Chromatographic Detection of 8-Hydroxy-2′-Deoxyguanosine in Leukocytes of Asbestos Exposed Workers for Assessing Past and Recent Carcinogen Exposures
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Population
2.2. DNA Extraction and Purification
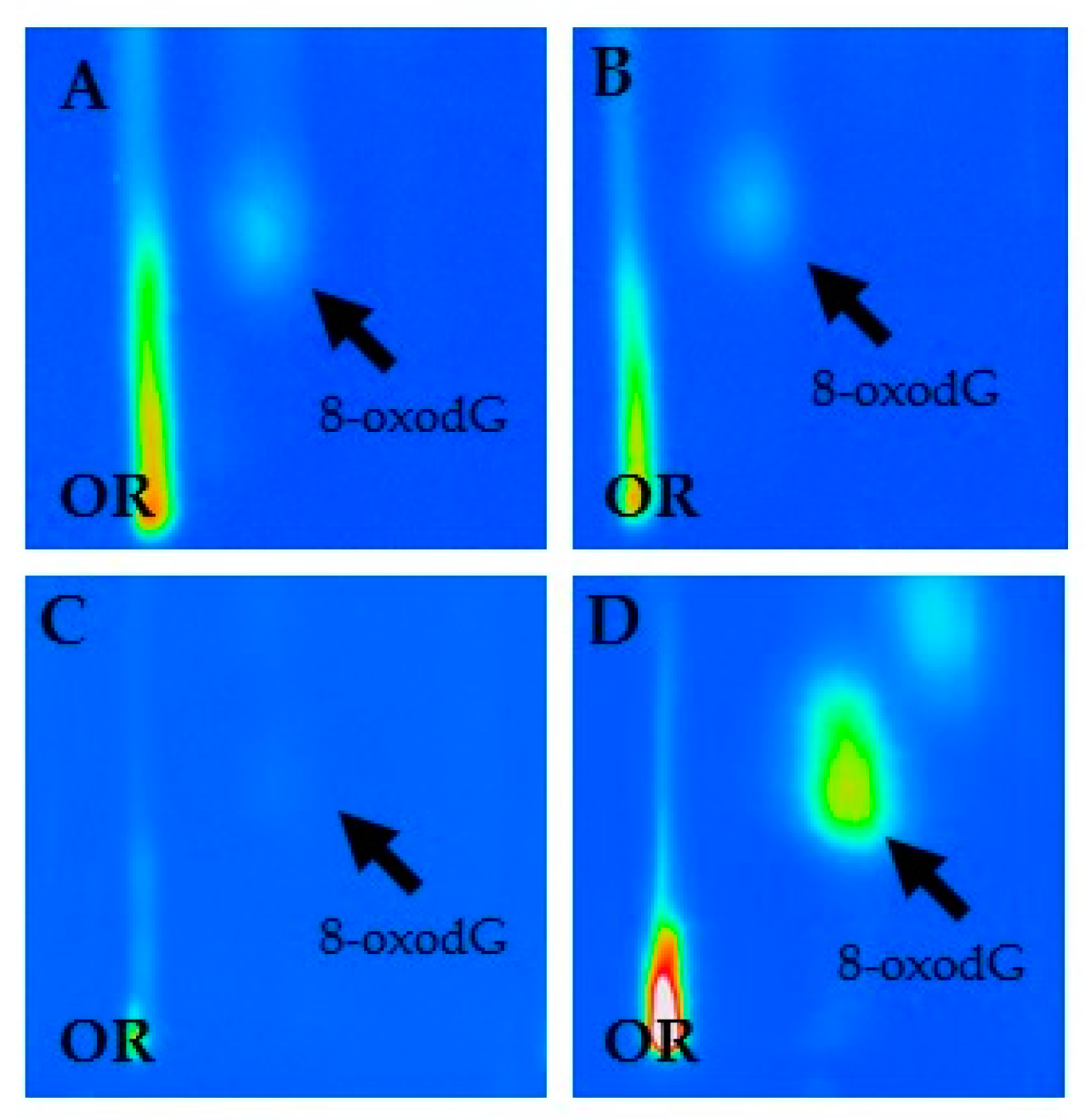
2.3. 32P-DNA Postlabeling
2.4. Statistical Analysis
3. Results
3.1. Demographic Variables
3.2. 8-OxodG and Asbestos Workers
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Afaghi, A.; Oryan, S.; Rahzani, K.; Abdollahi, M. Study on genotoxicity, oxidative stress biomarkers and clinical symptoms in workers of an asbestos-cement factory. Excli. J. 2015, 14, 1067–1077. [Google Scholar]
- Knox, J.F.; Holmes, S.; Doll, R.; Hill, I.D. Mortality from lung cancer and other causes among workers in an asbestos textile factory. Br. J. Ind. Med. 1968, 25, 293–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IARC. Overall evaluations of carcinogenicity: An updating of IARC Monographs volumes 1 to 42. IARC Monogr. Eval. Carcinog. Risk Chem. Hum. Suppl. 1987, 7, 1–440. [Google Scholar]
- Case, B.W.; Abraham, J.L.; Meeker, G.; Pooley, F.D.; Pinkerton, K.E. Applying definitions of “asbestos” to environmental and “low-dose” exposure levels and health effects, particularly malignant mesothelioma. J. Toxicol. Environ. Health Part B 2011, 14, 3–39. [Google Scholar] [CrossRef] [Green Version]
- Mossman, B.T.; Lippmann, M.; Hesterberg, T.W.; Kelsey, K.T.; Barchowsky, A.; Bonner, J.C. Pulmonary endpoints (lung carcinomas and asbestosis) following inhalation exposure to asbestos. J. Toxicol. Environ. Health Part B 2011, 14, 76–121. [Google Scholar] [CrossRef] [PubMed]
- Toyokuni, S. Iron addiction with ferroptosis-resistance in asbestos-induced mesothelial carcinogenesis: Toward the era of mesothelioma prevention. Free Radic. Biol. Med. 2019, 133, 206–215. [Google Scholar] [CrossRef]
- Park, J.-H.; Gelhaus, S.; Vedantam, S.; Oliva, A.L.; Batra, A.; Blair, I.A.; Troxel, A.B.; Field, J.; Penning, T.M. The pattern of p53 mutations caused by PAH o-quinones is driven by 8-oxo-dGuo formation while the spectrum of mutations is determined by biological selection for dominance. Chem. Res. Toxicol. 2008, 21, 1039–1049. [Google Scholar] [CrossRef]
- Unfried, K.; Schürkes, C.; Abel, J. Distinct Spectrum of Mutations Induced by Crocidolite Asbestos. Cancer Res. 2002, 62, 99. [Google Scholar]
- Loft, S.; Høgh Danielsen, P.; Mikkelsen, L.; Risom, L.; Forchhammer, L.; Møller, P. Biomarkers of oxidative damage to DNA and repair. Biochem. Soc. Trans. 2008, 36, 1071–1076. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-hydroxy-2′ -deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health. Part CEnviron. Carcinog. Ecotoxicol. Rev. 2009, 27, 120–139. [Google Scholar] [CrossRef] [Green Version]
- Brancato, B.; Munnia, A.; Cellai, F.; Ceni, E.; Mello, T.; Bianchi, S.; Catarzi, S.; Risso, G.G.; Galli, A.; Peluso, M.E. 8-Oxo-7, 8-dihydro-2-deoxyguanosine and other lesions along the coding strand of the exon 5 of the tumour suppressor gene P53 in a breast cancer case-control study. DNA Res. Int. J. Rapid Publ. Rep. Genes Genomes 2016, 23, 395–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, A.; Gulumian, M.; Hei, T.K.; Kamp, D.; Rahman, Q.; Mossman, B.T. Multiple roles of oxidants in the pathogenesis of asbestos-induced diseases. Free Radic. Biol. Med. 2003, 34, 1117–1129. [Google Scholar] [CrossRef]
- Huang, S.X.L.; Jaurand, M.-C.; Kamp, D.W.; Whysner, J.; Hei, T.K. Role of mutagenicity in asbestos fiber-induced carcinogenicity and other diseases. J. Toxicol. Environ. Health Part B Crit. Rev. 2011, 14, 179–245. [Google Scholar] [CrossRef] [PubMed]
- Marini, V.; Michelazzi, L.; Cioe, A.; Fucile, C.; Spigno, F.; Robbiano, L. Exposure to asbestos: Correlation between blood levels of mesothelin and frequency of micronuclei in peripheral blood lymphocytes. Mutat Res. 2011, 721, 114–117. [Google Scholar] [CrossRef]
- Zhao, X.H.; Jia, G.; Liu, Y.Q.; Liu, S.W.; Yan, L.; Jin, Y.; Liu, N. Association between polymorphisms of DNA repair gene XRCC1 and DNA damage in asbestos-exposed workers. Biomed. Environ. Sci. BES 2006, 19, 232–238. [Google Scholar]
- Govercin, M.; Tomatir, A.G.; Evyapan, F.; Acikbas, I.; Coskun, G.; Akdag, B. Elevated micronucleus frequencies in patients with pleural plaque secondary to environmental exposure to asbestos. Genet. Mol. Res. GMR 2014, 13, 598–604. [Google Scholar] [CrossRef]
- Marczynski, B.; Rozynek, P.; Kraus, T.; Schlosser, S.; Raithel, H.J.; Baur, X. Levels of 8-hydroxy-2′-deoxyguanosine in DNA of white blood cells from workers highly exposed to asbestos in Germany. Mutat. Res. 2000, 468, 195–202. [Google Scholar] [CrossRef]
- Tagesson, C.; Chabiuk, D.; Axelson, O.; Baranski, B.; Palus, J.; Wyszynska, K. Increased urinary excretion of the oxidative DNA adduct, 8-hydroxydeoxyguanosine, as a possible early indicator of occupational cancer hazards in the asbestos, rubber, and azo-dye industries. Pol. J. Occup. Med. Environ. Health 1993, 6, 357–368. [Google Scholar]
- Yoshida, R.; Ogawa, Y.; Shioji, I.; Yu, X.; Shibata, E.; Mori, I.; Kubota, H.; Kishida, A.; Hisanaga, N. Urinary 8-oxo-7, 8-dihydro-2′-deoxyguanosine and biopyrrins levels among construction workers with asbestos exposure history. Ind. Health 2001, 39, 186–188. [Google Scholar] [CrossRef]
- Vainio, H.U.; Oksa, P.; Tuomi, T.; Vehmas, T.; Wolff, H.J. Helsinki Criteria update 2014: Asbestos continues to be a challenge for disease prevention and attribution. Epidemiol. Prev. 2016, 40 (Suppl. 1), 15–19. [Google Scholar]
- Betti, M.; Casalone, E.; Ferrante, D.; Aspesi, A.; Morleo, G.; Biasi, A.; Sculco, M.; Mancuso, G.; Guarrera, S.; Righi, L.; et al. Germline mutations in DNA repair genes predispose asbestos-exposed patients to malignant pleural mesothelioma. Cancer Lett. 2017, 405, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Marinaccio, A.; Corfiati, M.; Binazzi, A.; Di Marzio, D.; Scarselli, A.; Ferrante, P.; Bonafede, M.; Verardo, M.; Mirabelli, D.; Gennaro, V.; et al. The epidemiology of malignant mesothelioma in women: Gender differences and modalities of asbestos exposure. Occup. Environ. Med. 2018, 75, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Mohamadkhani, A.; Pourshams, A.; Viti, J.; Cellai, F.; Mortazavi, K.; Sharafkhah, M.; Sotoudeh, M.; MalekZadeh, R.; Boffetta, P.; Peluso, M. Pancreatic cancer patients are prone to the higher oxidative DNA damage of peripheral leukocytes. Asian. Pac. J. Cancer Prev. 2017. [Google Scholar]
- Cristaudo, A.; Foddis, R.; Guglielmi, G. Methodology and results of an experience of medical surveillance of people previously exposed to asbestos in Tuscany. G. Ital. di Med. del Lav. ed Ergon. 2010, 32, 385–388. [Google Scholar]
- Zanoni, T.B.; Hudari, F.; Munnia, A.; Peluso, M.; Godschalk, R.W.; Zanoni, M.V.; den Hartog, G.J.; Bast, A.; Barros, S.B.; Maria-Engler, S.S.; et al. The oxidation of p-phenylenediamine, an ingredient used for permanent hair dyeing purposes, leads to the formation of hydroxyl radicals: Oxidative stress and DNA damage in human immortalized keratinocytes. Toxicol. Lett. 2015, 239, 194–204. [Google Scholar] [CrossRef]
- Godschalk, R.; Remels, A.; Hoogendoorn, C.; van Benthem, J.; Luijten, M.; Duale, N.; Brunborg, G.; Olsen, A.-K.; Bouwman, F.G.; Munnia, A.; et al. Paternal Exposure to Environmental Chemical Stress Affects Male Offspringns Hepatic Mitochondria. Toxicol. Sci. 2018, 162, 241–250. [Google Scholar] [CrossRef] [Green Version]
- Munnia, A.; Saletta, F.; Allione, A.; Piro, S.; Confortini, M.; Matullo, G.; Peluso, M. 32P-Post-labelling method improvements for aromatic compound-related molecular epidemiology studies. Mutagenesis 2007, 22, 381–385. [Google Scholar] [CrossRef] [Green Version]
- Van Houwelingen, H.C.; Arends, L.R.; Stijnen, T. Advanced methods in meta-analysis: Multivariate approach and meta-regression. Stat. Med. 2002, 21, 589–624. [Google Scholar] [CrossRef]
- Izzotti, A.; De Flora, S.; Cartiglia, C.; Are, B.M.; Longobardi, M.; Camoirano, A.; Mura, I.; Dore, M.P.; Scanu, A.M.; Rocca, P.C.; et al. Interplay between Helicobacter pylori and host gene polymorphisms in inducing oxidative DNA damage in the gastric mucosa. Carcinogenesis 2007, 28, 892–898. [Google Scholar] [CrossRef] [Green Version]
- Izzotti, A.; Cartiglia, C.; De Flora, S.; Sacca, S. Methodology for evaluating oxidative DNA damage and metabolic genotypes in human trabecular meshwork. Toxicol. Mech. Methods 2003, 13, 161–168. [Google Scholar] [CrossRef]
- Izzotti, A.; Balansky, R.M.; Dagostini, F.; Bennicelli, C.; Myers, S.R.; Grubbs, C.J.; Lubet, R.A.; Kelloff, G.J.; De Flora, S. Modulation of biomarkers by chemopreventive agents in smoke-exposed rats. Cancer Res. 2001, 61, 2472–2479. [Google Scholar] [PubMed]
- Balansky, R.; Izzotti, A.; D’Agostini, F.; Longobardi, M.; Micale, R.T.; La Maestra, S.; Camoirano, A.; Ganchev, G.; Iltcheva, M.; Steele, V.E.; et al. Assay of lapatinib in murine models of cigarette smoke carcinogenesis. Carcinogenesis 2014, 35, 2300–2307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Micale, R.T.; La Maestra, S.; Di Pietro, A.; Visalli, G.; Baluce, B.; Balansky, R.; Steele, V.E.; De Flora, S. Oxidative stress in the lung of mice exposed to cigarette smoke either early in life or in adulthood. Arch. Toxicol. 2013, 87, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Devanaboyina, U.; Gupta, R.C. Sensitive detection of 8-hydroxy-2′deoxyguanosine in DNA by 32P-postlabeling assay and the basal levels in rat tissues. Carcinogenesis 1996, 17, 917–924. [Google Scholar] [CrossRef] [Green Version]
- IARC. Biological agents. Volume 100 B. A review of human carcinogens. Iarc. Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 1–441. [Google Scholar]
- Sorensen, M.; Autrup, H.; Moller, P.; Hertel, O.; Jensen, S.S.; Vinzents, P.; Knudsen, L.E.; Loft, S. Linking exposure to environmental pollutants with biological effects. Mutat. Res. 2003, 544, 255–271. [Google Scholar] [CrossRef]
- Bonassi, S.; Cellai, F.; Munnia, A.; Ugolini, D.; Cristaudo, A.; Neri, M.; Milic, M.; Bonotti, A.; Giese, R.W.; Peluso, M.E. 3-(2-deoxy-β-d-erythro-pentafuranosyl) pyrimido [1, 2-α] purin-10 (3H)-one deoxyguanosine adducts of workers exposed to asbestos fibers. Toxic. Lett. 2017, 270, 1–7. [Google Scholar] [CrossRef]
- Svecova, V.; Topinka, J.; Solansky, I.; Sram, R.J. Personal exposure to volatile organic compounds in the Czech Republic. J. Expo. Sci. Environ. Epidemiol. 2012, 22, 455–460. [Google Scholar] [CrossRef] [Green Version]
- Peluso, M.E.; Munnia, A.; Giese, R.W.; Chellini, E.; Ceppi, M.; Capacci, F. Oxidatively damaged DNA in the nasal epithelium of workers occupationally exposed to silica dust in Tuscany region, Italy. Mutagenesis 2015, 30, 519–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossnerova, A.; Spatova, M.; Rossner, P.; Solansky, I.; Sram, R.J. The impact of air pollution on the levels of micronuclei measured by automated image analysis. Mutat. Res. 2009, 669, 42–47. [Google Scholar] [CrossRef]
- Topinka, J.; Sevastyanova, O.; Binkova, B.; Chvatalova, I.; Milcova, A.; Lnenickova, Z.; Novakova, Z.; Solansky, I.; Sram, R.J. Biomarkers of air pollution exposure—A study of policemen in Prague. Mutat. Res. 2007, 624, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Toyokuni, S. Mechanisms of asbestos-induced carcinogenesis. Nagoya. J. Med. Sci. 2009, 71, 1–10. [Google Scholar]
- Ceppi, M.; Munnia, A.; Cellai, F.; Bruzzone, M.; Peluso, M.E.M. Linking the generation of DNA adducts to lung cancer. Toxic In Vitro 2017, 390, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Milić, M.; Neri, M.; Ceppi, M.; Bruzzone, M.; Munnia, A.; Ugolini, D.; Cristaudo, A.; Bonotti, A.; Peluso, M.E.; Bonassi, S. DNA damage and genomic instability among workers formerly and currently exposed to asbestos. Scand. J. Work Environ. Health 2018. [Google Scholar]
- Ghio, A.J.; Churg, A.; Roggli, V.L. Ferruginous bodies: Implications in the mechanism of fiber and particle toxicity. Toxicol. Pathol. 2004, 32, 643–649. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, D.; Dunnigan, J.; Hesterberg, T.; Brown, R.; Velasco, J.A.; Barrera, R.; Hoskins, J.; Gibbs, A. Health risk of chrysotile revisited. Crit. Rev. Toxicol. 2013, 43, 154–183. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Liu, T.; Kamp, D.W.; Wang, Y.; He, H.; Zhou, X.; Li, D.; Yang, L.; Zhao, B.; Liu, G. AKT/mTOR and c-Jun N-terminal kinase signaling pathways are required for chrysotile asbestos-induced autophagy. Free Radic. Biol. Med. 2014, 72, 296–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, B.; Gogol, M.; Gaudenz, K.; Gerton, J.L. Improved transcription and translation with L-leucine stimulation of mTORC1 in Roberts syndrome. BMC Genom. 2016, 17, 25. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Liu, T.; Kamp, D.W.; Lin, Z.; Wang, Y.; Li, D.; Yang, L.; He, H.; Liu, G. The c-Jun N-terminal kinase signaling pathway mediates chrysotile asbestos-induced alveolar epithelial cell apoptosis. Mol. Med. Rep. 2015, 11, 3626–3634. [Google Scholar] [CrossRef] [Green Version]
- Peluso, M.; Munnia, A.; Ceppi, M.; Giese, R.W.; Catelan, D.; Rusconi, F.; Godschalk, R.W.; Biggeri, A. Malondialdehyde-deoxyguanosine and bulky DNA adducts in schoolchildren resident in the proximity of the Sarroch industrial estate on Sardinia Island, Italy. Mutagenesis 2013, 28, 315–321. [Google Scholar] [CrossRef] [Green Version]
- Nie, B.; Gan, W.; Shi, F.; Hu, G.-X.; Chen, L.-G.; Hayakawa, H.; Sekiguchi, M.; Cai, J.-P. Age-dependent accumulation of 8-oxoguanine in the DNA and RNA in various rat tissues. Oxidative Med. Cell. Longev. 2013, 2013, 303181. [Google Scholar] [CrossRef] [Green Version]
- Lodovici, M.; Caldini, S.; Luceri, C.; Bambi, F.; Boddi, V.; Dolara, P. Active and Passive Smoking and Lifestyle Determinants of 8-Oxo-7,8-Dihydro-2′-Deoxyguanosine Levels in Human Leukocyte DNA. Cancer Epidemiol. Prev. Biomark. 2005, 14, 2975. [Google Scholar] [CrossRef] [Green Version]
- Pilger, A.; Radiger, H.W. 8-Hydroxy-2-deoxyguanosine as a marker of oxidative DNA damage related to occupational and environmental exposures. Int. Arch. Occup. Environ. Health 2006, 80, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Risom, L.; Dybdahl, M.; Bornholdt, J.; Vogel, U.; Wallin, H.k.; Møller, P.; Loft, S. Oxidative DNA damage and defence gene expression in the mouse lung after short-term exposure to diesel exhaust particles by inhalation. Carcinogenesis 2003, 24, 1847–1852. [Google Scholar] [CrossRef] [Green Version]
- Jacob, K.D.; Noren Hooten, N.; Trzeciak, A.R.; Evans, M.K. Markers of oxidant stress that are clinically relevant in aging and age-related disease. Mech. Ageing Dev. 2013, 134, 139–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rundle, A.; Richards, C.; Neslund-Dudas, C.; Tang, D.; Rybicki, B.A. Neighborhood socioeconomic status modifies the association between individual smoking status and PAH-DNA adduct levels in prostate tissue. Environ. Mol. Mutagenesis 2012, 53, 384–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korkmaz, K.S.; Butuner, B.D.; Roggenbuck, D. Detection of 8-OHdG as a diagnostic biomarker. J. Lab. Precis. Med. 2018, 3. [Google Scholar] [CrossRef]
- Bolognesi, C.; Migliore, L.; Lista, F.; Caroli, S.; Patriarca, M.; De Angelis, R.; Capocaccia, R.; Amadori, S.; Pulliero, A.; Balia, C.; et al. Biological monitoring of Italian soldiers deployed in Iraq. Results of the SIGNUM project. Int. J. Hyg. Environ. Health 2016, 219, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Balansky, R.; Micale, R.T.; Pulliero, A.; La Maestra, S.; De Flora, S. Modulation of smoke-induced DNA and microRNA alterations in mouse lung by licofelone, a triple COX-1, COX-2 and 5-LOX inhibitor. Carcinogenesis 2020, 41, 91–99. [Google Scholar] [CrossRef] [Green Version]
- La Maestra, S.; D’Agostini, F.; Izzotti, A.; Micale, R.T.; Mastracci, L.; Camoirano, A.; Balansky, R.; Trosko, J.E.; Steele, V.E.; De Flora, S. Modulation by aspirin and naproxen of nucleotide alterations and tumors in the lung of mice exposed to environmental cigarette smoke since birth. Carcinogenesis 2015, 36, 1531–1538. [Google Scholar] [CrossRef] [Green Version]
- Peluso, M.; Bollati, V.; Munnia, A.; Srivatanakul, P.; Jedpiyawongse, A.; Sangrajrang, S.; Piro, S.; Ceppi, M.; Bertazzi, P.A.; Boffetta, P.; et al. DNA methylation differences in exposed workers and nearby residents of the Ma Ta Phut industrial estate, Rayong, Thailand. Int. J. Epidemiol. 2012, 41, 1753–1760. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peluso, M.E.; Munnia, A.; Bollati, V.; Srivatanakul, P.; Jedpiyawongse, A.; Sangrajrang, S.; Ceppi, M.; Giese, R.W.; Boffetta, P.; Baccarelli, A.A. Aberrant methylation of hypermethylated-in-cancer-1 and exocyclic DNA adducts in tobacco smokers. Toxicol. Sci. Off. J. Soc. Toxicol. 2014, 137, 47–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| 8-OxodG per 105 Deoxyguanosine (dG) | ||||
|---|---|---|---|---|
| N a | Mean ± Standard Error (SE) | MR, 95% C.I. | p-Values b | |
| Age (years) | ||||
| ≤52 | 67 | 2.3 ± 0.2 | Reference | |
| 53–58 | 63 | 2.1 ± 0.2 | 1.00 (0.77–1.29) | 0.978 |
| ≥59 | 55 | 2.1 ± 0.2 | 0.89 (0.68–1.17) | 0.408 |
| Educational level | ||||
| Primary education | 86 | 2.3 ± 0.2 | Reference | |
| Secondary education | 72 | 2.3 ± 0.2 | 0.84 (0.67–1.06) | 0.139 |
| University | 27 | 1.3 ± 0.2 | 0.65 (0.48–0.88) | 0.006 |
| Smoking status | ||||
| Non-smokers | 72 | 2.2 ± 0.2 | Reference | |
| Ex-smokers | 66 | 2.0 ± 0.2 | 0.98 (0.67–1.43) | 0.918 |
| Smokers | 46 | 2.4 ± 0.3 | 1.09 (0.74–1.60) | 0.668 |
| 8-OxodG per 105 Deoxyguanosine (dG) | ||||
|---|---|---|---|---|
| N a | Mean ± Standard Error (SE) | MR, 95% C.I. | p-Values b | |
| Asbestos exposure | ||||
| Unexposed controls | 87 | 1.3 ± 0.1 | Reference | |
| Asbestos workers | 98 | 3.0 ± 0.2 | 2.36 (1.99–2.81) | <0.001 |
| Current and past exposure | ||||
| Unexposed controls | 87 | 1.3 ± 0.1 | Reference | |
| Former asbestos workers | 59 | 2.9 ± 0.2 | 2.32 (1.88–2.86) | <0.001 |
| Current asbestos workers | 39 | 3.1 ± 0.3 | 2.43 (1.90–3.12) | <0.001 |
| ≤4 years asbestos workers | 18 | 2.8 ± 0.4 | 2.21 (1.64–2.99) | <0.001 |
| 5–9 years asbestos workers | 21 | 2.9 ± 0.3 | 2.40 (1.79–3.23) | <0.001 |
| ≥10 years asbestos workers | 16 | 3.5 ± 0.5 | 2.94 (2.11–4.11) | <0.001 |
| Occupational history | ||||
| ≤4 years | 18 | 2.8 ± 0.4 | Reference | |
| 5–9 years | 21 | 2.9 ± 0.3 | 1.09 (0.73–1.64) | 0.672 |
| ≥10 years | 16 | 3.5 ± 0.5 | 1.34 (0.85–2.10) | 0.197 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cellai, F.; Bonassi, S.; Cristaudo, A.; Bonotti, A.; Neri, M.; Ceppi, M.; Bruzzone, M.; Milić, M.; Munnia, A.; Peluso, M. Chromatographic Detection of 8-Hydroxy-2′-Deoxyguanosine in Leukocytes of Asbestos Exposed Workers for Assessing Past and Recent Carcinogen Exposures. Diagnostics 2020, 10, 239. https://doi.org/10.3390/diagnostics10040239
Cellai F, Bonassi S, Cristaudo A, Bonotti A, Neri M, Ceppi M, Bruzzone M, Milić M, Munnia A, Peluso M. Chromatographic Detection of 8-Hydroxy-2′-Deoxyguanosine in Leukocytes of Asbestos Exposed Workers for Assessing Past and Recent Carcinogen Exposures. Diagnostics. 2020; 10(4):239. https://doi.org/10.3390/diagnostics10040239
Chicago/Turabian StyleCellai, Filippo, Stefano Bonassi, Alfonso Cristaudo, Alessandra Bonotti, Monica Neri, Marcello Ceppi, Marco Bruzzone, Mirta Milić, Armelle Munnia, and Marco Peluso. 2020. "Chromatographic Detection of 8-Hydroxy-2′-Deoxyguanosine in Leukocytes of Asbestos Exposed Workers for Assessing Past and Recent Carcinogen Exposures" Diagnostics 10, no. 4: 239. https://doi.org/10.3390/diagnostics10040239







