The Value of Optical Coherence Tomography Angiography (OCT-A) in Neurological Diseases
Abstract
:1. Introduction
2. Method
3. OCT-A Findings in Neurological Diseases
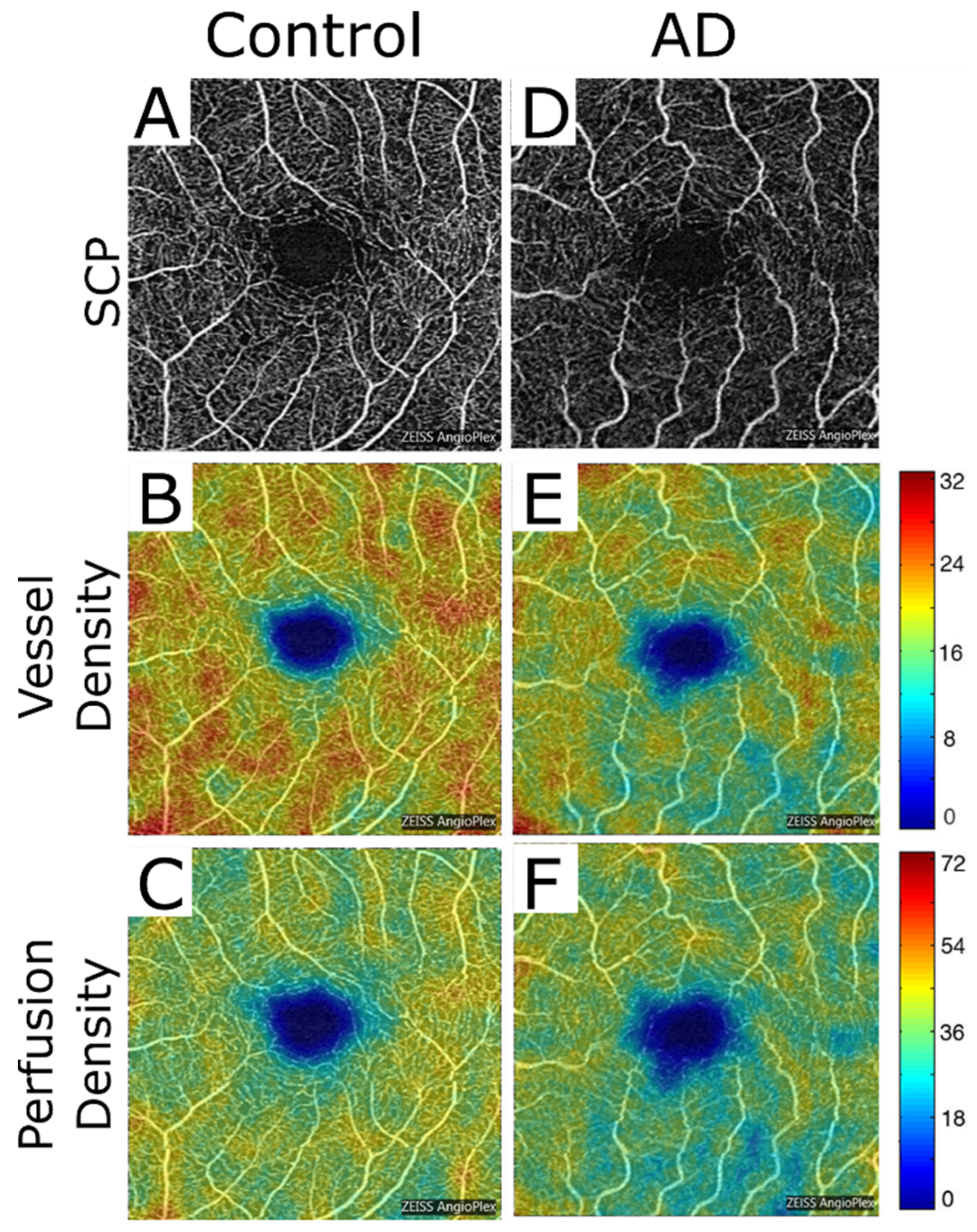
3.1. OCT-A in Alzheimer’s Disease (AD)
3.2. Parkinson’s Disease (PD)
3.3. Multiple Sclerosis (MS)
3.4. Glaucoma
3.5. Anterior Ischemic Optic Neuropathy (AION)
3.6. Papilloedema
3.7. Migraine
3.8. Stroke
3.9. Diabetic Neuropathy (Neurodegenerative Component)
3.10. Epilepsy
3.11. Leber’s Hereditary Optic Neuropathy (LHON)
3.12. Amyotrophic Lateral Sclerosis (ALS)
3.13. Wolfram Syndrome
3.14. Susac Syndrome
3.15. Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL)
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- London, A.; Benhar, I.; Schwartz, M. The retina as a window to the brain—From eye research to CNS disorders. Nat. Rev. Neurol. 2013, 9, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Tsokolas, G.; Tsaousis, K.T.; Diakonis, V.F.; Matsou, A.; Tyradellis, S. Optical coherence tomography angiography in neurodegenerative diseases: A review. Eye Brain 2020, 12, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Gabriele, M.L.; Wollstein, G.; Ishikawa, H.; Kagemann, L.; Xu, J.; Folio, L.S.; Schuman, J.S. Optical coherence tomography: History, current status, and laboratory work. Investig. Opthalmol. Vis. Sci. 2011, 52, 2425–2436. [Google Scholar] [CrossRef] [PubMed]
- Chan, N.C.Y.; Chan, C.K.M. The use of optical coherence tomography in neuro-ophthalmology. Curr. Opin. Ophthalmol. 2017, 28, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Zivadinov, R.; Ramanathan, M.; Weinstock-Guttman, B. Optical coherence tomography and neurodegeneration: Are eyes the windows to the brain? Expert Rev. Neurother. 2016, 16, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Lumbroso, B.; Jia, Y.; Waheed, N.K. Optical Coherencre Tomography Angiography of the Eye; SLACK Incorporated: West Deptford, NJ, USA, 2017. [Google Scholar]
- Murueta-Goyena, A.; Barrenechea, M.; Erramuzpe, A.; Teijeira-Portas, S.; Pengo, M.; Ayala, U.; Romero-Bascones, D.; Acera, M.; Del Pino, R.; Gómez-Esteban, J.C.; et al. Foveal remodeling of retinal microvasculature in Parkinson’s Disease. Front. Neurosci. 2021, 15, 708700. [Google Scholar] [CrossRef]
- Mirzaei, N.; Shi, H.; Oviatt, M.; Doustar, J.; Rentsendorj, A.; Fuchs, D.-T.; Sheyn, J.; Black, K.L.; Koronyo, Y.; Koronyo-Hamaoui, M. Alzheimer’s retinopathy: Seeing disease in the eyes. Front. Neurosci. 2020, 14, 921. [Google Scholar] [CrossRef]
- Koronyo-Hamaoui, M.; Koronyo, Y.; Ljubimov, A.V.; Miller, C.A.; Ko, M.K.; Black, K.L.; Schwartz, M.; Farkas, D.L. Identification of amyloid plaques in retinas from Alzheimer’s patients and noninvasive in vivo optical imaging of retinal plaques in a mouse model. Neuroimage 2011, 54 (Suppl. 1), S204–S217. [Google Scholar] [CrossRef] [Green Version]
- Schön, C.; Hoffmann, N.A.; Ochs, S.M.; Burgold, S.; Filser, S.; Steinbach, S.; Seeliger, W.M.; Arzberger, T.; Goedert, M.; Kretzschmar, H.A.; et al. Long-term in vivo imaging of fibrillar tau in the retina of P301S transgenic mice. PLoS ONE 2012, 7, e53547. [Google Scholar] [CrossRef]
- Shi, H.; Koronyo, Y.; Rentsendorj, A.; Fuchs, D.-T.; Sheyn, J.; Black, K.L.; Mirzaei, N.; Koronyo-Hamaoui, M. Retinal vasculopathy in Alzheimer’s disease. Front. Neurosci. 2021, 15, 731614. [Google Scholar] [CrossRef]
- Czakó, C.; Kovács, T.; Ungvari, Z.; Csiszar, A.; Yabluchanskiy, A.; Conley, S.; Csipo, T.; Lipecz, A.; Horváth, H.; Sándor, G.L.; et al. Retinal biomarkers for Alzheimer’s disease and vascular cognitive impairment and dementia (VCID): Implication for early diagnosis and prognosis. GeroScience 2020, 42, 1499–1525. [Google Scholar] [CrossRef] [PubMed]
- Rifai, O.M.; McGrory, S.; Robbins, C.B.; Grewal, D.S.; Liu, A.; Fekrat, S.; MacGillivray, T.J. The application of optical coherence tomography angiography in Alzheimer’s disease: A systematic review. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2021, 13, e12149. [Google Scholar] [CrossRef] [PubMed]
- O’Bryhim, B.E.; Lin, J.B.; Van Stavern, G.P.; Apte, R.S. OCT angiography findings in preclinical Alzheimer’s disease: 3-year follow-up. Ophthalmology 2021, 128, 1489–1491. [Google Scholar] [CrossRef]
- Yoon, S.P.; Grewal, D.; Thompson, A.C.; Polascik, B.; Dunn, C.; Burke, J.R.; Fekrat, S. Retinal microvascular and neurodegenerative changes in Alzheimer’s disease and mild cognitive impairment compared with control participants. Ophthalmol. Retin. 2019, 3, 489–499. [Google Scholar] [CrossRef]
- Garcia-Martin, E.; Bambo, M.P.; Marques, M.L.; Satue, M.; Otin, S.; Larrosa, J.M.; Polo, V.; Pablo, L.E. Ganglion cell layer measurements correlate with disease severity in patients with Alzheimer’s disease. Acta Ophthalmol. 2016, 94, e454–e459. [Google Scholar] [CrossRef]
- Zou, J.; Liu, K.; Li, F.; Xu, Y.; Shen, L.; Xu, H. Combination of optical coherence tomography (OCT) and OCT angiography increases diagnostic efficacy of Parkinson’s disease. Quant. Imaging Med. Surg. 2020, 10, 1930–1939. [Google Scholar] [CrossRef]
- Rascunà, C.; Russo, A.; Terravecchia, C.; Castellino, N.; Avitabile, T.; Bonfiglio, V.; Fallico, M.; Chisari, C.G.; Cicero, C.E.; Grillo, M.; et al. Retinal thickness and microvascular pattern in early Parkinson’s disease. Front. Neurol. 2020, 11, 533375. [Google Scholar] [CrossRef]
- Robbins, C.B.; Thompson, A.C.; Bhullar, P.K.; Koo, H.Y.; Agrawal, R.; Soundararajan, S.; Yoon, S.P.; Polascik, B.W.; Scott, B.L.; Grewal, D.S.; et al. Characterization of retinal microvascular and choroidal structural changes in Parkinson disease. JAMA Ophthalmol. 2021, 139, 182–188. [Google Scholar] [CrossRef]
- Robbins, C.B.; Grewal, D.S.; Thompson, A.C.; Soundararajan, S.; Yoon, S.P.; Polascik, B.W.; Scott, B.L.; Fekrat, S. Identifying peripapillary radial capillary plexus alterations in Parkinson’s disease using OCT angiography. Ophthalmol. Retin. 2021, 6, 29–36. [Google Scholar] [CrossRef]
- Yilmaz, H.; Ersoy, A.; Icel, E. Assessments of vessel density and foveal avascular zone metrics in multiple sclerosis: An optical coherence tomography angiography study. Eye 2019, 34, 771–778. [Google Scholar] [CrossRef]
- Farci, R.; Carta, A.; Cocco, E.; Frau, J.; Fossarello, M.; Diaz, G. Optical coherence tomography angiography in multiple sclerosis: A cross-sectional study. PLoS ONE 2020, 15, e0236090. [Google Scholar] [CrossRef] [PubMed]
- Cordon, B.; Vilades, E.; Orduna, E.; Satue, M.; Perez-Velilla, J.; Sebastian, B.; Polo, V.; Larrosa, J.M.; Pablo, L.E.; Garcia-Martin, E. Angiography with optical coherence tomography as a biomarker in multiple sclerosis. PLoS ONE 2020, 15, e0243236. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, G.; Carotenuto, A.; Montorio, D.; Petracca, M.; Moccia, M.; Melenzane, A.; Tranfa, F.; Lamberti, A.; Spiezia, A.L.; Servillo, G.; et al. Peripapillary vessel density as early biomarker in multiple sclerosis. Front. Neurol. 2020, 11, 542. [Google Scholar] [CrossRef] [PubMed]
- Aly, L.; Strauß, E.-M.; Feucht, N.; Weiß, I.; Berthele, A.; Mitsdoerffer, M.; Haass, C.; Hemmer, B.; Maier, M.; Korn, T.; et al. Optical coherence tomography angiography indicates subclinical retinal disease in neuromyelitis optica spectrum disorders. Mult. Scler. J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kleerekooper, I.; Houston, S.; Dubis, A.M.; Trip, S.A.; Petzold, A. Optical Coherence Tomography Angiography (OCTA) in multiple sclerosis and neuromyelitis optica spectrum disorder. Front. Neurol. 2020, 11, 604049. [Google Scholar] [CrossRef] [PubMed]
- Spain, R.I.; Liu, L.; Zhang, X.; Jia, Y.; Tan, O.; Bourdette, D.; Huang, D. Optical coherence tomography angiography enhances the detection of optic nerve damage in multiple sclerosis. Br. J. Ophthalmol. 2018, 102, 520–524. [Google Scholar] [CrossRef]
- Feucht, N.; Maier, M.; Lepennetier, G.; Pettenkofer, M.; Wetzlmair, C.; Daltrozzo, T.; Scherm, P.; Zimmer, C.; Hoshi, M.-M.; Hemmer, B.; et al. Optical coherence tomography angiography indicates associations of the retinal vascular network and disease activity in multiple sclerosis. Mult. Scler. J. 2018, 25, 224–234. [Google Scholar] [CrossRef]
- Jiang, H.; Gameiro, G.R.; Liu, Y.; Lin, Y.; Hernandez, J.; Deng, Y.; Gregori, G.; Delgado, S.; Wang, J. Visual function and disability are associated with increased retinal volumetric vessel density in patients with multiple sclerosis. Am. J. Ophthalmol. 2020, 213, 34–45. [Google Scholar] [CrossRef]
- Murphy, O.C.; Kwakyi, O.; Iftikhar, M.; Zafar, S.; Lambe, J.; Pellegrini, N.; Sotirchos, E.S.; Gonzalez-Caldito, N.; Ogbuokiri, E.; Filippatou, A.; et al. Alterations in the retinal vasculature occur in multiple sclerosis and exhibit novel correlations with disability and visual function measures. Mult. Scler. J. 2020, 26, 815–828. [Google Scholar] [CrossRef]
- WuDunn, D.; Takusagawa, H.L.; Sit, A.J.; Rosdahl, J.A.; Radhakrishnan, S.; Hoguet, A.; Han, Y.; Chen, T.C. OCT angiography for the diagnosis of glaucoma: A report by the American Academy of Ophthalmology. Ophthalmology 2021, 128, 1222–1235. [Google Scholar] [CrossRef]
- Ling, J.-W.; Yin, X.; Lu, Q.-Y.; Chen, Y.-Y.; Lu, P.-R. Optical coherence tomography angiography of optic disc perfusion in non-arteritic anterior ischemic optic neuropathy. Int. J. Ophthalmol. 2017, 10, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- Balducci, N.; Morara, M.; Veronese, C.; Barboni, P.; Casadei, N.L.; Savini, G.; Parisi, V.; Sadun, A.A.; Ciardella, A. Optical coherence tomography angiography in acute arteritic and non-arteritic anterior ischemic optic neuropathy. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 2255–2261. [Google Scholar] [CrossRef] [PubMed]
- Gaier, E.D.; Wang, M.; Gilbert, A.L.; Rizzo, J.F.; Cestari, D.M.; Miller, J.B. Quantitative analysis of optical coherence tomographic angiography (OCT-A) in patients with non-arteritic anterior ischemic optic neuropathy (NAION) corresponds to visual function. PLoS ONE 2018, 13, e0199793. [Google Scholar] [CrossRef] [PubMed]
- Mahdjoubi, A.; Haouas, M.; Caliot, J.; Jaillant, R.; Idlefqih, W.; Amari, F.Z.; Chahed, S. Optical coherence tomography angiography of macula in chronic anterior ischaemic optic neuropathy associated with giant cell arteritis. Neuro-Ophthalmol. 2018, 43, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Pierro, L.; Arrigo, A.; Aragona, E.; Cavalleri, M.; Bandello, F. Vessel density and vessel tortuosity quantitative analysis of arteritic and non-arteritic anterior ischemic optic neuropathies: An optical coherence tomography angiography study. J. Clin. Med. 2020, 9, 1094. [Google Scholar] [CrossRef] [PubMed]
- Rougier, M.-B.; Le Goff, M.; Korobelnik, J.-F. Optical coherence tomography angiography at the acute phase of optic disc edema. Eye Vis. 2018, 5, 15. [Google Scholar] [CrossRef]
- Fard, M.A.; Jalili, J.; Sahraiyan, A.; Khojasteh, H.; Hejazi, M.; Ritch, R.; Subramanian, P.S. Optical coherence tomography angiography in optic disc swelling. Am. J. Ophthalmol. 2018, 191, 116–123. [Google Scholar] [CrossRef]
- Fard, M.A.; Sahraian, A.; Jalili, J.; Hejazi, M.; Suwan, Y.; Ritch, R.; Subramanian, P.S. Optical coherence tomography angiography in papilledema compared with pseudopapilledema. Investig. Opthalmol. Vis. Sci. 2019, 60, 168–175. [Google Scholar] [CrossRef] [Green Version]
- Chonsui, M.; Le Goff, M.; Korobelnik, J.-F.; Rougier, M.-B. Quantitative analysis of radial peripapillary capillary network in patients with papilledema compared with healthy subjects using optical coherence tomography angiography. J. Neuro-Ophthalmol. 2021. [Google Scholar] [CrossRef]
- Puledda, F.; Messina, R.; Goadsby, P.J. An update on migraine: Current understanding and future directions. J. Neurol. 2017, 264, 2031–2039. [Google Scholar] [CrossRef] [Green Version]
- Güler, Ö.; Güler, M.; Tuğan Yıldız, C.B.; Hakkoymaz, H. Are retinal and peripapillary blood flows affected during migraine attack? Neuroophthalmology 2020, 44, 299–306. [Google Scholar] [CrossRef] [PubMed]
- van Dongen, R.M.; Haan, J. Symptoms related to the visual system in migraine. F1000Research 2019, 8, 1219. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.Y.; Phasukkijwatana, N.; Garrity, S.; Pineles, S.L.; Rahimi, M.; Sarraf, D.; Johnston, M.; Charles, A.; Arnold, A.C. Foveal and peripapillary vascular decrement in migraine with aura demonstrated by optical coherence tomography angiography. Investig. Opthalmol. Vis. Sci. 2017, 58, 5477–5484. [Google Scholar] [CrossRef] [Green Version]
- Bingöl Kızıltunç, P.; Özcan, G.; Özer, F.; Togay Işıkay, C.; Atilla, H. Evaluation of retinal vessel density and choriocapillaris flow in migraine patients with and without aura. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 2517–2521. [Google Scholar] [CrossRef] [PubMed]
- Özçift, S.G.; Aydın, E.; Eriş, E. Assessment of the choroidal thickness, central macular vascular and optic disk perfusion in migraine patients with optical coherence tomography angiography. Photodiagn. Photodyn. Ther. 2021, 35, 102397. [Google Scholar] [CrossRef]
- Hamurcu, M.S.; Gultekin, B.P.; Koca, S.; Ece, S.D. Evaluation of migraine patients with optical coherence tomography angiography. Int. Ophthalmol. 2021, 41, 3929–3933. [Google Scholar] [CrossRef]
- Kızıltunç, P.B.; Atilla, H. Vascular changes with optical coherence tomography angiography during aura of migraine: A case report. Eur. J. Ophthalmol. 2020, 31, NP54–NP57. [Google Scholar] [CrossRef]
- Katan, M.; Luft, A. Global burden of stroke. Semin. Neurol. 2018, 38, 208–211. [Google Scholar] [CrossRef] [Green Version]
- Barthels, D.; Das, H. Current advances in ischemic stroke research and therapies. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165260. [Google Scholar] [CrossRef]
- Kuriakose, D.; Xiao, Z. Pathophysiology and treatment of stroke: Present status and future perspectives. Int. J. Mol. Sci. 2020, 21, 7609. [Google Scholar] [CrossRef]
- Rim, T.H.; Teo, A.W.J.; Yang, H.H.S.; Cheung, C.Y.; Wong, T.Y. Retinal vascular signs and cerebrovascular diseases. J. Neuro-Ophthalmol. 2020, 40, 44–59. [Google Scholar] [CrossRef] [PubMed]
- Avery, M.B.; Magal, I.; Kherani, A.; Mitha, A.P. Risk of stroke in patients with ocular arterial occlusive disorders: A retrospective Canadian study. J. Am. Heart Assoc. 2019, 8, e010509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, B.; Hu, Y.; Ma, G.; Xiao, Y.; Zhang, B.; Liang, Y.; Zhong, P.; Zeng, X.; Lin, Z.; Kong, H.; et al. Reduced retinal microvascular perfusion in patients with stroke detected by optical coherence tomography angiography. Front. Aging Neurosci. 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Molero-Senosiain, M.; Vidal-Villegas, B.; Pascual-Prieto, J.; Valor-Suarez, C.; Saenz-Frances, F.; Santos-Bueso, E. Correlation between retrograde trans-synaptic degeneration of ganglion cells and optical coherence tomography angiography following ischemic stroke. Cureus 2021, 13, e19788. [Google Scholar] [CrossRef]
- Haritoglou, C.; Maier, M.; Augustin, A. Pathophysiology of diabetic macular edema—A background for current treatment modalities. Expert Rev. Ophthalmol. 2018, 13, 273–281. [Google Scholar] [CrossRef]
- Paul, K.C.; Jerrett, M.; Ritz, B. Type 2 diabetes mellitus and Alzheimer’s disease: Overlapping biologic mechanisms and environmental risk factors. Curr. Environ. Health Rep. 2018, 5, 44–58. [Google Scholar] [CrossRef]
- Simó, R.; Ciudin, A.; Simó-Servat, O.; Hernández, C. Cognitive impairment and dementia: A new emerging complication of type 2 diabetes—The diabetologist’s perspective. Acta Diabetol. 2017, 54, 417–424. [Google Scholar] [CrossRef]
- Simó, R.; Stitt, A.W.; Gardner, T.W. Neurodegeneration in diabetic retinopathy: Does it really matter? Diabetologia 2018, 61, 1902–1912. [Google Scholar] [CrossRef] [Green Version]
- Sundstrom, J.M.; Hernández, C.; Weber, S.R.; Zhao, Y.; Dunklebarger, M.; Tiberti, N.; Laremore, T.; Simó-Servat, O.; Garcia-Ramirez, M.; Barber, A.J.; et al. Proteomic analysis of early diabetic retinopathy reveals mediators of neurodegenerative brain diseases. Investig. Ophthalmology Vis. Sci. 2018, 59, 2264–2274. [Google Scholar] [CrossRef] [Green Version]
- Johannesen, S.K.; Viken, J.N.; Vergmann, A.S.; Grauslund, J. Optical coherence tomography angiography and microvascular changes in diabetic retinopathy: A systematic review. Acta Ophthalmol. 2019, 97, 7–14. [Google Scholar] [CrossRef]
- Palma, F.; Camacho, P. The role of optical coherence tomography angiography to detect early microvascular changes in diabetic retinopathy: A systematic review. J. Diabetes Metab. Disord. 2021, 20, 1957–1974. [Google Scholar] [CrossRef]
- Zhang, B.; Chou, Y.; Zhao, X.; Yang, J.; Chen, Y. Early detection of microvascular impairments with optical coherence tomography angiography in diabetic patients without clinical retinopathy: A meta-analysis. Am. J. Ophthalmol. 2020, 222, 226–237. [Google Scholar] [CrossRef]
- Bilen, N.B.; Titiz, A.P.; Bilen, S.; Gultekin, B.P.; Hamurcu, M.S.; Kalayci, D. Optical coherence tomography and neurodegeneration in epilepsy. Eur. J. Ophthalmol. 2019, 31, 252–257. [Google Scholar] [CrossRef]
- Kaplan, A.T.; Yalcın, S.O.; Sager, S.G. Evaluation of optical coherence tomography findings in adolescents with genetic generalized epilepsy. Eur. J. Ophthalmol. 2021. [Google Scholar] [CrossRef]
- Hilton, E.J.R.; Hosking, S.L.; Betts, T. Epilepsy patients treated with antiepileptic drug therapy exhibit compromised ocular perfusion characteristics. Epilepsia 2002, 43, 1346–1350. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Xu, H.; Huang, Y.; Gu, R.; Zong, Y.; Zhu, H.; Wang, M. Changes in retinal perfusion in Leber’s hereditary optic neuropathy: An optical coherence tomography-angiography study. Ophthalmic Res. 2021, 64, 863–870. [Google Scholar] [CrossRef]
- Kousal, B.; Kolarova, H.; Meliska, M.; Bydzovsky, J.; Diblik, P.; Kulhanek, J.; Votruba, M.; Honzik, T.; Liskova, P. Peripapillary microcirculation in Leber hereditary optic neuropathy. Acta Ophthalmol. 2019, 97, e71–e76. [Google Scholar] [CrossRef] [Green Version]
- Bingöl Kızıltunç, P.; Tüntaş Bilen, F.; Atilla, H. Optical coherence tomography angiography findings in long-term follow-up of Leber’s hereditary optic neuropathy: Report of two cases. Turk. J. Ophthalmol. 2020, 50, 313–316. [Google Scholar] [CrossRef]
- Matsuzaki, M.; Hirami, Y.; Uyama, H.; Kurimoto, Y. Optical coherence tomography angiography changes in radial peripapillary capillaries in Leber hereditary optic neuropathy. Am. J. Ophthalmol. Case Rep. 2018, 9, 51–55. [Google Scholar] [CrossRef]
- Yap, T.E.; Balendra, S.I.; Almonte, M.T.; Cordeiro, M.F. Retinal correlates of neurological disorders. Ther. Adv. Chronic Dis. 2019, 10. [Google Scholar] [CrossRef]
- Beswick, E.; Forbes, D.; Hassan, Z.; Wong, C.; Newton, J.; Carson, A.; Abrahams, S.; Chandran, S.; Pal, S. A systematic review of non-motor symptom evaluation in clinical trials for amyotrophic lateral sclerosis. J. Neurol. 2022, 269, 411–426. [Google Scholar] [CrossRef]
- Cerveró, A.; Casado, A.; Riancho, J. Retinal changes in amyotrophic lateral sclerosis: Looking at the disease through a new window. J. Neurol. 2021, 268, 2083–2089. [Google Scholar] [CrossRef]
- Urano, F. Wolfram syndrome: Diagnosis, management, and treatment. Curr. Diabetes Rep. 2016, 16, 6. [Google Scholar] [CrossRef] [Green Version]
- Asanad, S.; Wu, J.; Nassisi, M.; Ross-Cisneros, F.N.; Sadun, A.A. Optical coherence tomography-angiography in Wolfram syndrome: A mitochondrial etiology in disease pathophysiology. Can. J. Ophthalmol. 2019, 54, e27–e30. [Google Scholar] [CrossRef] [Green Version]
- Heng, L.Z.; Bailey, C.; Lee, R.; Dick, A.; Ross, A. A review and update on the ophthalmic implications of Susac syndrome. Surv. Ophthalmol. 2019, 64, 477–485. [Google Scholar] [CrossRef] [Green Version]
- Wirth, M.A.; Khan, H.M.; Chan, J.; Carruthers, R.; Lo, J.; Sarunic, M.V.; Navajas, E.V.; Sheldon, C.A. Investigating microangiopathy using swept-source optical coherence tomography angiography in patients with Susac syndrome. Retina 2021, 41, 2172–2178. [Google Scholar] [CrossRef]
- Leclaire, M.D.; Eter, N.; Alnawaiseh, M. Die optische Kohärenztomographie-Angiographie und Erkrankungen des kardiovaskulären Spektrums. Ein Überblick über die aktuelle Studienlage. Ophthalmologe 2021, 118, 1119–1127. [Google Scholar] [CrossRef]
- Alten, F.; Motte, J.; Ewering, C.; Osada, N.; Clemens, C.R.; Kadas, E.M.; Eter, N.; Paul, F.; Marziniak, M. Multimodal retinal vessel analysis in CADASIL patients. PLoS ONE 2014, 9, e112311. [Google Scholar] [CrossRef]
- Nelis, P.; Kleffner, I.; Burg, M.C.; Clemens, C.R.; Alnawaiseh, M.; Motte, J.; Marziniak, M.; Eter, N.; Alten, F. OCT-angiography reveals reduced vessel density in the deep retinal plexus of CADASIL patients. Sci. Rep. 2018, 8, 8148. [Google Scholar] [CrossRef]



| Group 1 |
|---|
| Alzheimer’s Disease (AD) |
| Parkinson’s Disease (PD) |
| Multiple Sclerosis (MS) |
| Glaucoma |
| AION + NAION |
| Papilloedema |
| Migraine |
| Stroke |
| Group 2 |
| Diabetic Retinopathy (DR) (neurodegenerative component) |
| Epilepsy |
| Group 3 |
| LHON |
| Amyotrophic lateral sclerosis (ALS) |
| Wolfram Syndrome |
| Susac Syndrome |
| CADASIL |
| Parameter | Definition |
|---|---|
| Vessel length density * | Length-based measurement of vessel density: total length of the perfused vasculature per unit area in the region of measurement (Zeiss definition) |
| Vessel density * | Area-based measurement of vessel density: expresses how much area is taken up by vessels (Optovue definition) |
| Perfusion density | Area-based measurement of vessel density: total area of the perfused vasculature per unit area in the region of measurement (Zeiss definition) |
| Fractal dimension | Describes shape or texture and determines complexity of an image |
| Lacunarity | Expresses patchiness or inhomogeneity of an image |
| Vessel perimeter index | Expresses vessel perimeter in relation to total image area |
| Foveal avascular zone (FAZ) | Avascular area in the center of the macula within the fovea |
| FAZ area | Area of the FAZ |
| FAZ diameter/perimeter | Diameter/Perimeter of the FAZ |
| FAZ circularity index | Index describing how circular the area of the FAZ is; values closer to 1 indicate higher circularity |
| Vessel tortuosity | Abnormal curvature of the vessels |
| Narrowed/dilated vessels | Morphologically obvious thinning or dilation of vessels |
| Branching complexity | Altered complexity of vessel branching |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Augustin, A.J.; Atorf, J. The Value of Optical Coherence Tomography Angiography (OCT-A) in Neurological Diseases. Diagnostics 2022, 12, 468. https://doi.org/10.3390/diagnostics12020468
Augustin AJ, Atorf J. The Value of Optical Coherence Tomography Angiography (OCT-A) in Neurological Diseases. Diagnostics. 2022; 12(2):468. https://doi.org/10.3390/diagnostics12020468
Chicago/Turabian StyleAugustin, Albert J., and Jenny Atorf. 2022. "The Value of Optical Coherence Tomography Angiography (OCT-A) in Neurological Diseases" Diagnostics 12, no. 2: 468. https://doi.org/10.3390/diagnostics12020468





