Clinical Value of Novel Echocardiographic Biomarkers Assessing Myocardial Work in Acute Heart Failure—Rationale and Design of the “Beyond Myo-HF Study”
Abstract
:1. Introduction
2. Methods and Materials
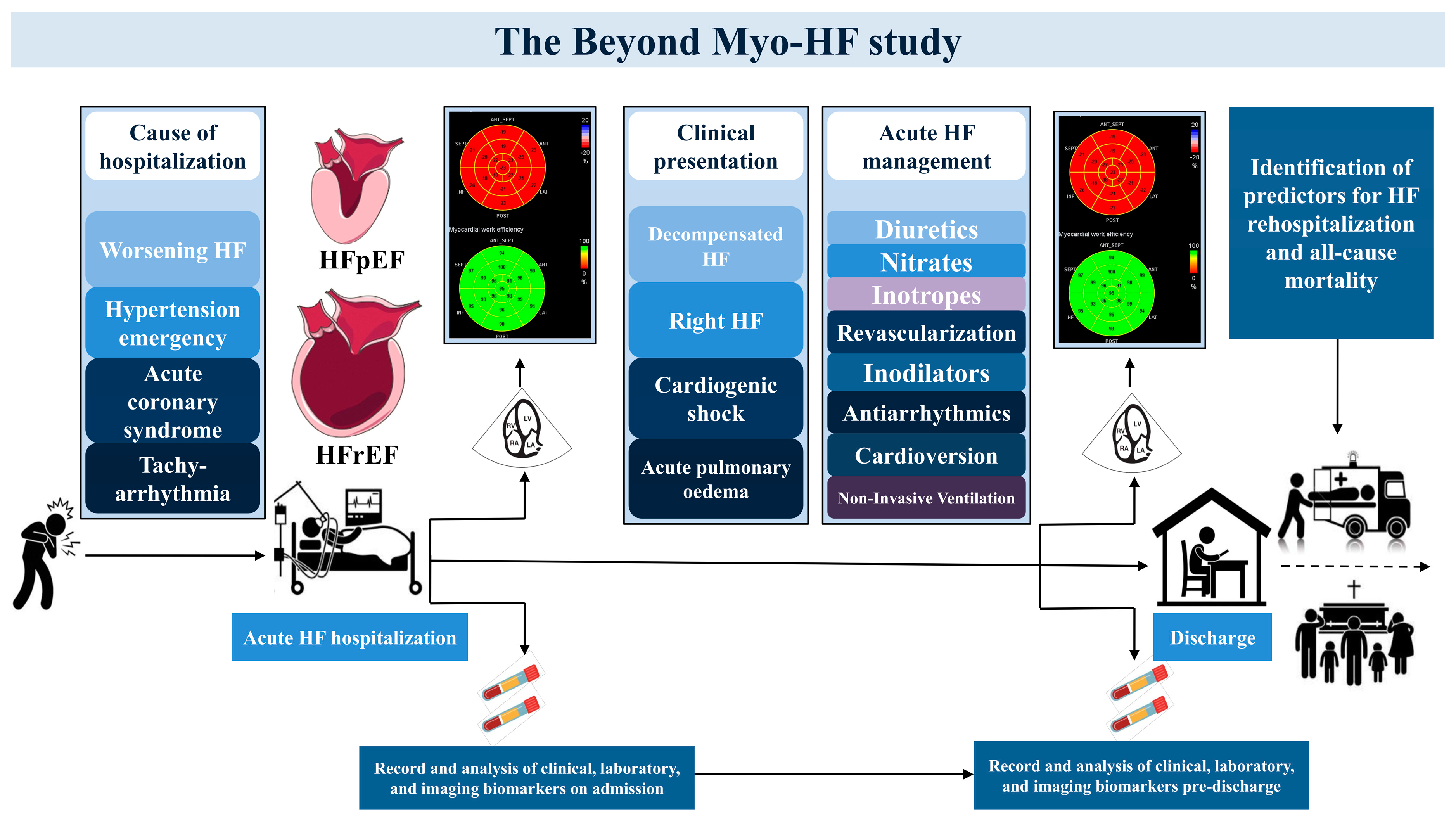
Study Design and Population
3. Materials and Equipment
3.1. Echocardiographic Analysis
3.2. Chamber Quantification Analysis
3.3. Strain Analysis
3.4. Left Ventricular Myocardial Work Analysis
3.5. Right Ventricular Myocardial Work Analysis
3.6. Statistical Analysis
4. Discussion and Expected Results
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| GLS | global longitudinal strain |
| GCW | global constructive work |
| GWE | global work efficiency |
| GWI | global work index |
| GWW | global wasted work |
| HF | heart failure |
| LV | left ventricle |
| LVEF | left ventricular ejection fraction |
| LVMW | left ventricular myocardial work |
| RV | right ventricle |
| RVMW | right ventricular myocardial work |
| RV-PA | right ventricle–pulmonary artery |
| PADP | pulmonary artry diastolic pressure |
| PAMP | pulmonary artery mean pressure |
| PASP | pulmonary artery systolic pressure |
| TAPSE | tricuspid annular plane systolic excursion |
| TTE | transthoracic echocardiographic assessment |
References
- Fonarow, G.C.; Stough, W.G.; Abraham, W.T.; Albert, N.M.; Gheorghiade, M.; Greenberg, B.H.; O’Connor, C.M.; Sun, J.L.; Yancy, C.W.; Young, J.B. Characteristics, treatments, and outcomes of patients with preserved systolic function hospitalized for heart failure: A report from the OPTIMIZE-HF Registry. J. Am. Coll. Cardiol. 2007, 50, 768–777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gheorghiade, M.; Pang, P.S. Acute heart failure syndromes. J. Am. Coll. Cardiol. 2009, 53, 557–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akiyama, E.; Cinotti, R.; Čerlinskaitė, K.; Van Aelst, L.N.L.; Arrigo, M.; Placido, R.; Chouihed, T.; Girerd, N.; Zannad, F.; Rossignol, P.; et al. Improved cardiac and venous pressures during hospital stay in patients with acute heart failure: An echocardiography and biomarkers study. ESC Heart Fail. 2020, 7, 996–1006. [Google Scholar] [CrossRef] [Green Version]
- Hullin, R.; Tzimas, G.; Barras, N.; Abdurashidova, T.; Soborun, N.; Aur, S.; Regamey, J.; Hugelshofer, S.; Lu, H.; Crisinel, V.; et al. Decongestion improving right heart function ameliorates prognosis after an acute heart failure episode. ESC Heart Fail. 2022, 9, 3814–3824. [Google Scholar] [CrossRef] [PubMed]
- Palazzuoli, A.; Ruocco, G.; Evangelista, I.; De Vivo, O.; Nuti, R.; Ghio, S. Prognostic Significance of an Early Echocardiographic Evaluation of Right Ventricular Dimension and Function in Acute Heart Failure. J. Card. Fail. 2020, 26, 813–820. [Google Scholar] [CrossRef]
- Verhaert, D.; Mullens, W.; Borowski, A.; Popović, Z.B.; Curtin, R.J.; Thomas, J.D.; Tang, W.H. Right ventricular response to intensive medical therapy in advanced decompensated heart failure. Circ. Heart Fail. 2010, 3, 340–346. [Google Scholar] [CrossRef] [Green Version]
- Russell, K.; Eriksen, M.; Aaberge, L.; Wilhelmsen, N.; Skulstad, H.; Remme, E.W.; Haugaa, K.H.; Opdahl, A.; Fjeld, J.G.; Gjesdal, O.; et al. A novel clinical method for quantification of regional left ventricular pressure-strain loop area: A non-invasive index of myocardial work. Eur. Heart J. 2012, 33, 724–733. [Google Scholar] [CrossRef] [Green Version]
- Butcher, S.C.; Fortuni, F.; Montero-Cabezas, J.M.; Abou, R.; El Mahdiui, M.; van der Bijl, P.; van der Velde, E.T.; Ajmone Marsan, N.; Bax, J.J.; Delgado, V. Right ventricular myocardial work: Proof-of-concept for non-invasive assessment of right ventricular function. Eur. Heart J. Cardiovasc. Imaging 2021, 22, 142–152. [Google Scholar] [CrossRef]
- Du Bois, D.; Du Bois, E.F. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition 1989, 5, 303–311, discussion 312–313. [Google Scholar]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Bohm, M.; Burri, H.; Butler, J.; Celutkiene, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 233–270. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F., 3rd; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 1321–1360. [Google Scholar] [CrossRef] [PubMed]
- Rudski, L.G.; Lai, W.W.; Afilalo, J.; Hua, L.; Handschumacher, M.D.; Chandrasekaran, K.; Solomon, S.D.; Louie, E.K.; Schiller, N.B. Guidelines for the echocardiographic assessment of the right heart in adults: A report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J. Am. Soc. Echocardiogr. 2010, 23, 685–713, quiz 786–788. [Google Scholar] [PubMed]
- Lancellotti, P.; Tribouilloy, C.; Hagendorff, A.; Moura, L.; Popescu, B.A.; Agricola, E.; Monin, J.L.; Pierard, L.A.; Badano, L.; Zamorano, J.L.; et al. European Association of Echocardiography recommendations for the assessment of valvular regurgitation. Part 1: Aortic and pulmonary regurgitation (native valve disease). Eur. J. Echocardiogr. 2010, 11, 223–244. [Google Scholar] [CrossRef] [Green Version]
- Lancellotti, P.; Moura, L.; Pierard, L.A.; Agricola, E.; Popescu, B.A.; Tribouilloy, C.; Hagendorff, A.; Monin, J.L.; Badano, L.; Zamorano, J.L.; et al. European Association of Echocardiography recommendations for the assessment of valvular regurgitation. Part 2: Mitral and tricuspid regurgitation (native valve disease). Eur. J. Echocardiogr. 2010, 11, 307–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feldman, T.; Kar, S.; Elmariah, S.; Smart, S.C.; Trento, A.; Siegel, R.J.; Apruzzese, P.; Fail, P.; Rinaldi, M.J.; Smalling, R.W.; et al. Randomized Comparison of Percutaneous Repair and Surgery for Mitral Regurgitation: 5-Year Results of EVEREST II. J. Am. Coll. Cardiol. 2015, 66, 2844–2854. [Google Scholar] [CrossRef] [Green Version]
- Aduen, J.F.; Castello, R.; Lozano, M.M.; Hepler, G.N.; Keller, C.A.; Alvarez, F.; Safford, R.E.; Crook, J.E.; Heckman, M.G.; Burger, C.D. An alternative echocardiographic method to estimate mean pulmonary artery pressure: Diagnostic and clinical implications. J. Am. Soc. Echocardiogr. 2009, 22, 814–819. [Google Scholar] [CrossRef]
- Namazi, F.; van der Bijl, P.; Hirasawa, K.; Kamperidis, V.; van Wijngaarden, S.E.; Mertens, B.; Leon, M.B.; Hahn, R.T.; Stone, G.W.; Narula, J.; et al. Prognostic Value of Left Ventricular Global Longitudinal Strain in Patients With Secondary Mitral Regurgitation. J. Am. Coll. Cardiol. 2020, 75, 750–758. [Google Scholar] [CrossRef]
- Kamperidis, V.; Marsan, N.A.; Delgado, V.; Bax, J.J. Left ventricular systolic function assessment in secondary mitral regurgitation: Left ventricular ejection fraction vs. speckle tracking global longitudinal strain. Eur. Heart J. 2016, 37, 811–816. [Google Scholar] [CrossRef]
- Kamperidis, V.; Joyce, E.; Debonnaire, P.; Katsanos, S.; van Rosendael, P.J.; van der Kley, F.; Sianos, G.; Bax, J.J.; Ajmone Marsan, N.; Delgado, V. Left ventricular functional recovery and remodeling in low-flow low-gradient severe aortic stenosis after transcatheter aortic valve implantation. J. Am. Soc. Echocardiogr. 2014, 27, 817–825. [Google Scholar] [CrossRef]
- Wang, C.L.; Chan, Y.H.; Wu, V.C.; Lee, H.F.; Hsiao, F.C.; Chu, P.H. Incremental prognostic value of global myocardial work over ejection fraction and global longitudinal strain in patients with heart failure and reduced ejection fraction. Eur. Heart J. Cardiovasc. Imaging 2021, 22, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Hedwig, F.; Nemchyna, O.; Stein, J.; Knosalla, C.; Merke, N.; Knebel, F.; Hagendorff, A.; Schoenrath, F.; Falk, V.; Knierim, J. Myocardial Work Assessment for the Prediction of Prognosis in Advanced Heart Failure. Front. Cardiovasc. Med. 2021, 8, 691611. [Google Scholar] [CrossRef] [PubMed]
- Sahiti, F.; Morbach, C.; Henneges, C.; Stefenelli, U.; Scholz, N.; Cejka, V.; Albert, J.; Heuschmann, P.U.; Ertl, G.; Frantz, S.; et al. Dynamics of Left Ventricular Myocardial Work in Patients Hospitalized for Acute Heart Failure. J. Card. Fail. 2021, 27, 1393–1403. [Google Scholar] [CrossRef]
- Sanz, J.; Sanchez-Quintana, D.; Bossone, E.; Bogaard, H.J.; Naeije, R. Anatomy, Function, and Dysfunction of the Right Ventricle: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 1463–1482. [Google Scholar] [CrossRef] [PubMed]
- Hahn, R.T. State-of-the-Art Review of Echocardiographic Imaging in the Evaluation and Treatment of Functional Tricuspid Regurgitation. Circ. Cardiovasc. Imaging 2016, 9, e005332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hahn, R.T.; Asch, F.; Weissman, N.J.; Grayburn, P.; Kar, S.; Lim, S.; Ben-Yehuda, O.; Shahim, B.; Chen, S.; Liu, M.; et al. Impact of Tricuspid Regurgitation on Clinical Outcomes: The COAPT Trial. J. Am. Coll. Cardiol. 2020, 76, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anastasiou, V.; Daios, S.; Moysidis, D.V.; Bazmpani, M.-A.; Zegkos, T.; Karamitsos, T.; Makedou, K.; Savopoulos, C.; Efthimiadis, G.; Ziakas, A.; et al. Clinical Value of Novel Echocardiographic Biomarkers Assessing Myocardial Work in Acute Heart Failure—Rationale and Design of the “Beyond Myo-HF Study”. Diagnostics 2023, 13, 1191. https://doi.org/10.3390/diagnostics13061191
Anastasiou V, Daios S, Moysidis DV, Bazmpani M-A, Zegkos T, Karamitsos T, Makedou K, Savopoulos C, Efthimiadis G, Ziakas A, et al. Clinical Value of Novel Echocardiographic Biomarkers Assessing Myocardial Work in Acute Heart Failure—Rationale and Design of the “Beyond Myo-HF Study”. Diagnostics. 2023; 13(6):1191. https://doi.org/10.3390/diagnostics13061191
Chicago/Turabian StyleAnastasiou, Vasileios, Stylianos Daios, Dimitrios V. Moysidis, Maria-Anna Bazmpani, Thomas Zegkos, Theodoros Karamitsos, Kali Makedou, Christos Savopoulos, Georgios Efthimiadis, Antonios Ziakas, and et al. 2023. "Clinical Value of Novel Echocardiographic Biomarkers Assessing Myocardial Work in Acute Heart Failure—Rationale and Design of the “Beyond Myo-HF Study”" Diagnostics 13, no. 6: 1191. https://doi.org/10.3390/diagnostics13061191
APA StyleAnastasiou, V., Daios, S., Moysidis, D. V., Bazmpani, M.-A., Zegkos, T., Karamitsos, T., Makedou, K., Savopoulos, C., Efthimiadis, G., Ziakas, A., & Kamperidis, V. (2023). Clinical Value of Novel Echocardiographic Biomarkers Assessing Myocardial Work in Acute Heart Failure—Rationale and Design of the “Beyond Myo-HF Study”. Diagnostics, 13(6), 1191. https://doi.org/10.3390/diagnostics13061191








