Imaging in Gastric Cancer: Current Practice and Future Perspectives
Abstract
:1. Introduction
2. Epidemiology, Risk Factors and Pathological Classification Systems
3. Diagnostic Techniques
3.1. Endoscopic Ultrasonography (EUS)
T and N Staging
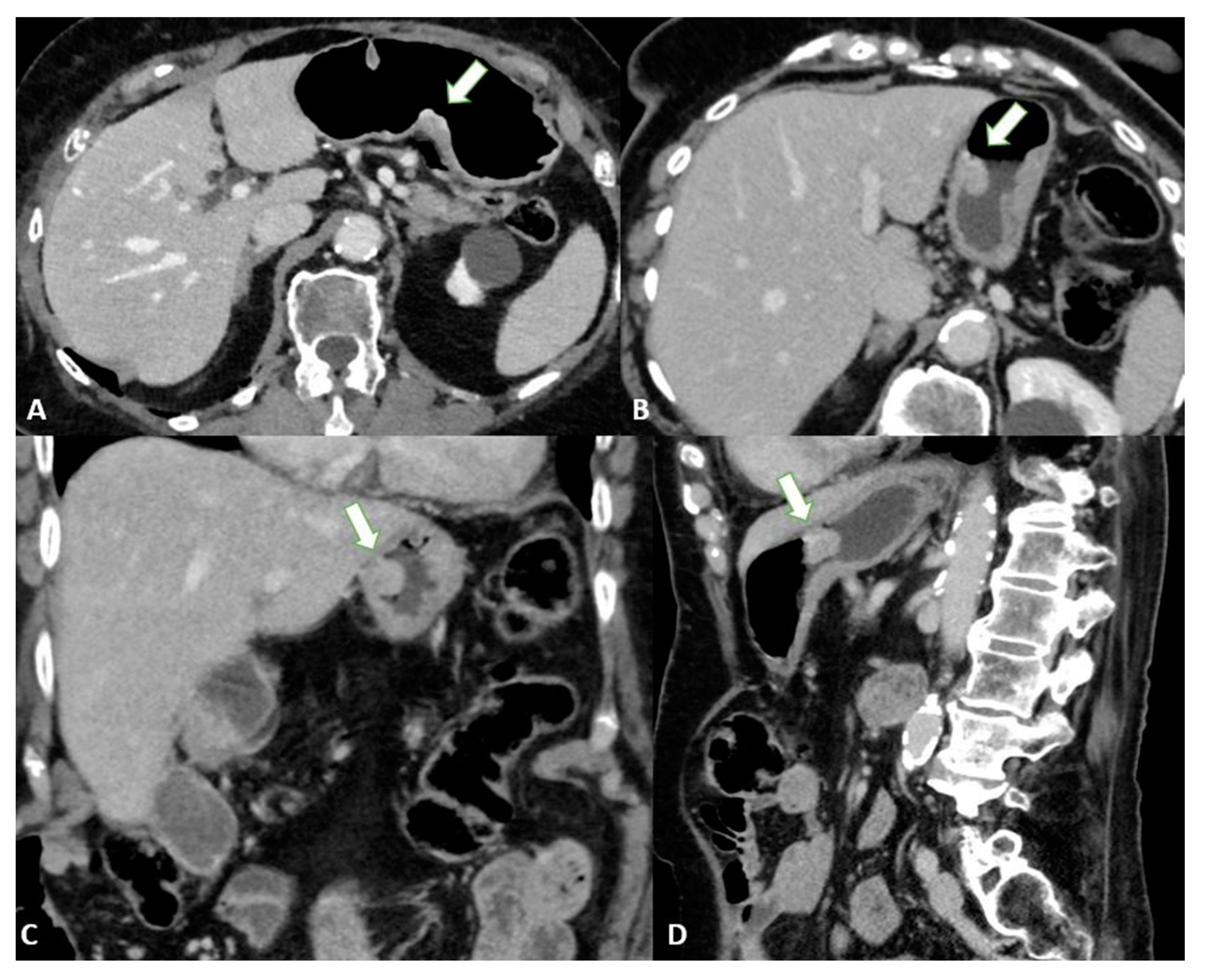
3.2. Computed Tomography (CT)
3.2.1. T and N Staging
3.2.2. M Staging
3.3. Magnetic Resonance Imaging (MRI)
TNM Staging
3.4. Comparison between Techniques
3.5. Positron Emission Tomography (PET)
4. New Frontiers
4.1. CT Volumetry
4.2. Perfusion CT (pCT)
4.3. Radiomics and Artifical Intelligence (AI)
5. Future Directions
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ajani, J.A.; D’Amico, T.A.; Bentrem, D.J.; Chao, J.; Cooke, D.; Corvera, C.; Das, P.; Enzinger, P.C.; Enzler, T.; Fanta, P.; et al. Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2022, 20, 167–192. [Google Scholar] [CrossRef]
- Smyth, E.C.; Nilsson, M.; Grabsch, H.I.; van Grieken, N.C.; Lordick, F. Gastric Cancer. Lancet 2020, 396, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Tsugane, S.; Sasazuki, S. Diet and the Risk of Gastric Cancer: Review of Epidemiological Evidence. Gastric Cancer 2007, 10, 75–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, C.; Pinheiro, H.; Figueiredo, J.; Seruca, R.; Carneiro, F. Familial Gastric Cancer: Genetic Susceptibility, Pathology, and Implications for Management. Lancet Oncol. 2015, 16, e60–e70. [Google Scholar] [CrossRef]
- Saragoni, L. Upgrading the Definition of Early Gastric Cancer: Better Staging Means More Appropriate Treatment. Cancer Biol. Med. 2015, 12, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Compton, C.C.; Greene, F.L. The Staging of Colorectal Cancer: 2004 and Beyond. CA A Cancer J. Clin. 2004, 54, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F. Surgical Treatment and Prognosis of Gastric Cancer in 2613 Patients. World J. Gastroenterol. 2004, 10, 3405–3408. [Google Scholar] [CrossRef]
- Young, J.J.; Pahwa, A.; Patel, M.; Jude, C.M.; Nguyen, M.; Deshmukh, M.; Huang, L.; Mohammad, S.F. Ligaments and Lymphatic Pathways in Gastric Adenocarcinoma. RadioGraphics 2019, 39, 668–689. [Google Scholar] [CrossRef]
- Fusaroli, P.; Caletti, G. Endoscopic Ultrasonography: Current Clinical Role. Eur. J. Gastroenterol. Hepatol. 2005, 17, 293–301. [Google Scholar] [CrossRef]
- Kwee, R.M.; Kwee, T.C. Imaging in Assessing Lymph Node Status in Gastric Cancer. Gastric Cancer 2009, 12, 6–22. [Google Scholar] [CrossRef]
- Kwee, R.M.; Kwee, T.C. The Accuracy of Endoscopic Ultrasonography in Differentiating Mucosal from Deeper Gastric Cancer. Am. J. Gastroenterol. 2008, 103, 1801–1809. [Google Scholar] [CrossRef] [PubMed]
- Botet, J.F.; Lightdale, C.J.; Zauber, A.G.; Gerdes, H.; Winawer, S.J.; Urmacher, C.; Brennan, M.F. Preoperative Staging of Gastric Cancer: Comparison of Endoscopic US and Dynamic CT. Radiology 1991, 181, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.A.; Scheiman, J.M. Initial Experience with an Electronic Radial Array Echoendoscope: Randomized Comparison with a Mechanical Sector Scanning Echoendoscope in Humans. Gastrointest. Endosc. 2002, 56, 573–577. [Google Scholar] [CrossRef]
- Noh, K.W.; Woodward, T.A.; Raimondo, M.; Savoy, A.D.; Pungpapong, S.; Hardee, J.D.; Wallace, M.B. Changing Trends in Endosonography: Linear Imaging and Tissue Are Increasingly the Issue. Dig. Dis. Sci. 2007, 52, 1014–1018. [Google Scholar] [CrossRef]
- Rösch, T. The Radial Echoendoscope: Here to Stay or Gone Tomorrow? Gastrointest. Endosc. 2009, 69, S159–S162. [Google Scholar] [CrossRef]
- Ahn, H.S.; Lee, H.-J.; Yoo, M.-W.; Kim, S.G.; Im, J.P.; Kim, S.H.; Kim, W.H.; Lee, K.U.; Yang, H.-K. Diagnostic Accuracy of T and N Stages with Endoscopy, Stomach Protocol CT, and Endoscopic Ultrasonography in Early Gastric Cancer. J. Surg. Oncol. 2009, 99, 20–27. [Google Scholar] [CrossRef]
- Kuntz, C.; Herfarth, C. Imaging Diagnosis for Staging of Gastric Cancer. Semin. Surg. Oncol. 1999, 17, 96–102. [Google Scholar] [CrossRef]
- Ilson, D.H. Advances in the Treatment of Gastric Cancer: 2019. Curr. Opin. Gastroenterol. 2019, 35, 551–554. [Google Scholar] [CrossRef]
- Puli, S.R.; Reddy, J.B.K.; Bechtold, M.L.; Antillon, M.R.; Ibdah, J.A. How Good Is Endoscopic Ultrasound for TNM Staging of Gastric Cancers? A Meta-Analysis and Systematic Review. World. J. Gastroenterol. 2008, 14, 4011–4019. [Google Scholar] [CrossRef]
- Hizawa, K.; Iwai, K.; Esaki, M.; Matsumoto, T.; Suekane, H.; Iida, M. Is Endoscopic Ultrasonography Indispensable in Assessing the Appropriateness of Endoscopic Resection for Gastric Cancer? Endoscopy 2002, 34, 973–978. [Google Scholar] [CrossRef]
- Hwang, S.W.; Lee, D.H.; Lee, S.H.; Park, Y.S.; Hwang, J.H.; Kim, J.W.; Jung, S.H.; Kim, N.Y.; Kim, Y.H.; Lee, K.H.; et al. Preoperative Staging of Gastric Cancer by Endoscopic Ultrasonography and Multidetector-Row Computed Tomography: Preoperative Staging of Gastric Cancer. J. Gastroenterol. Hepatol. 2010, 25, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Habermann, C.R.; Weiss, F.; Riecken, R.; Honarpisheh, H.; Bohnacker, S.; Staedtler, C.; Dieckmann, C.; Schoder, V.; Adam, G. Preoperative Staging of Gastric Adenocarcinoma: Comparison of Helical CT and Endoscopic US. Radiology 2004, 230, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Anand, D.; Barroeta, J.E.; Gupta, P.K.; Kochman, M.; Baloch, Z.W. Endoscopic Ultrasound Guided Fine Needle Aspiration of Non-Pancreatic Lesions: An Institutional Experience. J. Clin. Pathol. 2007, 60, 1254–1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.J.; Kim, A.Y.; Oh, S.T.; Kim, J.-S.; Kim, K.W.; Kim, P.N.; Lee, M.-G.; Ha, H.K. Gastric Cancer Staging at Multi–Detector Row CT Gastrography: Comparison of Transverse and Volumetric CT Scanning. Radiology 2005, 236, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Gossios, K.J.; Tsianos, E.V.; Demou, L.L.; Tatsis, C.K.; Papakostas, V.P.; Masalas, C.N.; Merkouropoulos, M.C.; Kontogiannis, D.S. Use of Water or Air as Oral Contrast Media for Computed Tomographic Study of the Gastric Wall: Comparison of the Two Techniques. Gastrointest. Radiol. 1991, 16, 293–297. [Google Scholar] [CrossRef]
- Kim, Y.M.; Baek, S.-E.; Lim, J.S.; Hyung, W.J. Clinical Application of Image-Enhanced Minimally Invasive Robotic Surgery for Gastric Cancer: A Prospective Observational Study. J. Gastrointest. Surg. 2013, 17, 304–312. [Google Scholar] [CrossRef]
- Minami, M.; Kawauchi, N.; Itai, Y.; Niki, T.; Sasaki, Y. Gastric Tumors: Radiologic-Pathologic Correlation and Accuracy of T Staging with Dynamic CT. Radiology 1992, 185, 173–178. [Google Scholar] [CrossRef]
- Fukuya, T.; Honda, H.; Kaneko, K.; Kuroiwa, T.; Yoshimitsu, K.; Irie, H.; Maehara, Y.; Masuda, K. Efficacy of Helical CT in T-Staging of Gastric Cancer. J. Comput. Assist. Tomogr. 1997, 21, 73–81. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Hsu, J.-S.; Wu, D.-C.; Kang, W.-Y.; Hsieh, J.-S.; Jaw, T.-S.; Wu, M.-T.; Liu, G.-C. Gastric Cancer: Preoperative Local Staging with 3D Multi–Detector Row CT—Correlation with Surgical and Histopathologic Results. Radiology 2007, 242, 472–482. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Wu, D.-C.; Kang, W.-Y.; Hsu, J.-S. Staging of Gastric Cancer with 16-Channel MDCT. Abdom. Imaging 2006, 31, 514–520. [Google Scholar] [CrossRef]
- Pan, Z.; Zhang, H.; Yan, C.; Du, L.; Ding, B.; Song, Q.; Ling, H.; Huang, B.; Chen, K. Determining Gastric Cancer Resectability by Dynamic MDCT. Eur. Radiol. 2010, 20, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Eun, H.W.; Hong, S.S.; Kim, Y.J.; Han, J.K.; Choi, B.I. Gastric Cancer Detection Using MDCT Compared with 2D Axial CT: Diagnostic Accuracy of Three Different Reconstruction Techniques. Abdom. Imaging 2012, 37, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.; Yuan, K.; Shen, Y.; Li, M.; Wang, Q.; Xing, L.; Shi, G. Clinical Value of Magnetic Resonance Imaging in Preoperative T Staging of Gastric Cancer and Postoperative Pathological Diagnosis. Oncol. Lett. 2014, 8, 275–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dorfman, R.E.; Alpern, M.B.; Gross, B.H.; Sandler, M.A. Upper Abdominal Lymph Nodes: Criteria for Normal Size Determined with CT. Radiology 1991, 180, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Smyth, E.C.; Verheij, M.; Allum, W.; Cunningham, D.; Cervantes, A.; Arnold, D. Gastric Cancer: ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2016, 27, v38–v49. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, X.; Shan, X.; Pan, D.; Jia, Y.; Ni, E.; Hu, Y.; Huang, H. Value of Multi-Slice Spiral Computed Tomography in the Diagnosis of Metastatic Lymph Nodes and N-Stage of Gastric Cancer. J. Int. Med. Res. 2019, 47, 281–292. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, H.-H.; Kim, Y.H.; Hwang, S.H.; Lee, H.S.; Park, D.J.; Kim, S.Y.; Lee, K.H. Peritoneal Metastasis: Detection with 16– or 64–Detector Row CT in Patients Undergoing Surgery for Gastric Cancer. Radiology 2009, 253, 407–415. [Google Scholar] [CrossRef]
- Kim, S.H.; Choi, Y.H.; Kim, J.W.; Oh, S.; Lee, S.; Kim, B.G.; Lee, K.L. Clinical Significance of Computed Tomography-Detected Ascites in Gastric Cancer Patients with Peritoneal Metastases. Medicine 2018, 97, e9343. [Google Scholar] [CrossRef]
- Saiz Martínez, R.; Dromain, C.; Vietti Violi, N. Imaging of Gastric Carcinomatosis. J. Clin. Med. 2021, 10, 5294. [Google Scholar] [CrossRef]
- Jacquet, P.; Sugarbaker, P.H. Clinical Research Methodologies in Diagnosis and Staging of Patients with Peritoneal Carcinomatosis. Cancer Treat. Res. 1996, 82, 359–374. [Google Scholar] [CrossRef]
- Paley, M.R.; Ros, P.R. MRI of the Gastrointestinal Tract. Eur. Radiol. 1997, 7, 1387–1397. [Google Scholar] [CrossRef]
- Kim, I.Y.; Kim, S.W.; Shin, H.C.; Lee, M.S.; Jeong, D.J.; Kim, C.J.; Kim, Y.T. MRI of Gastric Carcinoma: Results of T and N-Staging in an in Vitro Study. World J. Gastroenterol. 2009, 15, 3992–3998. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to Build a Bridge from a Population-Based to a More “Personalized” Approach to Cancer Staging: The Eighth Edition AJCC Cancer Staging Manual. CA A Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Seevaratnam, R.; Cardoso, R.; McGregor, C.; Lourenco, L.; Mahar, A.; Sutradhar, R.; Law, C.; Paszat, L.; Coburn, N. How Useful Is Preoperative Imaging for Tumor, Node, Metastasis (TNM) Staging of Gastric Cancer? A Meta-Analysis. Gastric Cancer 2012, 15 (Suppl. 1), 3–18. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Xie, D.H.; Guo, L.; Hu, C.H.; Fang, X.; Meng, Q.; Ping, X.X.; Lu, Z.W. The Utility of MRI for Pre-Operative T and N Staging of Gastric Carcinoma: A Systematic Review and Meta-Analysis. Br. J. Radiol. 2015, 88, 20140552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anzidei, M.; Napoli, A.; Zaccagna, F.; Di Paolo, P.; Zini, C.; Cavallo Marincola, B.; Geiger, D.; Catalano, C.; Passariello, R. Diagnostic Performance of 64-MDCT and 1.5-T MRI with Highresolution Sequences in the T Staging of Gastric Cancer: A Comparative Analysis with Histopathology. Radiol. Med. 2009, 114, 1065–1079. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Marcelli, G.; Al Ansari, N.; Zippi, M.; De Marco, V.; Kagarmanova, A.; Vestri, A.; Marcheggiano-Clarke, L.; Marini, M. Preoperative T and N Staging of Gastric Cancer: Magnetic Resonance Imaging (MRI) versus Multi Detector Computed Tomography (MDCT). Clin. Ter. 2010, 161, e57–e62. [Google Scholar]
- Laghi, A.; Bellini, D.; Rengo, M.; Accarpio, F.; Caruso, D.; Biacchi, D.; Di Giorgio, A.; Sammartino, P. Diagnostic Performance of Computed Tomography and Magnetic Resonance Imaging for Detecting Peritoneal Metastases: Systematic Review and Meta-Analysis. Radiol. Med. 2017, 122, 1–15. [Google Scholar] [CrossRef]
- Soydan, L.; Demir, A.A.; Torun, M.; Cikrikcioglu, M.A. Use of Diffusion-Weighted Magnetic Resonance Imaging and Apparent Diffusion Coefficient in Gastric Cancer Staging. Curr. Med. Imaging 2021, 16, 1278–1289. [Google Scholar] [CrossRef]
- Arslan, H.; Fatih Özbay, M.; Çallı, İ.; Doğan, E.; Çelik, S.; Batur, A.; Bora, A.; Yavuz, A.; Bulut, M.D.; Özgökçe, M.; et al. Contribution of Diffusion Weighted MRI to Diagnosis and Staging in Gastric Tumors and Comparison with Multi-Detector Computed Tomography. Radiol. Oncol. 2017, 51, 23–29. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; He, J.; Guan, W.; Li, Q.; Yu, H.; Zhou, Z.; Bao, S.; Zhou, Z. Added Value of Diffusion-Weighted MR Imaging to T2-Weighted and Dynamic Contrast-Enhanced MR Imaging in T Staging of Gastric Cancer. Clin. Imaging 2014, 38, 122–128. [Google Scholar] [CrossRef]
- Vreugdenburg, T.D.; Ma, N.; Duncan, J.K.; Riitano, D.; Cameron, A.L.; Maddern, G.J. Comparative Diagnostic Accuracy of Hepatocyte-Specific Gadoxetic Acid (Gd-EOB-DTPA) Enhanced MR Imaging and Contrast Enhanced CT for the Detection of Liver Metastases: A Systematic Review and Meta-Analysis. Int. J. Color. Dis. 2016, 31, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Li, K.; Peng, T. Diagnostic Value of Diffusion-Weighted Imaging/Magnetic Resonance Imaging for Peritoneal Metastasis from Malignant Tumor: A Systematic Review and Meta-Analysis. Medicine 2021, 100, e24251. [Google Scholar] [CrossRef] [PubMed]
- Kumano, S.; Murakami, T.; Kim, T.; Hori, M.; Iannaccone, R.; Nakata, S.; Onishi, H.; Osuga, K.; Tomoda, K.; Catalano, C.; et al. T Staging of Gastric Cancer: Role of Multi–Detector Row CT. Radiology 2005, 237, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.M.; Kim, H.C.; Jin, W.; Ryu, C.W.; Kang, J.H.; Park, C.H.; Kim, H.S.; Jung, D.H. 64 Multidetector-Row Computed Tomography for Preoperative Evaluation of Gastric Cancer: Histological Correlation. J. Comput. Assist. Tomogr. 2007, 31, 98–103. [Google Scholar] [CrossRef]
- Mehmedovi, A.; Mesihovi, R.; Saray, A.; Vanis, N. Gastric Cancer Staging: EUS And CT. Med. Arh. 2014, 68, 34–36. [Google Scholar] [CrossRef] [Green Version]
- Bhandari, S.; Sup Shim, C.; Hoon Kim, J.; Seop Jung, I.; Young Cho, J.; Seong Lee, J.; Sung Lee, M.; Sung Kim, B. Usefulness of Three-Dimensional, Multidetector Row CT (Virtual Gastroscopy and Multiplanar Reconstruction) in the Evaluation of Gastric Cancer: A Comparison with Conventional Endoscopy, EUS, and Histopathology. Gastrointest. Endosc. 2004, 59, 619–626. [Google Scholar] [CrossRef]
- Furukawa, K.; Miyahara, R.; Itoh, A.; Ohmiya, N.; Hirooka, Y.; Mori, K.; Goto, H. Diagnosis of the Invasion Depth of Gastric Cancer Using MDCT With Virtual Gastroscopy: Comparison with Staging with Endoscopic Ultrasound. Am. J. Roentgenol. 2011, 197, 867–875. [Google Scholar] [CrossRef]
- Giganti, F.; Orsenigo, E.; Arcidiacono, P.G.; Nicoletti, R.; Albarello, L.; Ambrosi, A.; Salerno, A.; Esposito, A.; Petrone, M.C.; Chiari, D.; et al. Preoperative Locoregional Staging of Gastric Cancer: Is There a Place for Magnetic Resonance Imaging? Prospective Comparison with EUS and Multidetector Computed Tomography. Gastric Cancer 2016, 19, 216–225. [Google Scholar] [CrossRef] [Green Version]
- Hwang, S.W. Is Endoscopic Ultrasonography Still the Modality of Choice in Preoperative Staging of Gastric Cancer? World J. Gastroenterol. 2014, 20, 13775–13782. [Google Scholar] [CrossRef]
- Ungureanu, B.S.; Sacerdotianu, V.M.; Turcu-Stiolica, A.; Cazacu, I.M.; Saftoiu, A. Endoscopic Ultrasound vs. Computed Tomography for Gastric Cancer Staging: A Network Meta-Analysis. Diagnostics 2021, 11, 134. [Google Scholar] [CrossRef]
- Marcus, C.; Subramaniam, R.M. PET/Computed Tomography and Precision Medicine. PET Clin. 2017, 12, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Findlay, J.M.; Antonowicz, S.; Segaran, A.; El Kafsi, J.; Zhang, A.; Bradley, K.M.; Gillies, R.S.; Maynard, N.D.; Middleton, M.R. Routinely Staging Gastric Cancer with 18F-FDG PET-CT Detects Additional Metastases and Predicts Early Recurrence and Death after Surgery. Eur. Radiol. 2019, 29, 2490–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foley, K.G.; Coomer, W.; Coles, B.; Bradley, K.M. The Impact of Baseline 18 F-FDG PET-CT on the Management and Outcome of Patients with Gastric Cancer: A Systematic Review. Br. J. Radiol. 2022, 95, 20220437. [Google Scholar] [CrossRef]
- Ma, D.; Zhang, Y.; Shao, X.; Wu, C.; Wu, J. PET/CT for Predicting Occult Lymph Node Metastasis in Gastric Cancer. Curr. Oncol. 2022, 29, 6523–6539. [Google Scholar] [CrossRef]
- Song, B.-I.; Kim, H.W.; Won, K.S.; Ryu, S.W.; Sohn, S.S.; Kang, Y.N. Preoperative Standardized Uptake Value of Metastatic Lymph Nodes Measured by 18F-FDG PET/CT Improves the Prediction of Prognosis in Gastric Cancer. Medicine 2015, 94, e1037. [Google Scholar] [CrossRef]
- Wang, X.; Wei, Y.; Xue, Y.; Lu, P.; Yu, L.; Shen, B. Predictive Role of the Number of 18F-FDG-Positive Lymph Nodes Detected by PET/CT for Pre-Treatment Evaluation of Locally Advanced Gastric Cancer. PLoS ONE 2016, 11, e0166836. [Google Scholar] [CrossRef] [Green Version]
- Qin, C.; Shao, F.; Gai, Y.; Liu, Q.; Ruan, W.; Liu, F.; Hu, F.; Lan, X. 68 Ga-DOTA-FAPI-04 PET/MR in the Evaluation of Gastric Carcinomas: Comparison with 18 F-FDG PET/CT. J. Nucl. Med. 2022, 63, 81–88. [Google Scholar] [CrossRef]
- Fu, L.; Huang, S.; Wu, H.; Dong, Y.; Xie, F.; Wu, R.; Zhou, K.; Tang, G.; Zhou, W. RETRACTED ARTICLE: Superiority of [68Ga]Ga-FAPI-04/[18F]FAPI-42 PET/CT to [18F]FDG PET/CT in Delineating the Primary Tumor and Peritoneal Metastasis in Initial Gastric Cancer. Eur. Radiol. 2022, 32, 6281–6290. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, W.; Xu, T.; Ding, H.; Li, Y.; Liu, H.; Huang, Y.; Liu, L.; Du, T.; Zhao, Y.; et al. Comparison of Diagnostic Efficacy of [68Ga]Ga-FAPI-04 and [18F]FDG PET/CT for Staging and Restaging of Gastric Cancer. Front. Oncol. 2022, 12, 925100. [Google Scholar] [CrossRef]
- Huh, J.; Lee, I.-S.; Kim, K.W.; Park, J.; Kim, A.Y.; Lee, J.S.; Yook, J.-H.; Kim, B.-S. CT Gastrography for Volumetric Measurement of Remnant Stomach after Distal Gastrectomy: A Feasibility Study. Abdom. Radiol. 2016, 41, 1899–1905. [Google Scholar] [CrossRef] [PubMed]
- Hallinan, J.T.P.D.; Venkatesh, S.K.; Peter, L.; Makmur, A.; Yong, W.P.; So, J.B.Y. CT Volumetry for Gastric Carcinoma: Association with TNM Stage. Eur. Radiol. 2014, 24, 3105–3114. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Chen, T.; Hu, J.; Guo, D.; Zhang, X.; Deng, D.; Li, H.; Chen, X.; Tang, H. Tumor Volume of Resectable Adenocarcinoma of the Esophagogastric Junction at Multidetector CT: Association with Regional Lymph Node Metastasis and N Stage. Radiology 2013, 269, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, S.; Sakuramoto, S.; Kobayashi, N.; Shimao, H.; Sakakibara, Y.; Sato, K.; Kakita, A. A New Staging System Based on Tumor Volume in Gastric Cancer. Anticancer Res. 2001, 21, 2933–2936. [Google Scholar]
- Yao, J.; Yang, Z.; Chen, T.; Li, Y.; Yang, L. Perfusion Changes in Gastric Adenocarcinoma: Evaluation with 64-Section MDCT. Abdom. Imaging 2010, 35, 195–202. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, Z.; Du, L.; Yan, C.; Ding, B.; Song, Q.; Ling, H.; Chen, K. Advanced Gastric Cancer and Perfusion Imaging Using a Multidetector Row Computed Tomography: Correlation with Prognostic Determinants. Korean J. Radiol. 2008, 9, 119–127. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Xu, F.; Chen, Q.; Hu, H.; Qi, F.; Zhao, J. The Value of CT-Based Radiomics Nomogram in Differential Diagnosis of Different Histological Types of Gastric Cancer. Phys. Eng. Sci. Med. 2022, 45, 1063–1071. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, L.; Liu, S.; You, J.; Chen, L.; Jin, Z.; Zhang, S.; Zhang, B. Radiomics in Precision Medicine for Gastric Cancer: Opportunities and Challenges. Eur. Radiol. 2022, 32, 5852–5868. [Google Scholar] [CrossRef]
- Xu, Q.; Sun, Z.; Li, X.; Ye, C.; Zhou, C.; Zhang, L.; Lu, G. Advanced Gastric Cancer: CT Radiomics Prediction and Early Detection of Downstaging with Neoadjuvant Chemotherapy. Eur. Radiol. 2021, 31, 8765–8774. [Google Scholar] [CrossRef]
- Huang, W.; Zhou, K.; Jiang, Y.; Chen, C.; Yuan, Q.; Han, Z.; Xie, J.; Yu, S.; Sun, Z.; Hu, Y.; et al. Radiomics Nomogram for Prediction of Peritoneal Metastasis in Patients with Gastric Cancer. Front. Oncol. 2020, 10, 1416. [Google Scholar] [CrossRef]
- Ashworth, S.; Spirito, A.; Colella, A.; Drew, C.B. A Pilot Suicidal Awareness, Identification, and Prevention Program. R. I. Med. J. 1976 1986, 69, 457–461. [Google Scholar] [PubMed]
- Jiang, Y.; Liang, X.; Wang, W.; Chen, C.; Yuan, Q.; Zhang, X.; Li, N.; Chen, H.; Yu, J.; Xie, Y.; et al. Noninvasive Prediction of Occult Peritoneal Metastasis in Gastric Cancer Using Deep Learning. JAMA Netw. Open 2021, 4, e2032269. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liang, X.; Han, Z.; Wang, W.; Xi, S.; Li, T.; Chen, C.; Yuan, Q.; Li, N.; Yu, J.; et al. Radiographical Assessment of Tumour Stroma and Treatment Outcomes Using Deep Learning: A Retrospective, Multicohort Study. Lancet Digit. Health 2021, 3, e371–e382. [Google Scholar] [CrossRef] [PubMed]








| Ref # | Manuscript Type | Main Findings |
|---|---|---|
| [19] | Meta-analysis |
|
| [20] | Original study |
|
| [21] | Original study |
|
| [22] | Original study |
|
| Stage | Pathological Features | CT Features |
|---|---|---|
| T1 | lesion invades the lamina propria, muscularis mucosae and submucosa |
|
| T2 | lesion invades the muscularis propria |
|
| T3 | lesion invades the subserosa |
|
| T4a | lesion invades the serosa |
|
| T4b | lesion invades adjacent structures |
|
| Ref # | Manuscript Type | Main Findings |
|---|---|---|
| [24] | Original study |
|
| [29] | Original study |
|
| [31] | Original study |
|
| [32] | Original study |
|
| [36] | Original study |
|
| [37] | Original study |
|
| [38] | Original study |
|
| Ref # | Manuscript Type | Main Findings |
|---|---|---|
| [44] | Meta-analysis |
|
| [45] | Meta-analysis |
|
| [46] | Original study |
|
| [48] | Meta-analysis |
|
| [49] | Original study |
|
| [50] | Original study |
|
| [51] | Original study |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giandola, T.; Maino, C.; Marrapodi, G.; Ratti, M.; Ragusi, M.; Bigiogera, V.; Talei Franzesi, C.; Corso, R.; Ippolito, D. Imaging in Gastric Cancer: Current Practice and Future Perspectives. Diagnostics 2023, 13, 1276. https://doi.org/10.3390/diagnostics13071276
Giandola T, Maino C, Marrapodi G, Ratti M, Ragusi M, Bigiogera V, Talei Franzesi C, Corso R, Ippolito D. Imaging in Gastric Cancer: Current Practice and Future Perspectives. Diagnostics. 2023; 13(7):1276. https://doi.org/10.3390/diagnostics13071276
Chicago/Turabian StyleGiandola, Teresa, Cesare Maino, Giuseppe Marrapodi, Michele Ratti, Maria Ragusi, Vittorio Bigiogera, Cammillo Talei Franzesi, Rocco Corso, and Davide Ippolito. 2023. "Imaging in Gastric Cancer: Current Practice and Future Perspectives" Diagnostics 13, no. 7: 1276. https://doi.org/10.3390/diagnostics13071276
APA StyleGiandola, T., Maino, C., Marrapodi, G., Ratti, M., Ragusi, M., Bigiogera, V., Talei Franzesi, C., Corso, R., & Ippolito, D. (2023). Imaging in Gastric Cancer: Current Practice and Future Perspectives. Diagnostics, 13(7), 1276. https://doi.org/10.3390/diagnostics13071276





