Head-to-Head Comparison between FDG and 11C-Methionine in Multiple Myeloma: A Systematic Review
Abstract
1. Introduction
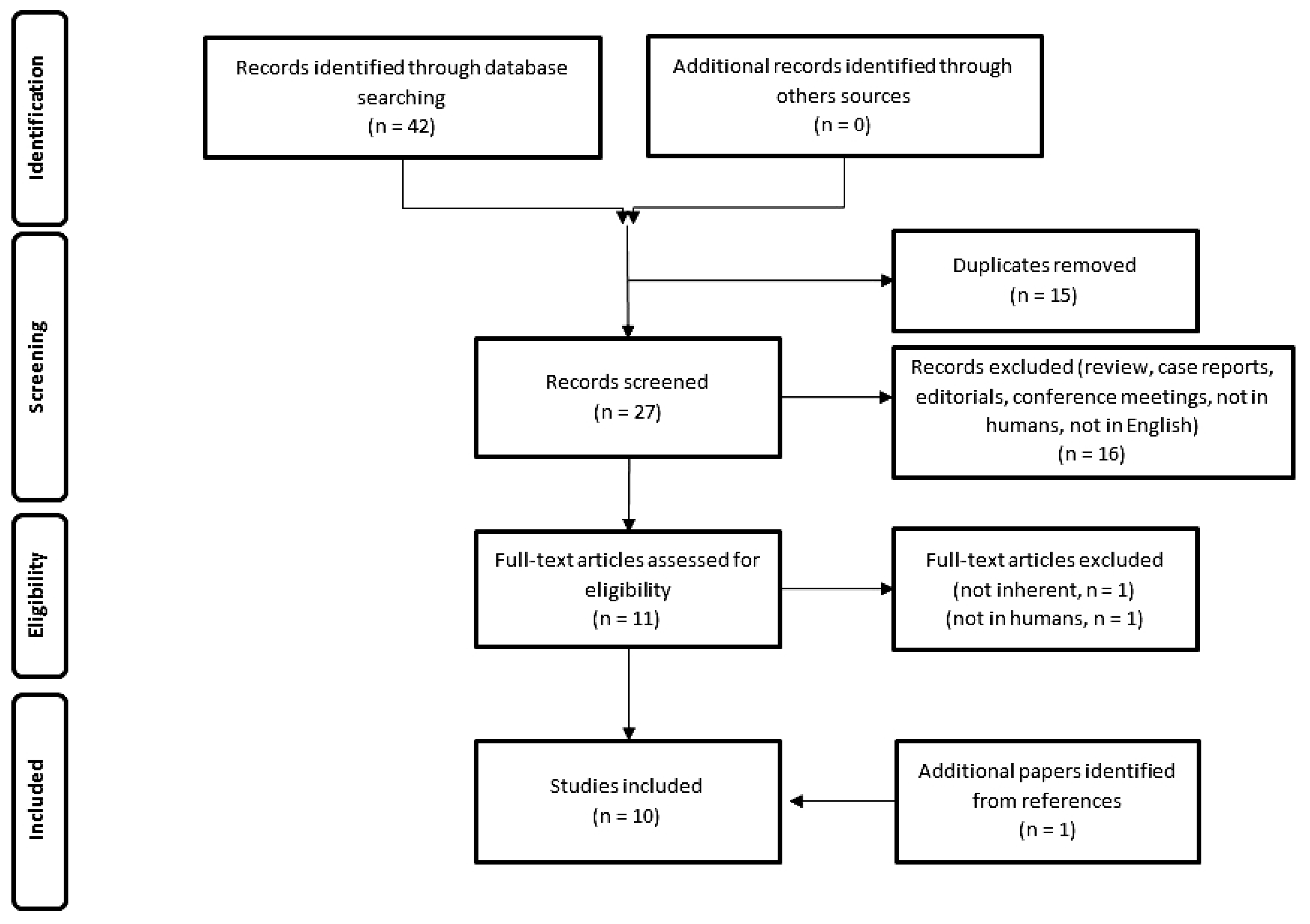
2. Materials and Methods
2.1. Search Strategy
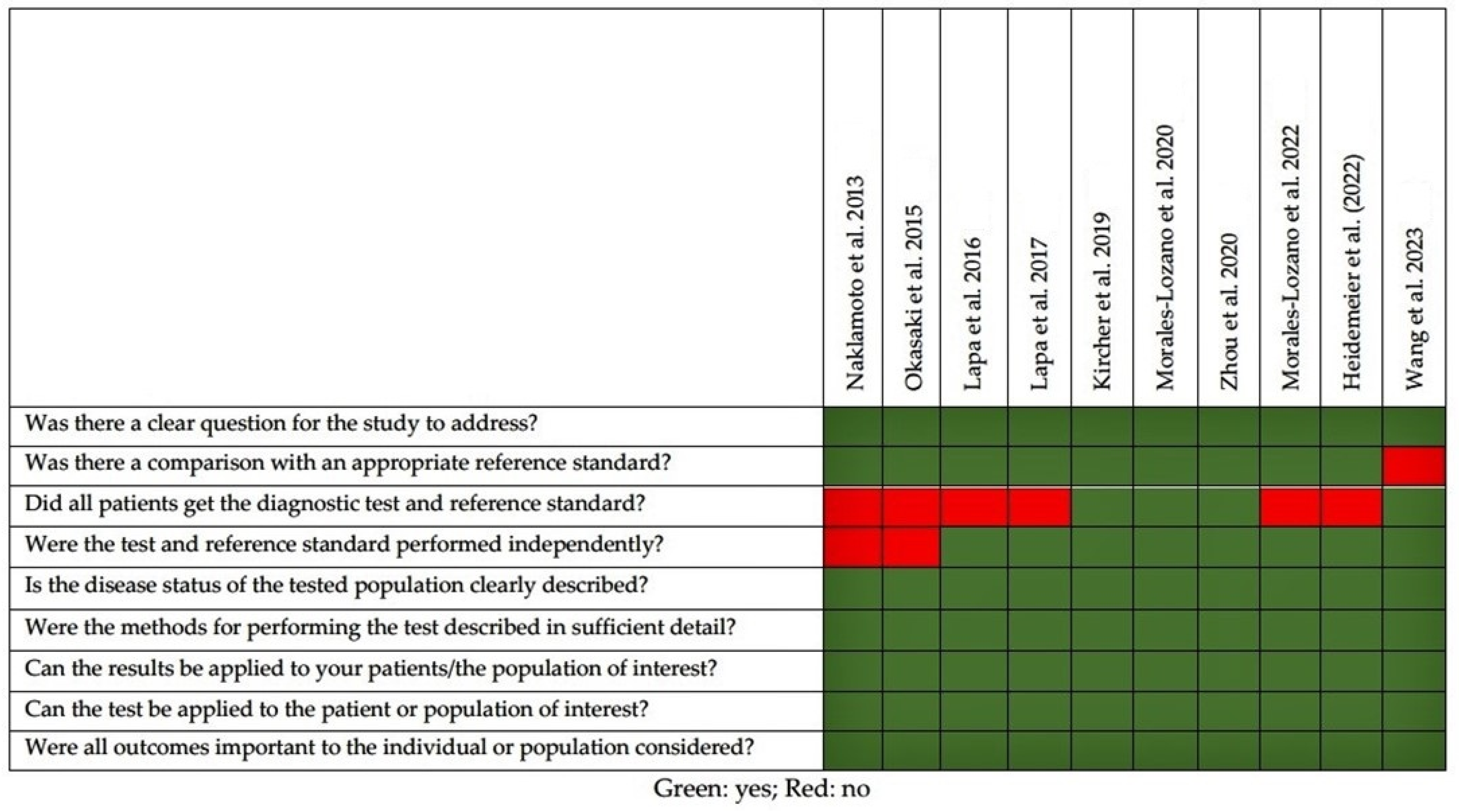
2.2. Quality of the Selected Studies
3. Results
3.1. Analysis of the Evidence
3.2. FDG and MET PET Head-to-Head Comparison
3.3. Diagnostic and Prognostic Impact of FDG and MET-Derived Volumetric Parameters
3.4. Comparison of FDG and MET with Other Radiopharmaceuticals or Other Imaging Modalities (MRI)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Padala, S.A.; Barsouk, A.; Barsouk, A.; Rawla, P.; Vakiti, A.; Kolhe, R.; Kota, V.; Ajebo, G.H. Epidemiology, Staging, and Management of Multiple Myeloma. Med. Sci. 2021, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Colmone, A.; Amorim, M.; Pontier, A.L.; Wang, S.; Jablonski, E.; Sipkins, D.A. Leukemic Cells Create Bone Marrow Niches That Disrupt the Behavior of Normal Hematopoietic Progenitor Cells. Science 2008, 322, 1861–1865. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.V.; Landgren, O.; Mateos, M.-V. Smoldering multiple myeloma. Blood 2015, 125, 3069–3075. [Google Scholar] [CrossRef] [PubMed]
- Eisfeld, C.; Kajüter, H.; Möller, L.; Wellmann, I.; Shumilov, E.; Stang, A. Time trends in survival and causes of death in multiple myeloma: A population-based study from Germany. BMC Cancer 2023, 23, 317. [Google Scholar] [CrossRef]
- Hari, P.N.; Zhang, M.-J.; Roy, V.; Pérez, W.S.; Bashey, A.; To, L.B.; Elfenbein, G.; Freytes, C.O.; Gale, R.P.; Gibson, J.; et al. Is the international staging system superior to the Durie–Salmon staging system? A comparison in multiple myeloma patients undergoing autologous transplant. Leukemia 2009, 23, 1528–1534. [Google Scholar] [CrossRef]
- Wu, F.; Bernard, S.; Fayad, L.M.; Ilaslan, H.; Messiou, C.; Moulopoulos, L.A.; Mulligan, M.E. Updates and Ongoing Challenges in Imaging of Multiple Myeloma: AJR Expert Panel Narrative Review. Am. J. Roentgenol. 2021, 217, 775–785. [Google Scholar] [CrossRef]
- Nanni, C. PET-FDG: Impetus. Cancers 2020, 12, 1030. [Google Scholar] [CrossRef]
- Bredella, M.A.; Steinbach, L.; Caputo, G.; Segall, G.; Hawkins, R. Value of FDG PET in the Assessment of Patients with Multiple Myeloma. Am. J. Roentgenol. 2005, 184, 1199–1204. [Google Scholar] [CrossRef]
- Mesguich, C.; Hulin, C.; Lascaux, A.; Bordenave, L.; Marit, G.; Hindié, E. Choline PET/CT in Multiple Myeloma. Cancers 2020, 12, 1394. [Google Scholar] [CrossRef]
- Matteucci, F.; Paganelli, G.; Martinelli, G.; Cerchione, C. PET/CT in Multiple Myeloma: Beyond FDG. Front. Oncol. 2021, 10, 622501. [Google Scholar] [CrossRef] [PubMed]
- Isoda, A.; Kaira, K.; Iwashina, M.; Oriuchi, N.; Tominaga, H.; Nagamori, S.; Kanai, Y.; Oyama, T.; Asao, T.; Matsumoto, M.; et al. Expression of L-type amino acid transporter 1 (LAT 1) as a prognostic and therapeutic indicator in multiple myeloma. Cancer Sci. 2014, 105, 1496–1502. [Google Scholar] [CrossRef]
- Brioli, A.; Morgan, G.J.; Durie, B.; Zamagni, E. The utility of newer imaging techniques as predictors of clinical outcomes in multiple myeloma. Expert Rev. Hematol. 2014, 7, 13–16. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, Y.; Kurihara, K.; Nishizawa, M.; Yamashita, K.; Nakatani, K.; Kondo, T.; Takaori-Kondo, A.; Togashi, K. Clinical value of 11C-methionine PET/CT in patients with plasma cell malignancy: Comparison with 18F-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Okasaki, M.; Kubota, K.; Minamimoto, R.; Miyata, Y.; Morooka, M.; Ito, K.; Ishiwata, K.; Toyohara, J.; Inoue, T.; Hirai, R.; et al. Comparison of 11C-4′-thiothymidine, 11C-methionine, and 18F-FDG PET/CT for the detection of active lesions of multiple myeloma. Ann. Nucl. Med. 2015, 29, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Lapa, C.; Knop, S.; Schreder, M.; Rudelius, M.; Knott, M.; Jörg, G.; Samnick, S.; Herrmann, K.; Buck, A.K.; Einsele, H.; et al. 11 C-Methionine-PET in Multiple Myeloma: Correlation with Clinical Parameters and Bone Marrow Involvement. Theranostics 2016, 6, 254–261. [Google Scholar] [CrossRef]
- Lapa, C.; Garcia-Velloso, M.J.; Lückerath, K.; Samnick, S.; Schreder, M.; Otero, P.R.; Schmid, J.-S.; Herrmann, K.; Knop, S.; Buck, A.K.; et al. 11 C-Methionine-PET in Multiple Myeloma: A Combined Study from Two Different Institutions. Theranostics 2017, 7, 2956–2964. [Google Scholar] [CrossRef]
- Kircher, S.; Stolzenburg, A.; Kortüm, K.M.; Kircher, M.; Da Via, M.; Samnick, S.; Buck, A.K.; Einsele, H.; Rosenwald, A.; Lapa, C. Hexokinase-2 Expression in 11 C-Methionine–Positive, 18 F-FDG–Negative Multiple Myeloma. J. Nucl. Med. 2019, 60, 348–352. [Google Scholar] [CrossRef]
- Zhou, X.; Dierks, A.; Kertels, O.; Kircher, M.; Schirbel, A.; Samnick, S.; Buck, A.K.; Knorz, S.; Böckle, D.; Scheller, L.; et al. 18F-FDG, 11C-Methionine, and 68Ga-Pentixafor PET/CT in Patients with Smoldering Multiple Myeloma: Imaging Pattern and Clinical Features. Cancers 2020, 12, 2333. [Google Scholar] [CrossRef]
- Morales-Lozano, M.I.; Viering, O.; Samnick, S.; Rodriguez-Otero, P.; Buck, A.K.; Marcos-Jubilar, M.; Rasche, L.; Prieto, E.; Kortüm, K.M.; San-Miguel, J.; et al. 18F-FDG and 11C-Methionine PET/CT in Newly Diagnosed Multiple Myeloma Patients: Comparison of Volume-Based PET Biomarkers. Cancers 2020, 12, 1042. [Google Scholar] [CrossRef]
- Heidemeier, A.; Schloetelburg, W.; Thurner, A.; Metz, C.; Heidemeier, H.; Rasche, L.; Martin Kortuem, K.; Boeckle, D.; Weiland, E.; Benkert, T.; et al. Multi-parametric whole-body MRI evaluation discerns vital from non-vital multiple myeloma lesions as validated by 18F-FDG and 11C-methionine PET/CT. Eur. J. Radiol. 2022, 155, 110493. [Google Scholar] [CrossRef]
- Morales-Lozano, M.I.; Rodriguez-Otero, P.; Sancho, L.; Nuñez-Cordoba, J.M.; Prieto, E.; Marcos-Jubilar, M.; Rosales, J.J.; Alfonso, A.; Guillen, E.F.; San-Miguel, J.; et al. 11C-Methionine PET/CT in Assessment of Multiple Myeloma Patients: Comparison to 18F-FDG PET/CT and Prognostic Value. Int. J. Mol. Sci. 2022, 23, 9895. [Google Scholar] [CrossRef]
- Wang, Y.; Yee, A.; Bernstein, Z.; O’Donnell, E.; Raje, N.; Mahmood, U. Carbon-11-Labeled Methionine PET/CT in Patients with FDG-Occult Multiple Myeloma: A Prospective Pilot Study. Am. J. Roentgenol. 2023, 220, 578–579. [Google Scholar] [CrossRef] [PubMed]
- Fanti, S.; Nanni, C.; Lopci, E.; Castellucci, P.; Rubello, D.; Farsad, M.; Boschi, S. Imaging with 11Carbon labelled PET tracers. Nucl. Med. Commun. 2010, 31, 613–616. [Google Scholar] [CrossRef] [PubMed]
- Filippi, L.; Bagni, O.; Schillaci, O. Digital PET/CT with 18F-FACBC in early castration-resistant prostate cancer: Our preliminary results. Expert Rev. Med. Devices 2022, 19, 591–598. [Google Scholar] [CrossRef]
- Stokke, C.; Nørgaard, J.N.; Feiring Phillips, H.; Sherwani, A.; Nuruddin, S.; Connelly, J.; Schjesvold, F.; Revheim, M.-E. Comparison of [18F]fluciclovine and [18F]FDG PET/CT in Newly Diagnosed Multiple Myeloma Patients. Mol. Imaging Biol. 2022, 24, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Czyż, J.; Małkowski, B.; Jurczyszyn, A.; Grząśko, N.; Łopatto, R.; Olejniczak, M.; Czyż, A.; Jachalska, A. 18F-fluoro-ethyl-tyrosine (18F-FET) PET/CT as a potential new diagnostic tool in multiple myeloma: A preliminary study. Contemp. Oncol. Poznan Pol. 2019, 23, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Sachpekidis, C.; Hillengass, J.; Goldschmidt, H.; Wagner, B.; Haberkorn, U.; Kopka, K.; Dimitrakopoulou-Strauss, A. Treatment response evaluation with 18F-FDG PET/CT and 18F-NaF PET/CT in multiple myeloma patients undergoing high-dose chemotherapy and autologous stem cell transplantation. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 50–62. [Google Scholar] [CrossRef]
- Zhou, X.; Dierks, A.; Kertels, O.; Samnick, S.; Kircher, M.; Buck, A.K.; Haertle, L.; Knorz, S.; Böckle, D.; Scheller, L.; et al. The Link between Cytogenetics/Genomics and Imaging Patterns of Relapse and Progression in Patients with Relapsed/Refractory Multiple Myeloma: A Pilot Study Utilizing 18F-FDG PET/CT. Cancers 2020, 12, 2399. [Google Scholar] [CrossRef]
- Langen Stokmo, H.; Aly, M.; Bowitz Lothe, I.M.; Borja, A.J.; Mehdizadeh Seraj, S.; Ghorpade, R.; Miao, X.; Hjortland, G.O.; Malinen, E.; Sorbye, H.; et al. Volumetric parameters from [18F] FDG PET / CT predicts survival in patients with high-grade gastroenteropancreatic neuroendocrine neoplasms. J. Neuroendocrinol. 2022, 34, e13170. [Google Scholar] [CrossRef]
- Gao, J.; Xu, S.; Ju, H.; Pan, Y.; Zhang, Y. The potential application of MR-derived ADCmin values from 68Ga-DOTATATE and 18F-FDG dual tracer PET/MR as replacements for FDG PET in assessment of grade and stage of pancreatic neuroendocrine tumors. EJNMMI Res. 2023, 13, 10. [Google Scholar] [CrossRef]
- Izuishi, K.; Yamamoto, Y.; Mori, H.; Kameyama, R.; Fujihara, S.; Masaki, T.; Suzuki, Y. Molecular mechanisms of [18F]fluorodeoxyglucose accumulation in liver cancer. Oncol. Rep. 2014, 31, 701–706. [Google Scholar] [CrossRef]
- Chiaravalloti, A.; Filippi, L.; Ricci, M.; Cimini, A.; Schillaci, O. Molecular Imaging in Pediatric Brain Tumors. Cancers 2019, 11, 1853. [Google Scholar] [CrossRef]
- Urso, L.; Panareo, S.; Castello, A.; Ambrosio, M.R.; Zatelli, M.C.; Caracciolo, M.; Tonini, E.; Valpiani, G.; Boschi, A.; Uccelli, L.; et al. Glucose Metabolism Modification Induced by Radioligand Therapy with [177Lu]Lu/[90Y]Y-DOTATOC in Advanced Neuroendocrine Neoplasms: A Prospective Pilot Study within FENET-2016 Trial. Pharmaceutics 2022, 14, 2009. [Google Scholar] [CrossRef] [PubMed]
- Filippi, L.; Urso, L.; Bianconi, F.; Palumbo, B.; Marzola, M.C.; Evangelista, L.; Schillaci, O. Radiomics and theranostics with molecular and metabolic probes in prostate cancer: Toward a personalized approach. Expert Rev. Mol. Diagn. 2023, 23, 243–255. [Google Scholar] [CrossRef]
- Urso, L.; Frantellizzi, V.; De Vincentis, G.; Schillaci, O.; Filippi, L.; Evangelista, L. Clinical applications of long axial field-of-view PET/CT scanners in oncology. Clin. Transl. Imaging 2023. [Google Scholar] [CrossRef]
- Mayerhoefer, M.E.; Archibald, S.J.; Messiou, C.; Staudenherz, A.; Berzaczy, D.; Schöder, H. MRI and PET/MRI in hematologic malignancies. J. Magn. Reson. Imaging 2020, 51, 1325–1335. [Google Scholar] [CrossRef]
- Rashidi, A.; Baratto, L.; Theruvath, A.J.; Greene, E.B.; Hawk, K.E.; Lu, R.; Link, M.P.; Spunt, S.L.; Daldrup-Link, H.E. Diagnostic Accuracy of 2-[18F]FDG-PET and whole-body DW-MRI for the detection of bone marrow metastases in children and young adults. Eur. Radiol. 2022, 32, 4967–4979. [Google Scholar] [CrossRef] [PubMed]
- Solnes, L.B.; Werner, R.A.; Jones, K.M.; Sadaghiani, M.S.; Bailey, C.R.; Lapa, C.; Pomper, M.G.; Rowe, S.P. Theranostics: Leveraging Molecular Imaging and Therapy to Impact Patient Management and Secure the Future of Nuclear Medicine. J. Nucl. Med. 2020, 61, 311–318. [Google Scholar] [CrossRef]
- Cimini, A.; Ricci, M.; Chiaravalloti, A.; Filippi, L.; Schillaci, O. Theragnostic Aspects and Radioimmunotherapy in Pediatric Tumors. Int. J. Mol. Sci. 2020, 21, 3849. [Google Scholar] [CrossRef] [PubMed]
- Filippi, L.; Bagni, O.; Nervi, C. Aptamer-based technology for radionuclide targeted imaging and therapy: A promising weapon against cancer. Expert Rev. Med. Devices 2020, 17, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xue, Q.; Yao, S. Nuclear Medicine Application of Pentixafor/Pentixather Targeting CXCR4for Imaging and Therapy in Related Disease. Mini-Rev. Med. Chem. 2023, 23, 787–803. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, J.; Liu, L.; Wang, J.; Zhang, C. The role of 68Ga-Pentixafor PET in multiple myeloma. Clin. Transl. Imaging 2023. [Google Scholar] [CrossRef]

| Authors | Country | Year of Pub | N of pts | Median Age (Range) | Male/ Female | Primary End-point | FDG/MET Sensitivity | FDG/MET Specificity | Comments |
|---|---|---|---|---|---|---|---|---|---|
| Nakamoto et al. [14] | Japan | 2013 | 20 | (45–80) | 11/9 | Compare FDG with MET in MM | 78%/89% * | 100%/100% * | MET revealed more lesions than FDG. MET is useful in case of inconclusive FDG |
| Okasaki et al. [15] | Japan | 2015 | 64 | (33–84) | 40/24 | Compare FDG and MET with 11C-4DST in detection of bone marrow in MM | 60/86.7% * | 76.1%/76.1% | MET can detect more active lesions than FDG in bone marrow |
| Lapa et al. [16] | Germany | 2016 | 43 | (39–82) | 24/19 | Compare FDG and MET in staging and restaging of MM | 76.7%/90.7% * | - | MET can detect more lesions than FDG both in medullary and extramedullary MM lesions |
| Lapa et al. [17] | Germany | 2017 | 78 (4 SP, 5 SSM and 69 sMM) | (31–76) | 55/33 | The superiority of MET than FDG in MM | 60.3%/75.6% * | - | MET can detect more lesions than FDG MET can detect more viable tissue than FDG |
| Kircher et al. [18] | Germany | 2019 | 15 | (51–73) | 10/5 | To understand the biological reason for the negative FDG PET in viable MM by using MET | 60%/100% * | - | No differences in HK2 expression was found among FDG-negative and FDG-positive MM. Other reasons can be linked with this finding |
| Zhou et al. [19] | Germany | 2020 | 10 | (41–74) | 8/2 | To explore the role of FDG, MET and 68Ga-Pentixafor PET/CT in SMM | - | - | MET and 68Ga-Pentixafor PET/CT demonstrate higher sensitivity than FDG PET/CT in detecting bone marrow involvement in SMM. |
| Morales-Lozano et al. [20] | Spain/Germany | 2020 | 22 | (37–79) | 14/6 | To compare MET suitability for the assessment of metabolic tumor burden in comparison to FDG PET. | 100%/100% * | - | MET seems to be a more sensitive and accurate surrogate for total myeloma burden as compared to FDG. |
| Heidemeier et al. [21] | Germany | 2022 | 24 | (49–78) | - | To identify a new wbMRI algorithm for bone marrow lesions, by using FDG and MET PET | - | - | DWI + CSI and FF + T2 sequences can help in identifying MM lesions |
| Morales-Lozano et al. [22] | Spain/Germany | 2022 | 8 SMM, 44 MM | 61 (37–83) | 28/24 | To compare MET with FDG PET/CT in SMM and MM | 84.6–96.1% | - | FDG has a prognostic role in MM MET can detect more lesions than FDG TMTV and TLMU at MET PET show a prognostic meaning |
| Wang et al. [23] | USA | 2023 | 7 | (58–93) | 5/2 | MET PET role in case of FDG negative scan | 0%/80% | 0%/100% | MET PET can be used in case of FDG negative scan for detecting focal avid lesions in MM patients |
| Parameter | FDG (Median and IQR) | Corr. with Clinical Variables | MET (Median and IQR) | Corr. with Clinical Variables |
|---|---|---|---|---|
| SUVmax | 8.76 (3.45–62.23) | beta-2-micro. | 16.40 (6–195.6) | - |
| SUVmean | 3.55 (1.82–7.74) | beta-2-micro. | 4.59 (2.79–8.35) | - |
| SUVpeak | 6.56 (2.82–39.85) | beta-2-micro. | 10.72 (4.64–126.50) | - |
| TMTV | 134.8 cm3 (5.6–524.9) | beta-2-micro. | 443.4 cm3 (145.2–1102.6) | beta-2-micro, M-component, BM infiltration |
| TLG | 598.4 g, (10.7–2086.4) | - | ||
| TLMU | - | 2021.4 g (761.6–6061.4) | beta-2-micro, M-component, BM infiltration |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filippi, L.; Frantellizzi, V.; Bartoletti, P.; Vincentis, G.D.; Schillaci, O.; Evangelista, L. Head-to-Head Comparison between FDG and 11C-Methionine in Multiple Myeloma: A Systematic Review. Diagnostics 2023, 13, 2009. https://doi.org/10.3390/diagnostics13122009
Filippi L, Frantellizzi V, Bartoletti P, Vincentis GD, Schillaci O, Evangelista L. Head-to-Head Comparison between FDG and 11C-Methionine in Multiple Myeloma: A Systematic Review. Diagnostics. 2023; 13(12):2009. https://doi.org/10.3390/diagnostics13122009
Chicago/Turabian StyleFilippi, Luca, Viviana Frantellizzi, Paola Bartoletti, Giuseppe De Vincentis, Orazio Schillaci, and Laura Evangelista. 2023. "Head-to-Head Comparison between FDG and 11C-Methionine in Multiple Myeloma: A Systematic Review" Diagnostics 13, no. 12: 2009. https://doi.org/10.3390/diagnostics13122009
APA StyleFilippi, L., Frantellizzi, V., Bartoletti, P., Vincentis, G. D., Schillaci, O., & Evangelista, L. (2023). Head-to-Head Comparison between FDG and 11C-Methionine in Multiple Myeloma: A Systematic Review. Diagnostics, 13(12), 2009. https://doi.org/10.3390/diagnostics13122009







