The Role of QRS Complex and ST-Segment in Major Adverse Cardiovascular Events Prediction in Patients with ST Elevated Myocardial Infarction: A 6-Year Follow-Up Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample and Group Formation
2.2. Electrocardiographic Data
2.3. Coronary Angiography
2.4. Echocardiography
2.5. Patient Follow-Up
2.6. Statistical Analysis
3. Results
3.1. Baseline Patients Characteristics
3.2. ECG Data
3.2.1. QRS Complex Width Measurement
3.2.2. ST-Segment Elevation
3.3. Follow-Up Data
3.3.1. 6-Month Follow-Up
3.3.2. 6-Year Follow-Up
3.4. ECG Data in MACE Prediction
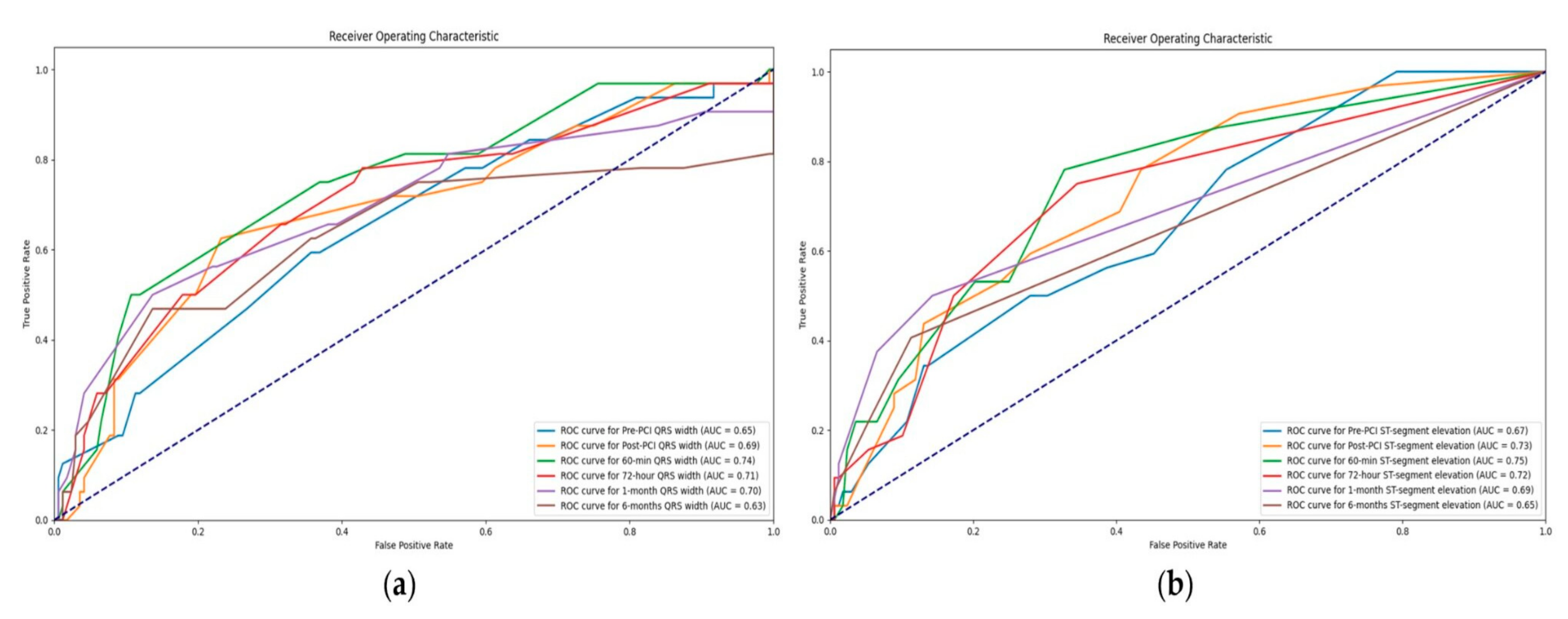
3.4.1. Value of QRS Complex Width in Association with MACE
3.4.2. Value of ST-Segment Elevation in Association with MACE
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, M.; Pan, J.; Meng, K.; Wang, Y.; Sun, X.; Ma, L.; Yu, X. Triglyceride-Glucose Body Mass Index Predicts Prognosis in Patients with ST-Elevation Myocardial Infarction. Sci. Rep. 2024, 14, 976. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Kong, L.; An, D.; Chen, B.; Zhao, C.; Li, Z.; Yang, F.; Dong, J.; Wei, L.; Shan, P.; et al. Prognostic Value of Segmental Strain After ST-Elevation Myocardial Infarction: Insights from the EARLY Assessment of MYOcardial Tissue Characteristics by Cardiac Magnetic Resonance (EARLY-MYO-CMR) Study. J. Magn. Reson. Imaging 2024. [Google Scholar] [CrossRef] [PubMed]
- Giustino, G.; Mehran, R.; Dangas, G.D.; Kirtane, A.J.; Redfors, B.; Généreux, P.; Brener, S.J.; Prats, J.; Pocock, S.J.; Deliargyris, E.N.; et al. Characterization of the Average Daily Ischemic and Bleeding Risk After Primary PCI for STEMI. J. Am. Coll. Cardiol. 2017, 70, 1846–1857. [Google Scholar] [CrossRef] [PubMed]
- Tsai, I.-T.; Wang, C.-P.; Lu, Y.-C.; Hung, W.-C.; Wu, C.-C.; Lu, L.-F.; Chung, F.-M.; Hsu, C.-C.; Lee, Y.-J.; Yu, T.-H. The Burden of Major Adverse Cardiac Events in Patients with Coronary Artery Disease. BMC Cardiovasc. Disord 2017, 17, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Poudel, I.; Tejpal, C.; Rashid, H.; Jahan, N. Major Adverse Cardiovascular Events: An Inevitable Outcome of ST-Elevation Myocardial Infarction? A Literature Review. Cureus 2019, 11, e5280. [Google Scholar] [CrossRef] [PubMed]
- Miao, B.; Hernandez, A.V.; Alberts, M.J.; Mangiafico, N.; Roman, Y.M.; Coleman, C.I. Incidence and Predictors of Major Adverse Cardiovascular Events in Patients with Established Atherosclerotic Disease or Multiple Risk Factors. J. Am. Heart Assoc. 2020, 9, e014402. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, F.; Butrymovich, V.; Kelbæk, H.; Wachtell, K.; Helqvist, S.; Kastrup, J.; Holmvang, L.; Clemmensen, P.; Engstrøm, T.; Grande, P.; et al. Short- and Long-Term Cause of Death in Patients Treated with Primary PCI for STEMI. J. Am. Coll. Cardiol. 2014, 64, 2101–2108. [Google Scholar] [CrossRef]
- Tanriverdi, Z.; Dursun, H.; Simsek, M.A.; Unal, B.; Kozan, O.; Kaya, D. The Predictive Value of Fragmented QRS and QRS Distortion for High-Risk Patients with STEMI and for the Reperfusion Success. Ann. Noninvasive Electrocardiol. 2015, 20, 578–585. [Google Scholar] [CrossRef]
- Savonitto, S.; Ardissino, D.; Granger, C.B.; Morando, G.; Prando, M.D.; Mafrici, A.; Cavallini, C.; Melandri, G.; Thompson, T.D.; Vahanian, A.; et al. Prognostic Value of the Admission Electrocardiogram in Acute Coronary Syndromes. JAMA 1999, 281, 707–713. [Google Scholar] [CrossRef]
- Rafie, N.; Kashou, A.H.; Noseworthy, P.A. ECG Interpretation: Clinical Relevance, Challenges, and Advances. Hearts 2021, 2, 505–513. [Google Scholar] [CrossRef]
- Alhamaydeh, M.; Gregg, R.; Ahmad, A.; Faramand, Z.; Saba, S.; Al-Zaiti, S. Identifying the Most Important ECG Predictors of Reduced Ejection Fraction in Patients with Suspected Acute Coronary Syndrome. J. Electrocardiol. 2020, 61, 81–85. [Google Scholar] [CrossRef] [PubMed]
- López-Castillo, M.; Aceña, Á.; Pello-Lázaro, A.M.; Viegas, V.; Merchán Muñoz, B.; Carda, R.; Franco-Peláez, J.; Martín-Mariscal, M.L.; Briongos-Figuero, S.; Tuñón, J. Prognostic Value of Initial QRS Analysis in Anterior STEMI: Correlation with Left Ventricular Systolic Dysfunction, Serum Biomarkers, and Cardiac Outcomes. Ann. Noninvasive Electrocardiol. 2021, 26, e12791. [Google Scholar] [CrossRef] [PubMed]
- Buszman, P.; Szafranek, A.; Kalarus, Z.; Gasior, M. Use of Changes in ST Segment Elevation for Prediction of Infarct Artery Recanalization in Acute Myocardial Infarction. Eur. Heart J. 1995, 16, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Matetzky, S.; Novikov, M.; Gruberg, L.; Freimark, D.; Feinberg, M.; Elian, D.; Novikov, I.; Di, S.E.; Agranat, O.; Har-Zahav, Y.; et al. The Significance of Persistent ST Elevation versus Early Resolution of ST Segment Elevation after Primary PTCA. J. Am. Coll. Cardiol. 1999, 34, 1932–1938. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Voigt, J.U. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. Off. Publ. Am. Soc. Echocardiogr. 2015, 28, 1–39.e14. [Google Scholar] [CrossRef] [PubMed]
- Resting Heart Rate and Long-Term Outcomes in Patients with Percutaneous Coronary Intervention: Results from a 10-Year Follow-Up of the CORFCHD-PCI Study. Available online: https://www.hindawi.com/journals/crp/2019/5432076/ (accessed on 28 February 2024).
- Noman, A.; Balasubramaniam, K.; Das, R.; Ang, D.; Kunadian, V.; Ivanauskiene, T.; Zaman, A.G. Admission Heart Rate Predicts Mortality Following Primary Percutaneous Coronary Intervention for ST-Elevation Myocardial Infarction: An Observational Study. Cardiovasc. Ther. 2013, 31, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Wah, W.; Pek, P.P.; Ho, A.F.W.; Fook-Chong, S.; Zheng, H.; Loy, E.Y.; Chua, T.S.J.; Koh, T.H.; Chow, K.Y.; Earnest, A.; et al. Symptom-to-Door Delay among Patients with ST-Segment Elevation Myocardial Infarction in Singapore. Emerg. Med. Australas. 2017, 29, 24–32. [Google Scholar] [CrossRef]
- Hafiz, A.M.; Naidu, S.S.; DeLeon, J.; Islam, S.; Alkhatib, B.; Lorenz, M.; D’Elia, A.; Rosenthal, B.; Marzo, K. Impact of First Contact on Symptom Onset–to-Door Time in Patients Presenting for Primary Percutaneous Coronary Intervention. Am. J. Emerg. Med. 2013, 31, 922–927. [Google Scholar] [CrossRef]
- De Luca, G.; Parodi, G.; Sciagrà, R.; Venditti, F.; Bellandi, B.; Vergara, R.; Migliorini, A.; Valenti, R.; Antoniucci, D. Time-to-Treatment and Infarct Size in STEMI Patients Undergoing Primary Angioplasty. Int. J. Cardiol. 2013, 167, 1508–1513. [Google Scholar] [CrossRef]
- Symptom Onset-to-Balloon Time and Mortality in the First Seven Years After STEMI Treated with Primary Percutaneous Coronary Intervention|Heart. Available online: https://heart.bmj.com/content/98/23/1738.short (accessed on 28 February 2024).
- Birdal, O.; Pay, L.; Aksakal, E.; Yumurtaş, A.Ç.; Çinier, G.; Yücel, E.; Tanboğa, İ.H.; Karagöz, A.; Oduncu, V. Naples Prognostic Score and Prediction of Left Ventricular Ejection Fraction in STEMI Patients. Angiology 2024, 75, 36–43. [Google Scholar] [CrossRef]
- Lazăr, M.-A.; Ionac, I.; Luca, C.-T.; Petrescu, L.; Vacarescu, C.; Crisan, S.; Gaiță, D.; Cozma, D.; Sosdean, R.; Arnăutu, D.-A.; et al. Reduced Left Ventricular Twist Early after Acute ST-Segment Elevation Myocardial Infarction as a Predictor of Left Ventricular Adverse Remodelling. Diagnostics 2023, 13, 2896. [Google Scholar] [CrossRef] [PubMed]
- Evaluation of a QRS Scoring System for Estimating Myocardial Infarct Size. I. Specificity and Observer Agreement. Circulation. Available online: https://www.ahajournals.org/doi/abs/10.1161/01.CIR.65.2.342 (accessed on 28 February 2024).
- QRS Score at Presentation Electrocardiogram Is Correlated with Infarct Size and Mortality in ST-Segment Elevation Myocardial Infarction Patients Undergoing Primary Percutaneous Coronary Intervention. Available online: https://www.jstage.jst.go.jp/article/circj/81/8/81_CJ-16-1255/_article/-char/ja/ (accessed on 28 February 2024).
- Das, M.K.; Michael, M.A.; Suradi, H.; Peng, J.; Sinha, A.; Shen, C.; Mahenthiran, J.; Kovacs, R.J. Usefulness of Fragmented QRS on a 12-Lead Electrocardiogram in Acute Coronary Syndrome for Predicting Mortality. Am. J. Cardiol. 2009, 104, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, Y.; Zhang, P.; Zhang, J.; Cao, X.; He, S.; Yang, D. Both Baseline Selvester QRS Score and Change in QRS Score Predict Prognosis in Patients with Acute ST-Segment Elevation Myocardial Infarction after Percutaneous Coronary Intervention. Coron. Artery Dis. 2020, 31, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Palomares, J.F.; Figueras-Bellot, J.; Descalzo, M.; Moral, S.; Otaegui, I.; Pineda, V.; del Blanco, B.G.; González-Alujas, M.T.; Evangelista Masip, A.; García-Dorado, D. Relation of ST-Segment Elevation Before and After Percutaneous Transluminal Coronary Angioplasty to Left Ventricular Area at Risk, Myocardial Infarct Size, and Systolic Function. Am. J. Cardiol. 2014, 113, 593–600. [Google Scholar] [CrossRef]
- Weaver, J.C.; Ramsay, D.D.; Rees, D.; Binnekamp, M.F.; Prasan, A.M.; McCrohon, J.A. Dynamic Changes in ST Segment Resolution After Myocardial Infarction and the Association with Microvascular Injury on Cardiac Magnetic Resonance Imaging. Heart Lung Circ. 2011, 20, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Dizon, J.M.; Brener, S.J.; Maehara, A.; Witzenbichler, B.; Biviano, A.; Godlewski, J.; Parise, H.; Dambrink, J.-H.; Mehran, R.; Gibson, C.M.; et al. Relationship between ST-Segment Resolution and Anterior Infarct Size after Primary Percutaneous Coronary Intervention: Analysis from the INFUSE-AMI Trial. Eur. Heart J. Acute Cardiovasc. Care 2014, 3, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Prognostic Impact of Early ST-Segment Resolution in Acute ST-Elevation Myocardial Infarction|Circulation. Available online: https://www.ahajournals.org/doi/full/10.1161/01.CIR.0000147778.05979.E6 (accessed on 28 February 2024).
- Rakowski, T.; Dziewierz, A.; Siudak, Z.; Mielecki, W.; Brzozowska-Czarnek, A.; Legutko, J.; Rzeszutko, L.; Urbanik, A.; Dubiel, J.S.; Dudek, D. ST-Segment Resolution Assessed Immediately after Primary Percutaneous Coronary Intervention Correlates with Infarct Size and Left Ventricular Function in Cardiac Magnetic Resonance at 1-Year Follow-Up. J. Electrocardiol. 2009, 42, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Haeck, J.D.E.; Verouden, N.J.W.; Kuijt, W.J.; Koch, K.T.; Majidi, M.; Hirsch, A.; Tijssen, J.G.P.; Krucoff, M.W.; De Winter, R.J. Impact of Early, Late, and No ST-Segment Resolution Measured by Continuous ST Holter Monitoring on Left Ventricular Ejection Fraction and Infarct Size as Determined by Cardiovascular Magnetic Resonance Imaging. J. Electrocardiol. 2011, 44, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Røsand, Ø.; Høydal, M.A. Cardiac Exosomes in Ischemic Heart Disease—A Narrative Review. Diagnostics 2021, 11, 269. [Google Scholar] [CrossRef]
- Fröhlich, G.M.; Meier, P.; White, S.K.; Yellon, D.M.; Hausenloy, D.J. Myocardial Reperfusion Injury: Looking beyond Primary PCI. Eur. Heart J. 2013, 34, 1714–1722. [Google Scholar] [CrossRef]
- Stone, G.W.; Selker, H.P.; Thiele, H.; Patel, M.R.; Udelson, J.E.; Ohman, E.M.; Maehara, A.; Eitel, I.; Granger, C.B.; Jenkins, P.L.; et al. Relationship Between Infarct Size and Outcomes Following Primary PCI: Patient-Level Analysis from 10 Randomized Trials. J. Am. Coll. Cardiol. 2016, 67, 1674–1683. [Google Scholar] [CrossRef] [PubMed]
- de Waha, S.; Patel, M.R.; Granger, C.B.; Ohman, E.M.; Maehara, A.; Eitel, I.; Ben-Yehuda, O.; Jenkins, P.; Thiele, H.; Stone, G.W. Relationship between Microvascular Obstruction and Adverse Events Following Primary Percutaneous Coronary Intervention for ST-Segment Elevation Myocardial Infarction: An Individual Patient Data Pooled Analysis from Seven Randomized Trials. Eur. Heart J. 2017, 38, 3502–3510. [Google Scholar] [CrossRef] [PubMed]
- Byrne, R.A.; Rossello, X.; Coughlan, J.J.; Barbato, E.; Berry, C.; Chieffo, A.; Claeys, M.J.; Dan, G.-A.; Dweck, M.R.; Galbraith, M.; et al. 2023 ESC Guidelines for the Management of Acute Coronary Syndromes. Eur. Heart J. 2023, 44, 3720–3826. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Wang, D.; Wan, Y.; Lai, S.; Ding, Y.; Wang, X. Prediction models for major adverse cardiovascular events after percutaneous coronary intervention: A systematic review. Front. Cardiovasc. Med. 2024, 10, 1287434. [Google Scholar] [CrossRef] [PubMed]
- van der Zwaan, H.B.; Stoel, M.G.; Roos-Hesselink, J.W.; Veen, G.; Boersma, E.; von Birgelen, C. Early versus late ST-segment resolution and clinical outcomes after percutaneous coronary intervention for acute myocardial infarction. Neth. Heart J. 2010, 18, 416–422. [Google Scholar] [CrossRef]
- Ndrepepa, G.; Alger, P.; Kufner, S.; Mehilli, J.; Schömig, A.; Kastrati, A. ST-segment resolution after primary percutaneous coronary intervention in patients with acute ST-segment elevation myocardial infarction. Cardiol. J. 2012, 19, 61–69. [Google Scholar] [CrossRef]
| Variables | Whole Sample (n = 200) n (%) Mean ± SD Median [IQR] | Symptoms Duration of Less than 6 h (n = 100) Median [IQR] % | Symptoms Duration of Less than 6 h (n = 100) Median [IQR] % | p Value |
|---|---|---|---|---|
| Sex | 1.000 | |||
| Female | 58 (29%) | 29 | 29 | |
| Male | 142 (71%) | 71 | 71 | |
| Age (years) | 60.611.39 | 58.5 [52.0–67.0] | 60.0 [52.0–74.0] | 0.154 |
| Height (cm) | 173.1 [165–180] | 175.0 [166.75–180.0] | 175.0 [165.0–180.0] | 0.463 |
| Weight (kg) | 82.6 [75–90] | 82.0 [75.0–90.0] | 80.0 [70.0–90.0] | 0.299 |
| Body mass index (kg/m2) | 27.5 [24.7–29.4] | 26.8 [25.24–29.55] | 26.45 [24.35–29.4] | 0.426 |
| Body surface area (m2) | 2.00.2 | 2.0 [1.87–2.08] | 1.98 [1.83–2.12] | 0.301 |
| Heart rate (beats/min) | 78.5 [65–85] | 77.5 [65.0–85.0] | 79.0 [70.0–86.25] | 0.035 |
| Systolic blood pressure (mmHg) | 141.0 [126.3–153.8] | 140.0 [120.0–150.0] | 140.0 [130.0–160.0] | 0.131 |
| Diastolic blood pressure (mmHg) | 82.4 [70–90] | 80.0 [70.0–90.0] | 80.0 [80.0–90.0] | 0.437 |
| Vascular risk factors and comorbidities | ||||
| Hypertension | 126 (63%) | 65 | 61 | 0.660 |
| Diabetes mellitus | 36 (18%) | 17 | 19 | 0.854 |
| Family history | 63 (32%) | 32 | 31 | 1.000 |
| Kidney function | ||||
| Creatinine (micromol/L) | 96.6 | 93.0 [79.0–108.25] | 94.0 [81.75–106.25] | 0.829 |
| Clearance (mL/min) | ||||
| >90 | 89 (45%) | 50 | 39 | 0.155 |
| 60–90 | 80 (40%) | 40 | 40 | 1.000 |
| 45–59 | 23 (12%) | 7 | 16 | 0.076 |
| 30–44 | 4 (2%) | 2 | 2 | 1.000 |
| 15–29 | 4 (2%) | 1 | 3 | 0.614 |
| Duration of the symptoms (min) | 304.3 [120–420] | 120.0 [90.0–180.0] | 420.0 [360.0–600.0] | 0.001 |
| Killip | ||||
| 1 | 128 (64%) | 72 | 56 | 0.027 |
| 1/2 | 4 (2%) | 1 | 3 | 0.614 |
| 2 | 56 (28%) | 24 | 32 | 0.270 |
| 2/3 | 6 (3%) | 1 | 5 | 0.214 |
| 3 | 4 (2%) | 0 | 4 | 0.130 |
| 4 | 2 (1%) | 2 | 0 | 0.477 |
| Post-MI NYHA status | ||||
| 1 | 175 (88%) | 89 | 86 | 0.669 |
| 2 | 18 (9%) | 11 | 7 | 0.459 |
| 2/3 | 2 (1%) | 0 | 2 | 0.477 |
| 3 | 1 (0.5%) | 0 | 1 | 1.000 |
| 4 | 4 (2%) | 0 | 4 | 0.130 |
| Post-MI CCS | ||||
| 0 | 188 (94%) | 95 | 93 | 0.766 |
| 1 | 9 (5%) | 4 | 5 | 1.000 |
| 2 | 1 (1%) | 0 | 1 | 1.000 |
| 3 | 1 (1%) | 0 | 1 | 1.000 |
| 4 | 1 (1%) | 1 | 0 | 1.000 |
| Myocardial injury assessment | ||||
| Creatine Kinase isoenzyme MB (CKMB) | ||||
| Pre-PCI | 61.5 [24–68.5] | 27.5 [21.0–35.25] | 59.0 [36.0–110.0] | 0.001 |
| 6-h | 285.1 [147.3–447.8] | 244.0 [122.25–376.25] | 345.5 [168.0–500.0] | 0.007 |
| 24-h | 148.1 [74.3–189.8] | 107.0 [72.25–188.0] | 139.5 [81.0–194.25] | 0.324 |
| 72-h | 38.7 [24–41] | 30.0 [21.75–38.0] | 32.0 [25.0–45.0] | 0.040 |
| Troponin I (TnI) | ||||
| Pre-PCI | 1.4 [0.1–1.5] | 0.04 [0.01–0.2] | 1.29 [0.37–3.46] | 0.001 |
| 6-h | 17.7 [6.6–30.0] | 11.43 [5.26–29.2] | 25.0 [10.1–30.0] | 0.001 |
| 24-h | 12.9 [4.3–16.1] | 7.65 [3.3–14.89] | 10.42 [5.5–16.9] | 0.575 |
| 72-h | 3.4 [1.1–4.3] | 2.21 [1.04–3.64] | 3.1 [1.2–4.77] | 0.074 |
| In-hospital stay length (days) | 6.2 [5–7] | 6.0 [5.0–7.0] | 5.0 [5.0–7.0] | 0.265 |
| Intrahospital complications | 0 (0%) | 0 | 0 | 1.000 |
| Without | 176 (88%) | 88 | 88 | 1.000 |
| Ventricular thrombus | 4 (2%) | 2 | 2 | 1.000 |
| Pericardial effusion | 5 (3%) | 1 | 4 | 0.365 |
| Ventricular fibrillation | 11 (6%) | 8 | 3 | 0.215 |
| Pulseless electrical activity | 1 (1%) | 1 | 0 | 1.000 |
| Cardiac tamponade | 1 (1%) | 0 | 1 | 1.000 |
| Stroke | 1 (1%) | 0 | 1 | 1.000 |
| In-hospital death | 1 (1%) | 0 | 1 | 1.000 |
| Echocardiographic assessment | ||||
| In-hospital FSLV | 35.07.3 | 34.7 [29.55–38.42] | 35.95 [32.6–40.15] | 0.254 |
| In-hospital EFLV | 48.97.4 | 50.0 [46.0–55.0] | 48.0 [42.0–55.0] | 0.024 |
| In-hospital LVIDs | 3.20.6 | 3.2 [2.9–3.5] | 3.15 [2.8–3.5] | 0.647 |
| In-hospital LVIDd | 4.9 | 4.9 [4.6–5.2] | 4.9 [4.6–5.22] | 0.823 |
| In-hospital LVEDV | 102.0 [81–117] | 94.0 [84.0–115.25] | 97.0 [78.5–121.5] | 0.787 |
| In-hospital LVESV | 52.6 [39–63] | 48.0 [40.88–60.0] | 50.0 [36.25–65.0] | 0.727 |
| In-hospital MADd | 3.0 [2.9–3.2] | 3.0 [2.9–3.2] | 3.0 [2.9–3.1] | 0.374 |
| In-hospital mitral regurgitation | ||||
| 0 | 59 (30%) | 26 | 33 | 0.352 |
| 0/1 | 5 (3%) | 4 | 1 | 0.365 |
| 1 | 64 (32%) | 38 | 26 | 0.095 |
| 1/2 | 31 (16%) | 11 | 20 | 0.118 |
| 2 | 34 (17%) | 19 | 15 | 0.572 |
| 2/3 | 7 (4%) | 2 | 5 | 0.442 |
| Variables | Whole Sample (n = 200) n (%) Mean ± SD Median [IQR] | Symptoms Duration of Less than 6 h (n = 100) Median [IQR] % | Symptoms Duration of Less than 6 h (n = 100 Median [IQR] % | p Value |
|---|---|---|---|---|
| Dynamics of the QRS complex (msec) | ||||
| Pre-PCI QRS width | 100.5 | 100.0 [90.0–110.0] | 100.0 [93.5–110.0] | 0.463 |
| Post-PCI QRS width | 98.815.1 | 95.0 [85.0–100.0] | 100.0 [90.0–110.0] | 0.111 |
| 60-min QRS width | 94.7 | 86.5 [80.0–100.0] | 100.0 [87.25–100.0] | 0.008 |
| 72-h QRS width | 89.8 | 81.0 [80.0–90.5] | 95.0 [85.0–100.0] | 0.001 |
| 1-month QRS width | 87.9 | 80.0 [80.0–90.0] | 90.0 [85.0–100.0] | 0.001 |
| 6-months QRS width | 87.7 | 80.0 [80.0–90.0] | 90.0 [80.0–100.0] | 0.001 |
| ST-segment elevation (mm) | ||||
| Pre-PCI | 3.[2–5] | 3.5 [2.5–5.0] | 3.0 [2.0–5.0] | 0.064 |
| Post-PCI | 1.9 [0.5–3] | 1.0 [0.0–2.5] | 2.0 [0.5–3.0] | 0.051 |
| 60-min | 1 [0–2] | 0.5 [0.0–1.0] | 1.0 [0.0–2.0] | 0.005 |
| 72-h | 0.5 [0–0.5] | 0.0 [0.0–0.5] | 0.5 [0.0–1.0] | 0.001 |
| 1-month | 0.2 [0–0.1] | 0.0 [0.0–0.0] | 0.0 [0.0–0.5] | 0.003 |
| 6-months | 0.1 [0–0.1] | 0.0 [0.0–0.0] | 0.0 [0.0–0.0] | 0.013 |
| ST-segment resolution (>50%) | ||||
| Post-PCI | 121 (61%) | 72 | 49 | 0.001 |
| 60-min | 98 (49%) | 48 | 50 | 0.888 |
| 72-h | 82 (41%) | 39 | 43 | 0.666 |
| 1-month | 52 (26%) | 23 | 29 | 0.420 |
| 6-months | 16 (8%) | 7 | 9 | 0.794 |
| Newly formed branch block | ||||
| No | 175 (88%) | 87 | 88 | 1.000 |
| Transitory RBBB | 2 (1%) | 1 | 1 | 1.000 |
| Transitory LBBB | 9 (5%) | 7 | 2 | 0.172 |
| Transitory LAHB | 4 (2%) | 2 | 2 | 1.000 |
| Permanent LAHB | 5 (3%) | 1 | 4 | 0.365 |
| Permanent ILBBB | 2 (1%) | 0 | 2 | 0.477 |
| Permanent RBBB | 2 (1%) | 2 | 0 | 0.477 |
| Variables | Whole Sample (n = 200) n (%) Mean ± SD Median [IQR] | Symptoms Duration of Less than 6 h (n = 100) (%) Median [IQR] | Symptoms Duration of Less than 6 h (n = 100) (%) Median [IQR] | p Value |
|---|---|---|---|---|
| 6-months follow-up data | ||||
| In-hospital death | 1 (1%) | 0 | 1 | 1.000 |
| Out-of-hospital death | 5 (3%) | 1 | 4 | 0.365 |
| Coronary event-related death | 6 (3%) | 1 | 5 | 0.214 |
| Reinfarction of the treated vessel | 1 (1%) | 1 | 0 | 1.000 |
| Manifested HF | 28 (14%) | 6 | 22 | 0.002 |
| Stent thrombosis | 4 (2%) | 2 | 2 | 1.000 |
| Clinically manifested restenosis | 2 (1%) | 0 | 2 | 0.477 |
| Ventricular thrombus | 1 (1%) | 1 | 0 | 1.000 |
| MACE | 32 (16%) | 9 | 23 | 0.012 |
| Dual Antiplatelet Treatment | ||||
| Acetylsalicylic acid + Ticagrelor | 79 (40%) | 27 | 52 | 0.001 |
| Acetylsalicylic acid + Clopidogrel | 109 (55%) | 69 | 40 | 0.001 |
| Acetylsalicylic acid + Ticagrelor + Oral anticoagulant | 2 (1%) | 2 | 0 | 0.477 |
| Acetylsalicylic acid + Clopidogrel + Oral anticoagulant | 9 (5%) | 2 | 7 | 0.172 |
| Acetylsalicylic acid + Clopidogrel + Direct oral anticoagulant | 1 (1%) | 0 | 1 | 1.000 |
| 6-months NYHA | ||||
| 1 | 171 (86%) | 94 | 77 | 0.001 |
| 1/2 | 1 (1%) | 1 | 0 | 1.000 |
| 2 | 19 (10%) | 4 | 15 | 0.016 |
| 2/3 | 1 (1%) | 0 | 1 | 1.000 |
| 3 | 2 (1%) | 0 | 2 | 0.477 |
| 3/4 | 1 (1%) | 0 | 1 | 1.000 |
| 6-months CCS | ||||
| 0 | 174 (87%) | 86 | 88 | 0.833 |
| 1 | 13 (7%) | 8 | 5 | 0.566 |
| 2 | 6 (3%) | 4 | 2 | 0.678 |
| 3 | 2 (1%) | 1 | 1 | 1.000 |
| Echocardiographic assessment | ||||
| 6-months FSLV | 35.7 [32.7–40.0] | 35.8 [33.3–40.0] | 36.0 [32.65–39.25] | 0.572 |
| 6-months EFLV | 51.8 [48.8–57] | 55.0 [50.0–57.5] | 50.0 [45.0–55.0] | 0.001 |
| 6-months LVIDs | 3.2 [2.9–3.5] | 3.1 [2.8–3.45] | 3.2 [2.9–3.5] | 0.368 |
| 6-months LVIDd | 5.00.5 | 5.0 [4.65–5.3] | 5.0 [4.5–5.35] | 0.502 |
| 6-months LVEDV | 104.030 | 95.0 [80.0–115.0] | 101.0 [80.5–124.0] | 0.251 |
| 6-months LVESV | 52.3 [37.5–60] | 47.0 [37.0–52.5] | 50.0 [40.0–67.0] | 0.023 |
| 6-months MADd | 3.1 [3–3.2] | 3.1 [3.0–3.2] | 3.0 [2.9–3.2] | 0.183 |
| 6-months mitral regurgitation | ||||
| 0 | 54 (27%) | 28 | 26 | 0.873 |
| 0/1 | 7 (4%) | 7 | 0 | 0.021 |
| 1 | 66 (33%) | 36 | 30 | 0.452 |
| 1/2 | 29 (15%) | 9 | 20 | 0.045 |
| 2 | 25 (13%) | 13 | 12 | 1.000 |
| 2/3 | 10 (5%) | 5 | 5 | 1.000 |
| 3 | 1 (1%) | 0 | 1 | 1.000 |
| 3/4 | 2 (1%) | 1 | 1 | 1.000 |
| 6-years follow-up data | ||||
| All-cause mortality | 22 (11%) | 7 | 15 | 0.114 |
| Cardiovascular death | 14 (7%) | 6 | 8 | 0.782 |
| Non-cardiovascular death | 8 (4%) | 1 | 7 | 0.071 |
| Hospitalization due to HF | 5 (3%) | 3 | 2 | 1.000 |
| Reinfarction | 7 (4%) | 6 | 1 | 0.124 |
| Stroke | 4 (2%) | 3 | 1 | 0.614 |
| Stent restenosis | 4 (2%) | 2 | 2 | 1.000 |
| Re-PCI of the non-culprit vessel | 10 (5%) | 5 | 5 | 1.000 |
| Number of days until the first MACE | 1745 | 2287.0 [1874.0–2476.0] | 1854.0 [615.0–2208.5] | 0.001 |
| Number of days until death | 2063 | 2361.0 [2272.5–2484.0] | 1942.0 [1601.75–2260.5] | 0.001 |
| QRS Complex | MACE | Width [msec] | p Value | OR [95% CI] | p Value |
|---|---|---|---|---|---|
| Pre-PCI | No | 100 [IQR 90–105] | 0.026 | 1.028 [1.003–1.053] | 0.029 |
| Yes | 102.5 [IQR 90–110] | ||||
| Right after the procedure | No | 95 [IQR 85–100] | 0.010 | 1.026 [1.004–1.048] | 0.020 |
| Yes | 100 [IQR 90–110] | ||||
| 1-h after the procedure | No | 94 [IQR 85–100] | 0.013 | 1.027 [1.003–1.052] | 0.025 |
| Yes | 95 [IQR 85–105] | ||||
| 72 h after the procedure | No | 85 [IQR 80–95] | 0.020 | 1.031 [1.002–1.060] | 0.034 |
| Yes | 89 [IQR 80–100] | ||||
| 1 month | No | 85 [IQR 80–90] | 0.034 | 1.042 [1.012–1.074] | 0.007 |
| Yes | 85 [IQR 80–100] | ||||
| 6 months | No | 85 [IQR 80–90] | 0.041 | 1.038 [1.009–1.069] | 0.011 |
| Yes | 87.5 [IQR 80–100] |
| ST-Segment | MACE | Elevation [mm] | p Value | OR [95% CI] | p Value |
|---|---|---|---|---|---|
| Pre-PCI | No | 3 [IQR 2–5] | 0.018 | 1.61 [1.024–1.317] | 0.020 |
| Yes | 3 [IQR 2.5–5] | ||||
| Right after the procedure | No | 1 [IQR 0–2.5] | 0.001 | 1.296 [1.103–1.523] | 0.002 |
| Yes | 2 [IQR 0.75–3] | ||||
| 1-h after the procedure | No | 0 [IQR 0–1] | 0.001 | 1.391 [1.111–1.741] | 0.004 |
| Yes | 0.5 [IQR 0.5–2] | ||||
| 72 h after the procedure | No | 0 [IQR 0–5] | 0.004 | 1.500 [1.037–2.170] | 0.031 |
| Yes | 0 [IQR 0–0.75] | ||||
| 1 month | No | 0 [IQR 0–0] | 0.001 | 3.256 [1.572–6.747] | 0.001 |
| Yes | 0 [IQR 0–0.5] | ||||
| 6 months | No | 0 [IQR 0–0] | 0.003 | 2.972 [1.268–6.965] | 0.012 |
| Yes | 0 [IQR 0–0.25] |
| MACE | Odds Ratio | Std. Err. | z | P > z | [95% Conf. | Interval] |
|---|---|---|---|---|---|---|
| QRS pre-PCI | 1.027325 | 0.0129865 | 2.13 | 0.033 | 1.002185 | 1.053096 |
| CPK MB pre-PCI | 1.003071 | 0.0034383 | 0.89 | 0.371 | 0.9963542 | 1.009832 |
| TNI 0 | 1.012977 | 0.0970325 | 0.13 | 0.893 | 0.8395824 | 1.222181 |
| _cons | 0.0243191 | 0.0316703 | −2.85 | 0.004 | 0.0018943 | 0.3122134 |
| MACE | Odds Ratio | Std. Err. | z | P > z | [95% Conf. | Interval] |
|---|---|---|---|---|---|---|
| ST-segment elevation pre-PCI | 1.165351 | 0.0758857 | 2.35 | 0.019 | 1.025718 | 1.323993 |
| CPK MB pre-PCI | 1.002997 | 0.0034725 | 0.86 | 0.387 | 0.996214 | 1.009826 |
| TNI 0 | 1.0225 | 0.0989468 | 0.23 | 0.818 | 0.8458492 | 1.236043 |
| _cons | 0.0243191 | 0.0316703 | −2.85 | 0.004 | 0.0018943 | 0.3122134 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maletin, S.; Petrović, M.; Stojšić-Milosavljević, A.; Miljković, T.; Milovančev, A.; Petrović, I.; Milosavljević, I.; Balenović, A.; Čanković, M. The Role of QRS Complex and ST-Segment in Major Adverse Cardiovascular Events Prediction in Patients with ST Elevated Myocardial Infarction: A 6-Year Follow-Up Study. Diagnostics 2024, 14, 1042. https://doi.org/10.3390/diagnostics14101042
Maletin S, Petrović M, Stojšić-Milosavljević A, Miljković T, Milovančev A, Petrović I, Milosavljević I, Balenović A, Čanković M. The Role of QRS Complex and ST-Segment in Major Adverse Cardiovascular Events Prediction in Patients with ST Elevated Myocardial Infarction: A 6-Year Follow-Up Study. Diagnostics. 2024; 14(10):1042. https://doi.org/10.3390/diagnostics14101042
Chicago/Turabian StyleMaletin, Srđan, Milovan Petrović, Anastazija Stojšić-Milosavljević, Tatjana Miljković, Aleksandra Milovančev, Ivan Petrović, Isidora Milosavljević, Ana Balenović, and Milenko Čanković. 2024. "The Role of QRS Complex and ST-Segment in Major Adverse Cardiovascular Events Prediction in Patients with ST Elevated Myocardial Infarction: A 6-Year Follow-Up Study" Diagnostics 14, no. 10: 1042. https://doi.org/10.3390/diagnostics14101042
APA StyleMaletin, S., Petrović, M., Stojšić-Milosavljević, A., Miljković, T., Milovančev, A., Petrović, I., Milosavljević, I., Balenović, A., & Čanković, M. (2024). The Role of QRS Complex and ST-Segment in Major Adverse Cardiovascular Events Prediction in Patients with ST Elevated Myocardial Infarction: A 6-Year Follow-Up Study. Diagnostics, 14(10), 1042. https://doi.org/10.3390/diagnostics14101042






