Abstract
In this work, MgAl-layered double hydroxides (LDH) were synthesized by co-precipitation method using a colloid mill and characterized by XRD and SEM. It was found that the environmentally friendly LDHs had greater performance than the traditional antioxidant. By adding LDHs into large electric shovel grease (GRK-A) in open-pit coal mine, the service lifetime of grease was extended by 20%. With the increase in LDH addition, the grease sample attains greater activation energy, and the thermal oxidation and decomposition resistance become stronger. Comparing the energy storage modulus and flow transition index at different temperatures, adding the right amount of LDHs needs close attention for the system oxidation resistance and viscoelasticity. For the electric shovel grease, the best oxidation resistance and rheological properties can be achieved by adding 2% of LDHs. The rheological viscosity–temperature curves show that the grease samples with different ratios of solid LDHs have better low-temperature properties than the mine grease. This work outlines a simple method for creating an environmentally sustainable lubricant additive with the use of LDH. LDH serves as a novel inorganic antioxidant additive that is optimal for open gear lubrication and sliding friction.
1. Introduction
Lubricating grease is a colloid-dispersed system in which base stock (mineral or synthetic oil) is trapped in a three-dimensional network of thickener [1]. Grease has obvious advantages over lubricating oil in terms of load-bearing capacity, sealing, and corrosion prevention because grease does not flow away from the contact surface [2,3]. Friction and wear are inevitable in daily production and life, which is also the main cause of mechanical failure, and grease can play a role in reducing friction and resistance, thus improving the normal operation and durability of mechanical parts [4,5,6]. Antioxidant is a key additive to protect lubricating oil from oxidative deterioration. At present, at least one antioxidant is added to industrial lubricating oil to improve its oxidation stability or enhance other properties, so as to meet the harsh requirements of lubricating oil in automotive, precision bearings, construction machinery, and other industrial fields [7,8]. Nowadays, countries around the world pay more and more attention to environmental protection and green development, and green chemical industry has become the trend in global chemical industry development. In recent years, due to environmental and resource problems, the promotion of new antioxidants that are green, low-cost, and high-efficiency has been the development trend of antioxidants, but there are few reports of green antioxidants that actually realize industrial applications [9,10,11,12].
Although additives represent a relatively small percentage of the total lubricant formulation, they play a critical role in reducing friction and wear, corrosion inhibition, and preventing oxidation. Common additives, such as ZDDP and molybdenum dialkyl dithiocarbamate (MoDTC), have been shown to have a synergistic effect on the performance of paraffinic mineral oils [13], effectively reducing friction and wear [14]. To improve the oxidative stability of biodiesel, Zhang et al. [15] prepared a bifunctional polymethacrylate (PMA)-type copolymer by grafting an amine antioxidant, 1-phenylnaphthylamine, onto PMA. The induction period (IP) of B20 (20 vol% soybean biodiesel + 80 vol% diesel fuel) treated with the 2000 ppm graft copolymer was prolonged by 17 h compared to pure B20. Majano et al. [16] accelerated oxidation experiments using pure base oil and base oil in the presence of LTL molecular sieves. The LTL molecular sieves showed the potential to slow down the oxidation rate of the oil and also resulted in a lower production rate of solid polymer residues. While the above additives have good antioxidant and tribological properties, their use in industry is limited by their high cost or lack of environmental friendliness.
Layered double hydroxides (LDH) are new types of two-dimensional nanomaterials [17], as compared with traditional layered materials, which possess good thermal stability [18], non-toxicity [19], and low production cost [20]. Their chemical formula can be expressed as: [M2+1–xM3+x(OH)2]x+(An−)x/n·mH2O. M2+ and M3+ are divalent and trivalent metal cations, respectively; An– is the interlayer anion for charge balance. Metal cations are located at the center of the hexagonal crystal of the laminates, while the hydroxide ions occupy the apexes of the hexagonal crystal. Most metal cations, such as Mg2+, Zn2+, Co2+, Al3+, Fe3+, etc., can achieve atomic-level dispersion in the laminates of LDHs. LDH materials and their composite products have been widely reported in the field of energy storage, ion exchange, drug delivery, flame-retardant additives, etc. [21,22,23,24,25,26,27,28]. There are abundant hydroxyl groups and a large number of unsaturated coordination structures on the surface and edge of the laminate, which can react with the free radicals in the lubricating oil and effectively quench alkyl free radicals and alkoxide free radicals generated by oil heating [29].
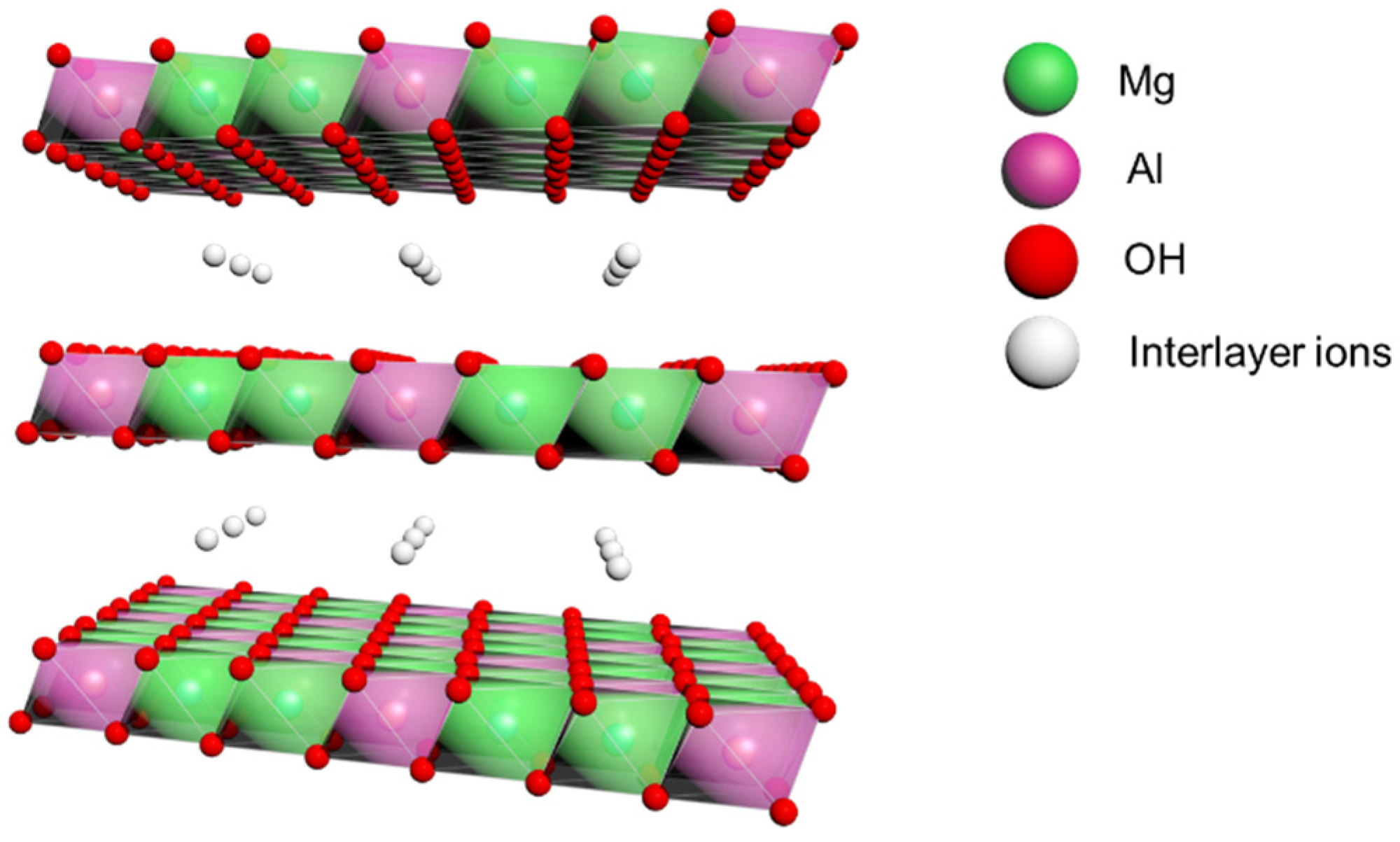
In this paper, we prepared MgAl LDH (schematically shown in Figure 1) by the co-precipitation method, using a colloid mill. The enhancement of service life, thermal oxidation resistance, and decomposition ability of large electric shovel grease by adding LDH was investigated. Subsequently, the changes in energy storage modulus and flow transition index at different temperatures for different LDH additions were compared to determine the optimal LDH additions for antioxidant and rheological properties. Finally, based on these results, the antioxidant behavior and rheological behavior of LDH as a grease additive were discussed.
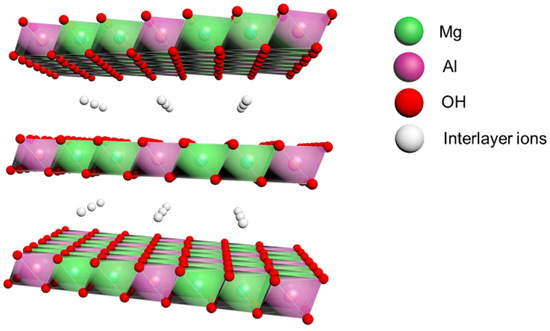
Figure 1.
Schematic diagram of the structure of LDH.
2. Materials and Methods
2.1. Materials
Mg(NO3)2·6H2O, Al(NO3)3·9H2O (analytical pure, >99.0%), NaOH (analytical pure, >96.0%), and Na2CO3 (analytical pure, >99.8%) were supplied by Aladdin (Shanghai, China). The water used in the experiment was deionized water. Base oil was supplied by SK Holdings (Seoul, Republic of Korea).
2.2. Synthesis of MgAl LDH
The MgAl LDH was prepared by the co-precipitation method, using a colloid mill [30]. In detail, 15.052 g Al(NO3)3·9H2O and 20.5128 g Mg(NO3)2·6H2O were dissolved in 200 mL deionized water to form a clear solution, A. NaOH (7.68 g) and Na2CO3 (8.48 g) were dissolved in 200 mL deionized water to form solution B. Solution A and B were poured into colloid mill simultaneously with rapid rotating (3000 rpm) for 3 min; and, then, the colloidal suspension was transferred into a reaction kettle and crystallized at 110 °C for 24 h. Subsequently, the product was washed with deionized water and alcohol several times using a centrifuge. Finally, MgAl-LDH powder was obtained after drying at room temperature.
2.3. Characterization
The chemical composition and crystal structure of the samples were analyzed by X-ray powder diffractometer (D8Discover25, Bruker, Mannheim, Germany). The Cu Kα ray wavelength was λ = 0.5416 nm, and the scanning step was 0.02°. The crystal structure and particle morphology of the prepared powders were characterized and observed by a scanning electron microscope (SEM; Zeiss SUPRA 55, Jena, Germany) with an accelerating voltage of 20 kV. The particle size distribution and size of samples were characterized by a laser particle size analyzer.
2.4. Tribological Experiments
The tribological performance of lubricating grease was detected by a four-ball friction tester, according to the standard methods ASTM D2596 and ASTM D2266. The testing parameters of wear protection were set at a speed of 1450 rpm, a stable applied load of 196 N, and a duration time of 30 min at a temperature of 75 °C. The testing parameters of extreme pressure performance were set at a speed of 1760 rpm and room temperature.
The cone penetration of the lubricating grease was tested by cone penetration meter at a temperature of 25 °C, according to the standard method ASTM D217. The drop point of the lubricating grease was tested by drop point apparatus, according to the standard method ASTM D566.
The corrosion resistance of the lubricating grease was determined by the standard methods ASTM D1743 and ASTM D4048. The corrosion resistance of the lubricating grease in relation to steel bearing was tested at a temperature of 52 °C and a time of 48 h. The corrosion resistance of the lubricating grease in relation to T2 copper was tested at a temperature of 100 °C and a time of 24 h.
Base oil viscosity was tested by a viscosity meter at a temperature of 100 °C, according to the standard method ASTM D445.
2.5. Performance of Anti-Oxidation and Bearing Lifetime
A Pressure Differential Scanning Calorimeter (PDSC) was used to determine the oxidation stability of lubricating oils, according to the standard method ASTM D5483, with a test temperature of 190 °C and a pressure of 3.5 MPa. The grease lifetime was assessed by using a high-speed bearing lifetime testing machine, and the test methods are 2ASTM D 3336 (“Determination of the lifetime of grease in ball bearings at high temperatures”) and 3DIN 51821-2 (“Determination of the effective lifetime of rolling bearing greases FE9 method”).
2.6. Viscosity-Temperature Property
The rheological properties of lubricating oil were tested by a DISCOVERY HR-3 Rotational Rheometer. The specific operation mode of the dynamic rheology test was as follows: the initial temperature was 200 °C, the cooling rate was 5 °C/min, the terminal temperature was −30 °C, the shear rate was 1.01/s, and one data point was taken every 60 s. The dynamic viscosity of four lubricating oils was measured as the temperature decreased.
3. Results and Discussion
3.1. Structural Characterization of MgAl LDHs Materials
3.1.1. XRD of MgAl LDHs
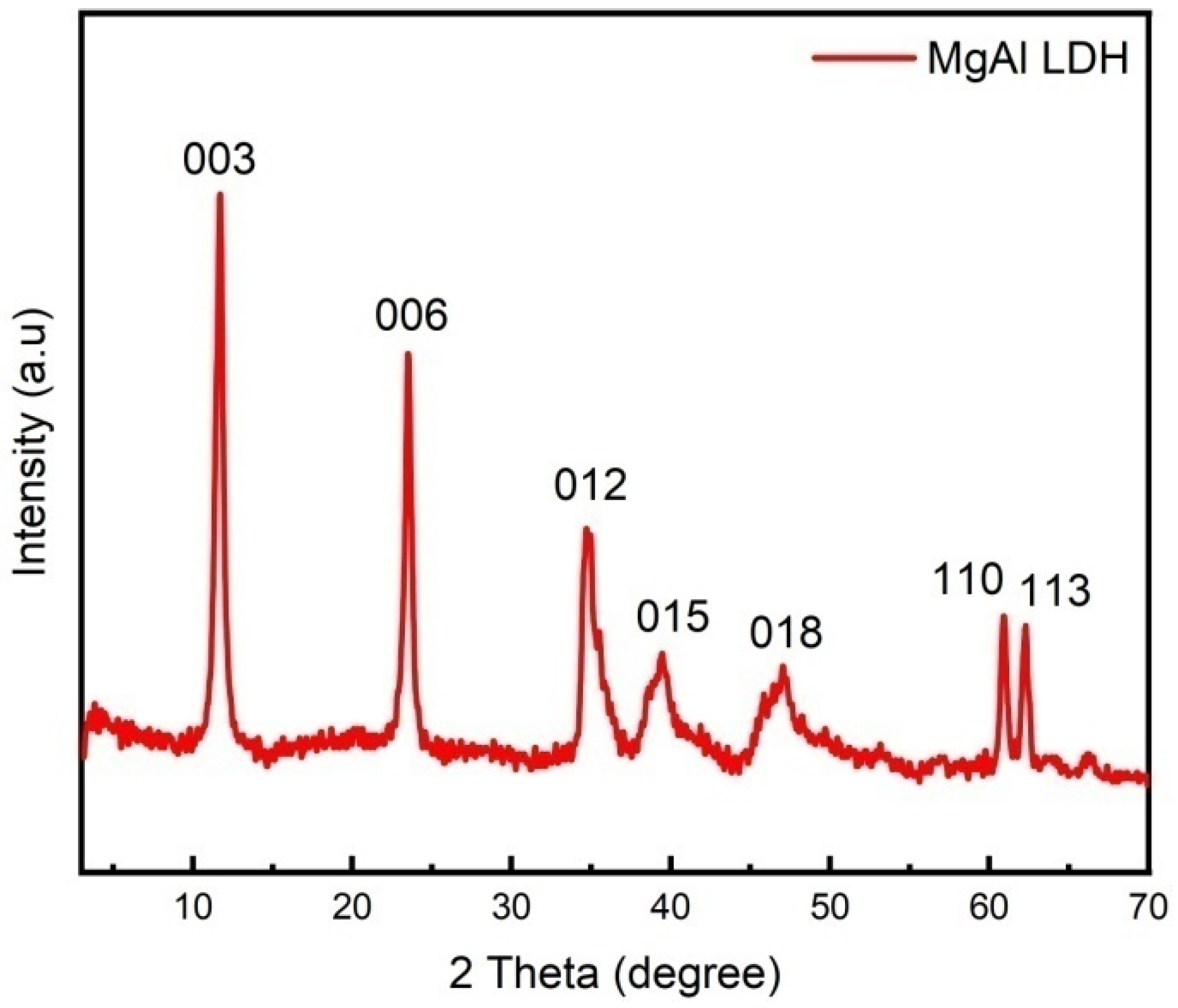
Figure 2 shows the XRD patterns of MgAl LDHs samples. It can be seen, from the figure, that the characteristic diffraction peaks of [003], [006], [009], [012], [015], [018], [110], and [113] of LDHs appear in all three samples. The diffraction peaks of the samples in the spectrogram are symmetrical and sharp, with high intensity and stable baseline, indicating that the synthesized LDHs have good crystal shape, high crystallinity, and high regularity between layers. The appearance of diffraction peaks on [1010] and [1011] crystal surfaces indicates that the LDHs have fine structures. It can be seen, from the position of the 003 peak, that the interlayer spacing is 0.75 nm, and the interlayer anion is CO32−.
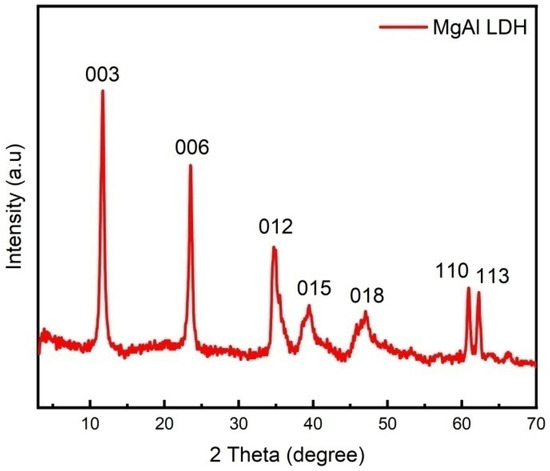
Figure 2.
XRD patterns of LDH.
3.1.2. SEM Image and Particle Size Analysis of MgAl LDHs
The SEM figure shows that LDH prepared by the co-precipitation method, using a colloid mill, has a hexagonal sheet structure, a smooth surface, and a sharp edge, indicating that the prepared LDHs have complete structure and high crystallinity, which are consistent with XRD characterization results. The particle size of the sample is mainly concentrated at 70−200 nm, with the average particle size being d0.5 = 100 nm. The particle size of MgAl LDH samples was prepared by the co-precipitation method, using a colloid mill that is nano scale, with small particle size, high crystallinity, and complete crystal shape (Figure 3). The average particle size is about 120 nm.

Figure 3.
SEM image and particle size distribution of MgAl LDHs.
3.2. Study on Properties of LDHs in Grease
3.2.1. Study on Oxidation Resistance of LDHs in Lithium Base Grease
Over the years, many oxidation bench tests have been developed and proven to be very effective for grease formulation selection, especially in the screening and formulation development of new antioxidants. Due to the limitations of the test site, a single test cannot simulate the real working conditions. Therefore, when examining grease formulation and antioxidant screening, an effective method is to use multiple test methods for comparative evaluation.
Bearing lifetime refers to the ability of grease to lubricate high temperature and high-speed ball bearings for extended periods of time under light load conditions. The test method is ASTM D 3336 (“Life of lubricating greases in ball bearings at elevated temperatures”).
To test the lifetime of the bearing, a set of steps are undertaken. Firstly, a 3.2 mL sample is loaded into the cleaned 6204 bearing, following which, the test bearing, support bearing, and finger spring are installed onto the main shaft, and the thermocouple is positioned and fixed carefully. Secondly, the device is turned on to raise the temperature of the bearing up to the measured temperature. At a speed of 10,000 r/min, the bearing is run continuously until either the grease sample fails or the specific running time is achieved. The running time (in hours) is then recorded as the lifetime of the specimen in the ball bearing.
Analyzing the data presented in Table 1 and Table 2, it is evident that, at a temperature of 149 °C, the bearing life of the grease sample is similar when the base grease is added with LDHs and diphenylamine at a concentration of 0.3%. Conversely, when the base grease was added with LDHs and T531 at a concentration of 0.5%, it was observed that the bearing life of the grease sample 4 (with added LDHs) doubled in comparison to sample 3 (with added T531). This indicates that the bearing life is heavily influenced by the addition of different types of antioxidants. Furthermore, these results suggest that LDH could potentially replace traditional organic antioxidants and pave the way towards sustainable, green development.

Table 1.
Proportion of LDHs in general lithium base grease.

Table 2.
Bearing lifetime test of LDHs in general lithium base grease.
Table 3 and Table 4 show the results of the bearing lifetime tester test, conducted by a different company, which found that the samples containing LDHs had significantly higher bearing lifetimes compared to the base grease. The base grease had a bearing lifetime of 40 h at 149 °C. However, adding 1% of LDHs increased the grease lifetime by a factor of three, and increasing the LDH content to 3% extended the lifespan by approximately four times. Further, adding 5% of LDHs increased the grease lifetime by nearly five times.

Table 3.
Formulas of different proportions of LDH general lithium base grease.

Table 4.
ASTM D 3336 bearing lifetime test of LDHs in general lithium base grease.
The FE9 method is a testing process that uses angular contact ball bearings in a specialized rolling bearing grease tester to determine the effective service lifetime of grease. This test method is defined by the DIN 51821-2 standard, titled “Testing of lubricants—Test using the FAG roller bearing grease testing apparatus FE9—Part 2: Test method”. The FE9 test steps involve filling five sets of clean test bearings with a certain amount of grease to be tested, and then the test stops involve installing these test bearings onto five test units. The test conditions are set in accordance with specific requirements, including a speed of 6000 r/min, axial force of 1500 N, and a temperature range of 120 °C to 220 °C (set as an integer multiple of 10 °C) under test conditions. The heating system is activated to control the temperature using a temperature controller. During the test, the friction torque of the bearing will increase as a result of poor lubrication. If this results in the power of the driving motor exceeding the limit value and lasting for 6 s to 8 s, the test is ended. After the test, the running times of each of the five test bearings are inputted into the Weibull distribution data evaluation software (version 9) to produce life failure probability distribution diagrams. The effective lifetime of the grease can be determined by reading the running time at the 50% failure probability mark on the diagram.
Through the test results in Table 3 and Table 5, it was found that the lifetime of FE9 bearings of the three samples added with LDHs are obviously better than those of the base grease, and with the increase in the amount of LDHs added, the bearing lifetime tends to increase regularly. Table 6 reveals that samples 1–4 used mineral oil, while samples 5–8 used a higher viscosity semi-synthetic oil, resulting in a significant improvement in the CRC bearing life test.

Table 5.
DIN 51821-2 bearing life test of LDHs in general lithium base grease.

Table 6.
The typical properties of eight greases.
3.2.2. Study on Oxidation Resistance of LDHs in Grease for Mine Electric Shovel
After the 1930s, electricity replaced other forms of power and became the only power for mining excavators, and the steam shovel was replaced by the electric shovel. At this time, the shovel was mainly used for stripping mining, and the large mining shovel used for stripping is called a “stripping shovel”. With the expansion of the scale of open-pit mining, the stripping shovel developed towards a larger scale. In the 1970s, as surface mining technology improved, smaller pick-up shovels gradually replaced larger stripper shovels for easier matching with mining trucks. So far, this type of electric shovel is still the main equipment for open-pit mining mines.
At the end of 2005, the WK-20 20 m3 electric shovel of Taiyuan Heavy Industry Co., Ltd. (TYHI) (Taiyuan, China) was successfully launched, breaking the long-term monopoly of American companies in the field of large mining electric shovels, with a bucket capacity of more than 20 m3. Since then, TYHI has developed WK-27, WK-35, WK-55, and WK-75 shovels, among which THE WK-75 shovel design standard, with a bucket capacity of 75 m3, is the largest types of shovel independently developed in China.
The shovel is one of the most important parts of equipment in open-pit mining technology. Lubrication of the gear mechanism of the shovel has always been a difficult problem. Improper selection of lubricant often causes serious pitting corrosion, abrasion, scratches, excessive wear, and other phenomena, which makes the gear enter overhaul before its service lifetime, and this increases maintenance costs. It is the bound duty of SINOPEC to prolong the use of grease in mine shovels and to reduce the surface pollution caused by exposed grease in open-pit coal mines.
According to Table 7 and Table 8, as well as Figure 4 and Figure 5, LDHs added to shovel grease can prolong the service lifetime of grease, while the overall performance of grease is not affected. After LDH is added, the anti-wear performance of grease is slightly improved (Figure 4), which may be caused by the two-dimensional structure and high crystallinity of LDH, forming a protective film on the surface of the friction pair.

Table 7.
Formula of LDHs in shovel grease.

Table 8.
Typical product data table.

Figure 4.
Surface morphology of four spheres of samples 9, 10, 11, and 12.
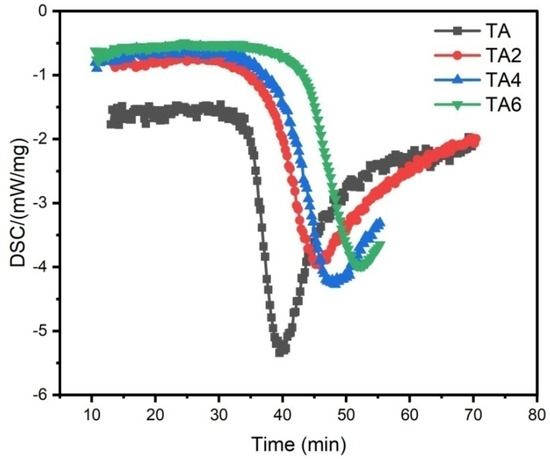
Figure 5.
PDSC measurements of TA, TA2, TA4, and TA6 (Note: sample 9 is TA, sample 10 is TA2, sample 11is TA4, and sample 12 is TA6).
The main premise of DSC to study reaction kinetics is that the degree of reaction is proportional to the thermal effect released or absorbed by the reaction, that is, it is proportional to the area under the DSC curve. This can be expressed by the following formula:
where; H—enthalpy, heat of reaction at temperature T; HT—total enthalpy of the reaction, —degree of reaction proceeding, —Total area under the DSC curve —Reaction process area under DSC curve, —The difference between the total area under the DSC curve and the area of the reaction process, T—reaction time. Thus, the basic kinetic equation of the reaction can be rewritten as:
On both sides of the exponential, the following holds:
By comparing DSC scanning curves with the same change rates of the two reactions at different rates, the activation energy E can be obtained:
If the change rate α is the same at peak temperature, it is further expressed as:
The Ea can be obtained through Equation (8), and the rate constant k is dependent on the reaction temperature, according to the Arrhenius Equation (9).
In these equations, β is the heating rate (K min−1), Tp is the temperature corresponding to the inflection point of the thermodegradation curves, which correspond to the maximum reaction rate (K), A is the pre-exponential factor (min−1), Ea is the activation energy (kJ mol−1), and R is the gas constant (8.314 J mol−1 K−1). The activation energy and pre-exponential factor can be obtained from the slope.
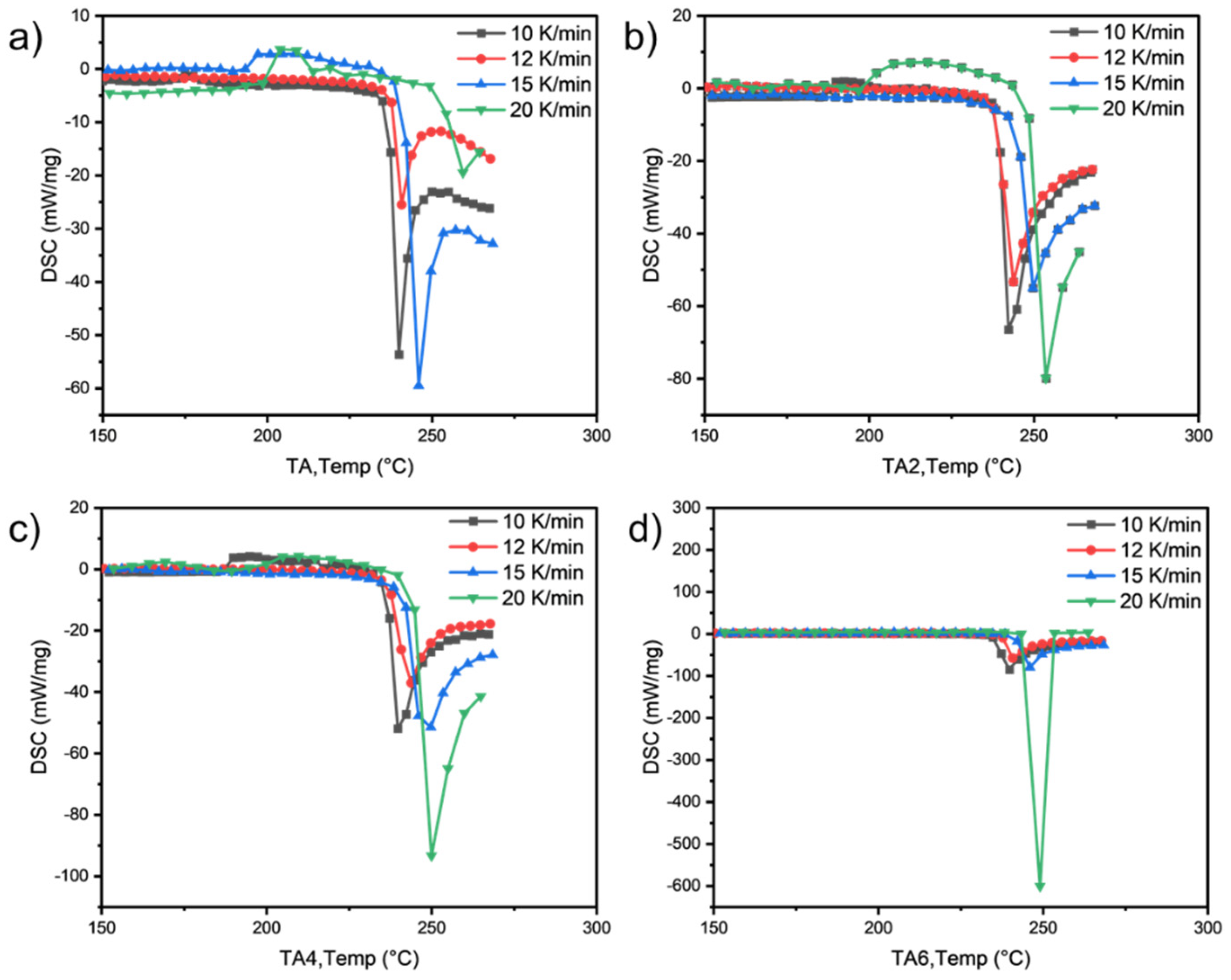
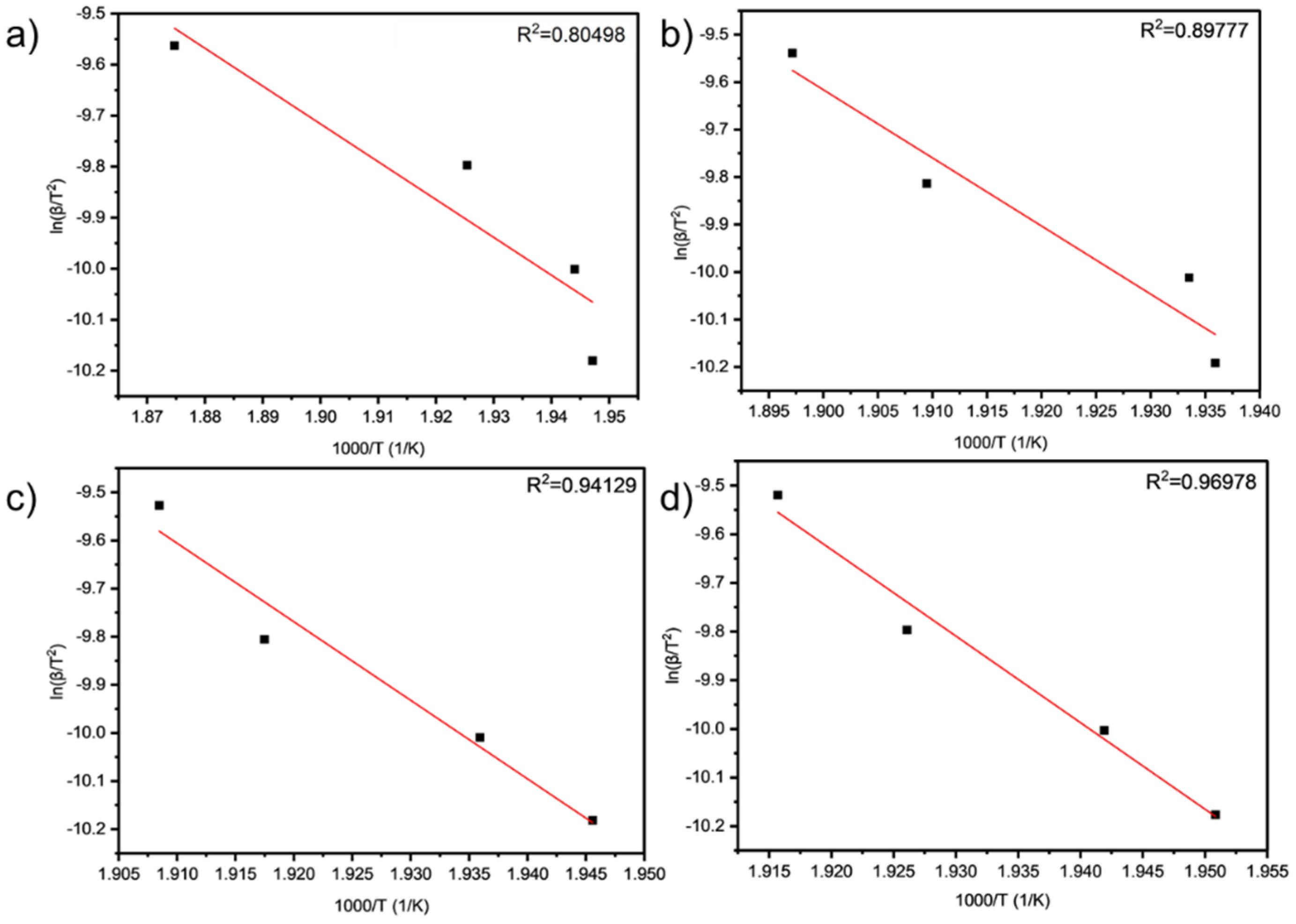
As can be seen from Table 9 and Table 10 and Figure 6 and Figure 7, the activation energies of the four samples all meet the increasing law of ETA < ETA2 < ETA4 < ETA6, indicating that, with the increase in the amount of LDHs added, the larger reaction activation energy of grease indicates the stronger that its thermal anti-oxidation and decomposition ability are.

Table 9.
PDSC situation of samples with different heating rates.

Table 10.
Correlation coefficients of different samples (R2), activation energy.
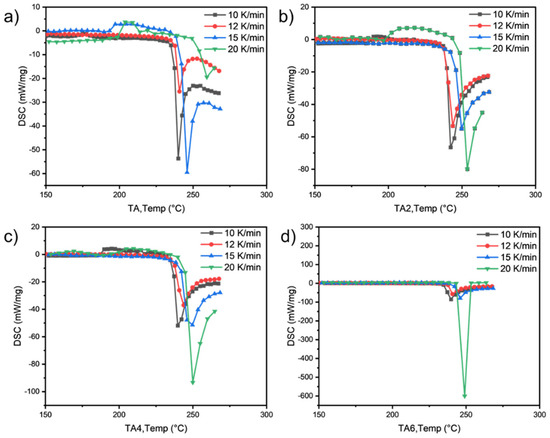
Figure 6.
PDSC curves of (a) TA, (b) TA2, (c) TA4, and (d) TA6 at different heating rates.
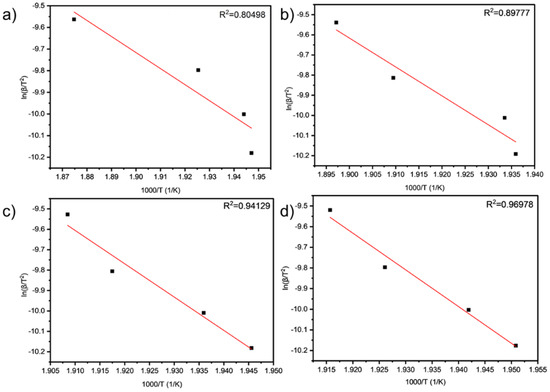
Figure 7.
Linear fit results of (a) TA, (b) TA2, (c) TA4, and (d) TA6 obtained by PDSC analysis.
3.2.3. Analysis of Rheological Properties of Electric Shovel Grease
Grease is a viscoelastic non-Newtonian fluid. The elastic part of viscoelasticity is represented by the energy storage modulus G′, which indicates that the stress energy can be temporarily stored and recovered during the experiment. The viscous part is represented by the loss modulus G″, which indicates thatthe grease loses energy during the initial flow, which is converted into shear heat, and the loss is irreversible. When rheological experiments are performed, the shear stress increases, and the point where the energy storage modulus G′ begins to decrease is usually defined as the end of the linear viscoelastic zone (LVE), called the yield point, and the shear stress at this point is also called the yield stress τy. The maximum elastic deformation that the grease can withstand can be determined by this point. Shear stress and deformation continue to increase, G′ line and G″ line intersect, the intersection point is called the “flow point”, the shear stress at this point is called the “flow stress”, τf, and, at this time, the grease thickener structure is more damaged, and the grease also begins to flow.
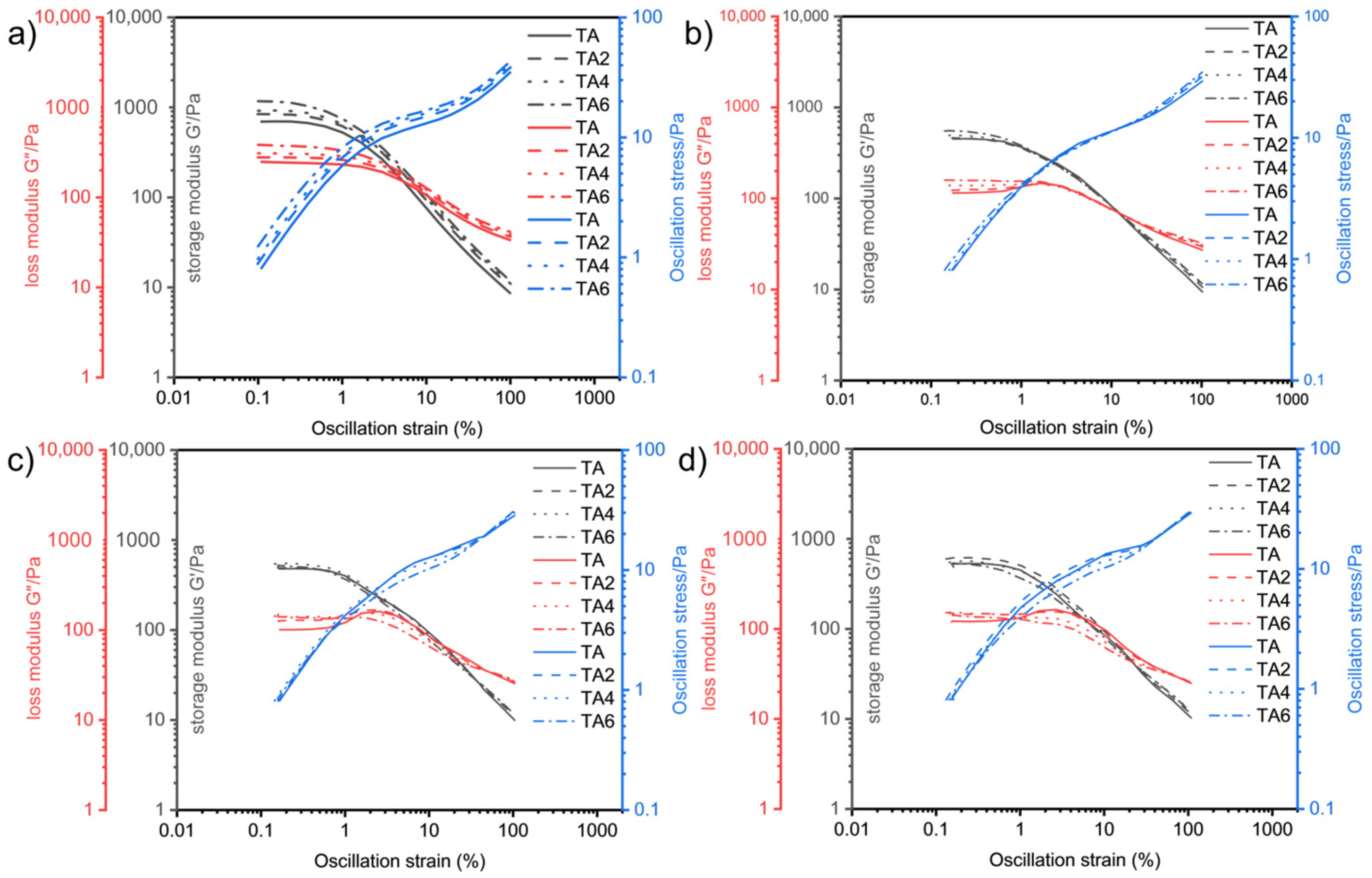
The oscillatory rheological experiments were performed on four grease samples using this method. Figure 8 shows the relationship between the storage modulus G′ and loss modulus G″, with the variation in shear deformation γ at different temperatures measured by changing the experimental temperatures (60, 80, 100, and 120 °C).
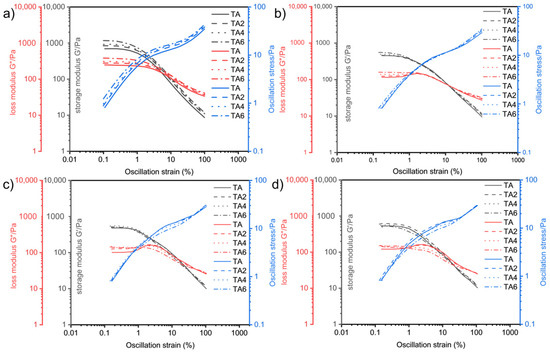
Figure 8.
Variation in storage modulus, loss modulus, and oscillation stress with strain at different temperatures: (a) 60 °C, (b) 80 °C, (c) 100 °C, and (d) 120 °C.
It can be seen, from Figure 8, that the overall pattern of the energy storage modulus G′ of the four greases entering the rheological transition zone at 60, 80, 100, and 120 °C is gradually decreasing with the increase in temperature. From the previous principle analysis, it is known that the larger the G′ is, the better the structure of the grease that is maintained before it is in gel state, and the lubricating performance of the grease inside the electric shovel gear is often carried out within a few seconds after the start of operation. Therefore, it is beneficial to improve the lubrication of gears by appropriately improving the energy storage modulus and fluidity performance of grease. At 60 °C, as the proportion of LDHs increases, the energy storage modulus of TA6 grease sample is the largest, and TA2 is the second-largest at 120 °C, and, as the proportion of LDHs increases, the energy storage modulus of TA6 grease sample is the largest, and TA4 is the second-largest; at 100 °C, the energy storage modulus of TA4 grease sample is the largest, and TA2 is the second-largest; at 120 °C, the energy storage modulus of TA2 grease sample is the largest, TA is the second-largest, and TA6 is the smallest; it means that, at low temperature, the energy storage modulus of grease increases simultaneously with the increase in LDH ratio. When there is an increase in temperature, the larger the LDHs ratio is, the larger the system elastic deformation damage is, so it is necessary to consider the effect of suitable ratio of LDHs on energy storage modulus. Combined with the flow transition index and energy storage moduli in different linear viscoelastic intervals, it can be seen that adding two-percent LDH is the best for the system’s elastic deformation and energy storage modulus. This indicates that the internal structure of sample TA2 is more stable under shear, and the fiber structure is better maintained, which is better for gear support and lubrication, and this is conducive to reducing gear wear.
The values of the energy storage modulus and the loss modulus, as well as shear stress and the corresponding strain values of the four greases at yield point and flow point at the two sets of experimental temperatures, can be calculated, and the numerical results are listed in Table 11.

Table 11.
G′ and G″ about four greases at yield point, as well as critical strains, corresponding to the flow point.
Table 11 shows that, at 60 °C, the yield point energy storage modulus G′ is ranked as G′ (TA6) > G′ (TA2) > G′ (TA4) > G′ (TA); at 80 °C, G′ (TA6) > G′ (TA4) > G′ (TA) > G′ (TA2); at 100 °C, G′ (TA4) > G′ (TA2) > G′ (TA) > G′ (TA6); at 120 °C, G′ (TA2) > G′ (TA) > G′ (TA4) > G′ (TA6).
At the flow point, the strain amplitude γ is ordered as follows: 60 °C, γ (TA6) > γ (TA4) > γ (TA2) > γ (TA); 80 °C, γ (TA4) > γ (TA2) > γ (TA6) > γ (TA); 100 °C, γ (TA4) > γ (TA6) > γ (TA2) > γ (TA); 120 °C, γ (TA2) > γ (TA6) > γ (TA4) > γ (TA). The energy storage modulus, G′, is ordered as follows: 60 °C, G′ (TA2) > G′ (TA4) > G′ (TA) > G′ (TA6); 80 °C, G′ (TA) > G′ (TA6) > G′ (TA2) > G′ (TA4); 100 °C, G′ (TA) >G′ (TA2) > G′ (TA4) > G′ (TA6); and, at 120 °C, G′ (TA) > G′ (TA4) > G′ (TA2) > G′ (TA6). The shear stress, τf, is sorted as follows: 60 °C, τf (TA6) > τf (TA4) > τf (TA2) > τf (TA); 80 °C, τf (TA4) > τf (TA2) > τf (TA6) > τf (TA); and, at 100 °C, τf (TA4) > τf (TA2) > τf (TA6) > τf (TA); finally, at 120 °C, τf (TA2) > τf (TA4) > τf (TA6) > τf (TA).
At the flow point, the flow transition index τf/τy is ordered as follows: τf/τy (TA2) > τf/τy (TA) > τf/τy (TA4) > τf/τy (TA6) for 60 °C; τf/τy (TA2) > τf/τy (TA4) > τf/τy (TA6) > τf/τy (TA) for 80 °C; τf/τy (TA2) > τf/τy (TA6) > τf/τy (TA4) > τf/τy (TA) for 100 °C; and τf/τy (TA2) > τf/τy (TA6) > τf/τy (TA4) > τf/τy (TA) for 120 °C.
The maximum value of τf/τy for sample TA2 at the test temperature indicates that, at this temperature, TA2 grease-like soap fibers have the best structural stability, and the soap fibers are less likely to fracture or break. Comparing the storage modulus and flow transition index at different temperatures, it can be seen that the addition of the appropriate amount of LDHs for the system oxidation resistance and viscoelasticity requires close attention. For the electric shovel grease system, adding 2% LDH can achieve the best oxidation resistance and rheological properties.
Since grease is a non-Newtonian fluid, curve fitting in this paper refers to the streamlining of flow curves of varying graphical complexity into an equation with two, three, or four coefficients. Curve fitting has the following advantages: in quality control, it is easier to mathematically set the tolerance range with the help of standard regression coefficients, and it is more difficult to visually compare the shape difference between the unique flow curve formed by the measured test point and the standard curve. After completing the procedural processing of the experimental data, the second step is obviously to continue the automatic evaluation of the samples for compliance with the technical specifications. Regression calculations help to solve this problem.
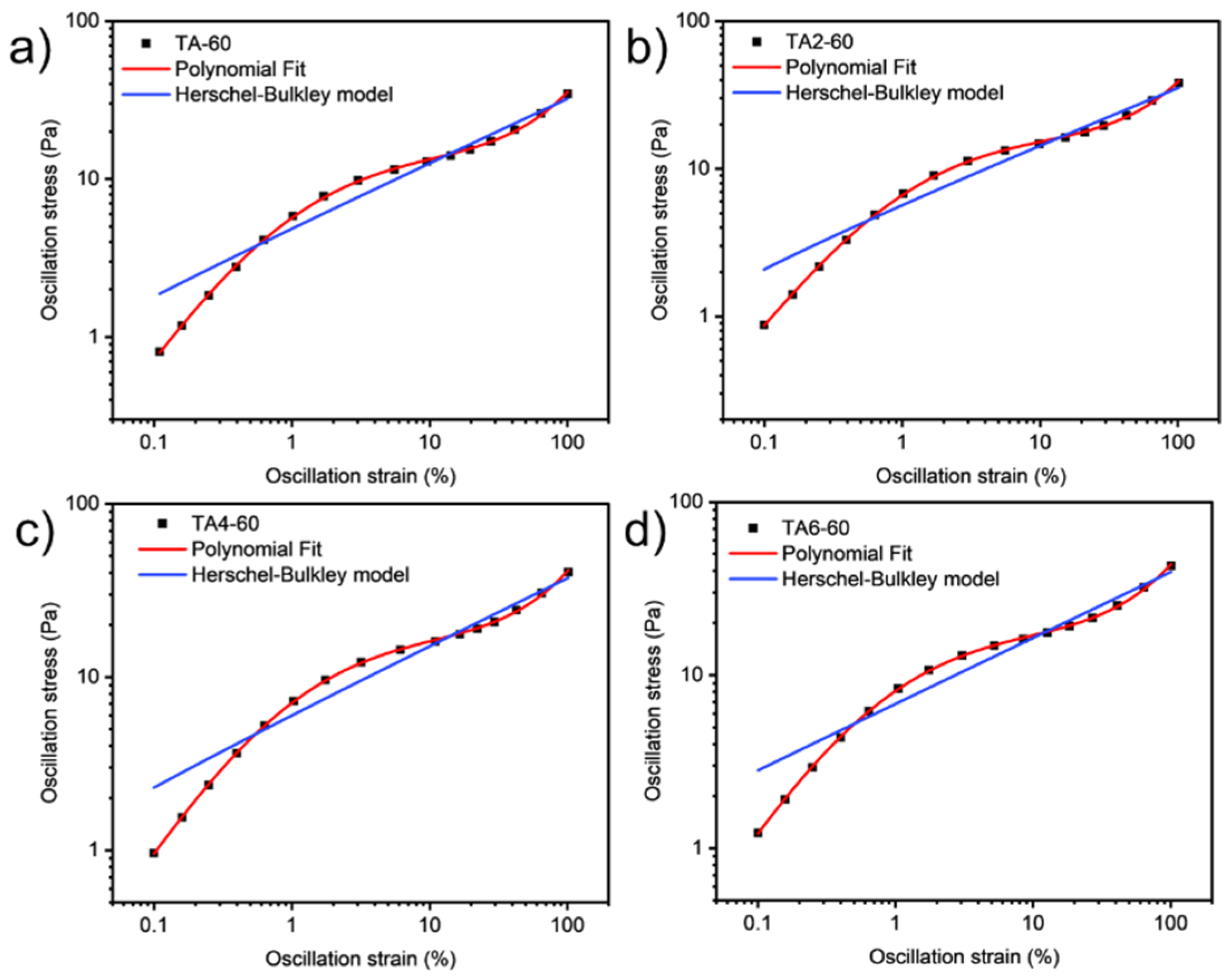
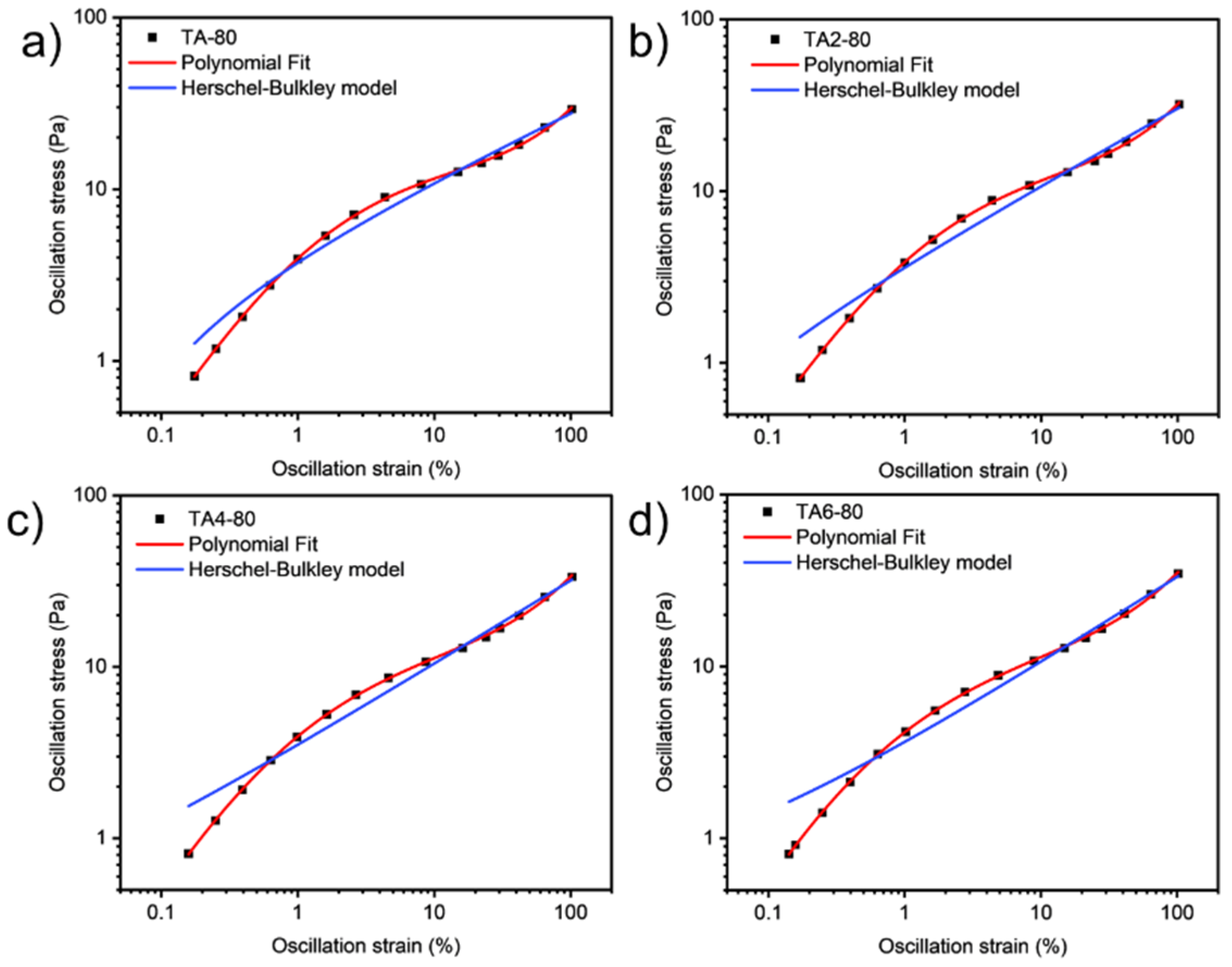
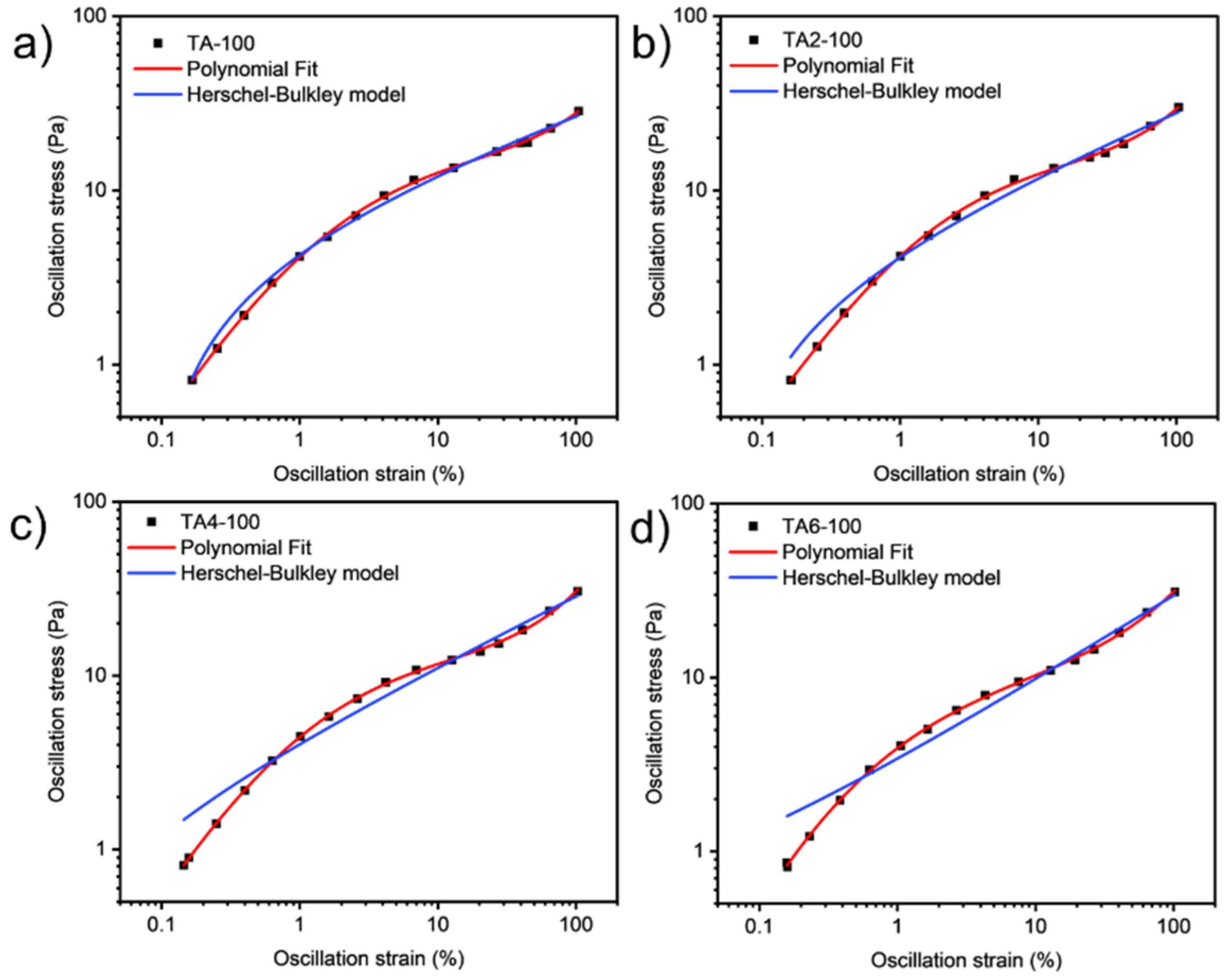
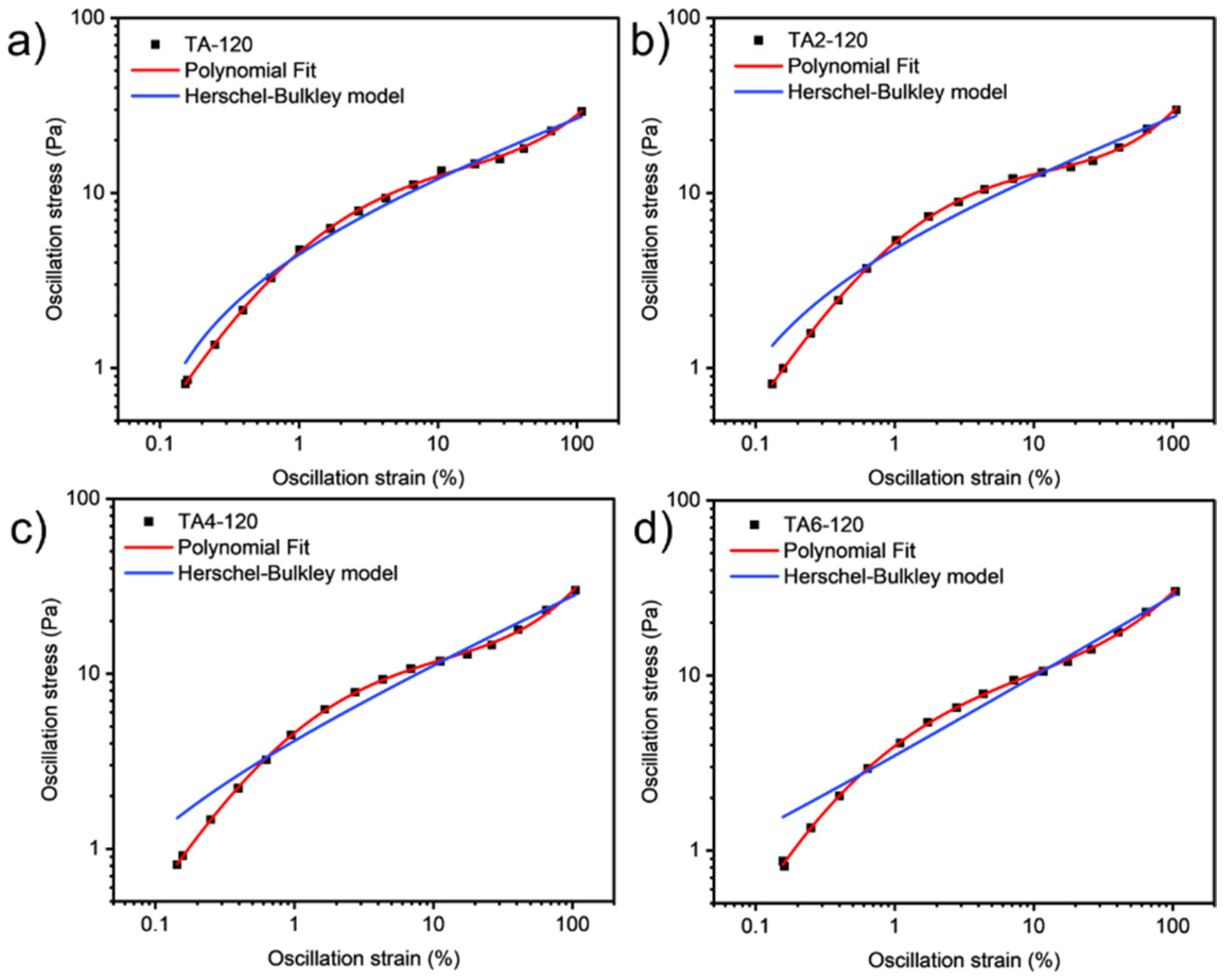
These data were not fitted to the Ostwald-de-Waele model (Equation (10)) because of R2. These presented a value of n not close to 1, as was expected for non-Newtonian fluids. The existence of a yield stress value can be recognized in Figure 9, Figure 10, Figure 11 and Figure 12, and the data were not satisfactorily fitted to a Herschel-Bulkley model (Equation (11)), and the results are collected in Table 12. The data were satisfactorily fitted to a polynomial model (Equation (12), R2 > 0.999), and the results are collected in Table 12.
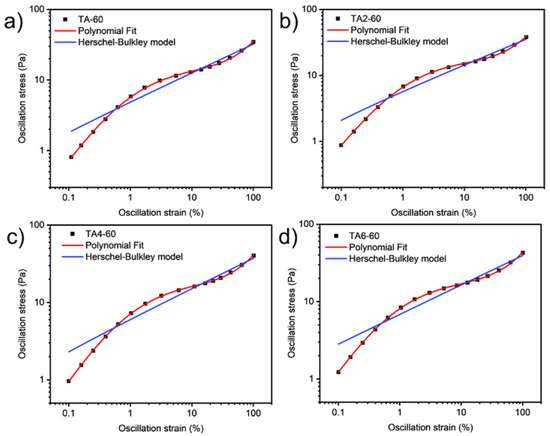
Figure 9.
Oscillation strain dependence on stress of GRK-A for (a) TA, (b) TA2, (c) TA4, and (d) TA6 at 60 °C.
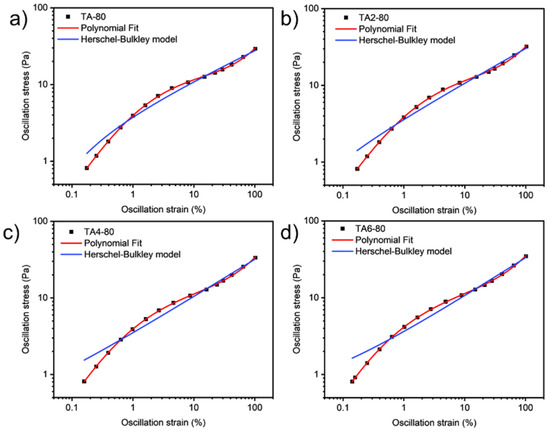
Figure 10.
Oscillation strain dependence on stress of GRK-A for (a) TA, (b) TA2, (c) TA4, and (d) TA6 at 80 °C.
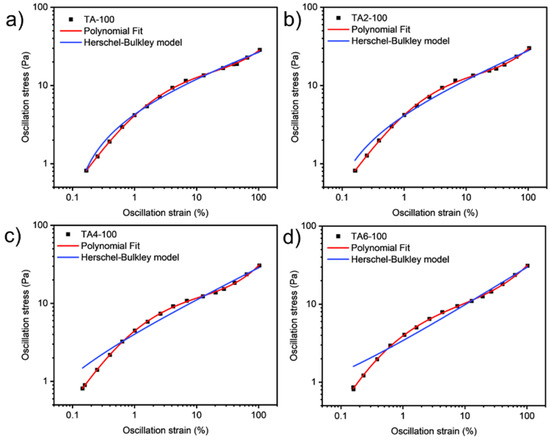
Figure 11.
Oscillation strain dependence on stress of GRK-A for (a) TA, (b) TA2, (c) TA4, and (d) TA6 at 100 °C.
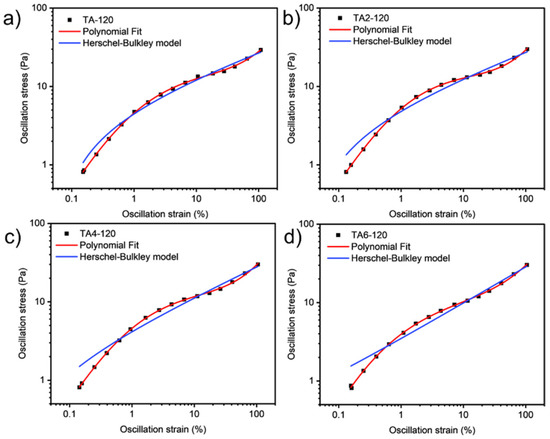
Figure 12.
Oscillation strain dependence on stress of GRK-A for (a) TA, (b) TA2, (c) TA4, and (d) TA6 at 120 °C.

Table 12.
Fitting parameters for different samples.
3.2.4. Electric Shovel Grease Viscosity and Temperature Analysis
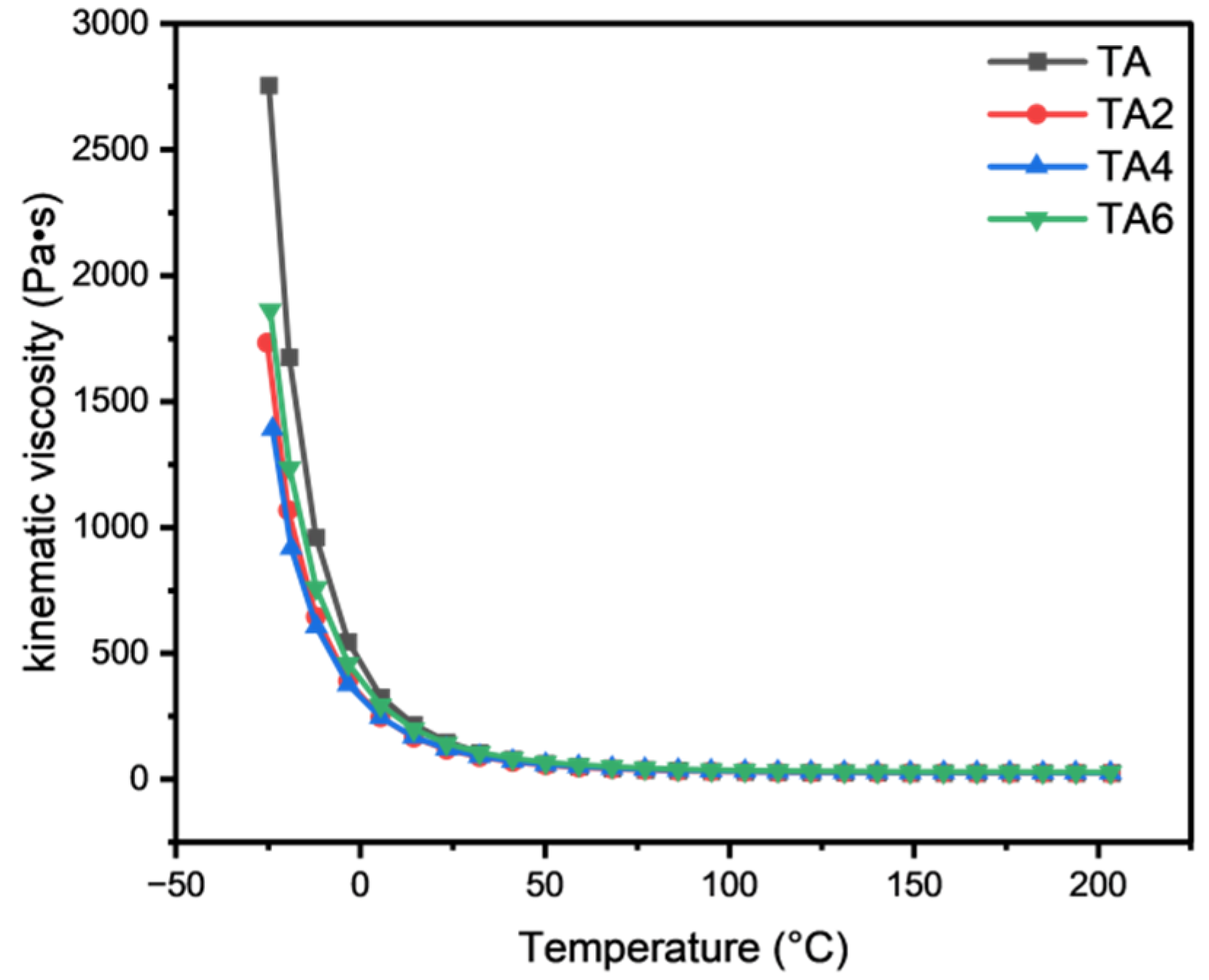
The viscosity–temperature property of grease is the performance of viscosity changed by the temperature. Viscosity usually decreases with the increase in temperature. The change in viscosity of a grease is highly correlated with temperature, and there is a pattern, and the viscosity reflects the rheology of the fluid. The most commonly used equations for calculating viscosity are derived from the corresponding state or statistical principles, but this method has some limitations.
The rheological properties of mine grease with different LDHs additions were investigated using a DISCOVERY HR-3 rotational rheometer. It can be found, by Figure 13, that the viscosity changes with temperature in an approximate exponential manner, and the kinematic viscosity of the four types of grease increases sharply when cooling from 200 °C to −30 °C, but the curve is relatively flat, and the decline becomes smaller from 200 °C to 5 °C. In the low-temperature working interval (−30 °C to −5 °C), the grease samples with different proportions of solid LDHs added are better than mine grease low temperature, indicating that the addition of solid LDH can change the low temperature performance of mine grease. In the high temperature working interval (40 °C~200 °C), the four grease samples’ power viscosities are comparable, indicating that, after adding nano-solid LDHs, the grease in the high temperature working interval viscosity declines, as a trend, and adding three proportions of LDH viscosity declines, as a trend, in the same way, especially after adding 6% of LDHs, which resulted in slightly higher viscosity.
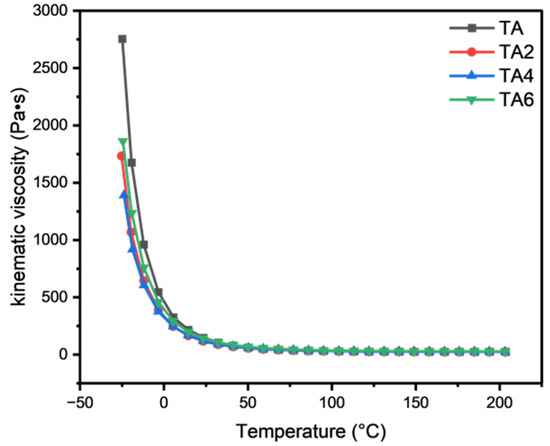
Figure 13.
Temperature curve of different samples.
4. Conclusions
In this paper, a new environmentally friendly LDH antioxidant was developed from theoretical design and mainly through experiments. The main research contents and conclusions of this paper are as follows:
- (1)
- LDH was prepared via the co-precipitation method and used as an environmentally friendly antioxidant in lithium base grease.
- (2)
- By adding LDHs into large electric shovel grease (GRK-A), the service lifetime of grease is extended by 20%, while the overall performance of grease is not affected. It was found that, with the increase in LDH addition, the greater the activation energy of the reaction of grease, the stronger the resistance to thermal oxidation and decomposition.
- (3)
- Comparing the energy storage modulus and flow transition index at different temperatures, it can be seen that adding the right amount of LDH needs close attention for system oxidation resistance and viscoelasticity. For electric shovel grease, the best oxidation resistance and rheological properties can be achieved by adding 2% LDH. The rheological viscosity–temperature curve shows that the grease samples with different ratios of solid LDHs are better than mine grease at low temperatures.
- (4)
- Non-Newtonian fluid grease is not suitable for the Ostwald-de-Waele model and the Herschel-Bulkley model, but it is more suitable for the polynomial model.
Author Contributions
Conceptualization, Y.L. and J.H.; methodology, Y.L., W.Z. and W.X.; software, W.Z., Y.H. and Q.Z.; formal analysis, W.Z., Y.H. and Q.Z.; investigation, W.Z. and W.X.; resources, Y.L. and J.H.; data curation, W.Z., Y.H. and Q.Z.; writing—original draft preparation, Y.L. and Q.Z.; writing—review and editing, Y.L. and J.H.; supervision, J.H.; project administration, J.H.; funding acquisition, J.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Fundamental Research Funds for the Central Universities (BHYC1702B).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
List of abbreviations
| LDH | Layered double hydroxide |
| LVE | Linear viscoelastic zone |
| H | Heat of reaction at temperature T |
| HT | Total enthalpy of the reaction |
| Degree of reaction proceeding | |
| Total area under the DSC curve | |
| Reaction process area under DSC curve | |
| Difference between the total area under the DSC Curve and the area of the reaction process | |
| t | Reaction time |
| β | Heating rate |
| Tp | Temperature corresponding to the inflection point of the Thermodegradation curves |
| A | Pre-exponential factor |
| Ea | Activation energy |
| R | Gas constant |
| G′ | Energy storage modulus |
| G″ | Loss modulus |
| τy | Yield stress |
| τf | Flow stress |
References
- Sommer, M.; Haas, W. A new approach on grease tribology in sealing technology: Influence of the thickener particles. Tribol. Int. 2016, 103, 574–583. [Google Scholar] [CrossRef]
- Lugt, P.M. A Review on Grease Lubrication in Rolling Bearings. Tribol. Trans. 2009, 52, 470–480. [Google Scholar] [CrossRef]
- Lugt, P.M. Modern advancements in lubricating grease technology. Tribol. Int. 2016, 97, 467–477. [Google Scholar] [CrossRef]
- Lisiecki, A. Tribology and Surface Engineering. Coatings 2019, 9, 663. [Google Scholar] [CrossRef]
- Minami, I. Ionic Liquids in Tribology. Molecules 2009, 14, 2286–2305. [Google Scholar] [CrossRef]
- Erdemir, A. Review of engineered tribological interfaces for improved boundary lubrication. Tribol. Int. 2005, 38, 249–256. [Google Scholar] [CrossRef]
- Meghana, K.N.; Ranjtha, R.; Ganesha, A.; Suraj, P. Synthesis and characterization of size controlled nano copper oxide structures for antioxidant study and as eco-friendly lubricant additive for bio-oils. Ceram. Int. 2022, 49, 10402–10410. [Google Scholar]
- Sonam, V.S.; Ganapati, D.Y. Synthesis of environment-friendly, sustainable, and nontoxic bio-lubricants: A critical review of advances and a path forward. Biofuels. Bioprod. Biorefin. 2022, 16, 1172–1195. [Google Scholar]
- Lu, Y.; Zhan, L.; Gong, Q.; Cai, Z.; Cheng, J.; Wen, X. Classification, mechanism and research progress of antioxidant. Plast. Addit. 2016, 116, 43–50. [Google Scholar]
- Brook, M.A.; Yepremyan, A.; Lu, G.; Melendez-Zamudio, M.; Hrabowyj, D.J.; Gale, C.B. Antioxidant silicone oils from natural antioxidants. Green Chem. 2022, 24, 8751–8759. [Google Scholar] [CrossRef]
- Cai, H. Research and application status of vitamin E as polymer antioxidant. China Plast. 2015, 29, 14–18. [Google Scholar]
- Wen, Y. Development and application progress of novel antioxidant. Shandong Chem. Ind. 2017, 46, 79–82. [Google Scholar]
- Liu, K.; Kang, J.-j.; Zhang, G.-a.; Lu, Z.-b.; Yue, W. Effect of temperature and mating pair on tribological properties of DLC and GLC coatings under high pressure lubricated by MoDTC and ZDDP. Friction 2020, 9, 1390–1405. [Google Scholar] [CrossRef]
- Morina, A.; Neville, A.; Priest, M.; Green, J.H. ZDDP and MoDTC interactions in boundary lubrication—The effect of temperature and ZDDP/MoDTC ratio. Tribol. Int. 2006, 39, 1545–1557. [Google Scholar] [CrossRef]
- Zhang, X.; Li, N.; Wei, Z.; Dai, B.; Lin, H.; Han, S. Enhanced the effects on improving the cold flow properties and oxidative stability of diesel-biodiesel blends by grafting antioxidant on PMA type pour point depressant. Fuel Process. Technol. 2022, 238, 107483–107494. [Google Scholar] [CrossRef]
- Majano, G.; Ng, E.-P.; Lakiss, L.; Mintova, S. Nanosized molecular sieves utilized as an environmentally friendly alternative to antioxidants for lubricant oils. Green Chem. 2011, 13, 2435–2440. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.; Lu, S. Preparation and application of layered double hydroxide nanosheets. RSC Adv. 2021, 11, 24254–24281. [Google Scholar] [CrossRef]
- Xu, W.; Wang, S.; Li, A.; Wang, X. Synthesis of aminopropyltriethoxysilane grafted/tripolyphosphate intercalated ZnAl LDHs and their performance in the flame retardancy and smoke suppression of polyurethane elastomer. RSC Adv. 2016, 6, 48189–48198. [Google Scholar] [CrossRef]
- Dragoi, B.; Uritu, C.M.; Agrigoroaie, L.; Lutic, D.; Hulea, V.; Postole, G.; Coroaba, A.; Carasevici, E. MnAl-Layered Double Hydroxide Nanosheets Infused with Fluorouracil for Cancer Diagnosis and Therapy. ACS Appl. Nano Mater. 2021, 4, 2061–2075. [Google Scholar] [CrossRef]
- Jiang, Y.; Yang, Z.; Su, Q.; Chen, L.; Wu, J.; Meng, J. Preparation of Magnesium-Aluminum Hydrotalcite by Mechanochemical Method and Its Application as Heat Stabilizer in poly (vinyl chloride). Materials 2020, 13, 5223. [Google Scholar] [CrossRef]
- Wang, X.B.; Bai, Z.M.; Zhao, D.; Zhao, F.Y. Friction behavior of Mg–Al–CO3 layered double hydroxide prepared by magnesite. Appl. Surf. Sci. 2013, 277, 134–138. [Google Scholar] [CrossRef]
- Kuang, Y.; Zhao, L.; Zhang, S.; Zhang, F.; Dong, M.; Xu, S. Morphologies, Preparations and Applications of Layered Double Hydroxide Micro-/Nanostructures. Materials 2010, 3, 5220–5235. [Google Scholar] [CrossRef] [PubMed]
- Meszáros, S.; Halász, J.; Kónya, Z.; Sipos, P.; Pálinkó, I. Reconstruction of Calcined MgAl- and NiMgAl-Layered Double Hydroxides during Glycerol Dehydration and Their Recycling Characteristics. Appl. Clay Sci. 2013, 80–81, 245–248. [Google Scholar] [CrossRef]
- Wang, H.D.; Liu, Y.H.; Liu, W.R.; Wang, R.; Wen, J.G.; Sheng, H.P.; Peng, J.F.; Erdemir, A.; Luo, J.B. Tribological behavior of NiAl-Layered double hydroxide nanoplatelets as oil-based lubricant additives. ACS Appl. Mater. Interfaces 2017, 9, 30891–30899. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.D.; Liu, Y.H.; Liu, W.R.; Liu, Y.M.; Wang, K.P.; Li, J.J.; Ma, T.B.; Eryilmaz, O.L.; Shi, Y.J.; Erdemir, A.; et al. Superlubricity of polyalkylene glycol aqueous solutions enabled by ultrathin layered double hydroxide nanosheets. ACS Appl. Mater. Interfaces 2019, 11, 20249–20256. [Google Scholar] [CrossRef]
- Malak-Polaczyk, A.; Vix-Guterl, C.; Frackowiak, E. Carbon/Layered Double Hydroxide (LDH) composites for super capacitor application. Energy Fuels 2010, 24, 3346–3351. [Google Scholar] [CrossRef]
- Ma, W.; Ma, R.Z.; Wang, C.X.; Liang, J.B.; Liu, X.H.; Zhou, K.C.; Sasaki, T. A superlattice of alternately stacked Ni–Fe hydroxide nanosheets and graphene for efficient splitting of water. ACS Nano 2015, 9, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, S.; O’Hare, D.; Seeley, G. Delamination of layered double hydroxides in polar monomers: New LDH-acrylate nanocomposites. Chem. Commun. 2002, 14, 1506–1507. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, W. Mechanism of lubricating oil antioxidant. Lubr. Oil 1998, 13, 55–58. [Google Scholar]
- Dou, Y.; Pan, T.; Xu, S.; Yan, H.; Han, J.; Wei, M.; Evans, D.G.; Duan, X. Transparent, Ultrahigh-Gas-Barrier Films with a Brick–Mortar–Sand Structure. Angew. Chem. Int. Ed. 2015, 54, 9673–9678. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).