Preimplantation Genetic Diagnosis of Neurodegenerative Diseases: Review of Methodologies and Report of Our Experience as a Regional Reference Laboratory
Abstract
:1. Introduction
1.1. Overview of Preimplantation Genetic Diagnosis
1.2. Current Status of PGD in Neurodegenerative Diseases
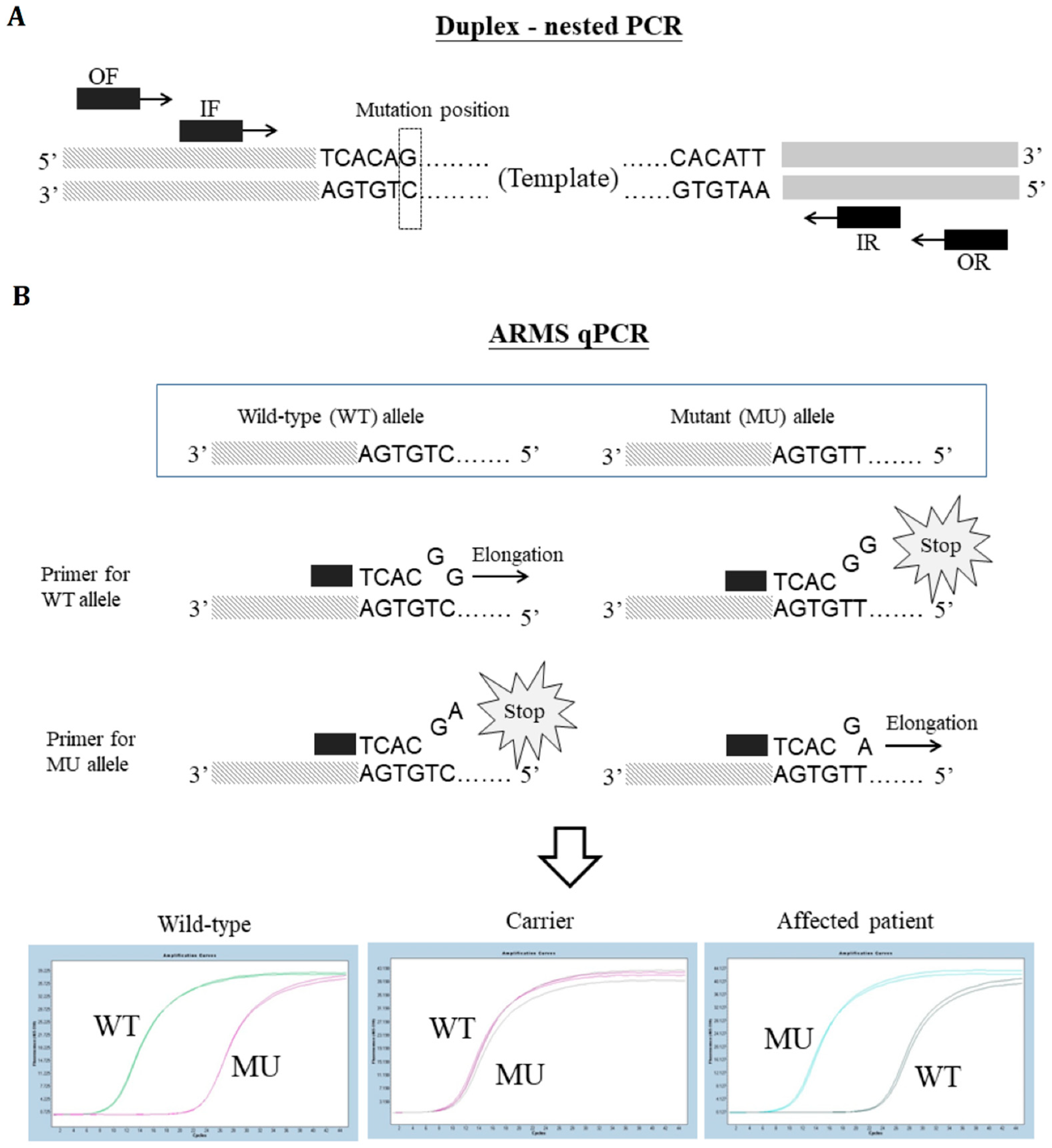
2. Materials and Methods
Genotyping Strategies Adopted in Our Laboratory
3. Results
3.1. Overall Outcomes
3.2. Example for ADO: The Index Family of FAP
4. Discussion
4.1. ADO in Our FAP PGD Case and Clinical Relevance
4.2. Ethical Aspects
4.3. Concurrent PGD/PGS
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Handyside, A.H.; Kontogianni, E.H.; Hardy, K.; Winston, R.M.L. Pregnancies from biopsied human preimplantation embryos sexed by Y-specific DNA amplification. Nature 1990, 344, 768–770. [Google Scholar] [CrossRef]
- Brezina, P.R.; Brezina, D.S.; Kearns, W.G. Preimplantation genetic testing. BMJ 2012, 345, e5908. [Google Scholar] [CrossRef] [PubMed]
- Harton, G.L.; De Rycke, M.; Fiorentino, F.; Moutou, C.; SenGupta, S.; Traeger-Synodinos, J.; Harper, J.C.; European Society for Human Reproduction and Embryology (ESHRE) PGD Consortium. ESHRE PGD consortium best practice guidelines for amplification-based PGD. Hum. Reprod. 2011, 26, 33–40. [Google Scholar] [CrossRef]
- Chen, H.-F.; Chang, S.-P.; Wu, S.-H.; Lin, W.-H.; Lee, Y.-C.; Ni, Y.-H.; Chen, C.-A.; Ma, G.-C.; Ginsberg, N.A.; You, E.-M.; et al. Validating a rapid, real-time, PCR-based direct mutation detection assay for preimplantation genetic diagnosis. Gene 2014, 548, 299–305. [Google Scholar] [CrossRef]
- Dreesen, J.; Destouni, A.; Kourlaba, G.; Degn, B.; Mette, W.C.; Carvalho, F.; Moutou, C.; Sengupta, S.; Dhanjal, S.; Renwick, P.; et al. Evaluation of PCR-based preimplantation genetic diagnosis applied to monogenic diseases: A collaborative ESHRE PGD consortium study. Eur. J. Hum. Genet. 2014, 22, 1012–1018. [Google Scholar] [CrossRef]
- Kuo, S.-J.; Ma, G.-C.; Chang, S.-P.; Wu, H.-H.; Chen, C.-P.; Chang, T.-M.; Lin, W.-H.; Wu, S.-H.; Lee, M.-H.; Hwu, W.-L.; et al. Preimplantation and prenatal genetic diagnosis of aromatic L-amino acid decarboxylase deficiency with an amplification refractory mutation system-quantitative polymerase chain reaction. Taiwan J. Obstet. Gynecol. 2011, 50, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Chang, S.-P.; Ma, G.-C.; Lin, W.-H.; Chen, H.-F.; Chen, S.-U.; Tsai, H.-D.; Tsai, F.-P.; Shen, M.-C. Preimplantation genetic diagnosis of hemophilia A. Thromb. J. 2016, 14, 768. [Google Scholar] [CrossRef]
- Chen, H.-F.; Chen, S.-U.; Ma, G.-C.; Hsieh, S.-T.; Tsai, H.-D.; Yang, Y.-S.; Chen, M. Preimplantation genetic diagnosis and screening: Current status and future challenges. J. Formos. Med. Assoc. 2018, 117, 94–100. [Google Scholar] [CrossRef]
- Sermon, K.; Goossens, V.; Seneca, S.; Lissens, W.; De Vos, A.; Vandervorst, M.; Van Steirteghem, A.; Liebaers, I. Preimplantation diagnosis for Huntington’s disease (HD): Clinical application and analysis of the HD expansion in affected embryos. Prenat. Diagn. 1998, 18, 1427–1436. [Google Scholar] [CrossRef]
- Stern, H.J.; Harton, G.L.; Sisson, M.E.; Jones, S.L.; Fallon, L.A.; Thorsell, L.P.; Getlinger, M.E.; Black, S.H.; Schulman, J.D. Non-disclosing preimplantation genetic diagnosis for Huntington disease. Prenat. Diagn. 2002, 22, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Sermon, K.; De Rijcke, M.; Lissens, W.; De Vos, A.; Platteau, P.; Bonduelle, M.; Devroey, P.; Van Steirteghem, A.; Liebaers, I. Preimplantation genetic diagnosis for Huntington’s disease with exclusion testing. Eur. J. Hum. Genet. 2002, 10, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.F.; Yeung, W.S.; Lau, E.Y.; Lam, S.T.; Tong, T.; Ng, E.H.; Ho, P.-C. Singleton birth after preimplantation genetic diagnosis for Huntington disease using whole genome amplification. Fertil. Steril. 2009, 92, 828.e7–828.e10. [Google Scholar] [CrossRef]
- Alberola, T.M.; Bautista-Llácer, R.; Fernández, E.; Vendrell, X.; Perez-Alonso, M. Preimplantation genetic diagnosis of P450 oxidoreductase deficiency and Huntington Disease using three different molecular approaches simultaneously. J. Assist. Reprod. Genet. 2009, 26, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Peciña, A.; Arana, M.D.L.; García-Lozano, J.C.; Borrego, S.; Antiñolo, G. One-step multiplex polymerase chain reaction for preimplantation genetic diagnosis of Huntington disease. Fertil. Steril. 2010, 93, 2411–2412. [Google Scholar] [CrossRef]
- Van Rij, M.C.; De Rademaeker, M.; Moutou, C.; Dreesen, J.C.F.M.; De Rycke, M.; Liebaers, I.; Geraedts, J.P.M.; Die-Smulders, C.E.M.D.; Viville, S. Preimplantation genetic diagnosis (PGD) for Huntington’s disease: The experience of three European centres. Eur. J. Hum. Genet. 2012, 20, 1010. [Google Scholar] [CrossRef]
- Perminov, D.; Voložonoka, L.; Korņejeva, L.; Jokste-Pțmane, E.; Blumberga, A.; Krasucka, S.; Seimuškina, N.; Kovaļova, I.; Fodina, V. First preimplantation genetic testing case for monogenic disease in Latvia. Gynecol. Endocrinol. 2017, 33, 47–49. [Google Scholar] [CrossRef]
- Moutou, C.; Nicod, J.-C.; Gardes, N.; Viville, S. Birth after pre-implantation genetic diagnosis (PGD) of spinocerebellar ataxia 2 (Sca2). Prenat. Diagn. 2008, 28, 126–130. [Google Scholar] [CrossRef]
- Drüsedau, M.; Dreesen, J.; De Die-Smulders, C.; Hardy, K.; Bras, M.; Dumoulin, J.; Evers, J.; Smeets, H.; Geraedts, J.; Herbergs, J. Preimplantation genetic diagnosis of spinocerebellar ataxia 3 by (CAG)n repeat detection. Mol. Hum. Reprod. 2004, 10, 71–75. [Google Scholar] [CrossRef]
- Sermon, K. PGD in the lab for triplet repeat diseases? myotonic dystrophy, Huntington’s disease and Fragile-X syndrome. Mol. Cell. Endocrinol. 2001, 183, 183. [Google Scholar] [CrossRef]
- Löfgren, A.; Vos, A.; Sermon, K.; Liebaers, I.; Van Steirteghem, A.; Van Broeckhoven, C. Preimplantation Diagnosis for Charcot-Marie-Tooth Type 1A. Ann. N. Y. Acad. Sci. 1999, 883, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Sermon, K.; De Rijcke, M.; Goossens, V.; Henderix, P.; Van Ranst, N.; Platteau, P.; Lissens, W.; Devroey, P.; Van Steirteghem, A.; Liebaers, I.; et al. Preimplantation genetic diagnosis for Charcot-Marie-Tooth disease type 1A. Mol. Hum. Reprod. 2003, 9, 429–435. [Google Scholar]
- Lee, H.-S.; Kim, M.J.; Ko, D.S.; Jeon, E.J.; Kim, J.Y.; Kang, I.S. Preimplantation genetic diagnosis for Charcot-Marie-Tooth disease. Clin. Exp. Reprod. Med. 2013, 40, 163–168. [Google Scholar] [CrossRef]
- Gui, B.; Yang, P.; Yao, Z.; Li, Y.; Liu, D.; Liu, N.; Lu, S.; Liang, D.; Wu, L. A New Next-Generation Sequencing-Based Assay for Concurrent Preimplantation Genetic Diagnosis of Charcot-Marie-Tooth Disease Type 1A and Aneuploidy Screening. J. Genet. Genom. 2016, 43, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Fallon, L.; Harton, G.L.; Sisson, M.E.; Rodriguez, E.; Field, L.K.; Fugger, E.F.; Geltinger, M.; Sun, Y.; Dorfmann, A.; Schoener, C.; et al. Preimplantation genetic diagnosis for spinal muscular atrophy type I. Neurology 1999, 53, 1087. [Google Scholar] [CrossRef]
- Pettigrew, R.; Abbs, S.; Lashwood, A.; O’Mahony, F.; Handyside, A.; Daniels, G.; Thornhill, A.; Mathew, C.; Braude, P. Six unaffected livebirths following preimplantation diagnosis for spinal muscular atrophy. Mol. Hum. Reprod. 2001, 7, 995–1000. [Google Scholar]
- Ren, Y.; Zhi, X.; Zhu, X.; Huang, J.; Lian, Y.; Li, R.; Jin, H.; Zhang, Y.; Zhang, W.; Nie, Y.; et al. Clinical applications of MARSALA for preimplantation genetic diagnosis of spinal muscular atrophy. J. Genet. Genom. 2016, 43, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Iacobelli, M.; Greco, E.; Rienzi, L.; Ubaldi, F.; Podini, D.; Nuccitelli, A.; Tesarik, J.; Baldi, M.; Fiorentino, F. Birth of a healthy female after preimplantation genetic diagnosis for Charcot’;Marie’;Tooth type X. Reprod. Biomed. 2003, 7, 558–562. [Google Scholar] [CrossRef]
- Borgulová, I.; Putzova, M.; Soldatova, I.; Stejskal, D. Preimplantation genetic diagnosis of X-linked Charcot-Marie-Tooth disease by indirect linkage analysis. Medicina Clínica (English Edition) 2018, 150, 215–219. [Google Scholar] [CrossRef]
- Uflacker, A.; Doraiswamy, P.M.; Rechitsky, S.; See, T.; Geschwind, M.; Tur-Kaspa, I. Preimplantation genetic diagnosis (PGD) for genetic prion disorder due to F198S mutation in the PRNP gene. JAMA Neurol. 2014, 71, 484–486. [Google Scholar] [CrossRef]
- Carvalho, F.; Sousa, M.; Fernandes, S.; Silva, J.; Saraiva, M.J.; Barros, A. Preimplantation genetic diagnosis for familial amyloidotic polyneuropathy (FAP). Prenat. Diagn. 2001, 21, 1093–1099. [Google Scholar] [CrossRef]
- Almeida, V.; Costa, P.; Moreira, P.; Goncalves, J.; Braga, J. Birth of two healthy females after preimplantation genetic diagnosis for familial amyloid polyneuropathy. Reprod. Biomed. 2005, 10, 641–644. [Google Scholar] [CrossRef]
- Bates, G.P.; Harper, P.S.; Jones, L. Huntington’s Disease; Oxford University Press: Oxford, UK, 2002. [Google Scholar]
- Novak, M.J.; Tabrizi, S.J. Huntington’s disease. BMJ 2010, 340, c3109. [Google Scholar] [CrossRef]
- Roos, R.A.; Roos, R.A. Huntington’s disease: A clinical review. Orphanet J. Rare Dis. 2010, 5, 40. [Google Scholar] [CrossRef]
- Araki, S.; Mawatari, S.; Ohta, M.; Nakajima, A.; Kuroiwa, Y. Polyneuritic Amyloidosis in a Japanese Family. Arch. Neurol. 1968, 18, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Ando, Y.; Araki, S.; Ando, M. Transthyretin and Familial Amyloidotic Polyneuropathy. Intern. Med. 1993, 32, 920–922. [Google Scholar] [CrossRef]
- Benson, M.D.; Uemichi, T. Transthyretin amyloidosis. Amyloid 1996, 3, 44–56. [Google Scholar] [CrossRef]
- Ando, Y.; Nakamura, M.; Araki, S. Transthyretin-Related Familial Amyloidotic Polyneuropathy. Arch. Neurol. 2005, 62, 1057–1062. [Google Scholar] [CrossRef]
- Benson, M.D. Pathogenesis of transthyretin amyloidosis. Amyloid 2012, 19, 14–15. [Google Scholar] [CrossRef]
- Hsieh, S.-T. Amyloid neuropathy with transthyretin mutations: Overview and unique Ala97Ser in Taiwan. Neurol. Taiwanica 2011, 20, 155–160. [Google Scholar]
- Yang, N.C.-C.; Lee, M.-J.; Chao, C.-C.; Chuang, Y.-T.; Lin, W.-M.; Chang, M.-F.; Hsieh, P.-C.; Kan, H.-W.; Lin, Y.-H.; Yang, C.-C.; et al. Clinical presentations and skin denervation in amyloid neuropathy due to transthyretin Ala97Ser. Neurology 2010, 75, 532–538. [Google Scholar] [CrossRef]
- Giménez, C.; Sarasa, J.; Arjona, C.; Vilamajó, E.; Martínez-Pasarell, O.; Wheeler, K.; Valls, G.; Garcia-Guixé, E.; Wells, D. Karyomapping allows preimplantation genetic diagnosis of a de-novo deletion undetectable using conventional PGD technology. Reprod. Biomed. Online 2015, 31, 770–775. [Google Scholar] [CrossRef]
- Sacks, G.C.; Altarescu, G.; Guedalia, J.; Varshaver, I.; Gilboa, T.; Levy-Lahad, E.; Eldar-Geva, T. Developmental neuropsychological assessment of 4-to 5-year-old children born following Preimplantation Genetic Diagnosis (PGD): A pilot study. Child Neuropsychol. 2016, 22, 458–471. [Google Scholar] [CrossRef]
- Schendelaar, P.; Gemert, S.L.B.-V.; Seggers, J.; Hadders-Algra, M.; Middelburg, K.; Bos, A.; Heineman, M.; Kok, J.; Heuvel, E.V.D. The effect of preimplantation genetic screening on neurological, cognitive and behavioural development in 4-year-old children: Follow-up of a RCT. Hum. Reprod. 2013, 28, 1508–1518. [Google Scholar] [CrossRef] [PubMed]
- Ando, Y.; Coelho, T.; Berk, J.L.; Cruz, M.W.; Ericzon, B.-G.; Ikeda, S.-I.; Lewis, W.D.; Obici, L.; Planté-Bordeneuve, V.; Rapezzi, C.; et al. Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J. Rare Dis. 2013, 8, 31. [Google Scholar] [CrossRef]
- Rowczenio, D.M.; Noor, I.; Gillmore, J.D.; Lachmann, H.J.; Whelan, C.; Hawkins, P.N.; Obici, L.; Westermark, P.; Grateau, G.; Wechalekar, A.D. Online Registry for Mutations in Hereditary Amyloidosis Including Nomenclature Recommendations. Hum. Mutat. 2014, 35, E2403–E2412. [Google Scholar] [CrossRef]
- Inês, M.; Coelho, T.; Conceição, I.; Duarte-Ramos, F.; De Carvalho, M.; Costa, J. Epidemiology of transthyretin familial amyloid polyneuropathy in Portugal. Orphanet J. Rare Dis. 2015, 10, P21. [Google Scholar] [CrossRef]
- Valdrez, K.; Alves, E.; Coelho, T.; Silva, S. Prevalence of use of preimplantation genetic diagnosis in Unidade Clínica de Paramiloidose from Centro Hospitalar do Porto. Acta Med. Port. 2014, 27, 710–716. [Google Scholar] [CrossRef]
- Girisha, K.; Adiga, S.; Kalthur, G.; Kumar, P. Preimplantation diagnosis of genetic diseases. J. Postgrad. Med. 2010, 56, 317. [Google Scholar] [CrossRef]
- Sengupta, S.B.; Da Delhanty, J. Preimplantation genetic diagnosis: Recent triumphs and remaining challenges. Rev. Mol. Diagn. 2012, 12, 585–592. [Google Scholar] [CrossRef]
- Wilton, L.; Goossens, V.; Moutou, C.; Budak, T.P.; Renwick, P.; De Rycke, M.; Harton, G.; Harper, J.; Traeger-Synodinos, J.; Sengupta, S.; et al. The ESHRE PGD Consortium: 10 years of data collection. Hum. Reprod. Update 2012, 18, 234–247. [Google Scholar]
- Belva, F.; Goossens, V.; Coonen, E.; De Rycke, M.; Moutou, C.; Sengupta, S.; Traeger-Synodinos, J. ESHRE PGD Consortium data collection XIII: Cycles from January to December 2010 with pregnancy follow-up to October 2011. Hum. Reprod. 2015, 30, 1763–1789. [Google Scholar]
- Robertson, J.A. Extending preimplantation genetic diagnosis: The ethical debate: Ethical issues in new uses of preimplantation genetic diagnosis. Hum. Reprod. 2003, 18, 465–471. [Google Scholar] [CrossRef]
- Zhao, M.; Chen, M.; Lee, C.G.; Chong, S.S. Identification of novel microsatellite markers <1 Mb from the HTT CAG repeat and development of a single-tube tridecaplex PCR panel of highly polymorphic markers for preimplantation genetic diagnosis of Huntington disease. Clin. Chem. 2016, 62, 1096–1105. [Google Scholar] [CrossRef]
- Treff, N.R.; Su, J.; Tao, X.; Levy, B.; Scott, R.T., Jr. Accurate single cell 24 chromosome aneuploidy screening using whole genome amplification and single nucleotide polymorphism microarrays. Fertil. Steril. 2010, 94, 2017–2021. [Google Scholar] [CrossRef]
- Schoolcraft, W.B.; Treff, N.R.; Stevens, J.M.; Ferry, K.; Katz-Jaffe, M.; Scott, R.T. Live birth outcome with trophectoderm biopsy, blastocyst vitrification, and single-nucleotide polymorphism microarray–based comprehensive chromosome screening in infertile patients. Fertil. Steril. 2011, 96, 638–640. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.; Rodrigo, L.; Mir, P.; Mateu, E.; Peinado, V.; Milán, M.; Al-Asmar, N.; Campos-Galindo, I.; Garcia, S.; Simon, C. Use of array comparative genomic hybridization (array-CGH) for embryo assessment: Clinical results. Fertil. Steril. 2013, 99, 1044–1048. [Google Scholar] [CrossRef]
- Treff, N.R.; Tao, X.; Ferry, K.M.; Su, J.; Taylor, D.; Scott, R.T. Development and validation of an accurate quantitative real-time polymerase chain reaction–based assay for human blastocyst comprehensive chromosomal aneuploidy screening. Fertil. Steril. 2012, 97, 819–824.e2. [Google Scholar] [CrossRef]
- Yang, Y.-S.; Chang, S.-P.; Chen, H.-F.; Ma, G.-C.; Lin, W.-H.; Lin, C.-F.; Tsai, F.-P.; Wu, C.-H.; Tsai, H.-D.; Lee, T.-H.; et al. Preimplantation genetic screening of blastocysts by multiplex qPCR followed by fresh embryo transfer: Validation and verification. Mol. Cytogenet. 2015, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Wells, D.; Kaur, K.; Grifo, J.; Glassner, M.; Taylor, J.C.; Fragouli, E.; Munne, S.; Grifo, J. Clinical utilisation of a rapid low-pass whole genome sequencing technique for the diagnosis of aneuploidy in human embryos prior to implantation. J. Med Genet. 2014, 51, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.-C.; Chen, H.-F.; Yang, Y.-S.; Lin, W.-H.; Tsai, F.-P.; Lin, C.-F.; Chiu, C.; Chen, M. A pilot proof-of-principle study to compare fresh and vitrified cycle preimplantation genetic screening by chromosome microarray and next generation sequencing. Mol. Cytogenet. 2016, 9, 595. [Google Scholar] [CrossRef]
- Mastenbroek, S.; Twisk, M.; Van Der Veen, F.; Repping, S. Preimplantation genetic screening: A systematic review and meta-analysis of RCTs. Hum. Reprod. Update 2011, 17, 454–466. [Google Scholar] [CrossRef]
- Goldman, K.N.; Nazem, T.; Berkeley, A.; Palter, S.; Grifo, J.A. Preimplantation Genetic Diagnosis (PGD) for Monogenic Disorders: The Value of Concurrent Aneuploidy Screening. J. Genet. Couns. 2016, 25, 1327–1337. [Google Scholar] [CrossRef]
- Zimmerman, R.S.; Jalas, C.; Tao, X.; Fedick, A.M.; Kim, J.G.; Pepe, R.J.; Northrop, L.E.; Scott, R.T., Jr.; Treff, N.R. Development and validation of concurrent preimplantation genetic diagnosis for single gene disorders and comprehensive chromosomal aneuploidy screening without whole genome amplification. Fertil. Steril. 2016, 105, 286–294. [Google Scholar] [CrossRef]
- Forman, E.J.; Hong, K.H.; Franasiak, J.M.; Scott, R.T. Obstetrical and neonatal outcomes from the BEST Trial: Single embryo transfer with aneuploidy screening improves outcomes after in vitro fertilization without compromising delivery rates. Am. J. Obstet. Gynecol. 2014, 210, 157. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.; Minasi, M.G.; Fiorentino, F. Healthy Babies after Intrauterine Transfer of Mosaic Aneuploid Blastocysts. N. Engl. J. Med. 2015, 373, 2089–2090. [Google Scholar] [CrossRef] [PubMed]
- Gleicher, N.; The International PGS Consortium Study Group; Vidali, A.; Braverman, J.; Kushnir, V.A.; Barad, D.H.; Hudson, C.; Wu, Y.-G.; Wang, Q.; Zhang, L.; et al. Accuracy of preimplantation genetic screening (PGS) is compromised by degree of mosaicism of human embryos. Reprod. Boil. Endocrinol. 2016, 14, 54. [Google Scholar]
- Orvieto, R.; Gleicher, N. Should preimplantation genetic screening (PGS) be implemented to routine IVF practice? J. Assist. Reprod. Genet. 2016, 33, 1445–1448. [Google Scholar] [CrossRef]
- Orvieto, R. Preimplantation genetic screening- the required RCT that has not yet been carried out. Reprod. Boil. Endocrinol. 2016, 14, 35. [Google Scholar] [CrossRef]
- Tortoriello, D.V.; Dayal, M.; Beyhan, Z.; Yakut, T.; Keskintepe, L. Reanalysis of human blastocysts with different molecular genetic screening platforms reveals significant discordance in ploidy status. J. Assist. Reprod. Genet. 2016, 33, 1467–1471. [Google Scholar] [CrossRef]

| Mutation Type | Disease (Gene) Examples | Genotyping Method | PGD Method |
|---|---|---|---|
| Inversion | Rare in neurodegenerative disorders | • I-PCR • Long-distance PCR • Southern blotting | • Linkage analysis |
| Point mutations | Common in neurodegenerative disorders, e.g., ALS (FUS), FAP (TTR) | • Direct DNA sequencing • DHPLC • TTGE | • ARMS-qPCR • Linkage analysis • Mini-sequencing |
| Small deletion/insertion (<1 exon) | Less common in neurodegenerative disorders, e.g., SMA (SMN1, SMN2) | • MLPA • Direct DNA sequencing | • ARMS-qPCR • Linkage analysis • Mini-sequencing • MARSALA |
| Large duplication (≥1 exon) | Charcot–Marie–Tooth 1A (PMP22) | • MLPA • High resolution aCGH | • Linkage analysis |
| Trinucleotide expansion | Very common in neurodegenerative disorders, e.g., Huntington’s disease (HTT), many subtypes of spinocerebellar ataxia (ATXN1, MJD1/ATXN3/SCA3), Poly Q diseases (ATN1, AR) | • Southern blotting • PCR followed by capillary electrophoresis (if CAG repeat number less than 100) | • Linkage analysis • Nested PCR followed by capillary electrophoresis (if CAG repeat number less than 100) |
| Disease | PGD Method | Strategy | Cycles | Pregnancy Rate Per Oocyte Retrieval | Pregnancy Rate Per Embryo Transfer | Notation | Reference |
|---|---|---|---|---|---|---|---|
| Triplet repeat expansion | |||||||
| Huntington’s disease | Single-cell PCR | Direct diagnosis | 9 | 1/153 | 1/13 | Sermon et al. [9] | |
| Huntington’s disease | Multiple-cell PCR | Direct diagnosis | 15 | NA | NA | Nondisclosure testing a | Stern et al. [10] |
| Huntington’s disease | Single-cell PCR | Exclusion testing | 7 | 1/67 | 1/6 | Sermon et al. [11] | |
| Huntington’s disease | Single-cell WGA followed by PCR | Direct diagnosis | 1 | 1/15 | 1/2 | Chow et al. [12] | |
| Huntington’s disease (and P450 oxidoreductase deficiency) | Single-cell PCR or two-cell PCR | Direct diagnosis | 2 | 0/18 | 0/2 | Alberola et al. [13] | |
| Huntington’s disease | Single-cell PCR | Direct diagnosis or exclusion testing | 7 | NA | 2/10 | Peciña et al. [14] | |
| Huntington’s disease | Single-cell PCR | Direct diagnosis or exclusion testing | 434 started; 389 continued to oocyte retrieval | 105/5218 | 105/511 | Van Rij et al. [15] | |
| Huntington’s disease | Direct diagnosis and Linkage analysis | 1 | 1/16 | 1/1 | Perminov et al. [16] | ||
| SCA2 | Single-cell PCR | Direct diagnosis | 2 | 2/36 | 2/4 | Moutou et al. [17] | |
| SCA3 | Single-cell PCR | Direct diagnosis | 1 | 1/10 | 1/2 | Drüsedau et al. [18] | |
| Huntington’s disease (and myotonic dystrophy, fragile X syndrome) | Two-cell PCR | Direct diagnosis | NA | NA | NA | No implantation data | Sermon et al. [19] |
| Gene dosage or rearrangement | |||||||
| Charcot–Marie–Tooth 1A | Single-cell PCR or two-cell PCR | Linkage analysis | 2 | 1/18 | 1/2 | Löfgren et al. [20] | |
| Charcot–Marie–Tooth 1A | Two-cell PCR | Direct diagnosis | 13 | 3/138 | 3/11 | De Vos et al. [21] | |
| Charcot–Marie–Tooth 1A | Single-cell PCR or two-cell PCR | Direct diagnosis | 6 | 4/117 | 4/13 | Lee et al. [22] | |
| Charcot–Marie–Tooth 1A | Single-cell WGA followed by NGS | Direct diagnosis | 1 | NA | NA | No implantation data | Gui et al. [23] |
| SMA 1 | Single-cell PCR or two-cell PCR | Direct diagnosis | 3 | NA | 2/7 | Fallon et al. [24] | |
| SMA | Single-cell PCR | Direct diagnosis | 5 | 6/62 | 6/9 | Daniels et al. [25] | |
| SMA | MARSALA | Direct diagnosis | 2 | NA | 1/1 | Ren et al. [26] | |
| Point mutation or small insertion/deletion | |||||||
| Charcot–Marie–Tooth X | Single-cell PCR or two-cell PCR | Direct diagnosis | 1 | 1/12 | 1/2 | Iacobelli et al. [27] | |
| Charcot–Marie–Tooth X | Single-cell WGA followed by PCR | Linkage analysis | 2 | 1/10 | 1/2 | Borgulová et al. [28] | |
| Charcot–Marie–Tooth 2F | Single-cell PCR or two-cell PCR | Direct diagnosis | 1 | 2/11 | 2/4 | Lee et al. [22] | |
| Gerstmann-Straussler- Scheinker disease | Polar body PCR | Direct diagnosis | NA | 2/14 | 2/2 | Uflacker et al. [29] | |
| FAP | Single-cell PCR | Direct diagnosis | 10 | 3/93 | 3/25 | Carvalho et al. [30] | |
| FAP | Two-cell PCR | Direct diagnosis | 1 | 2/10 | 2/3 | Almeida et al. [31] |
| Case No. | Maternal Age | Disease | Inheritance | Gene | Mutation Type | Diagnostic Methodology | No. of Oocyte Retrieval Cycle | Embryos Diagnosed | Diagnostic Result (Unaffected/Affected) | No. of Embryo Transfer Cycle (No. of Embryos Transferred) | Pregnancy Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 30–34 | SCA3 | AD | MJD1 | Trinucleotide expansion | Linkage analysis | 1 | 6 | 2/4 | 1 (2) | No implantation |
| 2 | 40–44 | SCA3 | AD | MJD1 | Trinucleotide expansion | Linkage analysis | 1 | 2 | 1/1 | 1 (1) | No implantation |
| 3 | 25–29 | SCA6 | AD | CACNA1A | Trinucleotide expansion | Linkage analysis | 2 | 12 | 6/6 | 2 (6) | No implantation |
| 4 | 35–39 | Charcot–Marie–Tooth 2E | AD | NEFL | Point mutation c.23C>G (p.P8R) | ARMS-qPCR + Linkage analysis | 2 | 13 | 8/5 | 2 (8) | No implantation |
| 5 | 25–29 | Charcot–Marie–Tooth 2E | AD | NEFL | Point mutation c.23C>G (p.P8R) | ARMS-qPCR | 1 | 7 | 5/2 | 2 (4) | Fraternal twins livebirth |
| 6 | 30–34 | Huntington’s disease | AD | HTT | Trinucleotide expansion | Linkage analysis | 1 | 1 | 1/0 | 1 (1) | No implantation |
| 7 | 30–34 | ALS | AD | FUS | Point mutation c.1562G>A (p.R521H) | ARMS-qPCR + Linkage analysis | 1 | 1 | 1/0 | 1 (1) | No implantation |
| 8 | 35–39 | FAP | AD | TTR | Point mutation c.349G>T (p.A117S ) | ARMS-qPCR + Linkage analysis | 1 | 2 | 0/2 | 0 (0) | No transfer |
| 9 * | 25–29 | FAP | AD | TTR | Point mutation c.349G>T (p.A117S ) | ARMS-qPCR | 2 | 6 | 2/4 | 1 (2) | Identical twins livebirth |
| 10 | 30–34 | SMA | AR | SMN1, SMN2 | Deletion in exon 7 of SMN1 | ARMS-qPCR + Linkage analysis | 1 | 9 | 3/6 | 1 (1) | Singleton livebirth |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, C.-H.; Chang, M.-Y.; Ma, G.-C.; Chang, S.-P.; Lin, C.-F.; Lin, W.-H.; Chen, H.-F.; Chen, S.-U.; Lee, Y.-C.; Chao, C.-C.; et al. Preimplantation Genetic Diagnosis of Neurodegenerative Diseases: Review of Methodologies and Report of Our Experience as a Regional Reference Laboratory. Diagnostics 2019, 9, 44. https://doi.org/10.3390/diagnostics9020044
Liao C-H, Chang M-Y, Ma G-C, Chang S-P, Lin C-F, Lin W-H, Chen H-F, Chen S-U, Lee Y-C, Chao C-C, et al. Preimplantation Genetic Diagnosis of Neurodegenerative Diseases: Review of Methodologies and Report of Our Experience as a Regional Reference Laboratory. Diagnostics. 2019; 9(2):44. https://doi.org/10.3390/diagnostics9020044
Chicago/Turabian StyleLiao, Chun-Hua, Ming-Yuh Chang, Gwo-Chin Ma, Shun-Ping Chang, Chi-Fang Lin, Wen-Hsiang Lin, Hsin-Fu Chen, Shee-Uan Chen, Yi-Chung Lee, Chi-Chao Chao, and et al. 2019. "Preimplantation Genetic Diagnosis of Neurodegenerative Diseases: Review of Methodologies and Report of Our Experience as a Regional Reference Laboratory" Diagnostics 9, no. 2: 44. https://doi.org/10.3390/diagnostics9020044






