Metabolic Alterations in Pancreatic Cancer Detected by In Vivo 1H-MR Spectroscopy: Correlation with Normal Pancreas, PET Metabolic Activity, Clinical Stages, and Survival Outcome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Image Acquisition and Analysis
- 1.
- Creatine (Cr) with separately fitted CH2 (Cr_2, 3.09 ppm) and CH3 (Cr_1, 3.03 ppm) groups;
- 2.
- N-Acetylaspartate (NAA, 2.02 ppm);
- 3.
- Glutamate (Glu) and Glutamine (Gln) (2.05–2.50 ppm): Glx as a combined Glu/Gln after separately fitted with each amplitude;
- 4.
- Two lipid/macromolecule lines with separately fitted CH3 (Lipid_1, 0.9 ppm) and CH2 (Lipid_2, 1.5 ppm) groups.
2.3. Statistical Analysis
3. Results
3.1. Clinical Treatment and Follow-Up
3.2. Comparison of MRS Metabolites between Pancreatic Cancer and Normal Pancreatic Parenchyma
3.3. Correlation of MRS Metabolites in Pancreatic Cancer with Pathologic Grade and Clinical TNM Stage
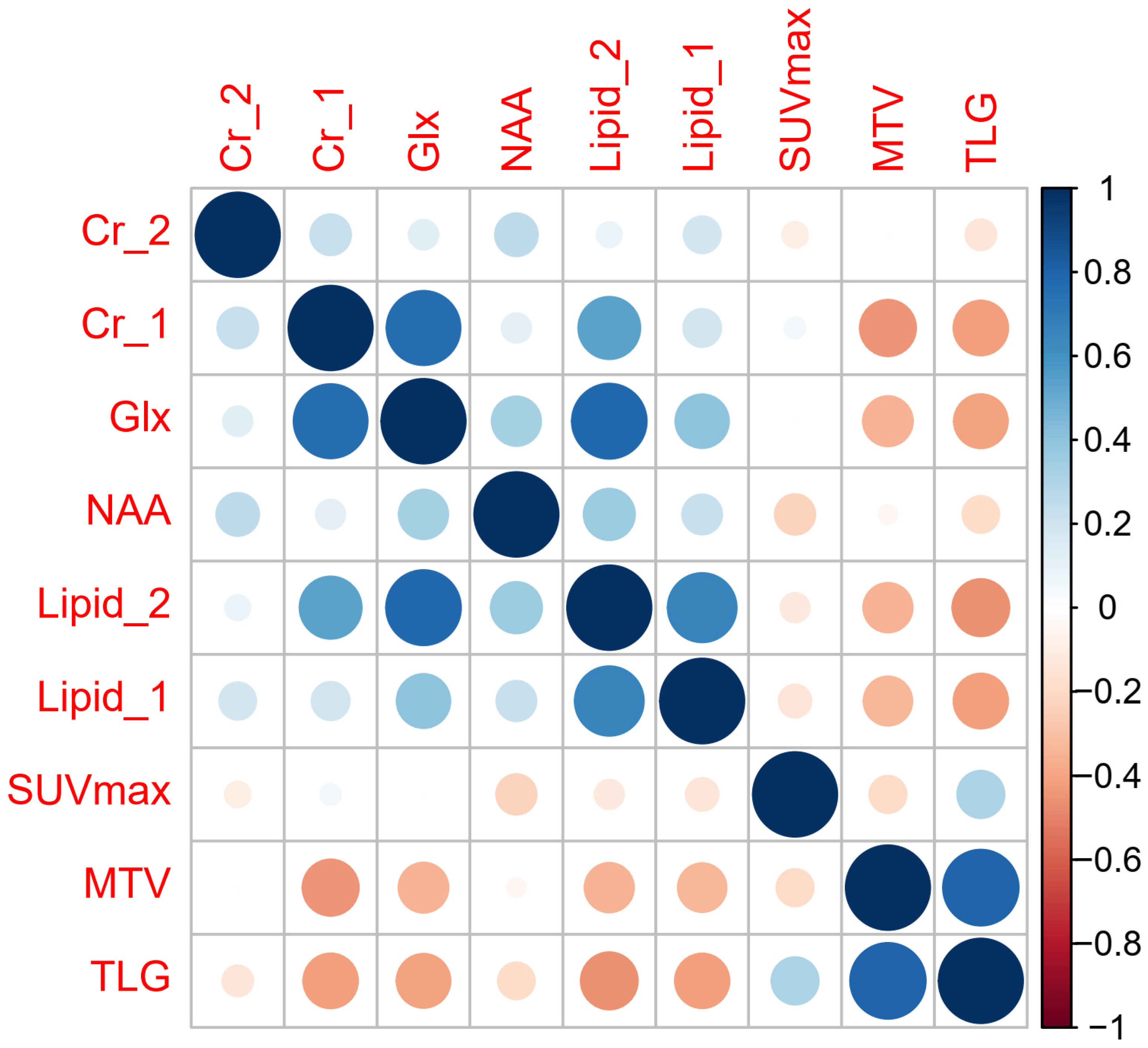
3.4. Correlation of MRS Metabolites in Pancreatic Cancer with PET Parameters
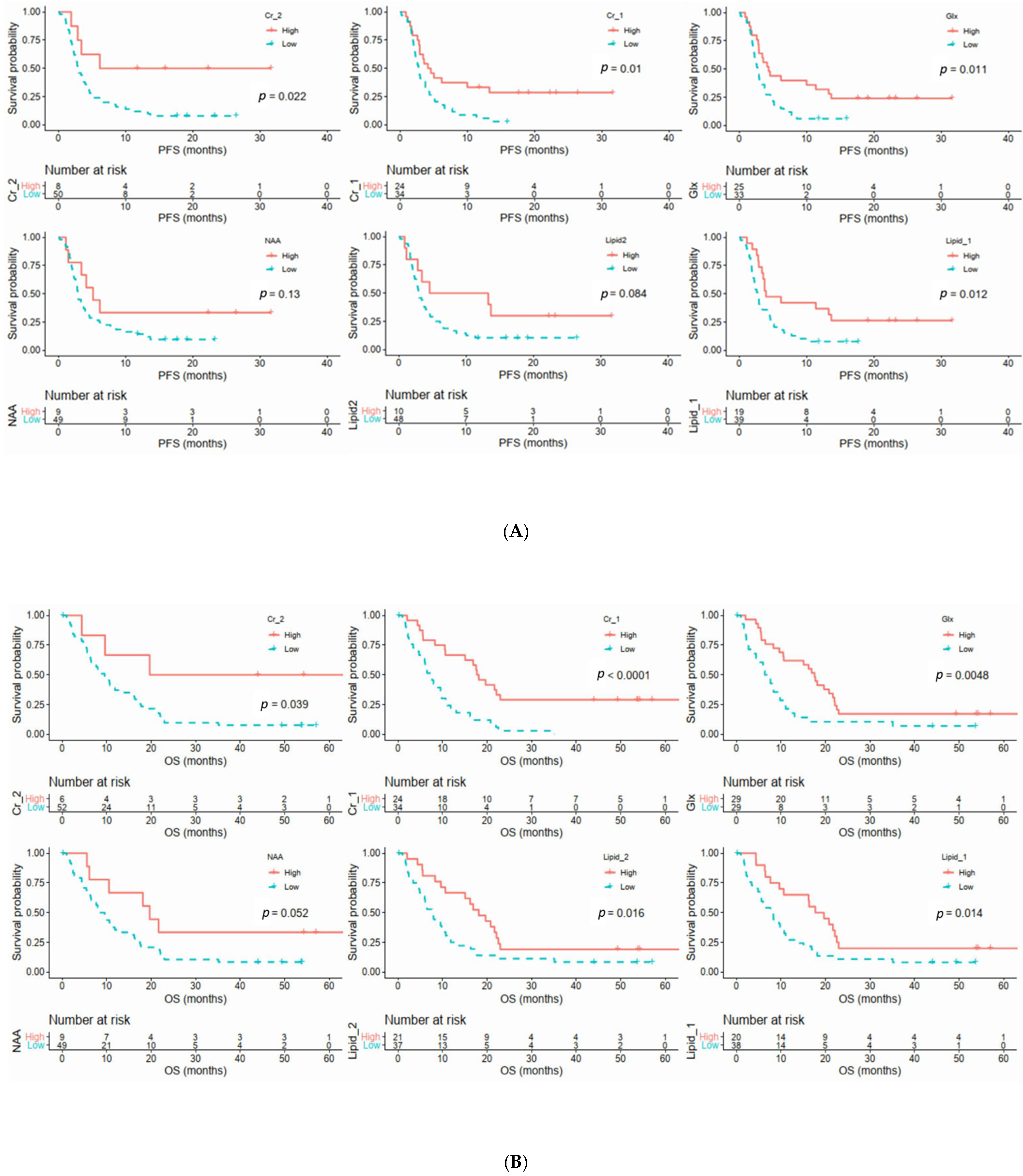
3.5. Relationships between Clinical Parameters and MRS Metabolites with Survival Outcomes
3.6. Subgroup Analysis in Patients with and without Curative Surgery
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.; Zheng, R.; Baade, P.; Zhang, S.; Zeng, H.; Bray, F.; Jemal, A.; Yu, X.Q.; He, J. Cancer statistics in China, 2015. CA Cancer J. Clin. 2016, 66, 115–132. [Google Scholar] [CrossRef] [Green Version]
- Chang, J.; Schomer, D.; Dragovich, T. Anatomical, Physiological, and Molecular Imaging for Pancreatic Cancer: Current Clini-cal Use and Future Implications. BioMed. Res. Int. 2015, 2015, 269641. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Chung, Y.-L. Current Opportunities and Challenges of Magnetic Resonance Spectroscopy, Positron Emission Tomography, and Mass Spectrometry Imaging for Mapping Cancer MetabolismIn Vivo. BioMed Res. Int. 2014, 2014, 625095. [Google Scholar] [CrossRef] [PubMed]
- Halbrook, C.J.; Lyssiotis, C.A. Employing Metabolism to Improve the Diagnosis and Treatment of Pancreatic Cancer. Cancer Cell 2017, 31, 5–19. [Google Scholar] [CrossRef] [Green Version]
- Biancur, D.E.; Kimmelman, A.C. The plasticity of pancreatic cancer metabolism in tumor progression and therapeutic resistance. Biochim. Biophys. Acta Rev. Cancer 2018, 1870, 67–75. [Google Scholar] [CrossRef]
- Karasinska, J.M.; Topham, J.T.; Kalloger, S.E.; Jang, G.H.; Denroche, R.E.; Culibrk, L.; Williamson, L.M.; Wong, H.-L.; Lee, M.K.; O’Kane, G.M.; et al. Altered Gene Expression along the Glycolysis–Cholesterol Synthesis Axis Is Associated with Outcome in Pancreatic Cancer. Clin. Cancer Res. 2020, 26, 135–146. [Google Scholar] [CrossRef] [Green Version]
- Daemen, A.; Peterson, D.; Sahu, N.; McCord, R.; Du, X.; Liu, B.; Kowanetz, K.; Hong, R.; Moffat, J.; Gao, M.; et al. Metabolite profiling stratifies pancreatic ductal adenocarcinomas into subtypes with distinct sensitivities to metabolic inhibitors. Proc. Natl. Acad. Sci. USA 2015, 112, E4410–E4417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, C.; Yang, G.; Yang, J.; Ren, B.; Wang, H.; Chen, G.; Zhao, F.; You, L.; Wang, W.; Zhao, Y. Metabolism of pancreatic cancer: Paving the way to better anticancer strategies. Mol. Cancer 2020, 19, 50. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.B.; Tien, Y.W.; Chang, M.C.; Cheng, M.F.; Chang, Y.T.; Wu, C.H.; Chen, X.J.; Kuo, T.C.; Yang, S.H.; Shih, I.L.; et al. PET/MRI in pancreatic and periampullary cancer: Correlating diffusion-weighted im-aging, MR spectroscopy and glucose metabolic activity with clinical stage and prognosis. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1753–1764. [Google Scholar] [CrossRef]
- Chen, B.-B.; Tien, Y.-W.; Chang, M.-C.; Cheng, M.-F.; Chang, Y.-T.; Yang, S.-H.; Wu, C.-H.; Kuo, T.-C.; Shih, I.-L.; Yen, R.-F.; et al. Multiparametric PET/MR imaging biomarkers are associated with overall survival in patients with pancreatic cancer. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.N.; Dankowski, T.; Ziegler, A. Unbiased split variable selection for random survival forests using maximally selected rank statistics. Stat. Med. 2017, 36, 1272–1284. [Google Scholar] [CrossRef] [Green Version]
- Wallimann, T.; Wyss, M.; Brdiczka, D.; Nicolay, K.; Eppenberger, H.M. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: The ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochem. J. 1992, 281, 21–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowry, O.H.; Berger, S.J.; Chi, M.M.; Carter, J.G.; Blackshaw, A.; Outlaw, W. Diversity of metabolic patterns in human brain tumors-I. High energy phosphate compounds and basic composition. J. Neurochem. 1977, 29, 959–977. [Google Scholar] [CrossRef] [PubMed]
- Papalazarou, V.; Zhang, T.; Paul, N.R.; Juin, A.; Cantini, M.; Maddocks, O.D. The creatine-phosphagen system is mechanoresponsive in pancreatic adenocarci-noma and fuels invasion and metastasis. Nat. Metab. 2020, 2, 62–80. [Google Scholar] [CrossRef]
- Hofer, D.C.; Zirkovits, G.; Pelzmann, H.J.; Huber, K.; Pessentheiner, A.R.; Xia, W.; Uno, K.; Miyazaki, T.; Kon, K.; Tsuneki, H.; et al. N-acetylaspartate availability is essential for juvenile survival on fat-free diet and determines metabolic health. FASEB J. 2019, 33, 13808–13824. [Google Scholar] [CrossRef] [Green Version]
- Zand, B.; Previs, R.A.; Zacharias, N.M.; Rupaimoole, R.; Mitamura, T.; Nagaraja, A.; Guindani, M.; Dalton, H.J.; Yang, L.; Baddour, J.; et al. Role of Increased n-acetylaspartate Levels in Cancer. J. Natl. Cancer Inst. 2016, 108, djv426. [Google Scholar] [CrossRef] [Green Version]
- Bogner-Strauss, J.G. N-Acetylaspartate Metabolism Outside the Brain: Lipogenesis, Histone Acetylation, and Cancer. Front. Endocrinol. 2017, 8, 240. [Google Scholar] [CrossRef] [Green Version]
- Cluntun, A.A.; Lukey, M.J.; Cerione, R.A.; Locasale, J.W. Glutamine Metabolism in Cancer: Understanding the Heterogeneity. Trends Cancer 2017, 3, 169–180. [Google Scholar] [CrossRef] [Green Version]
- Yao, W.; Maitra, A.; Ying, H. Recent insights into the biology of pancreatic cancer. EBioMedicine 2020, 53, 102655. [Google Scholar] [CrossRef]
- Roux, C.; Riganti, C.; Borgogno, S.F.; Curto, R.; Curcio, C.; Catanzaro, V.; Digilio, G.; Padovan, S.; Puccinelli, M.P.; Isabello, M.; et al. Endogenous glutamine decrease is associated with pancreatic cancer progression. Oncotarget 2017, 8, 95361–95376. [Google Scholar] [CrossRef] [PubMed]
- Biancur, D.E.; Paulo, J.A.; Małachowska, B.; Del Rey, M.Q.; Sousa, C.M.; Wang, X.; Sohn, A.S.W.; Chu, G.C.; Gygi, S.P.; Harper, J.; et al. Compensatory metabolic networks in pancreatic cancers upon perturbation of glutamine metabolism. Nat. Commun. 2017, 8, 15965. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Lai, L.A.; Sullivan, Y.; Wong, M.; Wang, L.; Riddell, J.; Jung, L.; Pillarisetty, V.G.; Brentnall, T.A.; Pan, S. Disrupting glutamine metabolic pathways to sensitize gemcitabine-resistant pancreatic cancer. Sci. Rep. 2017, 7, 7950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillies, R.J.; Morse, D.L. In Vivo Magnetic Resonance Spectroscopy in Cancer. Annu. Rev. Biomed. Eng. 2005, 7, 287–326. [Google Scholar] [CrossRef]
- Yao, X.; Zeng, M.; Wang, H.; Fei, S.; Rao, S.; Ji, Y. Metabolite detection of pancreatic carcinoma by in vivo proton MR spectroscopy at 3T: Initial results. Radiol. Med. 2011, 117, 780–788. [Google Scholar] [CrossRef]
- Cho, S.G.; Lee, D.H.; Lee, K.Y.; Ji, H.; Lee, K.H.; Ros, P.R.; Suh, C.H. Differentiation of Chronic Focal Pancreatitis From Pancreatic Carcinoma by In Vivo Proton Magnetic Resonance Spectroscopy. J. Comput. Assist. Tomogr. 2005, 29, 163–169. [Google Scholar] [CrossRef]
- Zhang, G.; He, P.; Tan, H.; Budhu, A.; Gaedcke, J.; Ghadimi, B.M.; Ried, T.; Yfantis, H.G.; Lee, D.H.; Maitra, A.; et al. Integration of Metabolomics and Transcriptomics Revealed a Fatty Acid Network Exerting Growth Inhibitory Effects in Human Pancreatic Cancer. Clin. Cancer Res. 2013, 19, 4983–4993. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.W.; Kang, C.M.; Choi, H.J.; Lee, W.J.; Song, S.Y.; Lee, J.H.; Lee, J.D. Prognostic Value of Metabolic Tumor Volume and Total Lesion Glycolysis on Preoperative 18F-FDG PET/CT in Patients with Pancreatic Cancer. J. Nucl. Med. 2014, 55, 898–904. [Google Scholar] [CrossRef] [Green Version]
- Julià-Sapé, M.; Candiota, A.P.; Arús, C. Cancer metabolism in a snapshot: MRS(I). NMR Biomed. 2018, 32, e4054. [Google Scholar] [CrossRef]
- García-Figueiras, R.; Baleato-González, S.; Padhani, A.R.; Oleaga, L.; Vilanova, J.C.; Luna, A.; Gómez, J.C.C. Proton magnetic resonance spectroscopy in oncology: The fin-gerprints of cancer? Diagn. Interv. Radiol. 2016, 22, 75–89. [Google Scholar] [CrossRef]
- Fukui, H.; Hori, M.; Fukuda, Y.; Onishi, H.; Nakamoto, A.; Ota, T.; Ogawa, K.; Ninomiya, K.; Tatsumi, M.; Osuga, K.; et al. Evaluation of fatty pancreas by proton density fat fraction using 3-T magnetic resonance imaging and its association with pancreatic cancer. Eur. J. Radiol. 2019, 118, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Öz, G.; Deelchand, D.K.; Wijnen, J.P.; Mlynarik, V.; Xin, L.; Mekle, R.; Noeske, R.; Scheenen, T.W.; Tkáč, I.; Andronesi, O.; et al. Advanced single voxel 1 H magnetic resonance spectroscopy techniques in humans: Experts’ consensus recommendations. NMR Biomed. 2021, 34, e4236. [Google Scholar] [CrossRef]
- Wilson, M.; Andronesi, O.; Barker, P.B.; Bartha, R.; Bizzi, A.; Bolan, P.J.; Brindle, K.M.; Choi, I.Y.; Cudalbu, C.; Dydak, U.; et al. Methodological consensus on clinical proton MRS of the brain: Review and recommendations. Magn. Reson. Med. 2019, 82, 527–550. [Google Scholar] [CrossRef] [Green Version]
- Pickup, S.; Lee, S.C.; Mancuso, A.; Glickson, J.D. Lactate imaging with Hadamard-encoded slice-selective multiple quantum co-herence chemical-shift imaging. Magn. Reson. Med. 2008, 60, 299–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lingvay, I.; Esser, V.; Legendre, J.L.; Price, A.L.; Wertz, K.M.; Adams-Huet, B.; Zhang, S.; Unger, R.H.; Szczepaniak, L.S. Noninvasive Quantification of Pancreatic Fat in Humans. J. Clin. Endocrinol. Metab. 2009, 94, 4070–4076. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Parameter | Variable |
|---|---|
| Age (years) * | 62.7 ± 12.1 (34~81) |
| Sex (Men/Women) * | |
| Men | 36 (62) |
| Women | 22 (38) |
| Tumor size (cm) * | 3.3 ± 1.4 |
| Tumor location † | |
| Head | 33 (57) |
| Neck | 7 (12) |
| Body | 12 (21) |
| Tail | 6 (10) |
| Surgery method † (n = 19) | |
| Whipple operation | 14 |
| Distal pancreatectomy | 2 |
| Exploratory laparotomy and biopsy | 1 |
| Bypass and biopsy | 2 |
| TNM staging † | |
| I | 4 (7) |
| II | 12 (21) |
| III | 9 (16) |
| IV | 33 (57) |
| Histology grades † (n = 20) | |
| Well-differentiated | 3 |
| Moderately differentiated | 8 |
| Poorly differentiated | 9 |
| Metabolites | ppm | Normal Pancreas | Pancreatic Cancer | p Value |
|---|---|---|---|---|
| Cr_2 | 3.9 | 11.2 ± 19.9 | 7.7 ± 13.9 | 0.200 |
| Cr_1 | 3.03 | 8.9 ± 15.8 | 4.5 ± 6.1 | 0.076 |
| Glx | 2.05–2.5 | 137.9 ± 201.4 | 66 ± 72.3 | 0.005 * |
| NAA | 2.02 | 78.9 ± 100.6 | 24.1 ± 41.7 | <0.001 * |
| Lipid_2 | 1.3 | 3119.4 ± 7427.6 | 1015.2 ± 1190.7 | <0.001 * |
| Lipid_1 | 0.9 | 886.3 ± 1218.7 | 181.4 ± 240.2 | <0.001 * |
| Metabolites | Pathological Grades | T Stage | N Stage | M Stage | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Well- and Moderately (n = 11) | Poorly Differentiated (n = 9) | p Value | T1–3 (n = 26) | T4 (n = 32) | p Value | N0 (n = 14) | N1 (n = 44) | p Value | M0 (n = 25) | M1 (n = 33) | p Value | |
| Cr_2 | 6 ± 5.7 | 6.3 ± 12.8 | 0.152 | 6.6 ± 9.2 | 8.2 ± 16.7 | 0.506 | 14.2 ± 22.4 | 5.3 ± 8.9 | 0.019 * | 5.6 ± 6.7 | 8.9 ± 17.3 | 0.85 |
| Cr_1 | 5.3 ± 6.5 | 2.6 ± 3.4 | 0.370 | 4.7 ± 6.1 | 4 ± 6.1 | 0.681 | 4.7 ± 6.2 | 4.2 ± 6.1 | 0.581 | 4.4 ± 5.5 | 4.3 ± 6.6 | 0.395 |
| Glx | 91.4 ± 87.5 | 46.6 ± 39.6 | 0.230 | 76.4 ± 75 | 57.5 ± 70.1 | 0.152 | 85.8 ± 83.4 | 59.7 ± 68.3 | 0.170 | 69.2 ± 70.5 | 63.6 ± 74.7 | 0.392 |
| NAA | 22.0 ± 21.9 | 15.1 ± 15.7 | 0.603 | 32.1 ± 55.8 | 17.1 ± 19.6 | 0.359 | 27.4 ± 28 | 22.7 ± 43.8 | 0.271 | 16.9 ± 15.6 | 29 ± 51.5 | 0.519 |
| Lipid_2 | 1337.6 ± 1278.2 | 1044.9 ± 1205.1 | 0.456 | 1139.3 ± 1246.1 | 860.4 ± 1128.6 | 0.101 | 1410.8 ± 1444 | 850.1 ± 1067.2 | 0.122 | 1088.9 ± 1192.3 | 907 ± 1183.6 | 0.282 |
| Lipid_1 | 197.2 ± 274.9 | 289.6 ± 264.8 | 0.503 | 222.3 ± 274.4 | 138.4 ± 200 | 0.038 * | 248.4 ± 253.7 | 153 ± 230.7 | 0.084 | 192.6 ± 213.6 | 163.5 ± 257 | 0.094 |
| PFS | OS | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariable | Multivariable | Univariable | Multivariable | ||||||||||
| Parameters | Cutoff | HR | 95% CI | p Value | HR | 95% CI | p Value | HR | 95% CI | p Value | HR | 95% CI | p Value |
| Age (y/o) | 64.5 | 1.001 | 0.574–1.748 | 0.996 | 1.025 | 0.588–1.787 | 0.935 | ||||||
| Sex (Women vs. men) | 1.222 | 0.691–2.162 | 0.490 | 0.731 | 0.411–1.301 | 0.287 | |||||||
| Size (cm) | 3.1 | 0.712 | 0.408–1.243 | 0.232 | 0.965 | 0.554–1.683 | 0.821 | ||||||
| TNM stage (4 vs. ≦ 3) | 2.096 | 1.164–3.774 | 0.014 * | 2.084 | 1.11–3.92 | 0.023 | 1.858 | 1.038–3.326 | 0.037 * | ||||
| MRS metabolites | |||||||||||||
| Cr_2 | 10.26/12.02 | 0.32 | 0.115–0.895 | 0.022 * | 0.299 | 0.10–0.86 | 0.025 | 0.309 | 0.096–1.001 | 0.039 * | |||
| Cr_1 | 1.89/1.89 | 0.466 | 0.256–0.846 | 0.01 * | 0.313 | 0.17–0.576 | <0.0001 * | 0.342 | 0.185–0.632 | <0.0001 | |||
| Glx | 45.36/39.86 | 0.472 | 0.261–0.854 | 0.011 * | 0.449 | 0.254–0.794 | 0.0048 * | ||||||
| NAA | 30.62/30.62 | 0.519 | 0.22–1.222 | 0.13 | 0.437 | 0.185–1.029 | 0.052 | ||||||
| Lipid_2 | 1720/835.7 | 0.496 | 0.22–1.117 | 0.084 | 0.489 | 0.27–0.887 | 0.016 * | ||||||
| Lipid_1 | 149.25/133.35 | 0.452 | 0.24–0.851 | 0.012 * | 0.575 | 0.295–1.117 | 0.102 | 0.478 | 0.261–0.873 | 0.014 * | 0.556 | 0.302–1.023 | 0.059 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-K.; Shih, T.T.-F.; Tien, Y.-W.; Chang, M.-C.; Chang, Y.-T.; Yang, S.-H.; Cheng, M.-F.; Chen, B.-B. Metabolic Alterations in Pancreatic Cancer Detected by In Vivo 1H-MR Spectroscopy: Correlation with Normal Pancreas, PET Metabolic Activity, Clinical Stages, and Survival Outcome. Diagnostics 2021, 11, 1541. https://doi.org/10.3390/diagnostics11091541
Chang C-K, Shih TT-F, Tien Y-W, Chang M-C, Chang Y-T, Yang S-H, Cheng M-F, Chen B-B. Metabolic Alterations in Pancreatic Cancer Detected by In Vivo 1H-MR Spectroscopy: Correlation with Normal Pancreas, PET Metabolic Activity, Clinical Stages, and Survival Outcome. Diagnostics. 2021; 11(9):1541. https://doi.org/10.3390/diagnostics11091541
Chicago/Turabian StyleChang, Chih-Kai, Tiffany Ting-Fang Shih, Yu-Wen Tien, Ming-Chu Chang, Yu-Ting Chang, Shih-Hung Yang, Mei-Fang Cheng, and Bang-Bin Chen. 2021. "Metabolic Alterations in Pancreatic Cancer Detected by In Vivo 1H-MR Spectroscopy: Correlation with Normal Pancreas, PET Metabolic Activity, Clinical Stages, and Survival Outcome" Diagnostics 11, no. 9: 1541. https://doi.org/10.3390/diagnostics11091541







