CT-Guided Radiofrequency Thermal Ablation for the Treatment of Atypical, Early-Onset Osteoid Osteoma in Children Younger than 4 Years Old: Single-Institution Experience and Literature Review
Abstract
:1. Introduction
2. Case Series Report from a Single Institution: Our Experience
2.1. Outcome Assessment
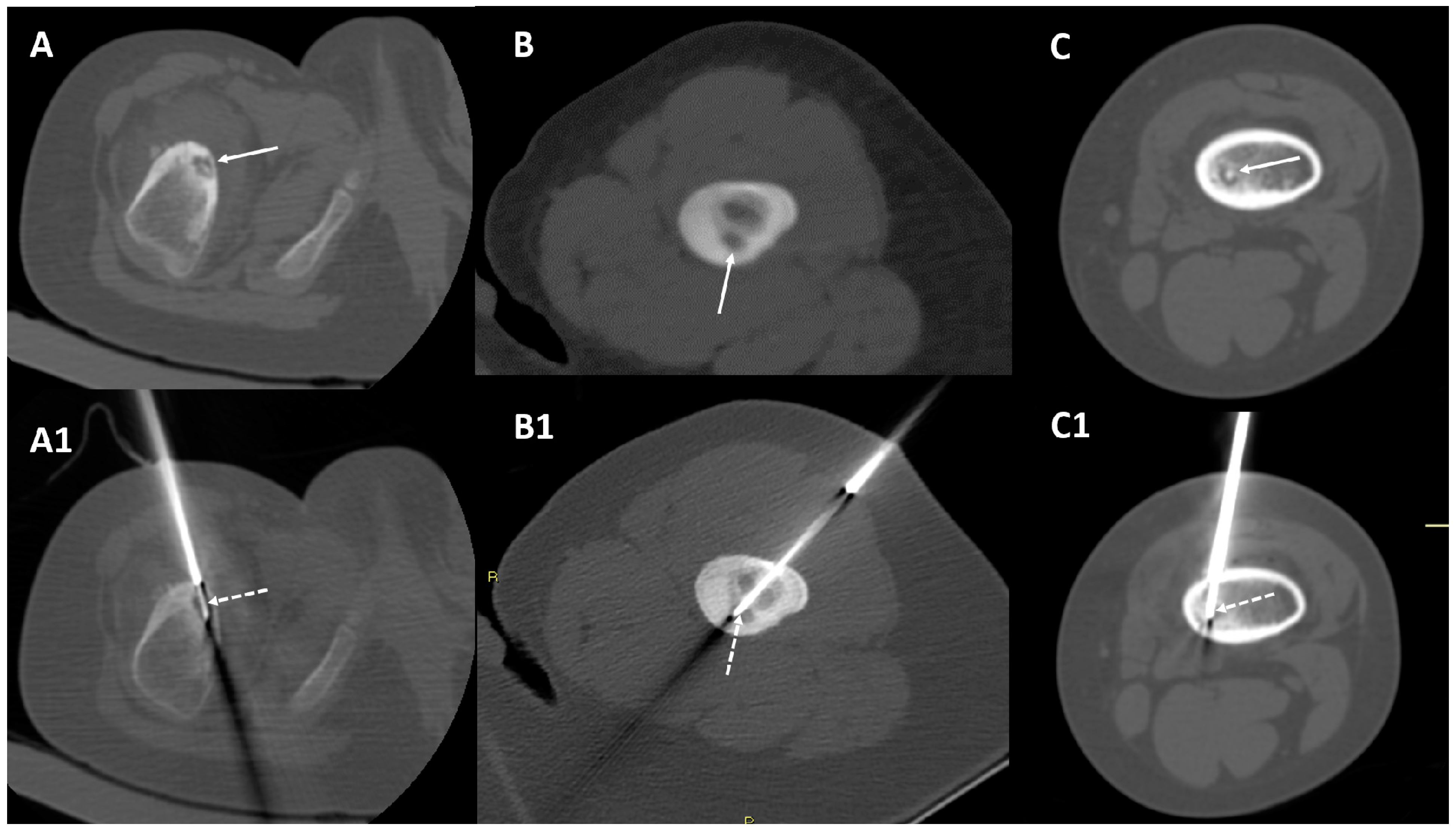
2.2. CT-Guided Radiofrequency Ablation
2.3. Patients Included in the Series (RFA for OO < 4 Years Old)
3. Literature Review
3.1. Literature Search Strategy
3.2. Literature Search Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jaffe, H. Osteoid osteoma: A benign osteoblastic tumor composed of osteoid and atypical bone. Arch Surg. 1935, 31, 709–728. [Google Scholar] [CrossRef]
- Lam, Y. Bone Tumors: Benign Bone Tumors. FP Essent. 2020, 493, 11–21. [Google Scholar] [PubMed]
- Frassica, F.J.; Waltrip, R.L.; Sponseller, P.D.; Ma, L.D.; McCarthy, E.F. Clinicopathologic features and treatment of osteoid osteoma and osteoblastoma in children and adolescents. Orthop. Clin. North Am. 1996, 27, 559–574. [Google Scholar] [CrossRef]
- Lee, E.H.; Shafi, M.; Hui, J.H.P. Osteoid osteoma: A current review. J. Pediatr. Orthop. 2006, 26, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Boscainos, P.J.; Cousins, G.R.; Kulshreshtha, R.; Oliver, T.B.; Papagelopoulos, P.J. Osteoid osteoma. Orthopedics 2013, 36, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.H.; Shankman, S. Osteoid osteoma: Radiologic and pathologic correlation. Skeletal Radiol. 1992, 21, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Gitelis, S.; Schajowicz, F. Osteoid osteoma and osteoblastoma. Orthop. Clin. North Am. 1989, 20, 313–325. [Google Scholar]
- Iyer, R.S.; Chapman, T.; Chew, F.S. Pediatric bone imaging: Diagnostic imaging of osteoid osteoma. AJR Am. J. Roentgenol. 2012, 198, 1039–1052. [Google Scholar] [CrossRef]
- Papathanassiou, Z.G.; Megas, P.; Petsas, T.; Papachristou, D.J.; Nilas, J.; Siablis, D. Osteoid osteoma:diagnosis and treatment. Orthopedics 2008, 31, 1118. [Google Scholar] [CrossRef]
- Parmeggiani, A.; Martella, C.; Ceccarelli, L.; Miceli, M.; Spinnato, P.; Facchini, G. Osteoid osteoma: Which is the best mininvasive treatment option? Eur. J. Orthop. Surg. Traumatol. 2021, 31, 1611–1624. [Google Scholar] [CrossRef]
- Spinnato, P.; Rimondi, E.; Facchini, G. Percutaneous CT-Guided Biopsy of the Craniovertebral Junction: Safety, Diagnostic Yield, and Technical Notes. Diagnostics 2022, 12, 168. [Google Scholar] [CrossRef] [PubMed]
- Tomasian, A.; Hillen, T.J.; Jennings, J.W. Percutaneous CT-Guided Skull Biopsy: Feasibility, Safety, and Diagnostic Yield. AJNR Am. J. Neuroradiol. 2019, 40, 309–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hiramatsu, M.; Nakayama, R.; Kasahara, T.; Nakagawa, R.; Hirozane, T.; Yamaguchi, S.; Mori, T.; Asano, N.; Okita, H.; Nakamura, M.; et al. Intramedullary osteoid osteoma in the humerus of a toddler-A case report and review of the literature. Radiol. Case Rep. 2022, 17, 3748–3753. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Solbiati, L.; Brace, C.L.; Breen, D.J.; Callstrom, M.R.; Charboneau, J.W.; Chen, M.-H.; Choi, B.I.; de Baère, T.; Dodd, G.D., III; et al. Image-guided tumor ablation: Standardization of terminology and reporting criteria--a 10-year update. J. Vasc. Interv. Radiol. 2014, 25, 1691–1705. [Google Scholar] [CrossRef]
- Bhat, I.; Zerin, J.M.; Bloom, D.A.; Mooney, J.F. 3rd Unusual presentation of osteoid osteoma mimicking osteomyelitis in a 27-month-old infant. Pediatr. Radiol. 2003, 33, 425–428. [Google Scholar] [CrossRef]
- Ekström, W.; Söderlund, V.; Brosjö, O. Osteoid osteoma in a 1-year-old boy-a case report. Acta Orthop. 2006, 77, 686–688. [Google Scholar] [CrossRef]
- Virayavanich, W.; Singh, R.; O’Donnell, R.J.; Horvai, A.E.; Goldsby, R.E.; Link, T.M. Osteoid osteoma of the femur in a 7-month-old infant treated with radiofrequency ablation. Skeletal Radiol. 2010, 39, 1145–1149. [Google Scholar] [CrossRef] [Green Version]
- Simon, A.L.; Ilharreborde, B.; Litzelmann, E.; Mazda, K.; Penneçot, G.F. Eight-Year Natural Course of a Femoral Neck Deformity Secondary to an Osteoid Osteoma Discovered at Fourteen Months of Age: A Case Report. JBJS Case Connect. 2013, 3, e119. [Google Scholar] [CrossRef]
- Sahin, C.; Oc, Y.; Ediz, N.; Hasanefendioglu Bayrak, A. Successful percutaneous treatment of osteoid osteoma in a 13 month-old boy with radiofrequency ablation under CT guidance. Acta Orthop. Traumatol. Turc. 2019, 53, 233–237. [Google Scholar] [CrossRef]
- Laliotis, N.; Chrysanthou, C.; Konstantinidis, P.; Papadopoulou, L. Osteoid Osteoma in Children Younger than 3 Years of Age. Case Rep. Orthop. 2019, 9, 8201639. [Google Scholar] [CrossRef]
- Cotta, A.; de Castro, R.C.R.; Paim, J.F.; Fiuza, L.S.; Lyra, M.H.F. Limb Length Discrepancy on an 11-Month-Old Boy with Osteoid Osteoma. Rev. Bras. Ortop. (Sao Paulo) 2019, 54, 210–213. [Google Scholar] [CrossRef]
- Gupta, S.; Sinha, S.; Narang, A.; Kanojia, R.K. Intramedullary osteoid osteoma in an 11-month-old child. J. Postgrad. Med. 2020, 66, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.C.; Monroe, E.J.; McKay, T.; Kanal, K.M.; Shivaram, G. Pediatric Percutaneous Osteoid Osteoma Ablation: Cone-Beam CT with Fluoroscopic Overlay versus Conventional CT Guidance. Cardiovasc. Interv. Radiol. 2017, 40, 1593–1599. [Google Scholar] [CrossRef] [PubMed]
- Gangi, A.; Alizadeh, H.; Wong, L.; Buy, X.; Dietemann, J.L.; Roy, C. Osteoid osteoma: Percutaneous laser ablation and follow-up in 114 patients. Radiology 2007, 242, 293–301. [Google Scholar] [CrossRef]
- Filippiadis, D.; Stefanou, D.; Mazioti, A.; Foti, L.; Tsochatzis, A.; Kelekis, N.; Kelekis, A. Computed tomography guided radiofrequency ablation of osteoid osteoma in children: A single center’s experience. Skeletal Radiol. 2022, 51, 855–861. [Google Scholar] [CrossRef]
- Rimondi, E.; Mavrogenis, A.F.; Rossi, G.; Ciminari, R.; Malaguti, C.; Tranfaglia, C.; Vanel, D.; Ruggieri, P. Radiofrequency ablation for non-spinal osteoid osteomas in 557 patients. Eur. Radiol. 2012, 22, 181–188. [Google Scholar] [CrossRef]
- Pipola, V.; Tedesco, G.; Spinnato, P.; Facchini, G.; Gala, R.B.; Bandiera, S.; Bròdano, G.B.; Terzi, S.; Ghermandi, R.; Evangelisti, G.; et al. Surgery Versus Radiofrequency Ablation in the Management of Spinal Osteoid Osteomas: A Spine Oncology Referral Center Comparison Analysis of 138 Cases. World Neurosurg. 2021, 145, e298–e304. [Google Scholar] [CrossRef]
- Albisinni, U.; Facchini, G.; Spinnato, P.; Gasbarrini, A.; Bazzocchi, A. Spinal osteoid osteoma: Efficacy and safety of radiofrequency ablation. Skeletal Radiol. 2017, 46, 1087–1094. [Google Scholar] [CrossRef]
- Somma, F.; Stoia, V.; D’Angelo, R.; Fiore, F. Imaging-guided radiofrequency ablation of osteoid osteoma in typical and atypical sites: Long term follow up. PLoS ONE 2021, 16, e0248589. [Google Scholar] [CrossRef]
- Arrigoni, F.; Napoli, A.; Bazzocchi, A.; Zugaro, L.; Scipione, R.; Bruno, F.; Palumbo, P.; Anzidei, M.; Mercatelli, D.; Gravina, G.L.; et al. Magnetic-resonance-guided focused ultrasound treatment of non-spinal osteoid osteoma in children: Multicentre experience. Pediatr. Radiol. 2019, 49, 1209–1216. [Google Scholar] [CrossRef]


| Age at Onset of Symptoms (Months) | Age at Time of Treatment (Months) | Main Clinical/Anamnestic Data | Osteoid Osteoma Location | Nidus Max Diameter | Electrode Active Tip | Technical Success | Clinical Success | Histopathological Diagnosis | First Follow-Up | Last Follow-Up |
|---|---|---|---|---|---|---|---|---|---|---|
| 30 | 38 | Pain at night and limping | Femur proximal metaphysis, endosteal | 9 mm | 10 mm | Yes | Yes | Non-diagnostic | 3 months after treatment: asymptomatic | 15 months after treatment: asymptomatic |
| 28 | 43 | Pain at night | Tibia proximal metaphysis, endosteal | 10 mm | 15 mm | Yes | Yes | Non-diagnostic | 1 month after treatment: asymptomatic | 6 months after treatment: asymptomatic |
| 27 | 33 | Continuous pain worsened at night | Femur distal metaphysis, intramedullary | 10 mm | 15 mm | Yes | Yes | Non-diagnostic | 6 months after treatment: asymptomatic | N/A |
| 30 | 35 | Continuous pain worsened at night | Tibia proximal metaphysis, cortical | 9 mm | 10 mm | Yes | Yes | Non-diagnostic | 3 months after treatment: asymptomatic | 18 months after treatment: asymptomatic |
| 39 | 46 | Pain at night and muscular hypotrophy | Femur proximal metaphysis, cortical-periosteal | 10 mm | 15 mm | Yes | Yes | Non-diagnostic | 6 months after treatment: asymptomatic | N/A |
| 22 | 37 | Pain at night | Humerus diaphysis, endosteal | 9 mm | 10 mm | Yes | Yes | Osteoid osteoma | 6 months after treatment: asymptomatic | N/A |
| 14 | 22 | Pain at night | Femur proximal metaphysis, endosteal | 9 mm | 10 mm | Yes | Yes | Osteoid osteoma | 12 months after treatment: asymptomatic | N/A |
| 20 | 28 | Continuous pain and functional limitation | Femur proximal metaphysis, endosteal | 7 mm | 10 mm | Yes | Yes | Non-diagnostic | 6 months after treatment: asymptomatic | 8 years after treatment: asymptomatic |
| 35 | 42 | Continuous pain worsened at night | Tibia diaphysis, cortical-periosteal | 12 mm | 15 mm | Yes | Yes | Osteoid osteoma | 4 months after treatment: asymptomatic | 7 years after treatment: asymptomatic |
| 34 | 39 | Pain at night | Femur proximal metaphysis, cortical-periosteal | 12 mm | 15 mm | Yes | Yes | Non-diagnostic | 4 months after treatment: asymptomatic | 12 months after treatment: asymptomatic |
| 32 | 34 | Pain at night and restlessness | Femur diaphysis, cortical-endosteal | 12 mm | 15 mm | Yes | Yes | Osteoid osteoma | 6 months after treatment: asymptomatic | N/A |
| 24 | 27 | Pain at night | Femur proximal metaphysis, cortical-periosteal | 6 mm | 10 mm | Yes | Yes | Non-diagnostic | 4 months after treatment: asymptomatic | 7 months after treatment: asymptomatic |
| First Author, Year, Reference Number | Study Design | Number of Patients | Patient Age | Diagnosis | Location | Clinical Findings | Treatment | Follow-Up | Recurrence |
|---|---|---|---|---|---|---|---|---|---|
| Bhat, 2003 Ref. [15] | Case report | 1 | 27 months | OO | Femur | Limp on the medial distal thigh, which was swollen and tender | N/A | N/A | N/A |
| Ekstrom, 2006 Ref. [16] | Case report | 1 | 7 months | OO | Femur | Restlessness and showed signs of pain at night but not during the day | CT-guided RFA | 6 months | No |
| Virayavanich, 2010 Ref. [17] | Case report | 1 | 7 months | OO | Femur | Decreased use of the right lower extremity due to pain | CT-guided RFA | 3 months | Yes |
| Simon, 2013 Ref. [18] | Case report | 1 | 14 months | OO | Femur | Pain, stiffness of the hip, and atrophy; coxa magna, limb discrepancy. | CT-guided RFA | 8 years | No |
| Sahin, 2018 Ref. [19] | Case report | 1 | 13 months | OO | Tibia | Restlessness for prior 6 months | CT-guided RFA | 16 months | No |
| Laliotis, 2019 Ref. [20] | Case series | 4 | 18 months–3 years | OO | 2 femur, 1 tibia, 1 fibula | Pain, functional limitation, atrophy of the muscles (only femur) | Curettage for tibia and fibula, CT-guided RF for femur | 12 months | No |
| Cotta, 2019 Ref. [21] | Case report | 1 | 11 months | OO | Tibia | Limb asymmetry | N/A | N/A | N/A |
| Gupta, 2020 Ref. [22] | Case report | 1 | 11 months | OO | Tibia | Inconsolable crying, swelling, and decreased use of left lower limb since the age of 8 months | Curettage | 12 months | No |
| Hiramatsu, 2022 Ref. [13] | Case report | 1 | 21 months | OO | Humerus | Affecter arm thinner than contralateral, restlessness for prior 5 months | Curettage | 2 years | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papalexis, N.; Peta, G.; Ponti, F.; Tuzzato, G.; Colangeli, M.; Facchini, G.; Spinnato, P. CT-Guided Radiofrequency Thermal Ablation for the Treatment of Atypical, Early-Onset Osteoid Osteoma in Children Younger than 4 Years Old: Single-Institution Experience and Literature Review. Diagnostics 2022, 12, 2812. https://doi.org/10.3390/diagnostics12112812
Papalexis N, Peta G, Ponti F, Tuzzato G, Colangeli M, Facchini G, Spinnato P. CT-Guided Radiofrequency Thermal Ablation for the Treatment of Atypical, Early-Onset Osteoid Osteoma in Children Younger than 4 Years Old: Single-Institution Experience and Literature Review. Diagnostics. 2022; 12(11):2812. https://doi.org/10.3390/diagnostics12112812
Chicago/Turabian StylePapalexis, Nicolas, Giuliano Peta, Federico Ponti, Gianmarco Tuzzato, Marco Colangeli, Giancarlo Facchini, and Paolo Spinnato. 2022. "CT-Guided Radiofrequency Thermal Ablation for the Treatment of Atypical, Early-Onset Osteoid Osteoma in Children Younger than 4 Years Old: Single-Institution Experience and Literature Review" Diagnostics 12, no. 11: 2812. https://doi.org/10.3390/diagnostics12112812
APA StylePapalexis, N., Peta, G., Ponti, F., Tuzzato, G., Colangeli, M., Facchini, G., & Spinnato, P. (2022). CT-Guided Radiofrequency Thermal Ablation for the Treatment of Atypical, Early-Onset Osteoid Osteoma in Children Younger than 4 Years Old: Single-Institution Experience and Literature Review. Diagnostics, 12(11), 2812. https://doi.org/10.3390/diagnostics12112812








