Abstract
Transjugular intrahepatic portosystemic shunt (TIPS) is the most effective measure to treat complications of portal hypertension. However, liver function may deteriorate after TIPS. Predictors of liver function and outcome after TIPS are therefore important for management of TIPS patients. The study aimed to evaluate the impact of liver volume on transplant-free survival (TFS) after TIPS, as well as the evolution of liver volume and its relationship with liver function after TIPS. A retrospective analysis of all consecutive patients who underwent TIPS in a tertiary care university liver center between 2012 and 2017 (n = 216) was performed; n = 72 patients with complete prior and follow-up (FU) computed tomography (CT) imaging studies were included in the study. Volumetry of the liver was performed by a semi-automatic 9-lobe image segmentation algorithm at baseline and FU (FU 1: 90–180 d; FU 2: 180–365 d; FU 3: 365–545 d; FU 4: 545–730 d; FU 5: >730 d). Output variables were total liver volume (TLV, cm3), left liver volume (LLV, cm3), right liver volume (RLV, cm3) and TLV/body weight ratio. CT derived liver volumes were correlated with liver function tests, portosystemic pressure gradient (PPG) measurements and survival. To assess predictors of liver volume change over time we fitted linear mixed models. Kaplan–Meier analysis was performed and validated by matched pair analysis followed by Cox regression to determine independent prognostic factors for survival. The median TLV at baseline was 1507.5 cm3 (773.7–3686.0 cm3). Livers with higher baseline liver volumes and larger TLV/weight ratios retained their volume after an initial loss while smaller livers continuously lost volume after TIPS. At the first follow-up period (90–180 d post-TIPS) lower liver volumes and TLV/weight ratios were associated with higher bilirubin levels. Within the final multivariable model containing time (days since TIPS), baseline INR and baseline TLV, the average loss of liver volume was 0.74 mL per day after TIPS. Twelve-month overall transplant-free survival was 89% and median overall TFS was 33 months. The median TFS for a baseline TLV/body weight ratio > 20 was significantly higher compared with ≤20 (40.0 vs. 27.0 months, p = 0.010) while there were no differences regarding the indication for TIPS or etiology of liver disease in the matched pair analysis. Lower TLV/weight ratios before TIPS were associated with shorter TFS and should therefore be critically considered when selecting patients for TIPS. In addition, this study provides first evidence of an effect of TIPS on subsequent liver volume change and associated liver function.
1. Introduction
Transjugular intrahepatic portosystemic shunt (TIPS) is an established treatment in the management of decompensated liver cirrhosis [1,2]. Technical progress has resulted in improved patency rates and reduced TIPS-related complications [1,3,4,5]. In terms of clinical outcome, TIPS significantly prolongs transplant-free survival in advanced cirrhosis [4]. In contrast, creating a portosystemic shunt alters the intrahepatic hemodynamics and results in decreased liver perfusion with possible impairment of liver function and progression to liver failure [5]. In cirrhotic patients, loss of liver volume leads to deterioration of liver function and is a poor prognostic factor in cirrhosis [6,7]. However, liver volume has not yet been extensively studied as a potential variable being suitable for patient selection for TIPS [6]. Despite a thorough patient evaluation proceeding for TIPS, including assessment of the Model of End-Stage Liver Disease (MELD), Freiburg index of post-TIPS survival (FIPS), Child–Pugh score and hepatic encephalopathy, there are still challenges regarding the appropriate workup for TIPS patients [8]. The assessment of the liver volume using pre-procedural imaging may have a predictive value for post-TIPS liver function and outcome and hence might be a simple tool in patient selection to optimize the balance of detriments and benefits achieved by TIPS [6,9,10,11].
CT volumetry is increasingly established in current clinical practice to accurately determine the liver volume [12]. It is useful in the pre-operative planning of major resection or living donor liver transplantation where the technical and clinical success depend on the future remnant liver volume or liver graft size in transplantation [12,13,14]. However, there are only limited data regarding the imaging-based assessment of liver volume in the patient selection for TIPS and follow-up of TIPS patients [6,15].
The aim of our study was to evaluate the impact of liver volume on transplant-free survival (TFS) after TIPS. Additionally, we aimed to analyze the evolution of liver volume and its relationship with liver function after TIPS.
2. Results
2.1. Patient Characteristics
Seventy-two patients with complete baseline and follow-up data were included in the study (median age, y: 60 (21–81); n = 40 male, n = 32 female; Table 1). The most frequent etiology of liver disease was alcohol abuse. Liver disease was mostly frequently graded as Child–Pugh B. Mean MELD-score pre-TIPS was 11.7 (SD: 6.0). Refractory ascites was the most frequent indication of TIPS insertion.

Table 1.
Patient characteristics.
2.2. Development of Liver Function and Liver Volume following TIPS Insertion
Liver function parameters and portal pressure gradient (PPG) were recorded at baseline before TIPS and modelled as repeated measurements after TIPS procedure at five follow-up (FU) periods. As shown in Table 2, there were significant alterations over time: MELD-score (p = 0.0224), bilirubin (p = 0.0008), International Normalized Ratio (INR, p = 0.0125), lactate dehydrogenase (LDH, p = 0.0004), quick (p = 0.0057) and sodium (p = 0.0003).

Table 2.
Baseline and follow-up laboratory data and pressure measurements regarding TIPS insertion (mean (SD)).
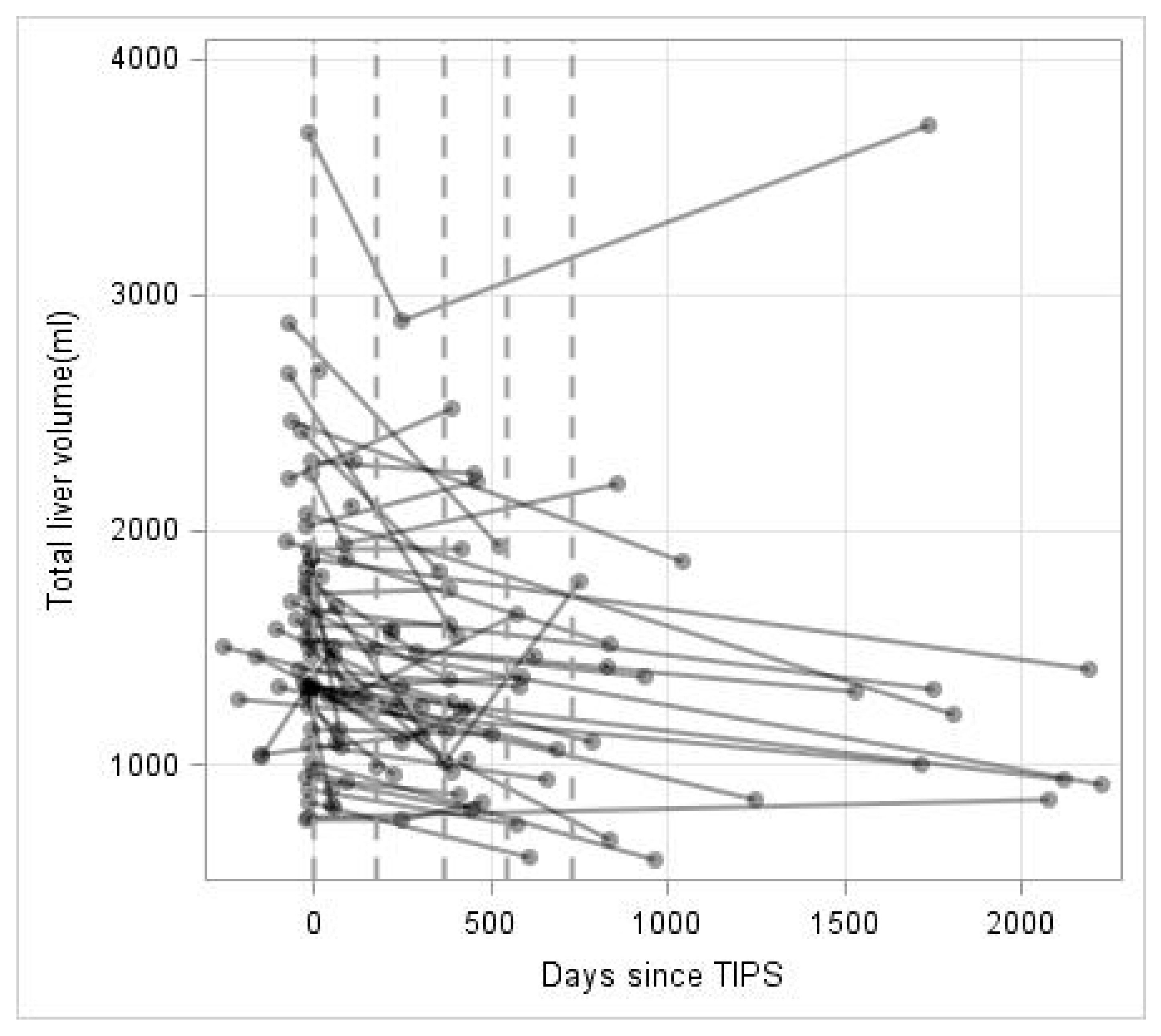
The median pre-procedural TLV was 1507.5 cm3 (773.7–3686.0 cm3). Figure 1 shows the liver volume data over time of all patients included. The dashed lines indicate the follow-up periods used for the descriptive analysis of the data indicating an overall TLV loss over time. Repeated volume measurements were analyzed for TLV, LLV, RLV and TLV/weight ratio. As shown in Table 3, TLV (p < 0.0001), LLV (p = 0.0102), RLV (p < 0.0001) and TLV/weight ratio (p < 0.0001) revealed significant decreases over time. The TLV reduced on average by 10.7% within the first 365 days. LLV and RLV reduced by 14.1% and 9.5%, respectively. The TLV/weight ratio decrease by 15.8% within the first 365 days after TIPS.
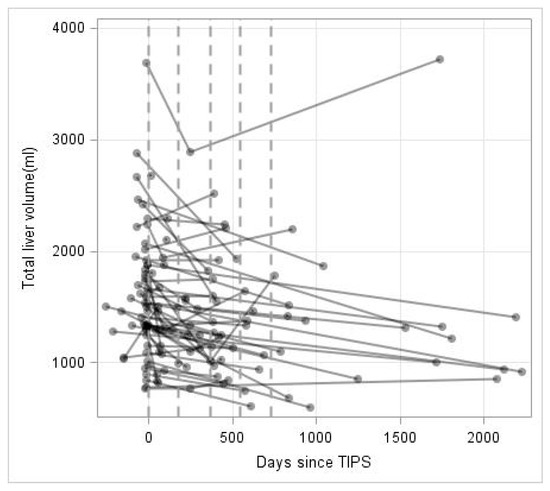
Figure 1.
Total liver volume (mL) over time (in days). Repeated measurements are connected by lines for each patient. Discretized assessment periods are marked by dashed vertical lines.

Table 3.
Baseline and follow-up liver volume regarding TIPS insertion (mean (SD)).
Next, we aimed to analyze liver function after TIPS in relation to liver volume. As shown in Table 4, a correlation analysis of total liver volume at the first follow-up period (FU1: 90–180 d) showed a negative correlation between TLV and bilirubin (Spearman correlation: −0.44, p = 0.03839), indicating that lower liver volume is associated with higher bilirubin levels. All other parameters could not be proved to be correlated with TLV. Similarly, TLV/weight ratio and bilirubin were correlated (Spearman correlation: −0.46, p = 0.04249), while TLV/weight ratio and the MELD-score showed a correlation descriptively (Spearman correlation: −0.42, p = 0.0649). None of the remaining parameters showed a significant correlation with TLV/weight ratio.

Table 4.
Correlation analysis of total liver volume (TLV) and liver function at first follow-up period (FU1: 90–180 d).
2.3. Analysis of Predictors for the Development of Liver Volume after TIPS
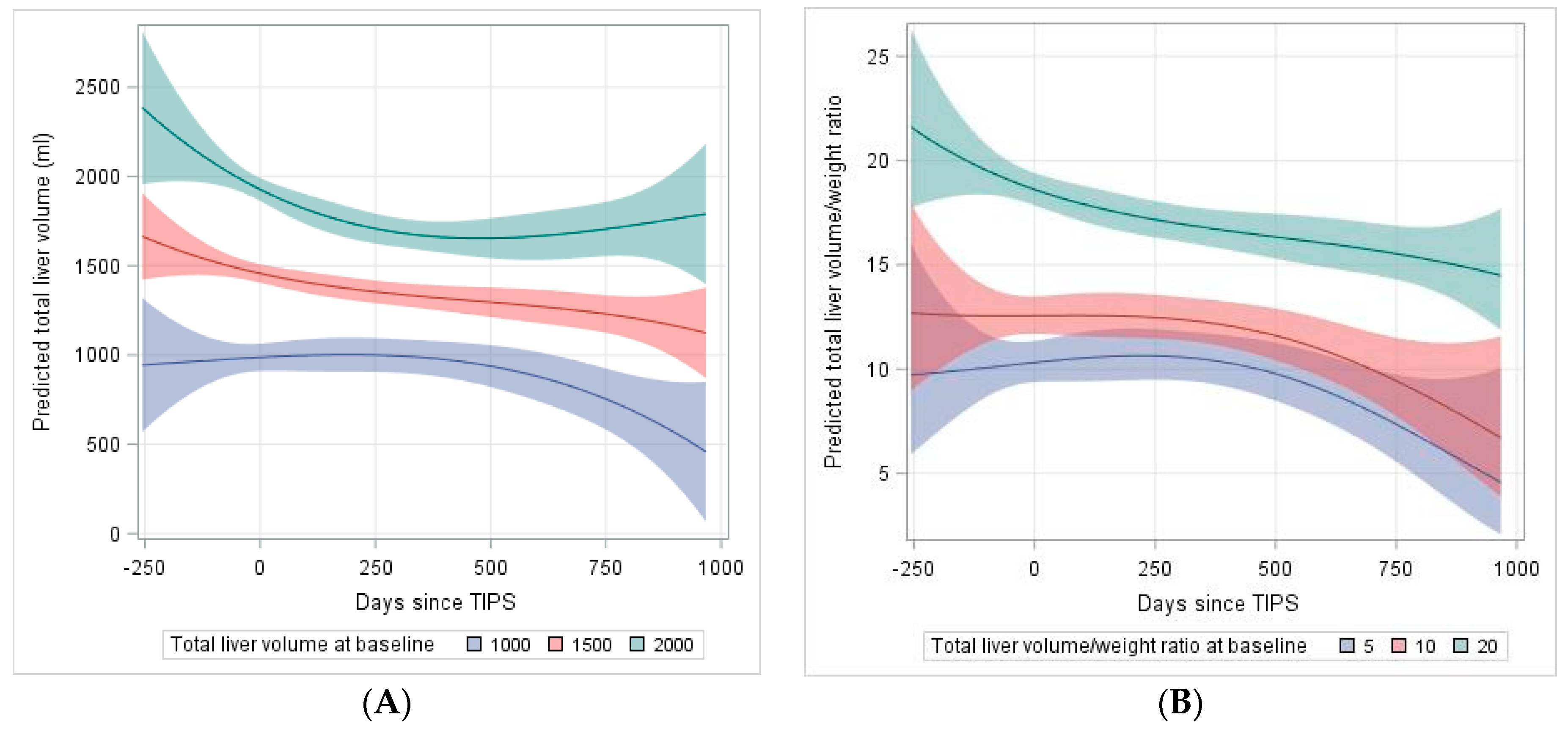
Based on follow-up liver volume data indicating an overall TLV loss over time, we fitted linear mixed models which predict the development of TLV following TIPS. As shown in Table 5, this model reveals a loss in TLV over time depending on the preexisting volume before TIPS (coefficient = −0.00143, interaction p = 0.0251). The higher order (Time3 × volume) term also shows a minor but significant contribution to the volume over time (coefficient = −14 × 10−11, interaction p ≤ 0.0001). Based on this model, Figure 2A presents the predicted means for the TLV over time and its course for three selected baseline volumes (1000 mL, 1500 mL, 2000 mL) over the first 1000 days after TIPS. While livers with higher baseline volumes retain their volume after initial loss, smaller livers (below 1500 mL) continue to lose volume over time. Similarly, we fitted a polynomial model for the TLV/weight ratio. Here, the time × TLV/weight ratio (before TIPS) does not show a significant decline over time, while the higher order terms indicate a decrease of TLV/weight ratio over time (Table 6) as shown in Figure 2B for three selected baseline ratios (5, 10, 20) over the first 1000 days after TIPS.

Table 5.
Regression coefficients for the explorative model for total liver volume and time.
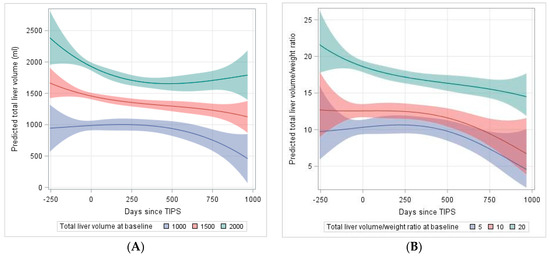
Figure 2.
Explorative model of (A) total liver volume pre-TIPS to 1000 days after TIPS dependent on baseline liver volume (B) total liver volume (TLV)/weight ratio pre-TIPS to 1000 days after TIPS dependent on baseline TLV/weight ratio. TIPS was applied at day 0. The underlying models are polynomial models of 3rd degree including interactions with baseline volume (volume/weight ratio).

Table 6.
Regression coefficients for the explorative model for TLV/weight ratio and time.
Finally, we aimed to develop a multivariable linear mixed model to stratify TIPS patients based on their baseline TLV and the expectable development of TLV following TIPS using routinely available clinical variables. From all available baseline variables, candidates were selected from the univariate analysis (p < 0.2). Here, age (interaction with time p = 0.0095), Alanine Aminotransferase (ALT, interaction p = 0.0479), INR (interaction p = 0.068), and baseline TLV (p < 0.0001) emerged as prominent covariates. A multivariable model was learned by backward selection by Schwarz Bayesian Criterion (SBC). The final multivariable model contains time (days since TIPS placement), INR before TIPS, the interaction between INR before TIPS and time, and finally baseline TLV. All factors were significantly associated with TLV after TIPS (all p < 0.02, Table 7). Within this multivariable model the average loss of liver volume is −0.74 mL/day (95%CI: −1.09 mL/day to −0.40 mL/day).

Table 7.
Regression coefficients for the final multivariate model for the development of total liver volume.
2.4. Survival Analysis Regarding Baseline Liver Volumes
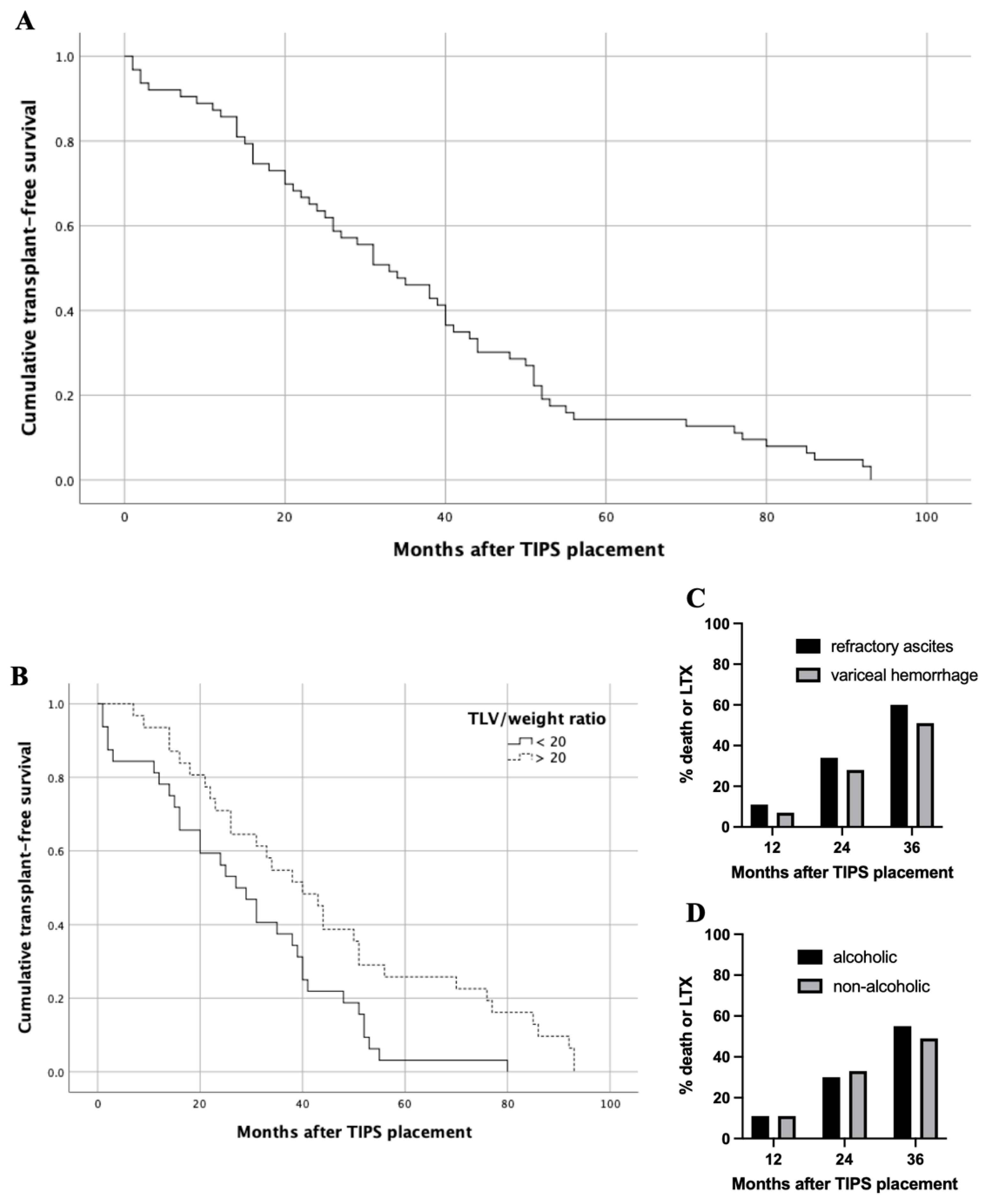
Twelve-month TFS was 89% and median TFS was 33 months (Figure 3A). The median TFS for a baseline TLV/weight ratio > 20 was significantly higher compared with ≤ 20 (40.0 vs. 27.0 months, p = 0.010; Figure 3B). There were no significant differences in the median survival regarding baseline TLV, LLV and RLV. Due to significant differences in the TFS regarding the TLV/weight ratio, we performed a matched pair analysis to validate the predictive value. Matched patients were sub-divided based on indication for TIPS (refractory ascites vs. variceal hemorrhage) or etiology of liver disease (alcoholic vs. non-alcoholic). Here, subgroup survival analysis revealed no significant differences concerning indication for TIPS or the etiology of liver cirrhosis in the matched pair cohort (Figure 3C,D).
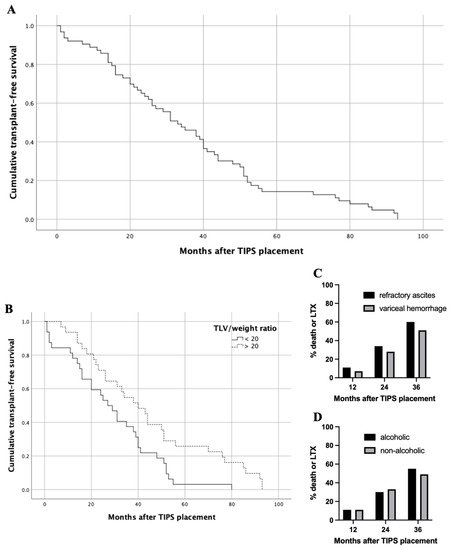
Figure 3.
Transplant-free survival (TFS) of entire patient cohort and subgroup analysis. (A) Twelve-month overall TFS was 89% and median overall TFS was 33 months. (B) Median TFS for a baseline TLV/body weight ratio > 20 was significantly higher compared with <20 (40.0 vs. 27.0 months, p = 0.010). (C) In matched pair analysis regarding indication for TIPS; death or LTX occurred in 11%, 34% and 60% of patients with refractory ascites 12, 24 and 36 months after TIPS, respectively. Death or LTX occurred in 7%, 28% and 51% of patients with variceal hemorrhage 12, 24 and 36 months after TIPS, respectively; (each p-value at 12, 24 and 36 months > 0.05). (D) In matched pair analysis regarding etiology of liver cirrhosis; death or LTX occurred in 11%, 30% and 55% of patients with alcoholic history 12, 24 and 36 months after TIPS, respectively. Death or LTX occurred in 11%, 33% and 49% of patients with non-alcoholic history 12, 24 and 36 months after TIPS, respectively; (each p-value at 12, 24 and 36 months > 0.05). Abbreviations: TIPS, transjugular intrahepatic portosystemic shunt; LTX, liver transplantation.
Model selection for multivariate analysis of baseline liver volume parameters on survival was done by forward and backward elimination with a threshold of p < 0.15 for inclusion in the model. A baseline TLV/weight ratio > 20 was found to be an independent parameter with a significant influence on survival (p = 0.013).
3. Discussion
Various models have been developed to predict survival after TIPS, while the MELD-score is one of the most commonly used in clinical routine [5,6,16,17,18,19]. Careful patient selection proceeding for TIPS continues to be a clinical challenge as some patients may develop post-TIPS liver failure despite initially preserved liver function as determined by laboratory data [6]. TIPS insertion alters the intrahepatic hemodynamics while the reduced portal blood supply may affect the reserve volume and function of the liver [20]. In cirrhotic patients, the decreasing liver volume results in an impairment of liver function while a smaller liver is a poor prognostic factor in cirrhosis [5,8]. Although imaging-based assessment of the liver volume is increasingly established in the workup before major liver resection or living donor liver transplantation, there is only limited data about a possible predictive value of the liver volume on post-TIPS morbidity and mortality [6,7,12,13,15]. Further on, liver volume can thereby be determined non-invasively in a stable and reproducible manner by cross sectional imaging with an accuracy of up to 5%, whereas the parameters for calculating the established Child- or MELD-scores tend to vary to a higher degree [6,21].
Within this study, we included n = 72 patients who underwent TIPS with a median pre-procedural TLV of 1507.5 cm3. In a recent study, however with older data, Lopera et al. performed a retrospective review of 80 patients, analyzing the effect of liver volume on morbidity and mortality after TIPS [6]. In a comparable manner, Lopera et al. performed a software-based semi-automatic CT- or MRI-based assessment of TLV before TIPS with comparable baseline data (median TLV: 1420 cm3), while they did not provide an analysis of liver volume subgroups and liver volume development [6]. Within their cohort, Lopera et al. were not able to identify a possible impact of pre-TIPS TLV on self-defined major adverse events (hepatic encephalopathy requiring hospital admission, increase in >2 points in MELD-score > 18 points, need for emergent liver transplantation and/or death) [6]. Within the patient cohort of Lopera et al., n = 15 (19%) deaths occurred within 6 months after TIPS, whereas no significant associations were found when modelling TLV in terms of death [6]. In contrast, in our study only n = 5 (7%) deaths occurred within the 6-month follow-up. We were able to show that median TFS for a baseline TLV/weight ratio > 20 was significantly higher compared with ≤20 (40.0 vs. 27.0 months). These results were validated in a matched pair analysis without differences regarding the indication for TIPS and etiology of liver disease. Beyond that baseline TLV/weight ratio > 20 was found to be an independent parameter with a significant influence on survival.
In another single-center study, Cohen et al. described n = 68 patients with cross sectional imaging (CT/MRI) liver volumetry prior to TIPS placement [15]. In contrast to our study, liver volume assessment was based on linear hepatic measurements, which is not as accurate as the segmentation method used in our study. The overall 30-day mortality of 13.2% was notably raised compared to our cohort (30-day mortality: 3%) [15]. Cohen et al. were able to delineate a smaller baseline liver volume as a predictive factor for increased 30-day mortality after TIPS creation [15].
To the best of our knowledge there are no comparable studies analyzing the development of liver volume after TIPS. Based on follow-up liver volume data after TIPS, we fitted a model which predicts the development of the TLV over time. Livers with higher baseline volumes or larger TLV/weight ratios retain their volume after initial loss, suggesting that in these cases the liver still seems to have some compensatory capacity after initial volume loss. In contrast, smaller livers (below 1500 mL) or lower TLV/weight ratios (below 20) continue to lose volume over time and revealed higher bilirubin levels in the follow-up, indicating that the small liver is more likely to have their reserve volume and thus their synthesis function compromised. Thus, baseline liver volume and TLV/weight ratio should be considered as important parameters for patient selection in TIPS. In particular, patients with a low liver volume (<1500 mL) should be evaluated particularly carefully, as low liver volume is potentially associated with poorer outcome as shown here for the baseline TLV/weight ratio < 20.
Moreover, we were able to develop a model to stratify TIPS patients based on their baseline TLV and the expectable development of TLV following TIPS using routinely available clinical variables. Before integration of this mathematical model prediction into routine workflows, further analyses are needed to establish a more standardized approach in the future. Nevertheless, combining a CT liver volume score as a simple, non-invasive tool with the assessment of the MELD, FIPS and Child–Pugh score and/or hepatic encephalopathy might optimize patient selection for TIPS.
This study is limited by its retrospective study design, including a potential selection bias. Future studies should include our suggested CT volumetry protocol to validate the prospective value of TLV in patients undergoing TIPS.
In conclusion, this study provides first evidence of an impact of TIPS placement on subsequent liver volume development and accompanied liver function. Baseline TLV/weight ratio before TIPS has a substantial effect on the transplant-free survival which has to be considered when selecting patients for shunt procedures.
4. Materials and Methods
4.1. Study Design and Patient Selection
The study was performed as a retrospective single-center observational trial in a tertiary care university liver center. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and the study protocol was approved by the local ethics committee (No.: 2016-046-f-S). Due to the retrospective character of this trial, informed consent was waived. The clinical data of all patients who underwent TIPS between 2012 and 2017 (n = 216) were collected; n = 144 patients had to be excluded due to missing baseline or follow-up CT imaging (n = 134) or liver transplantation (n = 10) before first routine follow-up following TIPS. Finally, n = 72 patients were included in the study.
4.2. TIPS Procedure
TIPS creation was performed according to standard operating procedures under general anesthesia. Single-shot antibiosis was performed by administration of 2 g ceftriaxone intravenously. All TIPS procedures were performed using PTFE-covered Stents (Viatorr, Gore Inc., Flagstaff, AZ, USA). Initially, shunts were sized to 8 mm diameter; in cases of insufficient reduction of the portosystemic gradient (>12 mmHg) the shunt lumen was further increased to 9 and 10 mm. Afterward, TIPS patients routinely were put on 100 mg acetylsalicylate acid daily for life.
4.3. Data Collection and Follow-Up
All patient and procedural data including laboratory parameters, invasive portosystemic pressure gradient measurements and follow-up/survival data were retrospectively acquired from the electronic patient records as well as from the Picture Archiving and Communications System (PACS). The electronic patient records were reviewed for baseline patient characteristics including age, sex, etiology of liver disease, Child’s grade and indication for TIPS insertion. Laboratory data and invasive portosystemic pressure gradient measurements were obtained, and MELD-score was calculated at baseline (n = 72) and 5 follow-up (FU) periods after TIPS procedure (FU 1: 90–180 d, n = 32; FU 2: 180–365 d, n = 36; FU 3: 365–545 d, n = 26; FU 4: 545–730 d, n = 10; FU 5: >730 d, n = 19). Survival data was collected until death, until liver transplantation or until end of follow-up. Transplant-free survival (TFS) was defined as survival free of death of any cause or until liver transplantation.
4.4. Imaging-Based Volumetry of the Liver
Volumetry of the liver was performed on contrast enhanced CT using commercial software (IntelliSpace Portal, Philips, Best, The Netherlands). This algorithm calculates the liver volume based on semi-automatic 9-lobe image segmentation [22]. Output variables were total liver volume (TLV, cm3), left liver volume (LLV, cm3) including segments I-III, right liver volume (RLV, cm3) including segments IV-VIII, and TLV/weight ratio calculated as ().
4.5. Statistics
Demographic parameters and liver volumes at baseline and during follow-up were reported as total number and percentage, mean and standard deviation or median and range or 95% confidence interval (CI), as appropriate. Analysis of variance was used to analyze the statistical significance for two paired/unpaired groups or multiple groups. Spearman’s correlation coefficient was used to assess correlation between liver volume and liver function. To assess predictors of liver volume over time we fitted linear mixed models. Each model contained the potential predictor variables as main effect and the interaction between predictor and assessment time. Repeated measurements were modelled in the random effect by using a power covariance structure. At first, univariate interaction models were fitted for each predictor variable. In a second step a multivariable model was fitted, using the predictor variables with a Wald-test p-value < 0.2 within a backward selection procedure. Kaplan–Meier analysis and log-rank test were performed to analyze survival. For validation analysis we performed a matched pair analysis. Propensity score matching was performed via logistic regression to create a propensity score for each patient, entering the following variables: age, sex, MELD-score and TLV/weight ratio. Matched patients were sub-divided based on indication for TIPS (refractory ascites vs. variceal hemorrhage) or etiology of liver disease (alcoholic vs. non-alcoholic) with a ratio of 2:1 (n = 28:n = 14) or 1:1 (n = 36:n = 36), respectively. A matched pair between the subgroups was obtained by use of nearest-neighborhood-matching using a caliper width of 0.2 without replacement as described elsewhere [23]. Model selection for multivariate analysis of baseline liver volumes on survival was done by using Cox regression with forward and backward elimination with a threshold of p < 0.15 for inclusion in the model; p-values < 0.05 were considered to be statistically significant. Statistical analysis was performed using the SPSS Statistics version 26 (SPSS Inc., Chicago, IL, USA) and SAS/STAT software version 9.4 (SAS Institute Inc., Cary, NC, USA).
Author Contributions
Conceptualization, P.S., M.W., H.H.; Data curation, P.S., A.R.; Formal analysis, P.S.; Investigation, P.S., A.R.; Methodology, P.S., M.M. (Max Masthoff), L.L.S.; Project administration, C.W., H.H., J.T., M.W.; Resources, H.H., J.T., M.W.; Software, D.G., J.K.; Supervision, M.K., M.S., H.H., J.T., M.W.; Validation, M.K., M.S.; Visualization, P.S., D.G., J.K.; Writing—original draft, P.S., A.R.; Writing—review and editing, D.G., J.K., M.M. (Miriam Maschmeier), M.M. (Max Masthoff), L.L.S., C.W., M.K., M.S., H.H., J.T., M.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and the study protocol was approved by the local ethics committee (No.: 2016-046-f-S).
Informed Consent Statement
Due to the retrospective character of this trial, informed consent was waived.
Data Availability Statement
Analyzed data are stored at the Clinic for Radiology, University Hospital in Muenster, Germany, and are available on reasonable request if not already included in this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hung, M.L.; Lee, E.W. Role of Transjugular Intrahepatic Portosystemic Shunt in the Management of Portal Hypertension: Review and Update of the Literature. Clin. Liver Dis. 2019, 23, 737–754. [Google Scholar] [CrossRef]
- Russo, M.W.; Sood, A.; Jacobson, I.M.; Brown, R.S. Transjugular intrahepatic portosystemic shunt for refractory ascites: An analysis of the literature on efficacy, morbidity, and mortality. Am. J. Gastroenterol. 2003, 98, 2521–2527. [Google Scholar] [CrossRef]
- Perry, B.C.; Kwan, S.W. Portosystemic Shunts: Stable Utilization and Improved Outcomes, Two Decades after the Transjugular Intrahepatic Portosystemic Shunt. J. Am. Coll. Radiol. 2015, 12, 1427–1433. [Google Scholar] [CrossRef]
- Bureau, C.; Thabut, D.; Oberti, F.; Dharancy, S.; Carbonell, N.; Bouvier, A.; Mathurin, P.; Otal, P.; Cabarrou, P.; Péron, J.M.; et al. Transjugular Intrahepatic Portosystemic Shunts with Covered Stents Increase Transplant-Free Survival of Patients with Cirrhosis and Recurrent Ascites. Gastroenterology 2017, 152, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Horhat, A.; Bureau, C.; Thabut, D.; Rudler, M. Transjugular intrahepatic portosystemic shunt in patients with cirrhosis: Indications and post-transjugular intrahepatic portosystemic shunt complications in 2020. United Eur. Gastroenterol. J. 2020, 9, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Lopera, J.E.; Speeg, K.V.; Young, C.; Garg, D.; Michalek, J.; Chen, Y.; Kroma, G.; Suri, R.; Garza-Berlanga, A. Effect of liver volume in morbidity and mortality after elective transjugular intrahepatic portosystemic shunt. Gastrointest. Interv. 2014, 3, 93–97. [Google Scholar] [CrossRef][Green Version]
- Schiano, T.D.; Bodian, C.; Schwartz, M.E.; Glajchen, N.; Min, A.D. Accuracy and significance of computed tomographic scan assessment of hepatic volume in patients undergoing liver transplantation. Transplantation 2000, 69, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Vizzutti, F.; Schepis, F.; Arena, U.; Fanelli, F.; Gitto, S.; Aspite, S.; Turco, L.; Dragoni, G.; Laffi, G.; Marra, F. Transjugular intrahepatic portosystemic shunt (TIPS): Current indications and strategies to improve the outcomes. Intern. Emerg. Med. 2020, 15, 37–48. [Google Scholar] [CrossRef]
- Tu, R.; Xia, L.P.; Yu, A.L.; Wu, L. Assessment of hepatic functional reserve by cirrhosis grading and liver volume measurement using CT. World J. Gastroenterol. 2007, 13, 3956–3961. [Google Scholar] [CrossRef]
- Saygili, O.B.; Tarhan, N.C.; Yildirim, T.; Serin, E.; Ozer, B.; Agildere, A.M. Value of computed tomography and magnetic resonance imaging for assessing severity of liver cirrhosis secondary to viral hepatitis. Eur. J. Radiol. 2005, 54, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Kok, B.; Abraldes, J.G. Patient Selection in Transjugular Intrahepatic Portosystemic Shunt (TIPS) for Refractory Ascites and Associated Conditions. Curr. Hepatol. Rep. 2019, 18, 197–205. [Google Scholar] [CrossRef]
- Lim, M.C.; Tan, C.H.; Cai, J.; Zheng, J.; Kow, A.W.C. CT volumetry of the liver: Where does it stand in clinical practice? Clin. Radiol. 2014, 69, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Kishi, Y.; Abdalla, E.K.; Chun, Y.S.; Zorzi, D.; Madoff, D.C.; Wallace, M.J.; Curley, S.A.; Vauthey, J.N. Three hundred and one consecutive extended right hepatectomies: Evaluation of outcome based on systematic liver volumetry. Ann. Surg. 2009, 250, 540–547. [Google Scholar] [CrossRef]
- Wei, L.; Zhu, Z.J.; Lü, Y.; Jiang, W.T.; Gao, W.; Zeng, Z.G.; Shen, Z.Y. Application of computer-assisted three-dimensional quantitative assessment and a surgical planning tool for living donor liver transplantation. Chin. Med. J. 2013, 126, 1288–1291. [Google Scholar] [PubMed]
- Cohen, B.; Tafreshi, S.; Tafreshi, R.; Cantos, A. Abstract No. 582 Liver volume as a predictor of 30-day mortality post transjugular intrahepatic portosystemic shunt (TIPS) creation. J. Vasc. Interv. Radiol. 2019, 30, S251. [Google Scholar] [CrossRef]
- García-Pagán, J.C.; Saffo, S.; Mandorfer, M.; Garcia-Tsao, G. Where does TIPS fit in the management of patients with cirrhosis? JHEP Rep. 2020, 2, 100122. [Google Scholar] [CrossRef] [PubMed]
- Ronald, J.; Wang, Q.; Choi, S.S.; Suhocki, P.V.; Hall, M.D.; Smith, T.P.; Kim, C.Y. Albumin-bilirubin grade versus MELD score for predicting survival after transjugular intrahepatic portosystemic shunt (TIPS) creation. Diagn. Interv. Imaging 2018, 99, 163–168. [Google Scholar] [CrossRef]
- Zhou, C.; Hou, C.; Cheng, D.; Tang, W.; Lv, W. Predictive accuracy comparison of meld and child-turcotte-pugh scores for survival in patients underwent TIPS placement: A systematic meta-analytic review. Int. J. Clin. Exp. Med. 2015, 8, 13464–13472. [Google Scholar] [PubMed]
- Bettinger, D.; Sturm, L.; Pfaff, L.; Hahn, F.; Kloeckner, R.; Volkwein, L.; Praktiknjo, M.; Lv, Y.; Han, G.; Huber, J.P.; et al. Refining prediction of survival after TIPS with the novel Freiburg index of post-TIPS survival. J. Hepatol. 2021, 74, 1362–1372. [Google Scholar] [CrossRef]
- Yao, X.; Zhou, H.; Huang, S.; Tang, S.H.; Qin, J.P. Effects of transjugular intrahepatic portosystemic shunt using the Viatorr stent on hepatic reserve function in patients with cirrhosis. World J. Clin. Cases 2021, 9, 1532–1542. [Google Scholar] [CrossRef]
- Vauthey, J.N.; Chaoui, A.; Do, K.A.; Bilimoria, M.M.; Fenstermacher, M.J.; Charnsangavej, C.; Hicks, M.; Alsfasser, G.; Lauwers, G.; Hawkins, I.F.; et al. Standardized measurement of the future liver remnant prior to extended liver resection: Methodology and clinical associations. Surgery 2000, 127, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Zahel, T.; Wildgruber, M.; Ardon, R.; Schuster, T.; Rummeny, E.J.; Dobritz, M. Rapid assessment of liver volumetry by a novel automated segmentation algorithm. J. Comput. Assist. Tomogr. 2013, 37, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm. Stat. 2011, 10, 150–161. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).