UV Fluorescence-Based Determination of Urinary Advanced Glycation End Products in Patients with Chronic Kidney Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Fluorescence
2.3. Routine Laboratory Parameters
2.4. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Busch, M.; Franke, S.; Ruster, C.; Wolf, G. Advanced glycation end-products and the kidney. Eur. J. Clin. Investig. 2010, 40, 742–755. [Google Scholar] [CrossRef]
- Byun, K.; Yoo, Y.; Son, M.; Lee, J.; Jeong, G.B.; Park, Y.M.; Salekdeh, G.H.; Lee, B. Advanced glycation end-products produced systemically and by macrophages: A common contributor to inflammation and degenerative diseases. Pharmacol. Ther. 2017, 177, 44–55. [Google Scholar] [CrossRef]
- Munch, G.; Keis, R.; Wessels, A.; Riederer, P.; Bahner, U.; Heidland, A.; Niwa, T.; Lemke, H.D.; Schinzel, R. Determination of advanced glycation end products in serum by fluorescence spectroscopy and competitive ELISA. Eur. J. Clin. Chem. Clin. Biochem. 1997, 35, 669–677. [Google Scholar] [CrossRef]
- Schmitt, A.; Schmitt, J.; Münch, G.; Gasic-Milencovic, J. Characterization of advanced glycation end products for biochemical studies: Side chain modifications and fluorescence characteristics. Anal. Biochem. 2005, 338, 201–215. [Google Scholar] [CrossRef]
- Hohmann, C.; Liehr, K.; Henning, C.; Fiedler, R.; Girndt, M.; Gebert, M.; Hulko, M.; Storr, M.; Glomb, M.A. Detection of free advanced glycation end products in vivo during hemodialysis. J. Agric. Food Chem. 2017, 65, 930–937. [Google Scholar] [CrossRef]
- Noordzij, M.J.; Lefrandt, J.D.; Smit, A.J. Advanced glycation end products in renal failure: An overview. J. Ren. Care. 2008, 34, 207–212. [Google Scholar] [CrossRef]
- Delgado-Andrade, C. Carboxymethyl-lysine: Thirty years of investigation in the field of AGE formation. Food Funct. 2016, 7, 46–57. [Google Scholar] [CrossRef]
- Galler, A.; Muller, G.; Schinzel, R.; Kratzsch, J.; Kiess, W.; Munch, G. Impact of metabolic control and serum lipids on the concentration of advanced glycation end products in the serum of children and adolescents with type 1 diabetes, as determined by fluorescence spectroscopy and nepsilon-(carboxymethyl)lysine ELISA. Diabetes Care 2003, 26, 2609–2615. [Google Scholar] [CrossRef] [Green Version]
- Sanaka, T.; Funaki, T.; Tanaka, T.; Hoshi, S.; Niwayama, J.; Taitoh, T.; Nishimura, H.; Higuchi, C. Plasma pentosidine levels measured by a newly developed method using ELISA in patients with chronic renal failure. Nephron 2002, 91, 64–73. [Google Scholar] [CrossRef]
- Hurtado-Sánchez Mdel, C.; Espinosa-Mansilla, A.; Rodríguez-Cáceres, M.I.; Martín-Tornero, E.; Durán-Merás, I. Development of a method for the determination of advanced glycation end products precursors by liquid chromatography and its application in human urine samples. J. Sep. Sci. 2012, 35, 2575–2584. [Google Scholar] [CrossRef]
- Verzijl, N.; De Groot, J.; Thorpe, S.R.; Bank, R.A.; Shaw, J.N.; Lyons, T.J.; Bijlsma, J.W.J.; Lafeberi, F.P.J.G.; Baynes, J.W.; Te Koppele, J.M. Effect of collagen turnover on the accumulation of advanced glycation endproducts. J. Biol. Chem. 2000, 275, 39027–39031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thornalley, P.J.; Battah, S.; Ahmed, N.; Karachalias, N.; Agalou, S.; Babaei-Jadidi, R.; Dawnay, A. Quantitative screening of advanced glycation endproducts in cellular and extracellular proteins by tandem mass spectrometry. Biochem. J. 2003, 375, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Liu, J.; Shi, B.; He, S.; Yao, X.; Willcox, D.M.P. Advanced glycation end product (AGE) modified proteins in tears of diabetic patients. Mol. Vis. 2010, 16, 1576–1584. [Google Scholar] [PubMed]
- Makita, Z.; Radoff, S.; Rayfield, E.J.; Yang, Z.; Skolnik, E.; Delaney, V.; Friedman, E.A.; Cerami, A.; Vlassara, H. Advanced glycosylation end products in patients with diabetic nephropathy. N. Engl. J. Med. 1991, 325, 836–842. [Google Scholar] [CrossRef]
- Yanagisawa, K.; Makita, Z.; Shiroshita, K.; Ueda, T.; Fusegawa, T.; Kuwajima, S.; Takeuchi, M.; Koike, T. Specific fluorescence assay for advanced glycation end products in blood and urine of diabetic patients. Metabolism 1998, 47, 1348–1853. [Google Scholar] [CrossRef]
- Nakamura, K.; Nakazawa, Y.; Ienaga, K. Acid-stable fluorescent advanced glycation end products: Vesperlysines A, B, and C are formed as crosslinked products in the Maillard reaction between lysine or proteins with glucose. Biochem. Biophys. Res. Commun. 1997, 232, 227–230. [Google Scholar] [CrossRef]
- Monnier, V.M.; Kohn, R.R.; Cerami, A. Accelerated age-related browning of human collagen in diabetes mellitus. Proc. Natl. Acad. Sci. USA 1984, 82, 583–587. [Google Scholar] [CrossRef] [Green Version]
- Suehiro, A.; Uchida, K.; Nakanishi, M.; Wakabayashi, I. Measurement of urinary advanced glycation end-products (AGEs) using a fluorescence assay for metabolic syndrome-related screening tests. Diabetes Metab. Syndr. 2016, 10, S110–S113. [Google Scholar] [CrossRef]
- Bucala, R.; Cerami, A. Advanced glycosylation: Chemistry, biology, and implications for diabetes and aging. Adv. Pharmacol. 1992, 23, 1–34. [Google Scholar]
- Forbes, J.M.; Cooper, M.E.; Oldfield, M.D.; Thomas, M.C. Role of advanced glycation end products in diabetic nephropathy. J. Am. Soc. Nephrol. 2003, 14, S254–S258. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, W.T.; Levy, R.I.; Frederickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [PubMed]
- Wagner, Z.; Wittmann, I.; Mazak, I.; Schinzel, R.; Heidland, A.; Kientsch-Engel, R.; Nagy, J. N(epsilon)-(carboxymethyl)lysine levels in patients with type 2 diabetes: Role of renal function. Am. J. Kidney Dis. 2001, 38, 785–791. [Google Scholar] [CrossRef]
- Kuzan, A.; Chwiłkowska1, A.; Maksymowicz, K.; Szydełko-Bronowicka, A.; Stach, K.; Pezowicz, C.; Gamian, A. Advanced glycation end products as a source of artifactsin immunoenzymatic methods. Glycoconj. J. 2018, 35, 95–103. [Google Scholar] [CrossRef] [Green Version]
- Bohlender, J.M.; Franke, S.; Stein, G.; Wolf, G. Advanced glycation end products and the kidney. Am. J. Physiol. Ren. Physiol. 2005, 289, F645–F659. [Google Scholar] [CrossRef]
- Rabbani, N.; Thornalley, P.J. Advanced glycation end products in the pathogenesis of chronic kidney disease. Kidney Int. 2018, 93, 803–813. [Google Scholar] [CrossRef] [Green Version]
- Nishino, T. Immunohistochemical detection of advanced glycosylation end products within the vascular lesions and glomeruli in diabetic nephropathy. Hum. Pathol. 1995, 26, 308–313. [Google Scholar] [CrossRef]
- Sakata, N.; Imanaga, Y.; Meng, J.; Tachikawa, Y.; Takebayashi, S.; Nagai, R.; Horiuchi, S. Increased advanced glycation end products in atherosclerotic lesions of patients with end-stage renal disease. Atherosclerosis 1999, 142, 67–77. [Google Scholar] [CrossRef]
- Stitt, A.W. Elevated AGE-modified ApoB in sera of euglycemic, normolipidemic patients with atherosclerosis: Relationship to tissue AGEs. Mol. Med. 1997, 3, 617–627. [Google Scholar] [CrossRef]
- Li, J.; Hou, F.; Guo, Z.; Shan, Y.; Zhang, X.; Liu, Z. Advanced glycation end products upregulate C-reactive protein synthesis by human hepatocytes through stimulation of monocyte IL-6 and IL-1 beta production. Scand. J. Immunol. 2007, 66, 555–562. [Google Scholar] [CrossRef]
- Chen, J.; Huang, L.; Song, M.; Yu, S.; Gao, P.; Jing, J. C-reactive protein upregulates receptor for advanced glycation end products expression and alters antioxidant defenses in rat endothelial progenitor cells. J. Cardiovasc. Pharmacol. 2009, 53, 359–367. [Google Scholar] [CrossRef]
- Ellulu, M.S.; Patimah, I.; Khaza’ai, H.; Rahmat, A.; Abed, Y. Obesity and inflammation: The linking mechanism and the complications. Arch. Med. Sci. 2017, 13, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Cai, W.; Peppa, M.; Goodman, S.; Ferrucci, L.; Striker, G.; Vlassara, H. Circulating glycotoxins and dietary advanced glycation endproducts: Two links to inflammatory response, oxidative stress, and aging. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 427–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fournet, M.; Bonté, F.; Desmoulière, A. Glycation damage: A possible hub for major pathophysiological disorders and aging. Aging Dis. 2018, 9, 880–900. [Google Scholar] [CrossRef] [Green Version]
- Tsukahara, H.; Sekine, K.; Uchiyama, M.; Kawakami, H.; Hata, I.; Todoroki, Y.; Hiraoka, M.; Kaji, M.; Yorifuji, T.; Momoi, T.; et al. Formation of advanced glycosylation end products and oxidative stress in young patients with type 1 diabetes. Pediatr. Res. 2003, 54, 419–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jud, P.; Sourij, H. Therapeutic options to reduce advanced glycation end products in patients with diabetes mellitus: A review. Diabetes Res. Clin. Pract. 2019, 148, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; He, J.C. The low AGE diet: A neglected aspect of clinical nephrology practice? Nephron 2015, 130, 48–53. [Google Scholar] [CrossRef]
- Tessier, F.J. The Maillard reaction in the human body. The main discoveries and factors that affect glycation. Pathol. Biol. 2010, 58, 214–219. [Google Scholar] [CrossRef]
- Kankova, K. Diabetic threesome (hyperglycaemia, renal function and nutrition) and advanced glycation end products: Evidence for the multiple-hit agent? Proc. Nutr. Soc. 2008, 67, 60–74. [Google Scholar] [CrossRef] [Green Version]
- Ramasamy, R.; Vannucci, S.J.; Yan, S.S.; Herold, K.; Yan, S.F.; Schmidt, A.M. Advanced glycation end products and RAGE: A common thread in aging, diabetes, neurodegeneration, and inflammation. Glycobiology 2005, 15, 16r–28r. [Google Scholar] [CrossRef]
- Uribarri, J.; Peppa, M.; Cai, W.; Goldberg, T.; Lu, M.; He, C.; Vlassara, H. Restriction of dietary glycotoxins reduces excessive advanced glycation end products in renal failure patients. J. Am. Soc. Nephrol. 2003, 14, 728–731. [Google Scholar] [CrossRef] [Green Version]
- Thomas, M.C.; Tsalamandris, C.; MacIsaac, R.; Medley, T.; Kingwell, B.; Cooper, M.E.; Jerums, G. Low-molecular-weight AGEs are associated with GFR and anemia in patients with type 2 diabetes. Kidney Int. 2004, 66, 1167–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uribarri, J.; Cai, W.; Sandu, O.; Peppa, M.; Goldberg, T.; Vlassara, H. Diet-derived advanced glycation end products are major contributors to the body’s AGE pool and induce inflammation in healthy subjects. Ann. N. Y. Acad. Sci. 2005, 1043, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Henle, T.; Deppisch, R.; Beck, W.; Hergesell, O.; Hansch, G.M.; Ritz, E. Advanced glycated end-products (AGE) during haemodialysis treatment: Discrepant results with different methodologies reflecting the heterogeneity of AGE compounds. Nephrol. Dial. Transplant. 1999, 14, 1968–1975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sivabalan, S.; Vedeswari, C.P.; Jayachandran, S.; Koteeswaran, D.; Pravda, C.; Aruna, P.R.; Ganesan, S. In vivo native fluorescence spectroscopy and nicotinamide adinine dinucleotide/flavin adenine dinucleotide reduction and oxidation states of oral submucous fibrosis for chemopreventive drug monitoring. J. Biomed. Opt. 2010, 15, 017010. [Google Scholar] [CrossRef] [PubMed]
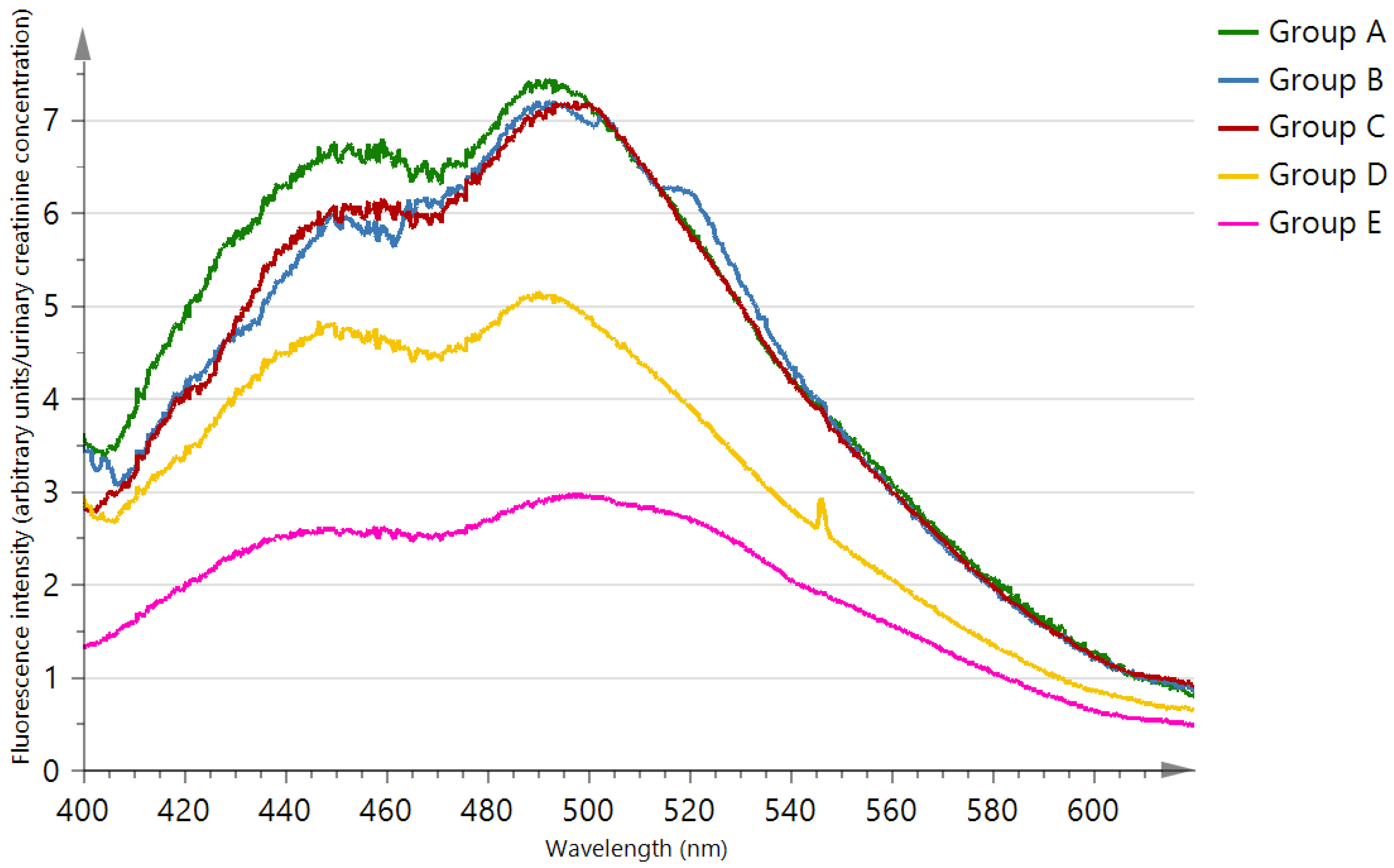

| Characteristics | Chronic Kidney Disease Patients | Healthy Subjects | p-Value | |||
|---|---|---|---|---|---|---|
| Group A | Group B | Group C | Group D | |||
| N | 46 | 27 | 45 | 46 | 31 | |
| Male/Female | 28/18 | 16/11 | 29/16 | 27/19 | 13/18 | |
| Age (years) | 68.5 (63–78) | 72 (66.5–79.8) | 68 (62–77.3) | 70 (64–77) | 30 (25–53.5) | <0.0001 |
| Diabetes mellitus | + | + | - | - | - | |
| Proteinuria | + | - | + | - | - | |
| eGFR (mL/min/1.73 m2) | 37.4 (29.4–42.3) | 33.0 (26.0–39.9) | 36.2 (29.0–46.7) | 41.2 (28.7–51.7) | >90 | <0.0001 |
| BMI (kg/m2) | 30.7 ± 4.8 | 30.8 ± 4.8 | 28.3 ± 4.2 | 27.4 ± 4.1 | 22.4 ± 3.3 | <0.0001 |
| Dependent Variable:Fluorescence Intensity | ||||
|---|---|---|---|---|
| Ln(440 nm) | Ln(490 nm) | |||
| Parameter | Correlation Coefficient | p-Value | Correlation Coefficient | p-Value |
| Age | 0.3930 | <0.0001 | 0.3806 | <0.0001 |
| BMI | 0.1418 | 0.0480 | 0.1181 | N.S. |
| Smoking | 0.1728 | 0.0171 | 0.1938 | 0.0074 |
| Ln(eGFR) | −0.3767 | <0.0001 | −0.3472 | <0.0001 |
| Ln(Urea) | 0.1822 | 0.0199 | 0.1453 | N.S. |
| Ln(CRP) | 0.2416 | 0.0008 | 0.2210 | 0.0023 |
| Hemoglobin | −0.1617 | 0.0392 | −0.1489 | N.S. |
| Treatment | ||||
| Aspirin | 0.1840 | 0.0110 | 0.1582 | 0.0293 |
| Peroral anticoagulant | 0.1711 | 0.0183 | 0.1464 | 0.0439 |
| ACE inhibitor or ARB | 0.1541 | 0.0337 | 0.1401 | N.S. |
| Beta blocker | 0.2023 | 0.0051 | 0.1724 | 0.0174 |
| Nondihydropyridine calcium | 0.1834 | 0.0113 | 0.1524 | 0.0358 |
| channel blocker | ||||
| Loop diuretic | 0.2570 | 0.0003 | 0.2243 | 0.0019 |
| Statin | 0.2053 | 0.0045 | 0.1990 | 0.0059 |
| Insulin | 0.2793 | 0.0001 | 0.2386 | 0.0009 |
| Vitamin D | 0.1505 | 0.0382 | 0.1219 | N.S. |
| Dependent Variable | Independent Variable | β (SE) | p-Value |
|---|---|---|---|
| Ln(Fluorescence intensity at emission wavelength 440 nm) R2 = 0.1970, p < 0.001 | Age (years) | 0.0107 (0.0046) | 0.0206 |
| Ln(eGFR) (mL/min/1.73 m2) | −0.2565 (0.1429) | 0.0743 | |
| Ln(CRP) (mg/L) | 0.1346 (0.0593) | 0.0245 | |
| Insulin treatment | 0.2798 (0.0844) | 0.0011 | |
| Ln(Fluorescence intensity at emission wavelength 490 nm) R2 = 0.1467, p < 0.001 | Age (years) | 0.0155 (0.0040) | 0.0001 |
| Ln(CRP) (mg/L) | 0.1166 (0.0632) | 0.0667 | |
| Insulin treatment | 0.2664 (0.0880) | 0.0028 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steenbeke, M.; De Bruyne, S.; Van Aken, E.; Glorieux, G.; Van Biesen, W.; Himpe, J.; De Meester, G.; Speeckaert, M.; Delanghe, J. UV Fluorescence-Based Determination of Urinary Advanced Glycation End Products in Patients with Chronic Kidney Disease. Diagnostics 2020, 10, 34. https://doi.org/10.3390/diagnostics10010034
Steenbeke M, De Bruyne S, Van Aken E, Glorieux G, Van Biesen W, Himpe J, De Meester G, Speeckaert M, Delanghe J. UV Fluorescence-Based Determination of Urinary Advanced Glycation End Products in Patients with Chronic Kidney Disease. Diagnostics. 2020; 10(1):34. https://doi.org/10.3390/diagnostics10010034
Chicago/Turabian StyleSteenbeke, Mieke, Sander De Bruyne, Elisabeth Van Aken, Griet Glorieux, Wim Van Biesen, Jonas Himpe, Gilles De Meester, Marijn Speeckaert, and Joris Delanghe. 2020. "UV Fluorescence-Based Determination of Urinary Advanced Glycation End Products in Patients with Chronic Kidney Disease" Diagnostics 10, no. 1: 34. https://doi.org/10.3390/diagnostics10010034








