced by Toxic Metals and Deltamethrin Acute Exposure. Sci. Total Environ. 2020, 698, 134299. [Google Scholar] [CrossRef] [PubMed]
- Haverinen, J.; Vornanen, M. Deltamethrin Is Toxic to the Fish (Crucian carp, Carassius carassius) Heart. Pestic. Biochem. Physiol. 2016, 129, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Aksakal, E.; Ceyhun, S.B.; Erdoğan, O.; Ekinci, D. Acute and Long-Term Genotoxicity of Deltamethrin to Insulin-like Growth Factors and Growth Hormone in Rainbow Trout. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 152, 451–455. [Google Scholar] [CrossRef]
- Velisek, J.; Zusková, E.; Máchová, J.; Svobodova, Z. Effects of Sub-Chronic Exposure to Terbutryn in Common Carp (Cyprinus carpio L.). Ecotoxicol. Environ. Saf. 2009, 73, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Calta, M.; Ural, M. Acute Toxicity of the Synthetic Pyrethroid Deltamethrin to Young Mirror Carp, Cyprinus carpio. Fresenius Environ. Bull. 2004, 13, 1179–1183. [Google Scholar]
- Kung, T.S.; Richardson, J.R.; Cooper, K.R.; White, L.A. Developmental Deltamethrin Exposure Causes Persistent Changes in Dopaminergic Gene Expression, Neurochemistry, and Locomotor Activity in Zebrafish. Toxicol. Sci. 2015, 146, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Zhang, T.; Li, S.; Ren, Z.; Yang, M.; Pan, H.; Xu, S.; Qi, L.; Chon, T.-S. Integrative Characterization of Toxic Response of Zebra Fish (Danio rerio) to Deltamethrin Based on AChE Activity and Behavior Strength. Biomed. Res. Int. 2016, 2016, 7309184. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, J.; Han, X.; Huang, T. The Use of Zebrafish (Danio rerio) Behavioral Responses in Identifying Sublethal Exposures to Deltamethrin. Int. J. Environ. Res. Public Health 2014, 11, 3650–3660. [Google Scholar] [CrossRef]
- DeMicco, A.; Cooper, K.R.; Richardson, J.R.; White, L.A. Developmental Neurotoxicity of Pyrethroid Insecticides in Zebrafish Embryos. Toxicol. Sci. 2010, 113, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Koc, N.D.; Teksöz, N.; Ural, M.; Akbulut, C. Histological structure of zebrafish (Danio rerio, Hamilton, 1822) testicles. Elixir Aquac. 2012, 46, 8117–8120. [Google Scholar]
- Liu, X.Y.; Zhang, Q.P.; Li, S.B.; Mi, P.; Chen, D.Y.; Zhao, X.; Feng, X.Z. Developmental toxicity and neurotoxicity of synthetic organic insecticides in zebrafish (Danio rerio): A comparative study of deltamethrin, acephate, and thiamethoxam. Chemosphere 2018, 199, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Strungaru, S.-A.; Plavan, G.; Ciobica, A.; Nicoara, M.; Robea, M.A.; Solcan, C.; Petrovici, A. Toxicity and Chronic Effects of Deltamethrin Exposure on Zebrafish (Danio rerio) as a Reference Model for Freshwater Fish Community. Ecotoxicol. Environ. Saf. 2019, 171, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Zolkipli-Cunningham, Z.; Falk, M.J. Clinical effects of chemical exposures on mitochondrial function. Toxicology 2017, 391, 90–99. [Google Scholar] [CrossRef]
- Rayner, T.S.; Creese, R.G. A Review of Rotenone Use for the Control of Non-Indigenous Fish in Australian Fresh Waters, and an Attempted Eradication of the Noxious Fish, Phalloceros caudimaculatus. N. Z. J. Mar. Freshw. Res. 2006, 40, 477–486. [Google Scholar] [CrossRef]
- National Toxicology Program. Available online: https://ntp.niehs.nih.gov (accessed on 9 April 2024).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 6758, Rotenone. 2024. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Rotenone (accessed on 9 April 2024).
- Gupta, R.C. Rotenone. In Veterinary Toxicology—Basic and Clinical Principles, 2nd ed.; Gupta, R.C., Ed.; Academic Press: New York, NY, USA, 2012; pp. 620–623. ISBN 978-0-12-385926-6. [Google Scholar]
- Hayes, W.J.; Laws, E.P. Handbook of Pesticide Toxicology; Academic Press: San Diego, CA, USA, 2010; Volume 3, ISBN 0-12-334163-9. [Google Scholar]
- Ott, K. Rotenone. In A Brief Review of Its Chemistry, Environmental Fate, and the Toxicity of Rotenone Formulations; New Mexico Council of Trout Unlimited: Albuquerque, NM, USA, 2006. [Google Scholar]
- Ling, N. Rotenone—A Review of Its Toxicity and Use for Fisheries Management; Department of Conservation: Wellington, New Zealand, 2003. [Google Scholar]
- Nass, A.; Nass, R.D. Actigraphic evidence for night-time hyperkinesia in Parkinson’s disease. Int. J. Neurosci. 2008, 118, 291–310. [Google Scholar] [CrossRef] [PubMed]
- Saybasili, H.; Akkentli, F. Rotenone is a pesticide controlling the habitat quality of aquatic ecosystems and has a negative impact on neuron activity. Rev. Hydrobiol. 2011, 4, 1–16. [Google Scholar]
- Melo, K.M.; Oliveira, R.; Grisolia, C.K.; Domingues, I.; Pieczarka, J.C.; de Souza Filho, J.; Nagamachi, C.Y. Short-Term Exposure to Low Doses of Rotenone Induces Developmental, Biochemical, Behavioral, and Histological Changes in Fish. Environ. Sci. Pollut. Res. Int. 2015, 22, 13926–13938. [Google Scholar] [CrossRef]
- Sherer, T.B.; Betarbet, R.; Testa, C.M.; Seo, B.B.; Richardson, J.R.; Kim, J.H.; Miller, G.W.; Yagi, T.; Matsuno-Yagi, A.; Greenamyre, J.T. Mechanism of Toxicity in Rotenone Models of Parkinson’s Disease. J. Neurosci. 2003, 23, 10756–10764. [Google Scholar] [CrossRef]
- Emerit, J.; Edeas, M.; Bricaire, F. Neurodegenerative Diseases and Oxidative Stress. Biomed. Pharmacother. 2004, 58, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.L.; James-Kracke, M.; Sun, G.Y.; Sun, A.Y. Oxidative and Inflammatory Pathways in Parkinson’s Disease. Neurochem. Res. 2009, 34, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Watabe, M.; Nakaki, T. Mitochondrial complex I inhibitor rotenone-elicited dopamine redistribution from vesicles to cytosol in human dopaminergic SH-SY5Y cells. J. Pharmacol. Exp. Ther. 2007, 323, 499–507. [Google Scholar] [PubMed]
- Ilie, O.-D.; Duta, R.; Balmus, I.-M.; Savuca, A.; Petrovici, A.; Nita, I.-B.; Antoci, L.-M.; Jijie, R.; Mihai, C.-T.; Ciobica, A.; et al. Assessing the Neurotoxicity of a Sub-Optimal Dose of Rotenone in Zebrafish (Danio rerio) and the Possible Neuroactive Potential of Valproic Acid, Combination of Levodopa and Carbidopa, and Lactic Acid Bacteria Strains. Antioxidants 2022, 11, 2040. [Google Scholar] [CrossRef] [PubMed]
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Noble, S.; Ekker, M. Modeling Neurodegeneration in Zebrafish. Curr. Neurol. Neurosci. Rep. 2011, 11, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of Embryonic Development of the Zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Wullimann, M.F.; Rink, E. Detailed Immunohistology of Pax6 Protein and Tyrosine Hydroxylase in the Early Zebrafish Brain Suggests Role of Pax6 Gene in Development of Dopaminergic Diencephalic Neurons. Brain Res. Dev. Brain Res. 2001, 131, 173–191. [Google Scholar] [CrossRef]
- Rink, E.; Wullimann, M.F. Connections of the Ventral Telencephalon and Tyrosine Hydroxylase Distribution in the Zebrafish Brain (Danio rerio) Lead to Identification of an Ascending Dopaminergic System in a Teleost. Brain Res. Bull. 2002, 57, 385–387. [Google Scholar] [CrossRef]
- Doyle, J.M.; Croll, R.P. Critical Review of Zebrafish Models of Parkinson’s Disease. Front. Pharmacol. 2022, 13, 835827. [Google Scholar] [CrossRef] [PubMed]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic Systemic Pesticide Exposure Reproduces Features of Parkinson’s Disease. Nat. Neurosci. 2000, 3, 1301–1306. [Google Scholar] [CrossRef] [PubMed]
- Parkinson’s Disease. Available online: https://www.researchgate.net/publication/233887624_Parkinson’s_Disease (accessed on 9 April 2024).
- Liu, Y.; Sun, J.-D.; Song, L.-K.; Li, J.; Chu, S.-F.; Yuan, Y.-H.; Chen, N.-H. Environment-Contact Administration of Rotenone: A New Rodent Model of Parkinson’s Disease. Behav. Brain Res. 2015, 294, 149–161. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, W.; Yang, J.; Wang, F.; Sima, Y.; Zhong, Z.-M.; Wang, H.; Hu, L.-F.; Liu, C.-F. Parkinson’s Disease-like Motor and Non-Motor Symptoms in Rotenone-Treated Zebrafish. Neurotoxicology 2017, 58, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Khotimah, H.; Sumitro, S.; Widodo, M. Zebrafish Parkinson’s Model: Rotenone Decrease Motility, Dopamine, and Increase α-Synuclein Aggregation and Apoptosis of Zebrafish Brain. Int. J. PharmTech Res. 2015, 8, 614–621. [Google Scholar]
- MDS Clinical Diagnostic Criteria for Parkinson’s Disease. Movement Disorders; Wiley Online Library. 2015. Available online: https://movementdisorders.onlinelibrary.wiley.com/doi/10.1002/mds.26424 (accessed on 9 April 2024).
- Ziemssen, T.; Reichmann, H. Non-Motor Dysfunction in Parkinson’s Disease. Park. Relat. Disord. 2007, 13, 323–332. [Google Scholar] [CrossRef]
- Pamies, D.; Block, K.; Lau, P.; Gribaldo, L.; Pardo, C.A.; Barreras, P.; Smirnova, L.; Wiersma, D.; Zhao, L.; Harris, G.; et al. Rotenone exerts developmental neurotoxicity in a human brain spheroid model. Toxicol. Appl. Pharmacol. 2018, 354, 101–114. [Google Scholar] [CrossRef]
- Kieburtz, K.; Wunderle, K.B. Parkinson’s Disease: Evidence for Environmental Risk Factors. Mov. Disord. 2013, 28, 8–13. [Google Scholar] [CrossRef]
- Hwang, O. Role of Oxidative Stress in Parkinson’s Disease. Exp. Neurobiol. 2013, 22, 11–17. [Google Scholar] [CrossRef]
- McCormack, A.L.; Atienza, J.G.; Langston, J.W.; Di Monte, D.A. Decreased Susceptibility to Oxidative Stress Underlies the Resistance of Specific Dopaminergic Cell Populations to Paraquat-Induced Degeneration. Neuroscience 2006, 141, 929–937. [Google Scholar] [CrossRef]
- Rappold, P.M.; Cui, M.; Chesser, A.S.; Tibbett, J.; Grima, J.C.; Duan, L.; Sen, N.; Javitch, J.A.; Tieu, K. Paraquat Neurotoxicity Is Mediated by the Dopamine Transporter and Organic Cation Transporter-3. Proc. Natl. Acad. Sci. USA 2011, 108, 20766–20771. [Google Scholar] [CrossRef] [PubMed]
- Ünal, İ.; Üstündağ, Ü.V.; Ateş, P.S.; Eğilmezer, G.; Alturfan, A.A.; Yiğitbaşı, T.; Emekli-Alturfan, E. Rotenone Impairs Oxidant/Antioxidant Balance Both in Brain and Intestines in Zebrafish. Int. J. Neurosci. 2019, 129, 363–368. [Google Scholar] [CrossRef] [PubMed]
| Doses (mg/L) | Exposure Time | Effects | Authors |
|---|---|---|---|
| 0.5, 1 and 2 | 96 h | Increased SOD and CAT activity Increased lipid peroxidation | [164] |
| 2.5, 7.5 and 15 | 72 h | Decreased cell proliferation | [184] |
| 0.4 and 0.8 | 5 days | Increased anxiety Disturbance of swimming behavior Increased lipid peroxidation | [178] |
| 0.33 and 0.8 | 5 days | Locomotor defects | [175] |
| Doses (mg/L) and Exposure Times | Effects | Authors |
|---|---|---|
| 0.0765 and 21.461 | Inhibited AChE activity Increased ROS levels | [183] |
| 0.125, 0.675 and 1.75 (96 h) | No locomotor disorders recorded No anxiety-like behaviors observed | [185] |
| 1.66 (96 h) | Increased ROS levels Increased lipid peroxidation Increased nitric oxide levels Decreased SOD, CAT, GPx levels | [186] |
| Area Analyzed | Histological Changes | Author |
|---|---|---|
| Telencephalon | Increased number of blood vessels (some being ectatic) and blood cell infiltration in both treatment groups | [158] |
| Diencephalon and Mesencephalon | Dilation of blood vessels and leukocyte infiltration in both treatment groups; central chromatolysis distinguished in large neurons in the oculomotor nucleus | |
| Rhombencephalon | Mild infiltration and neuronal damage were evident, especially in group of fish exposed to highest concentration | |
| Spinal cord | Only edema of pericardium was observed in some motor neurons, and intense vascularization | |
| Cerebellum | No obvious changes were observed |
| Behavioral Parameters Measured | Dose of Exposure (µg/L) | Results Recorded in Pre-Treatment Group | Results Recorded 2 h PostExposure | Effects | Author |
|---|---|---|---|---|---|
| Total swimming distance | DM 25 | 791.6 ± 264.9 cm | 337.9 ± 218.6 cm | Total swimming distance decreased post-exposure in both experimental groups | [224] |
| DM 12.5 | 721.3 ± 259.7 cm | 251 ± 137 cm | |||
| Variable swim velocity | DM 25 | 3.2 ± 1.2 cm/s | 1.06 ± 0.56 cm/s | Variable swimming speed decreased significantly | |
| DM 12.5 | 3.03 ± 1.07 cm/s | 1.06 ± 0.56 cm/s | |||
| Active swimming | DM 25 | 215.7 ± 35.17 s | 157.17 ± 57.79 s | Zebrafish exposed to these concentrations showed lethargic behavior and became less active | |
| DM 12.5 | 216.3 ± 49.2 s | 159.4 ± 57.7 s | |||
| Counterclockwise rotations | DM 25 | 5.25 ± 4.2 | 1.87 ± 1.12 | Counterclockwise movement decreased significantly | |
| DM 12.5 | 4.73 ± 3.53 | 2 ± 1.6 | |||
| Clockwise rotations | DM 25 | 5.13 ± 2.2 | 3 ± 3 | Clockwise revolutions decreased significantly | |
| DM 12.5 | 6.7 ± 4.3 | 1.6 ± 0.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buzenchi Proca, T.M.; Solcan, C.; Solcan, G. Neurotoxicity of Some Environmental Pollutants to Zebrafish. Life 2024, 14, 640. https://doi.org/10.3390/life14050640
Buzenchi Proca TM, Solcan C, Solcan G. Neurotoxicity of Some Environmental Pollutants to Zebrafish. Life. 2024; 14(5):640. https://doi.org/10.3390/life14050640
Chicago/Turabian StyleBuzenchi Proca, Teodora Maria, Carmen Solcan, and Gheorghe Solcan. 2024. "Neurotoxicity of Some Environmental Pollutants to Zebrafish" Life 14, no. 5: 640. https://doi.org/10.3390/life14050640






