Fractional Flow Reserve-Guided Stent Optimisation in Focal and Diffuse Coronary Artery Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Procedures
2.3. Characterisation of CAD Patterns
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Baseline Characteristics
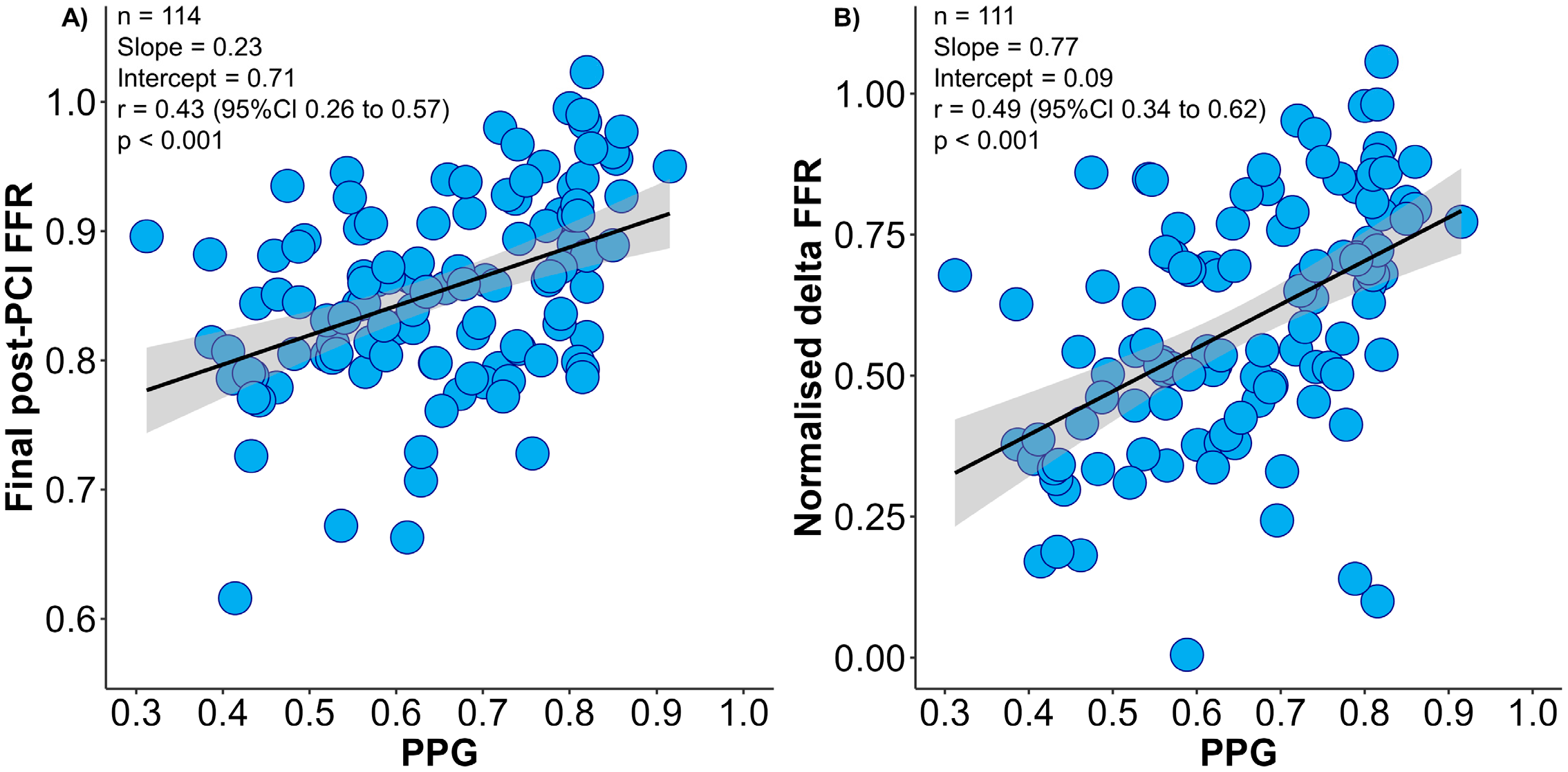
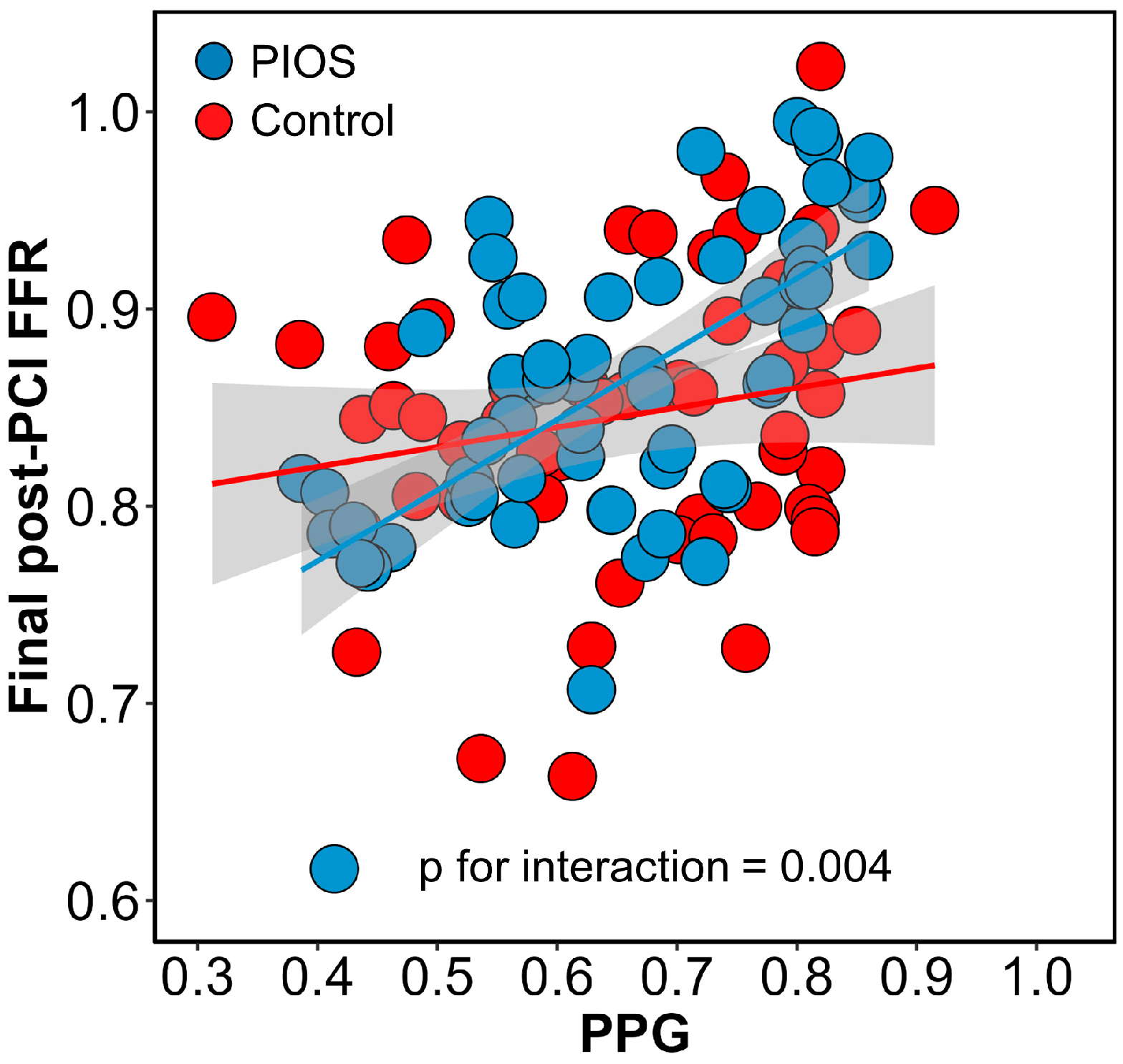
3.3. Baseline CAD Patterns and Final Post-PCI FFR
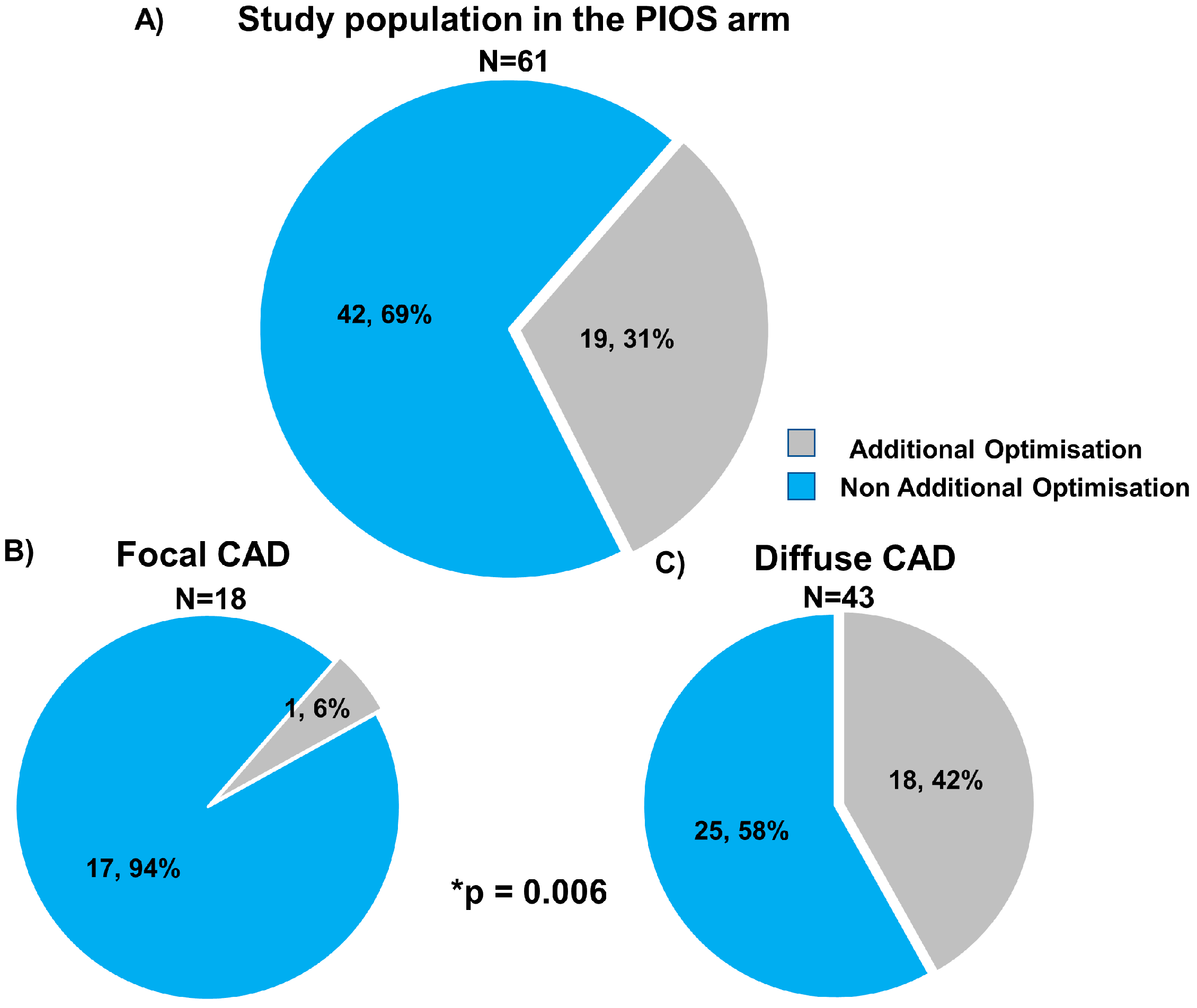
3.4. Stent Optimisation in Focal and Diffuse Disease
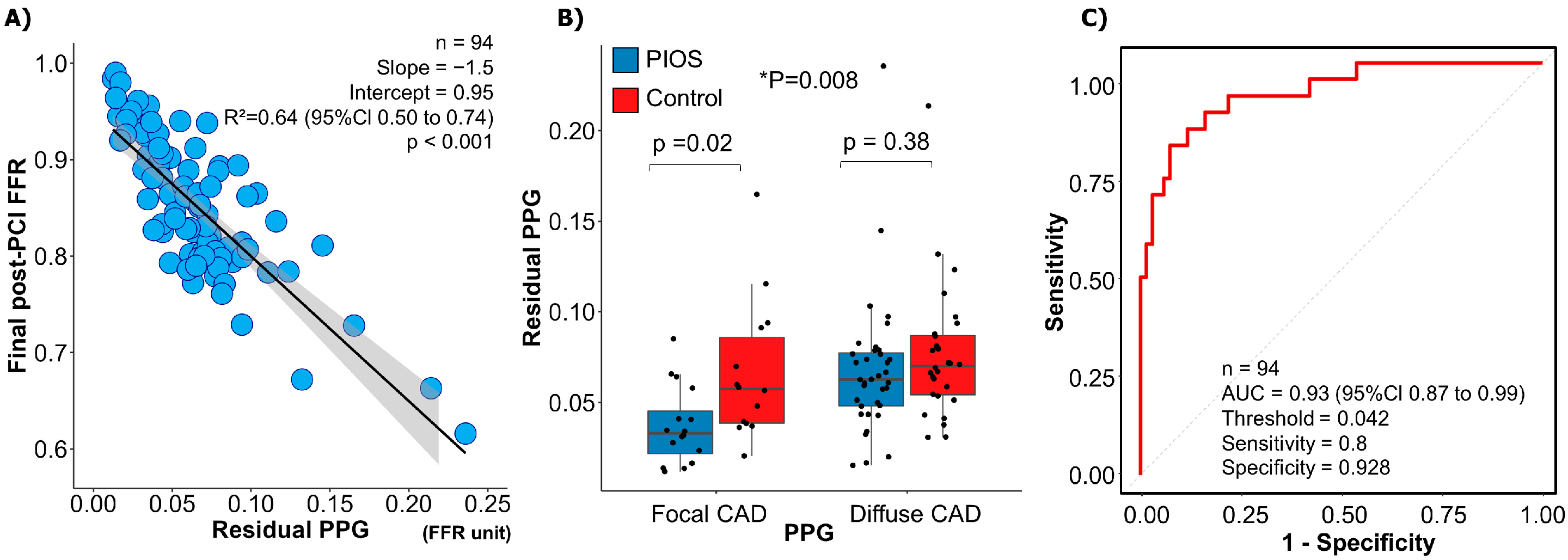
3.5. Residual PPG and Final Post-PCI FFR
3.6. Clinical Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Raber, L.; Mintz, G.S.; Koskinas, K.C.; Johnson, T.W.; Holm, N.R.; Onuma, Y.; Radu, M.D.; Joner, M.; Yu, B.; Jia, H. Clinical use of intracoronary imaging. Part 1: Guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. Eur. Heart J. 2018, 39, 3281–3300. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.W.; Raber, L.; Di Mario, C.; Bourantas, C.V.; Jia, H.; Mattesini, A.; Gonzalo, N.; de la Torre Hernandez, J.M.; Prati, F.; Koskinas, K.C. Clinical use of intracoronary imaging. Part2: Acute coronary syndromes, ambiguous coronary angiography findings, and guiding interventional decision-making: An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention 2019, 15, 434–451. [Google Scholar] [PubMed]
- Lee, J.M.; Choi, K.H.; Song, Y.B.; Lee, J.-Y.; Lee, S.-J.; Lee, S.Y.; Kim, S.M.; Yun, K.H.; Cho, J.Y.; Kim, C.J. Intravascular Imaging–Guided or Angiography-Guided Complex PCI. N. Engl. J. Med. 2023, 388, 1668–1679. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.P.; Toth, G.G.; Lai, D.; Zhu, H.; Acar, G.; Agostoni, P.; Appelman, Y.; Arslan, F.; Barbato, E.; Chen, S.L. Prognostic value of fractional flow reserve: Linking physiologic severity to clinical outcomes. J. Am. Coll. Cardiol. 2014, 64, 1641–1654. [Google Scholar] [CrossRef] [PubMed]
- Collison, D.; Didagelos, M.; Aetesam-Ur-Rahman, M.; Copt, S.; McDade, R.; McCartney, P.; Ford, T.J.; McClure, J.; Lindsay, M.; Shaukat, A.; et al. Post-stenting fractional flow reserve vs coronary angiography for optimization of percutaneous coronary intervention (TARGET-FFR). Eur. Heart J. 2021, 42, 4656–4668. [Google Scholar] [CrossRef] [PubMed]
- Rimac, G.; Fearon, W.F.; De Bruyne, B.; Ikeno, F.; Matsuo, H.; Piroth, Z.; Costerousse, O.; Bertrand, O.F. Clinical value of post-percutaneous coronary intervention fractional flow reserve value: A systematic review and meta-analysis. Am. Heart J. 2017, 183, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Koo, B.K.; Zhang, J.; Park, J.; Yang, S.; Kim, M.; Yun, J.P.; Lee, J.M.; Nam, C.W.; Shin, E.S.; et al. Prognostic Implications of Fractional Flow Reserve after Coronary Stenting: A Systematic Review and Meta-analysis. JAMA Netw. Open 2022, 5, e2232842. [Google Scholar] [CrossRef] [PubMed]
- Collet, C.; Sonck, J.; Vandeloo, B.; Mizukami, T.; Roosens, B.; Lochy, S.; Argacha, J.F.; Schoors, D.; Colaiori, I.; Di Gioia, G.; et al. Measurement of Hyperemic Pullback Pressure Gradients to Characterize Patterns of Coronary Atherosclerosis. J. Am. Coll. Cardiol. 2019, 74, 1772–1784. [Google Scholar] [CrossRef] [PubMed]
- Layland, J.; Carrick, D.; Lee, M.; Oldroyd, K.; Berry, C. Adenosine: Physiology, pharmacology, and clinical applications. JACC Cardiovasc. Interv. 2014, 7, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, C.A.; Shun-Shin, M.; Seligman, H.; Ahmad, Y.; Warisawa, T.; Cook, C.M.; Howard, J.P.; Ganesananthan, S.; Amarin, L.; Khan, C.; et al. Placebo-Controlled Efficacy of Percutaneous Coronary Intervention for Focal and Diffuse Patterns of Stable Coronary Artery Disease. Circ. Cardiovasc. Interv. 2021, 14, e009891. [Google Scholar] [CrossRef] [PubMed]
- Samady, H.; McDaniel, M.; Veledar, E.; De Bruyne, B.; Pijls, N.H.; Fearon, W.F.; Vaccarino, V. Baseline fractional flow reserve and stent diameter predict optimal post-stent fractional flow reserve and major adverse cardiac events after bare-metal stent deployment. JACC Cardiovasc. Interv. 2009, 2, 357–363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Piroth, Z.; Toth, G.G.; Tonino, P.A.L.; Barbato, E.; Aghlmandi, S.; Curzen, N.; Rioufol, G.; Pijls, N.H.J.; Fearon, W.F.; Juni, P.; et al. Prognostic Value of Fractional Flow Reserve Measured Immediately after Drug-Eluting Stent Implantation. Circ. Cardiovasc. Interv. 2017, 10, e005233. [Google Scholar] [CrossRef] [PubMed]
- Mizukami, T.; Sonck, J.; Sakai, K.; Ko, B.; Maeng, M.; Otake, H.; Koo, B.K.; Nagumo, S.; Norgaard, B.; Leipsic, J.; et al. Procedural Outcomes after Percutaneous Coronary Interventions in Focal and Diffuse Coronary Artery Disease. J. Am. Heart Assoc. 2022, 11, e026960. [Google Scholar] [CrossRef] [PubMed]
- Erlinge, D.; Gotberg, M. We need intracoronary physiology guidance before percutaneous coronary intervention, but do we need it post-stenting? Eur. Heart J. 2021, 42, 4669–4670. [Google Scholar] [CrossRef] [PubMed]
- Pijls, N.H.; Klauss, V.; Siebert, U.; Powers, E.; Takazawa, K.; Fearon, W.F.; Escaned, J.; Tsurumi, Y.; Akasaka, T.; Samady, H.; et al. Fractional Flow Reserve Post-Stent Registry, I. Coronary pressure measurement after stenting predicts adverse events at follow-up: A multicenter registry. Circulation 2002, 105, 2950–2954. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, T.; Matsumura, M.; Witzenbichler, B.; Metzger, D.C.; Rinaldi, M.J.; Duffy, P.L.; Weisz, G.; Stuckey, T.D.; Ali, Z.A.; Zhou, Z.; et al. Stent Expansion Indexes to Predict Clinical Outcomes: An IVUS Substudy From ADAPT-DES. JACC Cardiovasc. Interv. 2021, 14, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Biscaglia, S.; Tebaldi, M.; Brugaletta, S.; Cerrato, E.; Erriquez, A.; Passarini, G.; Ielasi, A.; Spitaleri, G.; Di Girolamo, D.; Mezzapelle, G.; et al. Prognostic Value of QFR Measured Immediately after Successful Stent Implantation: The International Multicenter Prospective HAWKEYE Study. JACC Cardiovasc. Interv. 2019, 12, 2079–2088. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Choi, G.; Koo, B.K.; Hwang, D.; Park, J.; Zhang, J.; Kim, K.J.; Tong, Y.; Kim, H.J.; Grady, L. Identification of High-Risk Plaques Destined to Cause Acute Coronary Syndrome Using Coronary Computed Tomographic Angiography and Computational Fluid Dynamics. JACC Cardiovasc. Imaging 2019, 12, 1032–1043. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, C.; Rao, S.; Zhang, Y.; Ghista, D.; Gao, Z.; Yang, G. Physiologically personalized coronary blood flow model to improve the estimation of noninvasive fractional flow reserve. Med. Phys. 2022, 49, 583–597. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Wang, X.; Sun, S.; Wu, D.; Bai, J.; Yin, Y.; Liu, X.; Zhang, H.; de Albuquerque, V.H.C. Learning physical properties in complex visual scenes: An intelligent machine for perceiving blood flow dynamics from static CT angiography imaging. Neural. Netw. 2020, 123, 82–93. [Google Scholar] [CrossRef] [PubMed]
| Variables | All | PIOS | Control | p-Value |
|---|---|---|---|---|
| N | 114 | 61 | 53 | |
| Clinical characteristics | ||||
| Gender (male), n (%) | 97 (85.1) | 53 (86.9) | 44 (83.0) | 0.75 |
| Age (years), mean ± SD | 59.8 ± 8.1 | 59.3 ± 8.0 | 60.4 ± 8.2 | 0.45 |
| BMI, mean ± SD | 29.7 ± 4.7 | 29.4 ± 4.3 | 30.1 ± 5.2 | 0.46 |
| Family history, n (%) | 75 (65.8) | 40 (65.6) | 35 (66.0) | 1.00 |
| Smoking, n (%) | 80 (70.2) | 44 (72.1) | 36 (67.9) | 0.78 |
| Hypertension, n (%) | 50 (43.9) | 29 (47.5) | 21 (39.6) | 0.51 |
| Dyslipidaemia, n (%) | 65 (57.0) | 33 (54.1) | 32 (60.4) | 0.63 |
| Diabetes mellitus, n (%) | 23 (20.2) | 10 (16.4) | 13 (24.5) | 0.40 |
| History of stroke, n (%) | 7 (6.1) | 5 (8.2) | 2 (3.8) | 0.56 |
| Chronic kidney disease, n (%) | 3 (2.6) | 2 (3.3) | 1 (1.9) | 1.00 |
| Prior PCI, n (%) | 51 (44.7) | 31 (50.8) | 20 (37.7) | 0.23 |
| Prior MI, n (%) | 50 (43.9) | 29 (47.5) | 21 (39.6) | 0.51 |
| Prior CABG, n (%) | 0 | 0 | 0 | NA |
| Symptomatic angina, n (%) | 95 (69.2) | 53 (86.9) | 42 (79.2) | 0.95 |
| CCS I | 21 (22.1) | 12 (22.6) | 9 (21.4) | |
| CCS II | 48 (50.5) | 26 (49.1) | 22 (52.4) | |
| CCS III | 26 (27.4) | 15 (28.3) | 11 (26.2) | |
| CCS IV | 0 (0) | 0 (0) | 0 (0) | |
| Clinical presentation, n (%) | 0.22 | |||
| Stable angina | 29 (25.4) | 13 (21.3) | 16 (30.2) | |
| ACS—NSTEMI | 44 (38.6) | 21 (34.4) | 23 (43.4) | |
| ACS—Unstable angina | 1 (0.9) | 1 (1.6) | 0 (0.0) | |
| Staged PCI | 40 (35.1) | 26 (42.6) | 14 (26.4) | |
| Procedural characteristics | ||||
| Vessel, n (%) | 0.99 | |||
| LAD | 72 (63.2) | 38 (62.3) | 34 (64.2) | |
| Non-LAD | 42 (36.8) | 23 (37.7) | 19 (35.8) | |
| QCA diameter stenosis (%), mean | 61.0 | 60.9 | 61.2 | 0.92 |
| QCA lesion length (mm), median (IQR) | 10.8 [7.96, 13.2] | 10.7 [8.34, 13.6] | 10.9 [6.93, 13.0] | 0.73 |
| Intravascular imaging, n (%) | 22 (19.3) | 10 (16.4) | 12 (22.6) | 0.55 |
| IVUS, n (%) | 19 (86.4) | 10 (100) | 9 (77.4) | |
| OCT, n (%) | 3 (13.6) | 0 (0.0) | 3 (23.1) | |
| Stent diameter (mm), median (IQR) | 3.00 [3.0, 3.5] | 3.00 [3.0, 3.5] | 3.00 [3.0, 3.5] | 0.45 |
| Stent length (mm), median (IQR) | 32.0 [23.0, 38.0] | 32.0 [23.0, 38.0] | 28.0 [23.0, 38.0] | 0.76 |
| Number of stents per patients, median (IQR) | 1.00 [1.00, 2.00] | 1.00 [1.00, 2.00] | 1.00 [1.00, 2.00] | 0.97 |
| Total stent length (mm), median (IQR) | 38.0 [25.0, 50.8] | 33.0 [28.0, 52.0] | 38.0 [24.0, 50.0] | 0.76 |
| Physiological characteristics | ||||
| Pre-PCI Pd/Pa*, median (IQR) | 0.85 [0.75, 0.90] | 0.86 [0.81, 0.91] | 0.83 [0.72, 0.89] | 0.11 |
| Pre-PCI CFR*, median (IQR) | 2.03 [1.47, 2.61] | 2.11 [1.49, 2.60] | 1.99 [1.31, 2.60] | 0.43 |
| Pre-PCI IMR*, median (IQR) | 22.9 [16.6, 31.8] | 24.0 [18.0, 33.5] | 21.7 [16.0, 31.3] | 0.31 |
| Pre-PCI FFR*, median (IQR) | 0.62 ± 0.14 | 0.65 ± 0.12 | 0.58 ± 0.15 | 0.04 |
| Final post-PCI FFR*, mean ± SD | 0.85 ± 0.07 | 0.86 ± 0.08 | 0.85 ± 0.07 | 0.27 |
| Normalised delta FFR* | 59.2 ± 21.6 | 59.4 ± 20.8 | 58.9 ± 22.7 | 0.91 |
| Final post-PCI FFR ≤ 0.80 (%), n (%) | 27 (23.7) | 13 (21.3) | 14 (26.4) | 0.68 |
| Final post-PCI FFR ≥ 0.80 (%), n (%) | 88 (77.2) | 48 (78.7) | 40 (75.5) | 0.85 |
| Final post-PCI FFR ≥ 0.90 (%), n (%) | 32 (28.1) | 22 (36.1) | 10 (18.9) | 0.07 |
| PPG, median (IQR) | 0.66 [0.55, 0.78] | 0.64 [0.56, 0.79] | 0.68 [0.54, 0.79] | 0.65 |
| Residual PPG* | 0.07 ± 0.04 | 0.06 ± 0.04 | 0.07 ± 0.04 | 0.06 |
| Variables | PIOS | Controls | p-Value *** | ||||
|---|---|---|---|---|---|---|---|
| Focal CAD | Diffuse CAD | p-Value * | Focal CAD | Diffuse CAD | p-Value ** | ||
| Number, (%) | 18 (29.5) | 43 (70.5) | 20 (37.7) | 33 (62.3) | |||
| Vessel (%) | <0.001 | <0.001 | <0.001 | ||||
| LAD, n (%) | 2 (11.1) | 36 (83.7) | 6 (30.0) | 28 (84.8) | |||
| LCx, n (%) | 10 (55.6) | 2 (4.7) | 3 (15.0) | 2 (6.1) | |||
| RCA, n (%) | 7 (33.3) | 3 (11.6) | 11 (55.0) | 3 (9.1) | |||
| Baseline coronary physiology | |||||||
| Pd/Pa, median (IQR) | 0.93 [0.83, 0.95] | 0.85 [0.81, 0.88] | 0.01 | 0.76 [0.69, 0.91] | 0.85 [0.76, 0.89] | 0.77 | 0.048 |
| CFR, median (IQR) | 2.00 [1.78, 2.21] | 2.25 [1.42, 2.69] | 0.33 | 1.71 [1.21, 2.012 | 2.24 [1.66 2.71] | 0.07 | 0.19 |
| IMR, median (IQR) | 25.1 [19.9, 37.5] | 23.9 [17.8, 32.0] | 0.55 | 24.1 [18.6, 31.8] | 20.8 [15.8, 28.5] | 0.42 | 0.59 |
| FFR, mean ± SD | 0.69 ± 0.13 | 0.63 ± 0.11 | 0.048 | 0.55 ± 0.15 | 0.61 ± 0.15 | 0.17 | 0.02 |
| PPG, median (IQR) | 0.81 [0.78, 0.82] | 0.58 [0.53, 0.66] | <0.001 | 0.81 [0.78, 0.82] | 0.59 [0.48, 0.66] | <0.001 | <0.001 |
| Immediately after stenting | |||||||
| Stent post-dilatation, n (%) | 18 (100.0) | 43 (100.0) | 1 | 18 (90.5) | 33 (100.0) | 0.27 | 0.02 |
| Intravascular imaging, n (%) | 1 (5.6) | 9 (20.9) | 0.26 | 0 (0) | 12 (36.4) | 0.006 | 0.004 |
| Pd/Pa, median (IQR) | 0.98 [0.97, 1.01] | 0.90 [0.88, 0.92] | <0.001 | 0.98 [0.93, 1.00] | 0.92 [0.89, 0.94] | 0.001 | <0.001 |
| CFR, median (IQR) | 4.44 [2.39, 5.81] | 2.71 [1.97, 4.00] | 0.01 | 3.28 [2.44, 5.41] | 2.74 [2.41, 4.13] | 0.25 | 0.04 |
| IMR, median (IQR) | 16.1 [13.2, 23.1] | 18.9 [13.1, 26.3] | 0.31 | 13.1 [10.7, 19.5] | 17.3 [13.1, 22.7] | 0.06 | 0.07 |
| FFR, mean ± SD | 0.93 ± 0.05 | 0.80 ± 0.09 | <0.001 | 0.87 ± 0.07 | 0.83 ± 0.07 | 0.09 | <0.001 |
| 1st PIOS treatments | |||||||
| 1st PIOS treatments performed, n (%) | 1 (5.6) | 18 (41.9) | 0.006 | - | - | - | |
| Additional stent post-dilatation, n (%) | 1 (5.6) | 11 (25.6) | 0.09 | - | - | - | |
| Additional lesion treated PCI, n (%) | 0 (0) | 8 (18.6) | 0.09 | - | - | - | |
| Additional intravascular imaging, n (%) | 0 (0.0) | 1 (2.3) | 1 | - | - | - | |
| Pd/Pa, median (IQR) | 0.98 [0.97, 1.01] | 0.90 [0.89, 0.93] | <0.001 | - | |||
| CFR, median (IQR) | 4.44 [2.59, 5.81] | 3.46 [2.01, 4.14] | 0.07 | - | - | - | |
| IMR, median (IQR) | 16.1 [13.2, 18.7] | 17.1 [13.1, 25.2] | 0.52 | - | |||
| FFR, mean ± SD | 0.93 ± 0.05 | 0.83 ± 0.07 | <0.001 | - | - | - | |
| 2nd PIOS treatments | |||||||
| 2nd PIOS treatments performed, n (%) | 0 (0) | 2 (4.7) | 1 | - | - | - | |
| Additional stent post-dilatation, n (%) | 0 (0) | 2 (4.7) | 1 | - | - | - | |
| Additional lesion treated, n (%) | 0 (0) | 2 (4.7) | 1 | - | - | - | |
| Additional intravascular imaging, n (%) | 0 (0) | 2 (4.7) | 1 | - | - | - | |
| Pd/Pa, median (IQR) | 0.98 [0.97, 1.01] | 0.91 [0.90, 0.93] | <0.001 | - | - | - | |
| CFR, median (IQR) | 4.44 [2.59, 5.81] | 3.23 [2.01, 4.11] | 0.051 | - | - | - | |
| IMR, median (IQR) | 16.1 [13.2, 18.7] | 17.6 [13.1, 25.2] | 0.49 | - | - | - | |
| FFR, mean ± SD | 0.93 ± 0.05 | 0.83 ± 0.07 | <0.001 | - | - | - | |
| Final coronary physiology | |||||||
| Number of stents per patients, median (IQR) | 1.00 [1.00, 1.00] | 1.00 [1.00, 2.00] | 0.14 | 1.00 [1.00, 2.00] | 1.00 [1.00, 2.00] | 0.28 | 0.34 |
| Total stent length (mm), median (IQR) | 32.0 [25.0, 36.8] | 38.0 [28.0, 56.0] | 0.12 | 38.0 [30.3, 48.0] | 38.0 [24.0, 56.0] | 0.93 | 0.47 |
| Pd/Pa, median (IQR) | 0.98 [0.97, 1.01] | 0.91 [0.90, 0.93] | <0.001 | 0.98 [0.93, 1.00] | 0.92 [0.89, 0.94] | 0.001 | <0.001 |
| CFR, median (IQR) | 4.44 [2.59, 6.14] | 3.23 [2.01, 4.11] | 0.051 | 3.28 [2.44, 5.41] | 2.74 [2.41, 4.13] | 0.25 | 0.18 |
| IMR, median (IQR) | 16.1 [13.2, 18.7] | 17.6 [13.01 25.2] | 0.53 | 13.1 [10.7, 19.5] | 17.3 [13.1, 22.7] | 0.07 | 0.17 |
| FFR, mean ± SD | 0.93 ± 0.05 | 0.83 ± 0.07 | <0.001 | 0.87 ± 0.07 | 0.83 ± 0.07 | 0.057 | <0.001 |
| Delta Pd/Pa, median (IQR) | 0.06 [0.04, 0.15] | 0.06 [0.03, 0.10] | 0.82 | 0.20 [0.07, 0.28] | 0.06 [0.04, 0.19] | 0.079 | 0.09 |
| Delta CFR, median (IQR) | 2.36 [0.79, 3.18] | 0.63 [−0.05, 1.92] | 0.01 | 1.60 [0.78, 2.90] | 1.04 [0.20, 1.97] | 0.11 | 0.03 |
| Delta IMR, median (IQR) | −8.23 [−13.2, −3.09] | −3.55 [−9.98, −0.28] | 0.19 | −11.3 [−15.1, −5.67] | −3.13 [−9.71, 1.26] | 0.04 | 0.10 |
| Normalised delta FFR (%), mean ± SD | 76.5 ± 15.5 | 52.2 ± 18.5 | <0.001 | 67.8 ± 23.5 | 53.0 ± 20.5 | 0.02 | <0.001 |
| FFR ≥ 0.90 (%), n (%) | 14 (77.8) | 8 (18.6) | <0.001 | 6 (30.0) | 4 (12.1) | 0.21 | <0.001 |
| FFR ≥ 0.80 (%), n (%) | 0 (0.0) | 13 (30.2) | 0.02 | 5 (25.0) | 9 (27.3) | 0.79 | 0.07 |
| Residual PPG | 0.04 ± 0.02 | 0.07 ± 0.04 | 0.004 | 0.07 ± 0.04 | 0.08 ± 0.04 | 0.44 | 0.008 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohashi, H.; Collison, D.; Mizukami, T.; Didagelos, M.; Sakai, K.; Aetesam-ur-Rahman, M.; Munhoz, D.; McCartney, P.; Ford, T.J.; Lindsay, M.; et al. Fractional Flow Reserve-Guided Stent Optimisation in Focal and Diffuse Coronary Artery Disease. Diagnostics 2023, 13, 2612. https://doi.org/10.3390/diagnostics13152612
Ohashi H, Collison D, Mizukami T, Didagelos M, Sakai K, Aetesam-ur-Rahman M, Munhoz D, McCartney P, Ford TJ, Lindsay M, et al. Fractional Flow Reserve-Guided Stent Optimisation in Focal and Diffuse Coronary Artery Disease. Diagnostics. 2023; 13(15):2612. https://doi.org/10.3390/diagnostics13152612
Chicago/Turabian StyleOhashi, Hirofumi, Damien Collison, Takuya Mizukami, Matthaios Didagelos, Koshiro Sakai, Muhammad Aetesam-ur-Rahman, Daniel Munhoz, Peter McCartney, Thomas J. Ford, Mitchell Lindsay, and et al. 2023. "Fractional Flow Reserve-Guided Stent Optimisation in Focal and Diffuse Coronary Artery Disease" Diagnostics 13, no. 15: 2612. https://doi.org/10.3390/diagnostics13152612
APA StyleOhashi, H., Collison, D., Mizukami, T., Didagelos, M., Sakai, K., Aetesam-ur-Rahman, M., Munhoz, D., McCartney, P., Ford, T. J., Lindsay, M., Shaukat, A., Rocchiccioli, P., Brogan, R., Watkins, S., McEntegart, M., Good, R., Robertson, K., O’Boyle, P., Davie, A., ... Collet, C. (2023). Fractional Flow Reserve-Guided Stent Optimisation in Focal and Diffuse Coronary Artery Disease. Diagnostics, 13(15), 2612. https://doi.org/10.3390/diagnostics13152612






