Effect of Temperature on Biobeneficiation of Bulk Copper-Nickel Concentrate with Thermoacidophilic Microbial Communities
Abstract
:1. Introduction
2. Materials and Methods
2.1. Copper–Nickel Concentrate
2.2. Microbial Communities and Cultivation Conditions
2.3. Analysis of Microbial Community
2.4. Bioleaching Procedure
2.5. Analytical Methods
3. Results and Discussion
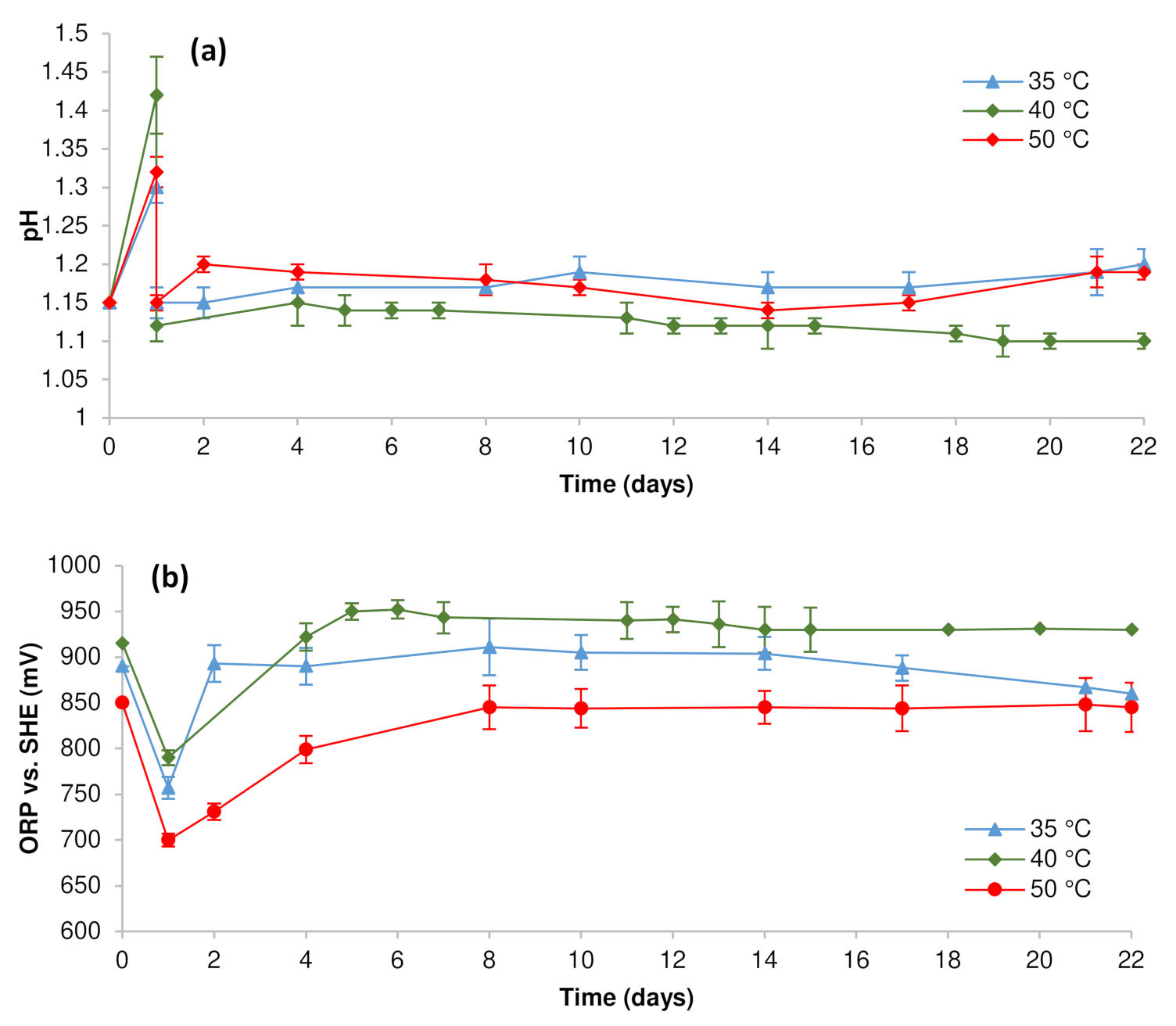
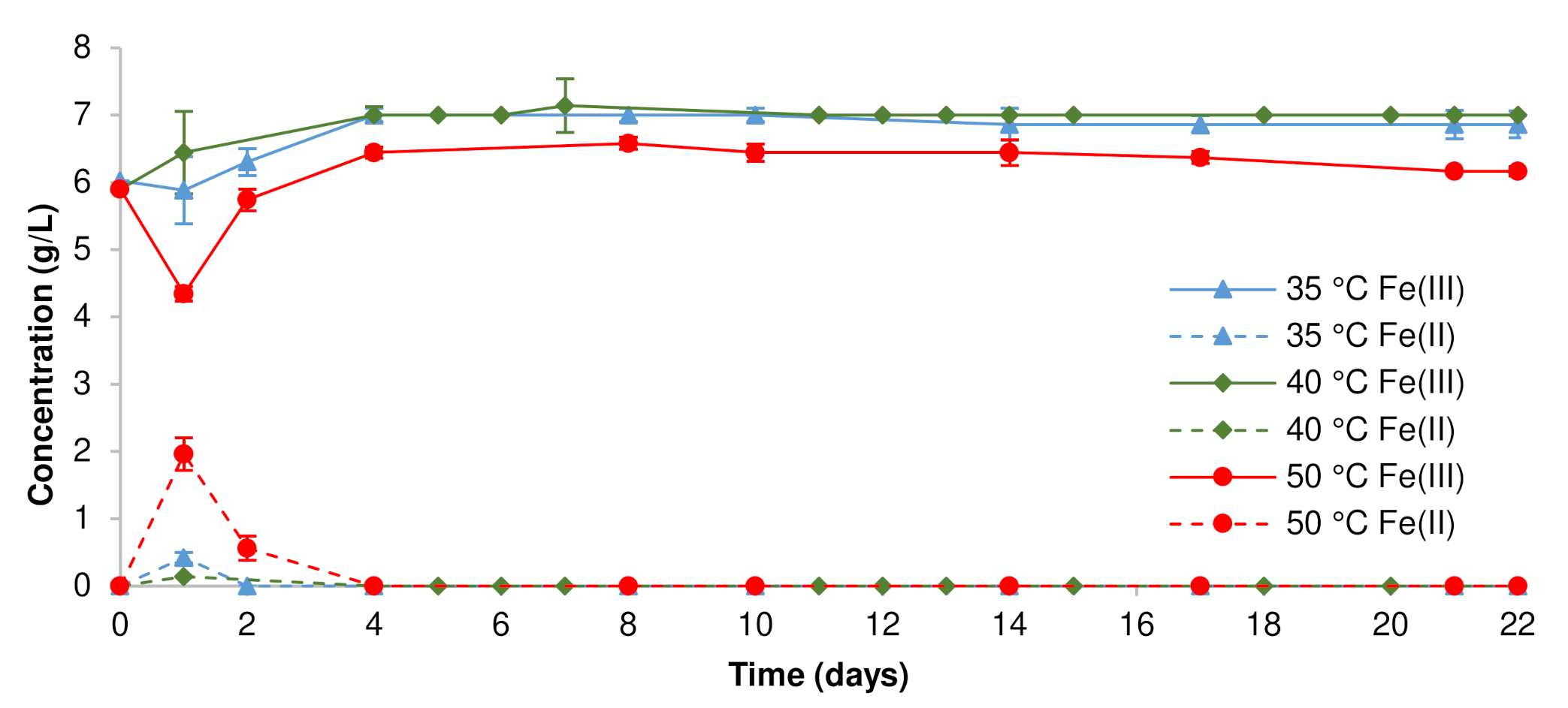
3.1. Pregnant Leach Solution Characteristics
3.2. Solids Composition
3.3. Microbial Community Structure
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wills, B.A.; Finch, J.A. Wills’ Mineral Processing Technology: An Introduction to the Practical Aspects of Ore Treatment and Mineral Recovery, 8th ed.; Butterworth-Heinemann: Oxford, UK, 2015. [Google Scholar]
- Frias, C.; Sánchez, F.; Van Staden, P.; Milanovic, D.; Kolehmainen, E. The IntMet project provides innovative hydro and biohydro- Technologies to deal efficiently with polymetallic and complex suphide ores. In Proceedings of the IMPC 2018–29th International Mineral Processing Congress, Moscow, Russia, 17–21 September 2018; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2019; pp. 2690–2699. [Google Scholar]
- Kaksonen, A.H.; Deng, X.; Bohu, T.; Zea, L.; Khaleque, H.N.; Gumulya, Y.; Boxall, N.J.; Morris, C.; Cheng, K.Y. Prospective directions for biohydrometallurgy. Hydrometallurgy 2020, 195, 105376. [Google Scholar] [CrossRef]
- Yin, S.-H.; Chen, W.; Fan, X.-L.; Liu, J.-M.; Wu, L.-B. Review and prospects of bioleaching in the Chinese mining industry. Int. J. Miner. Metall. Mater. 2021, 28, 1397–1412. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Lakaniemi, A.-M.; Tuovinen, O.H. Acid and ferric sulfate bioleaching of uranium ores: A review. J. Clean. Prod. 2020, 264, 121586. [Google Scholar] [CrossRef]
- Li, J.; Yang, H.; Tong, L.; Sand, W. Some aspects of industrial heap bioleaching technology: From basics to practice. Miner. Process. Extr. Metall. Rev. 2021, 1–9. [Google Scholar] [CrossRef]
- Kondrat’eva, T.F.; Pivovarova, T.A.; Tsaplina, I.A.; Fomchenko, N.V.; Zhuravleva, A.E.; Murav’ev, M.I.; Melamud, V.S.; Bulayev, A.G. Diversity of the communities of acidophilic chemolithotrophic microorganisms in natural and technogenic ecosystems. Microbiology 2012, 81, 1–24. [Google Scholar] [CrossRef]
- Pathak, A.; Morrison, L.; Healy, M.G. Catalytic potential of selected metal ions for bioleaching, and potential techno-economic and environmental issues: A critical review. Biores. Technol. 2017, 229, 211–221. [Google Scholar] [CrossRef] [Green Version]
- Potysz, A.; Pȩdziwiatr, A.; Hedwig, S.; Lenz, M. Bioleaching and toxicity of metallurgical wastes. J. Environ. Chem. Eng. 2020, 8, 104450. [Google Scholar] [CrossRef]
- Fomchenko, N.V.; Muravyov, M.I. Two-step biohydrometallurgical technology of copper-zinc concentrate processing as an opportunity to reduce negative impacts on the environment. J. Environ. Manag. 2018, 226, 270–277. [Google Scholar] [CrossRef]
- Mahmoud, A.; Cezac, P.; Hoadley, A.F.A.; Contamine, F.; D’Hugues, P. A review of sulfide minerals microbially assisted leaching in stirred tank reactors. Int. Biodeterior. Biodegradation 2017, 119, 118–146. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Mudunuru, B.M.; Hackl, R. The role of microorganisms in gold processing and recovery: A review. Hydrometallurgy 2014, 142, 70–83. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, Y.; Zhang, X.; Qian, L.; Sun, M.; Yang, Y.; Zhang, Y.; Wang, J.; Kim, H.; Qiu, G. The dissolution and passivation mechanism of chalcopyrite in bioleaching: An overview. Miner. Eng. 2019, 136, 140–154. [Google Scholar] [CrossRef]
- Harvey, T.J.; Yen, W.T.; Paterson, J.G. Selective zinc extraction from complex copper/zinc sulphide concentrates by pressure oxidation. Miner. Eng. 1992, 5, 975–992. [Google Scholar] [CrossRef]
- Fomchenko, N.V.; Muravyov, M.I. Selective leaching of zinc from copper-zinc concentrate. Appl. Biochem. Microbiol. 2017, 53, 73–77. [Google Scholar] [CrossRef]
- Fomchenko, N.; Uvarova, T.; Muravyov, M. Effect of mineral composition of sulfidic polymetallic concentrates on non-ferrous metals bioleaching. Miner. Eng. 2019, 138, 1–6. [Google Scholar] [CrossRef]
- Muravyov, M.I.; Panyushkina, A.E.; Melamud, V.S.; Bulaev, A.G.; Fomchenko, N.V. Ferric leaching of bulk sulfidic concentrates with biologically generated solution. Appl. Biochem. Microbiol. 2021, 57, 493–499. [Google Scholar] [CrossRef]
- Fomchenko, N.V.; Muravyov, M.I. Effect of sulfide mineral content in copper-zinc concentrates on the rate of leaching of non-ferrous metals by biogenic ferric iron. Hydrometallurgy 2019, 185, 82–87. [Google Scholar] [CrossRef]
- Arpalahti, A.; Lundström, M. The leaching behavior of minerals from a pyrrhotite-rich pentlandite ore during heap leaching. Miner. Eng. 2018, 119, 116–125. [Google Scholar] [CrossRef]
- Watling, H.R. The bioleaching of nickel-copper sulfides. Hydrometallurgy 2008, 91, 70–88. [Google Scholar] [CrossRef]
- Watling, H.R. The bioleaching of sulphide minerals with emphasis on copper sulphides: A review. Hydrometallurgy 2006, 84, 81–108. [Google Scholar] [CrossRef]
- Behera, S.K.; Manjaiah, M.; Sekar, S.; Panda, S.K.; Mavumengwana, V.; Mulaba-Bafubiandi, A.F. Optimization of microbial leaching of base metals from a South African sulfidic nickel ore concentrate by Acidithiobacillus ferrooxidans. Geomicrobiol. J. 2018, 35, 447–459. [Google Scholar] [CrossRef]
- Muravyov, M.; Panyushkina, A.; Bulaev, A.; Fomchenko, N. Biobeneficiation of bulk copper-zinc and copper-nickel concentrates at different temperatures. Miner. Eng. 2021, 170, 107040. [Google Scholar] [CrossRef]
- Gericke, M.; Govender, Y. Bioleaching strategies for the treatment of nickel-copper sulphide concentrates. Miner. Eng. 2011, 24, 1106–1112. [Google Scholar] [CrossRef]
- Muravyov, M.; Panyushkina, A. Distinct roles of acidophiles in complete oxidation of high-sulfur ferric leach product of zinc sulfide concentrate. Microorganisms 2020, 8, 386. [Google Scholar] [CrossRef] [Green Version]
- Panyushkina, A.; Bulaev, A.; Belyi, A.V. Unraveling the central role of sulfur-oxidizing Acidiphilium multivorum lms in industrial bioprocessing of gold-bearing sulfide concentrates. Microorganisms 2021, 9, 984. [Google Scholar] [CrossRef] [PubMed]
- Silverman, M.P.; Lundgren, D.G. Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. I. An improved medium and a harvesting procedure for securing high cell yields. J. Bacteriol. 1959, 77, 642–647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fadrosh, D.W.; Ma, B.; Gajer, P.; Sengamalay, N.; Ott, S.; Brotman, R.M.; Ravel, J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2014, 2, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pẽa, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.G.; Jacobsen, W.R. Determination of iron and iron-aluminum mixtures by titration with EDTA. Anal. Chem. 1960, 32, 215–217. [Google Scholar] [CrossRef]
- Filippova, N.A. Phazovy Analiz rud i Produktov ikh Pererabotki, 2nd ed.; Khimiya: Moscow, Russia, 1975. [Google Scholar]
- Rietveld, H.M. Line profiles of neutron powder-diffraction peaks for structure refinement. Acta Cryst. 1967, 22, 151–152. [Google Scholar] [CrossRef]
- Cruz, F.L.S.; Oliveira, V.A.; Guimarães, D.; Souza, A.D.; Leão, V.A. High-temperature bioleaching of nickel sulfides: Thermodynamic and kinetic implications. Hydrometallurgy 2010, 105, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Soe, K.M.; Ruan, R.; Jia, Y.; Tan, Q.; Wang, Z.; Shi, J.; Zhong, C.; Sun, H. Influence of jarosite precipitation on iron balance in heap bioleaching at monywa copper mine. J. Min. Inst. 2021, 247, 102–113. [Google Scholar] [CrossRef]
- Hedrich, S.; Joulian, C.; Graupner, T.; Schippers, A.; Guézennec, A.-G. Enhanced chalcopyrite dissolution in stirred tank reactors by temperature increase during bioleaching. Hydrometallurgy 2018, 179, 125–131. [Google Scholar] [CrossRef]
- Ranjbar, M.; Fazaelipoor, M.H.; Ranjbar Hamghavandi, M.; Schaffie, M.; Manafi, Z. Modeling study of the bio-dissolution of copper concentrate in a continuous bioreactors system. Miner. Eng. 2020, 153, 384. [Google Scholar] [CrossRef]
- Liu, H.-C.; Xia, J.-L.; Nie, Z.-Y.; Liu, L.-Z.; Wang, L.; Ma, C.-Y.; Zheng, L.; Zhao, Y.-D.; Wen, W. Comparative study of S, Fe and Cu speciation transformation during chalcopyrite bioleaching by mixed mesophiles and mixed thermophiles. Miner. Eng. 2017, 106, 22–32. [Google Scholar] [CrossRef]
- Tian, Z.; Li, H.; Wei, Q.; Qin, W.; Yang, C. Effects of redox potential on chalcopyrite leaching: An overview. Miner. Eng. 2021, 172, 107135. [Google Scholar] [CrossRef]
- Tanne, C.; Schippers, A. Electrochemical investigation of microbially and galvanically leached chalcopyrite. Hydrometallurgy 2021, 202, 105603. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, J.; Yang, C.; Hu, M.; Gan, X.; Tao, L.; Qin, W.; Qiu, G. Effect of redox potential on bioleaching of chalcopyrite by moderately thermophilic bacteria: An emphasis on solution compositions. Hydrometallurgy 2015, 151, 141–150. [Google Scholar] [CrossRef]
- Fu, K.; Ning, Y.; Chen, S.; Wang, Z. Bioleaching of different copper sulphide minerals and their physicochemical properties dependence. Trans. Inst. Min. Metall. Sect. C Miner. Process. Extr. Metall. 2016, 125, 1–4. [Google Scholar] [CrossRef]
- Mason, L.J.; Rice, N.M. The adaptation of Thiobacillus ferrooxidans for the treatment of nickel-iron sulphide concentrates. Miner. Eng. 2002, 15, 795–808. [Google Scholar] [CrossRef]
- Panyushkina, A.E.; Babenko, V.V.; Nikitina, A.S.; Selezneva, O.V.; Tsaplina, I.A.; Letarova, M.A.; Kostryukova, E.S.; Letarov, A.V. Sulfobacillus thermotolerans: New insights into resistance and metabolic capacities of acidophilic chemolithotrophs. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karavaiko, G.I.; Bogdanova, T.I.; Tourova, T.P.; Kondrat’eva, T.F.; Tsaplina, I.A.; Egorova, M.A.; Krasil’nikova, E.N.; Zakharchuk, L.M. Reclassification of “Sulfobacillus thermosulfidooxidans subsp. thermotolerans” strain K1 as Alicyclobacillus tolerans sp. nov. and Sulfobacillus disulfidooxidans Dufresne et al. 1996 as Alicyclobacillus disulfidooxidans comb. nov., and emended description. Int. J. Syst. Evol. Microbiol. 2005, 55, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Panyushkina, A.E.; Tsaplina, I.A.; Grigor’eva, N.V.; Kondrat’eva, T.F. Thermoacidophilic microbial community oxidizing the gold-bearing flotation concentrate of a pyrite-arsenopyrite ore. Microbiology 2014, 83, 539–549. [Google Scholar] [CrossRef]
- Holanda, R.; Hedrich, S.; Ňancucheo, I.; Oliveira, G.; Grail, B.M.; Johnson, D.B. Isolation and characterisation of mineral-oxidising “Acidibacillus” spp. from mine sites and geothermal environments in different global locations. Res. Microbiol. 2016, 167, 613–623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, D.B.; Bacelar-Nicolau, P.; Okibe, N.; Thomas, A.; Hallberg, K.B. Ferrimicrobium acidiphilum gen. nov., sp. nov. and Ferrithrix thermotolerans gen. nov., sp. nov.: Heterotrophic, iron-oxidizing, extremely acidophilic actinobacteria. Int. J. Syst. Evol. Microbiol. 2009, 59, 1082–1089. [Google Scholar] [CrossRef] [PubMed]
- Golyshina, O.V.; Lünsdorf, H.; Kublanov, I.V.; Goldenstein, N.I.; Hinrichs, K.-U.; Golyshin, P.N. The novel extremely acidophilic, cell-wall-deficient archaeon Cuniculiplasma divulgatum gen. nov., sp. nov. represents a new family, Cuniculiplasmataceae fam. nov., of the order Thermoplasmatales. Int. J. Syst. Evol. Microbiol. 2016, 66, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Bulaev, A.G.; Kanygina, A.V.; Manolov, A.I. Genome analysis of Acidiplasma sp. MBA-1, a polyextremophilic archaeon predominant in the microbial community of a bioleaching reactor. Microbiol. Russian Fed. 2017, 86, 89–95. [Google Scholar] [CrossRef]



| Cu | Ni | Fe | S | Ca | Si |
|---|---|---|---|---|---|
| 10.8 | 7.2 | 21.8 | 26.5 | 0.96 | 18.0 |
| Chalcopyrite | Pentlandite | Violarite | Pyrrhotite | Pyrite |
|---|---|---|---|---|
| 31 | 12 | 8 | 4 | 6 |
| Temperature at Bioleaching (°C) | Content (wt%) | ||||||
|---|---|---|---|---|---|---|---|
| Chalcopyrite | Pentlandite | Violarite | Pyrite | Pyrrhotite | Jarosite | Elemental Sulfur | |
| 35 | 35 | 2 | 0 | 2 | 0 | 25 | 1 |
| 40 | 45 | traces | 0 | 1 | 0 | 17 | 1 |
| 50 | 30 | traces | 0 | traces | 0 | 40 | 1 |
| Temperature at Bioleaching (°C) | Mass Loss (wt %) | Content (wt %) | |||
|---|---|---|---|---|---|
| Cu | Ni | Fe | S | ||
| 35 | 27 | 12.3 | 1.54 | 20.2 | 18.1 |
| 40 | 40 | 15.6 | 0.54 | 21.4 | 19.8 |
| 50 | 27 | 10.3 | 0.18 | 20.6 | 15.7 |
| Species | Original consortium 1 | Inoculum 2, 35 °C | Final 3, 35 °C | Inoculum, 40 °C | Final, 40 °C | Inoculum, 50 °C | Final, 50 °C |
|---|---|---|---|---|---|---|---|
| At. ferrooxidans | + 4 | N/A | N/A | N/A | N/A | – 5 | – |
| At. thiooxidans | + | N/A | N/A | N/A | N/A | – | – |
| At. caldus | + | N/A | N/A | N/A | N/A | – | – |
| Acidithiobacillus spp. | + | + | + | + | + | – | – |
| S. thermotolerans | + | + | + | + | + | + | + |
| S. thermosulfidooxidans | + | – | – | + | + | + | + |
| Al. tolerans | + | + | + | + | + | + | + |
| Ac. ferrooxidans | + | – | – | + | + | – | – |
| Fr. acidiphilum | + | + | + | + | – | – | – |
| Fr. acidarmanus | + | – | – | – | – | – | – |
| Acidiplasma sp. | + | – | – | – | – | + | + |
| Cuniculiplasma sp. | + | – | – | + | – | – | – |
| L. ferriphilum | + | + | – | + | + | – | – |
| Ap. multivorum | + | – | – | – | – | – | – |
| Fm. acidiphilium | + | – | – | + | – | – | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panyushkina, A.; Fomchenko, N.; Babenko, V.; Muravyov, M. Effect of Temperature on Biobeneficiation of Bulk Copper-Nickel Concentrate with Thermoacidophilic Microbial Communities. Metals 2021, 11, 1969. https://doi.org/10.3390/met11121969
Panyushkina A, Fomchenko N, Babenko V, Muravyov M. Effect of Temperature on Biobeneficiation of Bulk Copper-Nickel Concentrate with Thermoacidophilic Microbial Communities. Metals. 2021; 11(12):1969. https://doi.org/10.3390/met11121969
Chicago/Turabian StylePanyushkina, Anna, Natalya Fomchenko, Vladislav Babenko, and Maxim Muravyov. 2021. "Effect of Temperature on Biobeneficiation of Bulk Copper-Nickel Concentrate with Thermoacidophilic Microbial Communities" Metals 11, no. 12: 1969. https://doi.org/10.3390/met11121969






