Diagnostic Performance of Extrahepatic Protein Induced by Vitamin K Absence in the Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Caviglia, G.; Ciruolo, M.; Abate, M.; Carucci, P.; Rolle, E.; Rosso, C.; Olivero, A.; Troshina, G.; Risso, A.; Nicolosi, A.; et al. Alpha-fetoprotein, protein induced by vitamin K absence or antagonist II and glypican-3 for the detection and prediction of hepatocellular carcinoma in patients with cirrhosis of viral etiology. Cancers 2020, 12, 3218. [Google Scholar] [CrossRef]
- Venook, A.P.; Papandreou, C.; Furuse, J.; Ladrón de Guevara, L. The Incidence and Epidemiology of Hepatocellular Carcinoma: A Global and Regional Perspective. Oncologist 2010, 15, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Caviglia, G.P.; Ribaldone, D.G.; Abate, M.L.; Ciancio, A.; Pellicano, R.; Smedile, A.; Saracco, G.M. Performance of protein induced by vitamin K absence or antagonist-II assessed by chemiluminescence enzyme immunoassay for hepatocellular carcinoma detection: A meta-analysis. Scand. J. Gastroenterol. 2018, 53, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Caviglia, G.P.; Abate, M.L.; Pellicano, R.; Smedile, A. Chronic hepatitis B therapy: Available drugs and treatment guidelines. Minerva Gastroenterol. Dietol. 2015, 61, 61–70. [Google Scholar] [PubMed]
- Kanwal, F.; Kramer, J.; Asch, S.M.; Chayanupatkul, M.; Cao, Y.; El-Serag, H.B. Risk of Hepatocellular Cancer in HCV Patients Treated with Direct-Acting Antiviral Agents. Gastroenterology 2017, 153, 996–1005.e1. [Google Scholar] [CrossRef]
- Bhatti, A.B.H.; Naz, K.; Abbas, G.; Khan, N.Y.; Zia, H.H.; Ahmed, I.N. Clinical Utility of Protein Induced by Vitamin K Absence-II in Patients with Hepatocellular Carcinoma. Asian Pacific. J. Cancer. Prev. 2021, 22, 1731–1736. [Google Scholar] [CrossRef]
- Fan, J.; Chen, Y.; Zhang, D.; Yao, J.; Zhao, Z.; Jiang, Y.; Li, Y.; Guo, Y. Evaluation of the diagnostic accuracy of des-gamma-carboxy prothrombin and alpha-fetoprotein alone or in combination for hepatocellular carcinoma: A systematic review and meta-analysis. Surg. Oncol. 2020, 34, 245–255. [Google Scholar] [CrossRef]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Michelotti, A.; de Scordilli, M.; Palmero, L.; Guardascione, M.; Masala, M.; Roncato, R.; Foltran, L.; Ongaro, E.; Puglisi, F. NAFLD-Related Hepatocarcinoma: The Malignant Side of Metabolic Syndrome. Cells 2021, 10, 2034. [Google Scholar] [CrossRef]
- Sumida, Y.; Yoneda, M.; Seko, Y.; Ishiba, H.; Hara, T.; Toyoda, H.; Yasuda, S.; Kumada, T.; Hayashi, H.; Kobayashi, T.; et al. Surveillance of Hepatocellular Carcinoma in Nonalcoholic Fatty Liver Disease. Diagnostics 2020, 10, 579. [Google Scholar] [CrossRef]
- Bruix, J.; Sherman, M. Management of hepatocellular carcinoma: An update. Hepatology 2011, 53, 1020–1022. [Google Scholar] [CrossRef] [PubMed]
- Basile, U.; Miele, L.; Napodano, C.; Ciasca, G.; Gulli, F.; Pocino, K.; De Matthaeis, N.; Liguori, A.; De Magistris, A.; Marrone, G.; et al. The diagnostic performance of PIVKA-II in metabolic and viral hepatocellular carcinoma: A pilot study. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 12675–12685. [Google Scholar]
- Lombardi, A.; Grimaldi, A.; Zappavigna, S.; Misso, G.; Caraglia, M. Hepatocarcinoma: Genetic and epigenetic features. Minerva Gastroenterol. Dietol. 2017, 64, 14–27. [Google Scholar] [CrossRef]
- Choi, J.Y.; Jung, S.W.; Kim, H.Y.; Kim, M.; Kim, Y.; Kim, D.G.; Oh, E.-J. Diagnostic value of AFP-L3 and PIVKA-II in hepatocellular carcinoma according to total-AFP. World J. Gastroenterol. 2013, 19, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Izumi, N. Diagnostic and treatment algorithm of the Japanese society of hepatology: A consensus-based practice guideline. Oncology 2010, 78 (Suppl. 1), 78–86. [Google Scholar] [CrossRef] [PubMed]
- Saffroy, R.; Pham, P.; Reffas, M.; Takka, M.; Lemoine, A.; Debuire, B. New perspectives and strategy research biomarkers for hepatocellular carcinoma. Clin. Chem. Lab. Med. 2007, 45, 1169–1179. [Google Scholar] [CrossRef]
- Korean Liver Cancer Study Group. Practice guidelines for management of hepatocellular carcinoma 2009. Korean J. Hepatol. 2009, 15, 391. [Google Scholar] [CrossRef] [PubMed]
- Durazo, F.A.; Blatt, L.M.; Corey, W.G.; Lin, J.H.; Han, S.; Saab, S.; Busuttil, R.W.; Tong, M.J. Des-γ-carboxyprothrombin, α-fetoprotein and AFP-L3 in patients with chronic hepatitis, cirrhosis and hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2008, 23, 1541–1548. [Google Scholar] [CrossRef]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.-L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- European Association for The Study of The Liver; European Organization for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2012, 56, 908–943. [Google Scholar] [CrossRef]
- Caviglia, G.; Armandi, A.; Rosso, C.; Gaia, S.; Aneli, S.; Rolle, E.; Abate, M.; Olivero, A.; Nicolosi, A.; Guariglia, M.; et al. Biomarkers of oncogenesis, adipose tissue dysfunction and systemic inflammation for the detection of hepatocellular carcinoma in patients with nonalcoholic fatty liver disease. Cancers 2021, 13, 2305. [Google Scholar] [CrossRef]
- Liu, S.; Sun, L.; Yao, L.; Zhu, H.; Diao, Y.; Wang, M.; Xing, H.; Lau, W.Y.; Guan, M.; Pawlik, T.M.; et al. Diagnostic Performance of AFP, AFP-L3, or PIVKA-II for Hepatitis C Virus-Associated Hepatocellular Carcinoma: A Multicenter Analysis. J. Clin. Med. 2022, 11, 5075. [Google Scholar] [CrossRef]
- Chen, V.L.; Sharma, P. Role of Biomarkers and Biopsy in Hepatocellular Carcinoma. Clin. Liver. Dis. 2020, 24, 577–590. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Guo, Q.; Wei, L. The emerging influences of alpha-fetoprotein in the tumorigenesis and progression of hepatocellular carcinoma. Cancers 2021, 13, 5096. [Google Scholar] [CrossRef]
- Li, C.; Zhang, Z.; Zhang, P.; Liu, J. Diagnostic accuracy of des-gamma-carboxy prothrombin versus α-fetoprotein for hepatocellular carcinoma: A systematic review. Hepatol. Res. 2014, 44, E11–E25. [Google Scholar] [CrossRef]
- Sherman, M.; Peltekian, K.M.; Lee, C. Screening for hepatocellular carcinoma in chronic carriers of hepatitis B virus: Incidence and prevalence of hepatocellular carcinoma in a North American urban population. Hepatology 1995, 22, 432–438. [Google Scholar]
- Toyoda, H.; Kumada, T.; Tada, T. Highly sensitive Lens culinaris agglutinin-reactive α-fetoprotein: A new tool for the management of hepatocellular carcinoma. Oncology 2011, 81 (Suppl. 1), 61–65. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Asakawa, M.; Amemiya, H.; Fujii, H. Lens culinaris agglutinin-reactive fraction of AFP is a useful prognostic biomarker for survival after repeat hepatic resection for HCC. J. Gastroenterol. Hepatol. 2011, 26, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; He, J.; Zhang, F.; Wang, B.; Ding, H.; Shen, H.; Li, Y.; Chen, X. Diagnostic value of protein induced by vitamin K absence (PIVKAII) and hepatoma-specific band of serum gamma-glutamyl transferase (GGTII) as hepatocellular carcinoma markers complementary to α-fetoprotein. Br. J. Cancer. 2003, 88, 1878–1882. [Google Scholar] [CrossRef]
- Chi, X.; Jiang, L.; Yuan, Y.; Huang, X.; Yang, X.; Hochwald, S.; Liu, J.; Huang, H. A comparison of clinical pathologic characteristics between alpha-fetoprotein negative and positive hepatocellular carcinoma patients from Eastern and Southern China. BMC Gastroenterol. 2022, 22, 202. [Google Scholar] [CrossRef]
- Petrini, E.; Caviglia, G.; ABate, M.; Fagoonee, S.; Smedile, A.; Pellicano, R. MicroRNAs in HBV-related hepatocellular carcinoma: Functions and potential clinical applications. Panminerva Med. 2011, 47, 381–390. [Google Scholar]
- Jia, H.; Yu, H.; Liu, Q. Single nucleotide polymorphisms of MIR-149 gene rs2292832 contributes to the risk of hepatocellular carcinoma, but not overall cancer: A meta-analysis. Minerva Med. 2016, 107, 259–269. [Google Scholar]
- Taketa, K.; Sekiya, C.; Namiki, M.; Akamatsu, K.; Ohta, Y.; Endo, Y.; Kosaka, K. Lectin-reactive profiles of alpha-fetoprotein characterizing hepatocellular carcinoma and related conditions. Gastroenterology 1990, 99, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Taketa, K.; Endo, Y.; Sekiya, C.; Tanikawa, K.; Koji, T.; Taga, H.; Satomura, S.; Matsuura, S.; Kawai, T.; Hirai, H. A collaborative study for the evaluation of lectin-reactive alpha-fetoproteins in early detection of hepatocellular carcinoma. Cancer Res. 1993, 53, 5419–5423. [Google Scholar] [PubMed]
- Wu, Z.; Cheng, H.; Liu, J.; Zhang, S.; Zhang, M.; Liu, F.; Li, Y.; Huang, Q.; Jiang, Y.; Chen, S.; et al. The Oncogenic and Diagnostic Potential of Stanniocalcin 2 in Hepatocellular Carcinoma. J. Hepatocell. Carcinoma 2022, 9, 141–155. [Google Scholar] [CrossRef]
- Cao, L.; Cheng, H.; Jiang, Q.; Li, H.; Wu, Z. APEX1 is a novel diagnostic and prognostic biomarker for hepatocellular carcinoma. Aging 2020, 12, 4573–4591. [Google Scholar] [CrossRef]
- Liebman, H.A.; Furie, B.C.; Tong, M.J.; Blanchard, R.A.; Lo, K.-J.; Lee, S.-D.; Coleman, M.S.; Furie, B. Des-gamma-carboxi (abnormal) prothrombin as a serum marker of primary Hepatocellular carcinoma. N. Engl. J. Med. 1984, 310, 1427–1431. [Google Scholar] [CrossRef] [PubMed]
- Thomas, O.; Rein, H.; Strandberg, K.; Schött, U. Coagulative safety of epidural catheters after major upper gastrointestinal surgery: Advanced and routine coagulation analysis in 38 patients. Perioper. Med. 2016, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Wang, N.; Yang, Y.; Ma, L.; Du, Q.; Zhang, W.; Tran, A.; Jung, H.; Soh, A.; Zheng, Y.; et al. Review on Vitamin K Deficiency and its Biomarkers: Focus on the Novel Application of PIVKA-II in Clinical Practice. Clin Lab. 2018, 64, 413–424. [Google Scholar] [CrossRef]
- Kato, K.; Iwasaki, Y.; Taniguchi, M.; Onodera, K.; Matsuda, M.; Kawakami, T.; Higuchi, M.; Kato, K.; Kato, Y.; Furukawa, H. Primary colon cancer with a high serum PIVKA-II level. Int. J. Surg. Case Rep. 2015, 6, 95–99. [Google Scholar] [CrossRef]
- Yu, R.; Tan, Z.; Xiang, X.; Dan, Y.; Deng, G. Effectiveness of PIVKA-II in the detection of hepatocellular carcinoma based on real-world clinical data. BMC Cancer 2017, 17, 608. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, Y.; Tang, W.; Xu, H.; Wang, F.; Nakata, M.; Sugawara, Y.; Kokudo, N. Des-gamma-carboxyprothrombin: Clinical effectiveness and biochemical importance. Biosci. Trends. 2008, 2, 53–60. [Google Scholar] [PubMed]
- Baek, Y.-H.; Lee, J.-H.; Jang, J.-S.; Lee, S.-W.; Han, J.-Y.; Jeong, J.-S.; Choi, J.-C.; Kim, H.-Y.; Han, S.-Y. Diagnostic role and correlation with staging systems of PIVKA-II compared with AFP. Hepatogastroenterology 2009, 56, 763–767. [Google Scholar]
- Shirabe, K.; Itoh, S.; Yoshizumi, T.; Soejima, Y.; Taketomi, A.; Aishima, S.-I.; Maehara, Y. The predictors of microvascular invasion in candidates for liver transplantation with hepatocellular carcinoma—With special reference to the serum levels of des-gamma-carboxy prothrombin. J. Surg Oncol. 2007, 95, 235–240. [Google Scholar] [CrossRef]
- Kim, D.Y.; Paik, Y.H.; Ahn, S.H.; Youn, Y.J.; Choi, J.W.; Kim, J.K.; Lee, K.S.; Chon, C.Y.; Han, K.H. PIVKA-II is a useful tumor marker for recurrent hepatocellular carcinoma after surgical resection. Oncology 2007, 72 (Suppl. 1), 52–57. [Google Scholar] [CrossRef]
- Song, P.-P.; Xia, J.-F.; Inagaki, Y.; Hasegawa, K.; Sakamoto, Y.; Kokudo, N.; Tang, W. Controversies regarding and perspectives on clinical utility of biomarkers in hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 262–274. [Google Scholar] [CrossRef]
- Choi, J.; Park, Y.; Kim, J.H.; Kim, H.S. Evaluation of automated serum des-gamma-carboxyprothrombin (DCP) assays for detecting hepatocellular carcinoma. Clin. Biochem. 2011, 44, 1464–1468. [Google Scholar] [CrossRef] [PubMed]
- Arii, S.; Sata, M.; Sakamoto, M.; Shimada, M.; Kumada, T.; Shiina, S.; Yamashita, T.; Kokudo, N.; Tanaka, M.; Takayama, T.; et al. Management of hepatocellular carcinoma: Report of Consensus Meeting in the 45th Annual Meeting of the Japan Society of Hepatology (2009). Hepatol. Res. 2010, 40, 667–685. [Google Scholar] [CrossRef] [PubMed]
- Van Hees, S.; Michielsen, P.; Vanwolleghem, T. Circulating predictive and diagnostic biomarkers for hepatitis B virus-associated hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 8271–8282. [Google Scholar] [CrossRef]
- Vitale, A.; Morales, R.R.; Zanus, G.; Farinati, F.; Burra, P.; Angeli, P.; Frigo, A.C.; Del Poggio, P.; Rapaccini, G.; Di Nolfo, M.A.; et al. Barcelona Clinic Liver Cancer staging and transplant survival benefit for patients with hepatocellular carcinoma: A multicentre, cohort study. Lancet Oncol. 2011, 12, 654–662. [Google Scholar] [CrossRef]
- Forner, A.; Reig, M.E.; Rodriguez de Lope, C.; Bruix, J. Current Strategy for Staging and Treatment: The BCLC Update and Future Prospects. Semin. Liver Dis. 2010, 30, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Zhang, D. The 8th Edition American Joint Committee on Cancer Staging for Hepato-pancreato-biliary Cancer: A Review and Update. Arch. Pathol. Lab. Med. 2021, 145, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Schotten, C.; Ostertag, B.; Sowa, J.-P.; Manka, P.; Bechmann, L.; Hilgard, G.; Marquardt, C.; Wichert, M.; Toyoda, H.; Lange, C.; et al. Galad score detects early-stage hepatocellular carcinoma in a european cohort of chronic hepatitis b and c patients. Pharmaceuticals 2021, 14, 735. [Google Scholar] [CrossRef]
- Choi, J.; Kim, G.A.; Han, S.; Lee, W.; Chun, S.; Lim, Y.S. Longitudinal Assessment of Three Serum Biomarkers to Detect Very Early-Stage Hepatocellular Carcinoma. Hepatology 2019, 69, 1983–1994. [Google Scholar] [CrossRef]
- Best, J.; Bechmann, L.P.; Sowa, J.P.; Sydor, S.; Dechêne, A.; Pflanz, K.; Bedreli, S.; Schotten, C.; Geier, A.; Berg, T.; et al. GALAD Score Detects Early Hepatocellular Carcinoma in an International Cohort of Patients with Nonalcoholic Steatohepatitis. Clin. Gastroenterol. Hepatol. 2020, 18, 728–735. [Google Scholar] [CrossRef]
- Wu, M.; Liu, Z.; Li, X.; Zhang, A.; Li, N. Dynamic Changes in Serum Markers and Their Utility in the Early Diagnosis of all Stages of hepatitis B-associated Hepatocellular carcinoma. Onco Targets Ther. 2020, 13, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Malov, S.; Malov, I.; Kuvshinov, A.; Marche, P.; Decaens, T.; Macek-Jilkova, Z.; Yushchuk, N. Search for effective serum tumor markers for early diagnosis of hepatocellular carcinoma associated with hepatitis. Sovrem. Teh. V Med. 2021, 13, 27–34. [Google Scholar] [CrossRef]
- Chan, H.L.Y.; Vogel, A.; Berg, T.; De Toni, E.N.; Kudo, M.; Trojan, J.; Eiblmaier, A.; Klein, H.; Hegel, J.K.; Sharma, A.; et al. Performance evaluation of the Elecsys PIVKA-II and Elecsys AFP assays for hepatocellular carcinoma diagnosis. JGH Open. 2022, 6, 292–300. [Google Scholar] [CrossRef]
- Chalasani, N.P.; Porter, K.; Bhattacharya, A.; Book, A.J.; Neis, B.M.; Xiong, K.M.; Ramasubramanian, T.S.; Edwards, D.K.; Chen, I.; Johnson, S.; et al. Validation of a Novel Multitarget Blood Test Shows High Sensitivity to Detect Early Stage Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2022, 20, 173–182.e7. [Google Scholar] [CrossRef]
- Nouso, K.; Furubayashi, Y.; Shiota, S.; Miyake, N.; Oonishi, A.; Wakuta, A.; Kariyama, K.; Hiraoka, A.; Tsuji, K.; Itobayashi, E.; et al. Early detection of hepatocellular carcinoma in patients with diabetes mellitus. Eur. J. Gastroenterol. Hepatol. 2020, 32, 877–881. [Google Scholar] [CrossRef]
- Hemken, P.M.; Sokoll, L.J.; Yang, X.; Dai, J.; Elliott, D.; Gawel, S.H.; Lucht, M.; Feng, Z.; Marrero, J.A.; Srivastava, S.; et al. Validation of a novel model for the early detection of hepatocellular carcinoma. Clin. Proteom. 2019, 16, 2. [Google Scholar] [CrossRef]
- Unić, A.; Derek, L.; Duvnjak, M.; Patrlj, L.; Rakic, M.; Kujundžić, M.; Renjić, V.; Štoković, N.; Dinjar, P.; Jukic, A.; et al. Diagnostic specificity and sensitivity of PIVKAII, GP3, CSTB, SCCA1 and HGF for the diagnosis of hepatocellular carcinoma in patients with alcoholic liver cirrhosis. Ann. Clin. Biochem. 2018, 55, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wu, M.; Lin, D.; Li, N. Des-gamma-carboxyprothrombin is a favorable biomarker for the early diagnosis of alfa-fetoprotein-negative hepatitis B virus-related hepatocellular carcinoma. J. Int. Med. Res. 2020, 48, 300060520902575. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Wang, L.; Xin, R.; Zhang, L.; Tian, Y. Evaluation of serum AFP and DCP levels in the diagnosis of early-stage HBV-related HCC under different backgrounds. J. Int. Med. Res. 2020, 48, 0300060520969087. [Google Scholar] [CrossRef] [PubMed]
- Loglio, A.; Iavarone, M.; Facchetti, F.; Di Paolo, D.; Perbellini, R.; Lunghi, G.; Ceriotti, F.; Galli, C.; Sandri, M.T.; Viganò, M.; et al. The combination of PIVKA-II and AFP improves the detection accuracy for HCC in HBV caucasian cirrhotics on long-term oral therapy. Liver Int. 2020, 40, 1987–1996. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, Y.; Li, S.; Li, N.; Chen, Y.; Zhang, B.; Qu, C.; Ding, H.; Huang, J.; Dai, M. Direct comparison of five serum biomarkers in early diagnosis of hepatocellular carcinoma. Cancer Manag. Res. 2018, 10, 1947–1958. [Google Scholar] [CrossRef]
- Piratvisuth, T.; Tanwandee, T.; Thongsawat, S.; Sukeepaisarnjaroen, W.; Esteban, J.I.; Bes, M.; Köhler, B.; He, Y.; Lange, M.S.; Morgenstern, D.; et al. Multimarker Panels for Detection of Early Stage Hepatocellular Carcinoma: A Prospective, Multicenter, Case-Control Study. Hepatol. Commun. 2022, 6, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Wang, L.; Su, B.; Zeng, W.; Jiang, T.; Zhang, T.; Sun, G.; Wu, H. Diagnostic value of alpha-fetoprotein, Lens culinaris agglutinin-reactive alpha-fetoprotein, and des-gamma-carboxyprothrombin in hepatitis B virus-related hepatocellular carcinoma. J. Int. Med. Res. 2019, 48, 300060519889270. [Google Scholar] [CrossRef]
- Degasperi, E.; Perbellini, R.; D’Ambrosio, R.; Renteria, S.C.U.; Ceriotti, F.; Perego, A.; Orsini, C.; Borghi, M.; Iavarone, M.; Bruccoleri, M.; et al. Prothrombin induced by vitamin K absence or antagonist-II and alpha foetoprotein to predict development of hepatocellular carcinoma in Caucasian patients with hepatitis C-related cirrhosis treated with direct-acting antiviral agents. Aliment. Pharm. Ther. 2022, 55, 350–359. [Google Scholar] [CrossRef]
- Wu, J.; Xiang, Z.; Le Bai, L.; He, L.; Tan, L.; Hu, M.; Ren, Y. Diagnostic value of serum PIVKA-II levels for BCLC early hepatocellular carcinoma and correlation with HBV DNA. Cancer Biomark. 2018, 23, 235–242. [Google Scholar] [CrossRef]
- Qi, F.; Zhou, A.; Yan, L.; Yuan, X.; Wang, D.; Chang, R.; Zhang, Y.; Shi, F.; Han, X.; Hou, J.; et al. The diagnostic value of PIVKA-II, AFP, AFP-L3, CEA, and their combinations in primary and metastatic hepatocellular carcinoma. J. Clin. Lab. Anal. 2020, 34, e23158. [Google Scholar] [CrossRef]
- Li, Y.; Chen, J. Serum Des-Gamma-carboxi Prothrombin for diagnosis of adult primary cancer in liver. J. Coll. Physician Surg. Pak. 2018, 29, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Guan, M.; Ouyang, W.; Liu, S.; Sun, L.; Chen, W. Alpha-fetoprotein, protein induced by vitamin K absence or antagonist-II, lens culinaris agglutinin-reactive fraction of alpha-fetoprotein alone and in combination for early detection of hepatocellular carcinoma from nonalcoholic fatty liver disease: A multicenter analysis. Hepatobiliary Pancreat. Dis. Int. 2022, 21, 559–568. [Google Scholar] [PubMed]
- Si, Y.-Q.; Wang, X.-Q.; Fan, G.; Wang, C.-Y.; Zheng, Y.-W.; Song, X.; Pan, C.-C.; Chu, F.-L.; Liu, Z.-F.; Lu, B.-R.; et al. Value of AFP and PIVKA-II in diagnosis of HBV-related hepatocellular carcinoma and prediction of vascular invasion and tumor differentiation. Infect. Agent Cancer. 2020, 15, 70. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Liu, L.; Jiang, F.; Wen, X.; Zhang, Y.; Li, S.; Lou, J.; Wang, Y.; Liu, N.; Guo, Q.; et al. The clinical application of PIVKA-II in hepatocellular carcinoma and chronic liver diseases: A multi-center study in China. J. Clin. Lab. Anal. 2021, 35, e24013. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lu, X.; Du, Q.; Zhang, G.; Wang, D.; Wang, Q.; Guo, X. Diagnostic value of the γ-glutamyltransferase and alanine transaminase ratio, alpha-fetoprotein, and protein induced by vitamin K absence or antagonist II in hepatitis B virus-related hepatocellular carcinoma. Sci. Rep. 2020, 10, 13519. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, Q.; Zhang, X.; Lu, X.-L.; Du, Q.; Zhu, T.; Zhang, G.-Y.; Wang, D.-S.; Fan, Q.-M. Diagnostic value of gamma-glutamyltransferase/aspartate aminotransferase ratio, protein induced by Vitamin K absence or antagonist II, and alpha-fetoprotein in hepatitis B virus-related hepatocellular carcinoma. World J. Gastroenterol. 2019, 25, 5515–5529. [Google Scholar] [CrossRef]
- Li, T.; Li, H.; Wang, A.; Su, X.; Zhao, J.; Cui, Y.; Liu, J.; Hu, J. Development and validation of a simple model for detection of early hepatocellular carcinoma in a liver cirrhosis cohort. Cancer Manag. Res. 2019, 11, 9379–9386. [Google Scholar] [CrossRef]
- Feng, H.; Li, B.; Li, Z.; Wei, Q.; Ren, L. PIVKA-II serves as a potential biomarker that complements AFP for the diagnosis of hepatocellular carcinoma. BMC Cancer 2021, 21, 401. [Google Scholar] [CrossRef]
- Nguyen, H.B.; Le, X.T.T.; Nguyen, H.H.; Vo, T.T.; Le, M.K.; Nguyen, N.T.; Do-Nguyen, T.M.; Truong-Nguyen, C.M.; Nguyen, B.-S.T. Diagnostic Value of hTERT mRNA and in Combination With AFP, AFP-L3%, Des-γ-carboxyprothrombin for Screening of Hepatocellular Carcinoma in Liver Cirrhosis Patients HBV or HCV-Related. Cancer Inform. 2022, 21, 11769351221100730. [Google Scholar] [CrossRef] [PubMed]
- Lee, Q.; Yu, X.; Yu, W. The value of PIVKA-II versus AFP for the diagnosis and detection of postoperative changes in hepatocellular carcinoma. J. Interv. Med. 2021, 4, 77–81. [Google Scholar] [PubMed]
- Chen, J.; Tang, D.; Xu, C.; Niu, Z.; Li, H.; Li, Y.; Zhang, P. Evaluation of Serum GDF15, AFP, and PIVKA-II as Diagnostic Markers for HBV-Associated Hepatocellular Carcinoma. Lab. Med. 2021, 52, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Zhang, L.; He, W.; Song, D.; Ji, X.; Shao, J. The diagnostic value of serum PIVKA-II alone or in combination with AFP in Chinese hepatocellular carcinoma patients. Dis. Markers 2021, 2021, 8868370. [Google Scholar] [CrossRef]
- Peng, F.; Yuan, H.; Zhou, Y.F.; Wu, S.X.; Long, Z.Y.; Peng, Y.M. Diagnostic Value of Combined Detection via Protein Induced by Vitamin K Absence or Antagonist II, Alpha-Fetoprotein, and D-Dimer in Hepatitis B Virus-Related Hepatocellular Carcinoma. Int. J. Gen. Med. 2022, 15, 5763–5773. [Google Scholar] [CrossRef] [PubMed]
- Hadi, H.; Shuaib, W.; Ali, R.; Othman, H. Utility of PIVKA-II and AFP in Differentiating Hepatocellular Carcinoma from Non-malignant High-risk patients. Medicina 2022, 58, 1015. [Google Scholar] [CrossRef]
- Xing, H.; Yan, C.; Cheng, L.; Wang, N.; Dai, S.; Yuan, J.; Lu, W.; Wang, Z.; Han, J.; Zheng, Y.; et al. Clinical application of protein induced by vitamin K antagonist-II as a biomarker in hepatocellular carcinoma. Tumor. Biol. 2016, 37, 15447–15456. [Google Scholar] [CrossRef]
- Xing, H.; Zheng, Y.-J.; Han, J.; Zhang, H.; Li, Z.-L.; Lau, W.-Y.; Shen, F.; Yang, T. Protein induced by vitamin K absence or antagonist-II versus alpha-fetoprotein in the diagnosis of hepatocellular carcinoma: A systematic review with meta-analysis. Hepatobiliary Pancreat. Dis. Int. 2018, 17, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Tian, X.; Sun, J.; Meng, X. Evaluation of individual and combined applications of serum biomarkers for diagnosis of Hepatocellular carcinoma: A meta-analysis. Int. J. Mol. Sci. 2013, 14, 23559–23580. [Google Scholar] [CrossRef]
- Chen, J.; Wu, G.; Li, Y. Evaluation of serum des-gamma-carboxy prothrombin for the diagnosis of hepatitis B virus-related hepatocellular carcinoma: A meta-analysis. Dis. Markers 2018, 2018, 8906023. [Google Scholar] [CrossRef]
- Saitta, C.; Raffa, G.; Alibrandi, A.; Brancatelli, S.; Lombardo, D.; Tripodi, G.; Raimondo, G.; Pollicino, T. PIVKA-II is a useful tool for diagnostic characterization of ultrasound-detected liver nodules in cirrhotic patients. Medicine 2017, 96, e7266. [Google Scholar] [CrossRef]
- Burch, J.; Tort, S. For adults with chronic liver disease, how accurate are abdominal ultrasound and/or alpha—fetoprotein testing for diagnosing hepatocellular carcinoma? Cochrane. Libr. 2022, 4, CD013346. [Google Scholar] [CrossRef]
- Ludovico, A.; Luigi, B. New serum markers for detection of early hepatocellular carcinoma. Panminerva Med. 2017, 59, 281–282. [Google Scholar]
- Nomura, F.; Ishijima, M.; Kuwa, K.; Tanaka, N.; Nakai, T.; Ohnishi, K. Serum des-gamma-carboxy prothrombin levels determined by a new generation of sensitive immunoassays in patients with small-sized hepatocellular carcinoma. Am. J. Gastroenterol. 1999, 94, 650–654. [Google Scholar] [CrossRef]
- Chen, H.; Chen, S.; Li, S.; Chen, Z.; Zhu, X.; Dai, M.; Kong, L.; Lv, X.; Huang, Z.; Qin, X. Combining des-gamma-carboxyprothrombin and alpha-fetoprotein for hepatocellular carcinoma diagnosing: An update meta-analysis and validation study. Oncotarget 2017, 8, 90390–90401. [Google Scholar] [CrossRef]
- Fang, Y.-S.; Wu, Q.; Zhao, H.-C.; Zhou, Y.; Ye, L.; Liu, S.-S.; Li, X.-X.; Du, W.-D. Do combined assays of serum AFP, AFP-L3, DCP, GP73, and DKK-1 efficiently improve the clinical values of biomarkers in decision-making for hepatocellular carcinoma? A meta-analysis. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 1065–1076. [Google Scholar] [CrossRef] [PubMed]
- De, J.; Shen, Y.; Qin, J.; Feng, L.; Wang, Y.; Yang, L. A Systematic Review of Des-γ-Carboxy Prothrombin for the Diagnosis of Primary Hepatocellular Carcinoma. Medicine 2016, 95, e3448. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Li, Y.; Li, Z.; Li, N. Clinical utility of decarboxylation prothrombin combined with α-fetoprotein for diagnosing primary hepatocellular carcinoma. Biosci. Rep. 2018, 38, BSR20180044. [Google Scholar] [CrossRef]

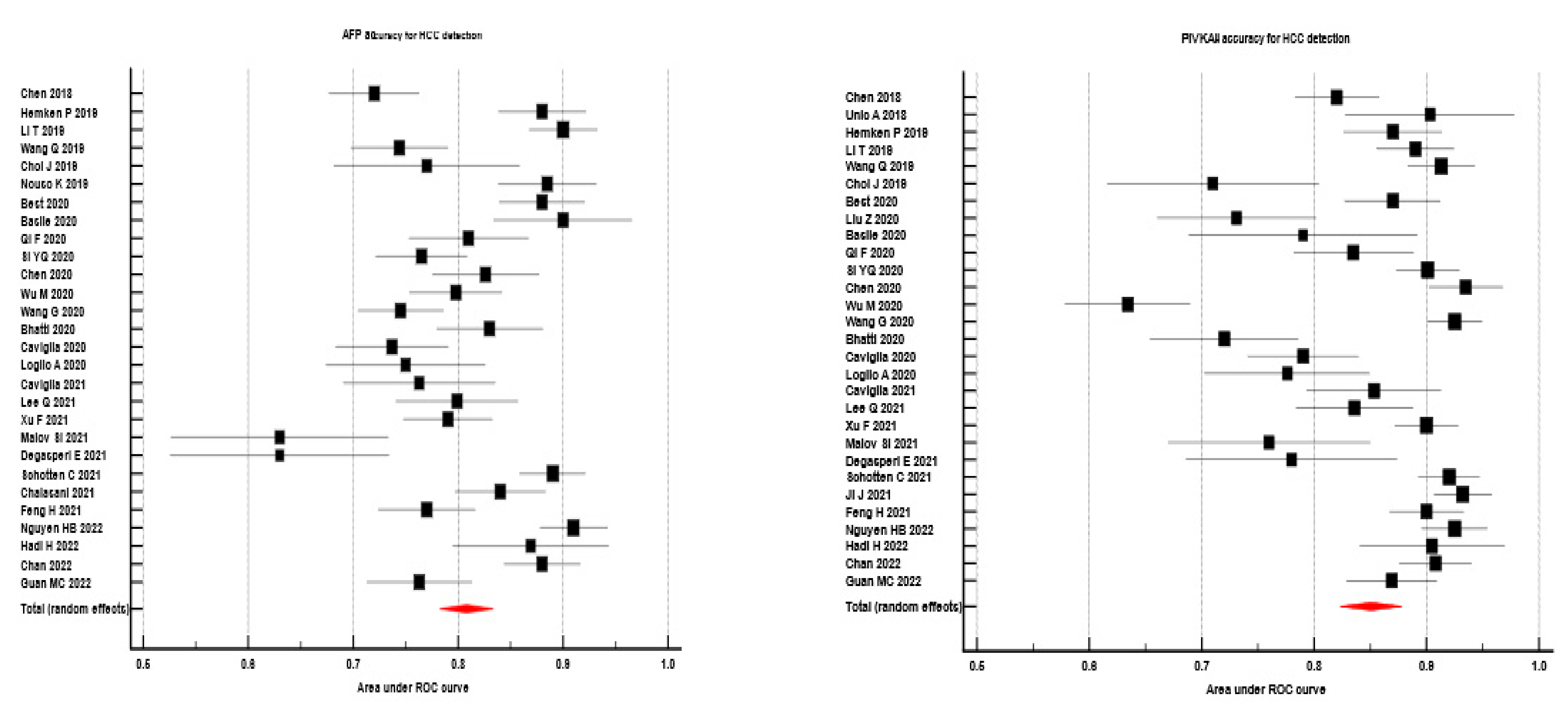
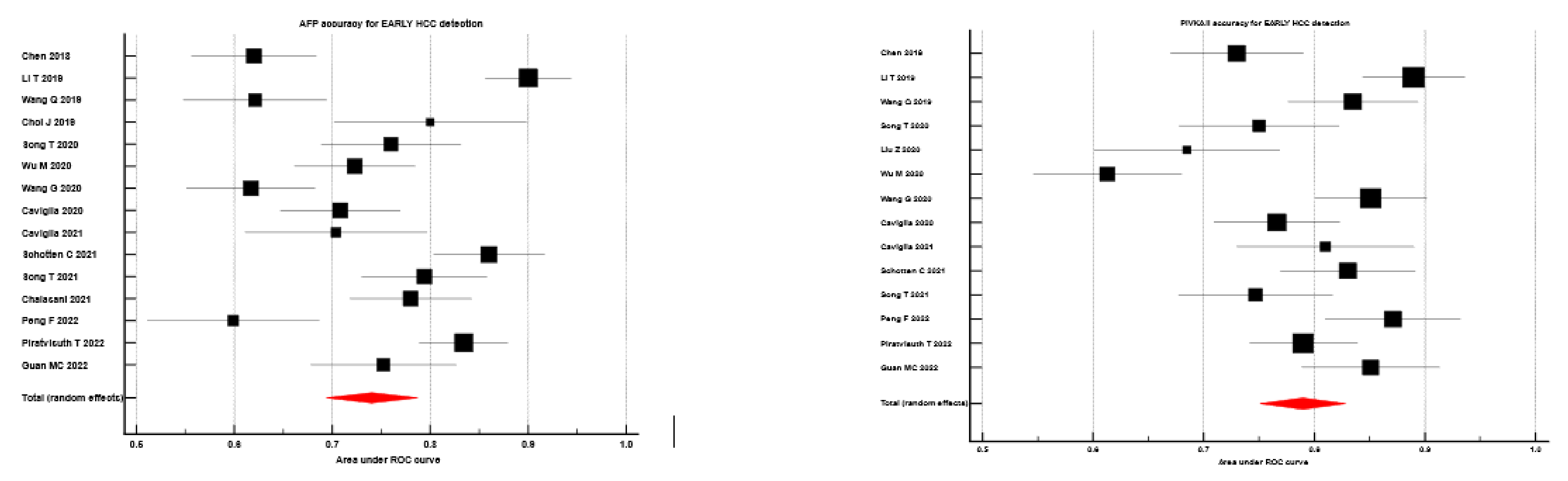
| Study | Period | Country | Study-Type | Patients | No | Cut-Off PIVKA II | Cut-Off AFP |
|---|---|---|---|---|---|---|---|
| Schotten C 2021 [53] | 2008–2020 | Germany | Retrospective study | 182 patients with HBV, 223 with HCV, 168 with other etiology, HCC—52 HBV, 84 HCV and 60 | 573 | NA | 20 ng/mL |
| Choi J 2019 [54] | NA | Korea | Matched case-control | 42 HCC; 168 cirrhosis or chronic B hepatitis | 210 | 20 mAU/mL | 5 ng/mL |
| Best 2020 [55] | 2005–2016 | Germany Japan | Multicenter case-control study | 126 patients with HCC; 231 patients without HCC, NASH controls | 357 | NA | NA |
| Wu M 2020 [56] | NA | China | Observational study | 176 healthy, CHB, LC; 198 very early HCC + early HCC + advance and HCC | 374 | NA | NA |
| Malov SI 2021 [57] | NA | Russia | Case-control study | 110 patients with chronic hepatitis C in the stage of liver cirrhosis 55 without HCC; 55 with HCC | 110 | 20 ng/mL | 20 ng/mL |
| Chan 2022 [58] | NA | China Germany Japan Thailand. | Multicenter prospective study | 168 HCC, 208 patients without HCC with an at-risk condition—cirrhosis, non-cirrhotic chronic hepatitis B virus (HBV), non-cirrhotic chronic hepatitis C virus (HCV), NASH | 376 | 28.4 ng/mL | 20 ng/mL |
| Chalasani 2021 [59] | NA | ClinicalTrials.gov | International, multicenter, case-control study | 136 HCC, 404 controls at-risk patients with chronic liver disease—HCV, NAFLD, ASH, HBV, other chronic liver disease | 540 | NA | 20 ng/mL |
| Basile 2020 [12] | NA | Italy | Case-control study | 20 metabolic, 40 viral newly diagnosed HCC, 20 healthy subjects | 80 | 38 mAU/mL, | 3.5 ng/mL |
| Nouso K 2019 [60] | 2001–2016 | Japan | Case-control study | 172 tumor-free diabetes mellitus, 93 consecutive NBNC-HCC patients | 265 | 20 mAU/mL | 3 ng/mL |
| Hemken P 2019 [61] | 2003–2016 | SUA | Retrospective case-control study | 119 HCC, 215 nonmalignant liver disease, 34 healthy | 368 | NA | NA |
| Unic A 2018 [62] | 2009–2011 | Croatia | Consecutively recruited study | 20 healthy volunteers, 31 patients with alcoholic liver cirrhosis, 32 patients with HCC. | 83 | 108 mAU/mL | NA |
| Liu Z 2020 [63] | 2010–2018 | China | Retrospective study | 87 AFP-negative HBV-related HCC, 123 control cases—benign liver disease, chronic HBV infection or liver cirrhosis | 210 | 45 mAU/mL | NA |
| Song T 2021 [64] | 2010–2020 | China | Cross-sectional study | 48 chronic HBV infection (CHB), 64 liver cirrhosis (LC), 33 early-stage CHB-HCC, 55 early-stage LC-HCC. | 200 | 44 mAU/mL | 5 ng/mL |
| Loglio A 2020 [65] | 2010–2020 | Italy | Cross-sectional, case-control study | 64 with HCC (cases), 148 HCC-free (control) | 212 | 48 mAU/mL | 4.2 ng/mL |
| Caviglia 2020 [1] | 2012–2018 | Italy | Cross-sectional study | 149 HCC, 200 cirrhosis of viral etiology | 349 | 73 mAU/mL | 9.7 ng/mL |
| Caviglia 2021 [21] | 2012–2020 | Italy | Retrospective case-control study | 191 NAFLD patients cohort, 72 of whom had a diagnosis of HCC, 119 non-HCC patients | 191 | 56 mAU/mL | 4.4 ng/mL |
| Chen H 2018 [66] | 2013–2014 | China | Cross-sectional, consecutively recruited study | 202 HCC patients, 226 liver cirrhosis patients, 215 chronic hepatitis B virus-infected 203 healthy | 846 | NA | NA |
| Piratvisuth T 2022 [67] | 2014–2016 | China Germany Spain Thailand | Case-control study | 308 HCC, 740 chronic liver disease—cirrhotic liver disease independent of etiology, noncirrhotic NASH, chronic HBV infection, chronic HCV infection | 1048 | NA | NA |
| Song T 2020 [68] | 2014–2017 | China | Prospective study | 100 HCC in patients with hepatitis B virus (HBV)—associated liver cirrhosis (LC), 67 LC | 167 | 38 mAU/mL | 10 ng/mL |
| Degasperi E 2021 [69] | 2014–2019 | Italy | Retrospective study | 34 HCC, 366 non-HCC patients | 400 | 47 mAU/mL | 17 ng/mL |
| Wu J 2018 [70] | 2016–2017 | China | Case-control study | 51 healthy, 37 chronic hepatitis, 43 cirrhotic; 143 HCC | 274 | 40 mAU/mL | 10 ng/mL |
| Qi F 2020 [71] | 2016–2018 | China | Prospective study | 120 HCC, 89 chronic liver disease—nonviral, autoimmune, fatty-liver, HBV, HCV | 209 | 33.08 mAU/mL | 11.88 ng/mL |
| Li Y 2019 [72] | 2016–2018 | China | Retrospective study | Group 1 non-cancer, Group 2 primary cancer in liver patients—not available the numbers | 1190 | 60.5 mAU/mL | NA |
| Guan MC 2022 [73] | 2016–2020 | China | Retrospective observational study | 139 HCC, 345 NAFLD | 484 | 40 mAU/mL | 20 ng/mL |
| Si YQ 2020 [74] | 2017–2018 | China | Case-control study | 266 cases with HBV-related HCC, 87 HBV DNA-positive benign liver disease, 80 healthy individuals | 433 | 41.74 mAU/mL | 21.8 ng/mL |
| Ji J 2021 [75] | 2017–2018 | China | Cross-sectional, multicenter study | 183 HCC-CHB- and HBV-related, 312 cases were chronic hepatitis and 289 cases were cirrhosis | 784 | 40 mAU/mL | 20 ng/mL |
| Wang G 2020 [76] | 2017–2018 | China | Retrospective study | 234 HBV-related HCC, 396 patients with chronic hepatitis B (CHB) | 630 | 87.63 mAU/mL | 499.80 ng/mL |
| Bhatti 2020 [6] | 2017–2019 | Pakistan | Retrospective study | Cirrhotic patients, surgical candidates—176 HCC, 68 non-HCC | 244 | 250 mAU/mL | 7.6 ng/mL |
| Wang Q 2019 [77] | 2017–2019 | China | Retrospective study | 176 HBV-related HCC, 359 patients with chronic hepatitis B. | 535 | 162.22 mAU/mL | 145.65 ng/mL |
| Li T 2019 [78] | 2017–2019 | China | Case-control study. | 169 newly diagnosed early HCC, 242 LC without HCC | 411 | NA | NA |
| Feng H 2021 [79] | 2017–2019 | China | Case-control study. | 168 HCC patients,150 benign liver disease, 153 healthy controls | 469 | 35.60 mAU/mL | 17.76 ng/mL |
| Nguyen HB 2022 [80] | 2018–2019 | Vietnam | Case-control study. | 170 chronic hepatitis B virus, hepatitis C virus, 170 HCC | 340 | 29.01 mAU/mL | 5.1 ng/mL |
| Lee Q 2021 [81] | 2018–2020 | China | Prospective study | 158 primary HCC in chronic hepatitis B, 62—chronic hepatitis B | 220 | 34.92 mAU/mL | 9.10 ng/mL |
| Chen J 2020 [82] | 2019 | China | Case-control study | 110 patients HBV-associated HCC, 70 HBV-related LC, 70 CBH, 110 healthy | 360 | 51.00 mAU/mL | 5.65 ng/mL |
| Xu F 2021 [83] | 2019 | China | Retrospective study | 308 HCC, 60 HBV-related LC, 60 benign liver disease | 428 | 40 mAU/mL | 25 ng/mL |
| Peng F 2022 [84] | 2020–2021 | China | Prospective study | 143 LC, 148 hepatitis B virus (HBV)-related hepatocellular carcinoma | 291 | NA | NA |
| Hadi H 2022 [85] | 2021–2022 | Malaysia | Cross-sectional study | HCC—in relationship with 26 HBV, 12 NASH, 2 HCV, 123 nonmalignant high-risk liver cirrhosis | 163 | 36.7 mAU/mL | 14.2 ng/mL |
| Study | AUC PIVKA II | AUC AFP | Se PIVKA II % | Sp PIVKA II % | Se AFP % | Sp AFP % |
|---|---|---|---|---|---|---|
| Schotten C 2021 [53] | 0.920 | 0.890 | - | - | - | - |
| Choi J 2019 [54] | 0.710 | 0.770 | 48.00 | 86.00 | 62.00 | 87.00 |
| Best 2020 [55] | 0.870 | 0.880 | - | - | - | - |
| Wu M 2020 [56] | 0.634 | 0.798 | 29.80 | 97.20 | 77.30 | 71.10 |
| Malov SI 2021 [57] | 0.760 | 0.630 | 54.60 | 88.60 | 45.50 | 94.50 |
| Chan 2022 [58] | 0.908 | 0.880 | 86.90 | 83.70 | 51.80 | 98.10 |
| Chalasani 2021 [59] | - | 0.840 | - | - | 46.00 | 88.00 |
| Basile 2020 [12] | 0.790 | 0.900 | - | - | - | - |
| Nouso K 2019 [60] | - | 0.885 | - | - | 81.80 | 82.60 |
| Hemken P 2019 [61] | 0.870 | 0.880 | 86.00 | 72.00 | 86.00 | 77.00 |
| Unic A 2018 [62] | 0.903 | - | 81.25 | 96.77 | - | - |
| Liu Z 2020 [63] | 0.731 | - | 50.60 | 94.30 | - | - |
| Loglio A 2020 [65] | 0.776 | 0.750 | 64.00 | 91.00 | 56.00 | 94.00 |
| Caviglia 2020 [1] | 0.790 | 0.737 | 68.00 | 84.00 | 72.00 | 66.00 |
| Caviglia 2021 [21] | 0.853 | 0.763 | 75.00 | 85.70 | 76.40 | 68.90 |
| Chen 2018 [66] | 0.820 | 0.720 | 65.20 | 90.00 | 43.70 | 90.00 |
| Degasperi E 2021 [69] | 0.780 | 0.630 | 76.00 | 79.00 | 29.00 | 97.00 |
| Qi F 2020 [71] | 0.835 | 0.810 | 83.50 | 71.60 | 73.60 | 80.70 |
| Guan MC 2022 [73] | 0.869 | 0.763 | 74.80 | 91.00 | 52.50 | 97.40 |
| Si YQ 2020 [74] | 0.901 | 0.765 | 81.20 | 88.50 | 51.50 | 89.70 |
| Ji J 2021 [75] | 0.932 | - | 84.08 | 90.43 | 61.33 | 91.15 |
| Wang G 2020 [76] | 0.925 | 0.745 | 86.80 | 90.20 | 52.10 | 91.40 |
| Bhatti 2020 [6] | 0.720 | 0.830 | 72.00 | 60.00 | 77.00 | 77.00 |
| Wang Q 2019 [77] | 0.913 | 0.744 | 81.30 | 93.60 | 64.80 | 77.20 |
| Li T 2019 [78] | 0.890 | 0.900 | 84.20 | 82.00 | 85.30 | 85.60 |
| Feng H 2021 [79] | 0.900 | 0.770 | 83.93 | 91.50 | 64.29 | 90.20 |
| Nguyen HB 2022 [80] | 0.925 | 0.910 | 91.00 | 76.00 | 73.00 | 92.00 |
| Lee Q 2021 [81] | 0.836 | 0.799 | 68.4 | 98.40 | 57.60 | 93.50 |
| Chen 2020 [82] | 0.935 | 0.826 | 85.00 | 93.00 | 84.10 | 70.90 |
| Xu F 2021 [83] | 0.900 | 0.790 | 89.00 | 91.70 | 68.80 | 87.60 |
| Hadi H 2022 [85] | 0.905 | 0.869 | 90.00 | 82.10 | 75.00 | 93.50 |
| Study | No | AUC PIVKA II | AUC AFP | Se PIVKA II % | Sp PIVKA II % | Se AFP % | Sp AFP % |
|---|---|---|---|---|---|---|---|
| Schotten C 2021 [53] | 70 | 0.830 | 0.860 | - | - | - | - |
| Choi J 2019 [54] | 31 | - | 0.800 | - | - | - | - |
| Wu M 2020 [56] | 113 | 0.613 | 0.723 | 25.70 | 97.20 | 67.30 | 71.70 |
| Chalasani 2021 [59] | 81 | - | 0.780 | - | - | - | - |
| Liu Z 2020 [63] | 62 | 0.685 | - | 43.50 | 94.30 | - | - |
| Song T 2021 [64] | 88 | 0.747 | 0.794 | 55.70 | 88.40 | 65.90 | 88.40 |
| Caviglia 2020 [1] | 115 | 0.766 | 0.708 | 65.00 | 84.00 | 67.00 | 66.00 |
| Caviglia 2021 [21] | 47 | 0.810 | 0.704 | - | - | - | - |
| Chen 2018 [66] | 94 | 0.730 | 0.620 | 48.30 | 90.00 | 30.60 | 90.00 |
| Piratvisuth T 2022 [67] | 125 | 0.790 | 0.834 | 56.00 | - | 53.60 | - |
| Song T 2020 [68] | 100 | 0.750 | 0.760 | 60.00 | 84.70 | 51.50 | 92.50 |
| Guan MC 2022 [73] | 60 | 0.851 | 0.752 | 75.00 | 89.60 | 46.70 | 97.40 |
| Wang G 2020 [76] | 94 | 0.851 | 0.617 | 72.30 | 90.20 | 33.00 | 92.40 |
| Wang Q 2019 [77] | 74 | 0.835 | 0.621 | 68.90 | 89.70 | 47.30 | 29.95 |
| Li T 2019 [78] | 95 | 0.890 | 0.900 | 84.20 | 82.00 | 85.30 | 85.60 |
| Peng F 2022 [84] | 59 | 0.871 | 0.599 | 61.20 | 95.80 | 28.81 | 97.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perne, M.G.; Sitar-Tăut, A.-V.; Alexescu, T.G.; Ciumărnean, L.; Milaciu, M.-V.; Coste, S.-C.; Vlad, C.-V.; Cozma, A.; Sitar-Tăut, D.-A.; Orăşan, O.H.; et al. Diagnostic Performance of Extrahepatic Protein Induced by Vitamin K Absence in the Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. Diagnostics 2023, 13, 816. https://doi.org/10.3390/diagnostics13050816
Perne MG, Sitar-Tăut A-V, Alexescu TG, Ciumărnean L, Milaciu M-V, Coste S-C, Vlad C-V, Cozma A, Sitar-Tăut D-A, Orăşan OH, et al. Diagnostic Performance of Extrahepatic Protein Induced by Vitamin K Absence in the Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. Diagnostics. 2023; 13(5):816. https://doi.org/10.3390/diagnostics13050816
Chicago/Turabian StylePerne, Mirela Georgiana, Adela-Viviana Sitar-Tăut, Teodora Gabriela Alexescu, Lorena Ciumărnean, Mircea-Vasile Milaciu, Sorina-Cezara Coste, Calin-Vasile Vlad, Angela Cozma, Dan-Andrei Sitar-Tăut, Olga Hilda Orăşan, and et al. 2023. "Diagnostic Performance of Extrahepatic Protein Induced by Vitamin K Absence in the Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis" Diagnostics 13, no. 5: 816. https://doi.org/10.3390/diagnostics13050816
APA StylePerne, M. G., Sitar-Tăut, A.-V., Alexescu, T. G., Ciumărnean, L., Milaciu, M.-V., Coste, S.-C., Vlad, C.-V., Cozma, A., Sitar-Tăut, D.-A., Orăşan, O. H., & Crăciun, A. (2023). Diagnostic Performance of Extrahepatic Protein Induced by Vitamin K Absence in the Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. Diagnostics, 13(5), 816. https://doi.org/10.3390/diagnostics13050816








