Papillary Thyroid Carcinoma with Spindle Cell Metaplasia: A Rare Encounter
Abstract
:1. Introduction
2. Case Presentation
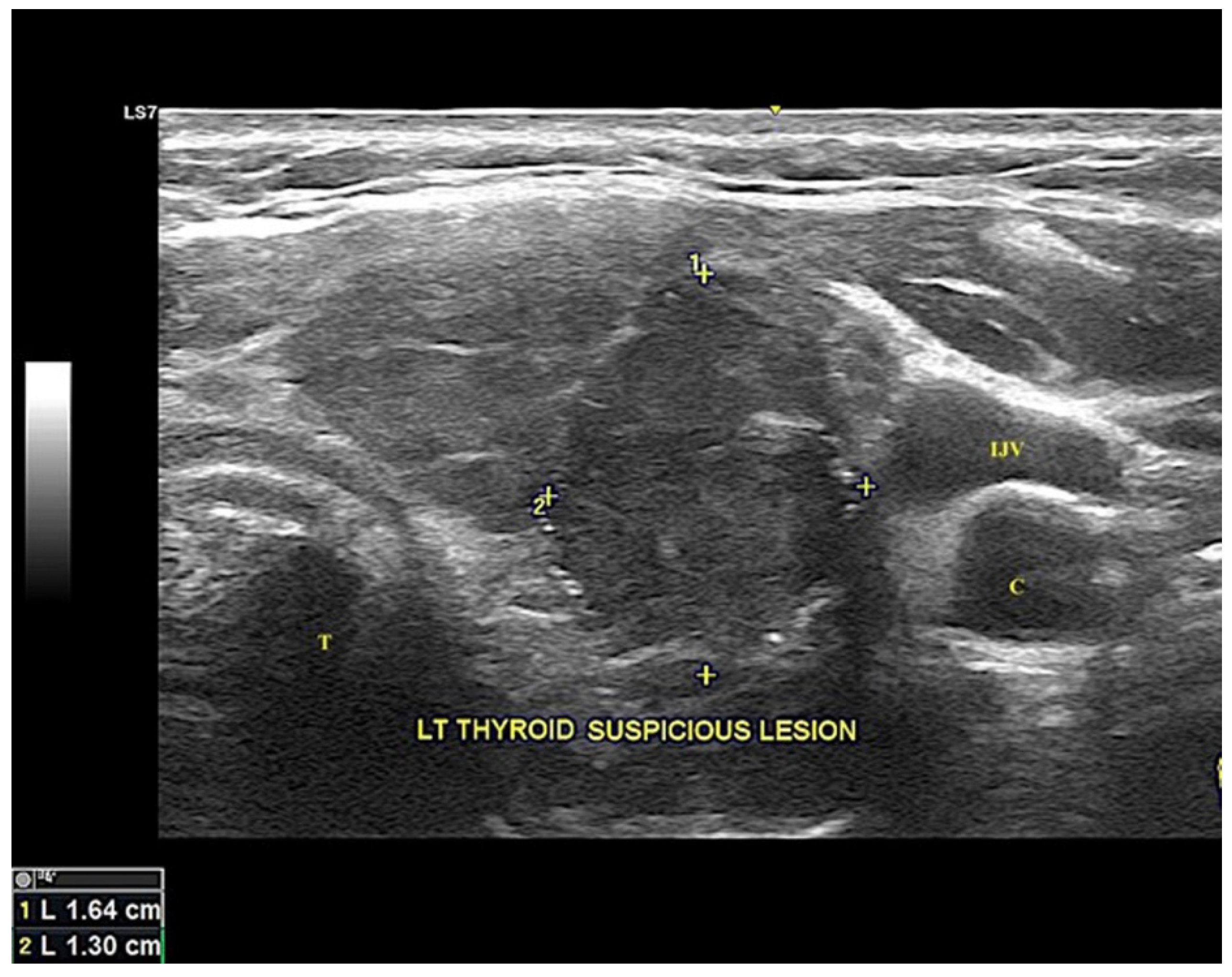
2.1. Clinical Presentation
2.2. Cytopathological and Histopathological Examination
3. Discussion
Histological Differential Diagnosis of Thyroid Lesions with Spindle Cell Morphology
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Rosai, J.; Albores Saavedra, J.; Asioli, S.; Baloch, Z.W.; Bogdanova, T.; Chen, H.; Delellis, R.A.; Erickson, L.A.; Fagin, J.A.; Franssila, K.O.; et al. Papillary thyroid carcinoma. In WHO Classification of Tumours of Endocrine Organs, 4th ed.; Lloyd, R.V., Osamura, R.Y., Kloppel, G., Rosai, J., Eds.; IARC Press: Lyon, France, 2017; pp. 81–91. [Google Scholar]
- Vergilio, J.; Baloch, Z.W.; LiVolsi, V.A. Spindle cell metaplasia of the thyroid arising in association with papillary carcinoma and follicular adenoma. Am. J. Clin. Pathol. 2002, 117, 199–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woenckhaus, C.; Cameselle-Teijeiro, J.; Ruiz-Ponte, C.; Abdulkader, I.; Reyes-Santías, R.; Sobrinho-Simões, M. Spindle cell variant of papillary thyroid carcinoma. Histopathology 2004, 45, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.D.; Martini, M.; Cingolani, N.; Ranaldi, R.; Fadda, G. Images in endocrine pathology: Spindle cell lesion of the thyroid gland. Endocr. Pathol. 2012, 23, 132–134. [Google Scholar] [CrossRef] [PubMed]
- Corrado, S.; Corsello, S.M.; Maiorana, A.; Rossi, E.D.; Pontecorvi, A.; Fadda, G.; Papi, G. Papillary thyroid carcinoma with predominant spindle cell component: Report of two rare cases and discussion on the differential diagnosis with other spindled thyroid neoplasm. Endocr. Pathol. 2014, 25, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Xia, C.; Liu, H.; Zhu, W. Primary thyroid spindle cell tumors: Spindle cell variant of papillary thyroid carcinoma? Int. J. Clin. Exp. Pathol. 2015, 8, 13528–13531. [Google Scholar] [PubMed]
- Matoso, A.; Easley, S.E.; Mangray, S.; Jacob, R.; DeLellis, R.A. Spindle cell foci in the thyroid gland: An immunohistochemical analysis. Appl. Immunohistochem. Mol. Morphol. 2011, 19, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Hutter, R.V.; Tollefsen, H.R.; De Cosse, J.J.; Foote, F.W., Jr.; Frazell, E.L. Spindle and giant cell metaplasia in papillary carcinoma of the thyroid. Am. J. Surg. 1965, 110, 660–668. [Google Scholar] [CrossRef]
- Rashid, F.A.; Tabassum, S.; Khan, M.S.; Ansari, H.R.; Asif, M.; Sheikh, A.K.; Aga, S.S. VE1 immunohistochemistry is an adjunct tool for detection of BRAFV600E mutation: Validation in thyroid cancer patients. J. Clin. Lab. Anal. 2021, 35, e23628. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.E.; Trevor, K.T. Epithelial-mesenchymal transitions during cell culture of primary thyroid tumors? Genes Chromosomes Cancer 1993, 6, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Shakib, H.; Rajabi, S.; Dehghan, M.H.; Mashayekhi, F.J.; Safari-Alighiarloo, N.; Hedayati, M. Epithelial-to-mesenchymal transition in thyroid cancer: A comprehensive review. Endocrine 2019, 66, 435–455. [Google Scholar] [CrossRef] [PubMed]
- Ku, E.J.; Park, J.W. A postoperative spindle cell tumor in the thyroid bed resembling recurrent papillary thyroid carcinoma. Int. J. Clin. Exp. Med. 2020, 13, 8127–8131. [Google Scholar]
- Zhang, Y.; Tang, H.; Hu, H.; Yong, X. A rare primary tumor of the thyroid gland: A new case of leiomyoma and literature review. Clin. Med. Insights Oncol. 2018, 12, 1179554918813535. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Liu, Z.; Su, C.; Guan, Q.; Wan, F.; Dong, B.; Bao, L.; Zhang, W.; Wang, Y.P.; Wang, G. Primary peripheral nerve sheath tumors of the thyroid gland: A case report and literature review. Mol. Clin. Oncol. 2016, 4, 209–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdulrahman, A.A.; Al-Loh Ashi, S.; Jain, M.; Hou, J.S. Spindle epithelial tumor with thymus-like differentiation (SETTLE): Case report with longest follow up & latency to metastasis. Hum. Pathol. Case Rep. 2018, 11, 60–64. [Google Scholar]
- Pusztaszeri, M.P.; Bongiovann, M.; Faquin, W.C. Update on the cytologic and molecular features of medullary thyroid carcinoma. Adv. Anat. Pathol. 2014, 21, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Brandwein-Gensler, M.S.; Wang, B.Y.; Urken, M.L. Spindle cell transformation of papillary carcinoma: An aggressive entity distinct from anaplastic thyroid carcinoma. Arch. Pathol. Lab. Med. 2004, 128, 87–89. [Google Scholar] [CrossRef] [PubMed]


| No. | Authors | Age(Years)/Gender | Clinical Presentation | Size (mm) | Histopathological Subtype | Immunohistochemistry Staining Pattern | |
|---|---|---|---|---|---|---|---|
| Follicular Cell Component | Spindle Cell Component | ||||||
| 1. | Vergilio et al. (2002) [2] | 25–61/6 F, 1 M | Mass at left/right thyroid lobe | 3–30 | Spindle cell variant (1%–95%) | NA | TG+, CK+/−, calcitonin−, SMA− |
| 2. | Woenckhaus et al. (2004) [3] | 32/F | Cold nodule over right thyroid lobe | 27 | Spindle cell variant (80%) + Follicular variant (20%) | TG+, CKAE1/AE3+, CAM5.2+, TTF-1+, vimentin+, S100+, BCL-2+, 34βE12+, E-cad+, PR+ *, ER−, calcitonin−, chromo−, synapto−, SMA−, CD34−, HMB45−, p53− | TG+, CKAE1/AE3+, CAM5.2+, TTF-1+, vimentin+, S100+, BCL-2+, 34βE12−, E-cad−, PR+ *, ER−, calcitonin−, chromo−, synapto−, SMA−, CD34−, HMB45−, p53− |
| 3. | Rossi et al. (2012) [4] | 51/M | Left thyroid nodule | 50 | Spindle cell variant (almost 100%) | NA | TG+, TTF-1+, HMBE-1+, galectin-3+, CK+, calcitonin−, CD34−, chromo−, synapto−, S100−, CEA−, p53−, Ki67 < 5% |
| 4. | Corrado et al. (2013) [5] | 56/M | Mass in left thyroid lobe | 45 | Spindle cell variant (80%) + Follicular variant (20%) | TTF-1+, CK19+ | TG+, TTF-1+, MIB < 1%, CKMNF116−, CK19−, CK7−, p63−, chromo−, calcitonin−, S100−, NSE−, SMA−, myosin−, desmin−, EMA−, CD1a−, CD3−, CD5−, CD21−, CD31−, CD34−, CD35−, CD68−, CD117−, BCL-2−, melan A−, HMB45−, p53− |
| 53/F | Mass in right thyroid lobe | 46 | Spindle cell variant (80%) + Follicular variant (20%) | TTF-1+, CK Fil7+8+ | TG+, TTF-1+, CKMNF116+, MIB < 2%, CK19−, CK7−, p63−, chromo−, calcitonin−, S100−, NSE−, SMA−, myosin−, desmin−, EMA−, CD1a−, CD3−, CD5−, CD21−, CD31−, CD34−, CD35−, CD68−, CD117−, BCL-2−, melan A−, HMB45−, p53− | ||
| 5. | Ma et al. (2015) [6] | 67/M | Mass in right thyroid | 25 | Spindle cell variant (almost 100%) with rare neoplastic follicular cells | TG+, TTF-1+, PAX-8+, vimentin+, BCL-2+, panCK+, CAM5.2+, CD34-, CD99−, CK7−, CK19−, ER−, PR−, calcitonin−, synapto−, chromo−, S100−, CD21−, CD23−, SMA−, HMB45−, p63−, p40−, p53− | TG+, TTF-1+, PAX-8+, vimentin+, BCL-2+, panCK−, CAM5.2−, CD34−, CD99−, CK7−, CK19−, ER−, PR−, calcitonin−, synapto−, chromo−, S100−, CD21−, CD23−, SMA−, HMB45−, p63−, p40−, p53−, Ki67 3% |
| 6. | Present case (2021) | 51/F | Cold nodule over left thyroid lobe | 30 | Classical type (70%) Spindle cell variant (30%) | TG+, TTF-1+, CKAE1/AE3+, CK19+, E-cad+, vimentin+, calcitonin−, S100−, desmin−, SMA−, chromo−, synapto− | TG+, TTF-1+, p53+, BRAF V600E+, CKAE1/AE3+, CK19−, E-cad−, vimentin+, calcitonin−, S100−, desmin−, SMA−, chromo−, synapto− |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leong, K.W.; Abdullah Suhaimi, S.N.; Tan, G.C.; Wong, Y.P. Papillary Thyroid Carcinoma with Spindle Cell Metaplasia: A Rare Encounter. Diagnostics 2022, 12, 855. https://doi.org/10.3390/diagnostics12040855
Leong KW, Abdullah Suhaimi SN, Tan GC, Wong YP. Papillary Thyroid Carcinoma with Spindle Cell Metaplasia: A Rare Encounter. Diagnostics. 2022; 12(4):855. https://doi.org/10.3390/diagnostics12040855
Chicago/Turabian StyleLeong, Ka Wen, Shahrun Niza Abdullah Suhaimi, Geok Chin Tan, and Yin Ping Wong. 2022. "Papillary Thyroid Carcinoma with Spindle Cell Metaplasia: A Rare Encounter" Diagnostics 12, no. 4: 855. https://doi.org/10.3390/diagnostics12040855
APA StyleLeong, K. W., Abdullah Suhaimi, S. N., Tan, G. C., & Wong, Y. P. (2022). Papillary Thyroid Carcinoma with Spindle Cell Metaplasia: A Rare Encounter. Diagnostics, 12(4), 855. https://doi.org/10.3390/diagnostics12040855






