Abstract
We aimed to compare dedicated coronary computed tomography angiography (CCTA) followed by high-pitch scanning and triple-rule-out computed tomography angiography (TRO-CTA) in terms of radiation dose, contrast media (CM) use, and image quality. Patients with acute chest pain were retrospectively enrolled and assigned to group A (n = 55; scanned with dedicated CCTA followed by high-pitch scanning) or group B (n = 45; with TRO-CTA). Patient characteristics, radiation dose, CM use, and quantitative parameters (CT value, image noise, signal-to-noise ratio, contrast-to-noise ratio, and image quality score) of pulmonary arteries (PAs), thoracic aortae (TAs), and coronary arteries (CAs) were compared. The total effective dose was significantly lower in group A (6.25 ± 2.94 mSv) than B (8.93 ± 4.08 mSv; p < 0.001). CM volume was significantly lower in group A (75.7 ± 8.9 mL) than B (95.0 ± 0 mL; p < 0.001). PA and TA image quality were significantly better in group B, whereas that of CA was significantly better in group A. Qualitative image scores of PA and TA scans rated by radiologists were similar, whereas that of CA scans was significantly higher in group A than B (p < 0.001). Dedicated CCTA followed by high-pitch scanning demonstrated lower radiation doses and CM volume without debasing qualities of PA, TA, and CA scans than did TRO-CTA.
1. Introduction
Over 8 million people visit emergency departments owing to chest pain yearly [1,2]. Chest pain remains a major diagnostic challenge in emergency departments [3]. The primary concern of emergency departments is to eliminate potential life-threatening causes of chest pain, pulmonary embolism (PE), aortic dissection (AD), and acute coronary syndrome (ACS) [4]. Triple-rule-out computed tomography angiography (TRO-CTA) has been used because of its rapid results, non-invasiveness, and superiority. It also allows the simultaneous imaging of the pulmonary arteries (PAs), thoracic aortae (TAs), and coronary arteries (CAs) [3].
However, TRO-CTA is often associated with a higher radiation dose and increased use of contrast media (CM), although it achieves a slightly higher diagnostic yield for PE and AD [5,6]. For example, 120 kV tube voltage was used on dual-source CT with an effective dose of as high as 32.6 mSv [7]; in contrast, coronary computed tomography angiography (CCTA) showed evidence of DNA damage with an effective dose of over 7.5 mSv [8]. Additionally, a large volume of iodinated CM ranging from 80 to 120 mL in TRO-CTA is usually needed to maintain good image quality [6,9,10,11]. Although the rapid injection of high-dose iodinated CM enhances the visualization of the right atrium and right ventricle artifacts, it can seriously affect the observation of the right coronary artery.
Despite the above concerns, the optimal dose or optimal TRO-CTA protocol remains unclear [5]. Therefore, this study aimed to compare dedicated CCTA followed by high-pitch scanning and traditional TRO-CTA performed in patients with chest pain to evaluate the image quality, radiation dose, and use of iodinated CM.
2. Methods
2.1. Patient Population
A total of 104 patients with acute chest pain in our hospital between January 2021 and January 2022 were retrospectively enrolled. Patients aged ≥18 years with acute chest pain who were clinically suspected of having ACS, PE, or AD were included. The exclusion criteria were as follows: (1) lack of patient information (height, weight, heart rate) or (2) lack of scanning parameters, such as dose-length product (DLP), injection delivery rate, and volume of CM.
This study was approved by the ethics committee of our hospital, and the need for informed consent was waived. Finally, 100 patients were enrolled and divided between group A (n = 55) and group B (n = 45) (Figure 1).

Figure 1.
Flowchart of patient enrolment.
2.2. Image Acquisition
All patients underwent imaging using a new generation of dual-source CT scanners (Somatom Drive, Siemens Healthcare, Forchheim, Germany). Dedicated CCTA followed by high-pitch scanning was used to scan the CAs first followed by the PAs and TA of the patients in group A. A calcium score (CS) was calculated to plan the scan range of CCTA using the following parameters: tube voltage, Sn100; automated anatomical tube current modulation, 268 mAsref.qual (CARE Dose 4D, Siemens Healthineers, Forchheim, Germany); and data acquisition window (R-R interval), 70%. For CCTA imaging, the collimation was 2 × 64 × 0.6 mm, and the rotation time was 0.28 s per rotation. A prospectively ECG-triggered sequence acquisition mode (step-and-shoot) was used, and scanning was performed from the cranium to the cauda. An automated anatomical tube current modulation technique with 380 mAsref.qual (CARE Dose 4D, Siemens Healthineers) and an automatic tube voltage selection with 120 kVref.qual (ATVS, CARE kVTM, Siemens Healthineers) were used, with the range of exposure dose (ECG-pulsing) set at 30–80%. A high-pitch scan or double high-pitch scan was performed to scan the entire chest in a cranial-to-caudal direction using an AVTS with 120 kVref.qual and a CARE Dose 4D with 100 mAsref.qual., with a pitch of 2.2. The bolus tracking technique was used for threshold monitoring at the aortic root for CAs with an enhancement threshold of 80 HU and a delay time of 7 s, and for threshold monitoring at the left atrium with an enhancement threshold of 120 HU and a delay time of 4 s. Pre-warmed CM (iobitridol, 350 mg iodine [mgI]/mL) was injected using an 18-G closed intravenous catheter system with an Ulrich high-pressure syringe. For CCTA, the CM was administered according to the patient’s weight (0.8 mL/kg with a 13 s injection duration), and saline was administered at the same delivery rate with a 10 s injection duration. For PE and AD, the CM and saline were fixed at 25 mL and 40 mL, respectively, with a fixed delivery rate of 4 mL/s.
A retrospective ECG-triggered acquisition mode with a pitch of 0.17 was used to scan the entire chest in a cranial-to-caudal direction for patients in group B. An automated anatomical tube current modulation technique with 290 mA (CARE Dose 4D, Siemens Healthineers) and an automatic tube voltage selection with 100 kVref.qual (ATVS, CARE kVTM, Siemens Healthineers) was used. The exposure dose (ECG-pulsing) range was set at 35–80% for the R-R interval. The bolus tracking technique was used for threshold monitoring at the aortic root, with an enhancement threshold of 100 HU and a delay time of 9 s. Pre-warmed CM (iobitridol, 350 mg iodine [mgI]/mL) was injected using an 18-G closed intravenous catheter system with an Ulrich high-pressure syringe. The CM was administered in two phases: a fixed 50 mL of CM at a delivery rate of 4 mL/s and a fixed 30 mL of saline at a delivery rate of 3.5 mL/s were delivered immediately followed by the first phase using a fixed 45 mL of CM delivered at a delivery rate of 4.5 mL/s.
2.3. Image Reconstruction and Evaluation
In group A, the slice thickness and image reconstruction interval with the advanced modeled iterative reconstruction (strength level 4) algorithm for CAs were both 0.6 mm, and the kernel used was I26f. The slice thickness and image reconstruction interval with the advanced modeled iterative reconstruction (strength level 3) algorithm for the PE and aorta were both 1.0 mm, and the kernel used was I30f. In group B, the slice thickness and image reconstruction interval with the advanced modeled iterative reconstruction (strength level 3) algorithm for CAs were both 0.6 mm, and the kernel used was I26f. The slice thickness and image reconstruction interval with the advanced modeled iterative reconstruction (strength level 3) algorithm for PE and aorta was both 1.0 mm, and the kernel used was I26f.
The CT attenuation value and standard deviation (SD) of the pulmonary trunk (PT), left pulmonary artery (LPA), right pulmonary artery (RPA), aortic root (AO), aortic arch (AA), descending aorta (DA), proximal left main coronary artery (LMCA-P), middle left anterior descending (LAD-M), distal left anterior descending (LAD-D), middle left circumflex (LCX-M), distal left circumflex (LCX-D), proximal right coronary artery (RCA-P), middle right coronary artery (RCA-M), distal coronary right artery (RCA-D), erector spinae muscle (ESM), and perivascular adipose tissue (PVAT) were measured on the axial images using a region of interest of 20–400 mm2, and the contrast-to-noise ratio (CNR) for vessels was calculated. For the PAs and aorta, CNR was calculated using muscle as the background with the SD of muscle as the background noise; for CAs, fat was used as the background with the SD value of fat as the background noise.
The qualitative image score was assessed blindly and independently by two radiologists (one with eight years of experience in chest imaging, and the other with over 15 years of experience in chest imaging). The image quality score of CAs was based on the 15-segment classification by the modified American Heart Association classification [12]. PAs and TAs were scored using a four-point scoring system as follows: excellent (a score of 1) was defined as the complete absence of motion artifacts, excellent signal-to-noise ratio, and clear delineation of vessel walls, with the ability to assess luminal stenosis and plaque characteristics; good (score of 2) was defined as nonlimiting motion artifacts, reduced signal-to-noise ratio, and/or presence of calcifications, with the preserved ability to assess luminal stenosis and plaque characteristics; adequate (a score of 3) was defined as reduced image quality due to any combination of noise, motion, poor contrast enhancement, or presence of calcium that significantly impaired ease of interpretation but where the image quality was sufficient to rule out significant stenosis; and nondiagnostic (a score of 4) was defined as reduced image quality that precludes adequate assessment of stenosis in the majority of vessels [6].
2.4. Radiation Dose
The radiation doses of CS scan, CCTA, PAs, and aorta were recorded; the radiation associated with the scout view, CS scan, and the automatic bolus tracking technique was not included. The DLP and volume CT dose index (CTDIvol) were automatically provided by the CT scanner. The effective dose (ED) was estimated by multiplying the DLP by a conversion factor of 0.017 mSv/(mGy × cm) for the chest [13] and 0.026 for the CAs [8].
2.5. Statistical Analysis
The statistical software used was SPSS 22.0 (IBM, Chicago, IL, USA). Quantitative data are expressed as the mean ± SD (x ± s) or median (minimum, maximum), and count data are expressed as absolute values and percentages. If the general data of the groups, the objective evaluation indicators of image quality, and the radiation dose conformed to the normal distribution, the independent sample t-test was used. If the normal distribution was not confirmed, the non-parametric rank-sum test (Mann–Whitney U-test), the subjective scores of image quality, and diagnostic accuracy were compared using the chi-squared test. The consistency of the subjective scores of the two physicians was tested using the Kappa value. A Kappa value of 0.21–0.40 indicates poor consistency, 0.41–0.60 indicates moderate consistency, and 0.61–0.80 indicates good consistency. p < 0.05 indicates statistical significance.
3. Results
A total of 100 patients were included in this study, of whom 55 patients was enrolled in group A and 45 in group B (Figure 1). There were no significant differences in sex, age, height, weight, body mass index, and heart rate between the two groups (p > 0.05).
In group A, the tube voltage for CS was Sn100; for CCTA, 2, 3, 9, and 41 patients were scanned using 120 kV, 110 kV, 100 kV, and 90 kV, respectively. For the high-pitch scan, 12 patients were scanned using a double high-pitch scan for both PA and TA evaluation, whereas the other 43 patients were scanned using only a single high-pitch scan; the tube voltage is shown in Table 1. The CTDIvol and DLP for groups A and B are shown in Table 2. The total ED in group A was significantly lower (6.25 ± 2.94 mSv; p < 0.001) than that in group B (8.93 ± 4.08 mSv). The volume of CM in group A was significantly lower (75.7 ± 8.9 mL; p < 0.001) than that in group B (95.0 ± 0 mL), while the saline volume in group B was significantly lower (p < 0.001). The details are listed in Table 2 and Table 3.

Table 1.
Tube voltage distribution.

Table 2.
Comparison of radiation dose parameters between two different TRO-CTA protocols.

Table 3.
Characteristics of patients examined by two different scan mode TRO-CTA protocols.
The CT attenuation values of PT, LPA, RPA, AO, AA, DA, LMCA-P, LAD-D, LCX-D, RCA-P, and ESM in group A were significantly different from those in group B (p < 0.05). The image noise of PT, LPA, RPA, AO, AA, and DA in group A was significantly higher than that in group B (p < 0.05), whereas that of LMCA-P, LAD-M, LAD-D, LCX-M, LCX-D, RCA-P, RCA-M, and RCA-D in group A was significantly lower than that in group B (p < 0.05). The SNR values of PT, LPA, RPA, AO, AA, and DA in group B were significantly better than those in group A, whereas those of LAD-M, LAD-D, LCX-D, and RCA-M in group A were significantly better than those in group B (p < 0.05). The CNR values of PT, LPA, RPA, AO, AA, and DA in group B were significantly better than those in group A, whereas those of LMCA-P, LAD-D, and LCX-D in group A were significantly better than those in group B (p < 0.05).
There was good inter-rater reliability between the two radiologists for the qualitative image score (κ = 0.89), and their average scores were used for further analysis. The qualitative image score showed that there was no significant difference in the PAs and CAs between groups, whereas the score for TAs in group A was significantly better than that in group B (Table 4, Figure 2 and Figure 3).

Table 4.
Results of the objective quality analysis.
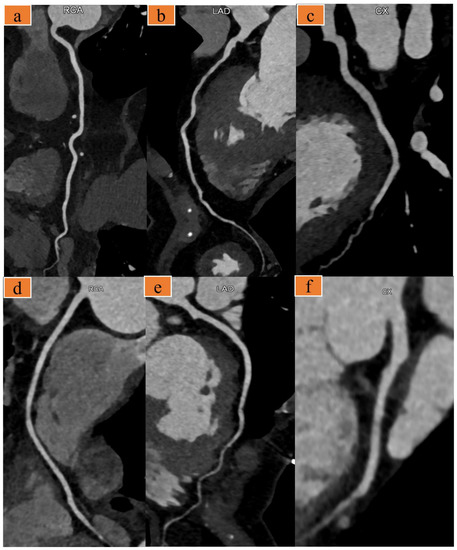
Figure 2.
(a–c) A 91-year-old man with chief complaint of chest pain (heart rate: 67 bpm). Images were acquired with the optimized scanning scheme. Curved multiplanar reformat computed tomography (CT) images show the right coronary artery (a), left anterior descending artery (b), and left circumflex artery (c). (d–f) A 63-year-old woman presented to the emergency room with several days of chest pain, heart rate 58 bpm. Images were acquired with the traditional triple-rule-out computed tomography angiography (TRO-CTA). Curved multiplanar reformat CT images of the right coronary (d), left anterior descending artery (e), and left circumflex artery (f).
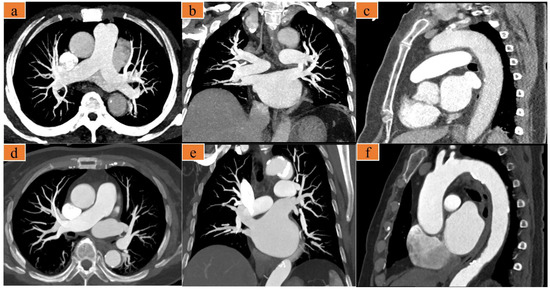
Figure 3.
(a–c) A 75-year-old woman with chest pain, heart rate 77 bpm. Images were acquired with the optimized scanning scheme. (d–f) A 67-year-old woman presents to the emergency room with several days of chest pain, heart rate 66 bpm. Images were acquired with the traditional triple-rule-out computed tomography angiography (TRO-CTA). (a,d) are axial maximum intensity projection (MIP) images showing the pulmonary artery; (b,e) are coronal MIP images showing the pulmonary artery; and (c,f) are sagittal MIP images showing the thoracic aorta.
4. Discussion
Our study demonstrated the feasibility of a dedicated CCTA imaging followed by a high-pitch scan to achieve better image quality and lower radiation dose and CM volume than one-scan TRO-CTA. Our study also revealed an alternative for TRO-CTA using a dual-source CT. Compared with one-scan TRO-CTA, this technique significantly reduces the radiation dose and the CM volume without debasing the image quality.
Burris et al. reported that the use and appropriateness of TRO-CTA in the clinical setting must be further defined because the higher benefits of PE and AD were accompanied with a higher radiation dose and CM volume [6,13,14]. Only a few studies reporting the optimal protocol for both radiation dose and CM use using dual-source CT have been conducted after Burris et al.’s study. In 2017, Si-Mohamed et al. demonstrated that the second generation dual-source CT using a 100-kVp combing sinogram affirmed the iterative reconstruction algorithm for TRO-CTA reduced the ED to 5.7 ± 2.7 mSv using a conversion factor of 0.017 mSv/(mGy × cm) [9]. Compared with their study, our study achieved a slightly higher radiation dose (6.25 ± 2.94 mSv vs. 5.65 ± 1.37 mSv) but better image quality and lower CM volume. In 2018, He et al. demonstrated a surprisingly lower ED of 2.67 ± 0.98 mSv using a conversion factor of 0.014 mSv/(mGy × cm) but with a 16-cm wide-detector CT [13]. Compared with He et al.’s study with less exposure time of the 16-cm coverage detector [13,15], our study demonstrated that TRO-CTA could be achieved by a dedicated step-and-shoot CCTA that combines the short scan time of high-pitch scan; both of them were widely reported to achieve a lower radiation dose with good image quality [16,17,18,19], and the step-and-shoot CCTA with AVTS has been adaptive to higher heart rate [20,21]. In addition, the tube voltage at Sn100 of CS reportedly achieves a lower radiation dose without influencing the accuracy of the calcium score [22,23].
For CM use, Takx et al. [10] injected 120 mL of CM (Imeron 400 mgI/mL) using a dual-source CT system; Si-Mohamed et al. injected 90 mL (Imeron 400 mgI/mL) using a single source 64-section CT system; and another study injected 113 ± 16 mL, even including 57.4% scanned using a high-pitch scan [6]. Our study demonstrated the second lowest CM volume (75.0 ± 9.0 mL), which was slightly more than that (73.9 ± 7.4 mL) of He et al.’s study [13], whereas the 16-cm coverage detector prioritizes less exposure time with wide coverage compared with the 64-row CT system [13,15]. Although the volume of CM in contrast-enhanced CT does not harm the patients, catheter-based angiography after CTA with CM may increase the burden on the kidneys [24]. Our results showed that CT values, image noise, SNR, and CNR of the PAs and TAs in group B were significantly better than those in group A, but group A obtained lower radiation dose and lower CM volume from the high-pitch scans. Previous studies have shown that the CT values for PE and AD diagnosis were suggested to be more than 200 HU [25] and 250 HU [26], respectively, which may also explain the similar qualitative image score (p > 0.05, Table 4). CT values, image noise, SNR, and CNR of CA in group A were significantly better than those in group B, which could be proven with better qualitative image scores (2 vs. 3, Table 4), indicating that our method succeeded in obtaining a better image quality with CCTA imaging than PA and TA imaging. Compared with previous studies [9,13], the CNR and SNR of group A were also better. TRO-CTA was reported to be associated with a slightly higher incidence of PE and AD along with a much higher radiation and contrast dose [6], while there was not enough evidence to compare TRO-CTA with pulmonary CTA or aorta CTA for the diagnosis of PE or AD [5]. When TRO-CTA instead of CCTA was chosen in the emergency department, PE and AD were rarely found together [5,14]. Based on the above, our study tried to prioritize the quality of CCTA while appropriately debasing the image quality for PE and AD to meet the balance between image quality, radiation, and CM dose compared with that of the one-scan TRO-CTA. Our study demonstrated that better CCTA imaging combined with slightly worse PA and TA imaging with lower ED and CM use achieved similar image quality compared with one-scan TRO-CTA.
This study has some limitations. First, this single-center study had a small sample size, and the retrospective nature of the study design could not distinguish how the patients were enrolled into different groups. Moreover, the history of the patients was unknown; however, the patient characteristics between the groups showed no difference, which could support our results. Second, our study did not compare the diagnostic accuracies of PE, AD, and ACS because for all patients with negative results for PE, AD, and coronary angiography, the diagnostic accuracy of ACS could not be achieved without performing coronary angiography, although ACS was diagnosed in both groups. Further studies should take this into account. Finally, the radiation dose of CCTA in group A remained higher owing to the wide (35–80% for the R-R interval) exposure dose (ECG-pulsing) range.
In conclusion, dedicated CCTA followed by a high-pitch scan could achieve better image quality and similar image quality of PAs and TAs with lower radiation and contrast doses than the traditional one-scan TRO-CTA.
Author Contributions
Conceptualization, K.W., L.J. and M.L.; data curation, K.W., S.Z. and C.L.; formal analysis, K.W., S.Z. and X.W.; funding acquisition, L.J. and M.L.; investigation, X.W. and L.J.; project administration, L.J.; resources, M.L. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Science and Technology Planning Project of Shanghai Science and Technology Commission [grant number, 22Y11910700], National Natural Science Foundation of China [grant number 61976238], Shanghai “Rising Stars of Medical Talent” Youth Development Program “Outstanding Youth Medical Talents” (SHWJRS [2021]-99), Emerging Talent Program (XXRC2213) and Leading Talent Program (LJRC2202) of Huadong hospital, and Excellent Academic Leaders of Shanghai(2022XD042).
Institutional Review Board Statement
This study was approved by the ethics committee of Huadong hospital (20210054 on 29 July 2021).
Informed Consent Statement
The need for informed consent was waived.
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.
Acknowledgments
We gratefully acknowledge the staff of this work for their efforts.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| Abbreviation | Meaning |
| TRO-CTA | Triple-Rule-Out Computed Tomography Angiography |
| CM | Contrast Media |
| CCTA | Coronary Computed Tomography Angiography |
| PA | Pulmonary Artery |
| TA | Thoracic Aorta |
| CA | Coronary Artery |
| PE | Pulmonary Embolism |
| AD | Aortic Dissection |
| ACS | Acute Coronary Syndrome |
| DLP | Dose-Length Product |
| CS | Calcium Score |
| ATVS | Automatic tube voltage selection |
| SD | Standard Deviation |
| PT | Pulmonary Trunk |
| LPA | Left Pulmonary Artery |
| RPA | Right Pulmonary Artery |
| AO | Aortic Root |
| AA | Aortic Arch |
| DA | Descending Aorta |
| LMCA-P | Proximal Left Main Coronary Artery |
| LAD-M | Middle Left Anterior Descending |
| LAD-D | Distal Left Anterior Descending |
| LCX-M | Middle Left Circumflex |
| LCX-D | Distal Left Circumflex |
| RCA-P | Proximal Right Coronary Artery |
| RCA-M | Middle Right Coronary Artery |
| RCA-D | Distal Coronary Right Artery |
| ESM | Erector Spinae Muscle |
| PVAT | Perivascular Adipose Tissue |
| SNR | Signal-to-Noise Ratio |
| CNR | Contrast-to-Noise Ratio |
| CTDIvol | CT dose index |
| ED | Effective Dose |
References
- Raff, G.L.; Hoffmann, U.; Udelson, J.E. Trials of imaging use in the emergency Department for Acute Chest Pain. JACC Cardiovasc. Imaging 2017, 10, 338–349. [Google Scholar] [CrossRef]
- Russo, V.; Sportoletti, C.; Scalas, G.; Attinà, D.; Buia, F.; Niro, F.; Modolon, C.; De Luca, C.; Monteduro, F.; Lovato, L. The triple rule out CT in acute chest pain: A challenge for emergency radiologists? Emerg. Radiol. 2021, 28, 735–742. [Google Scholar] [CrossRef]
- Chae, M.K.; Kim, E.K.; Jung, K.Y.; Shin, T.G.; Sim, M.S.; Jo, I.J.; Song, K.J.; Chang, S.A.; Song, Y.B.; Hahn, J.Y.; et al. Triple rule-out computed tomography for risk stratification of patients with acute chest pain. J. Cardiovasc. Comput. Tomogr. 2016, 10, 291–300. [Google Scholar] [CrossRef]
- DeLaney, M.C.; Neth, M.; Thomas, J.J. Chest pain triage: Current trends in the emergency departments in the United States. J. Nucl. Cardiol. 2017, 24, 2004–2011. [Google Scholar] [CrossRef]
- Hollander, J.E.; Chang, A.M. Triple rule out CTA scans or the right test for the right patient. JACC Cardiovasc. Imaging 2015, 8, 826–827. [Google Scholar] [CrossRef]
- Burris, A.C., 2nd; Boura, J.A.; Raff, G.L.; Chinnaiyan, K.M. Triple rule out versus coronary CT angiography in patients with acute chest pain: Results from the ACIC consortium. JACC Cardiovasc. Imaging 2015, 8, 817–825. [Google Scholar] [CrossRef]
- Ketelsen, D.; Luetkhoff, M.H.; Thomas, C.; Werner, M.; Buchgeister, M.; Tsiflikas, I.; Reimann, A.; Burgstahler, C.; Kopp, A.F.; Claussen, C.D.; et al. Estimation of the radiation exposure of a chest pain protocol with ECG-gating in dual-source computed tomography. Eur. Radiol. 2009, 19, 37–41. [Google Scholar] [CrossRef]
- Nguyen, P.K.; Lee, W.H.; Li, Y.F.; Hong, W.X.; Hu, S.; Chan, C.; Liang, G.; Nguyen, I.; Ong, S.G.; Churko, J.; et al. Assessment of the radiation effects of cardiac CT angiography using protein and genetic biomarkers. JACC Cardiovasc. Imaging 2015, 8, 873–884. [Google Scholar] [CrossRef]
- Si-Mohamed, S.; Greffier, J.; Bobbia, X.; Larbi, A.; Delicque, J.; Khasanova, E.; Beregi, J.P.; Macri, F. Diagnostic performance of a low dose triple rule-out CT angiography using SAFIRE in emergency department. Diagn. Interv. Imaging 2017, 98, 881–891. [Google Scholar] [CrossRef]
- Takx, R.A.P.; Krissak, R.; Fink, C.; Bachmann, V.; Henzler, T.; Meyer, M.; Nance, J.W.; Schoenberg, S.O.; Apfaltrer, P. Low-tube-voltage selection for triple-rule-out CTA: Relation to patient size. Eur. Radiol. 2017, 27, 2292–2297. [Google Scholar] [CrossRef]
- Gruettner, J.; Fink, C.; Walter, T.; Meyer, M.; Apfaltrer, P.; Schoepf, U.J.; Saur, J.; Sueselbeck, T.; Traunwieser, D.; Takx, R.; et al. Coronary computed tomography and triple rule out CT in patients with acute chest pain and an intermediate cardiac risk profile. Part 1: Impact on patient management. Eur. J. Radiol. 2013, 82, 100–105. [Google Scholar] [CrossRef]
- Wong, D.T.; Soh, S.Y.; Ko, B.S.; Cameron, J.D.; Crossett, M.; Nasis, A.; Troupis, J.; Meredith, I.T.; Seneviratne, S.K. Superior CT coronary angiography image quality at lower radiation exposure with second generation 320-detector row CT in patients with elevated heart rate: A comparison with first generation 320-detector row CT. Cardiovasc. Diagn. Ther. 2014, 4, 299–306. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Q.; Li, J.; Jia, Y.; Yang, Q.; He, T. Triple-rule-out CT angiography using two axial scans with 16 cm wide-detector for radiation dose reduction. Eur. Radiol. 2018, 28, 4654–4661. [Google Scholar] [CrossRef]
- Ripley, D.P.; Uddin, A.; Greenwood, J.P. Greenwood, CT triple rule-out examination: Higher radiation dose without clinical benefit. BMJ 2013, 347, f5118. [Google Scholar] [CrossRef]
- Jin, L.; Gao, Y.; Shan, Y.; Sun, Y.; Li, M.; Wang, Z. Qualitative and quantitative image analysis of 16 cm wide-coverage computed tomography compared to new-generation dual-source CT. J. Xray Sci. Technol. 2020, 28, 527–539. [Google Scholar] [CrossRef]
- Jin, L.; Jie, B.; Gao, Y.; Jiang, A.; Weng, T.; Li, M. Low dose contrast media in step-and-shoot coronary angiography with third-generation dual-source computed tomography: Feasibility of using 30 mL of contrast media in patients with body surface area <1.7 m2. Quant. Imaging Med. Surg. 2021, 11, 2598–2609. [Google Scholar] [CrossRef]
- Jin, L.; Gao, Y.; Sun, Y.; Li, C.; Gao, P.; Zhao, W.; Li, M. Contrast medium administration with a body surface area protocol in step-and-shoot coronary computed tomography angiography with dual-source scanners. Sci. Rep. 2020, 10, 16690. [Google Scholar] [CrossRef]
- Hou, D.J.; Tso, D.K.; Davison, C.; Inacio, J.; Louis, L.J.; Nicolaou, S.; Reimann, A.J. Clinical utility of ultra high pitch dual source thoracic CT imaging of acute pulmonary embolism in the emergency department: Are we one step closer towards a non-gated triple rule out? Eur. J. Radiol. 2013, 82, 1793–1798. [Google Scholar] [CrossRef]
- Lu, G.M.; Luo, S.; Meinel, F.G.; McQuiston, A.D.; Zhou, C.S.; Kong, X.; Zhao, Y.E.; Zheng, L.; Schoepf, U.J.; Zhang, L.J. High-pitch computed tomography pulmonary angiography with iterative reconstruction at 80 kVp and 20 mL contrast agent volume. Eur. Radiol. 2014, 24, 3260–3268. [Google Scholar] [CrossRef]
- Spearman, J.V.; Schoepf, U.J.; Rottenkolber, M.; Driesser, I.; Canstein, C.; Thierfelder, K.M.; Krazinski, A.W.; De Cecco, C.N.; Meinel, F.G. Effect of automated attenuation-based tube voltage selection on radiation dose at CT: An observational study on a global scale. Radiology 2016, 279, 167–174. [Google Scholar] [CrossRef]
- Mangold, S.; Wichmann, J.L.; Schoepf, U.J.; Poole, Z.B.; Canstein, C.; Varga-Szemes, A.; Caruso, D.; Bamberg, F.; Nikolaou, K.; De Cecco, C.N. Automated tube voltage selection for radiation dose and contrast medium reduction at coronary CT angiography using 3(rd) generation dual-source CT. Eur. Radiol. 2016, 26, 3608–3616. [Google Scholar] [CrossRef] [PubMed]
- Vonder, M.; Pelgrim, G.J.; Huijsse, S.E.; Meyer, M.; Greuter, M.J.; Henzler, T.; Flohr, T.G.; Oudkerk, M.; Vliegenthart, R. Feasibility of spectral shaping for detection and quantification of coronary calcifications in ultra-low dose CT. Eur. Radiol. 2017, 27, 2047–2054. [Google Scholar] [CrossRef] [PubMed]
- Tesche, C.; De Cecco, C.N.; Schoepf, U.J.; Duguay, T.M.; Albrecht, M.H.; Caruso, D.; Varga-Szemes, A.; Lesslie, V.W.; Ebersberger, U.; Canstein, C.; et al. Iterative beam-hardening correction with advanced modeled iterative reconstruction in low voltage CT coronary calcium scoring with tin filtration: Impact on coronary artery calcium quantification and image quality. J. Cardiovasc. Comput. Tomogr. 2017, 11, 354–359. [Google Scholar] [CrossRef]
- Wichmann, J.L.; Katzberg, R.W.; Litwin, S.E.; Zwerner, P.L.; De Cecco, C.N.; Vogl, T.J.; Costello, P.; Schoepf, U.J. Contrast-induced nephropathy. Circulation 2015, 132, 1931–1936. [Google Scholar] [CrossRef] [PubMed]
- Halpern, E.J. Triple-rule-out CT angiography for evaluation of acute chest pain and possible acute coronary syndrome. Radiology 2009, 252, 332–345. [Google Scholar] [CrossRef]
- Abbara, S.; Blanke, P.; Maroules, C.D.; Cheezum, M.; Choi, A.D.; Han, B.K.; Marwan, M.; Naoum, C.; Norgaard, B.L.; Rubinshtein, R.; et al. SCCT guidelines for the performance and acquisition of coronary computed tomographic angiography: A report of the society of cardiovascular Computed Tomography Guidelines Committee: Endorsed by the North American Society for Cardiovascular Imaging (NASCI). J. Cardiovasc. Comput. Tomogr. 2016, 10, 435–449. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).