Sorption Recovery of Platinum Metals from Production Solutions of Sulfate-Chloride Leaching of Chromite Wastes
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aleksandrova, T.N.; O’Connor, C. Processing of platinum group metal ores in Russia and south Africa: Current state and prospects. J. Min. Inst. 2020, 244, 462–473. [Google Scholar] [CrossRef]
- Katsuhiro, N.; Toru, H.O. Platinum group metals production. In Treatise on Process Metallurgy; Seetharama, S., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1071–1097. [Google Scholar]
- Saburbayeva, L.Y.; Boduen, A.Y.; Yu, P.S.; Ukraintsev, I.V. Study of pressure oxidation and bacterial leaching efficiency as a method of refractory gold concentrate breakdown. In Proceedings of the IMPC 2018–29th International Mineral Processing Congress, Moscow, Russia, 17–21 September 2018; pp. 2911–2921. [Google Scholar]
- Aleksandrova, T.N.; Nikolaeva, N.V.; Lvov, V.V.; Romashev, A.O. Ore processing efficiency improvements for precious metals based on process simulations. Obogashchenie Rud. 2019, 2, 8–13. [Google Scholar] [CrossRef]
- Lvov, V.V.; Chitalov, L.S. Modern trends in the design of comminution processes and equipment for non-ferrous metals ores. Tsvetnye Metally. 2020, 10, 20–26. [Google Scholar] [CrossRef]
- Buslaeva, T.M.; Simanova, S.A. The state of platinum metals in hydrochloric acid and chloride aqueous solutions. Palladium, platinum, rhodium, and iridium. J. Coord. Chem 1999, 25, 165–176. [Google Scholar]
- Petrov, G.V. Concentration of Platinum Metals in the Processing of Traditional and Non-Traditional Platinum-Metal Raw Materials; St. Petersburg State Mining Institute: St. Petersburg, Russia, 2001; p. 106. [Google Scholar]
- Zolotov, Y.A.; Varshal, G.M.; Ivanov, V.M. Analytical Chemistry of Platinum Group Metals: A Collection of Review Articles; KomKniga: Moscow, Russia, 2005; p. 592. [Google Scholar]
- Zakharchenko, E.A.; Mokhodoeva, O.B.; Myasoedova, G.V. Use of fibrous “filled” sorbents for dynamic concentration of precious metals. Res. J. Sorpt. Chromatogr. Process. 2006, 6, 233–241. [Google Scholar]
- Sipkina, E.I. Sorption materials for the extraction of platinum (IV) from chloride solutions. Appl. Chem. Biotechnol. 2015, 4, 7–15. [Google Scholar]
- Cheremisina, E.; Cheremisina, O.; Ponomareva, M.; Bolotov, V.; Fedorov, A. Kinetic features of the hydrogen sulfide sorption on the ferro-manganese material. Metals 2021, 11, 90. [Google Scholar] [CrossRef]
- Blokhin, A.A.; Abovsky, N.D.; Murashkin, Y.V.; Moloshneva, O.V.; Voronina, S.N. Sorption extraction of platinum and palladium from spent refining solutions with their low content. In Proceedings of the First International Congress and Exhibition Non-Ferrous Metals of Siberia-2009, Siberia, Russia, 8–10 September 2009; pp. 587–592. [Google Scholar]
- Borbat, V.F.; Schindler, A.A.; Jarosik, I.S.; Semeonova, O.N.; Petrov, A.F.; Mikhaylenko, M. Extraction of platinum (IV), palladium (II) and rhodium (III) by sorption from solutions and pulps with Purolite resins. In Proceedings of the First International Congress and Exhibition Non-Ferrous Metals of Siberia-2009, Siberia, Russia, 8–10 September 2009; pp. 313–331. [Google Scholar]
- Melnikov, N.D. Study of Ion-Exchange Equilibria and the Kinetics of Sorption of Platinum (II, IV) and Rhodium (III) in Chloride and Sulfate-Chloride Solutions for the Purpose of their Separation and Concentration. Ph.D. Thesis, University of Chemical Sciences, Irkutsk, 2013; p. 20. [Google Scholar]
- Kononova, O.N.; Melnikov, A.M.; Borisova, T.V. Simultaneous sorption recovery of platinum and rhodium from sulfate–chloride solutions. Hydrometallurgy 2012, 117, 101–107. [Google Scholar] [CrossRef]
- Kukushkin, Y.N. Chemistry of Coordination Compounds; Vysshaya Shkola: Moscow, Russia, 1985; p. 455. [Google Scholar]
- Buslaeva, T.M. Chemistry and Technology of Platinum metals; Nauka: Moscow, Russia, 1999; p. 79. [Google Scholar]
- Hartley, F.R. Chemistry of the Platinum Group Metals; Elsevier: Amsterdam, The Netherlands, 1991; p. 642. [Google Scholar]
- Egorov, S.A.; Blokhin, A.A.; Murashkin, Y.V. Features of the sorption of rhodium (III) from chloride solutions on an ion exchange resin with thiouronium functional groups. Russ. J. Appl. Chem. 2020, 93, 1359–1365. [Google Scholar] [CrossRef]
- Nikoloski, A.N.; Ang, K.-L.; Li, D. Recovery of platinum, palladium and rhodium from acidic chloride leach solution using ion exchange resins. Hydrometallurgy. 2015, 152, 20–32. [Google Scholar] [CrossRef] [Green Version]
- Dodin, D.A. Platinometallnye Deposits of Russia. World Mineral Resource Base of Noble and Non-Ferrous Metals: 1970–2000–2025; Migachev: Saint-Petersburg, Russia, 2000; p. 753. [Google Scholar]
- Dodin, D.A.; Izoitko, V.M. Superlarge man-made deposits of platinum metals. Obogashchenie Rud 2006, 6, 19–23. [Google Scholar]
- Nikolaeva, E.; Talovina, I.; Nikiforova, V.; Heide, G. Chemical composition and genesis of serpentinite group minerals in nickeliferous weathering crust of the elov deposit (Urals). In Scientific and Practical Studies of Raw Material Issues; Proceedings of the Russian-German Raw Materials Dialogue: A Collection of Young Scientists Papers and Discussion, Saint-Petersburg, Russia; CRC: Boca Raton, FL, USA, 2019; pp. 3–10. [Google Scholar]
- Ponomarenko, T.; Nevskaya, M.; Jonek-Kowalska, I. Mineral resource depletion assessment: Alternatives, problems, results. Sustainability 2021, 13, 862. [Google Scholar] [CrossRef]
- Stepanov, S.Y.; Palamarchuk, R.S.; Kozlov, A.V.; Khanin, D.A.; Varlamov, D.A.; Kiseleva, D.V. Platinum-group minerals of Pt-placer deposits associated with the svetloborsky Ural-Alaskan type massif, middle Urals, Russia. Minerals 2019, 9, 77. [Google Scholar] [CrossRef] [Green Version]
- Ivanik, S.A.; Ilyukhin, D.A. Flotation extraction of elemental sulfur from gold-bearing cakes. J. Min. Inst. 2020, 242, 202–208. [Google Scholar] [CrossRef]
- Zaitsev, A.Y. Methodological approach to substantiation of capital investments of gold fields based on unit costs. J. Min. Inst. 2019, 238, 459–464. [Google Scholar] [CrossRef] [Green Version]
- Fazlullin, M.I. Heap Leaching of Precious Metals; Publishing House of the Academy of Mining Sciences: Moscow, Russia, 2001; p. 647. [Google Scholar]
- Fazlullin, M.I. Heap Leaching of Noble Metals; Mining Science Academy Publ.: Moscow, Russia, 2001; p. 647. [Google Scholar]
- Kononova, O.N. Application of Chelated and Amphoteric Ionites in the Analysis and Technology of Rare Elements. Master’s Thesis, Kurgan State University, Krasnoyarsk, Russia, 1981. [Google Scholar]
- Helferich, F. Ionites. Fundamentals of Ion Exchange; Foreign Literature Publishing House: Moscow, Russia, 1962; p. 492. [Google Scholar]
- Lee, I.-H.; Kuan, Y.C.; Chern, J.M. Equilibrium and kinetics of heavy metal ion exchange. J. Chin. Inst. Chem. Eng. 2007, 38, 71–84. [Google Scholar] [CrossRef]
- Kokotov, Y.A.; Pasechnik, V.A. Equilibrium and kinetics of ion exchange. Chemistry 1970, 336. [Google Scholar]
- Kononova, O.N. Chromatographic methods of separation and concentration. Master’s Thesis, Krasnoyarsk State University, Krasnoyarsk, Russia, 1990. [Google Scholar]
- Billz, J.; Froelich, P. Kinetics and mechanisms of ion exchange on cheat-forming polymers. J. Phys. Chem. 1985, 3, 495–504. [Google Scholar]
- Krizhanovskaya, O.O.; Sinyaeva, L.A.; Karpov, S.I.; Selemenev, V.F.; Borodin, E.V.; Rossner, F. Kinetic models for the description of the sorption of fat-soluble physiologically active substances by highly ordered inorganic silicon-containing polymers. Sorpt. Chromatogr. Process. 2014, 5, 784–794. [Google Scholar]





| Ionite | Type | Physical Structure | Functional Groups | SEC for Cl- mmol/g |
|---|---|---|---|---|
| AB-17-8 | strong base | G | QAB | 3.4 |
| Purolite S 985 | weakly basic | MP | polyamines | 2.1 |
| AH-31 | weakly basic | G | AA | 2.8 |
| Ionite | Individual Sorption | Joint Sorption | ||||
|---|---|---|---|---|---|---|
| Quantitative Characteristics of Sorption | EC | D | R | EC | D | R |
| platinum | ||||||
| AH-31 | 0.010 | 213.16 | 81 | - | - | - |
| AB-17-8 | 0.011 | 334.62 | 87 | 0.010 | 188.10 | 79 |
| Purolite S 985 | 0.012 | 950.00 | 95 | 0.011 | 262.50 | 84 |
| iridium | ||||||
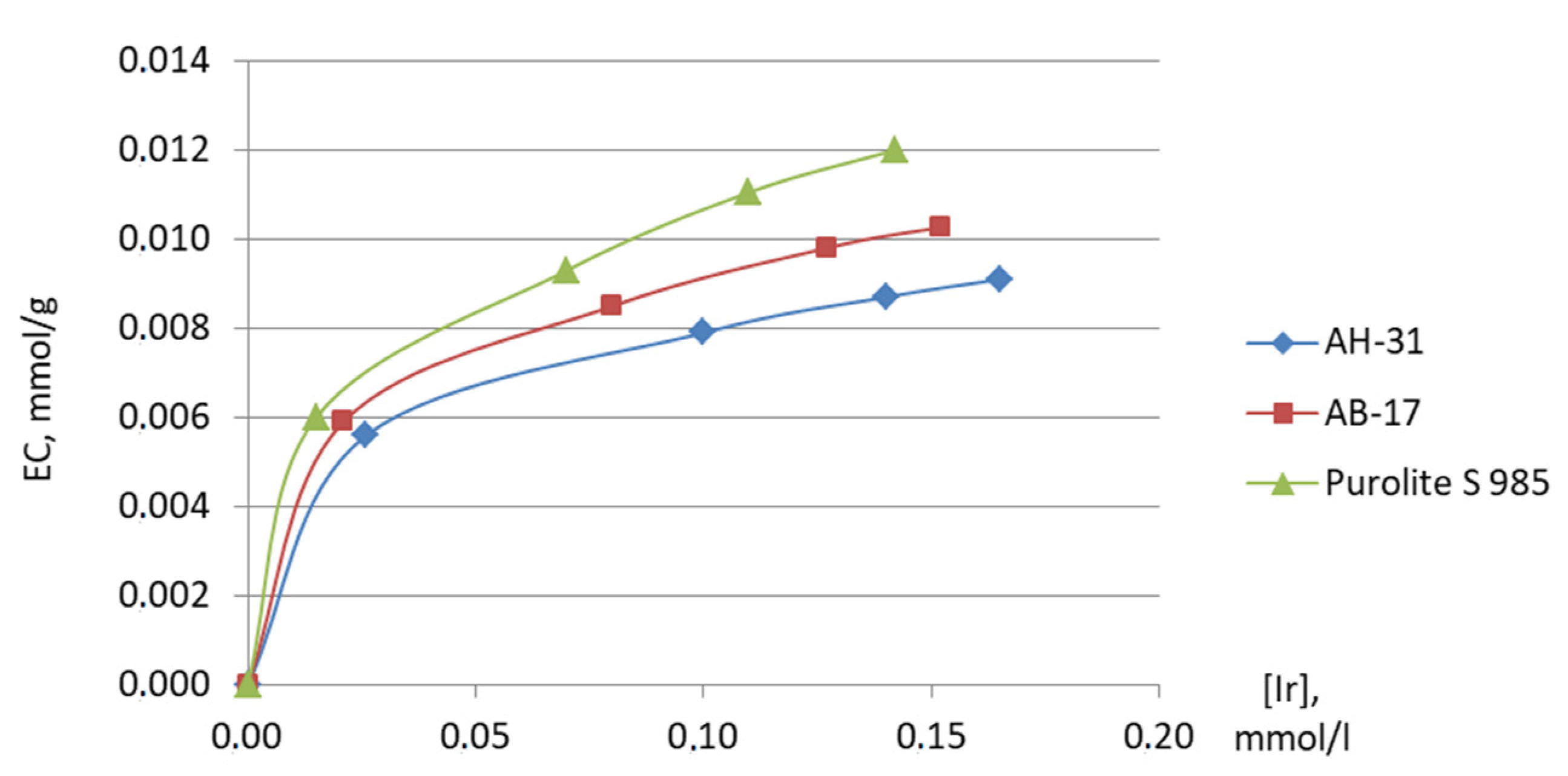
| AH-31 | 0.0078 | 78.21 | 61 | - | - | - |
| AB-17-8 | 0.0086 | 101.52 | 67 | 0.007 | 57.14 | 54 |
| Purolite S 985 | 0.0094 | 135.19 | 73 | 0.009 | 119.94 | 71 |
| Ionite | Individual Sorption | Joint Sorption | ||||
|---|---|---|---|---|---|---|
| Thermodynamic Parameters of Sorption | K | R2 | ΔG, kJ/mol | K | R2 | ΔG, kJ/mol |
| platinum | ||||||
| AH-31 | 16.02 | 0.986 | −6.76 | - | - | - |
| AB-17-8 | 20.4 | 0.9376 | −7.36 | 27.27 | 0.9934 | −8.05 |
| Purolite S 985 | 153.84 | 0.9861 | −12.27 | 67.67 | 0.9788 | −10.27 |
| iridium | ||||||
| AH-31 | 47.95 | 0.9796 | −9.43 | - | - | - |
| AB-17-8 | 53.66 | 0.9821 | −9.70 | 20.83 | 0.9979 | −7.40 |
| Purolite S 985 | 66.90 | 0.9681 | −10.24 | 27.34 | 0.997 | −8.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrov, G.; Zotova, I.; Nikitina, T.; Fokina, S. Sorption Recovery of Platinum Metals from Production Solutions of Sulfate-Chloride Leaching of Chromite Wastes. Metals 2021, 11, 569. https://doi.org/10.3390/met11040569
Petrov G, Zotova I, Nikitina T, Fokina S. Sorption Recovery of Platinum Metals from Production Solutions of Sulfate-Chloride Leaching of Chromite Wastes. Metals. 2021; 11(4):569. https://doi.org/10.3390/met11040569
Chicago/Turabian StylePetrov, Georgiy, Irina Zotova, Tatiana Nikitina, and Svetlana Fokina. 2021. "Sorption Recovery of Platinum Metals from Production Solutions of Sulfate-Chloride Leaching of Chromite Wastes" Metals 11, no. 4: 569. https://doi.org/10.3390/met11040569






