[68Ga]Ga-NODAGA-E[(cRGDyK)]2 Angiogenesis PET/MR in a Porcine Model of Chronic Myocardial Infarction
Abstract
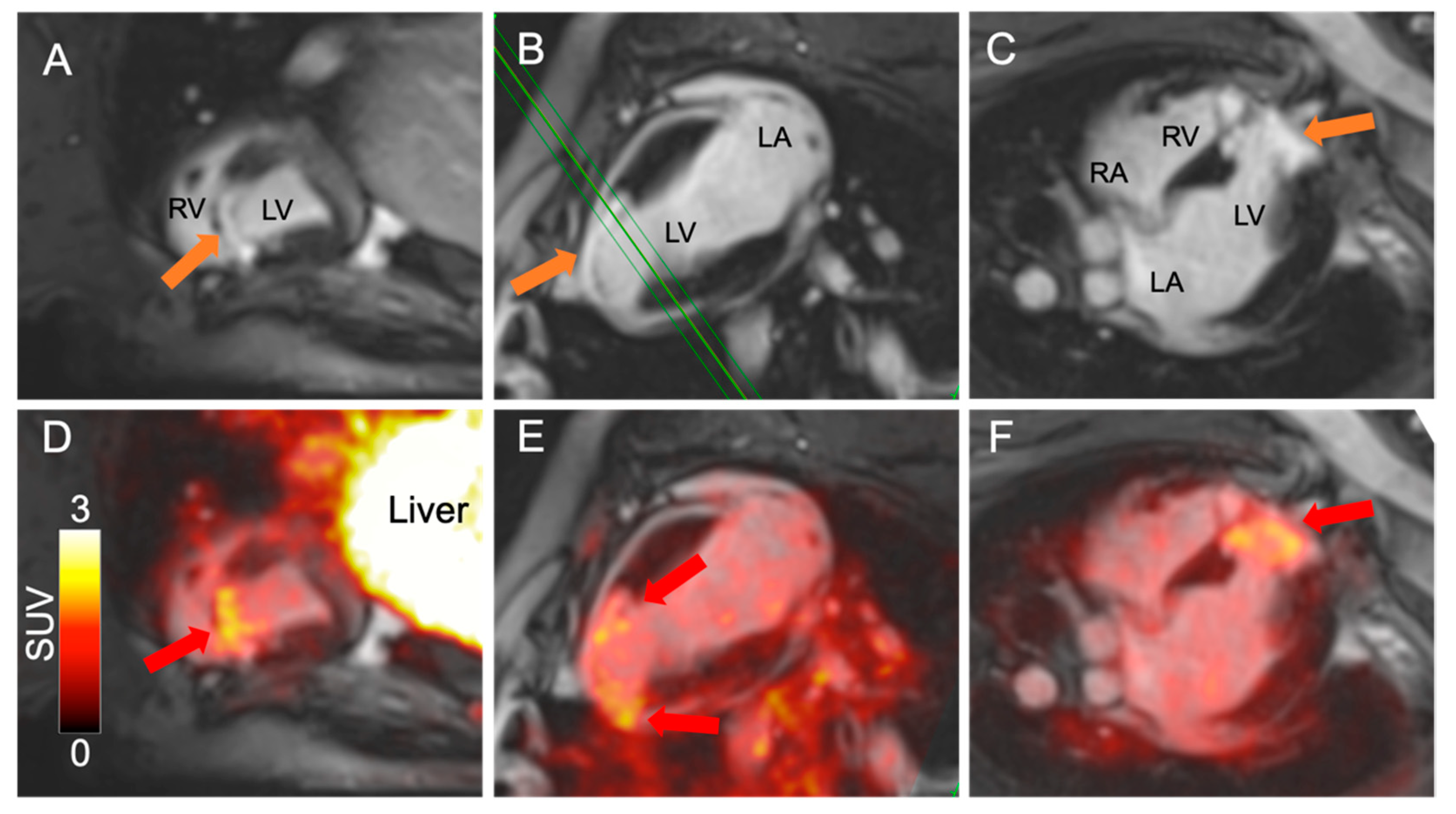
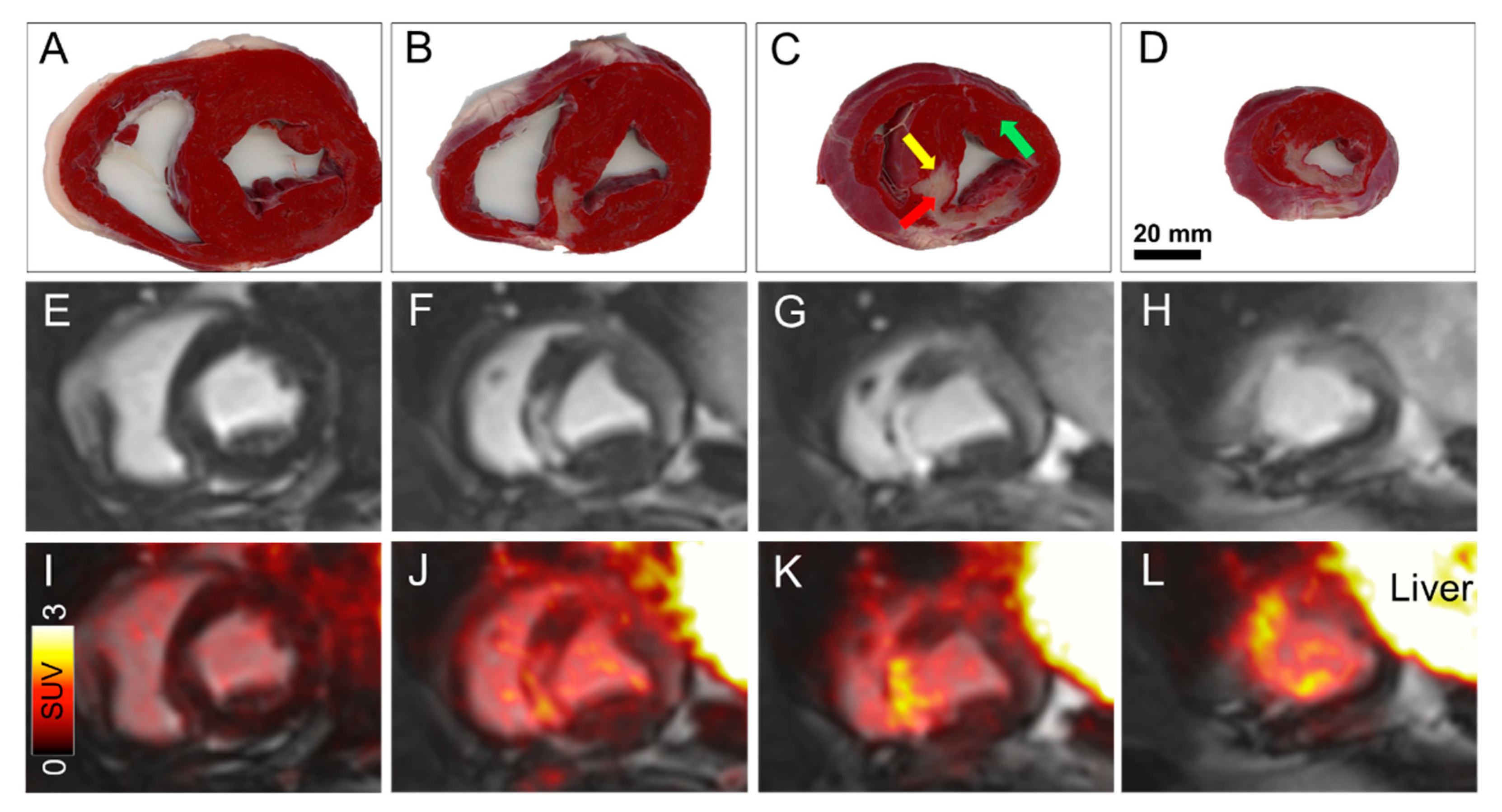
:
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dobrucki, L.W.; Sinusas, A.J. Imaging angiogenesis. Curr. Opin. Biotechnol. 2007, 18, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Kiugel, M.; Dijkgraaf, I.; Kytö, V.; Helin, S.; Liljenbäck, H.; Saanijoki, T.; Yim, C.-B.; Oikonen, V.; Saukko, P.; Knuuti, G.; et al. Dimeric [(68)Ga]DOTA-RGD peptide targeting alphavbeta3 integrin reveals extracellular matrix alterations after myocardial infarction. Mol. Imaging Biol. 2014, 16, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Schuleri, K.H.; Boyle, A.; Centola, M.; Amado, L.C.; Evers, R.; Zimmet, J.M.; Evers, K.S.; Ostbye, K.M.; Scorpio, D.G.; Hare, J.M.; et al. The Adult Gottingen Minipig as a Model for Chronic Heart Failure After Myocardial Infarction: Focus on Cardiovascular Imaging and Regenerative Therapies. Comp. Med. 2008, 58, 568–579. [Google Scholar] [PubMed]
- Chen, H.; Niu, G.; Wu, H.; Chen, X. Clinical Application of Radiolabeled RGD Peptides for PET Imaging of Integrin alphavbeta3. Theranostics 2016, 6, 78–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oxboel, J.; Schjoeth-Eskesen, C.; El-Ali, H.H.; Madsen, J.; Kjaer, A. (64)Cu-NODAGA-c(RGDyK) Is a Promising New Angiogenesis PET Tracer: Correlation between Tumor Uptake and Integrin alpha(V)beta(3) Expression in Human Neuroendocrine Tumor Xenografts. Int. J. Mol. Imaging 2012, 2012, 379807. [Google Scholar] [CrossRef] [PubMed] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bentsen, S.; Clemmensen, A.; Loft, M.; Flethøj, M.; Debes, K.P.; Ludvigsen, T.P.; Larsen, C.B.; Kirchhoff, J.; Olsen, L.H.; Møller, J.E.; et al. [68Ga]Ga-NODAGA-E[(cRGDyK)]2 Angiogenesis PET/MR in a Porcine Model of Chronic Myocardial Infarction. Diagnostics 2021, 11, 1807. https://doi.org/10.3390/diagnostics11101807
Bentsen S, Clemmensen A, Loft M, Flethøj M, Debes KP, Ludvigsen TP, Larsen CB, Kirchhoff J, Olsen LH, Møller JE, et al. [68Ga]Ga-NODAGA-E[(cRGDyK)]2 Angiogenesis PET/MR in a Porcine Model of Chronic Myocardial Infarction. Diagnostics. 2021; 11(10):1807. https://doi.org/10.3390/diagnostics11101807
Chicago/Turabian StyleBentsen, Simon, Andreas Clemmensen, Mathias Loft, Mette Flethøj, Karina Poulsdóttir Debes, Trine Pagh Ludvigsen, Cecilie Bjørstrup Larsen, Jeppe Kirchhoff, Lisbeth Høier Olsen, Jacob Eifer Møller, and et al. 2021. "[68Ga]Ga-NODAGA-E[(cRGDyK)]2 Angiogenesis PET/MR in a Porcine Model of Chronic Myocardial Infarction" Diagnostics 11, no. 10: 1807. https://doi.org/10.3390/diagnostics11101807
APA StyleBentsen, S., Clemmensen, A., Loft, M., Flethøj, M., Debes, K. P., Ludvigsen, T. P., Larsen, C. B., Kirchhoff, J., Olsen, L. H., Møller, J. E., Andersen, T. L., Johannesen, H. H., Jespersen, T., & Kjaer, A. (2021). [68Ga]Ga-NODAGA-E[(cRGDyK)]2 Angiogenesis PET/MR in a Porcine Model of Chronic Myocardial Infarction. Diagnostics, 11(10), 1807. https://doi.org/10.3390/diagnostics11101807







